The Cryptophlebia Leucotreta Granulovirus—10 Years of Commercial Field Use
Abstract
:1. Introduction
2. Results
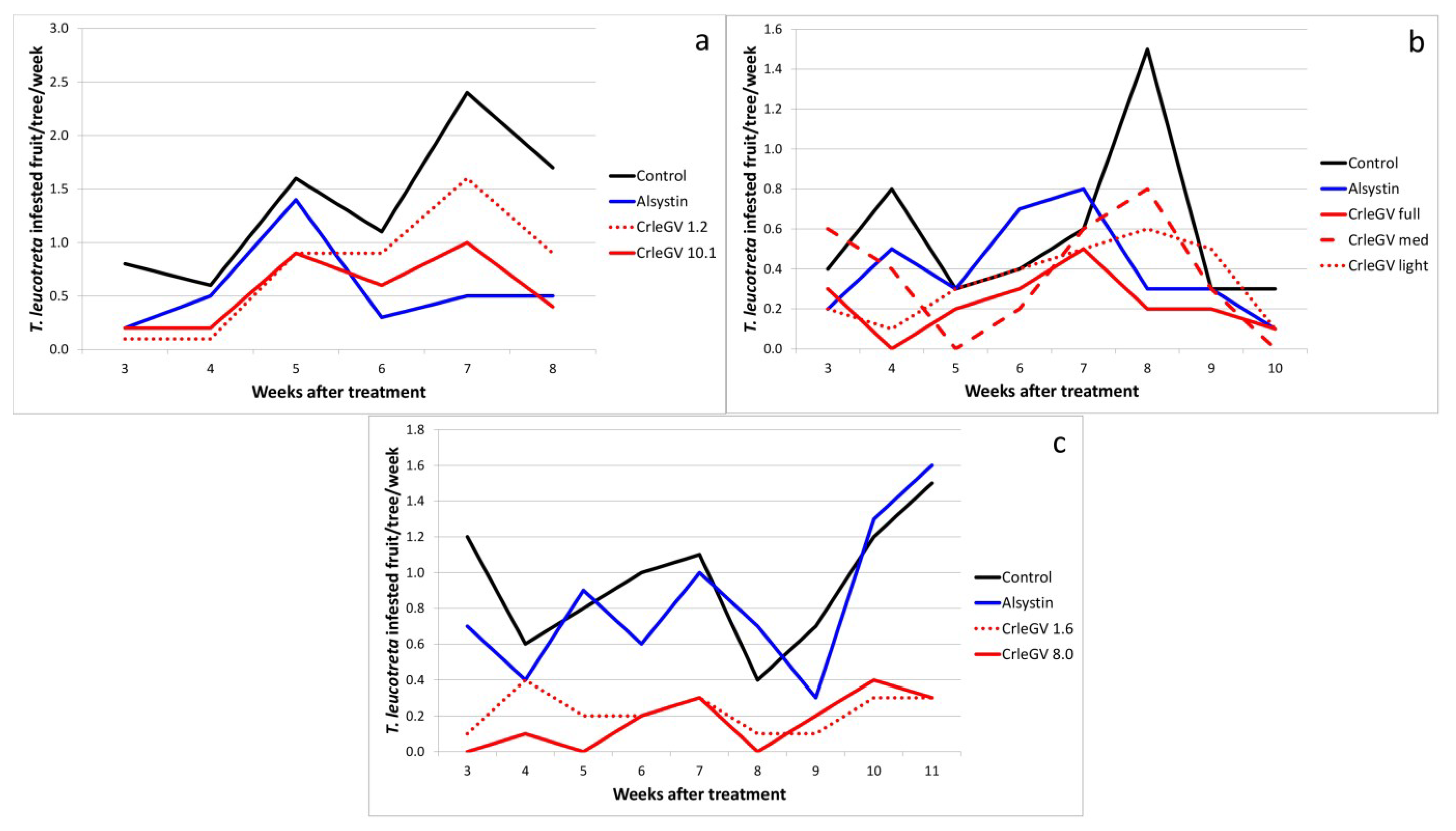
2.1. Unformulated CrleGV
| Site and year initiated | Treatment | Weeks evaluated | Fruit infested/tree/week | Reduction in infestation (%) | ||
|---|---|---|---|---|---|---|
| Mean 2 | SE | |||||
| Product/s | Concentration/s 1 | - | - | - | - | |
| Sun Orange 2001 | Control | - | 6 | 1.37a | 0.27 | - |
| CrleGV | 1.2 × 1014 | - | 0.75ab | 0.23 | 45.1 | |
| CrleGV | 1.0 × 1015 | - | 0.55b | 0.14 | 58.5 | |
| Alsystin | 20 mL | - | 0.57b | 0.17 | 59.8 | |
| Vergenoeg 2002 | Control | - | 8 | 0.57a | 0.15 | - |
| CrleGV full | 9.5 × 1014 | - | 0.22b | 0.05 | 60.9 | |
| CrleGV med | 1.2 × 1015 | - | 0.36ab | 0.10 | 37.0 | |
| CrleGV light | 1.2 × 1015 | - | 0.34ab | 0.07 | 41.3 | |
| Alsystin | 20 mL | - | 0.40ab | 0.09 | 30.4 | |
| Moosrivier 2003 | Control | - | 9 | 0.94a | 0.12 | - |
| CrleGV | 1.6 × 1014 | - | 0.22b | 0.04 | 76.5 | |
| CrleGV | 8.0 × 1014 | - | 0.17b | 0.05 | 82.3 | |
| Alsystin | 20 mL | - | 0.83a | 0.14 | 11.8 | |
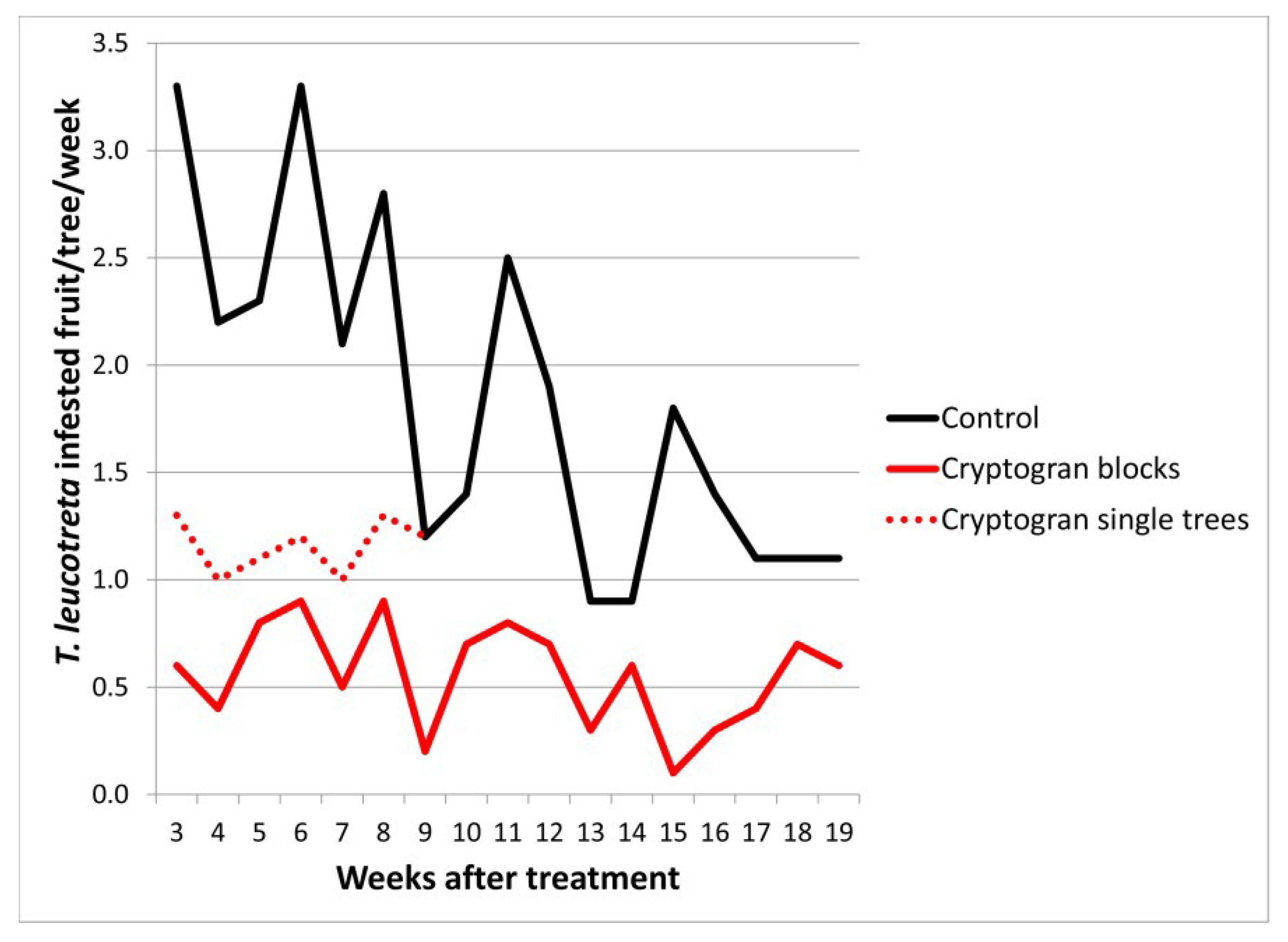
| Carden 2003 | Control | - | 7 | 2.46a | 0.28 | - |
| Cryptogran (blocks) | 6.6 × 1013 (10 mL) | - | 0.61b | 0.10 | 75.0 | |
| Cryptogran (single trees) | 5.2 × 1013 (10 mL) | - | 1.16c | 0.05 | 52.9 | |
| Control | - | 17 | 1.84a | 0.19 | - | |
| Cryptogran (blocks) | 6.6 × 1013 (10 mL) | - | 0.56b | 0.06 | 69.6 | |
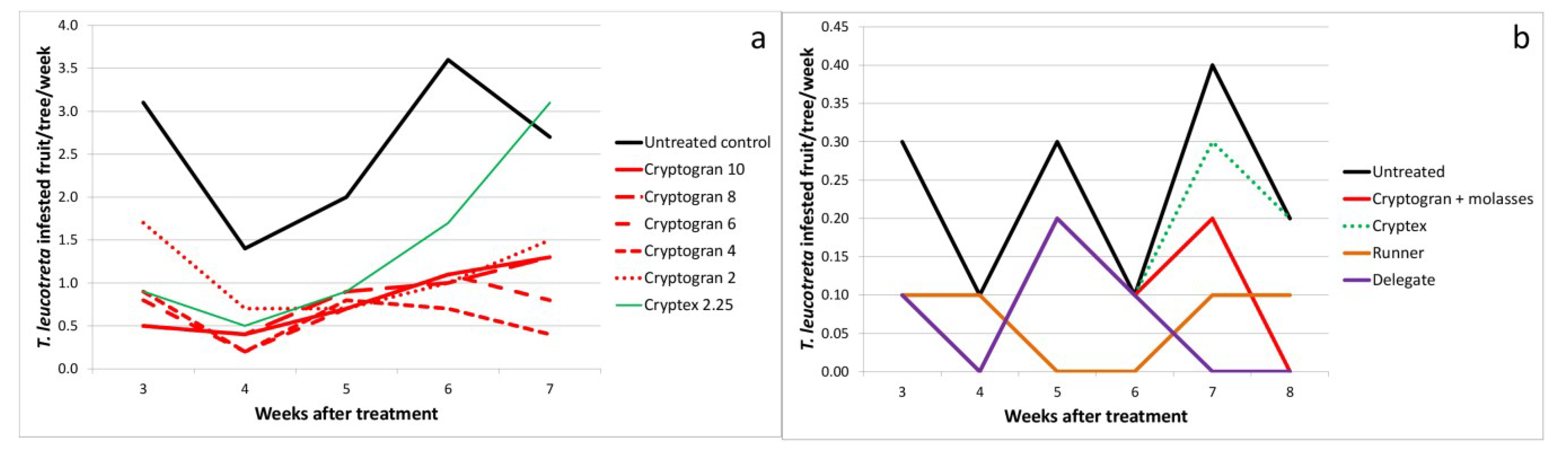
| Bernol 2004 | Control | - | 5 | 2.56a | 0.39 | - |
| Cryptogran | 4.5 × 1013 (10 mL) | - | 0.80bc | 0.17 | 68.7 | |
| Cryptogran | 3.6 × 1013 (8 mL) | - | 0.82bc | 0.16 | 68.0 | |
| Cryptogran | 2.7 × 1013 (6 mL) | - | 0.72bc | 0.15 | 71.9 | |
| Cryptogran | 1.8 × 1013 (4 mL) | - | 0.60b | 0.13 | 76.6 | |
| Cryptogran | 9.0 × 1012 (2 mL) | - | 1.12bc | 0.21 | 56.2 | |
| Cryptex | 4.0 × 1013 (2.25 mL) | - | 1.42c | 0.46 | 44.5 | |
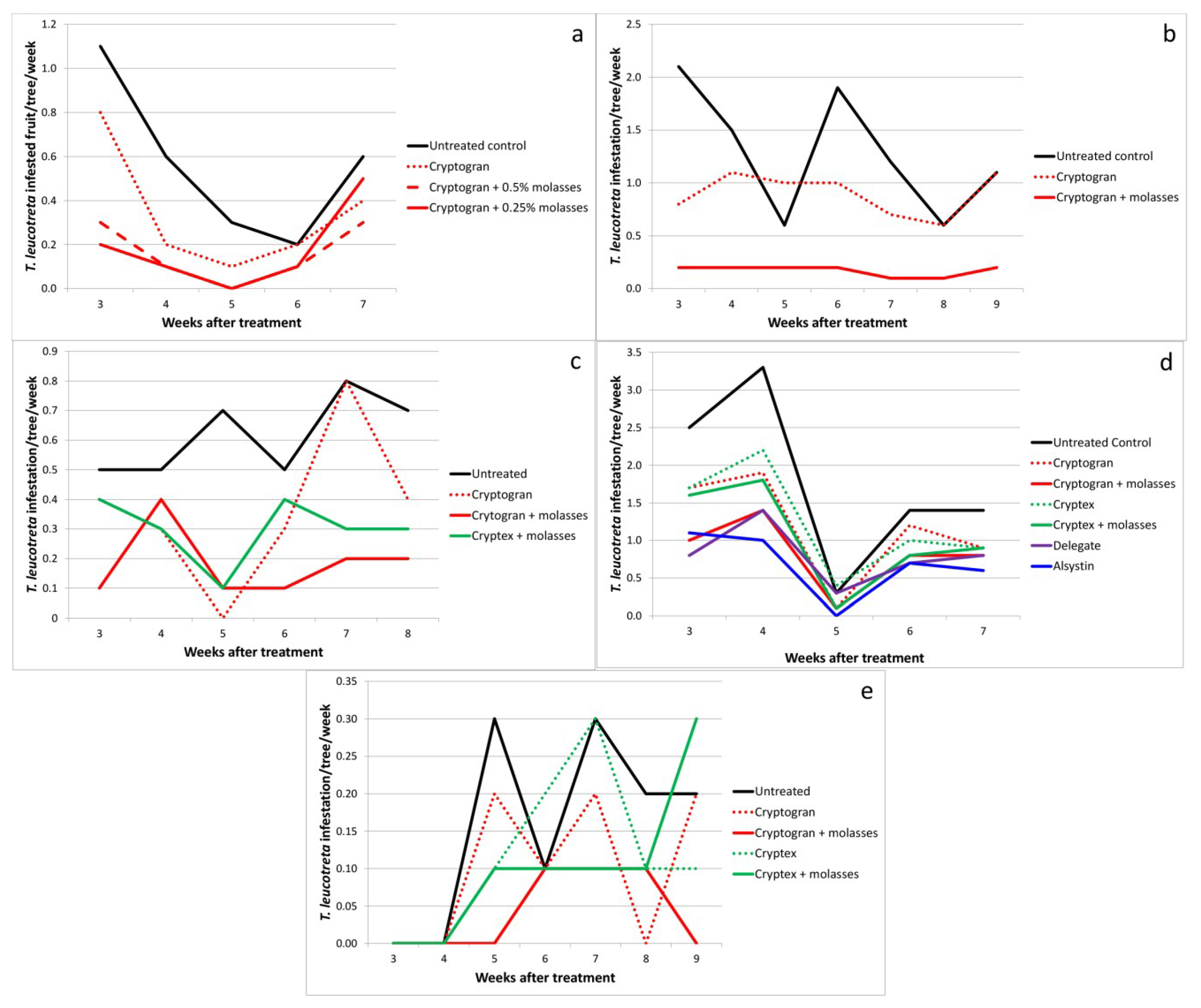
| Bernol 2005 | Control | - | 5 | 0.56a | 0.16 | - |
| Cryptogran | 5.4 × 1013 (10 mL) | - | 0.34ab | 0.12 | 39.3 | |
| Cryptogran + molasses | 5.4 × 1013 (10 mL) + 0.5 | - | 0.16b | 0.06 | 71.4 | |
| Cryptogran + molasses | 5.4 × 1013 (10 mL) + 0.25 | - | 0.18b | 0.09 | 67.9 | |
| Dunbrody 2006 | Control | - | 9 | 0.21a | 0.05 | - |
