Baculovirus Insecticides in Latin America: Historical Overview, Current Status and Future Perspectives
Abstract
:1. Introduction
1.1. Biological Control as an Essential Component of Integrated Pest Management Strategy
1.2. Baculoviruses: Molecular Biology, Ecology and Application as Biopesticides
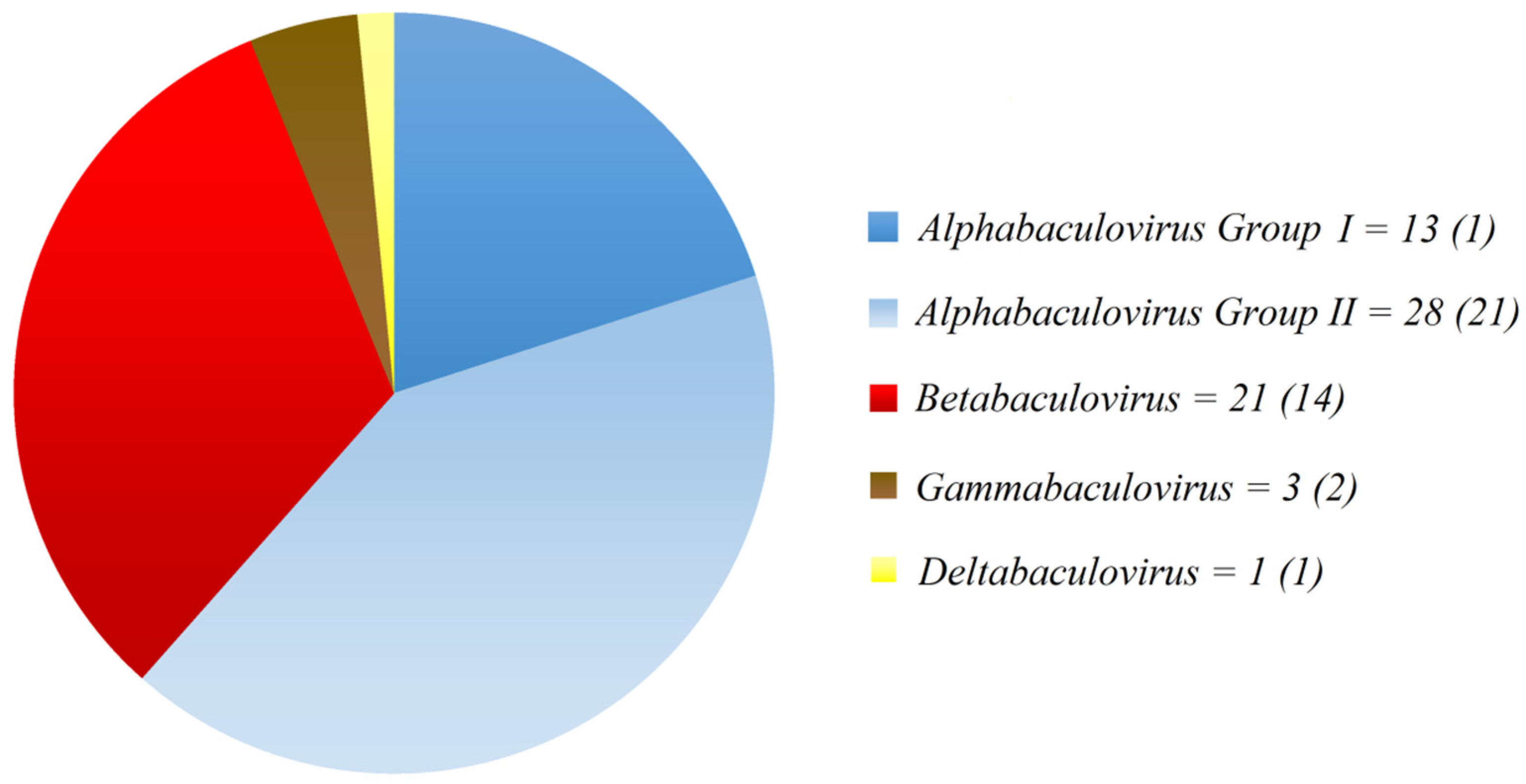
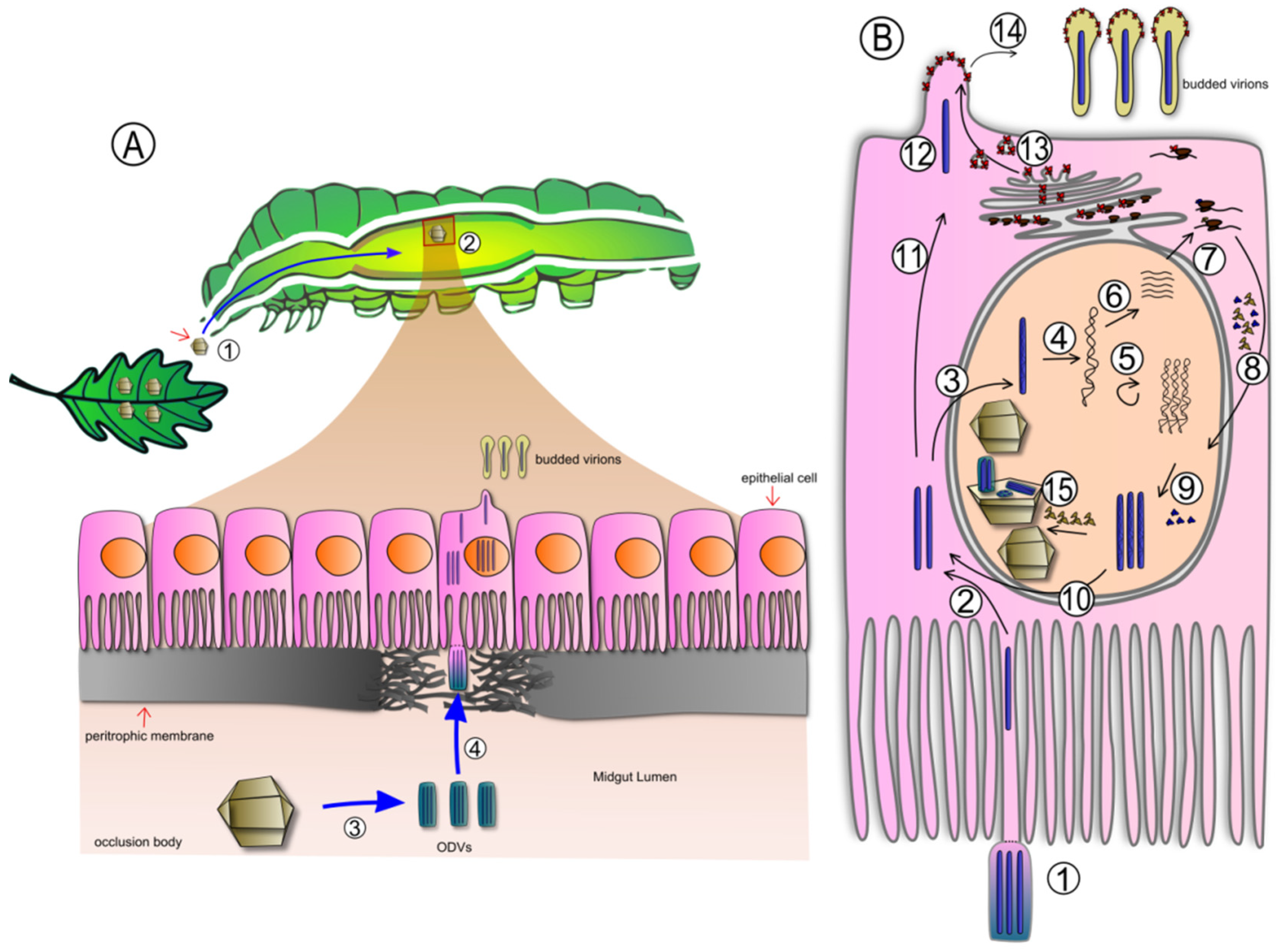
2. Examples of Baculovirus Control Programs in Latin America
| Virus | Host | Crops | Product | Country | Producer company |
|---|---|---|---|---|---|
| Anticarsia gemmatalis MNPV | Anticarsia gemmatalis | Soybean | Baculo-soja 1, Baculovirus Nitral 2, Coopervirus SC 3, protégé 4, Multigen 5 | Brazil | Nova Era Biotecnología Agrícola 1, Nitral Urbana 2, COODETEC 3, Milenia 4, EMBRAPA 5 |
| Autographa californica MNPV + Spodoptera albula NPV | Autographa califórnica Trichoplusia ni Pseudoplusia includens Heliothis virescens Spodoptera exigua Estigmene acrea Plutella xylostella | Alfalfa, vegetable crops | VPN-ULTRA | Guatemala | Agricola El Sol |
| Spodoptera sunia NPV | Spodoptera spp. | Vegetables | VPN 82 | Guatemala | Agricola El Sol |
| Cydia pomonella GV | Cydia pomonella, C. pomonella, Grapholita molesta | Apple, pear, walnut Apple, peach | Carpovirus Plus 6 Madex 7 Carpovirusine 6 Madex Twin 7 | Argentina 6 Argentina 7 Chile 6 Uruguay 7 | NPP-Arysta Life Science 6 Andermatt Biocontrol 7 |
| Erinnyis ello GV | Erinnyis ello | Cassava 8 Rubber trees 9 | Baculovirus erinnyis 8,9,10 | Brazil 8 Colombia 9 Colombia 10 | Empresa de Pesquisa Agropecuária e Extensão Rural de Santa Catarina S.A. 8 BioCaribe SA 9 CORPOICA 10 |
| Helicoverpa zea SNPV | Heliothis and Helicoverpa spp. | Maize, tomato, cotton and tobacco | Gemstar 11 HzNPV CCAB 12 | Mexico 11 Brazil12 | Certis USA 11 AgBiTech Australia 12 |
| Helicoverpa armigera NPV | Heliothis and Helicoverpa spp. | Tomato, sweet pepper, maize, soybean, tobacco, vegetable crops | Diplomata 13 Helicovex 14 | Brazil 13, 14 | Koppert 13 Andermatt Biocontrol 14 |
| Phthorimaea operculella GV | Phthorimaea operculella Tecia solanivora | Potato | Baculovirus Corpoica 15 PTM baculovirus 16, 17 | Colombia 15 Peru 16 Costa Rica 17 | CORPOICA 15 SENASA Peru 16 INTA Costa Rica 17 |
| Phthorimaea operculella GV + Bacillus thuringiensis | Phtorimaea operculella Tecia solanivora Symmetrischema tangolias | Potato | Matapol Plus 18 Bacu-Turin 19 | Bolivia 18 Ecuador 19 | PROINPA Foundation 18 INIAP, Ecuador 19 |
| Spodoptera exigua NPV | Spodoptera exigua | Tomato, chili, eggplant | SPOD-X LC | Mexico | Certis USA—SUMMIT AGRO Mexico |
| Spodoptera frugiperda MNPV | Spodoptera frugiperda | Maize, sorghum | - | Brazil | EMBRAPA (in development) |

2.1. AgMNPV for the Control of the Velvetbean Caterpillar in Soybean Crops
2.2. CpGV for the Control of the Codling Moth in Apple and Pear Orchards

2.3. Phthorimaea Operculella GV for the Control of the Potato Tuber Moth Complex
2.4. Spodoptera Frugiperda MNPV and GV (SfMNPV and SfGV) for the Control of the Fall Armyworm in Maize Crops
2.5. Erinnyis ello GV for the Control of the Cassava Hornworm
2.6. Other Baculoviruses Used as Biological Control Agents in Latin America
3. Production Technology and Product Formulations
| Component | Function | References |
|---|---|---|
| Surfactants | Reduce the surface tension of the drops allowing the drops to be retained on the leaves. Facilitate the spread of drops that reach the leaves Act as emulsifier agent, allowing the oil to be mixed with water in the formulation. | [182] |
| Adherents | Increase adherence of the drops to the leaf surface. | [183] |
| Thickeners | Keep the formulation as a homogeneous mix. | [182,184,185] |
| Binders | Increase the tendency of the OB to adhere with the carrier. | [186,187] |
| Baits and phagostimulants | Attract the larvae to ingest the formulated pesticide. Attract natural enemies to the formulation. | [188,189] |
| UV protectors | Avoid the UV inactivation of the OB. Some of them also damage peritrophic membrane structure. | [190] |
4. Genetic Improvement of Baculovirus Biopesticides. Possibilities in Latin America
5. Conclusions and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wood, R.J.; Bishop, J.A. Insecticide resistance: Populations and evolution. In Genetic Consequences of Man-Made Change; Bishop, J.A., Cook, L.M., Eds.; Academic Press: London, UK, 1981; pp. 97–127. [Google Scholar]
- Whalon, M.E.; Mota-Sanchez, D.; Hollingworth, R.M. Global Pesticide Resistance in Arthropods; CAB International: Wallingford, UK, 2008. [Google Scholar]
- Whalon, M.E.; Mota-Sanchez, D.; Hollingworth, R.M.; Duynslager, L. Arthropod Pesticide Resistance Database. Available online: http://www.pesticideresistance.com (accessed on 1 December 2014).
