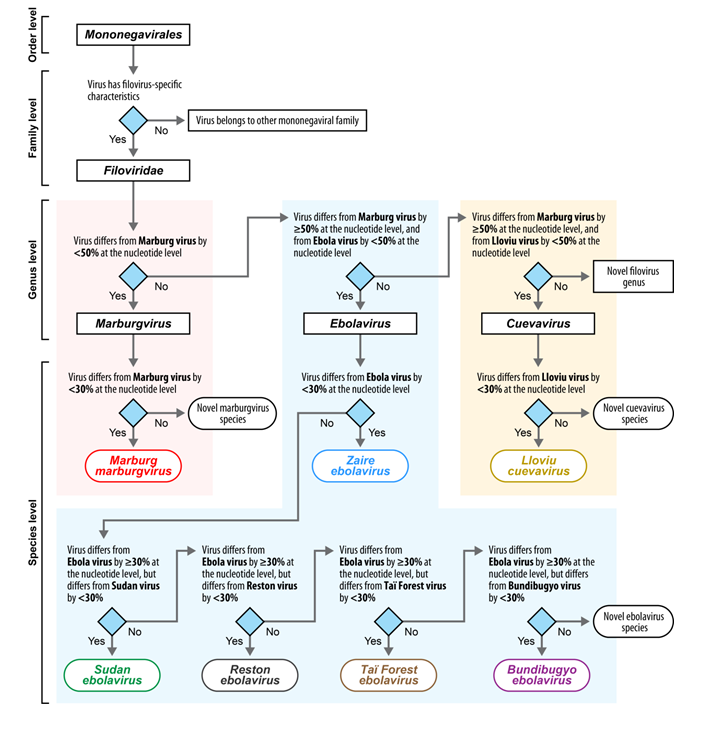
Filovirus RefSeq Entries: Evaluation and Selection of Filovirus Type Variants, Type Sequences, and Names
Abstract
:1. Introduction
2. Current Filovirus RefSeq Entries
| Current Taxonomy and Nomenclature (Ninth ICTV Report and Updates) |
|---|
| Order Mononegavirales |
| Family Filoviridae |
| Genus Marburgvirus |
| Species Marburg marburgvirus |
| Virus 1: Marburg virus (MARV) |
| Virus 2: Ravn virus (RAVV) |
| Genus Ebolavirus |
| Species Taï Forest ebolavirus |
| Virus: Taï Forest virus (TAFV) |
| Species Reston ebolavirus |
| Virus: Reston virus (RESTV) |
| Species Sudan ebolavirus |
| Virus: Sudan virus (SUDV) |
| Species Zaire ebolavirus |
| Virus: Ebola virus (EBOV) |
| Species Bundibugyo ebolavirus |
| Virus: Bundibugyo virus (BDBV) |
| Genus Cuevavirus |
| Species Lloviu cuevavirus |
| Virus: Lloviu virus (LLOV) |
| Filovirus Species | Type Virus of Species (Virus Abbreviation) | Type Variant and Isolate of Type Virus of Species | Type Sequence of Type Variant of Type Virus of Species (RefSeq) |
|---|---|---|---|
| Bundibugyo ebolavirus | Bundibugyo virus (BDBV) | Unnamed variant represented by isolate “811250”1 | NC_014373 |
| Lloviu cuevavirus | Lloviu virus (LLOV) | Unnamed variant represented by isolate “MS-Liver-86/2003”2 | NC_016144 |
| Marburg marburgvirus | Marburg virus (MARV) | Unnamed variant represented by isolate “Musoke” | NC_001608 |
| Reston ebolavirus | Reston virus (RESTV) | Unnamed variant represented by isolate “Pennsylvania” | NC_004161 |
| Sudan ebolavirus | Sudan virus (SUDV) | Unnamed variant represented by isolate “Boniface” [sic]3 | None |
| Taï Forest ebolavirus | Taï Forest virus (TAFV) | Unnamed variant represented by isolate “Côte d’Ivoire”4 | NC_014372 |
| Zaire ebolavirus | Ebola virus (EBOV) | Unnamed variant represented by isolate “Mayinga” | NC_002549 |
3. RefSeq Entry Reevaluation
3.1. Cuevavirus RefSeq Entries
| Full name: | Lloviu virus M.schreibersii-wt/ESP/2003/Asturias-Bat86 |
| Shortened name: | LLOV/M.sch/ESP/03/Ast-Bat86 |
| Abbreviated name: | LLOV/Ast-Bat86 |
3.2. Ebolavirus RefSeq Entries
3.2.1. Bundibugyo Virus
| Full name: | Bundibugyo virus H.sapiens-tc/UGA/2007/Butalya-811250 |
| Shortened name: | BDBV/H.sap/UGA/07/But-811250 |
| Abbreviated name: | BDBV/But-811250 |
3.2.2. Ebola Virus
| Full name: | Ebola virus H.sapiens-tc/COD/1976/Yambuku-Mayinga |
| Shortened name: | EBOV/H.sap/COD/76/Yam-May |
| Abbreviated name: | EBOV/Yam-May |
3.2.3. Reston Virus
| Full name: | Reston virus M.fascicularis-tc/USA/1989/Philippines89-Pennsylvania |
| Shortened name: | RESTV/M.fas/USA/89/Phi89-Pen |
| Abbreviated name: | RESTV/Phi89-Pen |
3.2.4. Sudan Virus
| Full name: | Sudan virus H.sapiens-tc/UGA/2000/Gulu-808892 |
| Shortened name: | SUDV/H.sap/UGA/00/Gul-808892 |
| Abbreviated name: | SUDV/Gul-808892 |
3.2.5. Taï Forest Virus
| Full name: | Taï Forest virus H.sapiens-tc/CIV/1994/Pauléoula-CI |
| Shortened name: | TAFV/H.sap/CIV/94/Pau-CI |
| Abbreviated name: | TAFV/Pau-CI |
