A Defective Interfering Influenza RNA Inhibits Infectious Influenza Virus Replication in Human Respiratory Tract Cells: A Potential New Human Antiviral
Abstract
:1. Introduction
2. Materials and Methods
2.1. Defective Interfering Virus
2.2. Cells
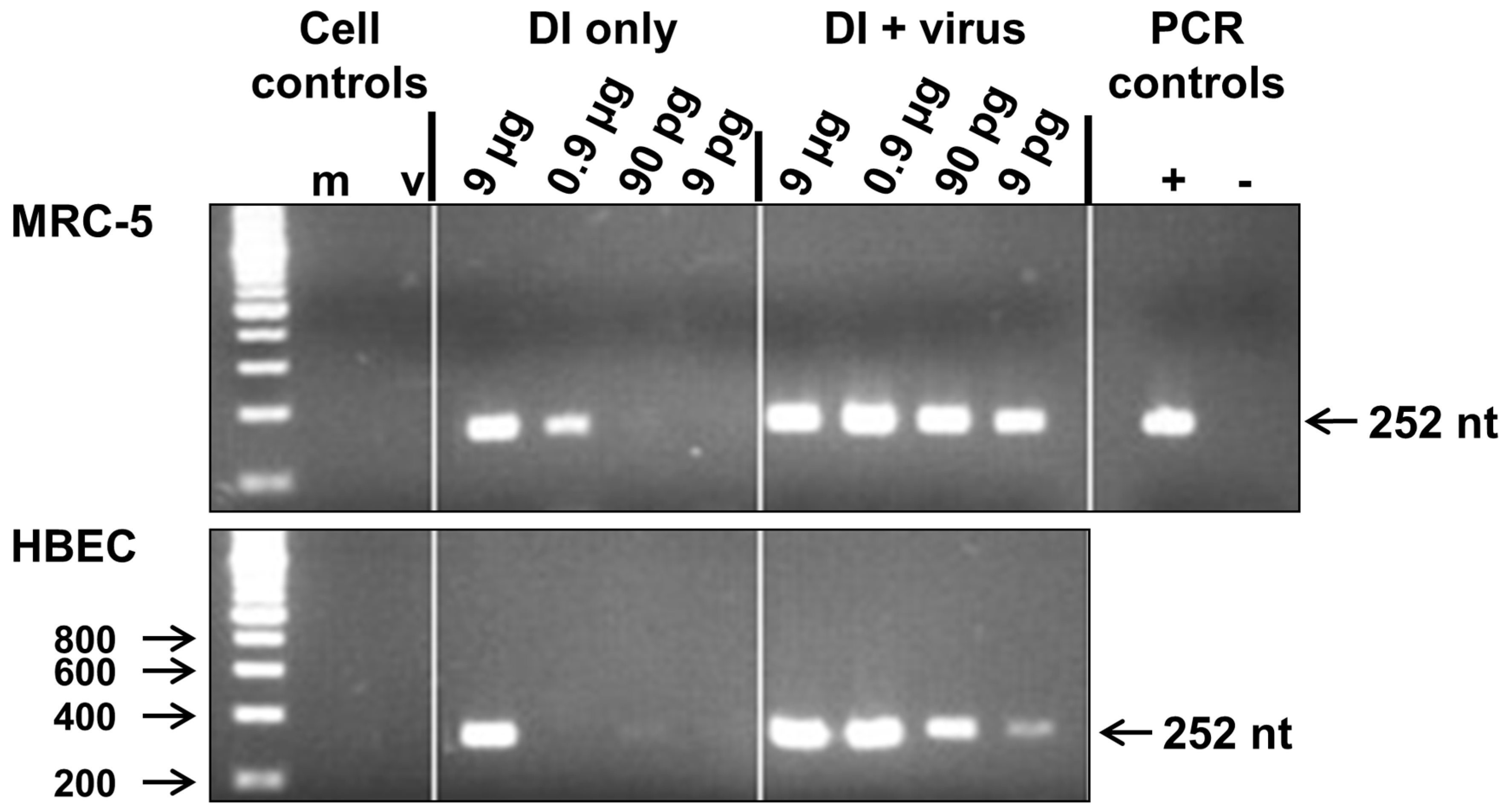
2.3. Reverse Transcription PCR (RT-PCR) Detection of 244/PR8 DI RNA
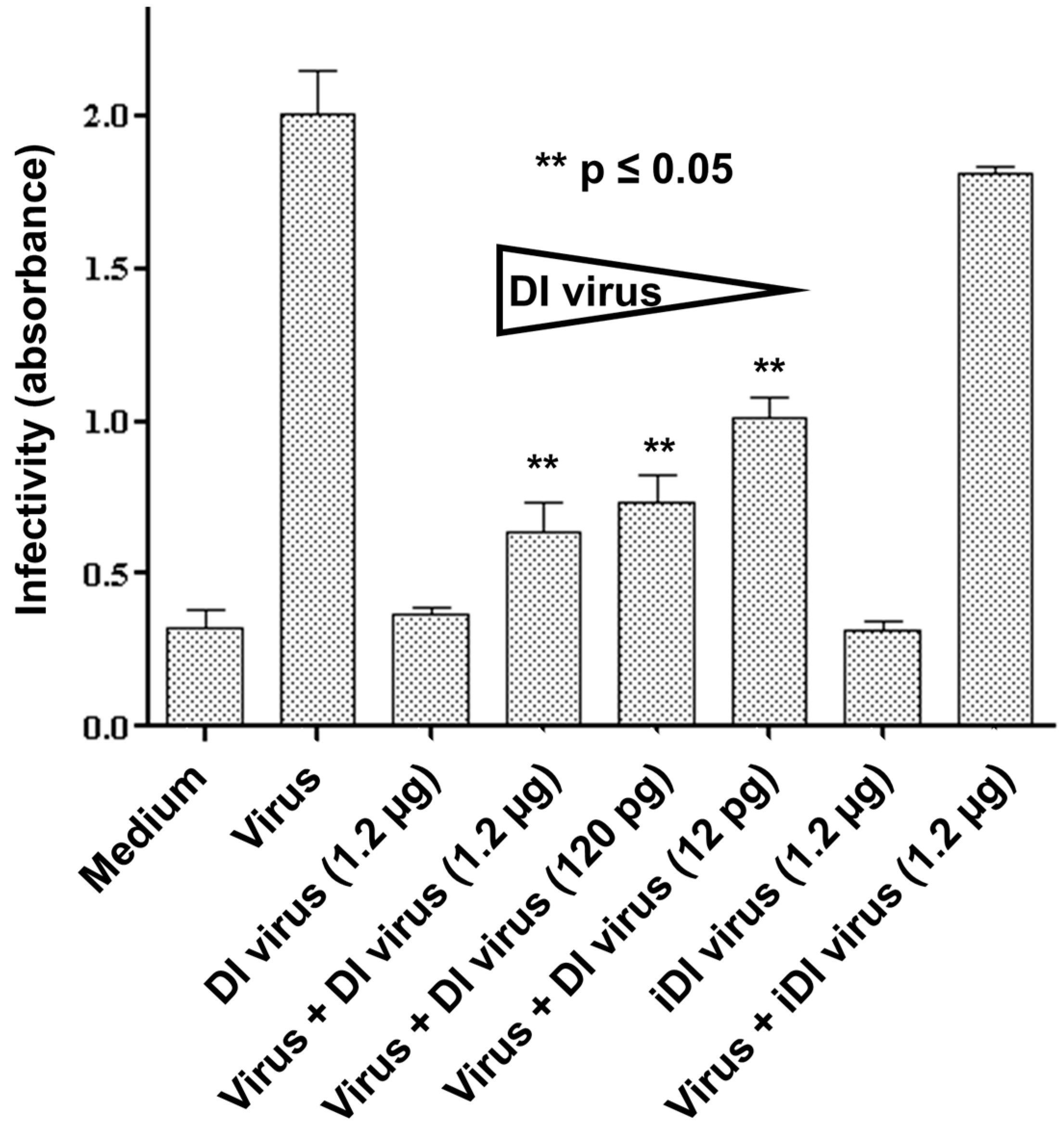
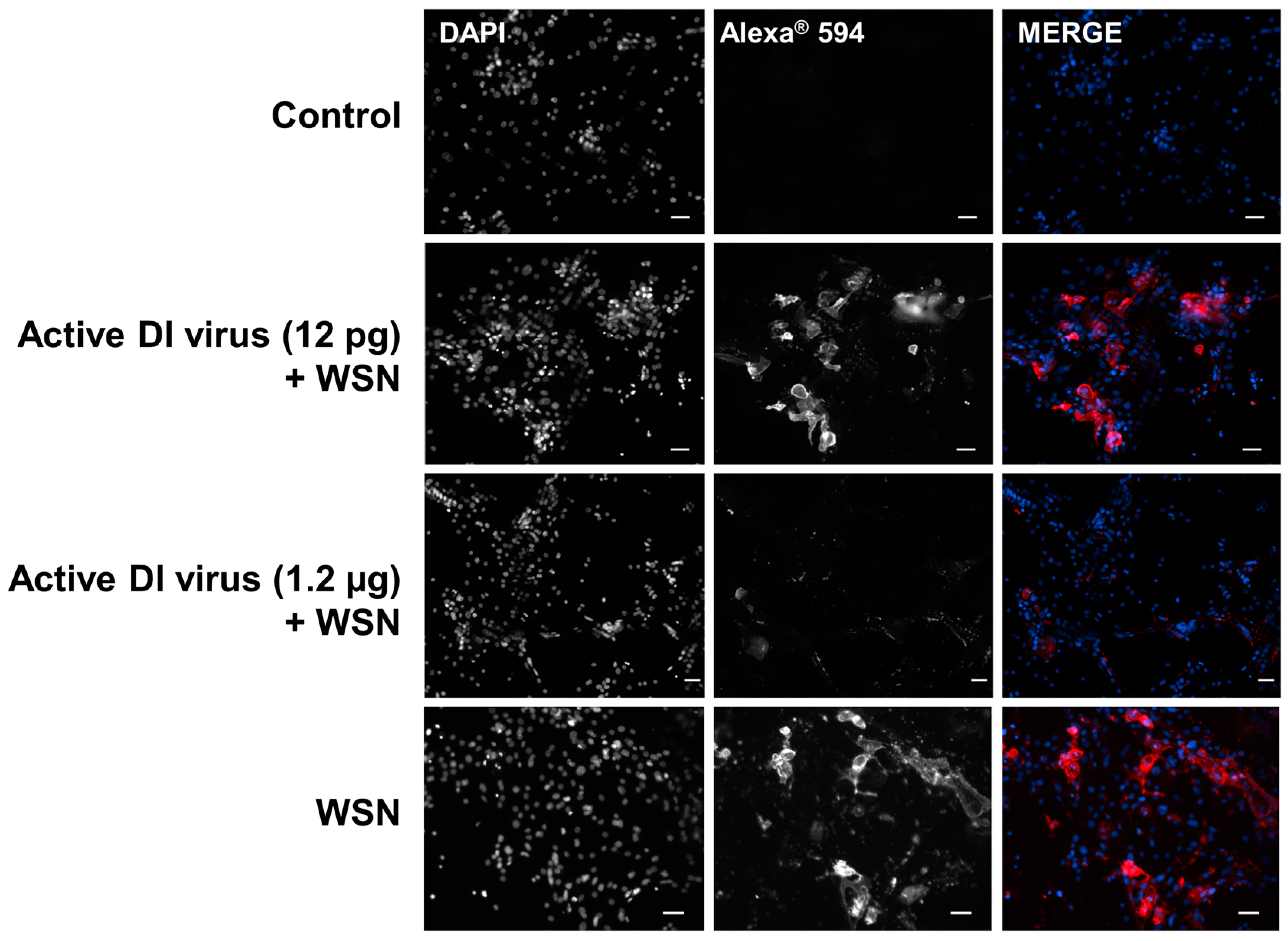
2.4. Inhibition of Infectious Virus Multiplication in Primary Nasal Basal Cells by 244/PR8 DI Virus
3. Results and Discussion
3.1. Replication of 244/PR8 DI RNA in MRC-5 and Human Bronchial Epithelial (HBE) Cells
3.2. Interference in Primary Human Nasal Basal Cells
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thompson, W.W.; Shay, D.K.; Weintraub, E.; Brammer, L.; Cox, N.; Anderson, L.J.; Fukuda, K. Mortality associated with influenza and respiratory syncytial virus in the united states. JAMA 2003, 289, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Thompson, W.W.; Viboud, C.G.; Ringholz, C.M.; Cheng, P.Y.; Steiner, C.; Abedi, G.R.; Anderson, L.J.; Brammer, L.; Shay, D.K. Hospitalizations associated with influenza and respiratory syncytial virus in the united states, 1993–2008. Clin. Infect Dis. 2012, 54, 1427–1436. [Google Scholar] [CrossRef] [PubMed]
- Chung, J.R.; Flannery, B.; Thompson, M.G.; Gaglani, M.; Jackson, M.L.; Monto, A.S.; Nowalk, M.P.; Talbot, H.K.; Treanor, J.J.; Belongia, E.A.; et al. Seasonal effectiveness of live attenuated and inactivated influenza vaccine. Pediatrics 2016, 137, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Flannery, B.; Clippard, J.; Zimmerman, R.K.; Nowalk, M.P.; Jackson, M.L.; Jackson, L.A.; Monto, A.S.; Petrie, J.G.; McLean, H.Q.; Belongia, E.A.; et al. Early estimates of seasonal influenza vaccine effectiveness—United States, January 2015. MMWR 2015, 64, 10–15. [Google Scholar] [PubMed]