- Fuller, E.; Elderd, B.D.; Dwyer, G. Pathogen persistence in the environment and insect-baculovirus interactions: Disease-Density thresholds, epidemic burnout, and insect outbreaks. Am. Nat. 2012, 179, E70–E96. [Google Scholar] [CrossRef] [PubMed]
- Saxena, H. Microbial Managment of Crop—Pest. J. Biopestic. 2008, 1, 32–37. [Google Scholar]
- Vasantharaj, D.B. Biotechnological approaches in IPM and their impact on environment. J. Biopestic. 2008, 1, 1–5. [Google Scholar]
- Entwistle, P.F.; Evans, H.F. Viral Control. In Conprehensive Insect Fisiology. Biochemestry and Farmacology; Gilbert, L.I., Kerkut, G.A., Eds.; Pergamon Press: Oxford, UK, 1985; Volume 12, pp. 347–412. [Google Scholar]
- Granados, R.R.; Federici, B.A. The Biology of Baculoviruses; CRC Press: Boca Raton, FL, USA, 1986; Volume 1. [Google Scholar]
- Moore, N.F.; King, L.A.; Possee, R.D. Viruses of insects. Insect Sci. Appl. 1987, 3, 275–289. [Google Scholar]
- Tanada, Y.; Kaya, H.K. Insect Pathology; Academic Press: San Diego, CA, USA, 1993; p. 666. [Google Scholar]
- Krieg, A.; Franz, J.M.; Groner, A.; Huber, J.; Miltenburger, H.G. Safety of entomopathogenic viruses for control of insect pests. Environ. Conserve. 1980, 7, 158–160. [Google Scholar] [CrossRef]
- Entwistle, P.F. Viruses for insect pest control. Span 1983, 26, 59–62. [Google Scholar]
- Cunningham, J.C. Insect Viruses and Pest Management; Hunter-Fujita, F.R., Entwistle, P.F., Evans, H.F., Crook, N.E., Eds.; John Wiley & Sons, Inc.: New York, NY, USA, 1998; pp. 313–331. [Google Scholar]
- Black, B.C.; Brennan, L.A.; Dierks, L.A.; Gard, I.E. Commercialization of baculoviral insecticides. In The Baculoviruses; Miller, L.K., Ed.; Plenum Press: New York, NY, USA; London, UK, 1997; pp. 341–388. [Google Scholar]
- McWilliam, A. Environmental Impact of Baculoviruses. FAO R7299_FTR_anx3. , 2007. Available online: http://www.fao.org/docs/eims/upload/agrotech/2003/R7299_FTR_anx3.pdf (accessed on 15 November 2014).
- Organization for Economic Co-operation and Development (OECD). Consensus Document on Information Used in the Assessment of Environmental Applications Involving Baculovirus. OECD Environment, Health and Safety Publications, Series on Harmonization of Regulatory Oversight in Biotechnology. Available online: http://www.rebeca-net.de/downloads/report/deliverable%2012.pdf (accessed on 15 November 2014).
- Rohrmann, G.F. Baculovirus Molecular Biology: Third Edition (Internet). Bethesda (MD): National Center for Biotechnology Information (US). 2013. Available online: http://www.ncbi.nlm.nih.gov/books/NBK114593/ (accessed on 15 November 2014). [Google Scholar]
- Herniou, E.A.; Arif, B.M.; Becnel, J.J.; Blissard, G.W.; Bonning, B.; Harrison, R.; Jehle, J.A.; Theilmann, D.A.; Vlak, J.M. Baculoviridae. In Virus Taxonomy: Classification and Nomenclature of Viruses: Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Elsevier Academic Press: San Diego, CA, USA, 2012; pp. 163–173. [Google Scholar]
- Bonning, B.C. Baculoviruses: Biology, biochemistry, and molecular biology. In Comprehensive Molecular Insect Science; Kostas, I., Lawrence, G., Sarjeet, G., Eds.; Elsevier Pergamon: Oxford, UK, 2005; pp. 233–270. [Google Scholar]
- Jehle, J.A.; Lange, M.; Wang, H.; Hu, Z.; Wang, Y.; Hauschild, R. Molecular identification and phylogenetic analysis of baculoviruses from Lepidoptera. Virology 2006, 346, 180–193. [Google Scholar] [CrossRef] [PubMed]
- Jehle, J.A.; Blissard, G.W.; Bonning, B.C.; Cory, J.S.; Herniou, E.A.; Rohrmann, G.F.; Theilmann, D.A.; Thiem, S.M.; Vlak, J.M. On the classification and nomenclature of baculoviruses: A proposal for revision. Arch. Virol. 2006, 151, 1257–1266. [Google Scholar] [CrossRef] [PubMed]
- Ardisson-Araújo, D.M.; de Melo, F.L.; Andrade, M.S.; Sihler, W.; Báo, S.N.; Ribeiro, B.M.; de Souza, M.L. Genome sequence of Erinnyis ello granulovirus (ErelGV), a natural cassava hornworm pesticide and the first sequenced sphingid-infecting betabaculovirus. BMC Genomics 2014, 15, e856. [Google Scholar] [CrossRef]
- Wennmann, J.T.; Gueli Alletti, G.; Jehle, J.A. The genome sequence of Agrotis segetum nucleopolyhedrovirus B (AgseNPV-B) reveals a new baculovirus species within the Agrotis baculovirus complex. Virus Genes 2014, 50, 260–276. [Google Scholar] [CrossRef] [PubMed]
- Cuartas, P.E.; Barrera, G.P.; Belaich, M.N.; Barreto, E.; Ghiringhelli, P.D.; Villamizar, L.F. The Complete Sequence of the First Spodoptera frugiperda Betabaculovirus Genome: A Natural Multiple Recombinant Virus. Viruses 2015, 7, 394–421. [Google Scholar] [CrossRef]
- Craveiro, S.R.; Inglis, P.W.; Togawa, R.C.; Grynberg, P.; Melo, F.L.; Ribeiro, M.A.; Ribeiro, B.M.; Báo, S.N.; Castro, M.L.B. The genome sequence of Pseudoplusia includens single nucleopolyhedrovirus and an analysis of p26 gene evolution in the baculoviruses. BMC Genomics 2015, 16, e127. [Google Scholar] [CrossRef]
- Adams, J.R.; McClintock, J.T. Baculoviridae, nuclear polyhedrosis viruses Part 1: Nuclear polyhedrosis viruses of insects. In Atlas of Invertebrate Viruses; Adams, J.R., Bonami, J.R., Eds.; CRC Press: Boca Raton, FL, USA, 1991; Chapter 6; pp. 87–180. [Google Scholar]
- Haase, S.; Ferrelli, M.L.; Pidre, M.L.; Romanowski, V. Genetic Engineering of Baculoviruses, Current Issues in Molecular Virology—Viral Genetics and Biotechnological Applications. Available online: http://www.intechopen.com/books/current-issues-in-molecular-virology-viral-genetics-and-biotechnological-applications/genetic-engineering-of-baculoviruses (accessed on 15 February 2015).