3.3. Marburgvirus RefSeq Entries
3.3.1. Marburg Virus
| Full name: | Marburg virus H.sapiens-tc/KEN/1980/Mt. Elgon-Musoke |
| Shortened name: | MARV/Hsap/KEN/80/MtE-Mus |
| Abbreviated name: | MARV/MtE-Mus |
3.3.2. Ravn Virus
| Full name: | Ravn virus H.sapiens-tc/KEN/1987/Kitum Cave-810040 |
| Shortened name: | RAVV/H.sap/KEN/87/KiC-810040 |
| Abbreviated name: | RAVV/KiC-810040 |
| Filovirus Species | Type Virus of Species (Virus Abbreviation) | Type Variant and Isolate of Type Virus of Species | Type Sequence of Type Variant of Type Virus of Species (RefSeq) |
|---|---|---|---|
| Bundibugyo ebolavirus | Bundibugyo virus (BDBV) | Bundibugyo virus H.sapiens-tc/UGA/2007/Butalya-811250 | NC_014373 |
| Lloviu cuevavirus | Lloviu virus (LLOV) | Lloviu virus M.schreibersii-wt/ESP/2003/Asturias-Bat861 | NC_016144 |
| Marburg marburgvirus | Marburg virus (MARV) | Marburg virus H.sapiens-tc/KEN/1980/Mt. Elgon-Musoke | NC_001608 |
| Reston ebolavirus | Reston virus (RESTV) | Reston virus M.fascicularis-tc/USA/1989/Philippines89-Pennsylvania | NC_004161 |
| Sudan ebolavirus | Sudan virus (SUDV) | Sudan virus H.sapiens-tc/UGA/2000/Gulu-808892 | NC_006432 |
| Taï Forest ebolavirus | Taï Forest virus (TAFV) | Taï Forest virus H.sapiens-tc/CIV/1994/Pauléoula-CI | NC_014372 |
| Zaire ebolavirus | Ebola virus (EBOV) | Ebola virus H.sapiens-tc/COD/1976/Yambuku-Mayinga | NC_002549 |
Acknowledgments
Author contributions
Conflicts of Interest
References
- Pruitt, K.D.; Tatusova, T.; Brown, G.R.; Maglott, D.R. NCBI Reference Sequences (RefSeq): Current status, new features and genome annotation policy. Nucl. Acids Res. 2012, 40, D130–D135. [Google Scholar] [CrossRef]
- Bao, Y.; Federhen, S.; Leipe, D.; Pham, V.; Resenchuk, S.; Rozanov, M.; Tatusov, R.; Tatusova, T. National center for biotechnology information viral genomes project. J. Virol. 2004, 78, 7291–7298. [Google Scholar] [CrossRef] [PubMed]
- Brister, J.R.; Le Mercier, P.; Hu, J.C. Microbial virus genome annotation-mustering the troops to fight the sequence onslaught. Virology 2012, 434, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Klimke, W.; O’Donovan, C.; White, O.; Brister, J.R.; Clark, K.; Fedorov, B.; Mizrachi, I.; Pruitt, K.D.; Tatusova, T. Solving the Problem: Genome Annotation Standards before the Data Deluge. Stand. Genomic Sci. 2011, 5, 168–193. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Jia, P.; Zhao, Z. VirusFinder: Software for efficient and accurate detection of viruses and their integration sites in host genomes through next generation sequencing data. PLoS One 2013, 8, e64465. [Google Scholar]
- Gaynor, A.M.; Nissen, M.D.; Whiley, D.M.; Mackay, I.M.; Lambert, S.B.; Wu, G.; Brennan, D.C.; Storch, G.A.; Sloots, T.P.; Wang, D. Identification of a novel polyomavirus from patients with acute respiratory tract infections. PLoS Pathog. 2007, 3, e64. [Google Scholar] [CrossRef]
