The Impact of Wolbachia on Virus Infection in Mosquitoes
Abstract
:1. Introduction
2. Wolbachia in Insects
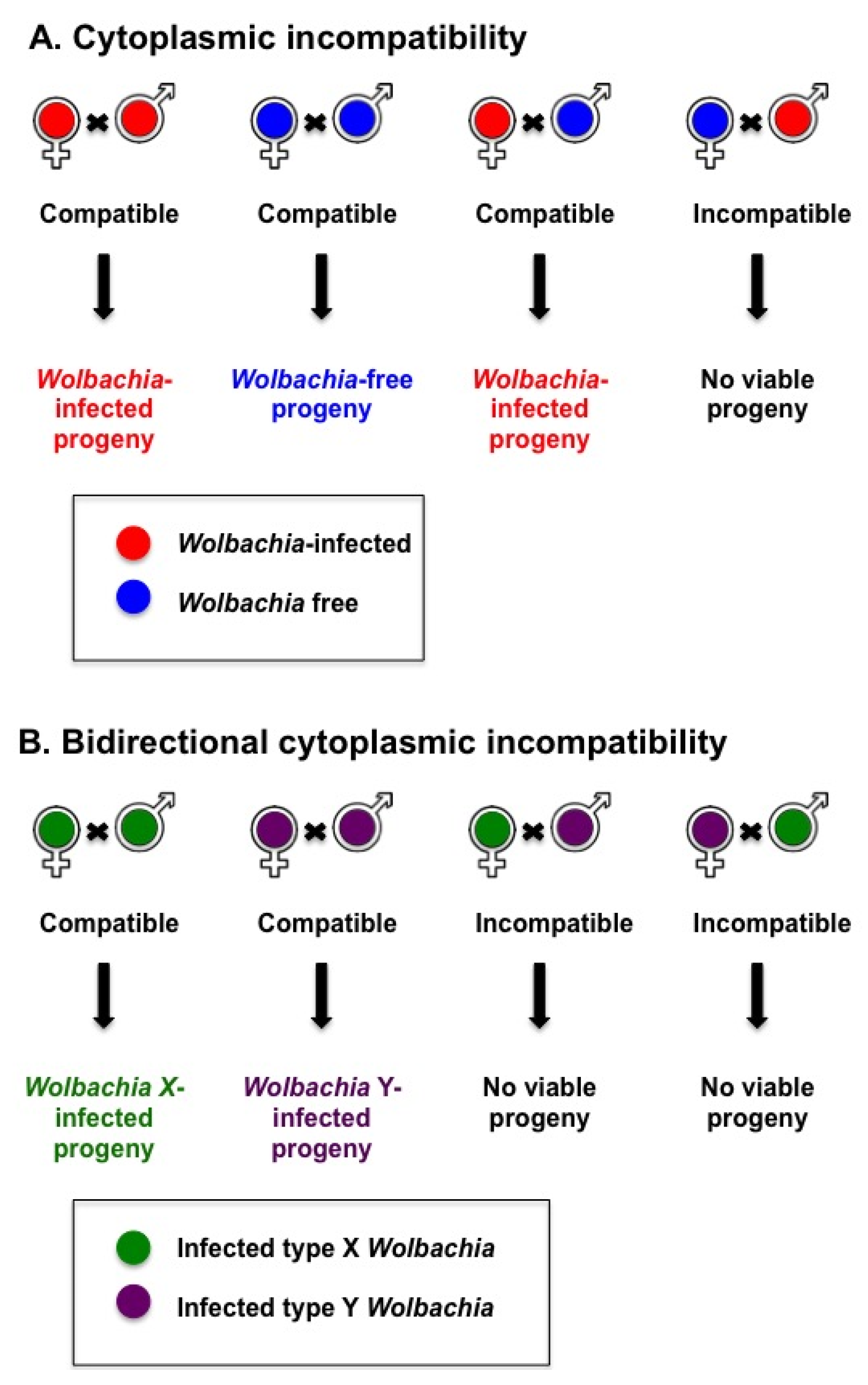
3. Wolbachia-Mediated Antiviral Protection
4. Arboviruses in Mosquitoes
| Virus Family (Genome Nucleic Acid) | Genera | Examples of Arboviruses | Common Vectors |
|---|---|---|---|
| Flaviviridae (ss (+) RNA) | Flavivirus | Dengue virus | Aedes aegypti, Aedes albopictus |
| Japanese encephalitis virus | Culex spp. | ||
| St Louis encephalitis virus | Culex spp. | ||
| West Nile virus | Culex spp. | ||
| Yellow fever virus | Aedes spp. | ||
| Togaviridae (ss (+) RNA) | Alphavirus | Chikungunya virus | Aedes albopictus, Aedes aegypti |
| O’nyong nyong virus | Anopheles spp. | ||
| Semliki Forest virus | Aedes spp. | ||
| Venezuelan equine encephalitis virus | Aedes spp., Culex spp. | ||
| Bunyaviridae (ss (−) RNA) | Orthobunyavirus | La Crosse virus | Aedes triseriatus |
| Phlebovirus | Rift Valley fever virus | Aedes spp., Culex spp. |
5. The Distribution of Wolbachia in Vector Mosquitoes
6. The Intrinsic Effects of Wolbachia on Virus Infection in Mosquitoes
| Host Species | Mode of Wolbachia Infection | Wolbachia Strain | Virus * | Antiviral Effect ** | Reference |
|---|---|---|---|---|---|
| Culex quinquefasciatus | Natural | wPip | WNV | Reduced virus load and transmission | [94] |
| Culex pipiens | Natural | Not typed | WNV | No effect | [95] |
| Culex tarsalis | Transient transinfection | wAlbB | WNV | Enhanced infection rate | [54] |
| Aedes albopictus | Natural | wAlbA and wAlbB | DENV | No effect | [34] |
| Natural | wAlbA and wAlbB | DENV | No effect on virus load, reduced dissemination | [52] | |
| CHIKV | No effect | [51] | |||
| Introgressed | wAlbA and wAlbB | CHIKV | No effect | [90] | |
| Stable transinfection | wMel | DENV | Reduced transmission | [50] | |
| CHIKV | Reduced transmission | [90] | |||
| Aedes polynesiensis | Stable transinfection | wAlbB | DENV | Decreased virus load, reduced transmission (compared to line naturally infected with wPolA) | [33] |
| Aedes aegypti | Stable transinfection | wMelPop | DENV | Reduced infection rate, virus load and transmission | [36] |
| CHIKV | Reduced infection rate and virus load | [36] | |||
| WNV | Reduced infection rate, viral load and transmission | [53] | |||
| YFV | Reduced infection rate and virus load | [46] | |||
| wMel | DENV | Reduced virus load, dissemination and transmission | [41] | ||
| CHIKV | Reduced virus load and transmission | [46] | |||
| WNV | Delayed virus accumulation, reduced transmission | [53] | |||
| YFV | Reduced virus load | [46] | |||
| wAlbB | DENV | Reduced infection rate, virus load and transmission | [49] |
7. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bourtzis, K.; Dobson, S.L.; Xi, Z.; Rasgon, J.L.; Calvitti, M.; Moreira, L.A.; Bossin, H.C.; Moretti, R.; Baton, L.A.; Hughes, G.L.; et al. Harnessing mosquito-Wolbachia symbiosis for vector and disease control. Acta Trop. 2014, 132, S150–S163. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.N. Bacteria and antiviral immunity in insects. Curr. Opin. Insect Sci. 2015, 8, 97–103. [Google Scholar] [CrossRef]