- Hegedus, D.; Erlandson, M.; Gillott, C.; Toprak, U. New insights into peritrophic matrix synthesis, architecture, and function. Annu. Rev. Entomol. 2009, 54, 285–302. [Google Scholar] [CrossRef] [PubMed]
- Barbehenn, R.V.; Martin, M.M. Peritrophic envelope permeability in herbivorous insects. J. Insect Physiol. 1995, 41, 301–311. [Google Scholar] [CrossRef]
- Wang, P.; Granados, R.R. An intestinal mucin is the target substrate for a baculovirus enhancin. Proc. Natl. Acad. Sci. USA 1997, 94, 6977–6982. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, Y.; Corsaro, B.G.; Granados, R.R. Location and nucleotide sequence of the gene encoding the viral enhancing factor of the Trichoplusia ni granulosis virus. J. Gen. Virol. 1991, 72, 2645–2652. [Google Scholar] [CrossRef] [PubMed]
- Slavicek, J.M.; Popham, H.J. The Lymantria dispar nucleopolyhedrovirus enhancins are components of occlusion-derived virus. J. Virol. 2005, 79, 10578–10588. [Google Scholar] [CrossRef] [PubMed]
- Derksen, A.C.G.; Granados, R.R. Alteration of a lepidopteran peritrophic membrane by baculoviruses and enhancement of viral infectivity. Virology 1988, 167, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Tanada, Y.; Hess, R.T.; Omi, E.M. Invasion of a nuclear polyhedrosis virus in midgut of the armyworm, Pseudaletia unipuncta, and the enhancement of a synergistic enzyme. J. Invertebr. Pathol. 1975, 26, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.K.; Lingg, A.J.; Bulla, L.A., Jr. Bacterial, viral, and fungal insecticides. Science 1983, 219, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Passarelli, A.L. Barriers to success: How baculovirus establish efficient systemic infections. J. Virol. 2011, 411, 383–392. [Google Scholar] [CrossRef]
- Van Oers, M.M.; Flipsen, J.T.; Reusken, C.B.; Vlak, J.M. Specificity of baculovirus p10 functions. Virology 1994, 200, 513–523. [Google Scholar] [CrossRef] [PubMed]
- Hawtin, R.E.; Zarkowska, T.; Arnold, K.; Thomas, C.J.; Gooday, G.W.; King, L.A.; Kuzio, J.A.; Possee, R.D. Liquefaction of Autographa californica Nucleopolyhedrovirus-infected insects is dependent on the integrity of virus-encoded chitinase and cathepsin genes. Virology 1997, 238, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, S.D. Alternative Routes for the Horizontal Transmission of a Nucleopolyhedrovirus. J. Invertebr. Pathol. 1996, 68, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Fuxa, J.R.; Richter, A.R.; Ameen, A.O.; Hammock, B.D. Vertical transmission of TnSNPV, TnCPV, AcMNPV, and possibly recombinant NPV in Trichoplusia ni. J. Invertebr. Pathol. 2002, 79, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Bird, J.T. The use of a virus disease in the biological control of the European pine sawfly, Neodiprion sertifer (Geoffr). Can. Entomol. 1953, 85, 437–446. [Google Scholar] [CrossRef]
- Bird, J.T.; Elgee, D.E. A virus disease and introduced parasites as factors controlling the European spruce sawfly, Diprion hercyniae Htd. in central New Brunswick. Can. Entomol. 1957, 89, 371–378. [Google Scholar] [CrossRef]
- Fuxa, J.R. Ecology of insect nucleopolyhedroviruses. Agric. Ecosyst. Environ. 2004, 103, 27–43. [Google Scholar] [CrossRef]
- Kogan, M.; Turnipseed, S.G.; Shepard, M.; Oliveira, E.B.; Borgo, A. Pilot insect pest management for soybean in southern Brazil. J. Econ. Entomol. 1977, 70, 659–663. [Google Scholar] [CrossRef]
- Allen, G.E.; Knell, J.D. A nuclear polyhedrosis virus of Anticarsia gemmatalis I: Ultrastructure, replication, and pathogenicity. Fla. Entomol. 1977, 60, 233–240. [Google Scholar] [CrossRef]
- Carner, G.R.; Turnipseed, S.G. Potential of a nuclearpolyhedrosis virus for the control of the velvetbean caterpillar in soybean. J. Econ. Entomol. 1977, 70, 608–610. [Google Scholar] [CrossRef]
- Moscardi, F. Assessment of the application of baculoviruses for the control of Lepidoptera. Ann. Rev. Entomol. 1999, 44, 257–289. [Google Scholar] [CrossRef]
- Moscardi, F. A Nucleopolyhedrovirus for control of the velvetbean caterpillar in Brazilian Soybeans. In Biological Control: A Global Perspective; Vincent, C., Goethel, M.S., Lazarovits, G., Eds.; CAB International: Oxford, UK, 2007; pp. 344–352. [Google Scholar]
- Moscardi, F.; de Souza Lobo, M.; de Castro Batista, M.E.; Moscardi, L.M.; Szewczyk, B. Baculovirus pesticides: Present state and future perspectives. In Microbes and Microbial Technology; Ahmad, I., Ahmad, F., Pichtel, P., Eds.; Springer: New York, NY, USA, 2011; pp. 415–445. [Google Scholar]
- Moscardi, F. Use of viruses for pest control in Brazil: The case of the nuclear polyhedrosis virus of the soybean caterpillar, Anticarsia gemmatalis. Mem. Inst. Oswaldo Cruz 1989, 84, 51–56. [Google Scholar] [CrossRef]
- Szewczyk, B.; Rabalski, L.; Krol, E.; Sihler, W.; Lobo de Souza, M. Baculovirus biopesticides—A safe alternative to chemical protection of plants. J. Biopestic. 2009, 2, 209–216. [Google Scholar]
- Sosa-Gómez, D.R.; Moscardi, F.; Santos, B.; Alves, L.F.A.; Alves, S.B. Produçao e uso de vírus para o controle de pragas na América Latina. In Controle Microbiano de Pragas na América Latina: Abanicos e Desafíos; Alves, S.A., Lopes, R.B., Eds.; FEALQ: Piracicaba, Brazil, 2008; pp. 49–68. (In Portuguese) [Google Scholar]
- Moscardi, F.; Sosa-Gómez, D.R. Microbial control of insect pests of soybeans. In Field Manual of Techniques in Invertebrate Pathology: Application and Evaluation of Pathogens for Control of Insects and Other Invertebrate Pests; Lacey, L.A., Kaya, H.K., Eds.; Springer: Heidelberg, Germany, 2000; pp. 447–466. [Google Scholar]
- Santos, B. Avanços na Produção Massal de Lagartas de Anticarsia gemmatalis Hübner 1818 (Lepidoptera: Noctuidae) Infectadas Com o Seu Vírus de Poliedrose Nuclear, em Laboratório e do Bioinseticida à Base Desse Vírus. Ph.D. Thesis, Universidade Federal do Paraná, Curitiba, Brazil, 2003. [Google Scholar]
- Corrêa-Ferreira, B.S.; Alexandre, T.M.; Pellizzaro, E.C.; Moscardi, F.; Bueno, A.F. Práticas de manejo de pragas utilizadas na soja e seu impacto sobre a cultura; Embrapa Soja: Londrina, Brasil, 2010. (In Portuguese) [Google Scholar]
- Bueno, R.C.O.F.; Parra, J.R.P.; Bueno, A.F.; Moscardi, F.; Oliveira, J.R.G.; Camillo, M.F. Sem barreira. Revista Cultivar 2007, 93, 12–15. [Google Scholar]
- Sosa-Gómez, D.R.; Centro Nacional de Pesquisa de Soja, Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA), Londrina, Brazil. Personal Communication, 2014.
- Kokubu, H. Brief overview of microorganisms used against agricultural insect pests. Available online: http://e-cucba.cucba.udg.mx/index.php/e-Cucba/article/view/10/pdf_brief (accessed on 24 April 2015).
- Williams, T.; Arredondo-Bernal, H.C.; Rodríguez-del-Bosque, L.A. Biological Pest Control in Mexico. Annu. Rev. Entomol. 2013, 58, 119–140. [Google Scholar] [CrossRef] [PubMed]
- Tanada, Y. A granulosis virus of the codling moth, Carpocapsa pomonella (Linnaeus) (Olethreutidae, Lepidoptera). J. Insect Pathol. 1964, 6, 378–380. [Google Scholar]
- Falcon, L.A.; Kane, W.R.; Bethel, R.S. Preliminary evaluation of a granulosis virus for control of the codling moth. J. Econ. Entomol. 1968, 61, 1208–1213. [Google Scholar] [CrossRef]
- Lacey, L.; Thomson, D.; Vincent, C.; Arthurs, S.P. Codling moth granulovirus: A comprehensive review. Biocontrol Sci. Technol. 2008, 18, 639–663. [Google Scholar] [CrossRef]
- Beas-Catena, A.; Sánchez-Mirón, A.; García-Camacho, F.; Contreras-Gómez, A.; Molina-Grima, E. Baculovirus biopesticides: An overview. J. Anim. Plant Sci. 2014, 24, 362–373. [Google Scholar]
- Quintana, G.; Alvarado, L. Carpovirus Plus: Primer insecticida biológico para el control de Cydia pomonella en montes comerciales de pera, manzana y nogal. In AgroInnova—La Innovación Tecnológica para Mejorar la Competitividad; SECyT-INTA: Rosario, Argentina, 2004; pp. 15–17. (In Spanish) [Google Scholar]
- Quintana, G. Control biológico de Cydia pomonella: Virus de la granulosis de Cydia pomonella: Una alternativa segura y eficaz de control. Rev. Redagrícola 2009, 27, 23. (In Spanish) [Google Scholar]
- Quintana, G. Uso del virus de la granulosis (CpGV) para el control de carpocapsa (Cydia pomonella L.) en Argentina. Resúmenes TAMIBIO 2013, DiMAyA. Asoc. Argent. Microbiol. 2013, pp. 21–22. (In Spanish)Available online: http://aam.org.ar/src/img_up/21072014.5.pdf (accessed on 15 February 2015).
