Filoviruses in Bats: Current Knowledge and Future Directions
Abstract
:1. Introduction and Background
1.1. Basic Virology
1.2. Viral Taxonomy and Phylogeny
1.3. Filovirus Outbreaks in Humans—Brief History Including Known Links to Bat Exposure
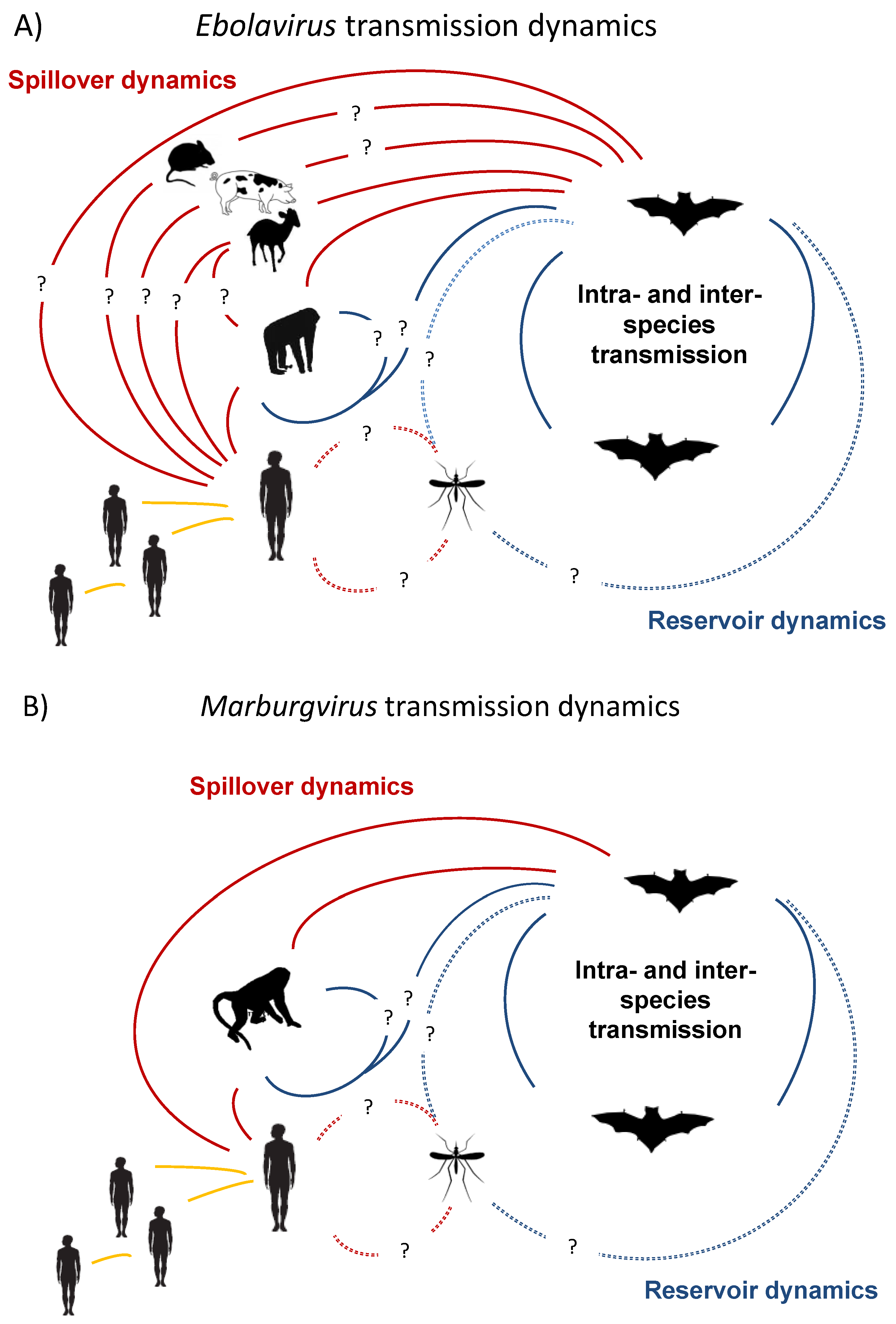
2. Natural Reservoirs
2.1. Investigations to Find the Natural Reservoir—Elusive for Decades and Ongoing
2.2. Role of Primates—Potential Reservoirs or Dead-End Hosts?
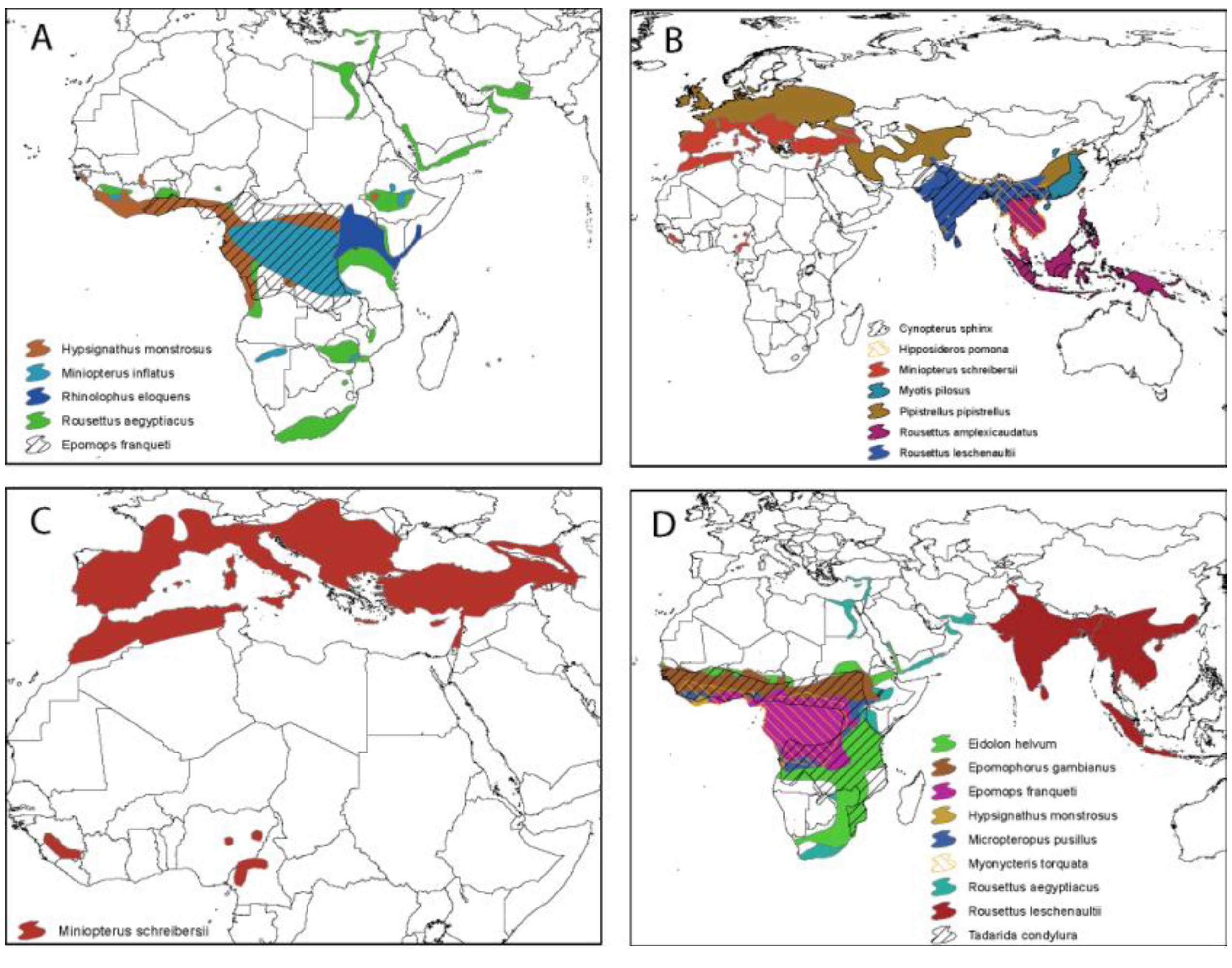
2.3. Evidence of Bats as Key Reservoirs—Ebola Viruses and Marburg in Africa
| Virus | Bat Species | Detection Method | References |
|---|---|---|---|
| Marburgvirus | Epomops franqueti | Antibodies | [77] |
| Hypsignathus monstrosus | Antibodies | [77] | |
| Miniopterus inflatus | Antibodies; PCR | [18,77] | |
| Rhinolophus eloquens | Antibodies; PCR | [18] | |
| Rousettus aegyptiacus | Antibodies; PCR; Viral Isolation | [18,19,28,77,78,79] | |
| Lloviu virus | Miniopterus schreibersii | PCR; HTS | [16] |
| Reston ebolavirus | Cynopterus sphinx | Antibodies | [80] |
| Hipposideros pomona | Antibodies | [80] | |
| Miniopterus schreibersii | Antibodies | [80] | |
| Myotis pilosus (=Myotis ricketti) | Antibodies | [80] | |
| Pipistrellus pipistrellus | Antibodies | [80] | |
| Rousettus amplexicaudatus | Antibodies | [64] | |
| Rousettus leschenaultii | Antibodies | [63,80] | |
| Zaire ebolavirus | Eidolon helvum | Antibodies | [76] |
| Epomops franqueti | Antibodies; PCR | [30,75,77,81] | |
| Epomophorus gambianus | Antibodies | [75] | |
| Hypsignathus monstrosus | Antibodies; PCR | [30,75,77,81] | |
| Micropteropus pusillus | Antibodies | [77] | |
| Tadarida condylura (=Mops condylurus) | Antibodies | [77] | |
| Myonycteris torquata | Antibodies; PCR | [30,77,81] | |
| Rousettus aegyptiacus | Antibodies | [77] | |
| Rousettus leschenaultii | Antibodies | [63] |
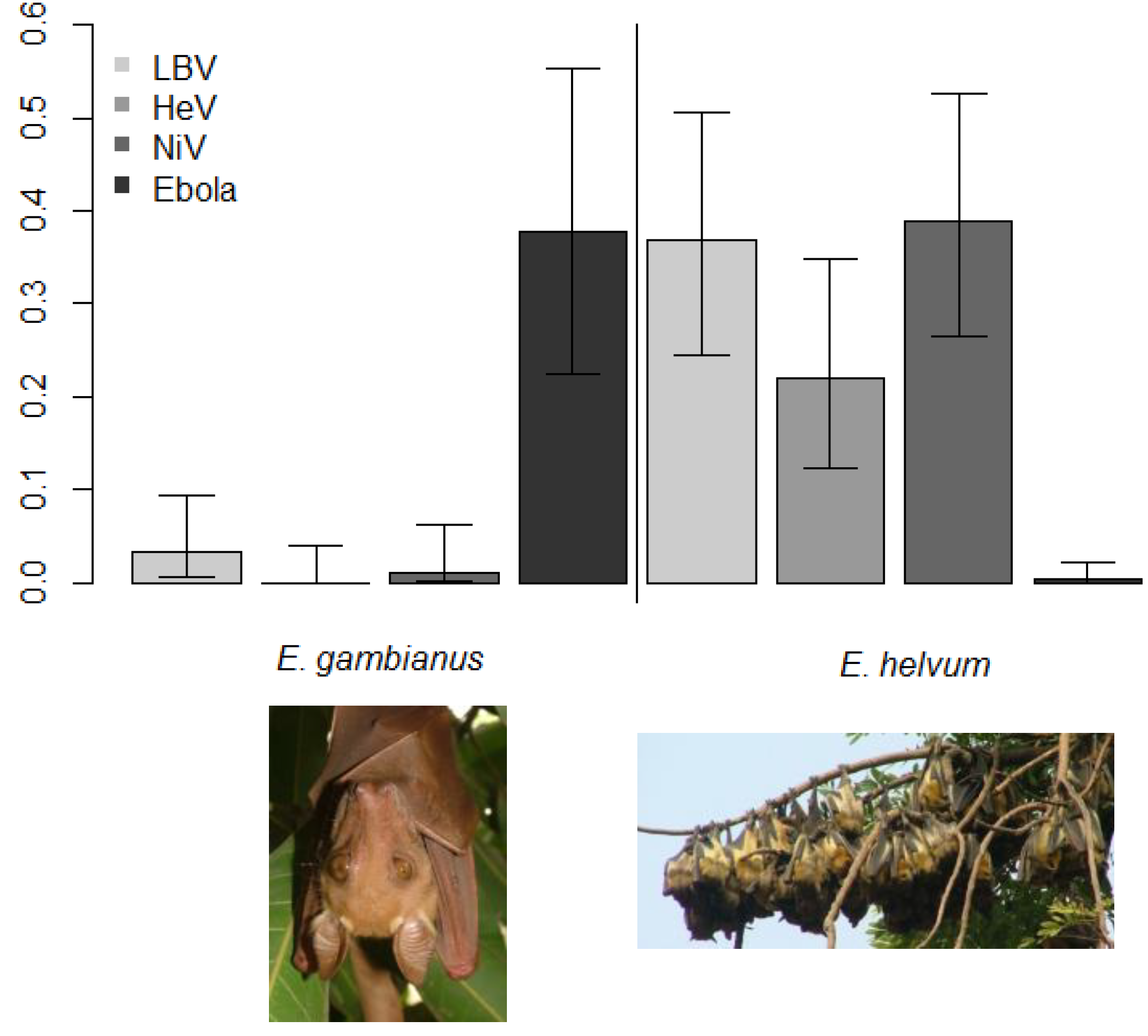
2.4. Evidence of Filoviruses from Bats in Asia and Europe
2.5. Experimental Research Supporting Bats as Reservoirs
3. Filovirus Dynamics and Ecology in Bats—What We Know and Don’t Know
3.1. Lessons to Learn from Other Bat Zoonoses
3.2. Seasonality of Infection Dynamics in Bats
3.3. Viral Shedding and Immunity in Bats
3.4. Multi-Host and Multi-Pathogen Dynamics in Bats
3.5. Meta Populations and Connectivity
4. Future Directions in Bat Filovirus Research
4.1. Unexplored Diversity and Geographic Gaps—A More Unified Surveillance Strategy
4.2. Develop More Sensitive, Non-Invasive Tools for Longitudinal Monitoring of Bat Populations
4.3. Genomics and Viral Fossils
4.4. Better Understanding Viral Shedding and Transmission in Bats
4.5. Better Understanding Host Ecology and Spillover Potential to Humans