- Rainey, S.M.; Shah, P.; Kohl, A.; Dietrich, I. Understanding the Wolbachia-mediated inhibition of arboviruses in mosquitoes: Progress and challenges. J. Gen. Virol. 2014, 95, 517–530. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.P. Wolbachia and arbovirus inhibition in mosquitoes. Future Microbiol. 2013, 8, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Moreira, L.A. Can Wolbachia be used to control malaria? Mem. Inst. Oswaldo Cruz 2011, 106, S212–S217. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Montgomery, B.L.; Popovici, J.; Iturbe-Ormaetxe, I.; Johnson, P.H.; Muzzi, F.; Greenfield, M.; Durkan, M.; Leong, Y.S.; Dong, Y.; et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 2011, 476, 454–457. [Google Scholar] [CrossRef] [PubMed]
- Zug, R.; Hammerstein, P. Still a host of hosts for Wolbachia: Analysis of recent data suggests that 40% of terrestrial arthropod species are infected. PLoS One 2012, 7, e38544. [Google Scholar] [CrossRef] [PubMed]
- Hilgenboecker, K.; Hammerstein, P.; Schlattmann, P.; Telschow, A.; Werren, J.H. How many species are infected with Wolbachia?—A statistical analysis of current data. FEMS Microbiol. Lett. 2008, 281, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Werren, J.H.; Baldo, L.; Clark, M.E. Wolbachia: Master manipulators of invertebrate biology. Nat. Rev. Microbiol. 2008, 6, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.Z.; Li, S.J.; Xue, X.; Yin, X.J.; Ren, S.X.; Jiggins, F.M.; Greeff, J.M.; Qiu, B.L. The intracellular bacterium Wolbachia uses parasitoid wasps as phoretic vectors for efficient horizontal transmission. PLoS Pathog. 2015, 10, e1004672. [Google Scholar] [CrossRef] [PubMed]
- Jiggins, F.M.; Hurst, G.D. Microbiology. Rapid insect evolution by symbiont transfer. Science 2011, 332, 185–186. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, S.L.; Giordano, R.; Colbert, A.M.; Karr, T.L.; Robertson, H.M. 16S rRNA phylogenetic analysis of the bacterial endosymbionts associated with cytoplasmic incompatibility in insects. Proc. Natl. Acad. Sci. USA 1992, 89, 2699–2702. [Google Scholar] [CrossRef] [PubMed]
- Werren, J.H.; Zhang, W.; Guo, L.R. Evolution and phylogeny of Wolbachia: Reproductive parasites of arthropods. Proc. R. Soc. London B Biol. Sci. 1995, 261, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Vavre, F.; Fleury, F.; Lepetit, D.; Fouillet, P.; Bouletreau, M. Phylogenetic evidence for horizontal transmission of Wolbachia in host-parasitoid associations. Mol. Biol. Evol. 1999, 16, 1711–1723. [Google Scholar] [CrossRef] [PubMed]
- Turelli, M.; Hoffmann, A.A. Rapid spread of an inherited incompatibility factor in California Drosophila. Nature 1991, 353, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.J.; Vavre, F.; Beukeboom, L.W. Manipulation of arthropod sex determination by endosymbionts: Diversity and molecular mechanisms. Sex. Dev. Genet. Mol. Biol. Evol. Endocrinol. Embryol. Pathol. Sex Determ. Differ. 2014, 8, 59–73. [Google Scholar] [CrossRef] [PubMed]
- Stouthamer, R.; Breeuwer, J.A.; Hurst, G.D. Wolbachia pipientis: Microbial manipulator of arthropod reproduction. Annu. Rev. Microbiol. 1999, 53, 71–102. [Google Scholar] [CrossRef] [PubMed]
- Cytoplasmic Incompatibility. Available online: http://dobsonserv.ca.uky.edu/DobsonSite/CI1.html (accessed on 25 July 2015).
