Properties of African Cassava Mosaic Virus Capsid Protein Expressed in Fission Yeast
Abstract
:1. Introduction
2. Materials and Methods
2.1. Construction of Expression Plasmid
2.2. Expression in Schizosaccharomyces pombe (S. pombe)
2.3. Preparation of Cell Extracts
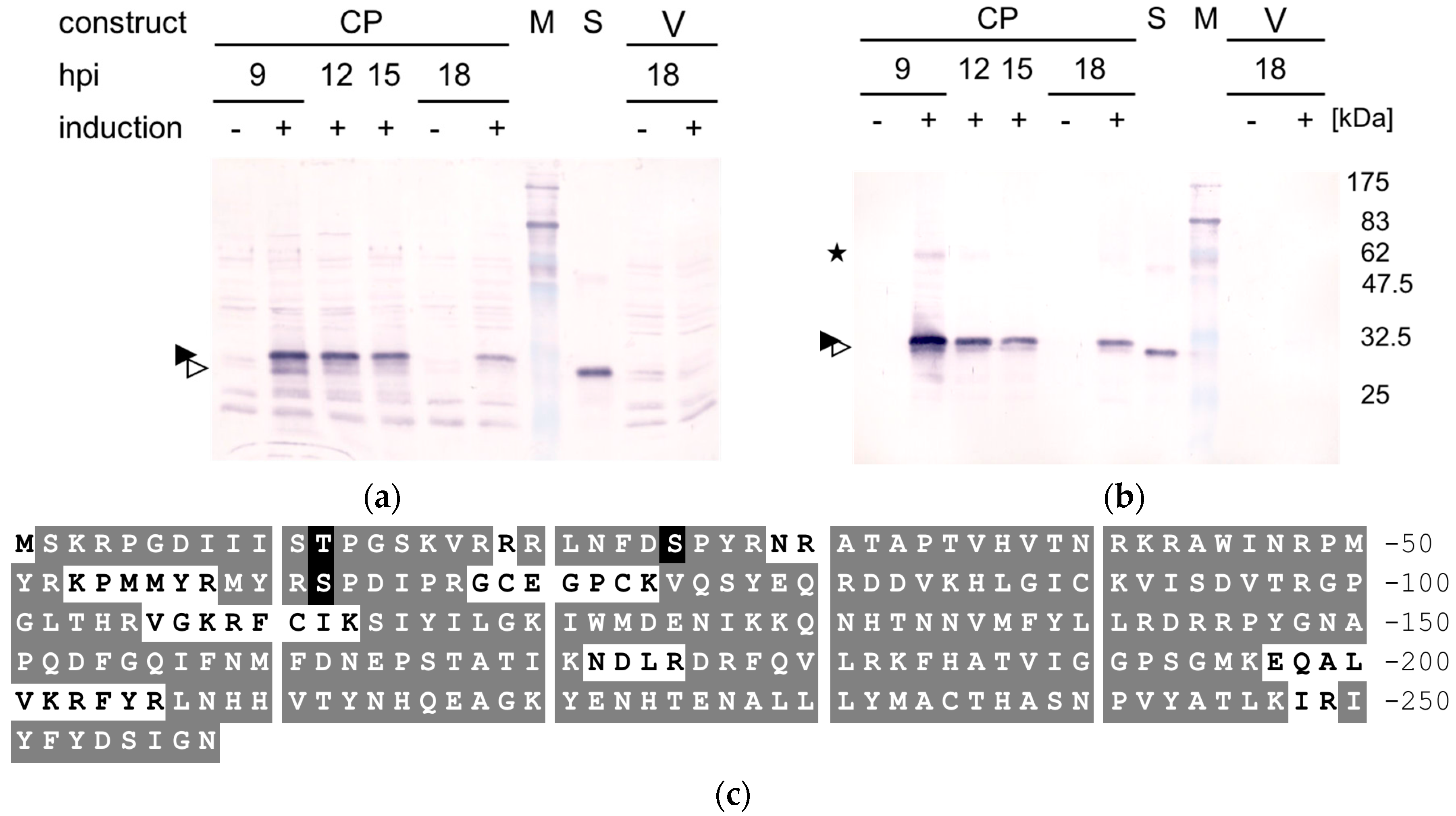
2.4. Western Blot Analysis
2.5. Mass Spectrometry
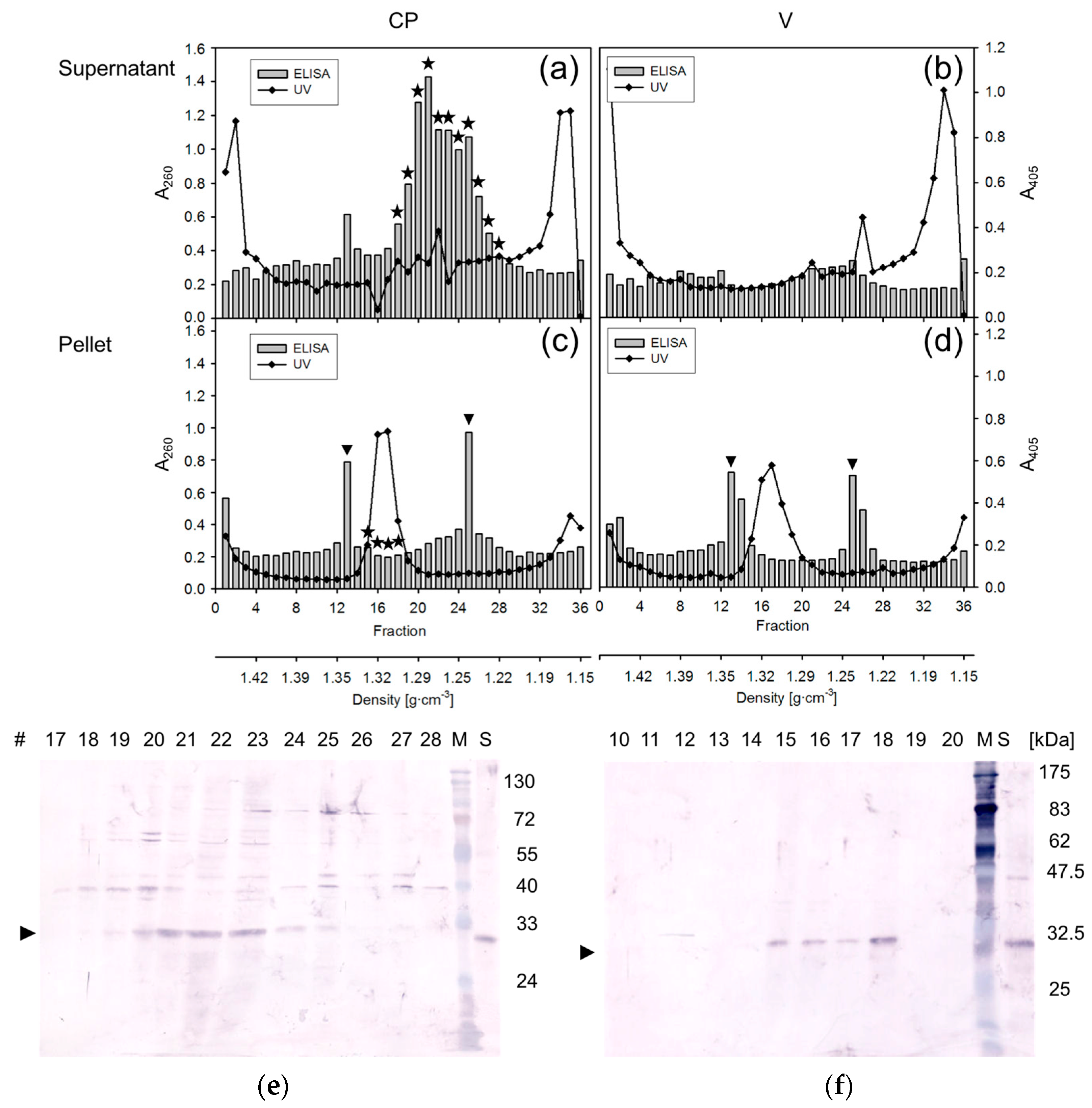
2.6. Density Gradient Centrifugation
2.7. Solubilization of Capsid Proteins (CPs)
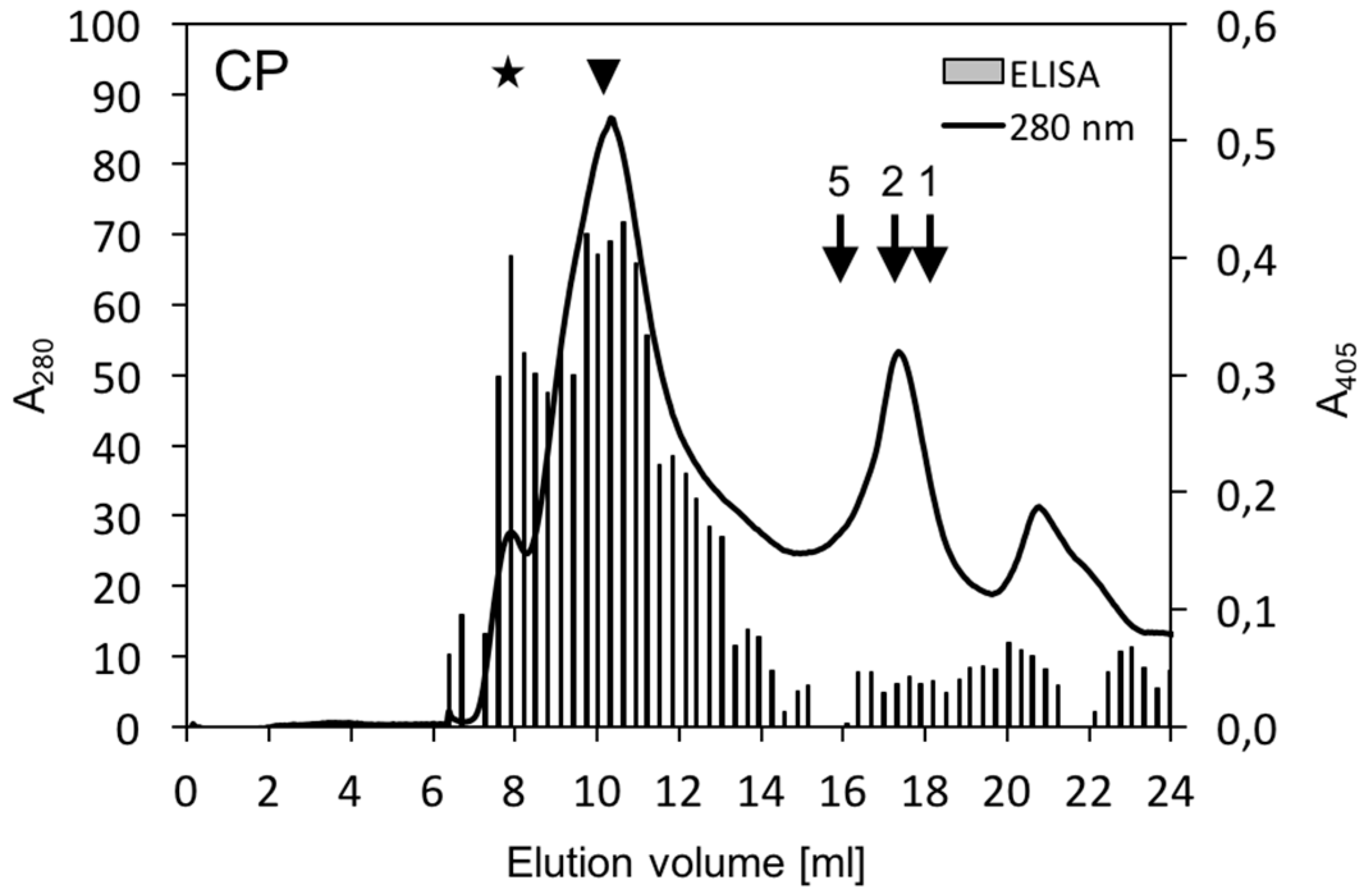
2.8. Size-Exclusion Chromatography
2.9. Enzyme-Linked Immunosorbent Assay (ELISA)
2.10. Preparation of ssDNA
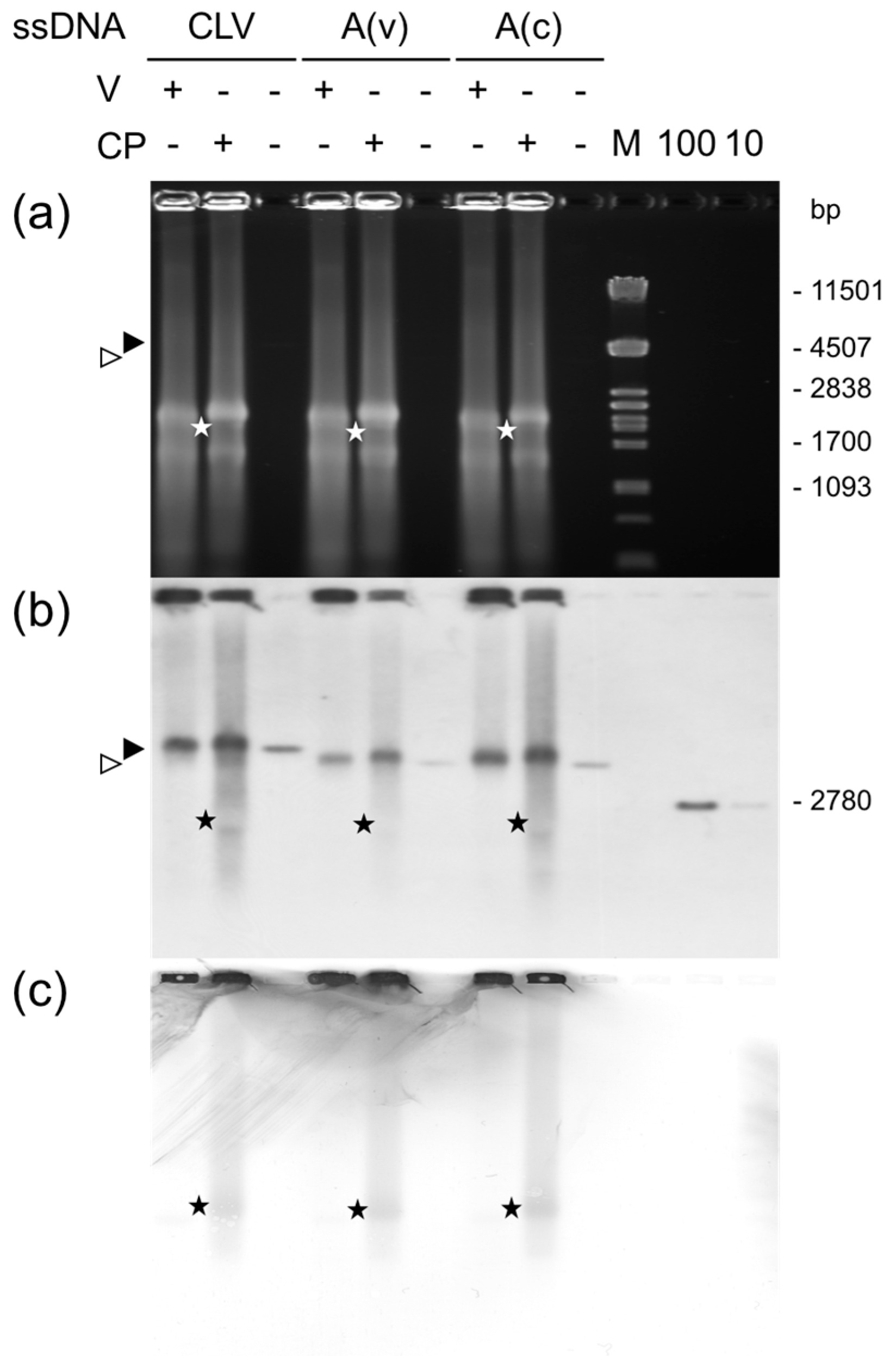
2.11. Binding Assays
2.11.1. Experimental Set 1
2.11.2. Experimental Set 2
3. Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Varsani, A.; Navas-Castillo, J.; Moriones, E.; Hernández-Zepeda, C.; Idris, A.; Brown, J.K.; Murilo Zerbini, F.; Martin, D.P. Establishment of three new genera in the family Geminiviridae: Becurtovirus, Eragrovirus and Turncurtovirus. Arch. Virol. 2014, 159, 2193–2203. [Google Scholar] [CrossRef] [PubMed]
- Legg, J.P.; Jeremiah, S.C.; Obiero, H.M.; Maruthi, M.N.; Ndyetabula, I.; Okao-Okuja, G.; Bouwmeester, H.; Bigirimana, S.; Tata-Hangy, W.; Gashaka, G.; et al. Comparing the regional epidemiology of the cassava mosaic and cassava brown streak virus pandemics in Africa. Virus Res. 2011, 159, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Patil, B.L.; Fauquet, C.M. Cassava mosaic geminiviruses: Actual knowledge and perspectives. Mol. Plant Pathol. 2009, 10, 685–701. [Google Scholar] [CrossRef] [PubMed]
- Rybicki, E.P. A Top Ten list for economically important plant viruses. Arch. Virol. 2015, 160, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Briddon, R.W.; Watts, J.; Markham, P.G.; Stanley, J. The coat protein of beet curly top virus is essential for infectivity. Virology 1989, 172, 628–633. [Google Scholar] [CrossRef]