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Leroy, E.M.; Kumulungui, B.; Pourrut, X.; Rouquet, P.; Hassanin, A.; Yaba, P.; Delicat, A.; Paweska, J.T.; Gonzalez, J.P.; Swanepoel, R. Fruit bats as reservoirs of Ebola virus. Nature 2005, 438, 575–576. [Google Scholar] [CrossRef]
- Kobinger, G.P.; Croyle, M.; Feldmann, H. Ebola and Marburg. In Vaccines for Biodefense and Emerging and Neglected Diseases; Barrett, A.D.T., Stanberry, L.R., Eds.; Academic Press: London, UK, 2009; pp. 325–337. [Google Scholar]
- Feldmann, H.; Geisbert, T.W. Ebola haemorrhagic fever. Lancet 2011, 377, 849–862. [Google Scholar] [CrossRef]
- Mohamadzadeh, M.; Chen, L.; Schmaljohn, A.L. How Ebola and Marburg viruses battle the immune system. Nat. Rev. Immunol. 2007, 7, 556–567. [Google Scholar] [CrossRef]
- MDPI Viruses Special Issue "Advances in Filovirus Research 2012". Available online: https://www.mdpi.com/journal/viruses/special_issues/Filovirus/ (accessed on 1 March 2014).
- Feldmann, H.; Jones, S.; Klenk, H.D.; Schnittler, H.J. Ebola virus: From discovery to vaccine. Nat. Rev. Immunol. 2003, 3, 677–685. [Google Scholar] [CrossRef]
- Kiley, M.P.; Bowen, E.T.; Eddy, G.A.; Isaacson, M.; Johnson, K.M.; McCormick, J.B.; Murphy, F.A.; Pattyn, S.R.; Peters, D.; Prozesky, O.W.; et al. Filoviridae: A taxonomic home for Marburg and Ebola viruses? Intervirology 1982, 18, 24–32. [Google Scholar] [CrossRef]
- Carroll, S.A.; Towner, J.S.; Sealy, T.K.; McMullan, L.K.; Khristova, M.L.; Burt, F.J.; Swanepoel, R.; Rollin, P.E.; Nichol, S.T. Molecular evolution of viruses of the family Filoviridae based on 97 whole-genome sequences. J. Virol. 2013, 87, 2608–2616. [Google Scholar] [CrossRef]
- Sanchez, A.; Geisbert, T.W.; Feldmann, H. Filoviridae: Marburg and Ebola Viruses. In Fields virology; Knipe, D.M., Howley, P.M., Eds.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2007; pp. 1279–1304. [Google Scholar]
- Feldmann, H.; Kiley, M.P. Classification, structure, and replication of filoviruses. Curr. Top. Microbiol. Immunol. 1999, 235, 1–21. [Google Scholar]
- Brauburger, K.; Hume, A.J.; Muhlberger, E.; Olejnik, J. Forty-Five Years of Marburg Virus Research. Viruses 2012, 4, 1878–1927. [Google Scholar] [CrossRef]
- Taylor, D.J.; Dittmar, K.; Ballinger, M.J.; Bruenn, J.A. Evolutionary maintenance of filovirus-like genes in bat genomes. BMC Evol. Biol. 2011, 11, 336. [Google Scholar] [CrossRef]
- Feldmann, H.; Volchkov, V.E.; Volchkova, V.A.; Stroher, U.; Klenk, H.D. Biosynthesis and role of filoviral glycoproteins. J. Gen. Virol. 2001, 82, 2839–2848. [Google Scholar]
