Tissue Barriers to Arbovirus Infection in Mosquitoes
Abstract
:1. Introduction
2. Infection Pattern of an Arbovirus in a Competent Mosquito Vector
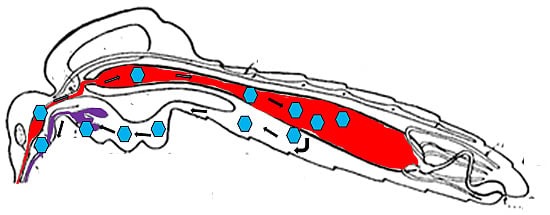
2.1. The Midgut as the Initial Site of Infection
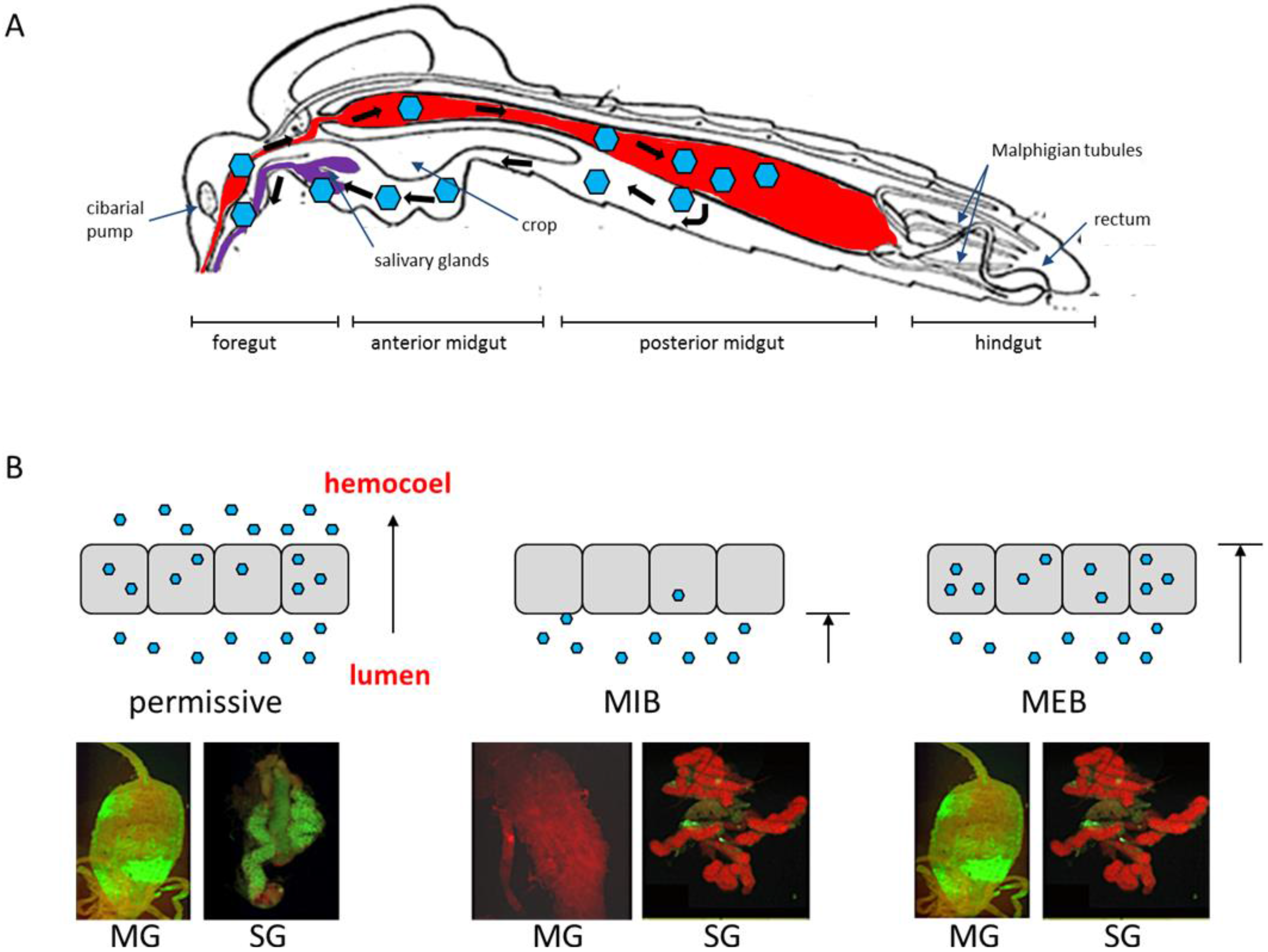
2.2. Virus Dissemination from Midgut to Secondary Tissues
2.3. Infection of Salivary Glands
3. Dose-dependent and -independent Tissue Barriers to Infection
4. The Midgut Infection Barrier
4.1. MIB for Alphaviruses
4.2. MIB for Flaviviruses Based on Studies with DENV
5. The Midgut Escape Barrier
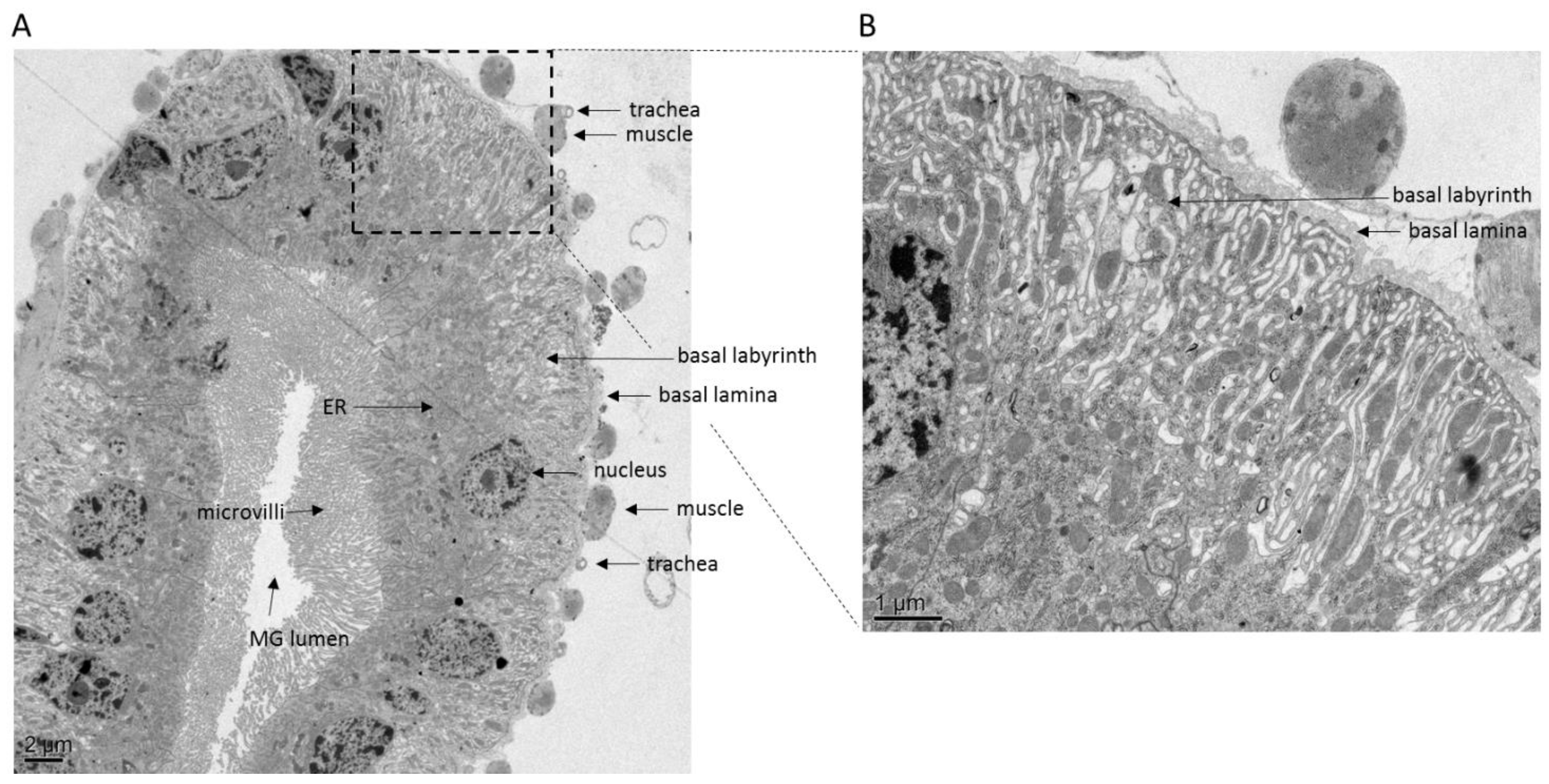
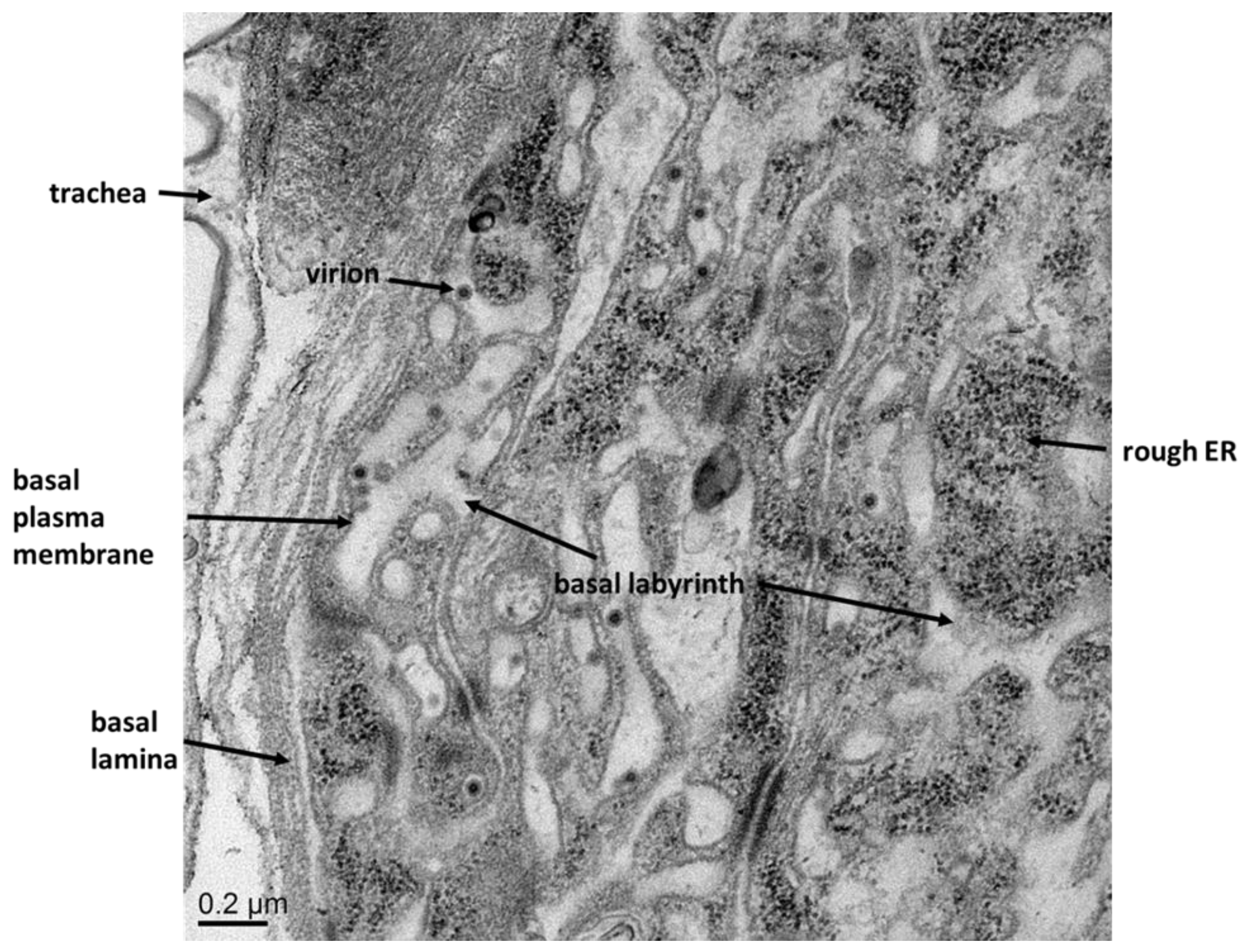
5.1. The Basal Lamina, a Physical Barrier for Virions
5.2. The Role of Tracheae in Virus Dissemination from Midgut
5.3. How Baculoviruses Escape from the Lepidopteran Midgut—A Model for Arboviruses?
5.4. Dissemination via Cardia/Intussuscepted Foregut
6. Salivary Gland Infection and Escape Barriers
7. What Role Do RNAi and Apoptosis Play in Barriers to Infection?
8. Summary and Conclusions
Acknowledgments
Author Contributions
Abbreviations
| CHIKV | chikungunya virus (Alphavirus, Togavirirdae) |
| DENV1–4 | dengue virus serotypes 1–4 (Flavivirus, Flaviviridae) |
| EEEV | eastern equine encephalitis virus (Alphavirus, Togavirirdae) |
| JEV | Japanese encephalitis virus (Flavivirus, Flaviviridae) |
| LACV | La Crosse virus (Orthobunyavirus, Bunyaviridae) |
| ONNV | O’nyong-nyong virus (Alphavirus, Togavirirdae) |
| RVFV | Rift Valley fever virus (Phlebovirus, Bunyaviridae) |
| SFV | Semliki Forest virus (Alphavirus, Togavirirdae) |
| SINV | Sindbis virus (Alphavirus, Togavirirdae) |
| SLEV | St. Louis encephalitis virus (Flavivirus, Flaviviridae) |
| VEEV | Venezuelan equine encephalitis virus (Alphavirus, Togavirirdae) |
| WEEV | western equine encephalitis virus (Alphavirus, Togavirirdae) |
| WNV | West Nile virus (Flavivirus, Flaviviridae) |
Conflicts of Interest
References
- Higgs, S.; Beaty, B.J. Natural cycles of vector-borne pathogens. In Biology of Disease Vectors; Marquardt, W.C., Ed.; Elsevier Academic Press: Burlington, MA, USA, 2005; pp. 167–186. [Google Scholar]
- Blair, C.D.; Adelman, Z.N.; Olson, K.E. Molecular strategies for interrupting arthropod-borne virus transmission by mosquitoes. Clin. Microbiol. Rev. 2000, 13, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Kuno, G.; Chang, G.J. Biological transmission of arboviruses: Reexamination of and new insights into components, mechanisms, and unique traits as well as their evolutionary trends. Clin. Microbiol. Rev. 2005, 18, 608–637. [Google Scholar] [CrossRef] [PubMed]
- Beaty, B.J.; Bernhardt, S.; Black, W.; Blair, C.; Eisen, L.; Elizondo-Quiroga, D.; Farfan-Ale, J.; Lozano-Fuente, S.; Franz, A.; Olson, K.; et al. Novel strategies to control Aedes aegypti and dengue. In Vector Biology, Ecology and Control; Atkinson, P.W., Ed.; Springer: New York, NY, USA, 2010; pp. 99–112. [Google Scholar]
- Gubler, D.J. The global emergence/resurgence of arboviral diseases as public health problems. Arch. Med. Res. 2002, 33, 330–342. [Google Scholar] [CrossRef]
- Weaver, S.C. Urbanization and geographic expansion of zoonotic arboviral diseases: Mechanisms and potential strategies for prevention. Trends Microbiol. 2013, 21, 360–363. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C. Arrival of chikungunya virus in the new world: Prospects for spread and impact on public health. PLoS Negl. Trop. Dis. 2014, 8, e2921. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Bisset, J.A.; Marin, R.; Rodriguez, M.M.; Severson, D.W.; Ricardo, Y.; French, L.; Diaz, M.; Perez, O. Insecticide resistance in two Aedes aegypti (Diptera: Culicidae) strains from Costa Rica. J. Med. Entomol. 2013, 50, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Saavedra-Rodriguez, K.; Suarez, A.F.; Salas, I.F.; Strode, C.; Ranson, H.; Hemingway, J.; Black, W.C. IV Transcription of detoxification genes after permethrin selection in the mosquito Aedes aegypti. Insect Mol. Biol. 2012, 21, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Villar, L.; Dayan, G.H.; Arredondo-Garcia, J.L.; Rivera, D.M.; Cunha, R.; Deseda, C.; Reynales, H.; Costa, M.S.; Morales-Ramirez, J.O.; Carrasquilla, G.; et al. Efficacy of a tetravalent dengue vaccine in children in Latin America. N. Engl. J. Med. 2015, 372, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L.; Andreasen, M. Dominant lethality and insect population control. Mol. Biochem. Parasitol. 2002, 121, 173–178. [Google Scholar] [CrossRef]
