Epidemiological Research on Hand, Foot, and Mouth Disease in Mainland China
Abstract
:1. Introduction
2. Basic Epidemiology
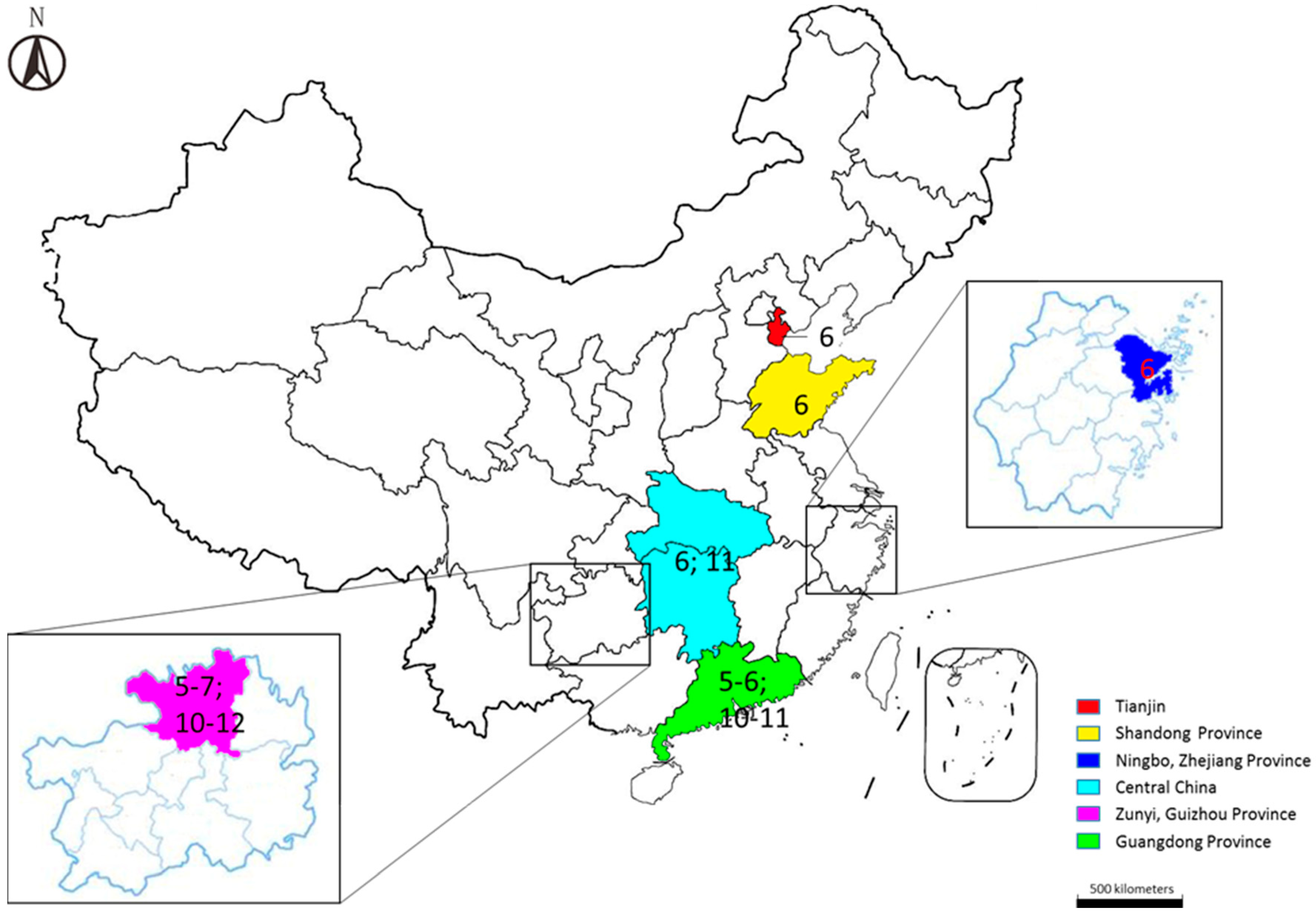
3. Etiology and Molecular Epidemiology
| Pathogen | Region | Year | Genotype |
|---|---|---|---|
| Enterovirus 71 | Jining, Shandong Province | 2010 | C1a |
| Central China | 2011–2012 | ||
| 54.8% (68/124) C1b | |||
| Shijiazhuang, Hebei Province | 2010–2012 | C4a | |
| Coxsackievirus A16 | Linyi, Shandong Province | 2009–2011 | B1a |
| Central China | 2011–2012 | ||
| 68.75% (55/80) B1b | |||
| Shijiazhuang, Hebei Province | 2010–2012 | B1 | |
| Coxsackievirus A6 * | Guangdong Province | 2013 | – |
| Tianjin | 2008–2013 | – | |
| Fujian | 2011–2013 | – | |
| Echovirus 30 | Shandong Province | 2010–2011 | D2 |
| Echovirus 25 | Shandong Province | 2010–2011 | D6 |
| Echovirus 6 | Shandong Province | 2010–2011 | C6 |
| Coxsackievirus B1 | Shandong Province | 2010–2011 | D6 |
| Coxsackievirus B3 | Shijiazhuang, Hebei Province | 2010–2012 | E |
| Coxsackievirus B4 | Shandong Province | 2010–2011 | D2 |
| Coxsackievirus B10 | Shijiazhuang, Hebei Province | 2010–2012 | C |
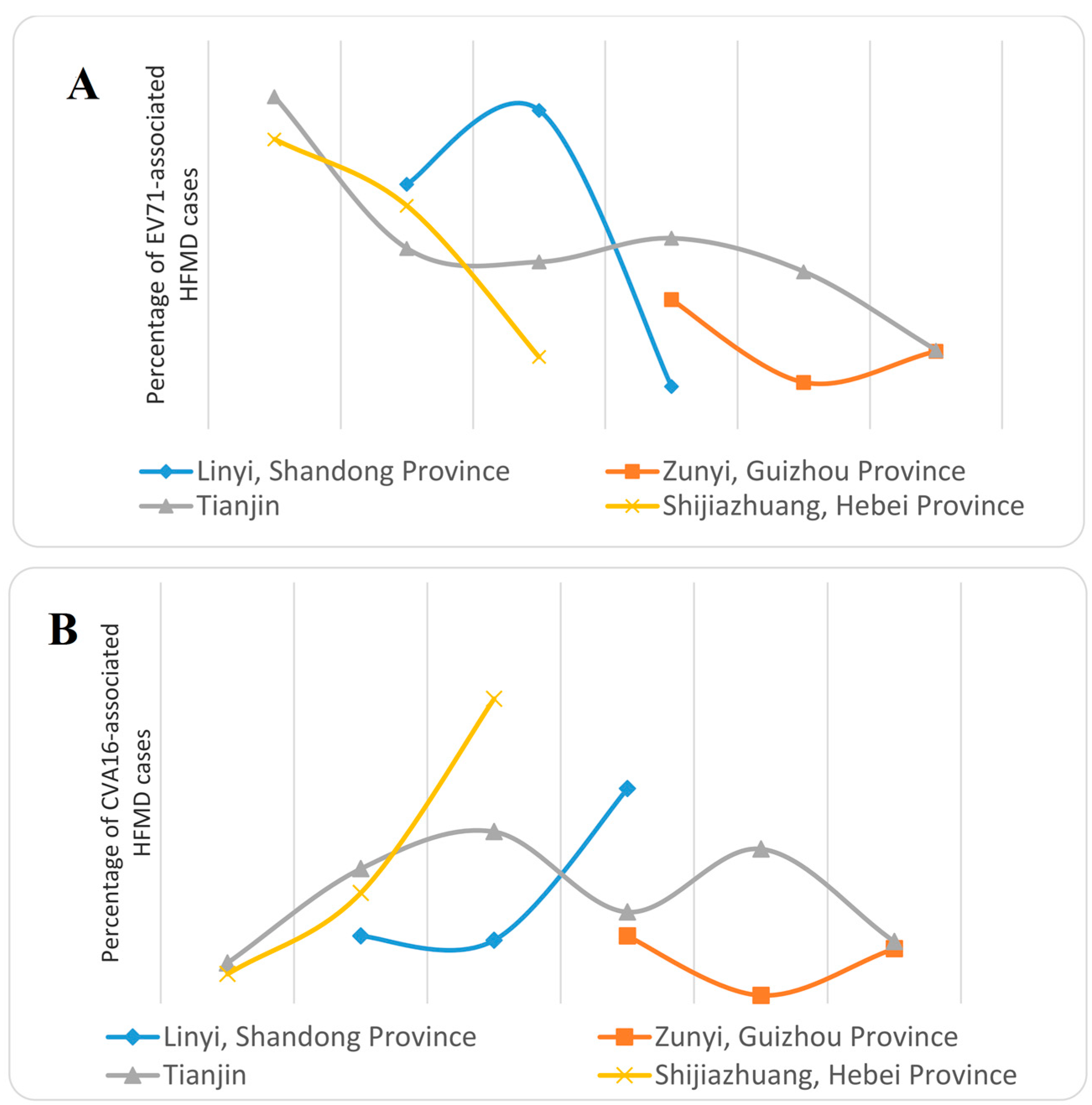
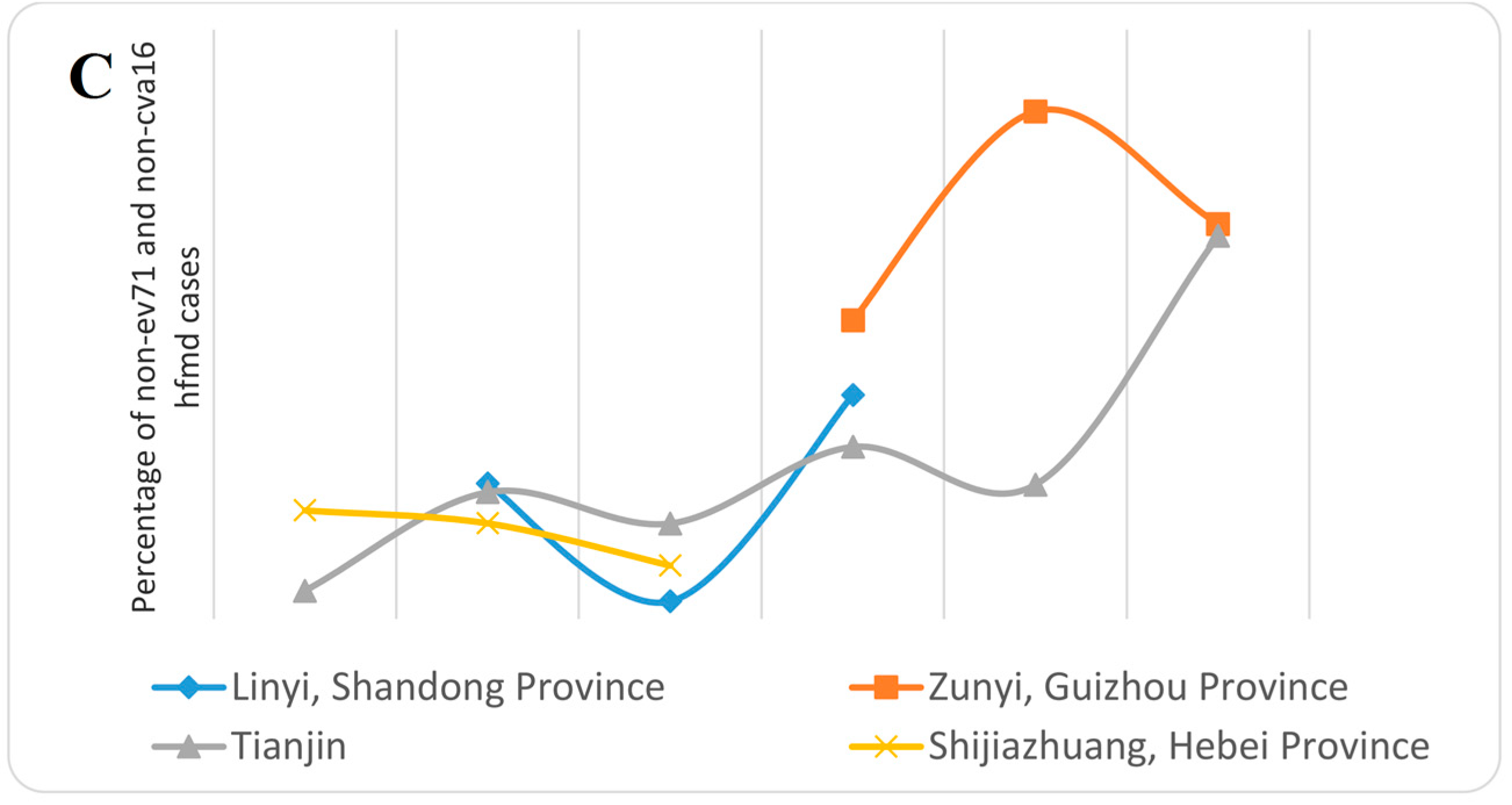
4. Influencing Factors
5. Detection and Surveillance
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Chan, L.G.; Parashar, U.D.; Lye, M.S.; Ong, F.G.; Zaki, S.R.; Alexander, J.P.; Ho, K.K.; Han, L.L.; Pallansch, M.A.; Suleiman, A.B.; et al. Deaths of children during an outbreak of hand, foot, and mouth disease in sarawak, malaysia: Clinical and pathological characteristics of the disease. For the Outbreak Study Group. Clin. Infect. Dis. 2000, 31, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Seiff, A. Cambodia unravels cause of mystery illness. Lancet 2012, 380. [Google Scholar] [CrossRef]
- Ma, E.; Chan, K.C.; Cheng, P.; Wong, C.; Chuang, S.K. The enterovirus 71 epidemic in 2008—Public health implications for Hong Kong. Int. J. Infect. Dis. 2010, 14, e775–e780. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, Z.; Yang, W.; Ren, J.; Tan, X.; Wang, Y.; Mao, N.; Xu, S.; Zhu, S.; Cui, A.; et al. An emerging recombinant human enterovirus 71 responsible for the 2008 outbreak of hand foot and mouth disease in Fuyang city of China. Virol. J. 2010, 7. [Google Scholar] [CrossRef] [PubMed]