- Briddon, R.W.; Pinner, M.S.; Stanley, J.; Markham, P.G. Geminivirus coat protein gene replacement alters insect specificity. Virology 1990, 177, 85–94. [Google Scholar] [CrossRef]
- Fischer, A.; Strohmeier, S.; Krenz, B.; Jeske, H. Evolutionary liberties of the Abutilon mosaic virus cluster. Virus Genes 2014. [Google Scholar] [CrossRef] [PubMed]
- Harrison, B.D.; Barker, H.; Bock, K.R.; Guthrie, E.J.; Meredith, G.; Atkinson, M. Plant-viruses with circular single-stranded DNA. Nature 1977, 270, 760–762. [Google Scholar] [CrossRef]
- Höhnle, M.; Höfer, P.; Bedford, I.D.; Briddon, R.W.; Markham, P.G.; Frischmuth, T. Exchange of three amino acids in the coat protein results in efficient whitefly transmission of a nontransmissible Abutilon mosaic virus isolate. Virology 2001, 290, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Kheyr-Pour, A.; Bananej, K.; Dafalla, G.A.; Caciagli, P.; Noris, E.; Ahoonmanesh, A.; Lecoq, H.; Gronenborn, B. Watermelon chlorotic stunt virus from the Sudan and Iran: Sequence comparison and identification of a whitefly-transmission determinant. Phytopathology 2000, 90, 629–635. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Briddon, R.W.; Bedford, I.D.; Pinner, M.S.; Markham, P.G. Identification of genes directly and indirectly involved in the insect transmission of African cassava mosaic geminivirus by Bemisia tabaci. Virus Genes 1999, 18, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Noris, E.; Vaira, A.M.; Caciagli, P.; Masenga, V.; Gronenborn, B.; Accotto, G.P. Amino acids in the capsid protein of tomato yellow leaf curl virus that are crucial for systemic infection, particle formation, and insect transmission. J. Virol. 1998, 72, 10050–10057. [Google Scholar] [PubMed]
- Böttcher, B.; Unseld, S.; Ceulemans, H.; Russell, R.B.; Jeske, H. Geminate structures of African cassava mosaic virus. J. Virol. 2004, 78, 6709–6714. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Olson, N.H.; Baker, T.S.; Faulkner, L.; Agbandje-McKenna, M.; Boulton, M.; Davies, J.W.; McKenna, R. Structure of the Maize streak virus geminate particle. Virology 2001, 279, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Hull, R. Matthews’ Plant Virology; Academic Press: San Diego, CA, USA, 2002. [Google Scholar]