- Quintana, G.; Cólica, J.J.; del Fernández Górgola, M.C.; Rivero, C.; Pérez, O.; Luna Mercado, L. Control de carpocapsa (Cydia pomonella L.) con un producto en base al virus de la granulosis (CpGV), en cultivos de nogal en Catamarca. Rev. CIZAS 2007, 8, 39–44. (In Spanish) [Google Scholar]
- Cólica, J.; Quintana, G.; del Fernández Górgola, M.C.; la Rossa, R. Evaluación preliminar de una formulación atracticida para el control de Cydia pomonella (L) en montes de nogal en Catamarca, Argentina. Revista del CIZAS. Facultad de Ciencias Agrarias, Universidad Nacional de Catamarca (UNCa). Rev. Cient. CIZAS 2008, 9, 15–22. (In Spanish) [Google Scholar]
- Sauphanor, B.; Berling, M.; Toubon, J.K.; Reyes, M.; Delnatte, J.; Allemoz, P. Carpocapse des pommes: Cas de resistance au virus de la granulose en vergers biologiques. Phytoma Def.Veg. 2006, 590, 24–27. (In French) [Google Scholar]
- Asser, K.S.; Fritsch, E.; Undorf, S.K.; Kienzle, J.; Eberle, K.E.; Gund, N.A.; Reineke, A.; Zebitz, C.P.; Heckel, D.G.; Huber, J.; et al. Rapid emergence of baculovirus resistance in codling moth due to dominant, sex-linked inheritance. Science 2007, 317, 1916–1918. [Google Scholar] [CrossRef] [PubMed]
- Eberle, K.E.; Jehle, J.A. Field resistance of codling moth against Cydia pomonella granulovirus (CpGV) is autosomal and incompletely dominant inherited. J. Invertebr. Pathol. 2006, 93, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, A.; Bisutti, I.L.; Ladurner, E.; Benuzzi, M.; Sauphanor, B.; Kienzle, J.; Zingg, D.; Undorf-Spahn, K.; Fritsch, E.; Huber, J.; et al. The occurrence and distribution of resistance of codling moth to Cydia pomonella granulovirus in Europe. J. Appl. Entomol. 2013, 137, 641–649. [Google Scholar] [CrossRef]
- Zichová, T.; Stará, J.; Kundu, J.; Eberle, K.E.; Jehle, J.A. Resistance to Cydia pomonella granulovirus follows a geographically widely distributed inheritance type within Europe. BioControl 2013, 58, 525–534. [Google Scholar] [CrossRef]
- Rezapanah, M.; Shojai-Estabragh, S.; Huber, J.; Jehle, J.A. Molecular and biological characterization of new isolates of Cydia pomonella granulovirus from Iran. J. Pest Sci. 2008, 81, 187–191. [Google Scholar] [CrossRef]
- Eberle, K.E.; Asser-Kaiser, S.; Sayed, S.M.; Nguyen, H.T.; Jehle, J.A. Overcoming the resistance of codling moth against conventional Cydia pomonella granulovirus (CpGV-M) by a new isolate CpGV-I12. J. Invertebr. Pathol. 2008, 98, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Berling, M.; Blachere-Lopez, C.; Soubabere, O.; Lery, X.; Bonhomme, A.; Sauphanor, B.; Lopez-Ferber, M. Cydia pomonella granulovirus Genotypes Overcome Virus Resistance in the Codling Moth and Improve Virus Efficiency by Selection against Resistant Hosts. Appl. Environm. Microbiol. 2009, 75, 925–930. [Google Scholar] [CrossRef]
- Arneodo, J.D.; de Anna, J.; Salvador, R.; Farinon, M.; Quintana, G.; Sciocco-Cap, A. Prospection and molecular analysis of CpGV isolates infecting Cydia pomonella at different geographical locations in Argentina. Ann. Appl. Biol. 2015, 166, 67–74. [Google Scholar] [CrossRef]
- Quintana, G.; Instituto de Microbiología y Zoología Agrícola, Instituto Nacional de Tecnología Agropecuaria, Castelar, Bs. As., Argentina. Personal Communication, 2014.
- Ríos-Velasco, C.; Sánchez-Valdez, V.M.; Gallegos-Morales, G.; Cambero-Campos, O.J. Evaluación en campo del granulovirus CpGV sobre Cydia pomonella L. (Lepidoptera: Tortricidae) CpGV field evaluation on Cydia pomonella L. (Lepidoptera: Tortricidae). Rev. Mex. Cienc. Agríc. 2012, 3, 797–804. (In Spanish) [Google Scholar]
- International Potato Center (CIP). Available online: http://cipotato.org (accessed on 10 February 2015).
- Barragán, A.R.; Onore, G.; Zeddam, J.L. Identificación, biología, y comportamiento de las polillas de la papa en el Ecuador. PROMSA-MAG PUCE, Ediciones Genesis: Quito, Ecuador, 2005. (In Spanish) [Google Scholar]
- Rondon, S.I. The potato tuber moth: A literature review of its biology, ecology and control. Am. J. Potato Res. 2010, 87, 149–166. [Google Scholar] [CrossRef]
- Raman, K.V.; Booth, R.H.; Palacios, M. Control of potato tuber moth Phthorimaea operculella (Zeller) in rustic potato stores. Trop. Sci. 1987, 27, 175–194. [Google Scholar]
- Kroschel, J.; Sporleder, M.; Tonnang, H.E.Z.; Juarez, H.; Carhuapoma, P.; Gonzales, J.C.; Simon, R. Predicting climate-change-caused changes in global temperature on potato tuber moth Phthorimaea operculella (Zeller) distribution and abundance using phenology modeling and GIS mapping. Agric. For. Meteorol. 2013, 170, 228–241. [Google Scholar] [CrossRef]
- Reed, E.M.; Springett, B.P. Large-scale field testing of a granulosis virus for the control of the potato moth (Phthorimaea operculella (Zell.) (Lep., Gelechiidae)). Bull. Entomol. Res. 1971, 61, 207–222. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Christian, R.L.; Grace, T.D.C.; Filshie, B.K. Large-scale field propagation and the purification of the granulosis virus of the potato moth, Phthorimaea operculella (Zeller) (Lepidoptera: Gelechiidae). Bull. Entomol. Res. 1978, 68, 385–391. [Google Scholar] [CrossRef]
- Alcázar, J.; Raman, K.V.; Salas, R. Un virus como agente de control de la polilla de la papa Phthorimaea operculella. Rev. Peru. Entomol. 1991, 34, 101–104. (In Spanish) [Google Scholar]
- Kroschel, J.; Kaack, H.J.; Fritsch, E.; Huber, J. Biological control of the potato tuber moth (Phthorimaea operculella Zeller) in the Republic of Yemen using granulosis virus: Propagation and effectiveness of the virus in field trials. Biocontrol Sci. Technol. 1996, 6, 217–226. [Google Scholar] [CrossRef]
- Zeddam, J.L.; Pollet, A.; Mangoendiharjo, S.; Ramadhan, T.H.; Lopez-Ferber, M. Occurrence and virulence of a granulosis virus in Phthorimaea operculella (Lep. Gelechiidae) populations in Indonesia. J. Invertebr. Pathol. 1999, 74, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Laarif, A.; Fattouch, S.; Essid, W.; Marzouki, N.; Salah, H.B.; Hammouda, M.H.B. Epidemiological survey of Phthorimaea operculella granulosis virus in Tunisia. EPPO Bull. 2003, 33, 335–338. [Google Scholar] [CrossRef]
- Lacey, L.A.; Kroschel, J.; Arthurs, S.P.; de la Rosa, F. Control microbiano de la palomilla de la papa Phthorimaea operculella (Lepidoptera: Gelechiidae). Rev. Colomb. Entomol. 2010, 36, 181–189. (In Spanish) [Google Scholar]
- Raman, K.V.; Alcazar, J.; Valdez, A. Biological Control of the Potato Tuber Moth Using Phthorimaea Baculovirus 1992.
- Barea, O.; Bejarano, C.; Calderón, R.; Crespo, L.; Franco, J.; Herbas, J.; Lino, V.; Martínez, E.; Ramos, J. Desarrollo de componentes del manejo integrado de las polillas de la papa (Phthorimaea operculella y Symmetrischema tangolias) en Bolivia y el Bioinsecticida Baculovirus (MATAPOL); PROINPA: Cochabamba, Bolivia, 2002; Available online: http://www.asocam.org/biblioteca/files/original/35b23c7f7ae28eae20b35f797ad89b5b.pdf (accessed on 10 January 2015).
- Suquillo, J.; Rodríguez, P.; Gallegos, P.; Orbe, K.; Zeddam, J.L. Manual Para la Elaboración del Bioinsecticida Bacu-Turin a Través de Premezclas Concentradas Para el Control de las Polillas de la Papa: Tecia Solanivora, Phthorimaea Operculella y Symmetrischema Tangolia. Instituto Nacional de Investigaciones Agropecuarias (INIAP): Carchi, Ecuador, 2012. (In Spanish) [Google Scholar]
- Briese, D.T.; Mende, H.A. Differences in susceptibility to a granulosis virus between field populations of the potato moth, Phthorimaea Operculella (Zeller) (Lepidoptera: Gelechiidae). Bull. Entomol. Res. 1981, 71, 11–18. [Google Scholar] [CrossRef]
- Vickers, J.M.; Cory, J.S.; Entwistle, P.F. DNA characterization of eight geographic isolates of granulosis virus from the potato tuber moth (Phthorimaea operculella) (lepidoptera, gelechiidae). J. Invertebr. Pathol. 1991, 57, 334–342. [Google Scholar] [CrossRef]
- Sporleder, M. The granulovirus of the potato tuber moth Phthorimaea operculella (Zeller): Characterization and prospects for effective mass production and pest control. In Advances in Crop Research Volume 3; Kroschel, J., Ed.; Margraf Verlag: Weikersheim, Germany, 2003; p. 196. [Google Scholar]
- Zeddam, J.L.; Léry, L.; Gómez-Bonilla, Y.; Espinel-Correal, C.; Páez, D.; Rebaudo, F.; López-Ferber, M. Responses of different geographic populations of two potato tuber moth species to genetic variants of Phthorimaea operculella granulovirus. Entomol. Exp. Appl. 2013, 149, 138–147. [Google Scholar]
- Ángeles, I.; Alcázar, J. Susceptibilidad de la polilla Scrobipalpuloides absoluta al virus de la granulosis de Phthorimaea operculella (PoVG). Rev. Peru. Entomol. 1995, 35, 65–70. (In Spanish) [Google Scholar]
- Niño, L.; Notz, A. Patogenicidad de un virus granulosis de la polilla de la papa Tecia solanivora (Povolny 1973) (Lepidoptera: Gelechiidae) en el estado de Mérida, Venezuela. Bol. Entomol. Venez. 2000, 15, 39–48. (In Spanish) [Google Scholar]
- Zeddam, J.L.; Vásquez, M.; Vargas, Z.; Lagnaoui, A. Producción viral y tasas de aplicación del granulovirus usado para el control biológico de las polillas de la papa Phthorimaea operculella y Tecia solanivora (Lepidoptera: Gelechiidae). Plagas 2003, 29, 659–667. (In Spanish) [Google Scholar]
- Lery, X.; Villamizar, L.; Espinel, C.; Zeddam, J.L.; Cotes, A.M.; López-Ferber, M. Analysis of several Colombian Phthorimaea operculella granuloviruses isolated from Tecia solanivora: Detection of a new variable region in the PhopGV genome. IOBC/WPRS Bull. 2008, 31, 83. [Google Scholar]