- Kuhn, J.H.; Bao, Y.; Bavari, S.; Becker, S.; Bradfute, S.; Brauburger, K.; Rodney Brister, J.; Bukreyev, A.A.; Cai, Y.; Chandran, K.; et al. Virus nomenclature below the species level: A standardized nomenclature for filovirus strains and variants rescued from cDNA. Arch. Virol. 2013. [Google Scholar] [CrossRef]
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Johnson, K.M.; Kawaoka, Y.; Lipkin, W.I.; Negredo, A.I.; Netesov, S.V.; Nichol, S.T.; et al. Proposal for a revised taxonomy of the family Filoviridae: Classification, names of taxa and viruses, and virus abbreviations. Arch. Virol. 2010, 155, 2083–2103. [Google Scholar] [CrossRef]
- Negredo, A.; Palacios, G.; Vazquez-Moron, S.; Gonzalez, F.; Dopazo, H.; Molero, F.; Juste, J.; Quetglas, J.; Savji, N.; de la Cruz Martinez, M.; et al. Discovery of an ebolavirus-like filovirus in europe. PLoS Pathog. 2011, 7, e1002304. [Google Scholar] [CrossRef]
- Towner, J.S.; Khristova, M.L.; Sealy, T.K.; Vincent, M.J.; Erickson, B.R.; Bawiec, D.A.; Hartman, A.L.; Comer, J.A.; Zaki, S.R.; Stroher, U.; et al. Marburgvirus genomics and association with a large hemorrhagic fever outbreak in Angola. J. Virol. 2006, 80, 6497–6516. [Google Scholar] [CrossRef]
- Swanepoel, R.; Smit, S.B.; Rollin, P.E.; Formenty, P.; Leman, P.A.; Kemp, A.; Burt, F.J.; Grobbelaar, A.A.; Croft, J.; Bausch, D.G.; et al. Studies of reservoir hosts for Marburg virus. Emerg. Infect. Dis. 2007, 13, 1847–1851. [Google Scholar] [CrossRef]
- Towner, J.S.; Amman, B.R.; Sealy, T.K.; Carroll, S.A.R.; Comer, J.A.; Kemp, A.; Swanepoel, R.; Paddock, C.D.; Balinandi, S.; Khristova, M.L.; et al. Isolation of Genetically Diverse Marburg Viruses from Egyptian Fruit Bats. PLoS Pathog. 2009, 5. [Google Scholar] [CrossRef]
- Lauber, C.; Gorbalenya, A.E. Genetics-Based Classification of Filoviruses Calls for Expanded Sampling of Genomic Sequences. Viruses 2012, 4, 1425–1437. [Google Scholar] [CrossRef]
- Bao, Y.; Chetvernin, V.; Tatusova, T. PAirwise Sequence Comparison (PASC) and Its Application in the Classification of Filoviruses. Viruses 2012, 4, 1318–1327. [Google Scholar] [CrossRef]
- Suzuki, Y.; Gojobori, T. The origin and evolution of Ebola and Marburg viruses. Mol. Biol. Evol. 1997, 14, 800–806. [Google Scholar] [CrossRef]
- Taylor, D.J.; Leach, R.W.; Bruenn, J. Filoviruses are ancient and integrated into mammalian genomes. BMC Evol. Biol. 2010, 10, 193. [Google Scholar] [CrossRef]
- Biek, R.; Walsh, P.D.; Leroy, E.M.; Real, L.A. Recent common ancestry of Ebola Zaire virus found in a bat reservoir. PLoS Pathog. 2006, 2, e90. [Google Scholar] [CrossRef]
- Grard, G.; Biek, R.; Tamfum, J.J.; Fair, J.; Wolfe, N.; Formenty, P.; Paweska, J.; Leroy, E. Emergence of divergent Zaire ebola virus strains in Democratic Republic of the Congo in 2007 and 2008. J. Infect. Dis. 2011, 204 (Suppl. 3), S776–S784. [Google Scholar] [CrossRef]