- Fu, G.; Lees, R.S.; Nimmo, D.; Aw, D.; Jin, L.; Gray, P.; Berendonk, T.U.; White-Cooper, H.; Scaife, S.; Kim Phuc, H.; et al. Female-specific flightless phenotype for mosquito control. Proc. Natl. Acad. Sci. USA 2010, 107, 4550–4554. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Sanchez-Vargas, I.; Raban, R.R.; Black, W.C., IV; James, A.A.; Olson, K.E. Fitness impact and stability of a transgene conferring resistance to dengue-2 virus following introgression into a genetically diverse Aedes aegypti strain. PLoS Negl. Trop. Dis. 2014, 8, e2833. [Google Scholar]
- Hardy, J.L.; Houk, E.J.; Kramer, L.D.; Reeves, W.C. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Annu. Rev. Entomol. 1983, 28, 229–262. [Google Scholar] [CrossRef] [PubMed]
- Romoser, W.S. The vector alimentary system. In The Biology of Disease Vectors; Beaty, B.J., Marquardt, W.C., Eds.; University Press of Colorado: Niwot, CO, USA, 1996; pp. 298–317. [Google Scholar]
- Okuda, K.; de Souza Caroci, A.; Ribolla, P.E.; de Bianchi, A.G.; Bijovsky, A.T. Functional morphology of adult female Culex quinquefasciatus midgut during blood digestion. Tissue Cell 2002, 34, 210–219. [Google Scholar] [CrossRef]
- Hecker, H. Structure and function of midgut epithelial cells in culicidae mosquitoes (Insecta, Diptera). Cell Tissue Res. 1977, 184, 321–341. [Google Scholar] [CrossRef] [PubMed]
- Romoser, W.S.; Wasieloski, L.P., Jr.; Pushko, P.; Kondic, J.P.; Lerdthusnee, K.; Neira, M.; Ludwig, G. Evidence for arbovirus dissemination conduits from the mosquito (Diptera: Culicidae) Midgut. J. Med. Entomol. 2004, 41, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Houk, E.J. Midgut ultrastructure of Culex tarsalis (Diptera: Culcidae) before and after a bloodmeal. Tissue Cell 1977, 9, 103–118. [Google Scholar] [CrossRef]
- Snodgrass, R.E. The alimentary canal. In The Anatomical Life of the Mosquito; Smithsonian Institution: Washington, DC, USA, 1959; pp. 73–76. [Google Scholar]
- Perrone, J.B.; Spielman, A. Time and site of assembly of the peritrophic membrane of the mosquito Aedes aegypti. Cell Tissue Res. 1988, 252, 473–478. [Google Scholar] [CrossRef] [PubMed]
- Kato, N.; Dasgupta, R.; Smartt, C.T.; Christensen, B.M. Glucosamine:fructose-6-phosphate aminotransferase: Gene characterization, chitin biosynthesis and peritrophic matrix formation in Aedes aegypti. Insect Mol. Biol. 2002, 11, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zheng, X.; Wu, Y.; Gan, M.; He, A.; Li, Z.; Liu, J.; Zhan, X. Quantitative analysis of replication and tropisms of Dengue virus type 2 in Aedes albopictus. Am. J. Trop. Med. Hyg. 2010, 83, 700–707. [Google Scholar] [CrossRef] [PubMed]
- Mourya, D.T.; Mishra, A.C. Antigen distribution pattern of Japanese encephalitis virus in Culex tritaeniorhynchus, C. vishnui & C. pseudovishnui. Indian J. Med. Res. 2000, 111, 157–161. [Google Scholar]
- Murphy, F.A.; Whitfield, S.G.; Sudia, W.D.; Chamberlain, R.W. Interactions of vector with vertebrate pathogenic viruses. In Invertebrate Immunity; Maramorosch, K., Shope, R.E., Eds.; Academic Press: New York, NY, USA, 1975; pp. 25–48. [Google Scholar]
- Whitfield, S.G.; Murphy, F.A.; Sudia, W.D. St. Louis encephalitis virus: An ultrastructural study of infection in a mosquito vector. Virology 1973, 56, 70–87. [Google Scholar] [CrossRef]
- Smith, D.R.; Adams, A.P.; Kenney, J.L.; Wang, E.; Weaver, S.C. Venezuelan equine encephalitis virus in the mosquito vector Aedes taeniorhynchus: Infection initiated by a small number of susceptible epithelial cells and a population bottleneck. Virology 2008, 372, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Scholle, F.; Girard, Y.A.; Zhao, Q.; Higgs, S.; Mason, P.W. Trans-Packaged West Nile virus-like particles: Infectious properties in vitro and in infected mosquito vectors. J. Virol. 2004, 78, 11605–11614. [Google Scholar] [CrossRef] [PubMed]