- Sitharam, M.; Agbandje-McKenna, M. Modeling virus self-assembly pathways: Avoiding dynamics using geometric constraint decomposition. J. Comput. Biol. 2006, 13, 1232–1265. [Google Scholar] [CrossRef] [PubMed]
- Kittelmann, K.; Jeske, H. Disassembly of African cassava mosaic virus. J. Gen. Virol. 2008, 89, 2029–2036. [Google Scholar] [CrossRef] [PubMed]
- Hallan, V.; Gafni, Y. Tomato yellow leaf curl virus (TYLCV) capsid protein (CP) subunit interactions: Implications for viral assembly. Arch. Virol. 2001, 146, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Yaakov, N.; Levy, Y.; Belausov, E.; Gaba, V.; Lapidot, M.; Gafni, Y. Effect of a single amino acid substitution in the NLS domain of Tomato yellow leaf curl virus-Israel (TYLCV-IL) capsid protein (CP) on its activity and on the virus life cycle. Virus Res. 2011, 158, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Malik, P.S.; Kumar, V.; Bagewadi, B.; Mukherjee, S.K. Interaction between coat protein and replication initiation protein of Mung bean yellow mosaic India virus might lead to control of viral DNA replication. Virology 2005, 337, 273–283. [Google Scholar] [CrossRef] [PubMed]
- Hehnle, S.; Wege, C.; Jeske, H. The interaction of DNA with the movement proteins of geminiviruses revisited. J. Virol. 2004, 78, 7698–7706. [Google Scholar] [CrossRef] [PubMed]
- Ingham, D.J.; Pascal, E.; Lazarowitz, S.G. Both bipartite geminivirus movement proteins define viral host range, but only BL1 determines viral pathogenicity. Virology 1995, 207, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.T.; Boulton, M.I.; Davies, J.W. Maize streak virus coat protein binds single- and double-stranded DNA in vitro. J. Gen. Virol. 1997, 78, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Palanichelvam, K.; Kunik, T.; Citovsky, V.; Gafni, Y. The capsid protein of tomato yellow leaf curl virus binds cooperatively to single-stranded DNA. J. Gen. Virol. 1998, 79, 2829–2833. [Google Scholar] [CrossRef] [PubMed]