- Cuartas, P.O.; Villamizar, L.; Espinel, C.C.; Cotes, A.M. Infección de granulovirus nativos sobre Tecia solanivora y Phthorimaea operculella (Lepidoptera: Gelechiidae). Rev. Colomb. Entomol. 2009, 35, 122–129. (In Spanish) [Google Scholar]
- Espinel-Correa, C.; Lery, X.; Villamizar, L.; Gómez, J.; Zeddam, J.L.; Cotes, A.M.; López-Ferber, M. Genetic and biological analysis of Colombian Phthorimaea operculella Granulovirus isolated from Tecia solanivora (Lepidoptera: Gelechiidae). Appl. Environ. Microbiol. 2010, 76, 7617–7625. [Google Scholar] [CrossRef] [PubMed]
- Espinel-Correa, C.; López-Ferber, M.; Zeddam, J.L.; Villamizar, L.; Gómez, J.; Cotes, A.M.; Léry, X. Experimental mixtures of Phthorimaea operculella granulovirus isolates provide high biological efficacy on both Phthorimaea operculella and Tecia solanivora (Lepidoptera: Gelechiidae). J. Invertebr. Pathol. 2012, 110, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Bonilla, Y.; López-Ferber, M.; Caballero, P.; Léry, X.; Muñoz, D. Characterization of a Costa Rican granulovirus strain highly pathogenic against its indigenous hosts, Phthorimaea operculella and Tecia solanivora. Entomol. Exp. Appl. 2011, 140, 238–246. [Google Scholar] [CrossRef]
- Carpio, C.; Olivier, D.; Dupas, S.; Léry, X.; Lopez-Ferber, M.; Orbe, K.; Páez, D.; Rebaudo, F.; Santillan, A.; Yangari, B.; et al. Development of a viral biopesticide for the control of the Guatemala potato tuber moth Tecia solanivora. J. Invertebr. Pathol. 2013, 112, 184–191. [Google Scholar] [CrossRef]
- Moura Mascarin, G.; Batista Alves, S.; Rampelotti-Ferreira, F.T.; Ragassi Urbano, M.; Borges Demétrio, C.G.; Delalibera, I., Jr. Potential of a granulovirus isolate to control Phthorimaea operculella (Lepidoptera: Gelechiidae). BioControl 2010, 55, 657–671. [Google Scholar] [CrossRef]
- Johnson, S.J. Migration and the life history strategy of the fall armyworm, Spodoptera frugiperda, in the Western hemisphere. Insect Sci. Appl. 1987, 8, 543–549. [Google Scholar]
- Clark, P.L.; Molina-Ochoa, J.; Martinelli, S.; Skoda, S.R.; Isenhour, D.I.; Lee, D.J.; Krumm, J.T.; Foster, J.E. Population variation of the fall armyworm, Spodoptera frugiperda, in western hemisphere. J. Insect Sci. 2007, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.J. Detection and biochemical characterization of insecticide resistance in fall armyworm (Lepidoptera: Noctuidae). J. Econ. Entomol. 1992, 85, 675–691. [Google Scholar] [CrossRef]
- Pacheco-Covarrubias, J.J. Monitoring insecticide resistance in Spodoptera frugiperda populations from the Yaqui Valley, Son., Mexico. Resist. Pest Manag. Newsl. 1993, 5, 3–4. [Google Scholar]
- Storer, N.P.; Babcock, J.M.; Schlenz, M.; Meade, T.; Thompson, G.D.; Bing, J.W.; Huckaba, R.M. Discovery and characterization of field resistance to Bt maize: Spodoptera frugiperda (Lepidoptera: Noctuidae) in Puerto Rico. J. Econ. Entomol. 2010, 103, 1031–1038. [Google Scholar] [CrossRef] [PubMed]
- Ríos-Díez, J.D.; Saldamando-Benjumea, C.I. Susceptibility of Spodoptera frugiperda (Lepidoptera: Noctuidae) strains from central Colombia to two insecticides, Methomyl and Lambda-Cyhalothrin: A study of the genetic basis of resistance. J. Econ. Entomol. 2011, 104, 1698–1705. [Google Scholar] [CrossRef] [PubMed]
- León-García, I.; Rodríguez-Leyva, E.; Ortega-Arenas, L.D.; Solís-Aguilar, J.E. Susceptibilidad de Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) a insecticidas asociada a césped en Quintana Roo, México. Agrociencia 2012, 46, 279–287. (In Spanish) [Google Scholar]
- Berretta, M.F.; Rios, M.L.; Sciocco de Cap, A. Characterization of a nuclear polyhedrosis virus of Spodoptera frugiperda from Argentina. J. Invertebr. Pathol. 1998, 71, 280–282. [Google Scholar] [CrossRef] [PubMed]
- Yasem de Romero, M.G.; Romero, E.; Sosa Gómez, D.; Willink, E. Evaluación de aislamientos de baculovirus para el control de Spodoptera frugiperda (Smith, 1797) Lep.: Noctuidae, plaga clave del maíz en el noroeste argentino. Rev. Ind. Agríc. Tucumán 2009, 86, 7–15. (In Spanish) [Google Scholar]
- Valicente, F.H.; Peixoto, M.J.V.V.D.; Paiva, E.; Kitajima, E.W. Identificação e purificação de um vírus da poliedrose nuclear da lagarta Spodoptera frugiperda (J.E. Smith, 1797) (Lepidoptera: Noctuidae). An. Soc. Entomol. Bras. 1989, 18, 71–81. (In Portuguese) [Google Scholar]
- Valicente, F.H.; da Costa, E.F. Controle da lagarta do cartucho Spodoptera frugiperda (J.E. Smith) com baculovirus spodoptera, aplicado via água de irrigação. An. Soc. Entomol. Bras. 1995, 24, 61–67. (In Portuguese) [Google Scholar]
- Arce Gómez, S.; Moscardi, F.; Sosa Gómez, D.R. Susceptibilidade de Spodoptera frugiperda a isolados geograficos de um virus de poliedrose nuclear. Pesqui. Agropecu. Bras. 1999, 34, 1539–1544. (In Portuguese) [Google Scholar]
- Gómez Valderrama, J.A.; Guevara Agudelo, E.J.; Barrera Cubillos, G.P.; Cotes Prado, A.M.; Villamizar Rivero, L.F. Aislamiento, identificación y caracterización de nucleopoliedrovirus nativos de Spodoptera frugiperda en Colombia. Rev. Fac. Nal. Agron. Medellín 2010, 63, 5511–5520. (In Spanish) [Google Scholar]
- Williams, T.; Goulson, D.; Caballero, P.; Cisneros, J.; Martínez, A.M.; Chapman, J.W.; Roman, D.X.; Cave, R. Evaluation of a baculovirus bioinsecticide for small-scale maize growers in Latin America. Biol. Control 1999, 14, 67–75. [Google Scholar] [CrossRef]
- Martínez, A.M.; Goulson, D.; Chapman, J.W.; Caballero, P.; Cave, R.D.; Williams, T. Is it feasible to use optical brightener technology with a baculovirus insecticide for resource-poor maize farmers in Mesoamerica? Biol. Control 2000, 17, 174–181. [Google Scholar] [CrossRef]
- Rangel Núñez, J.C.; Vázquez Ramírez, M.F.; del Rincón Castro, M.C. Caracterización biológica y molecular de cepas exóticas de Baculovirus SfNPV, con actividad bioinsecticida hacia una población mexicana del gusano cogollero del maíz Spodoptera frugiperda (Lepidóptera: Noctuidae). Interciencia 2014, 39, 320–326. (In Spanish) [Google Scholar]
- Ríos-Velasco, C.; Gallegos-Morales, G.; Berlanga-Reyes, D.; Cambero-Campos, J.; Romo-Chacón, A. Mortality and Production of Occlusion Bodies in Spodoptera frugiperda Larvae (Lepidoptera: Noctuidae) Treated with Nucleopolyhedrovirus. Fla. Entomol. 2012, 95, 752–757. [Google Scholar] [CrossRef]
- García-Gutiérrez, C.; Escobedo-Bonilla, C.M.; López, M.A. Infectivity of a Sinaloa Native Isolate of Multicapsid Nuclear Polyhedrosis Virus (SfMNPV) against Fall Armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae). Southwest. Entomol. 2013, 38, 597–604. [Google Scholar] [CrossRef]
- Vásquez, J.; Zeddam, J.L.; Tresierra, A.A. Control biológico del “cogollero del maíz” Spodoptera frugiperda, (Lepidoptera: Noctuidae) con el Baculovirus SfVPN, en Iquitos-Perú. Folia Amazon. 2002, 13, 25–39. (In Spanish) [Google Scholar]
- Agudelo, F.; Romano, M.; Wassink, H.; Cuello de Uzcategui, R. Una poliedrosis de Spodoptera frugiperda en Venezuela. Turrialba 1983, 33, 219–222. (In Spanish) [Google Scholar]
- Simón, O.; Williams, T.; López-Ferber, M.; Caballero, P. Genetic structure of a Spodoptera frugiperda nucleopolyhedrovirus population: High prevalence of deletion genotypes. Appl. Environ. Microbiol. 2004, 70, 5579–5588. [Google Scholar] [CrossRef] [PubMed]
- Simón, O.; Chevenet, F.; Williams, T.; Caballero, P.; López-Ferber, M. Physical and partial genetic map of Spodoptera frugiperda nucleopolyhedrovirus (SfMNPV) genome. Virus Genes 2005, 30, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Simón, O.; Williams, T.; Cerutti, M.; Caballero, P.; López-Ferber, M. Expression of a Peroral Infection Factor Determines Pathogenicity and Population Structure in an Insect Virus. PLoS ONE 2013, 8, e78834. [Google Scholar] [CrossRef] [PubMed]
- López-Ferber, M.; Simón, O.; Williams, T.; Caballero, P. Defective or effective? Mutualistic interactions between virus genotypes. Proc. R. Soc. B Biol. Sci. 2003, 270, 2249–2255. [Google Scholar] [CrossRef]
- Clavijo, G.; Williams, T.; Simón, O.; Muñoz, D.; Cerutti, M.; López-Ferber, M.; Caballero, P. Mixtures of complete and pif1- and pif2-deficient genotypes are required for increased potency of an insect nucleopolyhedrovirus. Virology 2009, 83, 5127–5136. [Google Scholar] [CrossRef]
- Clavijo, G.; Williams, T.; Muñoz, D.; Caballero, P.; López-Ferber, M. Mixed genotype transmission bodies and virions contribute to the maintenance of diversity in an insect virus. Proc. R. Soc. B 2010, 277, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Barrera, G.; Williams, T.; Villamizar, L.; Caballero, P.; Simón, O. Deletion Genotypes Reduce Occlusion Body Potency but Increase Occlusion Body Production in a Colombian Spodoptera frugiperda Nucleopolyhedrovirus Population. PLoS ONE 2013, 8, e77271. [Google Scholar] [CrossRef] [PubMed]
- Villamizar, L.; Guevara, J.; Espinel, C.; Gómez, M.; Gómez, J.; Cuartas, Paola.; Barrera, G.; Cruz, M.; Santos, A.; Uribe, L.; et al. Desarrollo de un Bioplaguicida a Base de Nucleopoliedrovirus Para el Control del Gusano Cogollero del Maíz, Spodoptera Frugiperda; Corpoica: Bogotá, Colombia, 2012; p. 100. (In Spanish) [Google Scholar]
- Gómez, J.; Guevara, J.; Cuartas, P.; Espinel, C.; Villamizar, L. Microencapsulated Spodoptera frugiperda nucleopolyhedrovirus: Insecticidal activity and effect on arthropod populations in maize. Biocontrol Sci. Technol. 2013, 23, 829–847. [Google Scholar] [CrossRef]
- Villamizar, L.; Corpoica (Corporación Colombiana de Investigación Agropecuaria), Bogotá, Colombia. Personal Communication, 2015.
