Development of a Triple-Color Pseudovirion-Based Assay to Detect Neutralizing Antibodies against Human Papillomavirus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells, Plasmids, Serum Samples, HPV Antibody Standards, and HPV Vaccines
2.2. Serum Samples
2.3. Preparation and Titration of HPV Pseudovirions
2.4. Pseudovirion-Based Neutralization Assay
3. Results
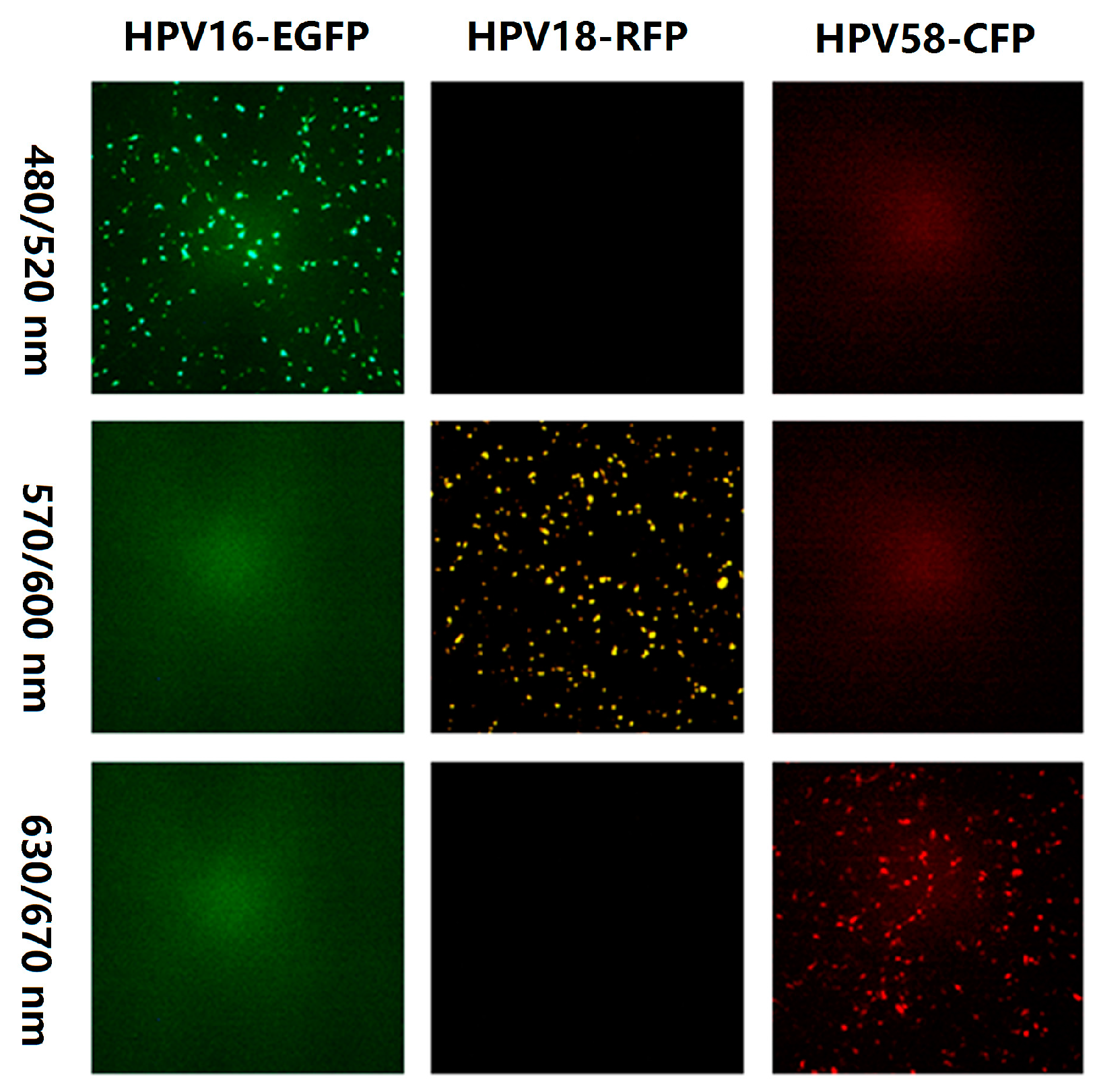
3.1. Selection of Fluorescent Proteins
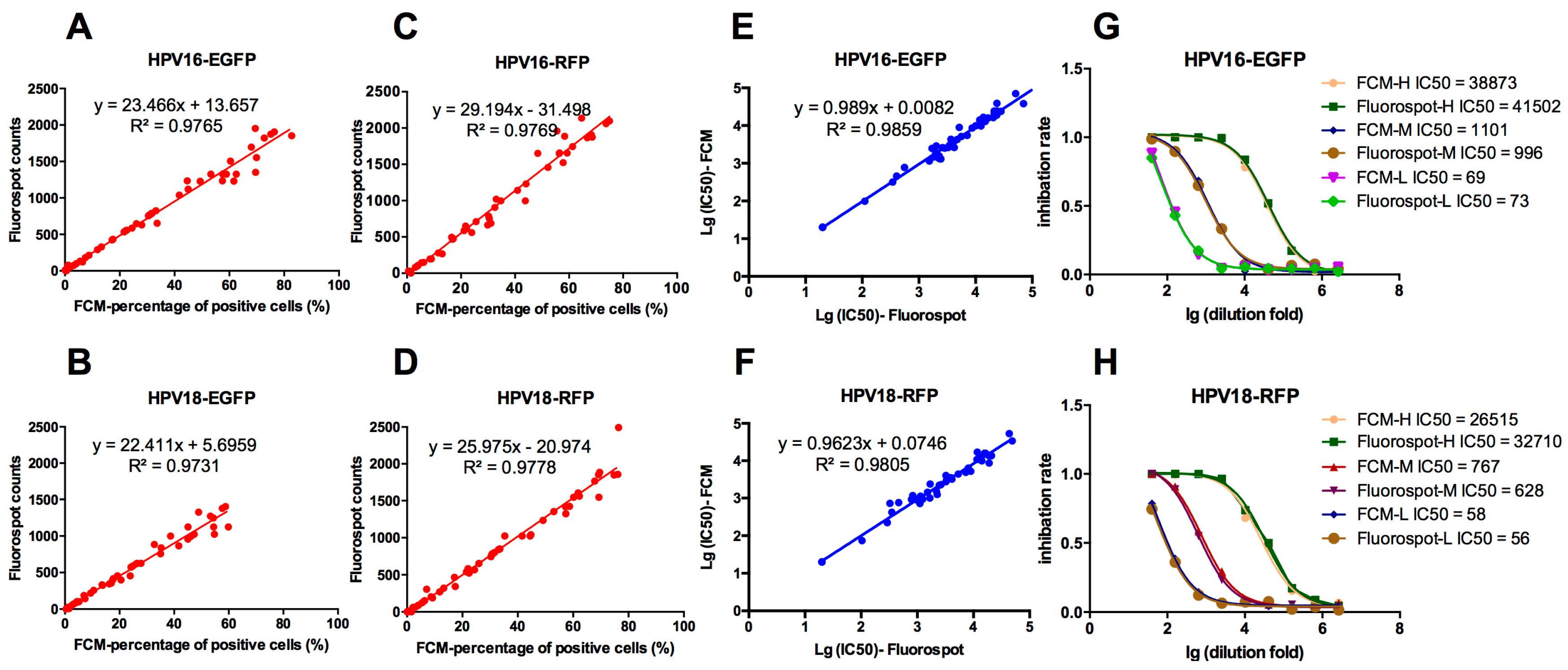
3.2. Correlation of the FluoroSpot and FCM Results
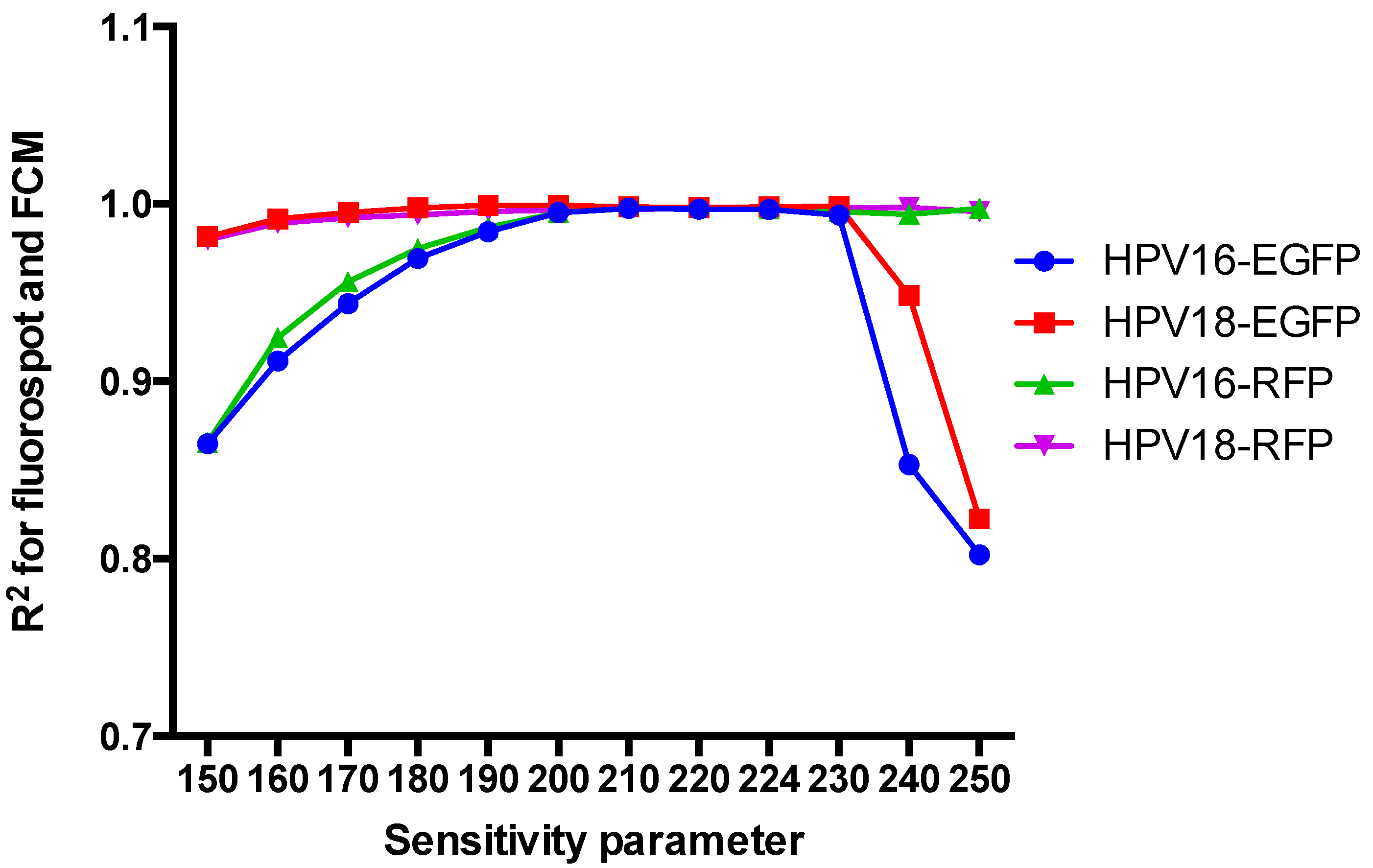
3.3. Impact of the Sensitivity Parameter on the Interpretation of the FluoroSpot Results
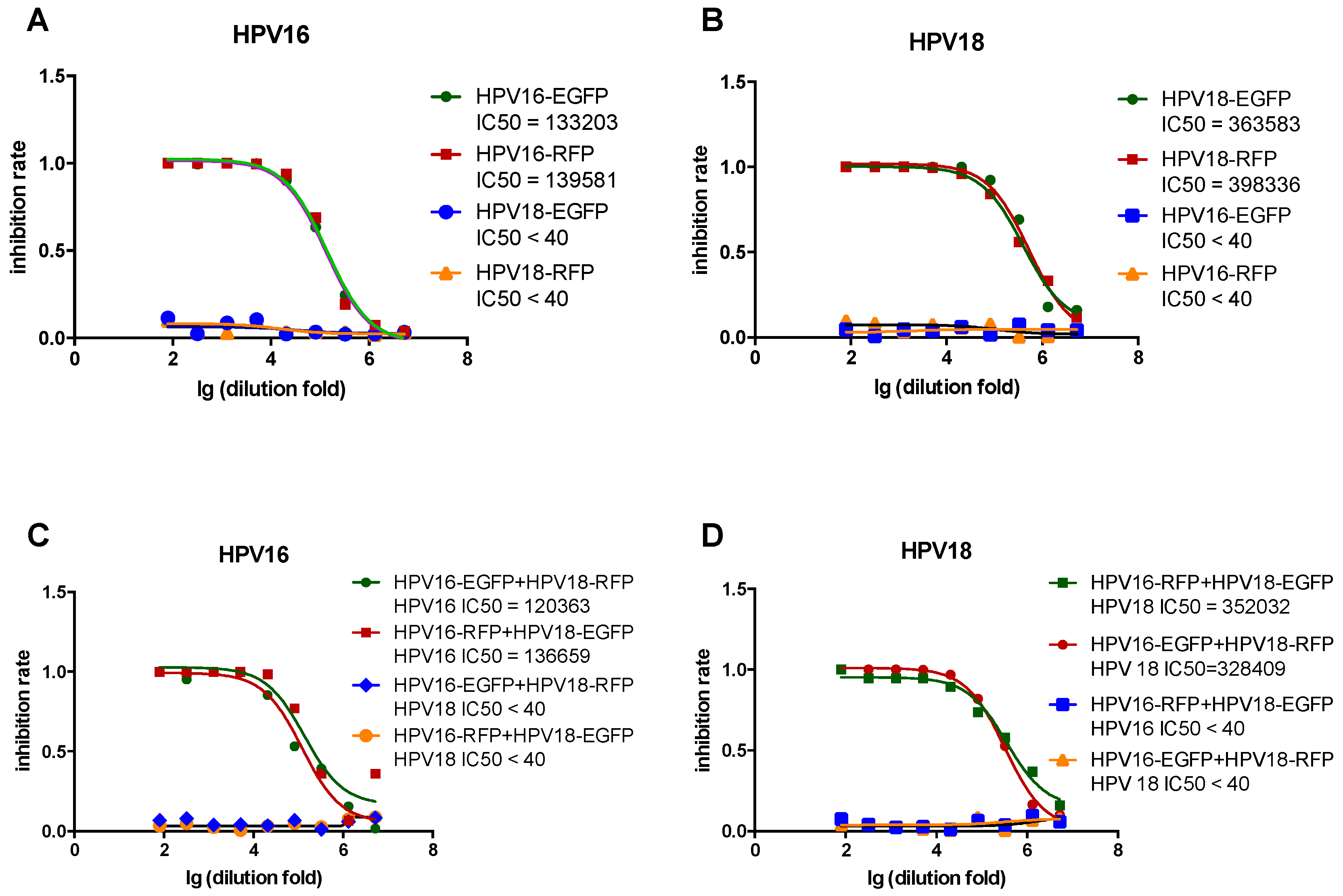
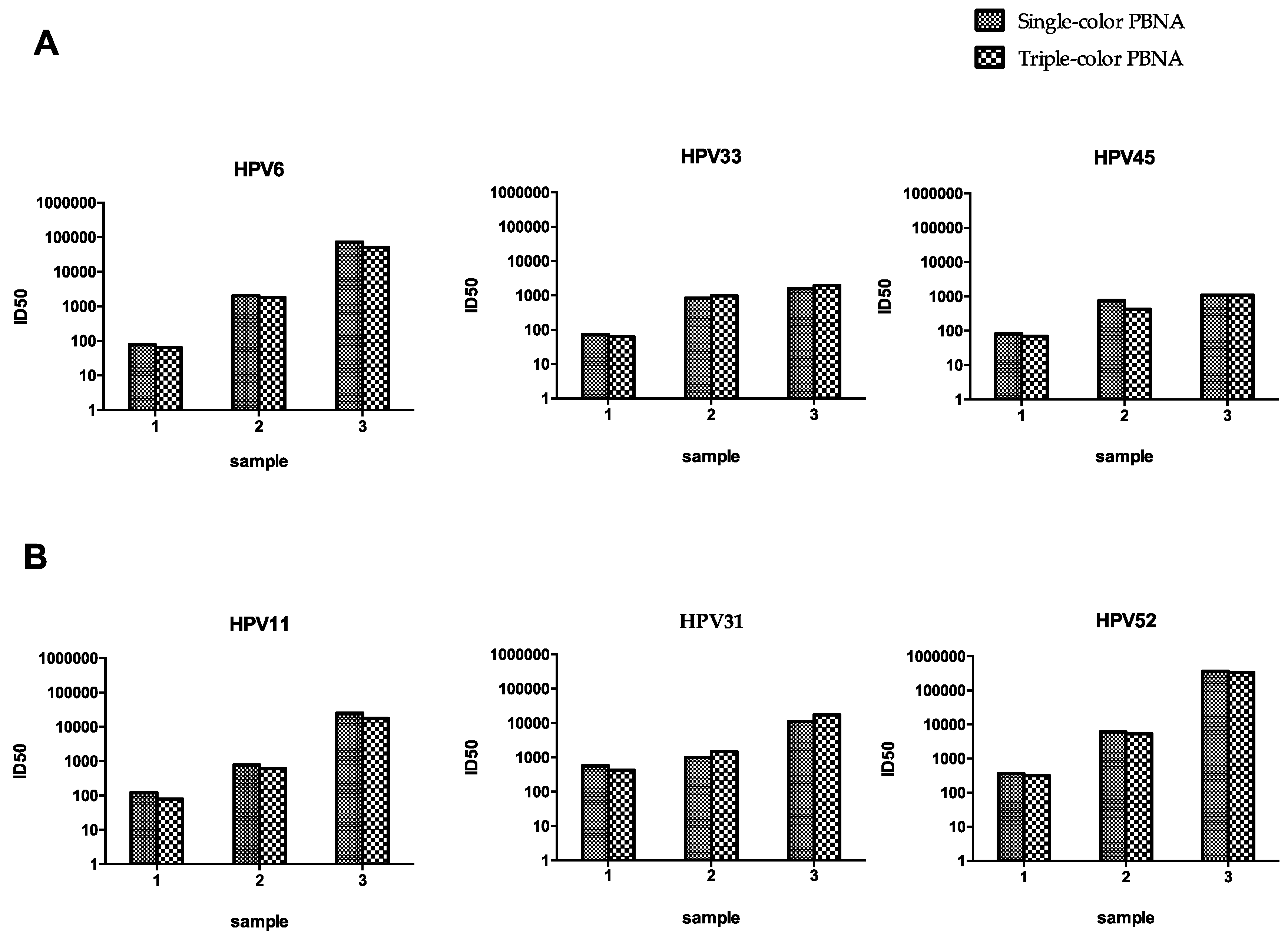
3.4. Comparison of Single- and Dual-Color PBNA
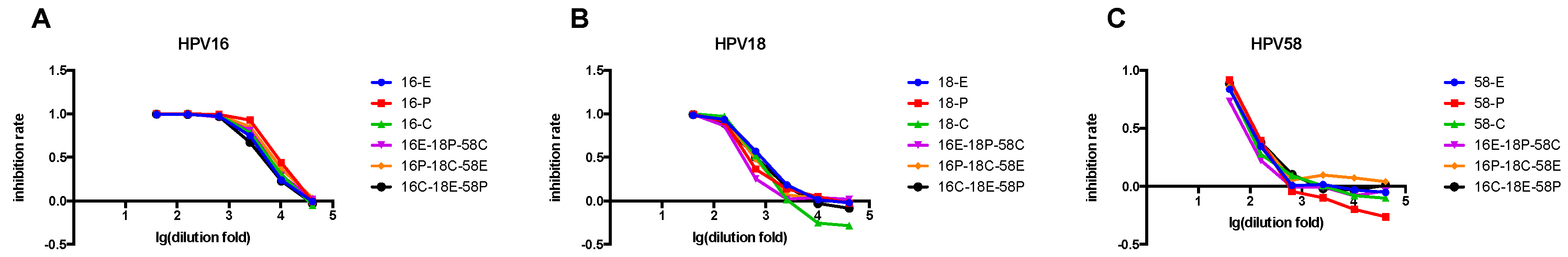
3.5. Comparison of Single- and Triple-Color PBNA
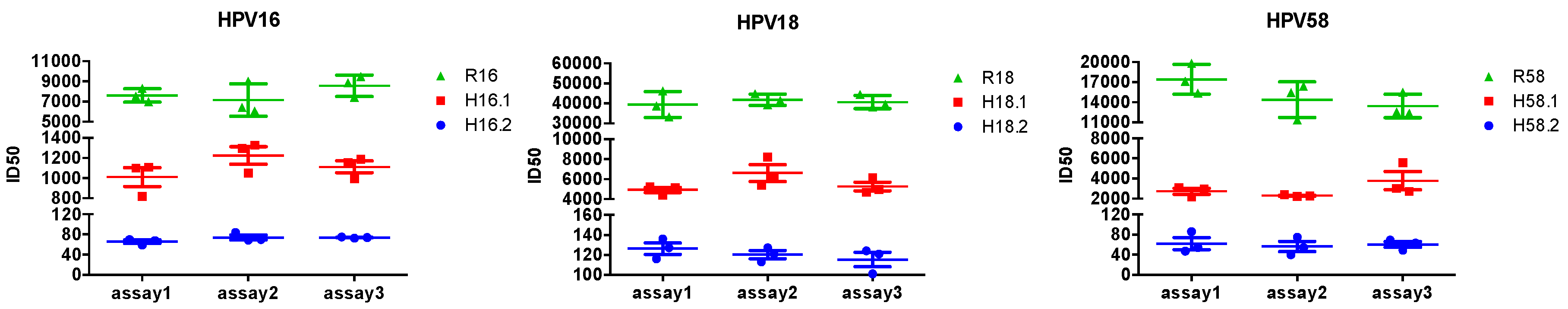
3.6. Reproducibility of the Triple-Color PBNA
3.7. Triple-Color PBNA for HPV6-33-45 and HPV11-31-52
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PBNA | Pseudovirion-based neutralization assay |
| HPV | Human papillomavirus |
| US | United States |
| FCM | Flow cytometry |
| FDA | Food and Drug Administration |
| cLIA | Competitive Luminex® immunoassay |
| VLP | Virus-like particle |
| VLP-MIA | Virus-like particle-based multiplex immunoassay |
| GST-L1-MIA | Glutathione S-transferase L1-based MIA |
| EGFP | Enhanced green fluorescent protein |
| GLuc | Gaussia luciferase |
| SEAP | Secreted alkaline phosphatase |
| HEPES | 4-(2-Hydroxyethyl)-1-piperazineethanesulfonic acid |