- Kostic, A.D.; Ojesina, A.I.; Pedamallu, C.S.; Jung, J.; Verhaak, R.G.; Getz, G.; Meyerson, M. PathSeq: Software to identify or discover microbes by deep sequencing of human tissue. Nat. Biotechnol. 2011, 29, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Holtz, L.R.; Finkbeiner, S.R.; Zhao, G.; Kirkwood, C.D.; Girones, R.; Pipas, J.M.; Wang, D. Klassevirus 1, a previously undescribed member of the family Picornaviridae, is globally widespread. Virol. J. 2009, 6, 86. [Google Scholar] [CrossRef] [PubMed]
- Borozan, I.; Watt, S.N.; Ferretti, V. Evaluation of alignment algorithms for discovery and identification of pathogens using RNA-Seq. PLoS One 2013, 8, e76935. [Google Scholar] [CrossRef]
- Adams, M.J.; Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2012). Arch. Virol. 2012, 157, 1411–1422. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Jahrling, P.B.; Kawaoka, Y.; Netesov, S.V.; Nichol, S.T.; Peters, C.J.; Volchkov, V.E.; et al. Family Filoviridae. In Virus Taxonomy—Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Elsevier/Academic Press: London, UK, 2011; pp. 665–671. [Google Scholar]
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Johnson, K.M.; Kawaoka, Y.; Lipkin, W.I.; Negredo, A.I.; Netesov, S.V.; Nichol, S.T.; et al. Proposal for a revised taxonomy of the family Filoviridae: Classification, names of taxa and viruses, and virus abbreviations. Arch. Virol. 2010, 155, 2083–2103. [Google Scholar]
- Bukreyev, A.A.; Chandran, K.; Dolnik, O.; Dye, J.M.; Ebihara, H.; Leroy, E.M.; Mühlberger, E.; Netesov, S.V.; Patterson, J.L.; Paweska, J.T.; et al. Discussions and decisions of the 2012–2014 International Committee on Taxonomy of Viruses (ICTV) Filoviridae Study Group, January 2012–June 2013. Arch. Virol. 2013, 159, 821–830. [Google Scholar]
- Adams, M.J.; Lefkowitz, E.J.; King, A.M.; Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2014). Arch. Virol. 2014, 159, 2831–2841. [Google Scholar] [CrossRef] [PubMed]
- Bao, Y.; Chetvernin, V.; Tatusova, T. PAirwise Sequence Comparison (PASC) and Its Application in the Classification of Filoviruses. Viruses 2012, 4, 1318–1327. [Google Scholar] [CrossRef] [PubMed]
- Lauber, C.; Gorbalenya, A.E. Genetics-based classification of filoviruses calls for expanded sampling of genomic sequences. Viruses 2012, 4, 1425–1437. [Google Scholar] [CrossRef] [PubMed]
- Towner, J.S.; Sealy, T.K.; Khristova, M.L.; Albariño, C.G.; Conlan, S.; Reeder, S.A.; Quan, P.L.; Lipkin, W.I.; Downing, R.; Tappero, J.W.; et al. Newly discovered Ebola virus associated with hemorrhagic fever outbreak in Uganda. PLoS Pathog. 2008, 4, e1000212. [Google Scholar]
- Negredo, A.; Palacios, G.; Vazquez-Morón, S.; González, F.; Dopazo, H.; Molero, F.; Juste, J.; Quetglas, J.; Savji, N.; de la Cruz Martínez, M.; et al. Discovery of an ebolavirus-like filovirus in europe. PLoS Pathog. 2011, 7, e1002304. [Google Scholar]
- Kuhn, J.H.; Bao, Y.; Bavari, S.; Becker, S.; Bradfute, S.; Brister, J.R.; Bukreyev, A.A.; Chandran, K.; Davey, R.A.; Dolnik, O.; et al. Virus nomenclature below the species level: A standardized nomenclature for natural variants of viruses assigned to the family Filoviridae. Arch. Virol. 2013, 158, 301–311. [Google Scholar]