- Priyadarshini, P.C.G.; Savithri, H.S. Kinetics of interaction of Cotton Leaf Curl Kokhran Virus-Dabawali (CLCuKV-Dab) coat protein and its mutants with ssDNA. Virology 2009, 386, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Kirthi, N.; Savithri, H.S. A conserved zinc finger motif in the coat protein of tomato leaf curl Bangalore virus is responsible for binding to ssDNA. Arch. Virol. 2003, 148, 2369–2380. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.W.; Ward, B.M.; Lazarowitz, S.G. The bipartite geminivirus coat protein aids BR1 function in viral movement by affecting the accumulation of viral single-stranded DNA. J. Virol. 1998, 72, 9247–9256. [Google Scholar] [PubMed]
- Unseld, S.; Höhnle, M.; Ringel, M.; Frischmuth, T. Subcellular targeting of the coat protein of African cassava mosaic geminivirus. Virology 2001, 286, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Unseld, S.; Frischmuth, T.; Jeske, H. Short deletions in nuclear targeting sequences of African cassava mosaic virus coat protein prevent geminivirus twinned particle formation. Virology 2004, 318, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Kotlizky, G.; Boulton, M.I.; Pitaksutheepong, C.; Davies, J.W.; Epel, B.L. Intracellular and intercellular movement of maize streak geminivirus V1 and V2 proteins transiently expressed as green fluorescent protein fusions. Virology 2000, 274, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Kunik, T.; Palanichelvam, K.; Czosnek, H.; Citovsky, V.; Gafni, Y. Nuclear import of the capsid protein of tomato yellow leaf curl virus (TYLCV) in plant and insect cells. Plant J. 1998, 13, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Kunik, T.; Mizrachy, L.; Citovsky, V.; Gafni, Y. Characterization of a tomato karyopherin alpha that interacts with the tomato yellow leaf curl virus (TYLCV) capsid protein. J. Exp. Bot. 1999, 50, 731–732. [Google Scholar] [CrossRef]