- Tram, U.; Fredrick, K.; Werren, J.H.; Sullivan, W. Paternal chromosome segregation during the first mitotic division determines Wolbachia-induced cytoplasmic incompatibility phenotype. J. Cell Sci. 2006, 119, 3655–3663. [Google Scholar] [CrossRef] [PubMed]
- Tram, U.; Sullivan, W. Role of delayed nuclear envelope breakdown and mitosis in Wolbachia-induced cytoplasmic incompatibility. Science 2002, 296, 1124–1126. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.B.; Stainton, K.; Harris, S.; Kambris, Z.; Sutton, E.R.; Bonsall, M.B.; Parkhill, J.; Sinkins, S.P. Transcriptional regulation of Culex pipiens mosquitoes by Wolbachia influences cytoplasmic incompatibility. PLoS Pathog. 2013, 9, e1003647. [Google Scholar] [CrossRef] [PubMed]
- Landmann, F.; Orsi, G.A.; Loppin, B.; Sullivan, W. Wolbachia-mediated cytoplasmic incompatibility is associated with impaired histone deposition in the male pronucleus. PLoS Pathog. 2009, 5, e1000343. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.A.; Iturbe-Ormaetxe, I.; Callahan, A.G.; Phillips, B.L.; Billington, K.; Axford, J.K.; Montgomery, B.; Turley, A.P.; O’Neill, S.L. Stability of the wMel Wolbachia infection following invasion into Aedes aegypti populations. PLoS Negl. Trop. Dis. 2014, 8, e3115. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Ferreira, A.; Ashburner, M. The bacterial symbiont Wolbachia induces resistance to RNA viral infections in Drosophila melanogaster. PLoS Biol. 2008, 6, e1000002. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Wolbachia and virus protection in insects. Science 2008, 322. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.E.; Leong, Y.S.; O’Neill, S.L.; Johnson, K.N. Variation in antiviral protection mediated by different Wolbachia strains in Drosophila simulans. PLoS Pathog. 2009, 5, e1000656. [Google Scholar] [CrossRef] [PubMed]
- Osborne, S.E.; Iturbe-Ormaetxe, I.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Antiviral protection and the importance of Wolbachia density and tissue tropism in Drosophila simulans. Appl. Environ. Microbiol. 2012, 78, 6922–6929. [Google Scholar] [CrossRef] [PubMed]
- Dobson, S.L.; Bourtzis, K.; Braig, H.R.; Jones, B.F.; Zhou, W.; Rousset, F.; O’Neill, S.L. Wolbachia infections are distributed throughout insect somatic and germ line tissues. Insect Biochem. Mol. Biol. 1999, 29, 153–160. [Google Scholar] [CrossRef]
- Dutton, T.J.; Sinkins, S.P. Strain-specific quantification of Wolbachia density in Aedes albopictus and effects of larval rearing conditions. Insect Mol. Biol. 2004, 13, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.J.; Riegler, M. Evolutionary dynamics of wAu-like Wolbachia variants in neotropical Drosophila spp. Appl. Environ. Microbiol. 2006, 72, 826–835. [Google Scholar] [CrossRef] [PubMed]
- Martinez, J.; Longdon, B.; Bauer, S.; Chan, Y.S.; Miller, W.J.; Bourtzis, K.; Teixeira, L.; Jiggins, F.M. Symbionts commonly provide broad spectrum resistance to viruses in insects: A comparative analysis of Wolbachia strains. PLoS Pathog. 2014, 10, e1004369. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Marialva, M.S.; Esteves, S.S.; Weinert, L.A.; Martinez, J.; Jiggins, F.M.; Teixeira, L. Wolbachia variants induce differential protection to viruses in Drosophila melanogaster: A phenotypic and phylogenomic analysis. PLoS Genet. 2013, 9, e1003896. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.W.; Zhou, G.L.; Lu, P.; Xi, Z.Y. Replacing a native Wolbachia with a novel strain results in an increase in endosymbiont load and resistance to Dengue virus in a mosquito vector. PLoS Negl. Trop. Dis. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Bian, G.W.; Pan, X.L.; Xi, Z.Y. Wolbachia induces density-dependent inhibition to Dengue virus in mosquito cells. PLoS Negl. Trop. Dis. 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Frentiu, F.D.; Robinson, J.; Young, P.R.; McGraw, E.A.; O’Neill, S.L. Wolbachia-mediated resistance to dengue virus infection and death at the cellular level. PLoS ONE 2010, 5, e13398. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.J.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A Wolbachia symbiont in Aedes aegypti limits infection with Dengue, Chikungunya, and Plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Aksoy, S. Tissue tropism, transmission and expression of foreign genes in vivo in midgut symbionts of tsetse flies. Insect Mol. Biol. 1999, 8, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Q.; Ruel, T.D.; Zhou, W.; Moloo, S.K.; Majiwa, P.; O’Neill, S.L.; Aksoy, S. Tissue distribution and prevalence of Wolbachia infections in tsetse flies, Glossina spp. Med. Vet. Entomol. 2000, 14, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Frydman, H.M.; Li, J.M.; Robson, D.N.; Wieschaus, E. Somatic stem cell niche tropism in Wolbachia. Nature 2006, 441, 509–512. [Google Scholar] [CrossRef] [PubMed]
- Min, K.T.; Benzer, S. Wolbachia, normally a symbiont of Drosophila, can be virulent, causing degeneration and early death. Proc. Natl. Acad. Sci. USA 1997, 94, 10792–10796. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Johnson, P.H.; Moreira, L.A.; Iturbe-Ormaetxe, I.; Frentiu, F.D.; McMeniman, C.J.; Leong, Y.S.; Dong, Y.; Axford, J.; Kriesner, P.; et al. The wMel Wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 2011, 476, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Jaenike, J. Coupled population dynamics of endosymbionts within and between hosts. Oikos 2009, 118, 353–362. [Google Scholar] [CrossRef]
- Graham, R.I.; Grzywacz, D.; Mushobozi, W.L.; Wilson, K. Wolbachia in a major African crop pest increases susceptibility to viral disease rather than protects. Ecol. Lett. 2012, 15, 993–1000. [Google Scholar] [CrossRef] [PubMed]
- Rances, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; O’Neill, S.L. The relative importance of innate immune priming in Wolbachia-mediated dengue interference. PLoS Pathog. 2012, 8, e1002548. [Google Scholar] [CrossRef] [PubMed]
- Shaw, A.E.; Veronesi, E.; Maurin, G.; Ftaich, N.; Guiguen, F.; Rixon, F.; Ratinier, M.; Mertens, P.; Carpenter, S.; Palmarini, M.; et al. Drosophila melanogaster as a model organism for Bluetongue virus replication and tropism. J. Virol. 2012, 86, 9015–9024. [Google Scholar] [CrossRef] [PubMed]
- Van den Hurk, A.F.; Hall-Mendelin, S.; Pyke, A.T.; Frentiu, F.D.; McElroy, K.; Day, A.; Higgs, S.; O’Neill, S.L. Impact of Wolbachia on infection with Chikungunya and Yellow Fever viruses in the mosquito vector Aedes aegypti. PLoS Negl. Trop. Dis. 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Longdon, B.; Fabian, D.K.; Hurst, G.D.D.; Jiggins, F.M. Male-killing Wolbachia do not protect Drosophila bifasciata against viral infection. BMC Microbiology 2012, 12. [Google Scholar] [CrossRef] [PubMed]