- Maruyama, J.; Miyamoto, H.; Kajihara, M.; Ogawa, H.; Maeda, K.; Sakoda, Y.; Yoshida, R.; Takada, A. Characterization of the envelope glycoprotein of a novel filovirus, Lloviu virus. J. Virol. 2014, 88, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Ladner, J.T.; Beitzel, B.; Chain, P.S.; Davenport, M.G.; Donaldson, E.F.; Frieman, M.; Kugelman, J.R.; Kuhn, J.H.; O'Rear, J.; Sabeti, P.C.; et al. Standards for sequencing viral genomes in the era of high-throughput sequencing. MBio 2014, 5, e01360–14. [Google Scholar]
- Albariño, C.G.; Shoemaker, T.; Khristova, M.L.; Wamala, J.F.; Muyembe, J.J.; Balinandi, S.; Tumusiime, A.; Campbell, S.; Cannon, D.; Gibbons, A.; et al. Genomic analysis of filoviruses associated with four viral hemorrhagic fever outbreaks in Uganda and the Democratic Republic of the Congo in 2012. Virology 2013, 442, 97–100. [Google Scholar]
- Kuhn, J.H. Filoviruses. A compendium of 40 years of Epidemiological, Clinical, and Laboratory Studies Archives of Virology Supplementum; SpringerWienNewYork: Vienna, Austria, 2008. [Google Scholar]
- Kuhn, J.H.; Bao, Y.; Bavari, S.; Becker, S.; Bradfute, S.; Brauburger, K.; Rodney Brister, J.; Bukreyev, A.A.; Caì, Y.; Chandran, K.; et al. Virus nomenclature below the species level: A standardized nomenclature for filovirus strains and variants rescued from cDNA. Arch. Virol. 2014, 159, 1229–1237. [Google Scholar]
- Kuhn, J.H.; Bao, Y.; Bavari, S.; Becker, S.; Bradfute, S.; Brister, J.R.; Bukreyev, A.A.; Caì, Y.; Chandran, K.; Davey, R.A.; et al. Virus nomenclature below the species level: A standardized nomenclature for laboratory animal-adapted strains and variants of viruses assigned to the family Filoviridae. Arch. Virol. 2013, 158, 1425–1432. [Google Scholar]
- Brown, C.S.; Lee, M.S.; Leung, D.W.; Wang, T.; Xu, W.; Luthra, P.; Anantpadma, M.; Shabman, R.S.; Melito, L.M.; Macmillan, K.S.; et al. In Silico Derived Small Molecules Bind the Filovirus VP35 Protein and Inhibit Its Polymerase Cofactor Activity. J. Mol. Biol. 2014, 426, 2045–2058. [Google Scholar]
- Binning, J.M.; Wang, T.; Luthra, P.; Shabman, R.S.; Borek, D.M.; Liu, G.; Xu, W.; Leung, D.W.; Basler, C.F.; Amarasinghe, G.K. Development of RNA Aptamers Targeting Ebola Virus VP35. Biochemistry 2013, 52, 8406–8419. [Google Scholar] [CrossRef] [PubMed]
- Prins, K.C.; Delpeut, S.; Leung, D.W.; Reynard, O.; Volchkova, V.A.; Reid, S.P.; Ramanan, P.; Cárdenas, W.B.; Amarasinghe, G.K.; Volchkov, V.E.; et al. Mutations abrogating VP35 interaction with double-stranded RNA render Ebola virus avirulent in guinea pigs. J. Virol. 2010, 84, 3004–3015. [Google Scholar]
- Leung, D.W.; Prins, K.C.; Borek, D.M.; Farahbakhsh, M.; Tufariello, J.M.; Ramanan, P.; Nix, J.C.; Helgeson, L.A.; Otwinowski, Z.; Honzatko, R.B.; et al. Structural basis for dsRNA recognition and interferon antagonism by Ebola VP35. Nat. Struct. Mol. Biol. 2010, 17, 165–172. [Google Scholar]