- Liu, H.; Boulton, M.I.; Thomas, C.L.; Prior, D.A.; Oparka, K.J.; Davies, J.W. Maize streak virus coat protein is karyophyllic and facilitates nuclear transport of viral DNA. Mol. Plant-Microbe Interact. 1999, 12, 894–900. [Google Scholar] [CrossRef] [PubMed]
- Wege, C.; Jeske, H. Abutilon mosaic geminivirus proteins expressed and phosphorylated in Escherichia coli. J. Phytopathol. 1998, 146, 613–621. [Google Scholar] [CrossRef]
- Frischmuth, S.; Wege, C.; Hülser, D.; Jeske, H. The movement protein BC1 promotes redirection of the nuclear shuttle protein BV1 of Abutilon mosaic geminivirus to the plasma membrane in fission yeast. Protoplasma 2007, 230, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Kittelmann, K.; Rau, P.; Gronenborn, B.; Jeske, H. Plant geminivirus Rep protein induces rereplication in fission yeast. J. Virol. 2009, 83, 6769–6778. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]
- Bjerrum, O.J. Buffer systems and transfer parameters for semidry electroblotting with a horizontal apparatus. In Proceedings of the fifth meeting of the International Electrophoresis Society, 1st ed.; Dunn, M.J., Ed.; VCH Verlagsgesellschaft: Weinheim, Germany, 1987; Volume 8, pp. 315–327. [Google Scholar]
- Townsend, R.; Stanley, J.; Curson, S.J.; Short, M.N. Major polyadenylated transcripts of cassava latent virus and location of the gene encoding coat protein. EMBO J. 1985, 4, 33–37. [Google Scholar] [PubMed]
- Klinkenberg, F.A.; Ellwood, S.; Stanley, J. Fate of African cassava mosaic virus coat protein deletion mutants after agroinoculation. J. Gen. Virol. 1989, 70, 1837–1844. [Google Scholar] [CrossRef]
- Stanley, J.; Gay, M.R. Nucleotide sequence of cassava latent virus DNA. Nature 1983, 305, 260–262. [Google Scholar] [CrossRef]