- Gaglani, M.; Pruszynski, J.; Murthy, K.; Clipper, L.; Robertson, A.; Reis, M.; Chung, J.R.; Piedra, P.A.; Avadhanula, V.; Nowalk, M.P.; et al. Influenza vaccine effectiveness against 2009 pandemic influenza A(H1N1) virus differed by vaccine type during 2013–2014 in the United States. J. Infect. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Gilca, R.; Skowronski, D.M.; Douville-Fradet, M.; Amini, R.; Boulianne, N.; Rouleau, I.; Martineau, C.; Charest, H.; De Serres, G. Mid-season estimates of influenza vaccine effectiveness against influenza A(H3N2) hospitalization in the elderly in Quebec, Canada, January 2015. PLoS ONE 2015, 10, e0132195. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, N.J.; Easton, A.J. Defective interfering influenza virus RNAs: Time to reevaluate their clinical potential as broad-spectrum antivirals? J. Virol. 2014, 88, 5217–5227. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, N.J.; Easton, A.J. Cloned defective interfering influenza RNA and a possible pan-specific treatment of respiratory virus diseases. Viruses 2015, 7, 3768–3788. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.S.; Baltimore, D. Defective viral particles and viral disease processes. Nature 1970, 226, 325–327. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.S.; Greenawalt, J.W.; Wagner, R.R. Defective t particles of vesicular stomatitis virus. I. Preparation, morphology, and some biologic properties. Virology 1966, 30, 161–172. [Google Scholar] [CrossRef]