- Tu, P.V.; Thao, N.T.; Perera, D.; Huu, T.K.; Tien, N.T.; Thuong, T.C.; How, O.M.; Cardosa, M.J.; McMinn, P.C. Epidemiologic and virologic investigation of hand, foot, and mouth disease, Southern Vietnam, 2005. Emerg. Infect. Dis. 2007, 13, 1733–1741. [Google Scholar] [PubMed]
- Chan, K.P.; Goh, K.T.; Chong, C.Y.; Teo, E.S.; Lau, G.; Ling, A.E. Epidemic hand, foot and mouth disease caused by human enterovirus 71, Singapore. Emerg. Infect. Dis. 2003, 9, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Ho, M.; Chen, E.R.; Hsu, K.H.; Twu, S.J.; Chen, K.T.; Tsai, S.F.; Wang, J.R.; Shih, S.R. An epidemic of enterovirus 71 infection in Taiwan. N. Engl. J. Med. 1999, 341, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Ang, L.W.; Tay, J.; Phoon, M.C.; Hsu, J.P.; Cutter, J.; James, L.; Goh, K.T.; Chow, V.T. Seroepidemiology of Coxsackievirus A6, Coxsackievirus A16, and Enterovirus 71 infections among children and adolescents in Singapore, 2008–2010. PLoS ONE 2015, 10, e0127999. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Park, K.; Shim, A.; Kwon, B.E.; Ahn, J.H.; Choi, Y.J.; Kim, J.K.; Yeo, S.G.; Yoon, K.; Ko, H.J. Complete sequence analysis and antiviral screening of medicinal plants for human coxsackievirus A16 isolated in Korea. Osong Public Health Res. Perspect. 2015, 6, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Chan, P.K.; Ho, I.I.; Lai, W.M. Enterovirus infection among patients admitted to hospital in Hong Kong in 2010: Epidemiology, clinical characteristics, and importance of molecular diagnosis. J. Med. Virol. 2013, 85, 1811–1817. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.T.; Pham, H.V.; Hoang, C.Q.; Nguyen, T.M.; Nguyen, L.T.; Phan, H.C.; Phan, L.T.; Vu, L.N.; Tran-Minh, N.N. Epidemiological and clinical characteristics of children who died from hand, foot and mouth disease in Vietnam, 2011. BMC Infect. Dis. 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Puenpa, J.; Mauleekoonphairoj, J.; Linsuwanon, P.; Suwannakarn, K.; Chieochansin, T.; Korkong, S.; Theamboonlers, A.; Poovorawan, Y. Prevalence and characterization of enterovirus infections among pediatric patients with hand foot mouth disease, herpangina and influenza like illness in Thailand, 2012. PLoS ONE 2014, 9, e98888. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Zeng, H.; Zheng, H.; Yi, L.; Guo, X.; Liu, L.; Sun, L.; Tan, X.; Li, H.; Ke, C.; et al. Hand, foot and mouth disease in Guangdong, China, in 2013: New trends in the continuing epidemic. Clin. Microbiol. Infect. 2014, 20, O442–O445. [Google Scholar] [CrossRef] [PubMed]