| RFP | Red fluorescent protein |
| E2-CFP | E2-crimson fluorescent protein |
| WHO | World Health Organization |
| NIBSC | National institute for Biological Standards and Control |
| YFP | Yellow fluorescent protein |
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Bravo, I.G.; Felez-Sanchez, M. Papillomaviruses: Viral evolution, cancer and evolutionary medicine. Evol. Med. Public Health 2015, 2015, 32–51. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, E.P. Prophylaxis of cervical cancer and related cervical disease: A review of the cost-effectiveness of vaccination against oncogenic HPV types. J. Manag. Care Pharm. 2010, 16, 217–230. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Lin, Z.-J.; Huang, S.-J.; Li, J.; Liu, X.-H.; Guo, M.; Zhang, J.; Xia, N.-S.; Pan, H.-R.; Wu, T.; Li, C.G. Correlation between ELISA and pseudovirion-based neutralisation assay for detecting antibodies against human papillomavirus acquired by natural infection or by vaccination. Hum. Vaccines Immunother. 2014, 10, 740–746. [Google Scholar] [CrossRef]
- Cassidy, B.; Schlenk, E.A. Uptake of the human papillomavirus vaccine: A review of the literature and report of a quality assurance project. J. Pediatr. Health Care 2012, 26, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Joura, E.A.; Giuliano, A.R.; Iversen, O.E.; Bouchard, C.; Mao, C.; Mehlsen, J.; Moreira, E.D., Jr.; Ngan, Y.; Petersen, L.K.; Lazcano-Ponce, E.; et al. A 9-valent HPV vaccine against infection and intraepithelial neoplasia in women. N. Engl. J. Med. 2015, 372, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Opalka, D.; Lachman, C.E.; MacMullen, S.A.; Jansen, K.U.; Smith, J.F.; Chirmule, N.; Esser, M.T. Simultaneous quantitation of antibodies to neutralizing epitopes on virus-like particles for human papillomavirus types 6, 11, 16, and 18 by a multiplexed luminex assay. Clin. Diagn. Lab. Immunol. 2003, 10, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Scherpenisse, M.; Mollers, M.; Schepp, R.M.; Boot, H.J.; de Melker, H.E.; Meijer, C.J.; Berbers, G.A.; van der Klis, F.R. Seroprevalence of seven high-risk HPV types in the Netherlands. Vaccine 2012, 30, 6686–6693. [Google Scholar] [CrossRef] [PubMed]
- Waterboer, T.; Sehr, P.; Michael, K.M.; Franceschi, S.; Nieland, J.D.; Joos, T.O.; Templin, M.F.; Pawlita, M. Multiplex human papillomavirus serology based on in situ-purified glutathione s-transferase fusion proteins. Clin. Chem. 2005, 51, 1845–1853. [Google Scholar] [CrossRef] [PubMed]
- Doorbar, J.; Quint, W.; Banks, L.; Bravo, I.G.; Stoler, M.; Broker, T.R.; Stanley, M.A. The biology and life-cycle of human papillomaviruses. Vaccine 2012, 30, F55–F70. [Google Scholar] [CrossRef] [PubMed]
- Buck, C.B.; Pastrana, D.V.; Lowy, D.R.; Schiller, J.T. Efficient intracellular assembly of papillomaviral vectors. J. Virol. 2004, 78, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Pastrana, D.V.; Buck, C.B.; Pang, Y.Y.; Thompson, C.D.; Castle, P.E.; FitzGerald, P.C.; Kruger Kjaer, S.; Lowy, D.R.; Schiller, J.T. Reactivity of human sera in a sensitive, high-throughput pseudovirus-based papillomavirus neutralization assay for HPV16 and HPV18. Virology 2004, 321, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zhang, C.; Feng, S.; Liu, C.; Li, Y.; Yang, Y.; Gao, J.; Li, H.; Meng, S.; Li, L.; et al. Detection of HPV types and neutralizing antibodies in Gansu province, China. J. Med. Virol. 