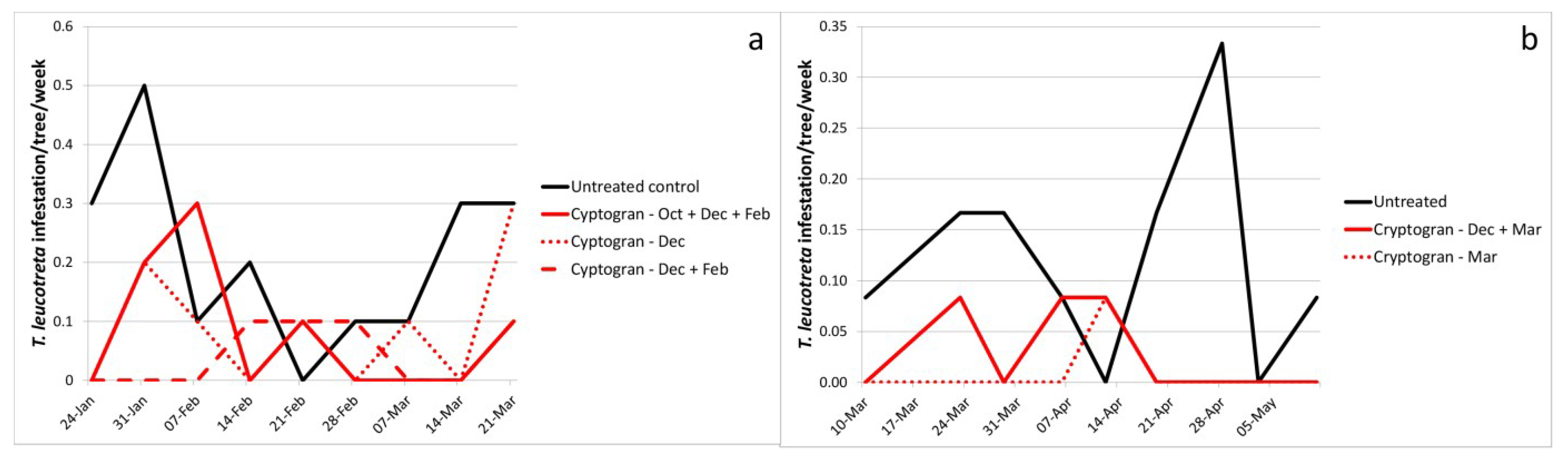
| Cryptogran October, December, February | 6.4 × 1013 (10 mL) | - | 0.08b | 0.04 | 63.2 | |
| Cryptogran December | 6.4 × 1013 (10 mL) | - | 0.09b | 0.03 | 57.9 | |
| Cryptogran December, February | 6.4 × 1013 (10 mL) | - | 0.04b | 0.02 | 78.9 | |
| Lone Tree 2007 | Control | - | 7 | 1.29a | 0.22 | - |
| Cryptogran | 6.1 × 1013 (10 mL) | - | 0.90a | 0.08 | 30.0 | |
| Cryptogran + molasses | 6.1 × 1013 (10 mL) | - | 0.17b | 0.02 | 86.7 | |
| Lone Tree 2008 | Control | - | 6 | 0.62a | 0.05 | - |
| Cryptogran | 4.2 × 1013 (10 mL) | - | 0.37b | 0.10 | 40.5 | |
| Cryptogran + molasses | 4.2 × 1013 (10 mL) | - | 0.18c | 0.05 | 70.3 | |
| Cryptex + molasses | 8.0 × 1012 (3.3 mL) | - | 0.30bc | 0.04 | 51.3 | |
| Welegelegen 2009 | Control | 9 | 0.12a | 0.03 | - | |
| Cryptogran December, March | 2.8 × 1013 (10 mL) | - | 0.03b | 0.01 | 76.9 | |
| Cryptogran March | 2.8 × 1013 (10 mL) | - | 0.01b | 0.01 | 92.3 | |
| Far Away 2010 | Control | - | 5 | 1.78a | 0.23 | - |
| Cryptogran | 3.0 × 1013 (10 mL) | - | 1.16a | 0.14 | 34.8 | |
| Cryptogran + molasses | 3.0 × 1013 (10 mL) | - | 0.82b | 0.09 | 53.9 | |
| Cryptex | 3.9 × 1012 (3.3 mL) | - | 1.24a | 0.14 | 30.3 | |
| Cryptex + molasses | 3.9 × 1012 (3.3 mL) | - | 1.04a | 0.14 | 41.6 | |
| Delegate | 20 g | - | 0.80b | 0.08 | 55.0 | |
| Alsystin | 20 mL | - | 0.68b | 0.09 | 61.8 | |
| Bernol 2011 | Control | - | 7 | 0.16a | 0.05 | - |
| Cryptogran | 6.7 × 1013 (10 mL) | - | 0.10a | 0.04 | 36.4 | |
| Cryptogran + molasses | 6.7 × 1013 (10 mL) + 0.25 | - | 0.04b | 0.02 | 72.7 | |
| Cryptex | 8.8 × 1012 (3.3 mL) | - | 0.11a | 0.04 | 27.3 | |
| Cryptex + molasses | 8.8 × 1012 (3.3 mL) + 0.5 | - | 0.10a | 0.04 | 36.4 | |
| Far Away 2013 | Control | - | 6 | 0.23a | 0.05 | - |
| Cryptogran + molasses | 3.2 × 1013 (10 mL) | - | 0.10b | 0.04 | 57.1 | |
| Cryptex | 4.3 × 1012 (3.3 mL) | - | 0.15a | 0.04 | 35.7 | |
| Runner | 60 mL | - | 0.07b | 0.02 | 71.4 | |
| Delegate | 20 g | - | 0.07b | 0.03 | 71.4 | |
2.2. CrleGV Efficacy in Block vs. Single Tree Treatments
2.3. Effect of Molasses as an Adjuvant
2.4. Dose Rate
2.5. CrleGV Spray Programs
2.6. Comparison with Chemical Insecticides
3. Discussion
4. Material and Methods
4.1. Source of CrleGV
4.2. Trial Sites