- Valicente, F.H.; Cruz, I. Controle Biológico da Lagarta do Cartucho, Spodoptera Frugiperda, Com o Baculovirus; Circular Técnica N° 15; EMBRAPA/CNPMS: Sete Lagoas, Brazil, 1991; p. 23. (In Portuguese) [Google Scholar]
- Valicente, F.H.; Tuelher, E.S.; Pena, R.C.; Andreazza, R.; Guimarães, M.R.F. Cannibalism and virus production in Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) larvae fed with two leaf substrates inoculated with baculovirus spodoptera. Neotrop. Entomol. 2013, 42, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Martínez, A.M.; Pineda, S.; Figueroa, J.I.; Chavarrieta, J.M.; Williams, T. Los baculovirus como bioinsecticidas: Evaluación de un nucleopoliedrovirus para el combate de Spodoptera frugiperda (Lepidoptera: Noctuidae) en México y Honduras. Cienc. Nicolaita. 2012, 35, pp. 35–47. Available online: http://www.cic.cn.umich.mx/index.php/cn/article/view/108/31 (accessed on 5 November 2014). (In Spanish)
- Zamora-Avilés, N.; Alonso-Vargas, J.; Pineda, S.; Isaac-Figueroa, J.; Lobit, P.; Martínez-Castillo, A.M. Effects of a nucleopolyhedrovirus in mixtures with azadirachtin on Spodoptera frugiperda (JE Smith) (Lepidoptera: Noctuidae) larvae and viral occlusion body production. Biocontrol Sci. Technol. 2013, 23, 521–534. [Google Scholar] [CrossRef]
- Pineda, S.; Pérez-Robledo, C.A.; Hernández, R.E.; de la Rosa, J.F.; Chavarrieta, J.M.; Martínez, A.M. Combined and individual effects of a nucleopolyhedrovirus and azadirachtin on the mortality and maize-leaf consumption of Spodoptera frugiperda. Phytoparasitica 2014, 42, 571–578. [Google Scholar] [CrossRef]
- Castillejos, V.; Trujillo, J.; Ortega, L.D.; Santizo, J.A.; Cisneros, J.; Penagos, D.I.; Valle, J.; Williams, T. Granular phagostimulant nucleopolyhedrovirus formulations for control of Spodoptera frugiperda in maize. Biol. Control 2002, 24, 300–310. [Google Scholar] [CrossRef]
- Cuartas, P.O.; Barrera, G.; Barreto, E.; Villamizar, L. Characterization of a Colombian granulovirus (Baculoviridae: Betabaculovirus) isolated from Spodoptera frugiperda (Lepidoptera: Noctuidae) larvae. Biocontrol Sci. Technol. 2014, 24, 1265–1285. [Google Scholar] [CrossRef]
- El-Sharkawy, M.A. Cassava biology and physiology. Plant Mol. Biol. 2004, 56, 481–501. [Google Scholar] [CrossRef] [PubMed]
- Bellotti, A.C.; Arias, B.; Guzman, O.L. Biological control of the cassava hornworm Erinnyis ello (Lepidoptera: Sphingidae). Fla. Entomol. 1992, 75, 506–515. [Google Scholar] [CrossRef]
- Bellotti, A.C.; Arias, B.; Reyes, J.A. Manejo de plagas de la yuca. In La Yuca en el Tercer Milenio. Sistemas Modernos de Producción, Procesamiento, Utilización y Comercialización; Ospina, B., Ceballos, H., Eds.; Centro Internacional de Agricultura Tropical (CIAT): Cali, Colombia, 2002; pp. 220–233. (In Spanish) [Google Scholar]
- Schmitt, A.T. Using Baculovirus erinnyis in the biological control of cassava hornworm. Cassava Newsl. 1988, 12, 1–4. [Google Scholar]
- Bellotti, A.C.; Arias, B. Biology, ecology and biological control of the cassava hornworm (Erinnyis ello). In Cassava Protection Workshop; Series CE-14; Brekelbaum, T., Bellotti, A., Lozano, J.C., Eds.; Centro Internacional de Agricultura Tropical (CIAT): Cali, Colombia, 1978; pp. 227–232. [Google Scholar]
- Laberry, R. La Aplicación de un Programa MIP en Producción Industrial de Yuca. En: Memorias del Congreso Biodiversidad y Micorrizas; CIAT; La Asociacion Colombiana de Fitopatologia y Ciencias Afines: Cali, Colombia, 1977; pp. 136–137. (In Spanish) [Google Scholar]
- Czepak, C.; Cordeiro Albernaz, K.; Vivan, L.M.; GuiMarães, H.O.; Carvalhais, T. Primeiro registro de ocorrência de Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) no Brasil. Pesqui. Agropecu. Trop., Goiâ. 2013, 43, 110–113. [Google Scholar] [CrossRef]
- Specht, A.; Sosa-Gómez, D.R.; Vieira de Paula-Moraes, S.; Cavaguchi Yano, S.A. Identificação morfológica e molecular de Helicoverpa armigera (Lepidoptera: Noctuidae) e ampliação de seu registro de ocorrência no Brasil. Pesqui. Agropecu. Bras. (Brasília) 2013, 48, 689–692. [Google Scholar] [CrossRef]
- Tay, W.T.; Soria, M.F.; Walsh, T.; Thomazoni, D.; Silvie, P.; Behere, G.T.; Anderson, C.; Downes, S. A Brave New World for an Old World Pest: Helicoverpa armigera (Lepidoptera: Noctuidae) in Brazil. PLoS ONE 2013, 8, e80134. [Google Scholar] [CrossRef]
- SENAVE. 2013. Available online: http://www.senave.gov.py/noticias-50-SENAVE-Reportan-presencia-de-peligrosa-plaga-en-nuestro-pais.html (accessed on 5 February 2015).
- Murúa, M.G.; Scalora, F.S.; Navarro, F.R.; Cazado, L.E.; Casmuz, A.; Villagrán, M.E.; Lobos, E.; Gastaminza, G. First Record of Helicoverpa armigera (Lepidoptera: Noctuidae) in Argentina. Fla. Entomol. 2014, 97, 854–856. [Google Scholar] [CrossRef]
- De Bueno, A.F.; Sosa-Gómez, D.R. The Old World Bollworm in the Neotropical Region: The experience of Brazilian Growers with Helicoverpa armigera. Outlooks Pest Manag. 2014, 25, 260–264. [Google Scholar]
- Coll, O.R.; Saini, E.D. Insectos y Ácaros Perjudiciales al Cultivo de la Yerba Mate en la Republica Argentina; Estación Experimental Agropecuaria Montecarlo, Misiones, Ediciones INTA: Buenos Aires, Argentina, 1992; p. 48. (In Spanish) [Google Scholar]
- Trujillo, M.R. Agroecosistema yerbatero de alta densidad: Plagas y sus enemigos naturales. In Erva-Mate: Biologia e Cultura no Cone Sul; Winge, H., Ferreira, A.G., Mariath, J.E.A., Tarasconi, L.C., Eds.; Editora da Universidade: Porto Alegre, Brazil, 1995; pp. 129–134. (In Portuguese) [Google Scholar]
- Alves, L.F.A.; Santana, D.L.Q.; Brancalhão, R.M.C. Ocorrência de Perigonia lusca (Fabr.) (Lep.: Sphingidae) em Erva-Mate (Ilex paraguariensis) no Brasil. Neotrop. Entomol. 2001, 30, 339–340. (In Portuguese) [Google Scholar] [CrossRef]
- Alves, L.F.A.; Brancalhão, R.M.C.; Santana, D.L.Q. Ocorrência de Baculovirus em Lagartas de Perigonia lusca (Fabr.) (Lep., Sphingidae) no Brasil. Neotrop. Entomol. 2001, 30, 493–494. (In Portuguese) [Google Scholar] [CrossRef]
- Sosa-Gómez, D.R.; Kitajima, E.W.; Rolón, M. First record of entomopathogenic diseases in Paraguay tea agroecosystem in Argentina. Fla. Entomol. 1994, 77, 378–382. [Google Scholar] [CrossRef]
- Melo, F. Personal Communication, Departamento de Biologia Celular, Universidade de Brasília-UnB: Brasília, Brazil, 2014.
- Sciocco-Cap, A.; Parola, A.; Goldberg, A.; Ghiringhelli, D.; Romanowski, V. Characterization of a granulovirus (EpapGV) isolated from Epinotia aporema (Lepidoptera: Tortricidae) larvae. Appl. Environ. Microbiol. 2001, 67, 3702–3706. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, A.; Romanowski, V.; Federici, B.; Sciocco-Cap, A. Effects of the EpapGV granulovirus on its host, Epinotia aporema. J. Invertebr. Pathol. 2002, 80, 10–21. [Google Scholar] [CrossRef]
- Ferrelli, M.L.; Salvador, R.; Biedma, M.E.; Berretta, M.F.; Haase, S.; Sciocco-Cap, A.; Ghiringhelli, P.D.; Romanowski, V. Genome of Epinotia aporema granulovirus (EpapGV), a polyorganotropic fast killing betabaculovirus with a novel thymidylate kinase gene. BMC Genomics 2012, 13, e548. [Google Scholar] [CrossRef]
- Parola, A.; Sciocco-Cap, A.; Glickmann, G.; Romanowski, V. An immunochemical method for quantification of Epinotia aporema granulovirus (EpapGV). J. Virol. Methods 2003, 112, 13–21. [Google Scholar] [CrossRef]
- Manzan, M.A.; Biedma, M.; Aljinovic, E.M.; Sciocco-Cap, A.; Romanowski, V.; Ghiringhelli, P.D. Multiplex PCR and quality control of Epinotia aporema Granulovirus (EpapGV) production. Virus Genes 2008, 37, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Moscardi, F.; Sosa-Gomez, D.R. Use of viruses against soybean caterpillars in Brazil. In Pest Management in Soybean; Copping, L.G., Green, M.B., Rees, R.T., Eds.; Springer: Heidelberg, Germany, 1992; pp. 98–109. [Google Scholar]
- Alexandre, T.M.; Ribeiro, Z.M.A.; Craveiro, S.R.; Cunha, F.; Fonseca, I.C.; Moscardi, F.; Castro, M.E. Evaluation of seven viral isolates as potential biocontrol agents against Pseudoplusia includens (Lepidoptera: Noctuidae) caterpillars. J. Invertebr. Pathol. 2010, 105, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Craveiro, S.R.; Melo, F.L.; Ribeiro, Z.M.A.; Ribeiro, B.M.; Báo, S.N.; Inglis, P.W.; Castro, M.E. Pseudoplusia includens single nucleopolyhedrovirus: Genetic diversity, phylogeny and hypervariability of the pif-2 gene. J. Invertebr. Pathol. 2013, 114, 258–267. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, V.A.; Belaich, M.N.; Quintana, G.; Sciocco-Cap, A.; Ghiringhelli, P.D. Isolation and Characterization of a Nucleopolyhedrovirus from Rachiplusia nu (Guenée) (Lepidoptera: Noctuidae). Int. J. Virol. Mol. Biol. 2012, 1, 28–34. [Google Scholar]
- Arneodo, J.; Jakubowicz, V.; Taibo, C.; Sciocco-Cap, A. Avances en la Caracterización de dos Baculovirus Aislados de la Oruga Medidora Rachiplusia nu (Lepidoptera: Noctuidae); XV Jornadas Argentinas de Microbiología: Córdoba, Argentina, 2014; p. 224. Available online: http://microbiologia2014.com.ar/website/wp-content/uploads/2014/08/Libro-de-Resúmenes-JAM-2014.pdf (accesses on 22 December 2014).