- Houk, E.J.; Kramer, L.D.; Hardy, J.L.; Chiles, R.E. Western equine encephalomyelitis virus: In vivo infection and morphogenesis in mosquito mesenteronal epithelial cells. Virus Res. 1985, 2, 123–138. [Google Scholar] [CrossRef]
- Houk, E.J.; Kramer, L.D.; Hardy, J.L.; Presser, S.B. An interspecific mosquito model for the mesenteronal infection barrier to western equine encephalomyelitis virus (Culex tarsalis and Culex pipiens). Am. J. Trop. Med. Hyg. 1986, 35, 632–641. [Google Scholar] [PubMed]
- Girard, Y.A.; Popov, V.; Wen, J.; Han, V.; Higgs, S. Ultrastructural study of West Nile virus pathogenesis in Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2005, 42, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Scott, T.W.; Lorenz, L.H.; Lerdthusnee, K.; Romoser, W.S. Togavirus-associated pathologic changes in the midgut of a natural mosquito vector. J. Virol. 1988, 62, 2083–2090. [Google Scholar] [PubMed]
- Girard, Y.A.; Klingler, K.A.; Higgs, S. West Nile virus dissemination and tissue tropisms in orally infected Culex pipiens quinquefasciatus. Vector Borne Zoonotic Dis. 2004, 4, 109–122. [Google Scholar] [CrossRef] [PubMed]
- Parikh, G.R.; Oliver, J.D.; Bartholomay, L.C. A haemocyte tropism for an arbovirus. J. Gen. Virol. 2009, 90, 292–296. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C. Electron microscopic analysis of infection patterns for Venezuelan equine encephalomyelitis virus in the vector mosquito, Culex (Melanoconion) taeniopus. Am. J. Trop. Med. Hyg. 1986, 35, 624–631. [Google Scholar] [PubMed]
- Wang, Z.; Lu, A.; Li, X.; Shao, Q.; Beerntsen, B.T.; Liu, C.; Ma, Y.; Huang, Y.; Zhu, H.; Ling, E. A systematic study on hemocyte identification and plasma prophenoloxidase from Culex pipiens quinquefasciatus at different developmental stages. Exp. Parasitol. 2011, 127, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Clements, A.N. Adult salivary glands and their secretions. In The Biology of Mosquitoes; Chapman and Hall: London, UK, 1996; pp. 251–262. [Google Scholar]
- Phattanawiboon, B.; Jariyapan, N.; Roytrakul, S.; Paemanee, A.; Sor-suwan, S.; Intakhan, N.; Chanmol, W.; Siriyasatien, P.; Saeung, A.; Choochote, W. Morphological and protein analyses of adult female salivary glands of Anopheles barbirostris species A1 (Diptera: Culicidae). Trop. Biomed. 2014, 31, 813–827. [Google Scholar] [PubMed]
- Bowers, D.F.; Abell, B.A.; Brown, D.T. Replication and tissue tropism of the alphavirus Sindbis in the mosquito Aedes albopictus. Virology 1995, 212, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Salazar, M.I.; Richardson, J.H.; Sanchez-Vargas, I.; Olson, K.E.; Beaty, B.J. Dengue virus type 2: Replication and tropisms in orally infected Aedes aegypti mosquitoes. BMC Microbiol. 2007, 7, e9. [Google Scholar] [CrossRef] [PubMed]
- Raquin, V.; Wannagat, M.; Zouache, K.; Legras-Lachuer, C.; Moro, C.V.; Mavingui, P. Detection of dengue group viruses by fluorescence in situ hybridization. Parasit. Vectors 2012, 5, e243. [Google Scholar] [CrossRef] [PubMed]
- Janzen, H.G.; Rhodes, A.J.; Doane, F.W. Chikungunya virus in salivary glands of Aedes aegypti (L.): An electron microscope study. Can. J. Microbiol. 1970, 16, 581–586. [Google Scholar]
- Gaidamovich, S.Y.; Khutoretskaya, N.V.; Lvova, A.I.; Sveshnikova, N.A. Immunofluorescent staining study of the salivary glands of mosquitoes infected with group A arboviruses. Intervirology 1973, 1, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Tchankouo-Nguetcheu, S.; Bourguet, E.; Lenormand, P.; Rousselle, J.C.; Namane, A.; Choumet, V. Infection by chikungunya virus modulates the expression of several proteins in Aedes aegypti salivary glands. Parasit. Vectors 2012, 5, e264. [Google Scholar] [CrossRef] [PubMed]
- Ciano, K.A.; Saredy, J.J.; Bowers, D.F. Heparan sulfate proteoglycan: An arbovirus attachment factor integral to mosquito salivary gland ducts. Viruses 2014, 6, 5182–5197. [Google Scholar] [CrossRef] [PubMed]
- Girard, Y.A.; Schneider, B.S.; McBee, C.E.; Wen, J.; Han, V.C.; Popov, V.; Mason, P.W.; Higgs, S. Salivary gland morphology and virus transmission during long-term cytopathologic West Nile virus infection in Culex mosquitoes. Am. J. Trop. Med. Hyg. 2007, 76, 118–128. [Google Scholar] [PubMed]