- Chomczynski, P.; Qasba, P.K. Alkaline transfer of DNA to plastic membrane. Biochem. Biophys. Res. Commun. 1984, 122, 340–344. [Google Scholar] [CrossRef]
- Maundrell, K. NMT1 of fission yeast. J. Biol. Chem. 1990, 265, 10857–10864. [Google Scholar] [PubMed]
- Abouzid, A.M.; Frischmuth, T.; Jeske, H. A putative replicative form of the Abutilon mosaic virus (gemini group) in a chromatin-like structure. Mol. Gen. Genet. 1988, 212, 252–258. [Google Scholar] [CrossRef]
- Pilartz, M.; Jeske, H. Abutilon mosaic geminivirus double-stranded DNA is packed into minichromosomes. Virology 1992, 189, 800–802. [Google Scholar] [CrossRef]
- Aberle, H.J.; Rütz, M.L.; Karayavuz, M.; Frischmuth, S.; Wege, C.; Hülser, D.; Jeske, H. Localizing BC1 movement proteins of Abutilon mosaic geminivirus in yeasts by subcellular fractionation and freeze-fracture immunolabelling. Arch. Virol. 2002, 147, 103–107. [Google Scholar] [CrossRef]
- Frischmuth, S.; Kleinow, T.; Aberle, H.-J.; Wege, C.; Hülser, D.; Jeske, H. Yeast two-hybrid systems confirm the membrane-association and oligomerization of BC1 but do not detect an interaction of the movement proteins BC1 and BV1 of Abutilon mosaic geminivirus. Arch. Virol. 2004, 149, 2349–2364. [Google Scholar] [CrossRef] [PubMed]
- Hipp, K.; Rau, P.; Schäfer, B.; Gronenborn, B.; Jeske, H. The RXL motif of the African cassava mosaic virus Rep protein is necessary for rereplication of yeast DNA and viral infection in plants. Virology 2014, 462–463, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Kleinow, T.; Holeiter, G.; Nischang, M.; Stein, M.; Karayavuz, M.; Wege, C.; Jeske, H. Post-translational modifications of Abutilon mosaic virus movement protein (BC1) in fission yeast. Virus Res. 2008, 131, 86–94. [Google Scholar] [CrossRef] [PubMed]
- Forsburg, S.L. The best yeast? Trends Genet. 1999, 15, 340–344. [Google Scholar] [CrossRef]