- Walsh, P.D.; Biek, R.; Real, L.A. Wave-like spread of Ebola Zaire. PLoS Biol. 2005, 3, e371. [Google Scholar] [CrossRef]
- Wittmann, T.J.; Biek, R.; Hassanin, A.; Rouquet, P.; Reed, P.; Yaba, P.; Pourrut, X.; Real, L.A.; Gonzalez, J.P.; Leroy, E.M. Isolates of Zaire ebolavirus from wild apes reveal genetic lineage and recombinants. Proc. Natl. Acad. Sci. USA 2007, 104, 17123–17127. [Google Scholar] [CrossRef]
- Amman, B.R.; Carroll, S.A.; Reed, Z.D.; Sealy, T.K.; Balinandi, S.; Swanepoel, R.; Kemp, A.; Erickson, B.R.; Comer, J.A.; Campbell, S.; et al. Seasonal Pulses of Marburg Virus Circulation in Juvenile Rousettus aegyptiacus Bats Coincide with Periods of Increased Risk of Human Infection. PLoS Pathog. 2012, 8. [Google Scholar] [CrossRef]
- Bausch, D.G.; Borchert, M.; Grein, T.; Roth, C.; Swanepoel, R.; Libande, M.L.; Talarmin, A.; Bertherat, E.; Muyembe-Tamfum, J.J.; Tugume, B.; et al. Risk factors for Marburg hemorrhagic fever, Democratic Republic of the Congo. Emerg. Infect. Dis. 2003, 9, 1531–1537. [Google Scholar] [CrossRef]
- Pourrut, X.; Kumulungui, B.; Wittmann, T.; Moussavou, G.; Delicat, A.; Yaba, P.; Nkoghe, D.; Gonzalez, J.P.; Leroy, E.M. The natural history of Ebola virus in Africa. Microbes Infect./Inst. Pasteur 2005, 7, 1005–1014. [Google Scholar] [CrossRef]
- Le Guenno, B.; Formenty, P.; Wyers, M.; Gounon, P.; Walker, F.; Boesch, C. Isolation and partial characterisation of a new strain of Ebola virus. Lancet 1995, 345, 1271–1274. [Google Scholar] [CrossRef]
- Towner, J.S.; Sealy, T.K.; Khristova, M.L.; Albarino, C.G.; Conlan, S.; Reeder, S.A.; Quan, P.L.; Lipkin, W.I.; Downing, R.; Tappero, J.W.; et al. Newly Discovered Ebola Virus Associated with Hemorrhagic Fever Outbreak in Uganda. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef]
- Leroy, E.M.; Epelboin, A.; Mondonge, V.; Pourrut, X.; Gonzalez, J.P.; Muyembe-Tamfum, J.J.; Formenty, P. Human Ebola Outbreak Resulting from Direct Exposure to Fruit Bats in Luebo, Democratic Republic of Congo, 2007. Vector-Borne Zoonotic Dis. 2009, 9, 723–728. [Google Scholar] [CrossRef]
- Albarino, C.G.; Shoemaker, T.; Khristova, M.L.; Wamala, J.F.; Muyembe, J.J.; Balinandi, S.; Tumusiime, A.; Campbell, S.; Cannon, D.; Gibbons, A.; et al. Genomic analysis of filoviruses associated with four viral hemorrhagic fever outbreaks in Uganda and the Democratic Republic of the Congo in 2012. Virology 2013, 442, 97–100. [Google Scholar] [CrossRef]
- W.H.O. Ebola Haemorrhagic Fever, Guinea. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4069-ebola-haemorrhagic-fever-guinea-27-march-2014.html/ (accessed on 27 March 2014).
