Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. Histopathology
2.3. DNA and RNA Extraction
2.4. Molecular Characterization
2.5. Quantitative Real-Time PCR
2.6. Genetic Diversity and Phylogenetic Analyses
3. Results
3.1. Detection of Herpesvirus Infection
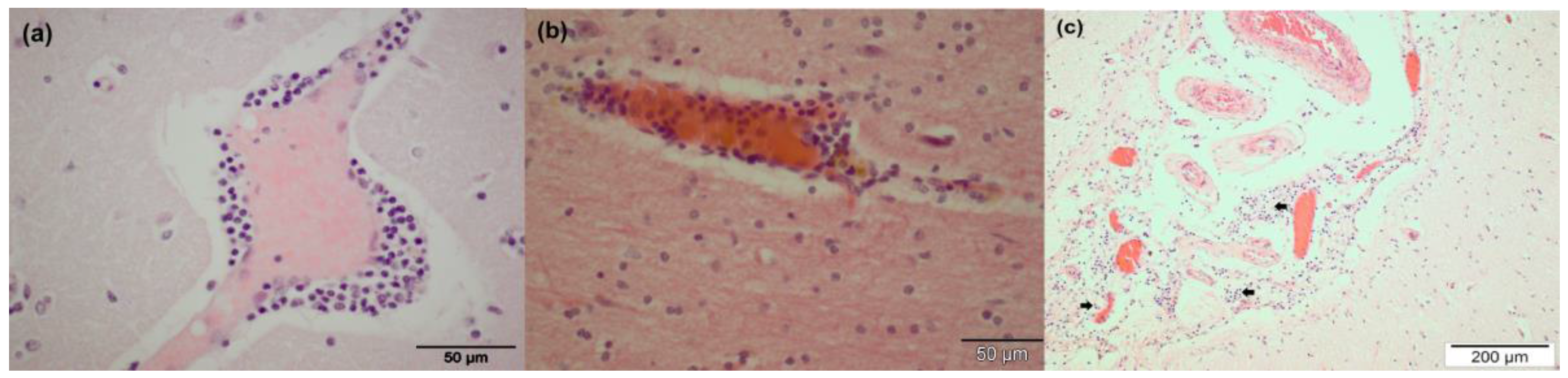
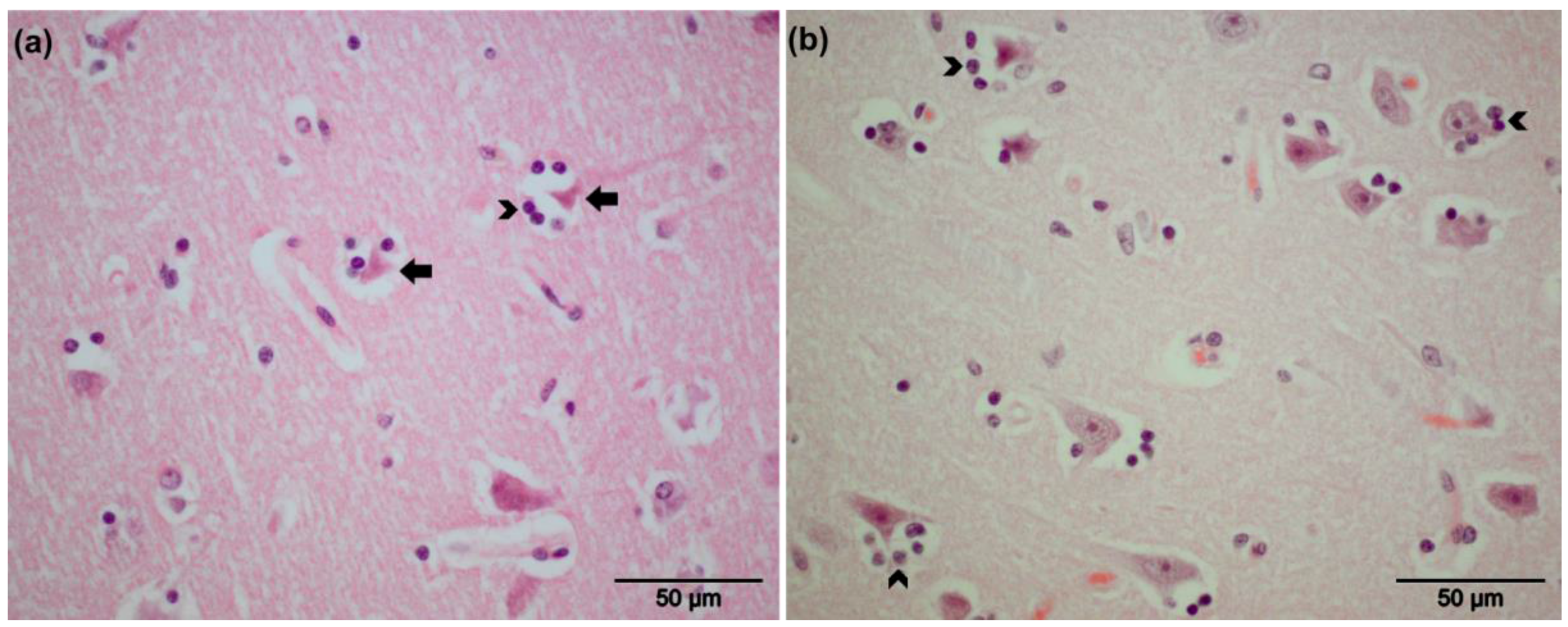
3.2. Pathological Findings
3.3. Detection of Latent Infection
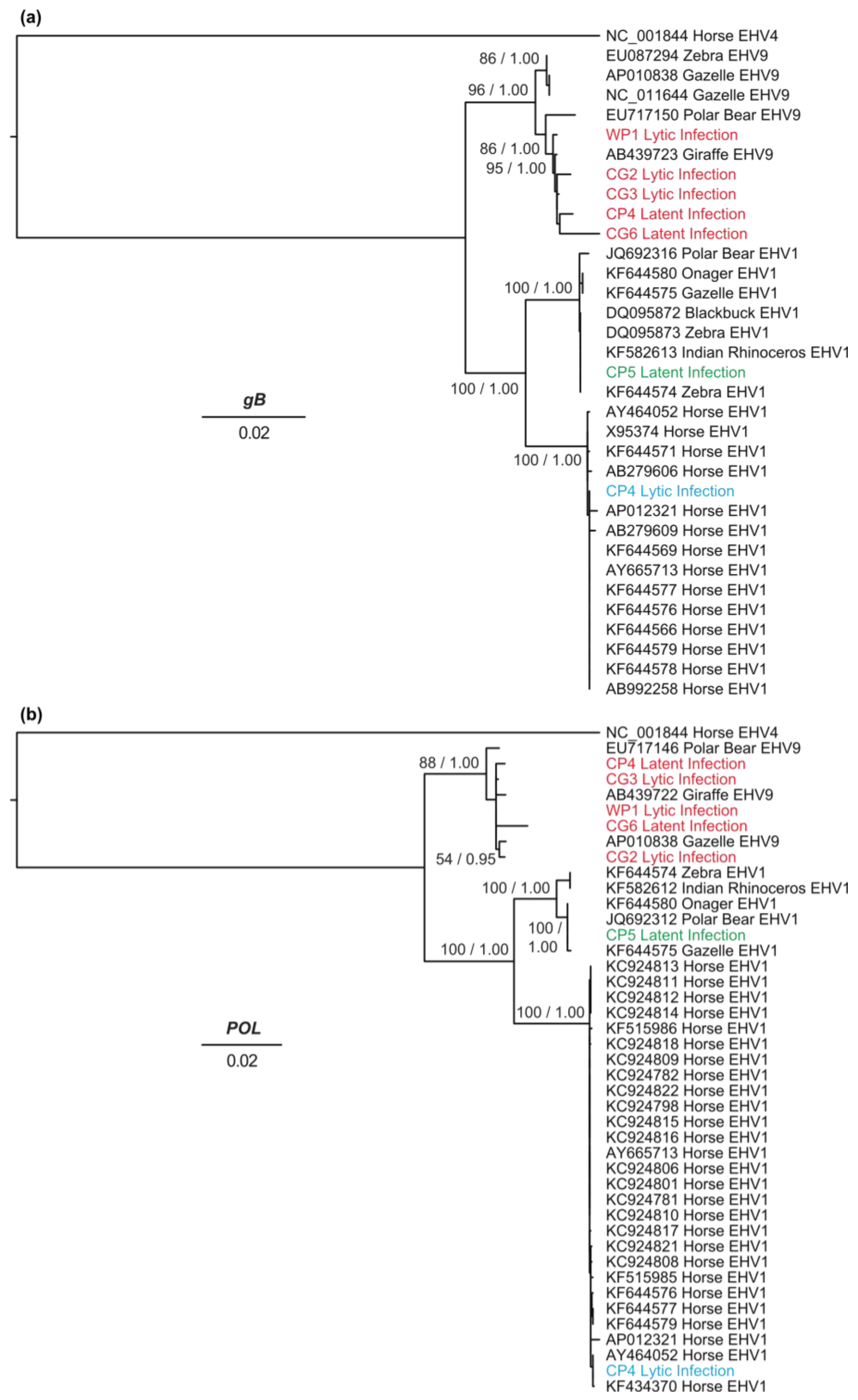
3.4. Genetic Diversity and Phylogenetic Analyses
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thiry, E.; Meurens, F.; Muylkens, B.; McVoy, M.; Gogev, S.; Thiry, J.; Vanderplasschen, A.; Epstein, A.; Keil, G.; Schynts, F. Recombination in alphaherpesviruses. Rev. Med. Virol. 2005, 15, 89–103. [Google Scholar] [CrossRef] [PubMed]
- Davison, A.J.; Eberle, R.; Ehlers, B.; Hayward, G.S.; McGeoch, D.J.; Minson, A.C.; Pellett, P.E.; Roizman, B.; Studdert, M.J.; Thiry, E. The order Herpesvirales. Arch. Virol. 2009, 154, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Fukushi, H.; Yamaguchi, T.; Yamada, S. Complete genome sequence of equine herpesvirus type 9. J. Virol. 2012, 86, 13822. [Google Scholar] [CrossRef] [PubMed]
- Schrenzel, M.D.; Tucker, T.A.; Donovan, T.A.; Busch, M.D.; Wise, A.G.; Maes, R.K.; Kiupel, M. New hosts for equine herpesvirus 9. Emerg. Infect. Dis. 2008, 14, 1616–1619. [Google Scholar] [CrossRef] [PubMed]
- Wohlsein, P.; Lehmbecker, A.; Spitzbarth, I.; Algermissen, D.; Baumgartner, W.; Boer, M.; Kummrow, M.; Haas, L.; Grummer, B. Fatal epizootic equine herpesvirus 1 infections in new and unnatural hosts. Vet. Microbiol. 2011, 149, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Fukushi, H.; Tomita, T.; Taniguchi, A.; Ochiai, Y.; Kirisawa, R.; Matsumura, T.; Yanai, T.; Masegi, T.; Yamaguchi, T.; Hirai, K. Gazelle herpesvirus 1: A new neurotropic herpesvirus immunologically related to equine herpesvirus 1. Virology 1997, 227, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Kasem, S.; Yamada, S.; Kiupel, M.; Woodruff, M.; Ohya, K.; Fukushi, H. Equine herpesvirus type 9 in giraffe with encephalitis. Emerg. Infect. Dis. 2008, 14, 1948–1949. [Google Scholar] [CrossRef] [PubMed]