- Huang, A.S. Defective interfering viruses. Annu. Rev. Microbiol. 1973, 27, 101–117. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Lott, W.B.; Lowry, K.; Jones, A.; Thu, H.M.; Aaskov, J. Defective interfering viral particles in acute dengue infections. PLoS ONE 2011, 6, e19447. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.T.; Lin, M.H.; Chen, D.S.; Shih, C. A defective interference-like phenomenon of human hepatitis B virus in chronic carriers. J. Virol. 1998, 72, 578–584. [Google Scholar] [PubMed]
- Yeh, C.T.; Lu, S.C.; Chu, C.M.; Liaw, Y.F. Molecular cloning of a defective hepatitis C virus genome from the ascitic fluid of a patient with hepatocellular carcinoma. J. Gen. Virol. 1997, 78, 2761–2770. [Google Scholar] [CrossRef] [PubMed]
- Nüesch, J.P.; de Chastonay, J.; Siegl, G. Detection of defective genomes in hepatitis A virus particles present in clinical specimens. J. Gen. Virol. 1989, 70, 3475–3480. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.D.; Meng, B.; Marriott, A.C.; Easton, A.J.; Dimmock, N.J. Defective interfering virus protects elderly mice from influenza. Virol. J. 2011, 8, 212. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, N.J.; Dove, B.K.; Scott, P.D.; Meng, B.; Taylor, I.; Cheung, L.; Hallis, B.; Marriott, A.C.; Carroll, M.W.; Easton, A.J. Cloned defective interfering influenza virus protects ferrets from pandemic 2009 influenza A virus and allows protective immunity to be established. PLoS ONE 2012, 7, e49394. [Google Scholar] [CrossRef] [PubMed]