- Unckless, R.L.; Jaenike, J. Maintenance of a male-killing Wolbachia in Drosophila innubila by male-killing dependent and male-killing independent mechanisms. Evolution 2012, 66, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.W.; Xu, Y.; Lu, P.; Xie, Y.; Xi, Z.Y. The endosymbiotic bacterium Wolbachia induces resistance to Dengue virus in Aedes aegypti. PLoS Pathog. 2010, 6, e1000833. [Google Scholar] [CrossRef] [PubMed]
- Blagrove, M.S.; Arias-Goeta, C.; Failloux, A.B.; Sinkins, S.P. Wolbachia strain wMel induces cytoplasmic incompatibility and blocks dengue transmission in Aedes albopictus. Proc. Natl. Acad. Sci. USA 2012, 109, 255–260. [Google Scholar] [CrossRef]
- Mousson, L.; Martin, E.; Zouache, K.; Madec, Y.; Mavingui, P.; Failloux, A.B. Wolbachia modulates Chikungunya replication in Aedes albopictus. Mol. Ecol. 2010, 19, 1953–1964. [Google Scholar] [CrossRef] [PubMed]
- Mousson, L.; Zouache, K.; Arias-Goeta, C.; Raquin, V.; Mavingui, P.; Failloux, A.B. The native Wolbachia symbionts limit transmission of dengue virus in Aedes albopictus. PLoS Negl. Trop. Dis. 2012, 6, e1989. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Lu, G.J.; Torres, S.; Edmonds, J.H.; Kay, B.H.; Khromykh, A.A.; Asgari, S. Effect of Wolbachia on replication of West Nile Virus in a mosquito cell line and adult mosquitoes. J. Virol. 2013, 87, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Dodson, B.L.; Hughes, G.L.; Paul, O.; Matacchiero, A.C.; Kramer, L.D.; Rasgon, J.L. Wolbachia enhances West Nile virus (WNV) infection in the mosquito Culex tarsalis. PLoS Negl. Trop. Dis. 2014, 8, e2965. [Google Scholar] [CrossRef] [PubMed]
- Caragata, E.P.; Rances, E.; Hedges, L.M.; Gofton, A.W.; Johnson, K.N.; O’Neill, S.L.; McGraw, E.A. Dietary cholesterol modulates pathogen blocking by Wolbachia. PLoS Pathog. 2013, 9, e1003459. [Google Scholar] [CrossRef] [PubMed]
- Kambris, Z.; Blagborough, A.M.; Pinto, S.B.; Blagrove, M.S.C.; Godfray, H.C.J.; Sinden, R.E.; Sinkins, S.P. Wolbachia stimulates immune gene expression and inhibits Plasmodium development in Anopheles gambiae. PLoS Pathog. 2010, 6, e1001143. [Google Scholar] [CrossRef] [PubMed]
- Kambris, Z.; Cook, P.E.; Phuc, H.K.; Sinkins, S.P. Immune activation by life-shortening Wolbachia and reduced filarial competence in mosquitoes. Science 2009, 326, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.L.; Zhou, G.L.; Wu, J.H.; Bian, G.W.; Lu, P.; Raikhel, A.S.; Xi, Z.Y. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar] [CrossRef] [PubMed]
- Wong, Z.S.; Brownlie, J.C.; Johnson, K.N. Oxidative stress correlates with Wolbachia-mediated antiviral protection in Wolbachia-Drosophila associations. Appl. Environ. Microbiol. 2015, 81, 3001–3005. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S. Role of microRNAs in arbovirus/vector interactions. Viruses 2014, 6, 3514–3534. [Google Scholar] [CrossRef] [PubMed]
- Asgari, S. Regulatory role of cellular and viral microRNAs in insect-virus interactions. Curr. Opin. Insect Sci. 2015, 8, 104–110. [Google Scholar] [CrossRef]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Tabachnick, W.J. Nature, nurture and evolution of intra-species variation in mosquito arbovirus transmission competence. Int. J. Environ. Res. Public Health 2013, 10, 249–277. [Google Scholar] [CrossRef] [PubMed]