2009, 81, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Sehr, P.; Rubio, I.; Seitz, H.; Putzker, K.; Ribeiro-Muller, L.; Pawlita, M.; Muller, M. High-throughput pseudovirion-based neutralization assay for analysis of natural and vaccine-induced antibodies against human papillomaviruses. PLoS ONE 2013, 8, e75677. [Google Scholar] [CrossRef] [PubMed]
- Nie, J.; Huang, W.; Wu, X.; Wang, Y. Optimization and validation of a high throughput method for detecting neutralizing antibodies against human papillomavirus (HPV) based on pseudovirons. J. Med. Virol. 2014, 86, 1542–1555. [Google Scholar] [CrossRef] [PubMed]
- Panicker, G.; Rajbhandari, I.; Gurbaxani, B.M.; Querec, T.D.; Unger, E.R. Development and evaluation of multiplexed immunoassay for detection of antibodies to HPV vaccine types. J. Immunol. Methods 2015, 417, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Hadjilaou, A.; Green, A.M.; Coloma, J.; Harris, E. Single-cell analysis of B cell/antibody cross-reactivity using a novel multicolor FluoroSpot assay. J. Immunol. 2015, 195, 3490–3496. [Google Scholar] [CrossRef] [PubMed]
- Leder, C.; Kleinschmidt, J.A.; Wiethe, C.; Muller, M. Enhancement of capsid gene expression: Preparing the human papillomavirus type 16 major structural gene L1 for DNA vaccination purposes. J. Virol. 2001, 75, 9201–9209. [Google Scholar] [CrossRef] [PubMed]