- Xing, W.; Liao, Q.; Viboud, C.; Zhang, J.; Sun, J.; Wu, J.T.; Chang, Z.; Liu, F.; Fang, V.J.; Zheng, Y.; et al. Hand, foot, and mouth disease in China, 2008-2012: An epidemiological study. Lancet Infect. Dis. 2014, 14, 308–318. [Google Scholar] [CrossRef]
- Yan, X.F.; Gao, S.; Xia, J.F.; Ye, R.; Yu, H.; Long, J.E. Epidemic characteristics of hand, foot, and mouth disease in Shanghai from 2009 to 2010: Enterovirus 71 subgenotype C4 as the primary causative agent and a high incidence of mixed infections with coxsackievirus A16. Scand. J. Infect. Dis. 2012, 44, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Ooi, M.H.; Wong, S.C.; Lewthwaite, P.; Cardosa, M.J.; Solomon, T. Clinical features, diagnosis, and management of enterovirus 71. Lancet Neurol. 2010, 9, 1097–1105. [Google Scholar] [CrossRef]
- Chou, A.H.; Liu, C.C.; Chang, J.Y.; Jiang, R.; Hsieh, Y.C.; Tsao, A.; Wu, C.L.; Huang, J.L.; Fung, C.P.; Hsieh, S.M.; et al. Formalin-inactivated EV71 vaccine candidate induced cross-neutralizing antibody against subgenotypes B1, B4, B5 and C4A in adult volunteers. PLoS ONE 2013, 8, e79783. [Google Scholar] [CrossRef] [PubMed]
- Mao, Q.; Cheng, T.; Zhu, F.; Li, J.; Wang, Y.; Li, Y.; Gao, F.; Yang, L.; Yao, X.; Shao, J.; et al. The cross-neutralizing activity of enterovirus 71 subgenotype c4 vaccines in healthy Chinese infants and children. PLoS ONE 2013, 8, e79599. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Jiang, L.; Liang, Z.; Mao, Q.; Su, W.; Zhang, H.; Li, X.; Jin, J.; Xu, L.; Zhao, D.; et al. Evaluation of monovalent and bivalent vaccines against lethal Enterovirus 71 and Coxsackievirus A16 infection in newborn mice. Hum. Vaccines Immunother. 2014, 10, 2885–2895. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.L.; Mao, Q.Y.; Wang, Y.P.; Zhu, F.C.; Li, J.X.; Yao, X.; Gao, F.; Wu, X.; Xu, M.; Wang, J.Z. Progress on the research and development of inactivated EV71 whole-virus vaccines. Hum. Vaccines Immunother. 2013, 9, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Deng, T.; Huang, Y.; Yu, S.; Gu, J.; Huang, C.; Xiao, G.; Hao, Y. Spatial-temporal clusters and risk factors of hand, foot, and mouth disease at the district level in Guangdong Province, China. PLoS ONE 2013, 8, e56943. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Huang, B.; She, C.; Liu, Y.; Tong, H.; Wang, F.; Wu, K. An epidemic analysis of hand, foot, and mouth disease in Zunyi, China between 2012 and 2014. Saudi Med. J. 2015, 36, 593–598. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, W.; Zhang, Y.; Yan, L.; Wang, S.; Zhang, J.; Sun, J.; Chang, Z.; Wang, Z. Clinical features of severe cases of hand, foot and mouth disease with EV71 virus infection in China. Arch. Med. Sci. 2014, 10, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Wang, L.; Yan, Y.; Hong, R.; Ou, J.; Huang, W.; Zhu, H.; Chen, G. A cohort study on the characteristics of the recurrent epidemics on hand, foot and mouth disease, in Fujian province. Chin. J. Epidemiol. 2014, 35, 1109–1114. (In Chinese) [Google Scholar]
- Zhou, J.; Wu, Y.D.; Chen, X.L.; Song, C.; Yan, J. Epidemiological investigation of hand-foot-and-mouth disease in children and exposed population in Hangzhou city. J. Zhejiang Univ. Med. Sci. 2014, 43, 212–217. [Google Scholar]
- Zhu, Q.; Hao, Y.; Ma, J.; Yu, S.; Wang, Y. Surveillance of hand, foot, and mouth disease in Mainland China (2008–2009). Biomed. Environ. Sci. 2011, 24, 349–356. [Google Scholar] [PubMed]
- Zou, X.N.; Zhang, X.Z.; Wang, B.; Qiu, Y.T. Etiologic and epidemiologic analysis of hand, foot, and mouth disease in Guangzhou city: A review of 4753 cases. Braz. J. Infect. Dis. 2012, 16, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Ni, H.; Yi, B.; Yin, J.; Fang, T.; He, T.; Du, Y.; Wang, J.; Zhang, H.; Xie, L.; Ding, Y.; et al. Epidemiological and etiological characteristics of hand, foot, and mouth disease in Ningbo, China, 2008–2011. J. Clin. Virol. 2012, 54, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wu, S.; Xiong, Y.; Li, T.; Wen, Z.; Yan, M.; Qin, K.; Liu, Y.; Wu, J. Co-circulation and genomic recombination of coxsackievirus A16 and enterovirus 71 during a large outbreak of hand, foot, and mouth disease in Central China. PLoS ONE 2014, 9, e96051. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Li, L.; Zhang, B.; Jorba, J.; Su, X.; Ji, T.; Yang, D.; Lv, L.; Li, J.; Xu, W. Molecular epidemiology of coxsackievirus A6 associated with outbreaks of hand, foot, and mouth disease in Tianjin, China, in 2013. Arch. Virol. 