- Westaway, E.G.; Mackenzie, J.M.; Kenney, M.T.; Jones, M.K.; Khromykh, A.A. Ultrastructure of Kunjin virus-infected cells: Colocalization of NS1 and NS3 with double-stranded RNA, and of NS2B with NS3, in virus-induced membrane structures. J. Virol. 1997, 71, 6650–6661. [Google Scholar] [PubMed]
- Mackenzie, J.M.; Jones, M.K.; Young, P.R. Immunolocalization of the dengue virus nonstructural glycoprotein NS1 suggests a role in viral RNA replication. Virology 1996, 220, 232–240. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, J.M.; Khromykh, A.A.; Jones, M.K.; Westaway, E.G. Subcellular localization and some biochemical properties of the flavivirus Kunjin nonstructural proteins NS2A and NS4A. Virology 1998, 245, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Forrester, N.L.; Coffey, L.L.; Weaver, S.C. Arboviral bottlenecks and challenges to maintaining diversity and fitness during mosquito transmission. Viruses 2014, 6, 3991–4004. [Google Scholar] [CrossRef] [PubMed]
- Schneider, B.S.; Higgs, S. The enhancement of arbovirus transmission and disease by mosquito saliva is associated with modulation of the host immune response. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Black, W.C., IV; Bennett, K.; Gorrochotegui-Escalante, N.; Barillas-Mury, C.V.; Fernandez-Salas, I.; de Lourdes Munoz, M.; Farfan-Ale, J.A.; Olson, K.E.; Beaty, B.J. Flavivirus susceptibility in Aedes aegypti. Arch. Med. Res. 2002, 33, 379–388. [Google Scholar]
- Khoo, C.C.H.; Piper, J.; Sanchez-Vargas, I.; Olson, K.E.; Franz, A.W.E. The RNA interference pathway affects midgut infection- and escape barriers for Sindbis virus in Aedes aegypti. BMC Microbiol. 2010, 10, e130. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.D.; Hardy, J.L.; Presser, S.B.; Houk, E.J. Dissemination barriers for western equine encephalomyelitis virus in Culex tarsalis infected after ingestion of low viral doses. Am. J. Trop. Med. Hyg. 1981, 30, 190–197. [Google Scholar] [PubMed]
- Forrester, N.L.; Guerbois, M.; Seymour, R.L.; Spratt, H.; Weaver, S.C. Vector-borne transmission imposes a severe bottleneck on an RNA virus population. PLoS Pathog. 2012, 8, e1002897. [Google Scholar] [CrossRef] [PubMed]
- Houk, E.J.; Arcus, Y.M.; Hardy, J.L.; Kramer, L.D. Binding of western equine encephalomyelitis virus to brush border fragments isolated from mesenteronal epithelial cells of mosquitoes. Virus Res. 1990, 17, 105–117. [Google Scholar] [CrossRef]
- Woodring, J.; Chandler, L.J.; Oray, C.T.; McGaw, M.M.; Blair, C.D.; Beaty, B.J. Short report: Diapause, transovarial transmission, and filial infection rates in geographic strains of La Crosse virus-infected Aedes triseriatus. Am. J. Trop. Med. Hyg. 1998, 58, 587–588. [Google Scholar] [PubMed]
- Smith, D.R.; Arrigo, N.C.; Leal, G.; Muehlberger, L.E.; Weaver, S.C. Infection and dissemination of Venezuelan equine encephalitis virus in the epidemic mosquito vector, Aedes taeniorhynchus. Am. J. Trop. Med. Hyg. 2007, 77, 176–187. [Google Scholar] [PubMed]
- Franz, A.W.E.; Sanchez-Vargas, I.; Adelman, Z.N.; Blair, C.D.; Beaty, B.J.; James, A.A.; Olson, K.E. Engineering RNA interference-based resistance to dengue virus type 2 in genetically modified Aedes aegypti. Proc. Natl. Acad. Sci. USA 2006, 103, 4198–4203. [Google Scholar] [CrossRef] [PubMed]
- Houk, E.J.; Obie, F.; Hardy, J.L. Peritrophic membrane formation and the midgut barrier to arboviral infection in the mosquito, Culex tarsalis Coquillett (Insecta, Diptera). Acta Trop. 1979, 36, 39–45. [Google Scholar] [PubMed]
- Kato, N.; Mueller, C.R.; Fuchs, J.F.; McElroy, K.; Wessely, V.; Higgs, S.; Christensen, B.M. Evaluation of the function of a type I peritrophic matrix as a physical barrier for midgut epithelium invasion by mosquito-borne pathogens in Aedes aegypti. Vector Borne Zoonotic Dis. 2008, 8, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.T.; Hernandez, R. Infection of cells by alphaviruses. Adv. Exp. Med. Biol. 2012, 726, 181–199. [Google Scholar] [PubMed]
- Mrkic, B.; Kempf, C. The fragmentation of incoming Semliki Forest virus nucleocapsids in mosquito (Aedes albopictus) cells might be coupled to virion uncoating. Arch. Virol. 1996, 141, 1805–1821. [Google Scholar] [CrossRef] [PubMed]