- Pastrana, D.V.; Gambhira, R.; Buck, C.B.; Pang, Y.Y.; Thompson, C.D.; Culp, T.D.; Christensen, N.D.; Lowy, D.R.; Schiller, J.T.; Roden, R.B. Cross-neutralization of cutaneous and mucosal Papillomavirus types with anti-sera to the amino terminus of L2. Virology 2005, 337, 365–372. [Google Scholar] [CrossRef] [PubMed]
- Mossadegh, N.; Gissmann, L.; Muller, M.; Zentgraf, H.; Alonso, A.; Tomakidi, P. Codon optimization of the human papillomavirus 11 (HPV 11) L1 gene leads to increased gene expression and formation of virus-like particles in mammalian epithelial cells. Virology 2004, 326, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Kieback, E.; Muller, M. Factors influencing subcellular localization of the human papillomavirus L2 minor structural protein. Virology 2006, 345, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Ishii, Y.; Ochi, H.; Matsumoto, T.; Yoshikawa, H.; Kanda, T. Neutralization of HPV16, 18, 31, and 58 pseudovirions with antisera induced by immunizing rabbits with synthetic peptides representing segments of the HPV16 minor capsid protein L2 surface region. Virology 2007, 358, 266–272. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.N.; Buck, C.B.; Thompson, C.D.; Kines, R.; Bernardo, M.; Choyke, P.L.; Lowy, D.R.; Schiller, J.T. Genital transmission of HPV in a mouse model is potentiated by nonoxynol-9 and inhibited by carrageenan. Nat. Med. 2007, 13, 857–861. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Ochi, H.; Matsumoto, T.; Yoshikawa, H.; Kanda, T. Modification of human papillomavirus-like particle vaccine by insertion of the cross-reactive L2-epitopes. J. Med. Virol. 2008, 80, 841–846. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.M.; Kines, R.C.; Roberts, J.N.; Lowy, D.R.; Schiller, J.T.; Day, P.M. Role of heparan sulfate in attachment to and infection of the murine female genital tract by human papillomavirus. J. Virol. 2009, 83, 2067–2074. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.L.; Zhang, C.T.; Zhu, X.K.; Wang, Y.C. Detection of HPV types and neutralizing antibodies in women with genital warts in Tianjin City, China. Virol. Sin. 2010, 25, 8–17. [Google Scholar] [CrossRef] [PubMed]
- Buck, C.B.; Thompson, C.D. Production of papillomavirus-based gene transfer vectors. Curr. Protoc. Cell Biol. 2007. [Google Scholar] [CrossRef]
- Matumoto, M. A note on some points of calculation method of LD50 by Reed and Muench. Jpn. J. Exp. Med. 1949, 20, 175–179. [Google Scholar] [PubMed]
- Ferguson, M.; Wilkinson, D.E.; Heath, A.; Matejtschuk, P. The first international standard for antibodies to HPV 16. Vaccine 2011, 29, 6520–6526. [Google Scholar] [CrossRef] [PubMed]
- Stanley, M.; Pinto, L.A.; Trimble, C. Human papillomavirus vaccines—Immune responses. Vaccine 2012, 30, F83–F87. [Google Scholar] [CrossRef] [PubMed]
- Ahlborg, N.; Axelsson, B. Dual- and triple-color fluorospot. Methods Mol. Biol. 2012, 792, 77–85. [Google Scholar] [PubMed]
- Wheeler, C.M.; Kjaer, S.K.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Perez, G.; Brown, D.R.; Koutsky, L.A.; Tay, E.H.; Garcia, P.; et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in sexually active women aged 16–26 years. J. Infect. Dis. 2009, 199, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.R.; Kjaer, S.K.; Sigurdsson, K.; Iversen, O.E.; Hernandez-Avila, M.; Wheeler, C.M.; Perez, G.; Koutsky, L.A.; Tay, E.H.; Garcia, P.; et