- Abdelgawad, A.; Hermes, R.; Damiani, A.; Lamglait, B.; Czirjak, G.A.; East, M.; Aschenborn, O.; Wenker, C.; Kasem, S.; Osterrieder, N.; et al. Comprehensive serology based on a peptide ELISA to assess the prevalence of closely related equine herpesviruses in zoo and wild animals. PLoS ONE 2015, 10, e0138370. [Google Scholar] [CrossRef] [PubMed]
- Borchers, K.; Frolich, K. Antibodies against equine herpesviruses in free-ranging mountain zebras from Namibia. J. Wildl. Dis. 1997, 33, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Borchers, K.; Lieckfeldt, D.; Ludwig, A.; Fukushi, H.; Allen, G.; Fyumagwa, R.; Hoare, R. Detection of equid herpesvirus 9 DNA in the trigeminal ganglia of a Burchell’s zebra from the Serengeti ecosystem. J. Vet. Med. Sci. 2008, 70, 1377–1381. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Izume, S.; Okada, A.; Ohya, K.; Kimura, T.; Fukushi, H. Full genome sequences of zebra-borne equine herpesvirus type 1 isolated from zebra, onager and Thomson’s gazelle. J. Vet. Med. Sci. 2014, 76, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Aleman, M.; Pickles, K.J.; Simonek, G.; Madigan, J.E. Latent equine herpesvirus-1 in trigeminal ganglia and equine idiopathic headshaking. J. Vet. Med. Sci. 2012, 26, 192–194. [Google Scholar] [CrossRef] [PubMed]
- Pusterla, N.; Wilson, W.D.; Mapes, S.; Finno, C.; Isbell, D.; Arthur, R.M.; Ferraro, G.L. Characterization of viral loads, strain and state of equine herpesvirus-1 using real-time PCR in horses following natural exposure at a racetrack in California. Vet. J. 2009, 179, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.J.; Iqbal, J.; Purewal, A.; Hamblin, A.S.; Edington, N. In vitro reactivation of latent equid herpesvirus-1 from CD5+/CD8+ leukocytes indirectly by IL-2 or chorionic gonadotrophin. J. Gen. Virol. 1998, 79 Pt 12, 2997–3004. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, R.; Poliani, P.L.; Levine, M.; Glorioso, J.C.; Fink, D.J. Detection of herpes simplex virus type 1 latency-associated transcript expression in trigeminal ganglia by in situ reverse transcriptase PCR. J. Virol. 1996, 70, 6519–6523. [Google Scholar] [PubMed]
- Baxi, M.K.; Efstathiou, S.; Lawrence, G.; Whalley, J.M.; Slater, J.D.; Field, H.J. The detection of latency-associated transcripts of equine herpesvirus 1 in ganglionic neurons. J. Gen. Virol. 1995, 76 Pt 12, 3113–3118. [Google Scholar] [CrossRef] [PubMed]