- Ruckert, C.; Bell-Sakyi, L.; Fazakerley, J.K.; Fragkoudis, R. Antiviral responses of arthropod vectors: An update on recent advances. Virusdisease 2014, 25, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Hertig, M.; Wolbach, S.B. Studies on Rickettsia-like micro-organisms in insects. J. Med. Res. 1924, 44, 329–374.7. [Google Scholar] [PubMed]
- Kittayapong, P.; Baisley, K.J.; Baimai, V.; O’Neill, S.L. Distribution and diversity of Wolbachia infections in Southeast Asian mosquitoes (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 340–345. [Google Scholar] [CrossRef] [PubMed]
- Ricci, I.; Cancrini, G.; Gabrielli, S.; D’Amelio, S.; Favi, G. Searching for Wolbachia (Rickettsiales: Rickettsiaceae) in mosquitoes (Diptera: Culicidae): Large polymerase chain reaction survey and new identifications. J. Med. Entomol. 2002, 39, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Rasgon, J.L.; Scott, T.W. An initial survey for Wolbachia (Rickettsiales: Rickettsiaceae) infections in selected California mosquitoes (Diptera: Culicidae). J. Med. Entomol. 2004, 41, 255–257. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.L.; Dodson, B.L.; Johnson, R.M.; Murdock, C.C.; Tsujimoto, H.; Suzuki, Y.; Patt, A.A.; Cui, L.; Nossa, C.W.; Barry, R.M.; et al. Native microbiome impedes vertical transmission of Wolbachia in Anopheles mosquitoes. Proc. Natl. Acad. Sci. USA 2014, 111, 12498–12503. [Google Scholar] [CrossRef] [PubMed]
- Baldini, F.; Segata, N.; Pompon, J.; Marcenac, P.; Robert Shaw, W.; Dabire, R.K.; Diabate, A.; Levashina, E.A.; Catteruccia, F. Evidence of natural Wolbachia infections in field populations of Anopheles gambiae. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.L.; Rasgon, J.L. Transinfection: A method to investigate Wolbachia-host interactions and control arthropod-borne disease. Insect Mol. Biol. 2014, 23, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Dean, J.L.; Khoo, C.; Dobson, S.L. Generation of a novel Wolbachia infection in Aedes albopictus (Asian tiger mosquito) via embryonic microinjection. Insect Biochem. Mol. Biol. 2005, 35, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Khoo, C.C.; Dobson, S.L. Interspecific transfer of Wolbachia into the mosquito disease vector Aedes albopictus. Proc. Biol. Sci. 2006, 273, 1317–1322. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Khoo, C.C.; Dobson, S.L. Wolbachia establishment and invasion in an Aedes aegypti laboratory population. Science 2005, 310, 326–328. [Google Scholar] [CrossRef] [PubMed]
- Bian, G.; Joshi, D.; Dong, Y.; Lu, P.; Zhou, G.; Pan, X.; Xu, Y.; Dimopoulos, G.; Xi, Z. Wolbachia invades Anopheles stephensi populations and induces refractoriness to Plasmodium infection. Science 2013, 340, 748–751. [Google Scholar] [CrossRef] [PubMed]