- Pletnev, S.V.; Zhang, W.; Mukhopadhyay, S.; Fisher, B.R.; Hernandez, R.; Brown, D.T.; Baker, T.S.; Rossmann, M.G.; Kuhn, R.J. Locations of carbohydrate sites on alphavirus glycoproteins show that E1 forms an icosahedral scaffold. Cell 2001, 105, 127–136. [Google Scholar] [CrossRef]
- Myles, K.M.; Pierro, D.J.; Olson, K.E. Deletions in the putative cell receptor-binding domain of Sindbis virus strain MRE16 E2 glycoprotein reduce midgut infectivity in Aedes aegypti. J. Virol. 2003, 77, 8872–8881. [Google Scholar] [CrossRef] [PubMed]
- Pierro, D.J.; Myles, K.M.; Foy, B.D.; Beaty, B.J.; Olson, K.E. Development of an orally infectious Sindbis virus transducing system that efficiently disseminates and expresses green fluorescent protein in Aedes aegypti. Insect Mol. Biol. 2003, 12, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Strauss, J.H.; Wang, K.S.; Schmaljohn, A.L.; Kuhn, R.J.; Strauss, E.G. Host-cell receptors for Sindbis virus. Arch. Virol. Suppl. 1994, 9, 473–484. [Google Scholar] [PubMed]
- Ludwig, G.V.; Kondig, J.P.; Smith, J.F. A putative receptor for Venezuelan equine encephalitis virus from mosquito cells. J. Virol. 1996, 70, 5592–5599. [Google Scholar] [PubMed]
- Mourya, D.T.; Ranadive, S.N.; Gokhale, M.D.; Barde, P.V.; Padbidri, V.S.; Banerjee, K. Putative chikungunya virus-specific receptor proteins on the midgut brush border membrane of Aedes aegypti mosquito. Indian J. Med. Res. 1998, 107, 10–14. [Google Scholar] [PubMed]
- Klimstra, W.B.; Nangle, E.M.; Smith, M.S.; Yurochko, A.D.; Ryman, K.D. DC-SIGN and l-SIGN can act as attachment receptors for alphaviruses and distinguish between mosquito cell- and mammalian cell-derived viruses. J. Virol. 2003, 77, 12022–12032. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, F.; Liu, J.; Xiao, X.; Zhang, S.; Qin, C.; Xiang, Y.; Wang, P.; Cheng, G. Transmission-blocking antibodies against mosquito C-type lectins for dengue prevention. PLoS Pathog. 2014, 10, e1003931. [Google Scholar] [CrossRef] [PubMed]
- Rose, P.P.; Hanna, S.L.; Spiridigliozzi, A.; Wannissorn, N.; Beiting, D.P.; Ross, S.R.; Hardy, R.W.; Bambina, S.A.; Heise, M.T.; Cherry, S. Natural resistance-associated macrophage protein is a cellular receptor for sindbis virus in both insect and mammalian hosts. Cell Host Microbe 2011, 10, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Gubler, D.J.; Nalim, S.; Tan, R.; Saipan, H.; Sulianti Saroso, J. Variation in susceptibility to oral infection with dengue viruses among geographic strains of Aedes aegypti. Am. J. Trop. Med. Hyg. 1979, 28, 1045–1052. [Google Scholar] [PubMed]
- Bosio, C.F.; Beaty, B.J.; Black, W.C., IV. Quantitative genetics of vector competence for dengue-2 virus in Aedes aegypti. Am. J. Trop. Med. Hyg. 1998, 59, 965–970. [Google Scholar]
- Mercado-Curiel, R.F.; Black, W.C., IV; Munoz Mde, L. A dengue receptor as possible genetic marker of vector competence in Aedes aegypti. BMC Microbiol. 2008, 8, e118. [Google Scholar]
- Smith, D.R. An update on mosquito cell expressed dengue virus receptor proteins. Insect Mol. Biol. 2012, 21, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Maguire, T.; Hileman, R.E.; Fromm, J.R.; Esko, J.D.; Linhardt, R.J.; Marks, R.M. Dengue virus infectivity depends on envelope protein binding to target cell heparan sulfate. Nat. Med. 1997, 3, 866–871. [Google Scholar] [CrossRef] [PubMed]
- Chu, J.J.; Leong, P.W.; Ng, M.L. Characterization of plasma membrane-associated proteins from Aedes albopictus mosquito (C6/36) cells that mediate West Nile virus binding and infection. Virology 2005, 339, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.L.; Cisneros, A.; Cruz, J.; Das, P.; Tovar, R.; Ortega, A. Putative dengue virus receptors from mosquito cells. FEMS Microbiol. Lett. 1998, 168, 251–258. [Google Scholar] [CrossRef]
- Mercado-Curiel, R.F.; Esquinca-Aviles, H.A.; Tovar, R.; Diaz-Badillo, A.; Camacho-Nuez, M.; Muñoz Mde, L. The four serotypes of dengue recognize the same putative receptors in Aedes aegypti midgut and Ae. albopictus cells. BMC Microbiol. 2006, 6, e85. [Google Scholar] [CrossRef] [PubMed]