- Dimmock, N.J.; Dove, B.K.; Meng, B.; Scott, P.D.; Taylor, I.; Cheung, L.; Hallis, B.; Marriott, A.C.; Carroll, M.W.; Easton, A.J. Comparison of the protection of ferrets against pandemic 2009 influenza A virus (H1N1) by 244 DI influenza virus and oseltamivir. Antivir. Res. 2012, 96, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Easton, A.J.; Scott, P.D.; Edworthy, N.L.; Meng, B.; Marriott, A.C.; Dimmock, N.J. A novel broad-spectrum treatment for respiratory virus infections: Influenza-based defective interfering virus provides protection against pneumovirus infection in vivo. Vaccine 2011, 29, 2777–2784. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.D.; Meng, B.; Marriott, A.C.; Easton, A.J.; Dimmock, N.J. Defective interfering influenza A virus protects in vivo against disease caused by a heterologous influenza A virus. J. Gen. Virol. 2011, 92, 2122–2132. [Google Scholar] [CrossRef] [PubMed]
- Duhaut, S.D.; Dimmock, N.J. Heterologous protection of mice from a lethal human H1N1 influenza A virus infection by H3N8 equine defective interfering virus: Comparison of defective RNA sequences isolated from the DI inoculum and mouse lung. Virology 1998, 248, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Jennings, P.A.; Finch, J.T.; Winter, G.; Robertson, J.S. Does the higher order structure of the influenza virus ribonucleoprotein guide sequence rearrangements in influenza viral RNA? Cell 1983, 34, 619–627. [Google Scholar] [CrossRef]
- Dimmock, N.J.; Rainsford, E.W.; Scott, P.D.; Marriott, A.C. Influenza virus protecting RNA: An effective prophylactic and therapeutic antiviral. J. Virol. 2008, 82, 8570–8578. [Google Scholar] [CrossRef] [PubMed]