- Ruang-Areerate, T.; Kittayapong, P. Wolbachia transinfection in Aedes aegypti: A potential gene driver of dengue vectors. Proc. Natl. Acad. Sci. USA 2006, 103, 12534–12539. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Song, S.; Sinkins, S.P. Wolbachia in the Culex pipiens group mosquitoes: Introgression and superinfection. J. Hered. 2009, 100, 192–196. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Lane, R.V.; Cass, B.N.; Fong, A.W.C.; Sidhu, M.; Wang, Y.F.; O’Neill, S.L. Stable introduction of a life-shortening Wolbachia infection into the mosquito Aedes aegypti. Science 2009, 323, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Suh, E.; Mercer, D.R.; Fu, Y.; Dobson, S.L. Pathogenicity of life-shortening Wolbachia in Aedes albopictus after transfer from Drosophila melanogaster. Appl. Environ. Microbiol. 2009, 75, 7783–7788. [Google Scholar] [CrossRef] [PubMed]
- Calvitti, M.; Moretti, R.; Lampazzi, E.; Bellini, R.; Dobson, S.L. Characterization of a new Aedes albopictus (Diptera: Culicidae)-Wolbachia pipientis (Rickettsiales: Rickettsiaceae) symbiotic association generated by artificial transfer of the wPip strain from Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 2010, 47, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Gavotte, L.; Mercer, D.R.; Dobson, S.L. Artificial triple Wolbachia infection in Aedes albopictus yields a new pattern of unidirectional cytoplasmic incompatibility. Appl. Environ. Microbiol. 2010, 76, 5887–5891. [Google Scholar] [CrossRef] [PubMed]
- Andrews, E.S.; Crain, P.R.; Fu, Y.; Howe, D.K.; Dobson, S.L. Reactive oxygen species production and Brugia pahangi survivorship in Aedes polynesiensis with artificial Wolbachia infection types. PLoS Pathog. 2012, 8, e1003075. [Google Scholar] [CrossRef] [PubMed]
- Calvitti, M.; Moretti, R.; Skidmore, A.R.; Dobson, S.L. Wolbachia strain wPip yields a pattern of cytoplasmic incompatibility enhancing a Wolbachia-based suppression strategy against the disease vector Aedes albopictus. Parasit. Vectors 2012, 5. [Google Scholar] [CrossRef] [PubMed]
- Hughes, G.L.; Vega-Rodriguez, J.; Xue, P.; Rasgon, J.L. Wolbachia strain wAlbB enhances infection by the rodent malaria parasite Plasmodium berghei in Anopheles gambiae mosquitoes. Appl. Environ. Microbiol. 2012, 78, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Turelli, M. Evolution of incompatibility-inducing microbes and their hosts. Evolution 1994, 48, 1500–1513. [Google Scholar] [CrossRef]
- McGraw, E.A.; Merritt, D.J.; Droller, J.N.; O’Neill, S.L. Wolbachia density and virulence attenuation after transfer into a novel host. Proc. Natl. Acad. Sci. USA 2002, 99, 2918–2923. [Google Scholar] [CrossRef] [PubMed]
- Carrington, L.B.; Hoffmann, A.A.; Weeks, A.R. Monitoring long-term evolutionary changes following Wolbachia introduction into a novel host: The Wolbachia popcorn infection in Drosophila simulans. Proc. Biol. Sci. 2010, 277, 2059–2068. [Google Scholar] [CrossRef] [PubMed]
- Weeks, A.R.; Turelli, M.; Harcombe, W.R.; Reynolds, K.T.; Hoffmann, A.A. From parasite to mutualist: Rapid evolution of Wolbachia in natural populations of Drosophila. PLoS Biol. 2007, 5, e114. [Google Scholar] [CrossRef] [PubMed]
- Woolfit, M.; Iturbe-Ormaetxe, I.; Brownlie, J.C.; Walker, T.; Riegler, M.; Seleznev, A.; Popovici, J.; Rances, E.; Wee, B.A.; Pavlides, J.; et al. Genomic evolution of the pathogenic Wolbachia strain, wMelPop. Genome Biol. Evol. 2013, 5, 2189–2204. [Google Scholar] [CrossRef] [PubMed]