- Sakoonwatanyoo, P.; Boonsanay, V.; Smith, D.R. Growth and production of the dengue virus in C6/36 cells and identification of a laminin-binding protein as a candidate serotype 3 and 4 receptor protein. Intervirology 2006, 49, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Salas-Benito, J.S.; del Angel, R.M. Identification of two surface proteins from C6/36 cells that bind dengue type 4 virus. J. Virol. 1997, 71, 7246–7252. [Google Scholar] [PubMed]
- Salas-Benito, J.; Reyes-Del Valle, J.; Salas-Benito, M.; Ceballos-Olvera, I.; Mosso, C.; del Angel, R.M. Evidence that the 45-kD glycoprotein, part of a putative dengue virus receptor complex in the mosquito cell line C6/36, is a heat-shock related protein. Am. J. Trop. Med. Hyg. 2007, 77, 283–290. [Google Scholar] [PubMed]
- Kuadkitkan, A.; Wikan, N.; Fongsaran, C.; Smith, D.R. Identification and characterization of prohibitin as a receptor protein mediating DENV-2 entry into insect cells. Virology 2010, 406, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Mde, L.; Limon-Camacho, G.; Tovar, R.; Diaz-Badillo, A.; Mendoza-Hernandez, G.; Black, W.C., IV. Proteomic identification of dengue virus binding proteins in Aedes aegypti mosquitoes and Aedes albopictus cells. Biomed. Res. Int. 2013, 2013, e875958. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.E.; Flick, D.; Fleming, K.H.; Jochim, R.; Beaty, B.J.; Black, W.C.T. Quantitative trait loci that control dengue-2 virus dissemination in the mosquito Aedes aegypti. Genetics 2005, 170, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Turell, M.J.; Gargan, T.P., II; Bailey, C.L. Replication and dissemination of Rift Valley fever virus in Culex pipiens. Am. J. Trop. Med. Hyg. 1984, 33, 176–181. [Google Scholar]
- Weaver, S.C.; Scherer, W.F.; Cupp, E.W.; Castello, D.A. Barriers to dissemination of Venezuelan encephalitis viruses in the Middle American enzootic vector mosquito, Culex (Melanoconion) taeniopus. Am. J. Trop. Med. Hyg. 1984, 33, 953–960. [Google Scholar] [PubMed]
- Yurchenco, P.D.; O’Rear, J.J. Basal lamina assembly. Curr. Opin. Cell. Biol. 1994, 6, 674–681. [Google Scholar] [CrossRef]
- Grimstad, P.R.; Walker, E.D. Aedes triseriatus (Diptera: Culicidae) and La Crosse virus. IV. Nutritional deprivation of larvae affects the adult barriers to infection and transmission. J. Med. Entomol. 1991, 28, 378–386. [Google Scholar]
- Thomas, R.E.; Wu, W.K.; Verleye, D.; Rai, K.S. Midgut basal lamina thickness and dengue-1 virus dissemination rates in laboratory strains of Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 1993, 30, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Chandler, L.J.; Blair, C.D.; Beaty, B.J. La Crosse virus infection of Aedes triseriatus (Diptera: Culicidae) ovaries before dissemination of virus from the midgut. J. Med. Entomol. 1998, 35, 567–572. [Google Scholar] [CrossRef] [PubMed]
- Engelhard, E.K.; Kam-Morgan, L.N.W.; Washburn, J.O.; Volkman, L.E. The insect tracheal system: A conduit for the systemic spread of Autographa californica M nuclear polyhedrosis virus. Proc. Natl. Acad. Sci. USA 1994, 91, 3224–3327. [Google Scholar] [CrossRef] [PubMed]
- Kirkpatrick, B.A.; Washburn, J.O.; Engelhard, E.K.; Volkman, L.E. Primary infection of insect tracheae by Autographa californica M nuclear polyhedrosis virus. Virology 1994, 203, 184–186. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Gopinathan, K.P. Systemic and in vitro infection process of Bombyx mori nucleopolyhedrovirus. Virus Res. 2004, 101, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Romoser, W.S.; Turell, M.J.; Lerdthusnee, K.; Neira, M.; Dohm, D.; Luldwig, G.; Wasieloski, L. Pathogenesis of Rift Valley fever virus in mosquitoes-tracheal conduits and the basal lamina as an extra-cellular barrier. Arch. Virol. Suppl. 2005, 19, 89–100. [Google Scholar] [PubMed]
- Oviedo, M.V.; Romoser, W.S.; James, C.B.; Mahmood, F.; Reisen, W.K. Infection dynamics of western equine encephalomyelitis virus (Togaviridae: Alphavirus) in four strains of Culex tarsalis (Diptera: Culicidae): An immunocytochemical study. Res. Rep. Trop. Med. 2011, 18, 65–77. [Google Scholar]