2015, 160, 1097–1104. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, X.; Pang, C.; Yuan, Z.; Li, H.; Xue, F. Spatio-temporal analysis of the relationship between climate and hand, foot, and mouth disease in Shandong province, China, 2008–2012. BMC Infect. Dis. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.D.; Wu, Y.; Liang, Q.; Hu, J.L.; Dai, Q.G.; Li, Y.; Ai, J.; Tang, F.Y. Epidemiological characteristics and temporal-spatial clustering analysis on hand-foot-mouth disease in Jiangsu province, 2009–2011. Chin. J. Epidemiol. 2012, 33, 813–817. (In Chinese) [Google Scholar]
- Ma, J.M.; Wei, H.Y.; Yu, H.J.; Li, K.; Huang, X.Y. Genetic characteristics of enterovirus 71 VP1 and epidemiology of hand-foot-mouth disease in Xinxiang in 2011. Chin. J. Virol. 2012, 28, 675–680. (In Chinese) [Google Scholar]
- Tian, H.; Zhang, Y.; Sun, Q.; Zhu, S.; Li, X.; Pan, Z.; Xu, W.; Xu, B. Prevalence of multiple enteroviruses associated with hand, foot, and mouth disease in Shijiazhuang City, Hebei province, China: Outbreaks of coxsackieviruses A10 and B3. PLoS ONE 2014, 9, e84233. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Du, J.; Xue, Y.; Su, H.; Yang, F.; Jin, Q. Epidemics and frequent recombination within species in outbreaks of human enterovirus B-associated hand, foot and mouth disease in Shandong China in 2010 and 2011. PLoS ONE 2013, 8, e67157. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.X.; Ou, Z.Y.; Xu, Y.; Zhou, R.; Xia, H.M. Phylogenetic analysis of the VP1 gene of Enterovirus 71 in Guangzhou during the high occurrence period of 2008. Virus Genes 2014, 48, 538–542. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Tan, X.; Li, J.; Jin, Y.; Gong, L.; Hong, M.; Shi, Y.; Zhu, S.; Zhang, B.; Zhang, S.; et al. Molecular epidemiology of coxsackievirus A16: Intratype and prevalent intertype recombination identified. PLoS ONE 2013, 8, e82861. [Google Scholar] [CrossRef] [PubMed]
- Han, J.F.; Yu, N.; Pan, Y.X.; He, S.J.; Xu, L.J.; Cao, R.Y.; Li, Y.X.; Zhu, S.Y.; Zhang, Y.; Qin, E.D.; et al. Phenotypic and genomic characterization of human coxsackievirus A16 strains with distinct virulence in mice. Virus Res. 2014, 179, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Wang, X.; Hu, Y.; Li, Z.; Li, Y.; Sun, S.; Yang, F.; Jin, Q. Changing aetiology of hand, foot and mouth disease in Linyi, China, 2009–2011. Clin. Microbiol. Infect. 