- Miranda, M.E.; Ksiazek, T.G.; Retuya, T.J.; Khan, A.S.; Sanchez, A.; Fulhorst, C.F.; Rollin, P.E.; Calaor, A.B.; Manalo, D.L.; et al. Epidemiology of Ebola (subtype Reston) virus in the Philippines, 1996. J. Infect. Dis. 1999, 179 (Suppl. 1), S115–S119. [Google Scholar] [CrossRef]
- Miranda, M.E.; Yoshikawa, Y.; Manalo, D.L.; Calaor, A.B.; Miranda, N.L.; Cho, F.; Ikegami, T.; Ksiazek, T.G. Chronological and spatial analysis of the 1996 Ebola Reston virus outbreak in a monkey breeding facility in the Philippines. Exp. Anim. 2002, 51, 173–179. [Google Scholar] [CrossRef]
- Miranda, M.E.G.; Miranda, N.L.J. Reston ebolavirus in Humans and Animals in the Philippines: A Review. J. Infect. Dis. 2011, 204, S757–S760. [Google Scholar] [CrossRef]
- Barrette, R.W.; Metwally, S.A.; Rowland, J.M.; Xu, L.Z.; Zaki, S.R.; Nichol, S.T.; Rollin, P.E.; Towner, J.S.; Shieh, W.J.; Batten, B.; et al. Discovery of Swine as a Host for the Reston ebolavirus. Science 2009, 325, 204–206. [Google Scholar] [CrossRef]
- Bausch, D.G.; Nichol, S.T.; Muyembe-Tamfum, J.J.; Borchert, M.; Rollin, P.E.; Sleurs, H.; Campbell, P.; Tshioko, F.K.; Roth, C.; Colebunders, R.; et al. Marburg hemorrhagic fever associated with multiple genetic lineages of virus. N. Engl. J. Med. 2006, 355, 909–919. [Google Scholar] [CrossRef]
- Olival, K.J.; Epstein, J.H.; Wang, L.F.; Field, H.E.; Daszak, P. Are bats unique viral reservoirs? In New Directions in Conservation Medicine: Applied Cases of Ecological Health, 2nd ed.; Aguirre, A.A., Ostfeld, R.S., Daszak, P., Eds.; Oxford University Press: Oxford, UK, 2012; pp. 195–212. [Google Scholar]
- Germain, M. Collection of mammals and arthropods during the epidemic of haemorrhagic fever in Zaire. In Ebola Virus Haemorrhagic Fever; Pattyn, S.R., Ed.; Elsevier: New York, NY, USA, 1978; pp. 185–189. [Google Scholar]
- Arata, A.A.; Johnson, B. Approaches toward studies on potential reservoirs of viral hemorrhagic fever in southern Sudan (1977). In Ebola Virus Haemorrhagic Fever; Pattyn, S.R., Ed.; Elsevier: New York, NY, USA, 1978; pp. 191–200. [Google Scholar]
- Leirs, H.; Mills, J.N.; Krebs, J.W.; Childs, J.E.; Akaibe, D.; Woollen, N.; Ludwig, G.; Peters, C.J.; Ksiazek, T.G. Search for the Ebola virus reservoir in Kikwit, Democratic Republic of the Congo: Reflections on a vertebrate collection. J. Infect. Dis. 1999, 179 (Suppl. 1), S155–S163. [Google Scholar]
- Swanepoel, R.; Leman, P.A.; Burt, F.J.; Zachariades, N.A.; Braack, L.E.; Ksiazek, T.G.; Rollin, P.E.; Zaki, S.R.; Peters, C.J. Experimental inoculation of plants and animals with Ebola virus. Emerg. Infect. Dis. 1996, 2, 321–325. [Google Scholar] [CrossRef]
- Turell, M.J.; Bressler, D.S.; Rossi, C.A. Short report: Lack of virus replication in arthropods after intrathoracic inoculation of Ebola Reston virus. Am. J. Trop. Med. Hyg. 1996, 55, 89–90. [Google Scholar]
- Kissling, R.E.; Robinson, R.Q.; Murphy, F.A.; Whitfield, S. Green monkey agent of disease. Science 1968, 161, 1364. [Google Scholar]
- Smith, C.E.; Simpson, D.I.; Bowen, E.T.; Zlotnik, I. Fatal human disease from vervet monkeys. Lancet 1967, 2, 1119–1121. [Google Scholar]
- Jahrling, P.B.; Geisbert, T.W.; Dalgard, D.W.; Johnson, E.D.; Ksiazek, T.G.; Hall, W.C.; Peters, C.J. Preliminary report: Isolation of Ebola virus from monkeys imported to USA. Lancet 1990, 335, 502–505. [Google Scholar] [CrossRef]
- Leroy, E.M.; Rouquet, P.; Formenty, P.; Souquiere, S.; Kilbourne, A.; Froment, J.M.; Bermejo, M.; Smit, S.; Karesh, W.; Swanepoel, R.; et al. Multiple Ebola virus transmission events and rapid decline of central African wildlife. Science 2004, 303, 387–390. [Google Scholar] [CrossRef]
- Smith, D.H.; Johnson, B.K.; Isaacson, M.; Swanapoel, R.; Johnson, K.M.; Killey, M.; Bagshawe, A.; Siongok, T.; Keruga, W.K. Marburg-virus disease in Kenya. Lancet 1982, 1, 816–820. [Google Scholar]
- Bermejo, M.; Rodriguez-Teijeiro, J.D.; Illera, G.; Barroso, A.; Vila, C.; Walsh, P.D. Ebola outbreak killed 5000 gorillas. Science 2006, 314, 1564. [Google Scholar] [CrossRef]
- Walsh, P.D.; Abernethy, K.A.; Bermejo, M.; Beyers, R.; de Wachter, P.; Akou, M.E.; Huijbregts, B.; Mambounga, D.I.; Toham, A.K.; Kilbourn, A.M.; et al. Catastrophic ape decline in western equatorial Africa. Nature 2003, 422, 611–614. [Google Scholar] [CrossRef]
- Nidom, C.A.; Nakayama, E.; Nidom, R.V.; Alamudi, M.Y.; Daulay, S.; Dharmayanti, I.N.; Dachlan, Y.P.; Amin, M.; Igarashi, M.; Miyamoto, H.; et al. Serological evidence of Ebola virus infection in Indonesian orangutans. PLoS One 2012, 7, e40740. [Google Scholar] [CrossRef]
- Gilbert, A.T.; Fooks, A.R.; Hayman, D.T.; Horton, D.L.; Muller, T.; Plowright, R.; Peel, A.J.; Bowen, R.; Wood, J.L.; Mills, J.; et al. Deciphering serology to understand the ecology of infectious diseases in wildlife. Ecohealth 2013, 10, 298–313. [Google Scholar] [CrossRef]
- Hayes, C.G.; Burans, J.P.; Ksiazek, T.G.; Del Rosario, R.A.; Miranda, M.E.; Manaloto, C.R.; Barrientos, A.B.; Robles, C.G.; Dayrit, M.M.; Peters, C.J. Outbreak of fatal illness among captive macaques in the Philippines caused by an Ebola-related filovirus. Am. J. Trop. Med. Hyg. 1992, 46, 664–671. [Google Scholar]
- Miranda, M.E.; White, M.E.; Dayrit, M.M.; Hayes, C.G.; Ksiazek, T.G.; Burans, J.P. Seroepidemiological study of filovirus related to Ebola in the Philippines. Lancet 1991, 337, 425–426. [Google Scholar]
- Rollin, P.E.; Williams, R.J.; Bressler, D.S.; Pearson, S.; Cottingham, M.; Pucak, G.; Sanchez, A.; Trappier, S.G.; Peters, R.L.; Greer, P.W.; et al. Ebola (subtype Reston) virus among quarantined nonhuman primates recently imported from the Philippines to the United States. J. Infect. Dis. 1999, 179 (Suppl. 1), S108–S114. [Google Scholar]
- Control, C.F.D. Update: Filovirus infection in animal handlers. Morb. Mortal. Wkly. Rep. 1990, 39, 221. [Google Scholar]
- W.H.O. Viral haemorrhagic fever in imported monkeys. Wkly. Epidemiol. Rec. 1992, 67, 183. [Google Scholar]
- Walsh, P.D.; Breuer, T.; Sanz, C.; Morgan, D.; Doran-Sheehy, D. Potential for Ebola transmission between gorilla and chimpanzee social groups. Am. Nat. 2007, 169, 684–689. [Google Scholar] [CrossRef]