al. The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naive women aged 16–26 years. J. Infect. Dis. 2009, 199, 926–935. [Google Scholar] [CrossRef] [PubMed]
- Paavonen, J.; Jenkins, D.; Bosch, F.X.; Naud, P.; Salmeron, J.; Wheeler, C.M.; Chow, S.N.; Apter, D.L.; Kitchener, H.C.; Castellsague, X.; et al. Efficacy of a prophylactic adjuvanted bivalent L1 virus-like-particle vaccine against infection with human papillomavirus types 16 and 18 in young women: An interim analysis of a phase III double-blind, randomised controlled trial. Lancet 2007, 369, 2161–2170. [Google Scholar] [CrossRef]
| Sample | ||||||
|---|---|---|---|---|---|---|
| Pseudovirion | A | B | C | D | E | |
| HPV16 | 16E | 113 | 967 | 5025 | 99 | <40 |
| 16P | 95 | 1945 | 8608 | 109 | <40 | |
| 16C | 75 | 1225 | 5786 | 75 | <40 | |
| 16E-18P-58C | 93 | 1016 | 5866 | 118 | <40 | |
| 16P-18C-58E | 74 | 1328 | 7039 | 97 | <40 | |
| 16C-18E-58P | <40 | 761 | 4381 | 55 | <40 | |
| CV | 18% | 34% | 25% | 25% | 0% | |
| HPV18 | 18E | 120 | 16,957 | 15,630 | <40 | 826 |
| 18P | 80 | 5641 | 14,820 | <40 | 824 | |
| 18C | 78 | 6875 | 13,912 | <40 | 674 | |
| 16E-18P-58C | 102 | 6500 | 15,366 | <40 | 801 | |
| 16P-18C-58E | 98 | 11,296 | 5106 | <40 | 1067 | |
| 16C-18E-58P | 98 | 18,427 | 9519 | <40 | 628 | |
| CV | 16% | 51% | 34% | 0% | 19% | |
| HPV58 | 58E | 103 | 1778 | 8799 | <40 | <40 |
| 58P | 121 | 2029 | 7422 | <40 | <40 | |
| 58C | 93 | 1098 | 3077 | <40 | <40 | |
| 16E-18P-58C | 76 | 1395 | 3686 | <40 | <40 | |