2014, 20, O47–O49. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, H.; Ding, S.; Wang, X.; Chen, X.; Wo, Y.; Wang, L.; Huang, D.; Liu, W.; Cao, W. Prevalence of enteroviruses in children with and without hand, foot, and mouth disease in China. BMC Infect. Dis. 2013, 13, 606. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Guo, J.; Zhou, J.Z.; Tang, G.P.; Wang, D.M. Analysis on molecular epidemiological features of enterovirus type 71 in Guizhou Province, 2008–2011. Chin. J. Virol. 2013, 29, 176–179. (In Chinese) [Google Scholar]
- Tan, X.; Huang, X.; Zhu, S.; Chen, H.; Yu, Q.; Wang, H.; Huo, X.; Zhou, J.; Wu, Y.; Yan, D.; et al. The persistent circulation of enterovirus 71 in People’s Republic of China: Causing emerging nationwide epidemics since 2008. PLoS ONE 2011, 6, e25662. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.H.; Wang, S.; Wei, L.L.; Wu, J.; Qi, Z.; Liu, H.; Gou, W.M.; Chen, C.; Tan, X.J.; Zhang, Y.; Zhang, Y.; et al. Multiple viral transmission chains of enterovirus 71 co-circulated in Jilin Province during 2009–2010. Chin. J. Exp. Clin. Virol. 2012, 26, 273–275. [Google Scholar]
- Chen, Y.J.; Chang, S.C.; Tsao, K.C.; Shih, S.R.; Yang, S.L.; Lin, T.Y.; Huang, Y.C. Comparative genomic analysis of coxsackievirus A6 strains of different clinical disease entities. PLoS ONE 2012, 7, e52432. [Google Scholar] [CrossRef] [PubMed]
- Mirand, A.; Henquell, C.; Archimbaud, C.; Ughetto, S.; Antona, D.; Bailly, J.L.; Peigue-Lafeuille, H. Outbreak of hand, foot and mouth disease/herpangina associated with coxsackievirus A6 and A10 infections in 2010, France: A large citywide, prospective observational study. Clin. Microbiol. Infect. 2012, 18, E110–E118. [Google Scholar] [CrossRef] [PubMed]
- Fujimoto, T.; Iizuka, S.; Enomoto, M.; Abe, K.; Yamashita, K.; Hanaoka, N.; Okabe, N.; Yoshida, H.; Yasui, Y.; Kobayashi, M.; et al. Hand, foot, and mouth disease caused by coxsackievirus A6, Japan, 2011. Emerg. Infect. Dis. 2012, 18, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Cabrerizo, M.; Tarrago, D.; Munoz-Almagro, C.; del Amo, E.; Dominguez-Gil, M.; Eiros, J.M.; Lopez-Miragaya, I.; Perez, C.; Reina, J.; Otero, A.; et al. Molecular epidemiology of enterovirus 71, coxsackievirus A16 and A6 associated with hand, foot and mouth disease in Spain. Clin. Microbiol. Infect. 