- Haydon, D.T.; Cleaveland, S.; Taylor, L.H.; Laurenson, M.K. Identifying reservoirs of infection: A conceptual and practical challenge. Emerg. Infect. Dis. 2002, 8, 1468–1473. [Google Scholar] [CrossRef]
- Olival, K.J.; Islam, A.; Yu, M.; Anthony, S.J.; Epstein, J.H.; Khan, S.A.; Khan, S.U.; Crameri, G.; Wang, L.F.; Lipkin, W.I.; et al. Ebola virus antibodies in fruit bats, Bangladesh. Emerg. Infect. Dis. 2013, 19, 270–273. [Google Scholar] [CrossRef]
- Taniguchi, S.; Watanabe, S.; Masangkay, J.S.; Omatsu, T.; Ikegami, T.; Alviola, P.; Ueda, N.; Iha, K.; Fujii, H.; Ishii, Y.; et al. Reston ebolavirus antibodies in bats, the Philippines. Emerg. Infect. Dis. 2011, 17, 1559–1560. [Google Scholar]
- Formenty, P.; Boesch, C.; Wyers, M.; Steiner, C.; Donati, F.; Dind, F.; Walker, F.; le Guenno, B. Ebola virus outbreak among wild chimpanzees living in a rain forest of Cote d’Ivoire. J. Infect. Dis. 1999, 179 (Suppl. 1), S120–S126. [Google Scholar]
- Wyers, M.; Formenty, P.; Cherel, Y.; Guigand, L.; Fernandez, B.; Boesch, C.; le Guenno, B. Histopathological and immunohistochemical studies of lesions associated with Ebola virus in a naturally infected chimpanzee. J. Infect. Dis. 1999, 179 (Suppl. 1), S54–S59. [Google Scholar]
- Johnson, E.D.; Johnson, B.K.; Silverstein, D.; Tukei, P.; Geisbert, T.W.; Sanchez, A.N.; Jahrling, P.B. Characterization of a new Marburg virus isolated from a 1987 fatal case in Kenya. Arch. Virol. Suppl. 1996, 11, 101–114. [Google Scholar]
- Bertherat, E.; Talarmin, A.; Zeller, H. Democratic Republic of the Congo: Between civil war and the Marburg virus. International Committee of Technical and Scientific Coordination of the Durba Epidemic. Med. Trop. (Mars) 1999, 59, 201–204. [Google Scholar]
- Colebunders, R.; Tshomba, A.; van Kerkhove, M.D.; Bausch, D.G.; Campbell, P.; Libande, M.; Pirard, P.; Tshioko, F.; Mardel, S.; Mulangu, S.; et al. Marburg hemorrhagic fever in Durba and Watsa, Democratic Republic of the Congo: Clinical documentation, features of illness, and treatment. J. Infect. Dis. 2007, 196 (Suppl. 2), S148–S153. [Google Scholar] [CrossRef]
- Zeller, H. Lessons from the Marburg virus epidemic in Durba, Democratic Republic of the Congo (1998–2000). Med. Trop. (Mars) 2000, 60, 23–26. [Google Scholar]
- Peterson, A.T.; Bauer, J.T.; Mills, J.N. Ecologic and geographic distribution of filovirus disease. Emerg. Infect. Dis. 2004, 10, 40–47. [Google Scholar] [CrossRef]
- Peterson, A.T.; Carroll, D.S.; Mills, J.N.; Johnson, K.M. Potential mammalian filovirus reservoirs. Emerg. Infect. Dis. 2004, 10, 2073–2081. [Google Scholar] [CrossRef]
- Peterson, A.T.; Lash, R.R.; Carroll, D.S.; Johnson, K.M. Geographic potential for outbreaks of Marburg hemorrhagic fever. Am. J. Trop. Med. Hyg. 2006, 75, 9–15. [Google Scholar]
- Morvan, J.M.; Deubel, V.; Gounon, P.; Nakoune, E.; Barriere, P.; Murri, S.; Perpete, O.; Selekon, B.; Coudrier, D.; Gautier-Hion, A.; et al. Identification of Ebola virus sequences present as RNA or DNA in organs of terrestrial small mammals of the Central African Republic. Microbes Infect./Inst. Pasteur 1999, 1, 1193–1201. [Google Scholar] [CrossRef]
- Hayman, D.T.S.; Yu, M.; Crameri, G.; Wang, L.F.; Suu-Ire, R.; Wood, J.L.N.; Cunningham, A.A. Ebola Virus Antibodies in Fruit Bats, Ghana, West Africa. Emerg. Infect. Dis. 2012, 18, 1207–1209. [Google Scholar] [CrossRef]
- Hayman, D.T.S.; Emmerich, P.; Yu, M.; Wang, L.F.; Suu-Ire, R.; Fooks, A.R.; Cunningham, A.A.; Wood, J.L.N. Long-Term Survival of an Urban Fruit Bat Seropositive for Ebola and Lagos Bat Viruses. PLoS One 2010, 5. [Google Scholar] [CrossRef]
- Pourrut, X.; Souris, M.; Towner, J.S.; Rollin, P.E.; Nichol, S.T.; Gonzalez, J.P.; Leroy, E. Large serological survey showing cocirculation of Ebola and Marburg viruses in Gabonese bat populations, and a high seroprevalence of both viruses in Rousettus aegyptiacus. BMC Infect. Dis. 2009, 9. [Google Scholar] [CrossRef]
- Towner, J.S.; Pourrut, X.; Albarino, C.G.; Nkogue, C.N.; Bird, B.H.; Grard, G.; Ksiazek, T.G.; Gonzalez, J.P.; Nichol, S.T.; Leroy, E.M. Marburg Virus Infection Detected in a Common African Bat. PLoS One 2007, 2. [Google Scholar] [CrossRef]
- Kuzmin, I.V.; Niezgoda, M.; Franka, R.; Agwanda, B.; Markotter, W.; Breiman, R.F.; Shieh, W.J.; Zaki, S.R.; Rupprecht, C.E. Marburg Virus in Fruit Bat, Kenya. Emerg. Infect. Dis. 2010, 16, 352–354. [Google Scholar] [CrossRef]
- Yuan, J.F.; Zhang, Y.J.; Li, J.L.; Zhang, Y.Z.; Wang, L.F.; Shi, Z.L. Serological evidence of ebolavirus infection in bats, China. Virol. J. 2012, 9. [Google Scholar] [CrossRef]
- Pourrut, X.; Delicat, A.; Rollin, P.E.; Ksiazek, T.G.; Gonzalez, J.P.; Leroy, E.M. Spatial and temporal patterns of Zaire ebolavirus antibody prevalence in the possible reservoir bat species. J. Infect. Dis. 2007, 196, S176–S183. [Google Scholar] [CrossRef]
- Hayman, D.T.; Suu-Ire, R.; Breed, A.C.; McEachern, J.A.; Wang, L.; Wood, J.L.; Cunningham, A.A. Evidence of henipavirus infection in West African fruit bats. PLoS One 2008, 3, e2739. [Google Scholar]
- De Jong, C.; Field, H.; Tagtag, A.; Hughes, T.; Dechmann, D.; Jayme, S.; Epstein, J.; Smith, C.; Santos, I.; Catbagan, D.; et al. Foraging Behaviour and Landscape Utilisation by the Endangered Golden-Crowned Flying Fox (Acerodon jubatus), The Philippines. PLoS One 2013, 8, e79665. [Google Scholar]
- Khan, M.S.U.; Hossain, J.; Gurley, E.S.; Nahar, N.; Sultana, R.; Luby, S.P. Use of Infrared Camera to Understand Bats’ Access to Date Palm Sap: Implications for Preventing Nipah Virus Transmission. Ecohealth 2011, 7, 517–525. [Google Scholar]
- Paweska, J.T.; Jansen van Vuren, P.; Masumu, J.; Leman, P.A.; Grobbelaar, A.A.; Birkhead, M.; Clift, S.; Swanepoel, R.; Kemp, A. Virological and serological findings in Rousettus aegyptiacus experimentally inoculated with vero cells-adapted hogan strain of Marburg virus. PLoS One 2012, 7, e45479. [Google Scholar] [CrossRef]
- Towner, J.S. Bats as reservoirs of filoviruses (data from recent challenges). In Proceedings of the 16th International Bat Research Conference, San Jose, Costa Rica, 11–15 August 2013.