- Castro, M.E.; Ribeiro, Z.M.; Santos, A.C.; Souza, M.L.; Machado, E.B.; Sousa, N.J.; Moscardi, F. Identification of a new nucleopolyhedrovirus from naturally-infected Condylorrhiza vestigialis (Guenee) (Lepidoptera: Crambidae) larvae on poplar plantations in South Brazil. J. Invertebr. Pathol. 2009, 102, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Williams, T.; Cisneros, J. Formulación y aplicación de los baculovirus bioinsecticidas. In Los Baculovirus y Sus Aplicaciones Como Bioinsecticidas; Caballero, P., López Ferber, M., Williams, T., Eds.; Phytoma—Universidad Pública de Navarra: Pamplona, España, 2001. (In Spanish) [Google Scholar]
- Steinke, W.E.; Giles, D.K. Delivery systems for biorational agents. In Biorational Pest Control Agents: Formulation and Delivery; American Chemical Society Symposium Series 595; Hall, F.R., Barry, J.W., Eds.; American Chemical Society: Washington, DC, USA, 1995. [Google Scholar]
- Jones, K.A.; Westby, A.; Reilly, P.J.A.; Jeger, M.J. The exploitation of microorganisms in the developing countries of the tropics. In Exploitation of Micro-Organisms; Jones, D.G., Ed.; Chapman and Hall: London, UK, 1993; pp. 343–370. [Google Scholar]
- Shapiro, M. In vivo mass production of insect viruses. In Microbial and Viral Pesticides; Kurstak, E., Ed.; Marcel Dekker: New York, NY, USA, 1982; pp. 463–492. [Google Scholar]
- Dulmage, H.T.; Martínez, A.J.; Correa, J.A. Recovery of the nuclear polyhedrosis virus of the cabbage looper Trichoplusia ni by coprecipitation with lactose. J. Invertebr. Pathol. 1970, 16, 80–83. [Google Scholar] [CrossRef] [PubMed]
- Couch, T.L.; Ignoffo, C.M. Formulation of insect pathogens. In Microbial Control of Insect Pests and Plant Diseases; Burges, H.D., Ed.; Academic Press: London, UK, 1981; pp. 621–634. [Google Scholar]
- Mendugo, C.C.; Ferraz, G.; Maia, A.H.N.; Freitas, C.C.L. Evaluation of a wettable powder formulation for the nuclear polyhedrosis virus of Anticarsia gemmatalis (Lep.: Noctuidae). Pestic. Sci. 1997, 51, 153–156. [Google Scholar] [CrossRef]
- Smith, D.B.; Hostetter, D.L.; Ignoffo, C.M. Formulation and equipment effects on application of a viral (Baculovirus heliothis) insecticide. J. Econ. Entomol. 1978, 71, 633–637. [Google Scholar] [CrossRef]
- Ignoffo, C.M.; García, C.; Saathoff, S.G. Sunlight stability and rain-fastness of formulations of Baculovirus Heliothis. Environ. Entomol. 1977, 26, 1470–1474. [Google Scholar] [CrossRef]
- Smith, D.B.; Hostetter, D.L.; Pinnell, R.E.; Ignoffo, C.M. Laboratory formulation comparisons for a bacterial (Bacillus thuringiensis) and a viral (Baculovirus heliothis) insecticide. J. Econ. Entomol. 1980, 73, 18–21. [Google Scholar] [CrossRef]
- Smith, D.B.; Hostetter, D.L.; Pinnell, R.E.; Ignoffo, C.M. Laboratory studies of aerial adjuvants: Formulation development. J. Econ. Entomol. 1982, 75, 16–20. [Google Scholar] [CrossRef]
- Henry, J.E. Experimental application of Nosema locustae for control of grasshoppers. J. Invertebr. Pathol. 1971, 18, 389–394. [Google Scholar] [CrossRef]
- Hostetter, D.L.; Pinnell, R.E. Laboratory evaluation of plant-derived granules for bollworm control with virus. J. Ga. Entomol. Soc. 1983, 18, 155–159. [Google Scholar]
- Bell, M.R.; Kranavel, R.F. Tobacco budworm: Development of a spray adjuvant to increase effectiveness of a nuclear polyhedrosis virus. J. Econ. Entomol. 1978, 71, 350–352. [Google Scholar] [CrossRef]
- Cañas, L.A.; O’Neil, R.J. Applications of sugar solutions to maize and the impact of natural enemies on fall armyworm. Int. J. Pest Manag. 1998, 44, 59–64. [Google Scholar] [CrossRef]
- Shapiro, M. Use of optical brighteners as radiation protectants for the gypsy moth (Lepidoptera: Lymantriidae) nuclear polyhedrovirus. J. Econ. Entomol. 1992, 85, 1682–1686. [Google Scholar] [CrossRef]
- Wringley, G. Mineral oils as carriers for ultra-low-volume (UVL) spraying. Proc. Natl. Acad. Sci. U.S.A. 1973, 19, 54–61. [Google Scholar]
- Hamm, J.J.; Shapiro, M. Infectivity of fall armyworm (Lepidoptera: Noctuidae) nuclear polyhedrosis virus enhanced by a fluorescent brightener. J. Econ. Entomol. 1992, 85, 2149–2152. [Google Scholar] [CrossRef]
- Mondragón, G.; Pineda, A.; Martínez, A.; Martínez, A.M. Optical brightener Tinopal C1101 as an ultraviolet protectant for a nucleopolyhedrovirus. Commun. Agric. Biol. Sci. 2007, 72, 243–247. [Google Scholar]
- Martínez, A.M.; Simón, O.; Williams, T.; Caballero, P. Effect of optical brighteners on the insecticidal activity of a nucleopolyhedrovirus in three instars of Spodoptera frugiperda (Lepidoptera: Noctuidae). Entomol. Exp. Appl. 2003, 109, 139–146. [Google Scholar] [CrossRef]
- Washburn, J.O.; Kirkpatrick, B.A.; Haas-Stapleton, E.; Volkman, L.E. Evidence that the stilbene-derived optical brightener M2R enhances Autographa californica M nucleopolyhedrovirus infection of Trichoplusia ni and Heliothis virescens by preventing sloughing of infected midgut epithelial cells. Biol. Control 1998, 11, 58–69. [Google Scholar] [CrossRef]
- Wang, P.; Granados, R. Calcofluor disrupts the midgut defense system in insects. Insect Biochem. Mol. Biol. 2000, 30, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Morales, L.; Moscardi, F.; Sosa-Gómez, D.R.; Paro, F.E.; Soldorio, I.L. Enhanced activity of Anticarsia gemmatalis Hüb. (Lepidoptera: Noctuidae) nuclear polyhedrosis virus by boric acid in the laboratory. An. Soc. Entomol. Bras. 1997, 26, 115–120. [Google Scholar] [CrossRef]
- Cisneros, J.; Pérez, J.A.; Penagos, D.I.; Ruiz, J.; Goulson, D.; Caballero, P.; Cave, R.D.; Williams, T. Formulation of a nucleopolyhedrovirus with boric acid for control of Spodoptera frugiperda (Lepidoptera: Noctuidae) in maize. Biol. Control 2002, 23, 87–95. [Google Scholar] [CrossRef]
- Ignoffo, C.M.; Shasha, B.S.; Shapiro, M. Sunlight ultraviolet protection of the Heliothis nuclear polyhedrosis virus through starch-encapsulation technology. J. Invertebr. Pathol. 1991, 57, 134–136. [Google Scholar] [CrossRef]
- Morales-Ramos, L.H.; McGuire, M.R.; Galán-Wong, L.J. Utilization of several biopolymers of granular formulations of Bacillus thuringiensis. J. Econ. Entomol. 1998, 91, 1109–1113. [Google Scholar] [CrossRef]
- Morales-Ramos, L.H.; McGuire, M.R.; Galán-Wong, L.J.; Franco-Castro, R. Evaluation of pectin, gelatin, and starch granular formulations of Bacillus thuringiensis. Southwest. Entomol. 2000, 25, 59–67. [Google Scholar]
- Villamizar, L.; Barrera, G.; Cotes, A.M.; Martínez, F. Eudragit S100 microparticles containing Spodoptera frugiperda nucleopolyehedrovirus: Physicochemical characterization, photostability and in vitro virus release. J. Microencapsul. 2010, 27, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Tamez-Guerra, P.; McGuire, M.R.; Behle, R.W.; Hamm, J.J.; Sumner, H.R.; Shasha, B.S. Sunlight persistence and rainfastness of spray-dried formulations of the Anagrapha falcifera baculovirus. J. Econ. Entomol. 2000, 93, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Gioria, V.; Beccaría, A.; Claus, J.D. Crecimiento, metabolismo y producción de baculovirus en cultivos en suspensión de una línea celular del insecto lepidóptero Anticarsia gemmatalis. Quim. Viva 2006, 1, 27–45. (In Spanish) [Google Scholar]
- Claus, J.D.; Remondetto, G.; Guerrero, S.; Demonte, A.; Murguía, M.; Mancipar, A. Anticarsia gemmatalis nuclear polyhedrosis virus replication in serum-free and serum-reduced insect cell cultures. J. Biotechnol. 1993, 31, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Micheloud, G.A.; Gioria, V.V.; Pérez, G.; Claus, J.D. Production of occlusion bodies of Anticarsia gemmatalis multiple nucleopolyhedrovirus in serum-free suspension cultures of the saUFL-AG-286 cell line: Influence of infection conditions and statistical optimization. J. Virol. Methods 2009, 162, 258–266. [Google Scholar] [CrossRef]
- Shuler, M.A.; Kargi, F. Bioprocess Engineering—Basic Concepts, 2nd ed.; Prentice Hall of India: Upper Saddle River, NJ, USA, 2006. [Google Scholar]
- Micheloud, G.A.; Gioria, V.V.; Eberhardt, I.; Visnovsky, G.; Claus, J.D. Production of the Anticarsia gemmatalis multiple nucleopolyhedrovirus in serum-free suspension cultures of the saUFL-AG-286 cell line in stirred reactor and airlift reactor. J. Virol. Methods 2011, 178, 106–116. [Google Scholar] [CrossRef]