- Leung, D.W.; Ginder, N.D.; Fulton, D.B.; Nix, J.; Basler, C.F.; Honzatko, R.B.; Amarasinghe, G.K. Structure of the Ebola VP35 interferon inhibitory domain. Proc. Natl. Acad. Sci. USA 2009, 106, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Malashkevich, V.N.; Schneider, B.J.; McNally, M.L.; Milhollen, M.A.; Pang, J.X.; Kim, P.S. Core structure of the envelope glycoprotein GP2 from Ebola virus at 1.9-Å resolution. Proc. Natl. Acad. Sci. USA 1999, 96, 2662–2667. [Google Scholar] [CrossRef]
- Lee, J.E.; Fusco, M.L.; Hessell, A.J.; Oswald, W.B.; Burton, D.R.; Saphire, E.O. Structure of the Ebola virus glycoprotein bound to an antibody from a human survivor. Nature 2008, 454, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Bornholdt, Z.A.; Noda, T.; Abelson, D.M.; Halfmann, P.; Wood, M.R.; Kawaoka, Y.; Saphire, E.O. Structural rearrangement of Ebola virus VP40 begets multiple functions in the virus life cycle. Cell 2013, 154, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Hartlieb, B.; Muziol, T.; Weissenhorn, W.; Becker, S. Crystal structure of the C-terminal domain of Ebola virus VP30 reveals a role in transcription and nucleocapsid association. Proc. Natl. Acad. Sci. USA 2007, 104, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Ebola haemorrhagic fever in Zaire, 1976. Bull. World Health Organ. 1978, 56, 271–293.
- Garrett, L. Yambuku—Ebola. In The Coming Plague—Newly Emerging Disease in a World out of Balance; Garrett, L., Ed.; Farrar, Straus & Giroux: New York, USA, 1994; pp. 100–152. [Google Scholar]
- Miranda, M.E.; Miranda, N.L. Reston ebolavirus in Humans and Animals in the Philippines: A Review. J. Infect. Dis. 2011, 204, S757–S760. [Google Scholar] [CrossRef]
- Groseth, A.; Marzi, A.; Hoenen, T.; Herwig, A.; Gardner, D.; Becker, S.; Ebihara, H.; Feldmann, H. The Ebola virus glycoprotein contributes to but is not sufficient for virulence in vivo. PLoS Pathog. 2012, 8, e1002847. [Google Scholar] [CrossRef]
- Kimberlin, C.R.; Bornholdt, Z.A.; Li, S.; Woods, V.L., Jr.; MacRae, I.J.; Saphire, E.O. Ebolavirus VP35 uses a bimodal strategy to bind dsRNA for innate immune suppression. Proc. Natl. Acad. Sci. USA 2010, 107, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.W.; Shabman, R.S.; Farahbakhsh, M.; Prins, K.C.; Borek, D.M.; Wang, T.; Mühlberger, E.; Basler, C.F.; Amarasinghe, G.K. Structural and functional characterization of Reston Ebola virus VP35 interferon inhibitory domain. J. Mol. Biol. 2010, 399, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Clifton, M.C.; Kirchdoerfer, R.N.; Atkins, K.; Abendroth, J.; Raymond, A.; Grice, R.; Barnes, S.; Moen, S.; Lorimer, D.; Edwards, T.E.; et al. Structure of the Reston ebolavirus VP30 C-terminal domain. Acta Crystallogr. F Struct. Biol. Commun. 2014, 70, 457–460. [Google Scholar] [CrossRef]
- Zhang, A.P.; Bornholdt, Z.A.; Liu, T.; Abelson, D.M.; Lee, D.E.; Li, S.; Woods, V.L., Jr.; Saphire, E.O. The Ebola Virus Interferon Antagonist VP24 Directly Binds STAT1 and Has a Novel, Pyramidal Fold. PLoS Pathog. 2012, 8, e1002550. [Google Scholar] [CrossRef]