| 16P-18C-58E | 189 | 952 | 10,764 | <40 | <40 | |
| 16C-18E-58P | 115 | 1608 | 7599 | <40 | <40 | |
| CV | 34% | 28% | 43% | 0% | 0% | |
| Triple-Color PBNA | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Single-Color PBNA | HPV16 | HPV18 | HPV58 | ||||||
| − | + | Total | − | + | Total | − | + | Total | |
| − | 26 | 0 | 26 | 35 | 0 | 35 | 29 | 1 | 30 |
| + | 2 | 20 | 22 | 2 | 11 | 13 | 0 | 18 | 18 |
| Total | 28 | 16 | 48 | 37 | 11 | 48 | 29 | 19 | 48 |
| Agreement (%) | 95.8 | 95.8 | 97.9 | ||||||
| Negative Predictive Value (%) | 92.9 | 94.6 | 100 | ||||||
| Positive Predictive Value (%) | 100 | 100 | 94.7 | ||||||
| Kappa score | 0.92 | 0.89 | 0.96 | ||||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nie, J.; Liu, Y.; Huang, W.; Wang, Y. Development of a Triple-Color Pseudovirion-Based Assay to Detect Neutralizing Antibodies against Human Papillomavirus. Viruses 2016, 8, 107. https://doi.org/10.3390/v8040107
Nie J, Liu Y, Huang W, Wang Y. Development of a Triple-Color Pseudovirion-Based Assay to Detect Neutralizing Antibodies against Human Papillomavirus. Viruses. 2016; 8(4):107. https://doi.org/10.3390/v8040107
Chicago/Turabian StyleNie, Jianhui, Yangyang Liu, Weijin Huang, and Youchun Wang. 2016. "Development of a Triple-Color Pseudovirion-Based Assay to Detect Neutralizing Antibodies against Human Papillomavirus" Viruses 8, no. 4: 107. https://doi.org/10.3390/v8040107
APA StyleNie, J., Liu, Y., Huang, W., & Wang, Y. (2016). Development of a Triple-Color Pseudovirion-Based Assay to Detect Neutralizing Antibodies against Human Papillomavirus. Viruses, 8(4), 107. https://doi.org/10.3390/v8040107