- Margueron, R.; Reinberg, D. Chromatin structure and the inheritance of epigenetic information. Nat. Rev. Genet. 2010, 11, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Morris, C.A.; Moazed, D. Centromere assembly and propagation. Cell 2007, 128, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Russell, P.; Nurse, P. Schizosaccharomyces pombe and Saccharomyces cerevisiae: A look at yeasts divided. Cell 1986, 45, 781–782. [Google Scholar] [CrossRef]
- Volpe, T.A.; Kidner, C.; Hall, I.M.; Teng, G.; Grewal, S.I.S.; Martienssen, R.A. Regulation of heterochromatic silencing and histone H3 lysine-9 methylation by RNAi. Science 2002, 297, 1833–1837. [Google Scholar] [CrossRef] [PubMed]
- Wixon, J. Featured organism: Schizosaccharomyces pombe, the fission yeast. Comp. Funct. Genom. 2002, 3, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Abouzid, A.; Jeske, H. The purification and characterization of gemini particles from Abutilon mosaic virus infected Malvaceae. J. Phytopathol. 1986, 115, 344–353. [Google Scholar] [CrossRef]
- Jeske, H.; Gotthardt, D.; Kober, S. In planta cloning of geminiviral DNA: The true Sida micrantha mosaic virus. J. Virol. Methods 2010, 163, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Jovel, J.; Reski, G.; Rothenstein, D.; Ringel, M.; Frischmuth, T.; Jeske, H. Sida micrantha mosaic is associated with a complex infection of begomoviruses different from Abutilon mosaic virus. Arch. Virol. 2004, 149, 829–841. [Google Scholar] [CrossRef] [PubMed]
- Jovel, J.; Preiß, W.; Jeske, H. Characterization of DNA intermediates of an arising geminivirus. Virus Res. 2007, 130, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Gorovits, R.; Moshe, A.; Ghanim, M.; Czosnek, H. Recruitment of the host plant heat shock protein 70 by Tomato yellow leaf curl virus coat protein is required for virus infection. PLoS ONE 2013, 8, e70280. [Google Scholar] [CrossRef] [PubMed]
- Gorovits, R.; Moshe, A.; Kolot, M.; Sobol, I.; Czosnek, H. Progressive aggregation of Tomato yellow leaf curl virus coat protein in systemically infected tomato plants, susceptible and resistant to the virus. Virus Res. 2013, 171, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Stachorski, L.; (University of Stuttgart, Stuttgart, Germany). Personal Communication, 2011.