- Telford, E.A.; Watson, M.S.; Perry, J.; Cullinane, A.A.; Davison, A.J. The DNA sequence of equine herpesvirus-4. J. Gen. Virol. 1998, 79 Pt 5, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Dunowska, M. A review of equid herpesvirus 1 for the veterinary practitioner. Part B: Pathogenesis and epidemiology. N. Z. Vet. J. 2014, 62, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Welch, H.M.; Bridges, C.G.; Lyon, A.M.; Griffiths, L.; Edington, N. Latent equid herpesviruses 1 and 4: Detection and distinction using the polymerase chain reaction and co-cultivation from lymphoid tissues. J. Gen. Virol. 1992, 73 Pt 2, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Losada, M.; Arenas, M.; Galán, JC.; Palero, F.; González-Candelas, F. Recombination in viruses: Mechanisms, methods of study, and evolutionary consequences. Infect. Genet. Evol. 2015, 30, 296–307. [Google Scholar] [CrossRef] [PubMed]
- Norberg, P.; Depledge, D.P.; Kundu, S.; Atkinson, C.; Brown, J.; Haque, T.; Hussaini, Y.; MacMahon, E.; Molyneaux, P.; Papaevangelou, V.; et al. Recombination of globally circulating varicella-zoster virus. J. Virol. 2015, 89, 7133–7146. [Google Scholar] [CrossRef] [PubMed]
- Pagamjav, O.; Sakata, T.; Matsumura, T.; Yamaguchi, T.; Fukushi, H. Natural recombinant between equine herpesviruses 1 and 4 in the ICP4 gene. Microbiol. Immunol. 2005, 49, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Vaz, P.K.; Horsington, J.; Hartley, C.A.; Browning, G.F.; Ficorilli, N.P.; Studdert, M.J.; Gilkerson, J.R.; Devlin, J.M. Evidence of widespread natural recombination among field isolates of equine herpesvirus 4 but not among field isolates of equine herpesvirus 1. J. Gen. Virol. 2016, 97, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Abdelgawad, A.; Azab, W.; Damiani, A.M.; Baumgartner, K.; Will, H.; Osterrieder, N.; Greenwood, A.D. Zebra-borne equine herpesvirus type 1 (EHV-1) infection in non-African captive mammals. Vet. Microbiol. 2014, 169, 102–106. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, A.D.; Tsangaras, K.; Ho, S.Y.; Szentiks, C.A.; Nikolin, V.M.; Ma, G.; Damiani, A.; East, M.L.; Lawrenz, A.; Hofer, H.; et al. A potentially fatal mix of herpes in zoos. Curr. Biol. 2012, 22, 1727–1731. [Google Scholar] [CrossRef] [PubMed]