- Albarino, C.G.; Uebelhoer, L.S.; Vincent, J.P.; Khristova, M.L.; Chakrabarti, A.K.; McElroy, A.; Nichol, S.T.; Towner, J.S. Development of a reverse genetics system to generate recombinant Marburg virus derived from a bat isolate. Virology 2013, 446, 230–237. [Google Scholar] [CrossRef]
- Crameri, G.; Todd, S.; Grimley, S.; McEachern, J.A.; Marsh, G.A.; Smith, C.; Tachedjian, M.; De Jong, C.; Virtue, E.R.; Yu, M.; et al. Establishment, Immortalisation and Characterisation of Pteropid Bat Cell Lines. PLoS One 2009, 4. [Google Scholar] [CrossRef]
- Jordan, I.; Munster, V.J.; Sandig, V. Authentication of the R06E Fruit Bat Cell Line. Viruses 2012, 4, 889–900. [Google Scholar] [CrossRef]
- Maruyama, J.; Miyamoto, H.; Kajihara, M.; Ogawa, H.; Maeda, K.; Sakoda, Y.; Yoshida, R.; Takada, A. Characterization of the envelope glycoprotein of a novel filovirus, lloviu virus. J. Virol. 2014, 88, 99–109. [Google Scholar] [CrossRef]
- Banyard, A.C.; Hayman, D.; Johnson, N.; McElhinney, L.; Fooks, A.R. Bats and lyssaviruses. Adv. Virus Res. 2011, 79, 239–289. [Google Scholar] [CrossRef]
- Hoffmann, M.; Muller, M.A.; Drexler, J.F.; Glende, J.; Erdt, M.; Gutzkow, T.; Losemann, C.; Binger, T.; Deng, H.; Schwegmann-Wessels, C.; et al. Differential sensitivity of bat cells to infection by enveloped RNA viruses: Coronaviruses, paramyxoviruses, filoviruses, and influenza viruses. PloS One 2013, 8, e72942. [Google Scholar] [CrossRef]
- Hayman, D.T.; Bowen, R.A.; Cryan, P.M.; McCracken, G.F.; O’Shea, T.J.; Peel, A.J.; Gilbert, A.; Webb, C.T.; Wood, J.L. Ecology of Zoonotic Infectious Diseases in Bats: Current Knowledge and Future Directions. Zoonoses Public Health 2013, 60, 2–21. [Google Scholar] [CrossRef]
- Bogich, T.L.; Olival, K.J.; Hosseini, P.; Zambrana-Torrelio, C.; Loh, E.; Funk, S.; Brito, I.; Epstein, J.H.; Brownstein, J.S.; Joly, D.O.; et al. Using Mathematical Models in a Unified Approach to Predicting the Next Emerging Infectious Disease. In New Directions in Conservation Medicine: Applied Cases of Ecological Health; Aguirre, A.A., Ostfeld, R.S., Daszak, P., Eds.; Oxford University Press: Oxford, UK, 2012; pp. 607–618. [Google Scholar]
- Wood, J.L.; Leach, M.; Waldman, L.; Macgregor, H.; Fooks, A.R.; Jones, K.E.; Restif, O.; Dechmann, D.; Hayman, D.T.; Baker, K.S.; et al. A framework for the study of zoonotic disease emergence and its drivers: Spillover of bat pathogens as a case study. Philos. Trans. R. Soc. Lond. Ser. B, Biol. Sci. 2012, 367, 2881–2892. [Google Scholar] [CrossRef]
- Daszak, P.; Plowright, R.; Epstein, J.H.; Pulliam, J.H.; Abdul Rahman, S.; Field, H.E.; Smith, C.S.; Olival, K.J.; Luby, S.; Halpin, K.; et al. The emergence of Nipah and Hendra virus: Pathogen dynamics across a wildlife-livestock-human continuum. In Disease Ecology: Community Structure and Pathogen Dynamics; Collinge, S., Ray, C., Eds.; Oxford University Press: Oxford, UK, 2006; pp. 188–203. [Google Scholar]
- Epstein, J.H.; Olival, K.J.; Pulliam, J.R.C.; Smith, C.; Westrum, J.; Hughes, T.; Dobson, A.P.; Zubaid, A.; Rahman, S.A.; Basir, M.M.; et al. Pteropus vampyrus, a hunted migratory species with a multinational home-range and a need for regional management. J. Appl. Ecol. 2009, 46, 991–1002. [Google Scholar] [CrossRef]
- Rahman, S.A.; Hassan, S.S.; Olival, K.J.; Mohamed, M.; Chang, L.-Y.; Hassan, L.; Suri, A.S.; Saad, N.M.; Shohaimi, S.A.; Mamat, Z.C.; et al. HERG Characterization of Nipah Virus from Naturally Infected Pteropus vampyrus Bats, Malaysia. Emerg. Infect. Dis. 2010, 16, 1990–1993. [Google Scholar] [CrossRef]
- Plowright, R.K.; Foley, P.; Field, H.E.; Dobson, A.P.; Foley, J.E.; Eby, P.; Daszak, P. Urban habituation, ecological connectivity and epidemic dampening: The emergence of Hendra virus from flying foxes (Pteropus spp.). Proc. R. Soci. B-Biol. Sci. 2011, 278, 3703–3712. [Google Scholar] [CrossRef]
- Pulliam, J.R.; Epstein, J.H.; Dushoff, J.; Rahman, S.A.; Bunning, M.; Jamaluddin, A.A.; Hyatt, A.D.; Field, H.E.; Dobson, A.P.; Daszak, P. Agricultural intensification, priming for persistence and the emergence of Nipah virus: A lethal bat-borne zoonosis. J. R. Soc. Interface 2012, 9, 89–101. [Google Scholar] [CrossRef]
- Rahman, S.A.; Hassan, L.; Epstein, J.H.; Mamat, Z.C.; Yatim, A.M.; Hassan, S.S.; Field, H.E.; Hughes, T.; Westrum, J.; Naim, M.S.; et al. Risk factors for Nipah virus infection among pteropid bats, Peninsular Malaysia. Emerg. Infect. Dis. 2013, 19, 51–60. [Google Scholar] [CrossRef]
- Thomas, D.W. Annual migration of three species of West African fruit bats (Chiroptera: Pteropodidae). Can. J. Zool. 1983, 61, 2266–2272. [Google Scholar] [CrossRef]