| Years (Citrus season) in which trial was conducted | Farm name | Coordinates | Cultivar | Tree age (years) | Trees/ha | Trial layout |
|---|---|---|---|---|---|---|
| 2000/01 | Sun Orange | 33°28'06"S 25°39'00"E | Palmer Navel | 21 | 383 | STRB |
| 2001/02 | Vergenoeg | 33°45'45"S 24°48'59"E | Robyn Navel | 17 | 555 | STRB |
| 2002/03 | Moosrivier | 25°01'24"S 29°22'22"E | Robyn Navel | 15 | 340 | STRB |
| 2003/04 | Carden | 33°28'13"S 25°41'23"E | Palmer Navel | 11 | 555 | STRB SCB |
| 2004/05 | Bernol | 33°28'26"S 25°36'43"E | Washington Navel | 6 | 595 | SCB |
| 2004/05 | Bernol | 33°28'26"S 25°36'43"E | Palmer Navel | 7 | 555 | STRB |
| 2006/07 | Dunrody | 33°27'59"S 25°31'30"E | Lane Late Navel | 10 | 833 | SCB |
| 2007/08 | Lone Tree | 33°51'56"S 25°41'31"E | Palmer | 8 | 555 | STRB |
| 2008/09 | Lone Tree | 33°51'56"S 25°41'31"E | Palmer Navel | 9 | 555 | STRB |
| 2009/10 | Welgelegen | 25°27'51"S 31°53'02"E | Turkey Valencia | 8 | 555 | SCB |
| 2010/11 | Far Away | 33°29'07"S 25°40'34"E | Newhall | 3 | 555 | STRB |
| 2011/12 | Bernol | 33°28'26"S 25°36'43"E | Palmer Navel | 6 | 555 | STRB |
| 2012/13 | Far Away | 33°29'07"S 25°40'34"E | Witkrans Navel | 5 | 555 | STRB |
4.3. Trial Layout
4.4. Treatment Application
4.5. Trial Details
| Farm | Treatment application date | Type of CrleGV | Concentration for commercial products (mL/100 L Water) | OBs/ha | Concentration of molasses (%) 1 | Volume applied (mean L/tree) | Spray method |
|---|---|---|---|---|---|---|---|
| Sun Orange | 5 April 2001 | Unformulated | - | 1.22 × 1014 | - | 38.3 | Handguns |
| - | - | - | - | 1.01 × 1015 | - | - | - |
| Vergenoeg | 14 March 2002 | Unformulated | - | 9.52 × 1014 | - | 27.0 | Handguns |
| - | - | - | - | 1.24 × 1015 | - | 22.0 | - |
| - | - | - | - | 1.23 × 1015 | - | 15.0 | - |
| Moosrivier | 16 January 2003 | Unformulated | - | 1.60 × 1014 | 0.5 | 35.0 | Handguns |
| - | - | - | - | 8.00 × 1014 | - | - | - |
| Carden | 3 December 2003 | Cryptogran | 10 | 6.59 × 1013 | 0.5 | 20.1 (STRB)2 | Handguns |
| - | - | - | 10 | 5.20 × 1013 | 0.25 | 15.3 (SCB)3 | Oscillating tower |
| Bernol | 1 December 2004 | Cryptogran | 10 | 4.49 × 1013 | 0.5 | 15.1 | Oscillating tower |
| - | - | - | 8 | 3.59 × 1013 | - | - | - |
| - | - | - | 6 | 2.69 × 1013 | - | - | - |
| - | - | - | 4 | 1.80 × 1013 | - | - | - |