- Klieforth, R.; Maalouf, G.; Stalis, I.; Terio, K.; Janssen, D.; Schrenzel, M. Malignant catarrhal fever-like disease in Barbary red deer (Cervus elaphus barbarus) naturally infected with a virus resembling alcelaphine herpesvirus 2. J. Clin. Microbiol. 2002, 40, 3381–3390. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.N.; Khawaja, H.; Chen, Y.W. Culture conditions affect expression of DUX4 in FSHD myoblasts. Molecules 2015, 20, 8304–8315. [Google Scholar] [CrossRef] [PubMed]
- VanDevanter, D.R.; Warrener, P.; Bennett, L.; Schultz, E.R.; Coulter, S.; Garber, R.L.; Rose, T.M. Detection and analysis of diverse herpesviral species by consensus primer PCR. J. Clin. Microbiol. 1996, 34, 1666–1671. [Google Scholar] [PubMed]
- Goodman, L.B.; Loregian, A.; Perkins, G.A.; Nugent, J.; Buckles, E.L.; Mercorelli, B.; Kydd, J.H.; Palu, G.; Smith, K.C.; Osterrieder, N.; et al. A point mutation in a herpesvirus polymerase determines neuropathogenicity. PLoS Pathog. 2007, 3, e160. [Google Scholar] [CrossRef] [PubMed]
- D’Haene, B.; Vandesompele, J.; Hellemans, J. Accurate and objective copy number profiling using real-time quantitative PCR. Methods 2010, 50, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Allen, G.P.; Kydd, J.H.; Slater, J.D.; Smith, K.C. Equid herpesvirus-1 (EHV-1) and -4 (EHV-4) infections. In Infectious Diseases of Livestock; Coetzer, J.A.W., Tustin, R.C., Eds.; Oxford Press: Cape Town, South Africa, 2004; Chapter 76; pp. 829–859. [Google Scholar]
- Nei, M.; Jin, L. Variances of the average numbers of nucleotide substitutions within and between populations. Mol. Biol. Evol. 1989, 6, 290–300. [Google Scholar] [PubMed]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Escalera-Zamudio, M.; Zepeda-Mendoza, M.L.; Loza-Rubio, E.; Rojas-Anaya, E.; Mendez-Ojeda, M.L.; Arias, C.F.; Greenwood, A.D. The evolution of bat nucleic acid-sensing Toll-like receptors. Mol. Ecol. 2015, 24, 5899–5909. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Higgins, D.G.; Gibson, T.J. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Hohna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Azab, W.; Osterrieder, N. Equine herpesviruses type 1 (EHV-1) and 4 (EHV-4)—Masters of co-evolution and a constant threat to equids and beyond. Vet. Microbiol. 2013, 167, 123–134. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, A.; Fukushi, H.; Matsumura, T.; Yanai, T.; Masegi, T.; Hirai, K. Pathogenicity of a new neurotropic equine herpesvirus 9 (gazelle herpesvirus 1) in horses. J. Vet. Med. Sci. 2000, 62, 215–218. [Google Scholar] [CrossRef] [PubMed]
- Fukushi, H.; Taniguchi, A.; Yasuda, K.; Yanai, T.; Masegi, T.; Yamaguchi, T.; Hirai, K. A hamster model of equine herpesvirus 9 induced encephalitis. J. Neurovirol. 2000, 6, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Kodama, A.; Yanai, T.; Yomemaru, K.; Sakai, H.; Masegi, T.; Yamada, S.; Fukushi, H.; Kuraishi, T.; Hattori, S.; Kai, C. Acute neuropathogenicity with experimental infection of equine herpesvirus 9 in common marmosets (Callithrix jacchus). J. Med. Primatol. 2007, 36, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, A.; Fukushi, H.; Yanai, T.; Masegi, T.; Yamaguchi, T.; Hirai, K. Equine herpesvirus 9 induced lethal encephalomyelitis in experimentally infected goats. Arch. Virol. 2000, 145, 2619–2627. [Google Scholar] [CrossRef] [PubMed]
- Yanai, T.; Fujishima, N.; Fukushi, H.; Hirata, A.; Sakai, H.; Masegi, T. Experimental infection of equine herpesvirus 9 in dogs. Vet. Pathol. 2003, 40, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Yanai, T.; Tujioka, S.; Sakai, H.; Fukushi, H.; Hirai, K.; Masegi, T. Experimental infection with equine herpesvirus 9 (EHV-9) in cats. J. Comp. Pathol. 2003, 128, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Bowden, R.; Sakaoka, H.; Donnelly, P.; Ward, R. High recombination rate in herpes simplex virus type 1 natural populations suggests significant co-infection. Infect. Genet. Evol. 2004, 4, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Norberg, P. Divergence and genotyping of human alpha-herpesviruses: an overview. Infect. Genet. Evol. 2010, 10, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Norberg, P.; Bergstrom, T.; Liljeqvist, J.A. Genotyping of clinical herpes simplex virus type 1 isolates by use of restriction enzymes. J. Clin. Microbiol. 2006, 44, 4511–4514. [Google Scholar] [CrossRef] [PubMed]