- Passarelli, A.L. Barriers to success: How baculoviruses establish systemic infections. Virology 2011, 411, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.M.; Pierro, D.J.; Olson, K.E. Comparison of the transmission potential of two genetically distinct Sindbis viruses after oral infection of Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 2004, 41, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Granados, R.R.; Lawler, K.A. In vivo pathway of Autographa californica baculovirus invasion and infection. Virology 1981, 108, 297–308. [Google Scholar] [CrossRef]
- Means, J.C.; Passarelli, A.L. Viral fibroblast growth factor, matrix metalloproteases, and caspases are associated with enhancing infection by baculoviruses. Proc. Natl. Acad. Sci. USA 2010, 107, 9825–9830. [Google Scholar] [CrossRef] [PubMed]
- Romoser, W.S.; Faran, M.E.; Bailey, C.L.; Lerdthusnee, K. An immunocytochemical study of the distribution of Rift Valley fever virus in the mosquito Culex pipiens. Am. J. Trop. Med. Hyg. 1992, 46, 489–501. [Google Scholar] [PubMed]
- Lerdthusnee, K.; Romoser, W.S.; Faran, M.E.; Dohm, D.J. Rift Valley fever virus in the cardia of Culex pipiens: An immunocytochemical and ultrastructural study. Am. J. Trop. Med. Hyg. 1995, 53, 331–337. [Google Scholar] [PubMed]
- Brault, A.C.; Foy, B.D.; Myles, K.M.; Kelly, C.L.; Higgs, S.; Weaver, S.C.; Olson, K.E.; Miller, B.R.; Powers, A.M. Infection patterns of o’nyong nyong virus in the malaria-transmitting mosquito, Anopheles gambiae. Insect Mol. Biol. 2004, 13, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Linthicum, K.J.; Platt, K.; Myint, K.S.; Lerdthusnee, K.; Innis, B.L.; Vaughn, D.W. Dengue 3 virus distribution in the mosquito Aedes aegypti: An immunocytochemical study. Med. Vet. Entomol. 1996, 10, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Foy, B.D.; Myles, K.M.; Pierro, D.J.; Sanchez-Vargas, I.; Uhlirova, M.; Jindra, M.; Beaty, B.J.; Olson, K.E. Development of a new Sindbis virus transducing system and its characterization in three Culicine mosquitoes and two lepidopteran species. Insect Mol. Biol. 2004, 13, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Paulson, S.L.; Grimstad, P.R.; Craig, G.B., Jr. Midgut and salivary gland barriers to La Crosse virus dissemination in mosquitoes of the Aedes triseriatus group. Med. Vet. Entomol. 1989, 3, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Scott, T.W.; Lorenz, L.H.; Weaver, S.C. Susceptibility of Aedes albopictus to infection with eastern equine encephalomyelitis virus. J. Am. Mosq. Control Assoc. 1990, 6, 274–278. [Google Scholar] [PubMed]
- Turell, M.J.; Mores, C.N.; Dohm, D.J.; Komilov, N.; Paragas, J.; Lee, J.S.; Shermuhemedova, D.; Endy, T.P.; Kodirov, A.; Khodjaev, S. Laboratory transmission of Japanese encephalitis and West Nile viruses by molestus form of Culex pipiens (Diptera: Culicidae) collected in Uzbekistan in 2004. J. Med. Entomol. 2006, 43, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Dourado, L.A.; Ribeiro, L.F.; Brancalhao, R.M.; Tavares, J.; Borges, A.R.; Fernandez, M.A. Silkworm salivary glands are not susceptible to Bombyx mori nuclear polyhedrosis virus. Genet. Mol. Res. 2011, 10, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Barreau, C.; Conrad, J.; Fischer, E.; Lujan, H.D.; Vernick, K.D. Identification of surface molecules on salivary glands of the mosquito, Aedes aegypti, by a panel of monoclonal antibodies. Insect Biochem. Mol. Biol. 1999, 29, 515–526. [Google Scholar] [CrossRef]
- Juhn, J.; Naeem-Ullah, U.; Maciel Guedes, B.A.; Majid, A.; Coleman, J.; Paolucci Pimenta, P.F.; Akram, W.; James, A.A.; Marinotti, O. Spatial mapping of gene expression in the salivary glands of the dengue vector mosquito, Aedes aegypti. Parasit. Vectors 2011, 4, e1. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, P.A.; Spielman, A.J. Fluid transport across the ducts of the salivary glands of a mosquito. Insect Physiol. 1982, 28, 579–583. [Google Scholar] [CrossRef]
- Beaty, B.J.; Holterman, M.; Tabachnick, W.; Shope, R.E.; Rozhon, E.J.; Bishop, D.H. Molecular basis of bunyavirus transmission by mosquitoes: Role of the middle-sized RNA segment. Science 1981, 211, 1433–1435. [Google Scholar] [CrossRef] [PubMed]