| - | - | - | 2 | 8.98 × 1012 | - | - | - |
| - | - | Cryptex | 2.25 | 4.04 × 1012 | 0.5 | - | - |
| Bernol | 22 March 2005 | Cryptogran | 10 | 5.41 × 1013 | - | 19.5 | Handguns |
| - | - | - | 10 | 5.41 × 1013 | 0.5 | - | - |
| - | - | - | 10 | 5.41 × 1013 | 0.25 | - | - |
| Dunbrody | 26 October + 5 December 2006 + 5 February 2007 | Cryptogran | 10 | 6.37 × 1013 | 0.25 | 15.3 | Oscillating tower |
| - | 5 December 2006 | - | 10 | 6.37 × 1013 | 0.25 | - | - |
| - | 5 December 2006 + 5 February 2007 | - | 10 | 6.37 × 1013 | 0.25 | - | - |
| Lone Tree | 10 December 2007 | Cryptogran | 10 | 6.08 × 1013 | - | 21.9 | Handguns |
| - | - | - | 10 | 6.08 × 1013 | 0.25 | - | - |
| Lone Tree | 10 December 2008 | Cryptogran | 10 | 4.25 × 1013 | - | 21.9 | Handguns |
| - | - | Cryptogran | 10 | 4.25 × 1013 | 0.25 | - | - |
| - | - | Cryptex | 3.3 | 8.02 × 1012 | 0.25 | - | - |
| Welgelegen | 8 December 2009 + 15 March 2010 | Cryptogran | 10 | 2.80 × 1013 | 0.25 | 10.1 | Oscillating tower |
| - | 16 March 2010 | Cryptogran | 10 | 2.80 × 1013 | 0.25 | - | - |
| Far Away | 7 December 2010 | Cryptogran | 10 | 2.97 × 1013 | - | 10.7 | Handguns |
| - | - | Cryptogran | 10 | - | 0.25 | - | - |
| - | - | Cryptex | 3.3 | 3.91 × 1012 | - | - | - |
| - | - | Cryptex | 3.3 | - | 0.5 | - | - |
| Bernol | 19 December 2011 | Cryptogran | 10 | 6.66 × 1013 | - | 24.0 | Handguns |
| - | - | Cryptogran | 10 | 0.25 | - | - | |
| - | - | Cryptex | 3.3 | 8.79 × 1012 | - | - | - |
| - | - | Cryptex | 3.3 | - | 0.5 | - | - |
| Far Away | 24 April 2013 | Cryptogran | 10 | 3.25 × 1013 | 0.25 | 11.7 | Handguns |
| - | - | Cryptex | 3.3 | 4.29 × 1012 | - | - | - |
4.6. Trial Evaluation
4.7. Data Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Newton, P.J. False codling moth Cryptophlebia leucotreta (Meyrick). In Citrus Pests in the Republic of South Africa; Bedford, E.C.G., van den Berg, M.A., de Villiers, E.A., Eds.; Dynamic Ad: Nelspruit, South Africa, 1998. [Google Scholar]
- Grout, T.G.; Moore, S.D. Citrus. In Insects of Cultivated Plants and Natural Pastures in Southern Africa; Prinsloo, G.L., Uys, G.M., Eds.; Entomological Society of Southern Africa: Pretoria, South Africa, 2015; pp. 447–501. [Google Scholar]
- Hepburn, G.A.; Bishop, H.J. The insect pests of citrus in South Africa. Union S. Afr. Departm. Agric. Bull. 1954, 41, 15–22. [Google Scholar]