2014, 20, O150–O156. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yeo, A.; Phoon, M.C.; Tan, E.L.; Poh, C.L.; Quak, S.H.; Chow, V.T. The largest outbreak of hand; foot and mouth disease in Singapore in 2008: The role of enterovirus 71 and coxsackievirus A strains. Int. J. Infect. Dis. 2010, 14, e1076–e1081. [Google Scholar] [CrossRef] [PubMed]
- Puenpa, J.; Chieochansin, T.; Linsuwanon, P.; Korkong, S.; Thongkomplew, S.; Vichaiwattana, P.; Theamboonlers, A.; Poovorawan, Y. Hand, foot, and mouth disease caused by coxsackievirus A6, Thailand, 2012. Emerg. Infect. Dis. 2013, 19, 641–643. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Weng, Y.W.; He, W.X.; Zhang, Y.J.; Yang, X.H.; Meng, H.; Xie, J.F.; Wang, J.Z.; Zheng, K.C.; Yan, Y.S. Molecular epidemiology of HFMD-associated pathogen coxsackievirus A6 in Fujian Province, 2011–2013. Chin. J. Virol. 2014, 30, 624–629. (In Chinese) [Google Scholar]
- Wang, H.; Du, Z.; Wang, X.; Liu, Y.; Yuan, Z.; Liu, Y.; Xue, F. Detecting the association between meteorological factors and hand, foot, and mouth disease using spatial panel data models. Int. J. Infect. Dis. 2015, 34, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Li, Z.; Wang, J.; Jia, L.; Liao, Y.; Lai, S.; Guo, Y.; Zhao, D.; Yang, W. Determinants of the incidence of hand, foot and mouth disease in China using geographically weighted regression models. PLoS ONE 2012, 7, e38978. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Yang, Z.; Di, B.; Wang, M. Hand-foot-and-mouth disease and weather factors in Guangzhou, southern China. Epidemiol. Infect. 2014, 142, 1741–1750. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.C.; Song, C.; Wang, J.F.; Li, X.W. Using an autologistic regression model to identify spatial risk factors and spatial risk patterns of hand, foot and mouth disease (HFMD) in Mainland China. BMC Public Health 2014, 14. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Sun, Y.; Lin, C.; Jia, L.; Wu, Q.; Li, X.; Wang, Q. A case-control study to identify environmental risk factors for hand, foot, and mouth disease outbreaks in Beijing. Japan. J. Infect. Dis. 2014, 67, 95–99. [Google Scholar]