- Scott, P.D.; Meng, B.; Marriott, A.C.; Easton, A.J.; Dimmock, N.J. Defective interfering influenza virus confers only short-lived protection against influenza virus disease: Evidence for a role for adaptive immunity in DI virus-mediated protection in vivo. Vaccine 2011, 29, 6584–6591. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.M.; Kulkarni, H.; Radhakrishnan, P.; Rutman, A.; Bankart, M.J.; Williams, G.; Hirst, R.A.; Easton, A.J.; Andrew, P.W.; O’Callaghan, C. Ciliary dyskinesia is an early feature of respiratory syncytial virus infection. Eur. Respir. J. 2014, 43, 485–496. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smith, C.M.; Scott, P.D.; O’Callaghan, C.; Easton, A.J.; Dimmock, N.J. A Defective Interfering Influenza RNA Inhibits Infectious Influenza Virus Replication in Human Respiratory Tract Cells: A Potential New Human Antiviral. Viruses 2016, 8, 237. https://doi.org/10.3390/v8080237
Smith CM, Scott PD, O’Callaghan C, Easton AJ, Dimmock NJ. A Defective Interfering Influenza RNA Inhibits Infectious Influenza Virus Replication in Human Respiratory Tract Cells: A Potential New Human Antiviral. Viruses. 2016; 8(8):237. https://doi.org/10.3390/v8080237
Chicago/Turabian StyleSmith, Claire M., Paul D. Scott, Christopher O’Callaghan, Andrew J. Easton, and Nigel J. Dimmock. 2016. "A Defective Interfering Influenza RNA Inhibits Infectious Influenza Virus Replication in Human Respiratory Tract Cells: A Potential New Human Antiviral" Viruses 8, no. 8: 237. https://doi.org/10.3390/v8080237
APA StyleSmith, C. M., Scott, P. D., O’Callaghan, C., Easton, A. J., & Dimmock, N. J. (2016). A Defective Interfering Influenza RNA Inhibits Infectious Influenza Virus Replication in Human Respiratory Tract Cells: A Potential New Human Antiviral. Viruses, 8(8), 237. https://doi.org/10.3390/v8080237