- Grimstad, P.R.; Paulson, S.L.; Craig, G.B., Jr. Vector competence of Aedes hendersoni (Diptera: Culicidae) for La Crosse virus and evidence of a salivary-gland escape barrier. J. Med. Entomol. 1985, 22, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Jupp, P.G. Culex theileri and Sindbis virus; salivary glands infection in relation to transmission. J. Am. Mosq. Control Assoc. 1985, 1, 374–376. [Google Scholar] [PubMed]
- Paulson, S.L.; Poirier, S.J.; Grimstad, P.R.; Craig, G.B., Jr. Vector competence of Aedes hendersoni (Diptera: Culicidae) for La Crosse virus: Lack of impaired function in virus-infected salivary glands and enhanced virus transmission by sporozoite-infected mosquitoes. J. Med. Entomol. 1992, 29, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Turell, M.J.; Britch, S.C.; Aldridge, R.L.; Kline, D.L.; Boohene, C.; Linthicum, K.J. Potential for mosquitoes (Diptera: Culicidae) from Florida to transmit Rift Valley fever virus. J. Med. Entomol. 2013, 50, 1111–1117. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Keene, K.M.; Brakney, D.E.; Olson, K.E.; Blair, C.D.; Wilusz, J.; Foy, B.D. Aedes aegypti uses RNA interference in defense against Sindbis virus infection. BMC Microbiol. 2008, 8, e47. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Doty, J.B.; Heersink, M.S.; Olson, K.E.; Franz, A.W. Transgene-mediated suppression of the RNA interference pathway in Aedes aegypti interferes with gene silencing and enhances Sindbis virus and dengue virus type 2 replication. Insect Mol. Biol. 2013, 22, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vargas, I.; Scott, J.C.; Poole-Smith, B.K.; Franz, A.W.; Barbosa-Solomieu, V.; Wilusz, J.; Olson, K.E.; Blair, C.D. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquito’s RNA interference pathway. PLoS Pathog. 2009, 5, e1000299. [Google Scholar] [CrossRef] [PubMed]
- Kelly, E.M.; Moon, D.C.; Bowers, D.F. Apoptosis in mosquito salivary glands: Sindbis virus-associated and tissue homeostasis. J. Gen. Virol. 2012, 93, 2419–2424. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, R.; Scott, T.W. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 2006, 11, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Gort, T.; Boyle, D.L.; Clem, R.J. Effects of manipulating apoptosis on Sindbis virus infection of Aedes aegypti mosquitoes. J. Virol. 2012, 86, 6546–6554. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Clem, R.J. Defining the core apoptosis pathway in the mosquito disease vector Aedes aegypti the roles of iap1, ark, dronc, and effector caspases. Apoptosis 2011, 16, 105–113. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, K.; Olson, B.J.; Huang, N.; Unis, D.; Clem, R.J. Rapid selection against arbovirus-induced apoptosis during infection of a mosquito vector. Proc. Natl. Acad. Sci. USA 2015, 112, E1152–E1161. [Google Scholar] [CrossRef] [PubMed]
- Pierro, D.J.; Powers, E.L.; Olson, K.E. Genetic determinants of Sindbis virus strain TR339 affecting midgut infection in the mosquito Aedes aegypti. J. Gen. Virol. 2007, 88, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Pierro, D.J.; Powers, E.L.; Olson, K.E. Genetic determinants of Sindbis virus mosquito infection are associated with a highly conserved alphavirus and flavivirus envelope sequence. J. Virol. 2008, 82, 2966–2974. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Doty, J.B.; Held, N.L.; Olson, K.E.; Franz, A.W. Isolation of midgut escape mutants of two American genotype dengue 2 viruses from Aedes aegypti. Virol. J. 2013, 10, e257. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franz, A.W.E.; Kantor, A.M.; Passarelli, A.L.; Clem, R.J. Tissue Barriers to Arbovirus Infection in Mosquitoes. Viruses 2015, 7, 3741-3767. https://doi.org/10.3390/v7072795
Franz AWE, Kantor AM, Passarelli AL, Clem RJ. Tissue Barriers to Arbovirus Infection in Mosquitoes. Viruses. 2015; 7(7):3741-3767. https://doi.org/10.3390/v7072795
Chicago/Turabian StyleFranz, Alexander W. E., Asher M. Kantor, A. Lorena Passarelli, and Rollie J. Clem. 2015. "Tissue Barriers to Arbovirus Infection in Mosquitoes" Viruses 7, no. 7: 3741-3767. https://doi.org/10.3390/v7072795
APA StyleFranz, A. W. E., Kantor, A. M., Passarelli, A. L., & Clem, R. J. (2015). Tissue Barriers to Arbovirus Infection in Mosquitoes. Viruses, 7(7), 3741-3767. https://doi.org/10.3390/v7072795