- Zhu, Q.; Li, Y.; Li, N.; Han, Q.; Liu, Z.; Li, Z.; Qiu, J.; Zhang, G.; Li, F.; Tian, N. Prolonged exclusive breastfeeding, autumn birth and increased gestational age are associated with lower risk of fever in children with hand, foot, and mouth disease. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2197–2202. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, X.; Zhang, Y.; Gong, L.; Mao, H.; Feng, C.; Ojcius, D.M.; Yan, J. Rapid and sensitive identification of RNA from the emerging pathogen, coxsackievirus A6. Virol. J. 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, J.; Yan, Q.; He, S.; Zhou, W.; Ge, S.; Xia, N. A one-step, triplex, real-time RT-PCR assay for the simultaneous detection of enterovirus 71, coxsackie A16 and pan-enterovirus in a single tube. PLoS ONE 2014, 9, e102724. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Zhou, L.; Tan, L.; Jiang, H.; Wang, Y.; Wei, S.; Nie, S. Application of a new hybrid model with seasonal auto-regressive integrated moving average (ARIMA) and nonlinear auto-regressive neural network (NARNN) in forecasting incidence cases of HFMD in Shenzhen, China. PLoS ONE 2014, 9, e98241. [Google Scholar] [CrossRef] [PubMed]
- Feng, H.; Duan, G.; Zhang, R.; Zhang, W. Time series analysis of hand-foot-mouth disease hospitalization in Zhengzhou: Establishment of forecasting models using climate variables as predictors. PLoS ONE 2014, 9, e87916. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhuang, Z.-C.; Kou, Z.-Q.; Bai, Y.-J.; Cong, X.; Wang, L.-H.; Li, C.; Zhao, L.; Yu, X.-J.; Wang, Z.-Y.; Wen, H.-L. Epidemiological Research on Hand, Foot, and Mouth Disease in Mainland China. Viruses 2015, 7, 6400-6411. https://doi.org/10.3390/v7122947
Zhuang Z-C, Kou Z-Q, Bai Y-J, Cong X, Wang L-H, Li C, Zhao L, Yu X-J, Wang Z-Y, Wen H-L. Epidemiological Research on Hand, Foot, and Mouth Disease in Mainland China. Viruses. 2015; 7(12):6400-6411. https://doi.org/10.3390/v7122947
Chicago/Turabian StyleZhuang, Zhi-Chao, Zeng-Qiang Kou, Yong-Juan Bai, Xiang Cong, Li-Hong Wang, Chun Li, Li Zhao, Xue-Jie Yu, Zhi-Yu Wang, and Hong-Ling Wen. 2015. "Epidemiological Research on Hand, Foot, and Mouth Disease in Mainland China" Viruses 7, no. 12: 6400-6411. https://doi.org/10.3390/v7122947
APA StyleZhuang, Z.-C., Kou, Z.-Q., Bai, Y.-J., Cong, X., Wang, L.-H., Li, C., Zhao, L., Yu, X.-J., Wang, Z.-Y., & Wen, H.-L. (2015). Epidemiological Research on Hand, Foot, and Mouth Disease in Mainland China. Viruses, 7(12), 6400-6411. https://doi.org/10.3390/v7122947