- Moore, S.D. The Development and Evaluation of Cryptophlebia Leucotreta Granulovirus (CrleGV) as a Biological Control Agent for the Management of False Codling Moth, Cryptophlebia Leucotreta, on Citrus. Ph.D. Thesis, Rhodes University, Grahamstown, South Africa, 12 March 2002; p. 311. [Google Scholar]
- Moore, S.D.; Hattingh, V. A review of current Pre-harvest Control Options for False Codling Moth in Citrus in Southern Africa. S. Afr. Fruit J. 2012, 11, 82–85. [Google Scholar]
- Moore, S.D.; Kirkman, W.; Stephen, P. Cryptogran: A virus for the biological control of false codling moth. S. Afr. Fruit J. 2004, 3, 35–39. [Google Scholar]
- Moore, S.D.; Hendry, D.A.; Richards, G.I. Virulence of a South African isolate of the Cryptophlebia leucotreta granulovirus (CrleGV-SA) to Thaumatotibia leucotreta neonate larvae. BioControl 2011, 56, 341–352. [Google Scholar] [CrossRef]
- Angelini, A.; Amargier, A.; Vandamme, P.; Duthoit, J.L. Une virose á granules chez le lepidoptére Argyroploce leucotreta. Coton Fibres Trop. 1965, 20, 277–282. [Google Scholar]
- Komai, F. A taxonomic review of the genus Grapholita and allied genera (Lepidoptera, Tortricidae) in the Palaerctic region. Entomol. Scand. 1999, 55, 1–219. [Google Scholar]
- Angelini, A.; Le Rumeur, C. Sur une maladie à virus d’ Argyroploce leucotreta découverte en Côte d’Ivoire. Coton Fibres Trop. 1962, 17, 291–296. [Google Scholar]
- Mück, O. Biologie, verhalten und wirtshcaftliche bedeutaung von parasiten schädlicher Lepidopteren auf den Kapverden. Neue Entomol. Nachrichten 1985, 18, 168. [Google Scholar]
- Whitlock, V.H. Long virus-like rods associated with a granulosis virus of the false codling moth, Cryptophlebia leucotreta (Meyr.). Phytophylactica 1980, 12, 181–186. [Google Scholar]
- Jehle, A.J.; Backhaus, H.; Fritsch, E.; Huber, J. Physical map of the Cryptophlebia leucotreta granulosis virus genome and its relationship to the genome of Cydia pomonella granulosis virus. J. Gen. Virol. 1992, 73, 1621–1626. [Google Scholar] [CrossRef] [PubMed]
- Fritsch, E.; Huber, J. The granulosis virus of the false codling moth, Cryptophlebia leucotreta (Meyr). In Proceedings of the 4th International Colloquium for Invertebrate Pathology, Veldhoven, The Netherlands, 18–22 August 1986; p. 112.