- McGarry, R.C.; Barron, Y.D.; Carvalho, M.F.; Hill, J.E.; Gold, D.; Cheung, E.; Kraus, W.L.; Lazarowitz, S.G. A novel Arabidopsis acetyltransferase interacts with the geminivirus movement protein NSP. Plant Cell 2003, 15, 1605–1618. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-M.; Crothers, D.M. The locus of sequence-directed and protein-induced DNA bending. Nature 1984, 308, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Ortega, M.E.; Catalano, C.E. Bacteriophage lambda gpNu1 and Escherichia coli IHF proteins cooperatively bind and bend viral DNA: Implications for the assembly of a genome-packaging motor. Biochemistry (Mosc.) 2006, 45, 5180–5189. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-L.; Wei, X.-M.; Ye, X.-D.; Xu, H.-X.; Zhou, X.-P.; Liu, S.-S.; Wang, X.-W. Expression and functional characterisation of a soluble form of Tomato yellow leaf curl virus coat protein. Pest Manag. Sci. 2014, 70, 1624–1631. [Google Scholar] [CrossRef] [PubMed]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hipp, K.; Schäfer, B.; Kepp, G.; Jeske, H. Properties of African Cassava Mosaic Virus Capsid Protein Expressed in Fission Yeast. Viruses 2016, 8, 190. https://doi.org/10.3390/v8070190
Hipp K, Schäfer B, Kepp G, Jeske H. Properties of African Cassava Mosaic Virus Capsid Protein Expressed in Fission Yeast. Viruses. 2016; 8(7):190. https://doi.org/10.3390/v8070190
Chicago/Turabian StyleHipp, Katharina, Benjamin Schäfer, Gabi Kepp, and Holger Jeske. 2016. "Properties of African Cassava Mosaic Virus Capsid Protein Expressed in Fission Yeast" Viruses 8, no. 7: 190. https://doi.org/10.3390/v8070190
APA StyleHipp, K., Schäfer, B., Kepp, G., & Jeske, H. (2016). Properties of African Cassava Mosaic Virus Capsid Protein Expressed in Fission Yeast. Viruses, 8(7), 190. https://doi.org/10.3390/v8070190