| Animal Species | Sample ID | Sex | Age (Years) | Observations | Infection |
|---|---|---|---|---|---|
| Plains zebra | WP1 | f | 2 | unknown | Lytic EHV-9 |
| Plains zebra | CP4 | m | 24 | Signs of ataxia and tremors without improvement after the course of medication (complete rest and anti-inflammatory drug, Meloxicam) | Lytic EHV-1+ Latent EHV-9 |
| Plains zebra | CP5 | m | 2 | Cardiomyopathy was noticed during necropsy | Latent zebra-borne EHV-1 |
| Grevy’s zebra | CG2 | f | 23 | Found lying in the barn. No clinical signs were observed. Intramuscular bleeding along the femoral shaft was found during necropsy | Lytic EHV-9 |
| Grevy’s zebra | CG3 | f | 18 | Central nervous disorders | Lytic EHV-9 |
| Grevy’s zebra | CG6 | m | 11 | Massive intra-abdominal hemorrhage followed by hypovolemic shock as a result of surgical complications | Latent EHV-9 |
| Samples ID | Viral Genomic DNA (gB), (copies/million cells) | Transcriptional Activity of gB (mRNA) | Transcriptional Activity of LATs (copies/million cells) | Virus Species |
|---|---|---|---|---|
| CP4 | 8 | Negative | 107 | EHV-9 |
| CP5 | 65.3 | Negative | 124 | EHV-1 |
| CG6 | 33.3 | Negative | 174 | EHV-9 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelgawad, A.; Damiani, A.; Ho, S.Y.W.; Strauss, G.; Szentiks, C.A.; East, M.L.; Osterrieder, N.; Greenwood, A.D. Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections. Viruses 2016, 8, 262. https://doi.org/10.3390/v8090262
Abdelgawad A, Damiani A, Ho SYW, Strauss G, Szentiks CA, East ML, Osterrieder N, Greenwood AD. Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections. Viruses. 2016; 8(9):262. https://doi.org/10.3390/v8090262
Chicago/Turabian StyleAbdelgawad, Azza, Armando Damiani, Simon Y. W. Ho, Günter Strauss, Claudia A. Szentiks, Marion L. East, Nikolaus Osterrieder, and Alex D. Greenwood. 2016. "Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections" Viruses 8, no. 9: 262. https://doi.org/10.3390/v8090262
APA StyleAbdelgawad, A., Damiani, A., Ho, S. Y. W., Strauss, G., Szentiks, C. A., East, M. L., Osterrieder, N., & Greenwood, A. D. (2016). Zebra Alphaherpesviruses (EHV-1 and EHV-9): Genetic Diversity, Latency and Co-Infections. Viruses, 8(9), 262. https://doi.org/10.3390/v8090262