- Fritsch, E. Das Granulosevirus des Falschen Apfelwicklers, Cryptophlebia Leucotreta (Meyrick). Ph.D. Thesis, Technische Universität, Darmstadt, Germany, 1 December 1989. [Google Scholar]
- Jehle, J.A.; Backhaus, H. The granulin gene region of Cryptophlebia leucotreta granulosis virus: Sequence analysis and phylogenetic considerations. J. Gen. Virol. 1994, 75, 3667–3671. [Google Scholar] [CrossRef] [PubMed]
- Jehle, J.A.; Backhaus, H. Genome organisation of the DNA-binding protein gene region of Cryptophlebia leucotreta granulosis virus is closely related to that of nuclear polyhedrosis viruses. J. Gen. Virol. 1994, 75, 1815–1820. [Google Scholar] [CrossRef] [PubMed]
- Jehle, A.J.; Fritsch, E.; Backhaus, H.; Huber, J. Risk assessment on genetic engineering of baculoviruses: In vivo integration of host DNA into the genome of granulosis viruses. In Proceedings of the 2nd International Symposium on the Biosafety Results of Field Tests of Genetically Modified Plants and Microorganisms, Goslar, Germany, 11–14 May 1992; Casper, R., Landsmann, J., Eds.; 1992; pp. 250–256. [Google Scholar]
- Fritsch, E.; Huber, J.; Backhaus, H. CpGV as a tool in the risk assessment of genetically engineered baculoviruses. In Proceedings of the 5th International Colloquium on Invertebrate Pathology and Microbial Control, Adelaide, Australia, 20–24 August 1990; pp. 439–443.
- Lange, M.; Jehle, J.A. The Genome of the Cryptophlebia leucotreta granulovirus. Virology 2003, 317, 220–236. [Google Scholar] [CrossRef] [PubMed]
- Reiser, M.; Gröner, A.; Sander, E. Cryptophlebia leucotreta (Lep.: Tortricidae)—A promising alternate host for mass production of the Cydia pomonella granulosis virus (CpGV) for biological pest control. J. Plant Dis. Protect. 1993, 100, 586–598. [Google Scholar]
- Cross, J.V.; Solomon, M.G.; Chandler, D.; Jarrett, P.; Richardson, P.N.; Winstanley, D.; Bathon, H.; Huber, J.; Keller, B.; Langenbruch, G.A.; et al. Biocontrol of pests of apples and pears in northern and central Europe: 1. Microbial agents and nematodes. Biocontrol Sci. Technol. 1999, 9, 125–149. [Google Scholar] [CrossRef]
- Chambers, C. Production of Cydia Pomonella Granulovirus (CpGV) in a Heterologous Host, Thaumatotibia Leucotreta (Meyrick) (False Codling Moth). Ph.D. Thesis, Rhodes University, Grahamstown, South Africa, 2015. [Google Scholar]
- Dhladhla, B.I. Enumeration of Insect Viruses Using Microscopic and Molecular Analyses; South African Isolate of Cryptophlebia Leucotreta Granulovirus As a Case Study. MSc Thesis, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa, 2012. [Google Scholar]
- Falcon, L.A.; Kane, W.R.; Bethell, R.S. Preliminary evaluation of a granulosis virus for control of the codling moth. J. Econ. Entomol. 1968, 61, 1208–1213. [Google Scholar] [CrossRef]
- Lacey, L.A.; Thomson, D.; Vincent, C.; Arthurs, S.P. Codling moth granulovirus: A comprehensive review. Biocontrol Sci. Technol. 2008, 18, 639–663. [Google Scholar] [CrossRef]
- Fritsch, E. Biologische bekämpfung des falschen apfelwicklers, Cryptophlebia leucotreta (Meyrick) (Lep., Tortricidae), mit granuloseviren. Mitt. Dtsch. Ges. Allg. Angew. Ent. 1988, 6, 280–283. [Google Scholar]
- Singh, S.; Moore, S.D.; Spillings, B.; Hendry, D.A. South African Isolate of Cryptophlebia leucotreta granulovirus. J. Invertebr. Pathol. 2003, 83, 249–252. [Google Scholar] [CrossRef] [PubMed]
- Ludewig, M.H. The Establishment of a Virus Free Laboratory Colony of Cryptophlebia Leucotreta (False Codling Moth) and Characterisation of Cryptophlebia Leucotreta Granulovirus (CrleGV) Genes. MSc Thesis, Rhodes University, Grahamstown, South Africa, 2003. [Google Scholar]
- Opoku-Debrah, J.K.; Hill, M.P.; Knox, C.; Moore, S.D. Overcrowding of false codling moth, Thaumatotibia leucotreta (Meyrick) leads to the isolation of five new Cryptophlebia leucotreta granulovirus (CrleGV-SA) isolates. J. Invertebr. Pathol. 2013, 112, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.D.; Richards, G.I.; Chambers, C.; Hendry, D. An improved larval diet for commercial mass rearing of the false codling moth, Thaumatotibia leucotreta (Meyrick) (Lepidoptera: Tortricidae). Afr. Entomol. 2014, 22, 216–219. [Google Scholar] [CrossRef]
- Tanada, Y.; Kaya, H.K. Insect Pathology; Academic Press, Inc.: San Diego, CA, USA, 1993. [Google Scholar]
- Kirkman, W. Understanding and Improving the Residual Efficacy of the Cryptophlebia Leucotreta Granulovirus (CRYPTOGRAN). MSc Thesis, Rhodes University, Grahamstown, EC, South Africa, 15 December 2007. [Google Scholar]
- Kessler, P.; Zingg, D. New baculovirus products offer solutions for the biological control of Cydia pomonella and Cryptophlebia leucotreta. In Proceedings of the 23rd International Congress of Entomology, Durban, South Africa, 6–12 July 2008.
- Grove, T.; de Beer, M.S.; Joubert, P.H. Developing a Systems Approach for Thaumatotibia leucotreta (Lepidoptera: Tortricidae) on “Hass” Avocado in South Africa. J. Econ. Entomol. 2010, 103, 1112–1128. [Google Scholar]
- Goble, T. The Genetic Characterisation and Biological Activity of the South African Cryptophlebia Leucotreta Granulovirus (CrleGV-SA) in two Biopesticides, Cryptogran and Cryptex. BSc Honours Project. Rhodes University: Grahamstown, South Africa, 2007. [Google Scholar]
- Opoku-Debrah, J.K.; Moore, S.D.; Hill, M.P.; Knox, C. Characterisation of novel CrleGV isolates for false codling moth control-lessons learnt from codling moth resistance to CpGV. In Proceedings of the Insect Pathogens and Entomoparasitic Nematodes IOBC-WPRS Bulletin, Zagreb, Croatia, 16–20 June 2013; Volume 90, pp. 155–159.