- Bale, S.; Julien, J.P.; Bornholdt, Z.A.; Krois, A.S.; Wilson, I.A.; Saphire, E.O. Ebolavirus VP35 coats the backbone of double-stranded RNA for interferon antagonism. J. Virol. 2013, 87, 10385–10388. [Google Scholar] [CrossRef] [PubMed]
- Bale, S.; Dias, J.M.; Fusco, M.L.; Hashiguchi, T.; Wong, A.C.; Liu, T.; Keuhne, A.I.; Li, S.; Woods, V.L., Jr.; Chandran, K.; et al. Structural basis for differential neutralization of ebolaviruses. Viruses 2012, 4, 447–470. [Google Scholar]
- Dias, J.M.; Kuehne, A.I.; Abelson, D.M.; Bale, S.; Wong, A.C.; Halfmann, P.; Muhammad, M.A.; Fusco, M.L.; Zak, S.E.; Kang, E.; et al. A shared structural solution for neutralizing ebolaviruses. Nat. Struct. Mol. Biol. 2011, 18, 1424–1427. [Google Scholar] [CrossRef]
- Le Guenno, B.; Formenty, P.; Wyers, M.; Gounon, P.; Walker, F.; Boesch, C. Isolation and partial characterisation of a new strain of Ebola virus. Lancet 1995, 345, 1271–1274. [Google Scholar]
- Albariño, C.G.; Uebelhoer, L.S.; Vincent, J.P.; Khristova, M.L.; Chakrabarti, A.K.; McElroy, A.; Nichol, S.T.; Towner, J.S. Development of a reverse genetics system to generate recombinant Marburg virus derived from a bat isolate. Virology 2013, 446, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Johnson, B.K.; Isaacson, M.; Swanapoel, R.; Johnson, K.M.; Killey, M.; Bagshawe, A.; Siongok, T.; Keruga, W.K. Marburg-virus disease in Kenya. Lancet 1982, 1, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Preston, R. The Hot Zone—A Terrifying New Story; Random House: New York, NY, USA, 1994. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kuhn, J.H.; Andersen, K.G.; Bào, Y.; Bavari, S.; Becker, S.; Bennett, R.S.; Bergman, N.H.; Blinkova, O.; Bradfute, S.; Brister, J.R.; et al. Filovirus RefSeq Entries: Evaluation and Selection of Filovirus Type Variants, Type Sequences, and Names. Viruses 2014, 6, 3663-3682. https://doi.org/10.3390/v6093663
Kuhn JH, Andersen KG, Bào Y, Bavari S, Becker S, Bennett RS, Bergman NH, Blinkova O, Bradfute S, Brister JR, et al. Filovirus RefSeq Entries: Evaluation and Selection of Filovirus Type Variants, Type Sequences, and Names. Viruses. 2014; 6(9):3663-3682. https://doi.org/10.3390/v6093663
Chicago/Turabian StyleKuhn, Jens H., Kristian G. Andersen, Yīmíng Bào, Sina Bavari, Stephan Becker, Richard S. Bennett, Nicholas H. Bergman, Olga Blinkova, Steven Bradfute, J. Rodney Brister, and et al. 2014. "Filovirus RefSeq Entries: Evaluation and Selection of Filovirus Type Variants, Type Sequences, and Names" Viruses 6, no. 9: 3663-3682. https://doi.org/10.3390/v6093663
APA StyleKuhn, J. H., Andersen, K. G., Bào, Y., Bavari, S., Becker, S., Bennett, R. S., Bergman, N. H., Blinkova, O., Bradfute, S., Brister, J. R., Bukreyev, A., Chandran, K., Chepurnov, A. A., Davey, R. A., Dietzgen, R. G., Doggett, N. A., Dolnik, O., Dye, J. M., Enterlein, S., ... Nichol, S. T. (2014). Filovirus RefSeq Entries: Evaluation and Selection of Filovirus Type Variants, Type Sequences, and Names. Viruses, 6(9), 3663-3682. https://doi.org/10.3390/v6093663