- Claus, J.D.; Gioria, V.V.; Micheloud, G.A.; Visnovsky, G. Production of Insecticidal Baculoviruses in Insect Cell Cultures: Potential and Limitations. In Insecticides—Basic and Other Applications; Soloneski, S., Larramendy, M., Eds.; InTech: Rijeka, Croatia, 2012; pp. 127–152. [Google Scholar]
- Van Beek, N.; Davis, D.C. Baculovirus insecticide production in insect larvae. Methods Mol. Biol. 2014, 388, 367–378. [Google Scholar] [CrossRef]
- Keeley, L.L.; Hayes, T.K. Speculations on biotechnology applications for insect neuroendocrine research. Insect Biochem. 1987, 17, 639–651. [Google Scholar] [CrossRef]
- Maeda, S. Increased insecticidal effect by a recombinant baculovirus carrying a synthetic diuretic hormone gene. Biochem. Biophys. Res. Commun. 1989, 165, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Menn, J.J.; Borkovec, A.B. Insect neuropeptides—Potential new insect control agents. J. Agric. Food Chem. 1989, 37, 271–278. [Google Scholar] [CrossRef]
- Carbonell, L.F.; Hodge, M.R.; Tomalski, M.D.; Miller, L.K. Synthesis of a gene coding for an insect specific scorpion neurotoxin and attempts to express it using baculovirus vectors. Gene 1988, 73, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, J.A.; Bonning, B.C.; Harrison, R.L. Expression, Delivery and Function of Insecticidal Proteins Expressed by Recombinant Baculoviruses. Viruses 2015, 7, 422–455. [Google Scholar] [CrossRef] [PubMed]
- Bonning, B.C.; Hammock, B.D. Development of recombinant baculoviruses for insect control. Annu. Rev. Entomol. 1996, 41, 191–210. [Google Scholar] [CrossRef] [PubMed]
- Wood, H.A. Genetically enhanced baculovirus insecticides. In Molecular Biology of the Biological Control of Pests and Diseases of Plants; Gunasekaran, M., Weber, D.J., Eds.; CRC Press: Boca Raton, FL, USA, 1996; pp. 91–104. [Google Scholar]
- Coast, G.M.; Orchard, I.; Phillips, J.E.; Schooley, D.A. Insect diuretic and antidiuretic hormones. Adv. Insect Physiol. 2002, 29, 279–400. [Google Scholar]
- Gade, G. Regulation of intermediary metabolism and water balance of insects by neuropeptides. Annu. Rev. Entomol. 2004, 49, 93–113. [Google Scholar] [CrossRef] [PubMed]
- Holman, G.M.; Nachman, R.J.; Wright, M.S. Insect neuropeptides. Annu. Rev. Entomol. 1990, 35, 201–217. [Google Scholar] [CrossRef] [PubMed]
- O’Reilly, D.R. Baculovirus-encoded ecdysteroid UDP-glucosyltransferases. Insect Biochem. Mol. Biol. 1995, 25, 541–550. [Google Scholar] [CrossRef]
- O’Reilly, D.R.; Miller, L.K. A baculovirus blocks insect molting by producing ecdysteroid UDP-glucosyltransferase. Science 1989, 245, 1110–1112. [Google Scholar] [CrossRef] [PubMed]
- Slavicek, J.M. Baculovirus Enhancins and Their Role in Viral Pathogenicity. In Molecular Virology; Adoga, M.P., Ed.; InTech: Rijeka, Croatia, 2012; pp. 147–168. [Google Scholar]
- Arana, E.I.; Albariño, C.G.; O’Reilly, D.; Ghiringhelli, P.D.; Romanowski, V. Generation of a recombinant Anticarsia gemmatalis multicapsid nucleopolyhedrovirus expressing a foreign gene under the control of a very late promoter. Virus Genes 2001, 22, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, B.M.; Gatti, C.D.; Costa, M.H.; Moscardi, F.; Maruniak, J.E.; Possee, R.D.; Zanotto, P.M. Construction of a recombinant Anticarsia gemmatalis nucleopolyhedrovirus (AgMNPV-2D) harbouring the beta-galactosidase gene. Arch. Virol. 2001, 146, 1355–1367. [Google Scholar] [CrossRef] [PubMed]
- Pinedo, F.J.R.; Moscardi, F.; Luque, T.; Olszewski, J.A.; Ribeiro, B.M. Inactivation of the ecdysteroid UDP-glucosyltransferase (egt) gene of Anticarsia gemmatalis nucleopolyhedrovirus (AgMNPV) improves its virulence towards its insect host. Biol. Control 2003, 27, 336–344. [Google Scholar] [CrossRef]
- Haase, S.; McCarthy, C.B.; Ferrelli, M.L.; Pidre, M.L.; Sciocco-Cap, A.; Romanowski, V. Development of a recombination system for the generation of occlusion positive genetically modified Anticarsia gemmatalis Multiple Nucleopolyhedrovirus. Viruses 2015, 7, 1599–1612. [Google Scholar] [CrossRef] [PubMed]
- Haase, S.; López, M.G.; Jaramillo, C.; Sciocco-Cap, A.; Taboga, O.; Romanowski, V. Study of polyhedrin functional complementation among nucleopolyhedroviruses. Manuscript in preparation.
- Inceoglu, A.B.; Kamita, S.G.; Hammock, B.D. Genetically modified baculoviruses: A historical overview and future outlook. Adv. Virus Res. 2006, 68, 323–360. [Google Scholar]
- Wood, H.A.; Hughes, P.R.; Shelton, A. Field studies of the co-occlusion strategy with a genetically altered isolate of the Autographa californica nuclear polyhedrosis virus. Environ. Entomol. 1994, 23, 211–219. [Google Scholar] [CrossRef]
- Lima, A.A.; Aragao, C.W.; de Castro, M.E.; Oliveira, J.V.; Sosa Gomez, D.R.; Ribeiro, B.M. A recombinant Anticarsia gemmatalis MNPV harboring chiA and v-cath genes from Choristoneura fumiferana defective NPV induce host liquefaction and increased insecticidal activity. PLoS ONE 2013, 8, e74592. [Google Scholar] [CrossRef] [PubMed]
- Simón, O.; Williams, T.; Asensio, A.C.; Ros, S.; Gaya, A.; Caballero, P.; Possee, R.D. Sf29 gene of Spodoptera frugiperda multiple nucleopolyhedrovirus is a viral factor that determines the number of virions in occlusion bodies. J. Virol. 2008, 82, 7897–7904. [Google Scholar] [CrossRef] [PubMed]
- Bonning, B.C.; Boughton, A.J.; Jin, H.; Harrison, R.L. Genetic enhancement of baculovirus insecticides. In Advances in Microbial Control of Insect Pests; Upadhyay, K., Ed.; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2002. [Google Scholar]
- Cory, J.S.; Hirst, M.L.; Williams, T.; Halls, R.S.; Goulson, D.; Green, B.M.; Carty, T.M.; Possee, R.D.; Cayley, P.J.; Bishop, D.H.L.; et al. Field trial of a genetically improved baculovirus insecticide. Nature (Lond.) 1994, 370, 138–140. [Google Scholar] [CrossRef]
- Ardisson-Araújo, D.M.; Melo, F.L.; de Souza Andrade, M.; Brancalhão, R.M.C.; Báo, S.N.; Ribeiro, B.M. Complete genome sequence of the first non-Asian isolate of Bombyx mori nucleopolyhedrovirus. Virus Genes 2014, 49, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.V.C.; Wolf, J.L.C.; Garcia-Muriak, A.; Ribeiro, B.M.; de Castro, M.E.; de Souza, M.L.; Moscardi, F.; Maruniak, J.E.; Zanotto, P.M. Genome of the most widely used viral biopesticide: Anticarsia gemmatalis multiple nucleopolyhedrovirus. J. Gen. Virol. 2006, 87, 3233–3250. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J.L.; Valicente, F.H.; Martins, R.; Oliveira, J.V.; Zanotto, P.M. Analysis of the genome of Spodoptera frugiperda nucleopolyhedrovirus (SfMNPV-19) and of the high genomic heterogeneity in group II nucleopolyhedroviruses. J. Gen. Virol. 2008, 89, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Meister, G.; Tuschl, T. Mechanisms of gene silencing by double-stranded RNA. Nature 2004, 431, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Sander, J.D.; Joung, J.K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat. Biotechnol. 2014, 32, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Herbert, T.; Vonada, R.; Jenkins, M.; Byon, R.; Frausto Leyva, J.M. Environmental Funds and Payments for Ecosystems Services: RedLAC Capacity Building Project for Environmental Funds; RedLAC: Rio de Janeiro, Brazil, 2010. Available online: http://www.forest-trends.org/documents/files/doc_2627.pdf (accessed on 20 December 2014).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haase, S.; Sciocco-Cap, A.; Romanowski, V. Baculovirus Insecticides in Latin America: Historical Overview, Current Status and Future Perspectives. Viruses 2015, 7, 2230-2267. https://doi.org/10.3390/v7052230
Haase S, Sciocco-Cap A, Romanowski V. Baculovirus Insecticides in Latin America: Historical Overview, Current Status and Future Perspectives. Viruses. 2015; 7(5):2230-2267. https://doi.org/10.3390/v7052230
Chicago/Turabian StyleHaase, Santiago, Alicia Sciocco-Cap, and Víctor Romanowski. 2015. "Baculovirus Insecticides in Latin America: Historical Overview, Current Status and Future Perspectives" Viruses 7, no. 5: 2230-2267. https://doi.org/10.3390/v7052230
APA StyleHaase, S., Sciocco-Cap, A., & Romanowski, V. (2015). Baculovirus Insecticides in Latin America: Historical Overview, Current Status and Future Perspectives. Viruses, 7(5), 2230-2267. https://doi.org/10.3390/v7052230