- Pereira-da-Conceicoa, L.L.; Hill, M.P.; Moore, S.D. Development of a droplet-dose bioassay laboratory technique for Thaumatotibia leucotreta (Lepidoptera: Tortricidae). Afr. Entomol. 2012, 20, 187–190. [Google Scholar] [CrossRef]
- Fritsch, E.; Undorf-Spahn, K.; Kienzle, J.; Zebitz, C.P.W.; Huber, J. Codling moth granulovirus: First indications of variations in the susceptibility of local codling moth populations. Nachrichtenbl. Deut. Pflanzenschutzd. 2005, 57, 29–34. [Google Scholar]
- Sauphanor, B.; Berling, M.; Toubon, J.F.; Reyes, M.; Delnatte, J.; Allemoz, P. Carpocapse des pommes. Cas de resistance au virus de la granulose en vergers biologique. Phytoma–La Defense des Vegetaux 2006, 590, 24–27. [Google Scholar]
- Eberle, K.E.; Jehle, J.A. Field resistance of codling moth against Cydia pomonella granulovirus (CpGV) is autosomal and incompletely dominant inherited. J. Invertebr. Pathol. 2006, 93, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Berling, M.; Blanchere-Lopez, C.; Soubabere, O.; Lery, X.; Bonhomme, A.; Sauphanor, B.; Lopez-Ferber, M. Cydia pomonella granulovirus genotypes overcome virus resistance in codling moth and improve virus efficacy by selection against resistant hosts. Appl. Environ. Microb. 2009, 75, 925–930. [Google Scholar] [CrossRef]
- Stotter, R.L.; Samways, M.J.; Hattingh, V. Preparing the way for sterile insect release: Determination of false codling moth distribution across a landscape mosaic. Crop Prot. 2014, 60, 1–4. [Google Scholar] [CrossRef]
- Love, C.N.; Hill, M.P.; Moore, S.D. Thaumatotibia leucotreta and the Navel orange: Ovipositional preferences and host susceptibility. J. Appl. Entomol. 2014, 138, 600–611. [Google Scholar] [CrossRef]
- Hofmeyr, J.H.; Pringle, K.L. Resistance of false codling moth, Cryptophlebia leucotreta (Meyrick) (Lepidoptera: Tortricidae), to the chitin synthesis inhibitor, triflumuron. Afr. Entomol. 1998, 6, 373–375. [Google Scholar]
- Citrus Growers’ Association. Key Industry Statistics; Citrus Growers’ Association: Durban, South Africa, 2013. [Google Scholar]
- Grout, T.G.; Scholtz, K.C.; Tate, B.A. Citrus Research International, Nelspruit, South Africa. Unpublished work, 2003. [Google Scholar]
- Hattingh, V.; Ware, A.B.; Grout, T.G. The development of a non-target evaluation system for Southern African citrus. In Proceedings of the International Society of Citriculture, Foz do Iguassu, Brazil, 20–26 August 2000; pp. 795–797.
- Prinsloo, G.L. An illustrated guide to the parasitic wasps associated with citrus pests in the Republic of South Africa. Sci. Bull. Departm. Agric. Repub. S. Afr. 1984, 402, 1–119. [Google Scholar]
- Newton, P.J.; Odendaal, W.J. Commercial inundative releases of Trichogrammatoidea cryptophlebiae (Hym: Trichogrammatidae) against Cryptophlebia leucotreta (Lepidoptera: Tortricidae) in Citrus. Entomophaga 1990, 35, 545–556. [Google Scholar] [CrossRef]
- Moore, S.D.; Hattingh, V. Augmentation of natural enemies for control of citrus pests in South Africa: A guide for growers. S. Afr. Fruit J. 2004, 3, 45–47, 51, 53. [Google Scholar]
- Arthurs, S.; Lacey, L.A. Field evaluation of commercial formulations of the codling moth granulovirus: Persistence of activity and success of seasonal applications against natural infestations of codling moth in Pacific Northwest apple orchards. Biol. Control 2004, 31, 388–397. [Google Scholar] [CrossRef]
- Osenberg, C.W.; Sarnelle, O.; Goldberg, D.E. Meta-analysis in ecology: Concepts, statistics, and applications. Ecology 1999, 80, 1103–1104. [Google Scholar] [CrossRef]
- Huber, J.; Dickler, E. Codling moth granulosis virus: Its efficiency in the field in comparison with organophosphorous insecticides. J. Econ. Entomol. 1977, 70, 557–561. [Google Scholar] [CrossRef]
- Jaques, R.P.; Hardman, J.M.; Laing, J.E.; Smith, R.F.; Bent, E. Orchard trials in Canada on control of Cydia pomonella (Lep.: Tortricidae) by granulosis virus. Entomophaga 1994, 39, 281–292. [Google Scholar] [CrossRef]
- Sheppard, R.F.; Stairs, G.R. Effects of dissemination of low dosage levels of a granulosis virus in populations of the codling moth. J. Econ. Entomol. 1976, 69, 583–586. [Google Scholar] [CrossRef]
- Stará, J.; Kocourek, F. Evaluation of efficiency of Cydia pomonella granulovirus (CpGV) to control the codling moth (Cydia pomonella L., Lep.: Tortricidae) in field trials. Plant Protect. Sci. 2003, 39, 117–125. [Google Scholar]
- Arthurs, S.; Lacey, L.A.; Fritts, J., Jr. Optimizing the use of the Codling Moth Granulovirus: Effects of application rate and spraying frequency on control of Codling Moth larvae in Pacific Northwest apple orchards. J. Econ. Entomol. 2005, 98, 1459–1468. [Google Scholar] [CrossRef] [PubMed]
- Glen, D.M.; Clark, J. Death of Cydia pomonella larvae and damage to apple fruit, after field application of codling moth granulosis virus. Entomol. Exp. Appl. 1985, 38, 93–96. [Google Scholar] [CrossRef]
- Huber, J. Field persistence of the codling moth granulosis virus Laspeyresia pomonella. IOBC/WPRS Bull. 1980, 3, 58–59. [Google Scholar]
- Jaques, R.P.; Laing, J.W.; Laing, D.R.; Yu, D.S.K. Effectiveness and persistence of the granulosis virus of the codling moth Cydia pomonella (L.) (Lepidoptera: Olethreutidae) on apple. Can. Entomol. 1987, 119, 1063–1067. [Google Scholar] [CrossRef]
- Jaques, R.P. Effectiveness of the granulosis virus of the codling moth in orchard trials in Canada. In Proceedings of the 5th International Colloquium on Invertebrate Pathology and Microbial Control, Adelaide, Australia, 20–24 August 1990; pp. 428–430.