- Blagrove, M.S.; Arias-Goeta, C.; di Genua, C.; Failloux, A.B.; Sinkins, S.P. A Wolbachia wMel transinfection in Aedes albopictus is not detrimental to host fitness and inhibits Chikungunya virus. PLoS Negl. Trop. Dis. 2013, 7, e2152. [Google Scholar] [CrossRef] [PubMed]
- Baton, L.A.; Pacidonio, E.C.; Goncalves, D.S.; Moreira, L.A. wFlu: Characterization and evaluation of a native Wolbachia from the mosquito Aedes fluviatilis as a potential vector control agent. PLoS ONE 2013, 8, e59619. [Google Scholar] [CrossRef] [PubMed]
- Zele, F.; Nicot, A.; Berthomieu, A.; Weill, M.; Duron, O.; Rivero, A. Wolbachia increases susceptibility to Plasmodium infection in a natural system. Proc. Biol. Sci. 2014, 281. [Google Scholar] [CrossRef] [PubMed]
- Murdock, C.C.; Blanford, S.; Hughes, G.L.; Rasgon, J.L.; Thomas, M.B. Temperature alters Plasmodium blocking by Wolbachia. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.L.; Meola, M.A. The native Wolbachia endosymbionts of Drosophila melanogaster and Culex quinquefasciatus increase host resistance to West Nile virus infection. PLoS ONE 2010, 5, e11977. [Google Scholar] [CrossRef] [PubMed]
- Micieli, M.V.; Glaser, R.L. Somatic Wolbachia (Rickettsiales: Rickettsiaceae) levels in Culex quinquefasciatus and Culex pipiens (Diptera: Culicidae) and resistance to West Nile virus infection. J. Med. Entomol. 2014, 51, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Wong, Z.S.; Hedges, L.M.; Brownlie, J.C.; Johnson, K.N. Wolbachia-mediated antibacterial protection and immune gene regulation in Drosophila. PLoS ONE 2011, 6, e25430. [Google Scholar] [CrossRef] [PubMed]
- Chrostek, E.; Marialva, M.S.; Yamada, R.; O’Neill, S.L.; Teixeira, L. High anti-viral protection without immune upregulation after interspecies Wolbachia transfer. PLoS One 2014, 9, e99025. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Yamada, R.; O’Neill, S.L.; Johnson, K.N. The small interfering RNA pathway is not essential for Wolbachia-mediated antiviral protection in Drosophila melanogaster. Appl. Environ. Microbiol. 2012, 78, 6773–6776. [Google Scholar] [CrossRef] [PubMed]
- Rances, E.; Johnson, T.K.; Popovici, J.; Iturbe-Ormaetxe, I.; Zakir, T.; Warr, C.G.; O’Neill, S.L. The Toll and Imd pathways are not required for Wolbachia-mediated dengue virus interference. J. Virol. 2013, 87, 11945–11949. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.G.; Naylor, H.; Esteves, S.S.; Pais, I.S.; Martins, N.E.; Teixeira, L. The Toll-dorsal pathway is required for resistance to viral oral infection in Drosophila. PLoS Pathog. 2014, 10, e1004507. [Google Scholar] [CrossRef] [PubMed]
- Bull, J.J.; Turelli, M. Wolbachia versus dengue: Evolutionary forecasts. Evol. Med. Public Health 2013, 2013, 197–207. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johnson, K.N. The Impact of Wolbachia on Virus Infection in Mosquitoes. Viruses 2015, 7, 5705-5717. https://doi.org/10.3390/v7112903
Johnson KN. The Impact of Wolbachia on Virus Infection in Mosquitoes. Viruses. 2015; 7(11):5705-5717. https://doi.org/10.3390/v7112903
Chicago/Turabian StyleJohnson, Karyn N. 2015. "The Impact of Wolbachia on Virus Infection in Mosquitoes" Viruses 7, no. 11: 5705-5717. https://doi.org/10.3390/v7112903
APA StyleJohnson, K. N. (2015). The Impact of Wolbachia on Virus Infection in Mosquitoes. Viruses, 7(11), 5705-5717. https://doi.org/10.3390/v7112903