- Charmillot, P.J.; Pasquier, D.; Scalo, A. Le virus de la granulose du carpocapse Cydia pomonella: 2. Efficacite´ en microparcelles, rémanence et rôle des adjuvants. Rev. Suisse Viticul. Arboricul. Horticul. 1998, 30, 61–64. [Google Scholar]
- Pasquier, D.; Charmillot, P.J. Le virus de la granulose du carpocapse Cydia pomonella. 3. Essai pratique de longue durée. Rev. Suisse Vitic. Arboric. Hortic. 1998, 30, 185–187. [Google Scholar]
- Lacey, L.A.; Arthurs, S.; Knight, A.; Becker, K.; Headrick, H. Efficacy of codling moth granulovirus: Effect of adjuvants on persistence of activity and comparison with other larvicides in a Pacific Northwest apple orchard. J. Entomol. Sci. 2004, 39, 500–513. [Google Scholar]
- Glen, D.M.; Payne, C.C. Production and field evaluation of codling moth granulosis virus for control fo Cydia pomonella in the United Kingdom. Ann. Appl. Biol. 1984, 104, 87–98. [Google Scholar] [CrossRef]
- Keller, S. Microbiological control of the codling moth (Laspeyresia pomonella (L.)) (=Carpocapsa pomonella) with specific granulosis virus. Z. Ang. Entomol. 1973, 73, 137–181. [Google Scholar] [CrossRef]
- Dickler, E.; Huber, J. Il virus della granulosi della carpocapsa; situazione attuale della ricerce possibilita di una sua introduzione nella difensa dei frutteti. Difesa Delle Piante 1986, 9, 297–304. [Google Scholar]
- Fritsch, E.; Huber, J. Comparative field persistence of granulosis viruses under tropical and European conditions. IOBC WPRS Bull. 1989, 12, 84–87. [Google Scholar]
- Mwanza, P. Determination of the Effects of Sunlight and UV Irradiation on the Structure, Viability and Reapplication Frequency of the Biopesticide Cryptophlebia Leucotreta Granulovirus in the Protection Against False Codling Moth Infestation of Citrus Crops. MSc Thesis, Nelson Mandela Metropolitan University, Port Elizabeth, South Africa, 9 February 2015; p. 148. [Google Scholar]
- Timm, A.E.; Geertsema, H.; Warnich, L. Population genetic structure of economically important Tortricidae (Lepidoptera) in South Africa: A comparative analysis. Bull. Entomol. Res. 2010, 100, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Briese, D.T.; Mende, H.A. Differences in susceptibility to a granulosis virus between field populations of the potato tuber moth, Phthorimaea operculella (Zeller) (Lepidoptera: Gelechiidae). Bull. Entomol. Res. 1981, 71, 11. [Google Scholar] [CrossRef]
- Briese, D.T. Genetic basis for resistance to a granulosis virus in the potato tuber moth, Phthorimaea operculella. J. Invertebr. Pathol. 1982, 39, 215–218. [Google Scholar]
- Fuxa, J.R. Insect resistance to viruses. In Parasites and Pathogens of Insects; Volume II, Beckage, N., Thomson, S., Federici, B.A., Eds.; Academic Press Inc.: San Diego, CA, USA, 1993; pp. 197–209. [Google Scholar]
- Abot, A.R.; Moscardi, F.; Fuxa, J.R.; Sosa-Gomez, D.R.; Ritcher, A.R. Development of resistance by Anticarsia gemmatalis from Brazil and the United States to a nuclear polyhedrosis virus under laboratory selection pressure. Biol. Control 1996, 7, 126–130. [Google Scholar] [CrossRef]
- Gebhardt, M.M.; Eberle, K.E.; Radtke, P.; Jehle, J.A. Baculovirus resistance in codling moth is virus isolate-dependent and the consequence of a mutation in viral gene pe38. Proc. Natl. Acad. Sci. USA 2014, 111, 15711–15716. [Google Scholar] [CrossRef] [PubMed]
- Eberle, K.E. Novel Isolates of Cydia Pomonella Granulovirus (CpGV): Deciphering the Molecular Mechanism for Overcoming CpGV Resistance in Codling Moth (Cydia pomonella). Ph.D. Thesis, Johannes Gutenberg University, Mainz, Germany, 23 September 2010; p. 185. [Google Scholar]
- Eberle, K.E.; Sayed, S.; Rezapanah, M.; Shojai-Estabragh, S.; Jehle, J.A. Diversity and evolution of the Cydia pomonella granulovirus. J. Gen. Virol. 2009, 90, 662–671. [Google Scholar] [CrossRef] [PubMed]
- Gund, N.A.; Wagner, A.; Timm, A.E.; Schulze-Bopp, S.; Jehle, J.A.; Johannesen, J.; Reineke, A. Genetic analysis of Cydia pomonella (Lepidoptera: Tortricidae) populations with different levels of sensitivity towards the Cydia pomonella granulovirus (CpGV). Genetica 2012, 140, 235–247. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, E.M.; Guthrie, K.P.; Shapiro, M. Optical Brighteners Provide Baculovirus Activity Enhancement and UV Radiation Protection. Biol. Control 1996, 7, 71–74. [Google Scholar] [CrossRef]
- Farrar, R.R.; Shapiro, M.; Javaid, I. Photostabilized titanium dioxide and a fluorescent brightener as adjuvants for a nucleopolyhedrovirus. BioControl 2003, 48, 543–560. [Google Scholar] [CrossRef]
- Asano, S. Ultraviolet protection of granulovirus product using iron oxide. Appl. Entomol. Zool. 2005, 40, 359–364. [Google Scholar] [CrossRef]
- Arthurs, S.P.; Lacey, L.A.; Behle, R.W. Evaluation of spray-dried lignin-based formulations and adjuvants as solar protectants for the granulovirus of the codling moth, Cydia pomonella (L). J. Invert. Pathol. 2006, 93, 88–95. [Google Scholar] [CrossRef]
- Wu, Z.-W.; Fan, J.-B.; Yu, H.; Wang, D.; Zhang, Y.-L. Ultraviolet protection of the Cydia pomonella granulovirus using zinc oxide and titanium dioxide. Biocontrol Sci. Technol. 2014, 25, 97–107. [Google Scholar] [CrossRef]
- Arthurs, S.P.; Lacey, L.A.; Behle, R.W. Evaluation of lignins and particle films as solar protectants for the granulovirus of the codling moth, Cydia pomonella. Biocontrol Sci. Technol. 2008, 18, 829–839. [Google Scholar] [CrossRef]
- Grout, T.G. Use of plant protection products. In Citrus Research International Integrated Production Guidelines; Volume 3, Integrated Pest and Disease Management; Grout, T.G., Ed.; Citrus Research International: Nelspruit, South Africa, 2014. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moore, S.D.; Kirkman, W.; Richards, G.I.; Stephen, P.R. The Cryptophlebia Leucotreta Granulovirus—10 Years of Commercial Field Use. Viruses 2015, 7, 1284-1312. https://doi.org/10.3390/v7031284
Moore SD, Kirkman W, Richards GI, Stephen PR. The Cryptophlebia Leucotreta Granulovirus—10 Years of Commercial Field Use. Viruses. 2015; 7(3):1284-1312. https://doi.org/10.3390/v7031284
Chicago/Turabian StyleMoore, Sean D., Wayne Kirkman, Garth I. Richards, and Peter R. Stephen. 2015. "The Cryptophlebia Leucotreta Granulovirus—10 Years of Commercial Field Use" Viruses 7, no. 3: 1284-1312. https://doi.org/10.3390/v7031284
APA StyleMoore, S. D., Kirkman, W., Richards, G. I., & Stephen, P. R. (2015). The Cryptophlebia Leucotreta Granulovirus—10 Years of Commercial Field Use. Viruses, 7(3), 1284-1312. https://doi.org/10.3390/v7031284