- Mutere, F.A. Breeding cycles in tropical bats in Uganda. J. Anim. Ecol. 1968, 37, 8–9. [Google Scholar]
- Hayman, D.T.S.; McCrea, R.; Restif, O.; Suu-Ire, R.; Fooks, A.R.; Wood, J.L.N.; Cunningham, A.A.; Rowcliffe, J.M. Demography of straw-colored fruit bats in Ghana. J. Mammal. 2012, 93, 1393–1404. [Google Scholar] [CrossRef]
- Mondul, A.M.; Krebs, J.W.; Childs, J.E. Trends in national surveillance for rabies among bats in the United States (1993–2000). J. Am. Vet. Med. Assoc. 2003, 222, 633–639. [Google Scholar] [CrossRef]
- Drexler, J.F.; Corman, V.M.; Wegner, T.; Tateno, A.F.; Zerbinati, R.M.; Gloza-Rausch, F.; Seebens, A.; Muller, M.A.; Drosten, C. Amplification of emerging viruses in a bat colony. Emerg. Infect. Dis 2011, 17, 449–456. [Google Scholar] [CrossRef]
- Patyk, K.; Turmelle, A.; Blanton, J.D.; Rupprecht, C.E. Trends in national surveillance data for bat rabies in the United States: 2001–2009. Vector Borne Zoonotic Dis. 2012, 12, 666–673. [Google Scholar] [CrossRef]
- Anderson, R.M.; May, R.M. Population biology of infectious diseases: Part I. Nature 1979, 280, 361–367. [Google Scholar] [CrossRef]
- Porter, T.A.; Wilkinson, G.S. Birth synchrony in greater spear-nosed bats (Phyllostomus hastatus). J. Zool. 2001, 253, 383–390. [Google Scholar] [CrossRef]
- Heideman, P.D.; Deoraj, P.; Bronson, F.H. Seasonal Reproduction of a Tropical Bat, Anoura-Geoffroyi, in Relation to Photoperiod. J. Reprod. Fertil. 1992, 96, 765–773. [Google Scholar] [CrossRef]
- Fleming, T.H.; Hooper, E.T.; Wilson, D.E. Three Central American bat communities: Structure, reproductive cycles and movement patterns. Ecology 1972, 53, 555–569. [Google Scholar] [CrossRef]
- Racey, P.A.; Entwistle, A.C. Life-History and Reproductive Strategies of Bats; Academic Press: London, UK, 2000; pp. 363–414. [Google Scholar]
- Peel, A.J.; Pulliam, J.R.C.; Luis, A.D.; Plowright, R.K.; O’Shea, T.J.; Hayman, D.T.S.; Wood, J.L.N.; Webb, C.T.; Restif, O. The effect of seasonal birth pulses on pathogen persistence in wild mammal populations. 2014; submitted for publication. [Google Scholar]
- Wacharapluesadee, S.; Boongird, K.; Wanghongsa, S.; Ratanasetyuth, N.; Supavonwong, P.; Saengsen, D.; Gongal, G.N.; Hemachudha, T. A longitudinal study of the prevalence of Nipah virus in Pteropus lylei bats in Thailand: Evidence for seasonal preference in disease transmission. Vector Borne Zoonotic Dis. 2010, 10, 183–190. [Google Scholar] [CrossRef]
- Peel, A.J.; Sargan, D.R.; Baker, K.S.; Hayman, D.T.; Barr, J.A.; Crameri, G.; Suu-Ire, R.; Broder, C.C.; Lembo, T.; Wang, L.F.; et al. Continent-wide panmixia of an African fruit bat facilitates transmission of potentially zoonotic viruses. Nat. Commun. 2013, 4, 2770. [Google Scholar]
- Ksiazek, T.G.; Rollin, P.E.; Williams, A.J.; Bressler, D.S.; Martin, M.L.; Swanepoel, R.; Burt, F.J.; Leman, P.A.; Khan, A.S.; Rowe, A.K.; et al. Clinical virology of Ebola hemorrhagic fever (EHF): Virus, virus antigen, and IgG and IgM antibody findings among EHF patients in Kikwit, Democratic Republic of the Congo, 1995. J. Infect. Dis. 1999, 179 (Suppl. 1), S177–S187. [Google Scholar]
- Chen, J.P.; Rossiter, S.J.; Flanders, J.R.; Sun, Y.H.; Hua, P.Y.; Miller-Butterworth, C.; Liu, X.S.; Rajan, K.E.; Zhang, S.Y. Contrasting Genetic Structure in Two Co-Distributed Species of Old World Fruit Bat. PLoS One 2010, 5, e13903. [Google Scholar] [CrossRef]
- Olival, K.J. Population Genetic Structure and Phylogeography of Southeast Asian Flying Foxes: Implications for Conservation and Disease Ecology; Columbia University: New York, NY, USA, 2008. [Google Scholar]
- Olival, K.J.; Dick, C.W.; Simmons, N.B.; Morales, J.C.; Melnick, D.J.; Dittmar, K.; Perkins, S.L.; Daszak, P.; Desalle, R. Lack of population genetic structure and host specificity in the bat fly, Cyclopodia horsfieldi, across species of Pteropus bats in Southeast Asia. Parasit Vectors 2013, 6, 231. [Google Scholar] [CrossRef]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef]
- Anthony, S.J.; Epstein, J.H.; Murray, K.A.; Navarrete-Macias, I.; Zambrana-Torrelio, C.M.; Solovyov, A.; Ojeda-Flores, R.; Arrigo, N.C.; Islam, A.; Ali Khan, S.; et al. A strategy to estimate unknown viral diversity in mammals. MBio 2013, 4. [Google Scholar] [CrossRef]
- Levinson, J.; Bogich, T.L.; Olival, K.J.; Epstein, J.H.; Johnson, C.K.; Karesh, W.; Daszak, P. Targeting surveillance for zoonotic virus discovery. Emerg. Infect. Dis. 2013, 19, 743–747. [Google Scholar] [CrossRef]
- Turmelle, A.S.; Olival, K.J. Correlates of viral richness in bats (Order Chiroptera). Ecohealth 2009, 6, 522–539. [Google Scholar] [CrossRef]
- Luis, A.D.; Hayman, D.T.S.; O’Shea, T.J.; Cryan, P.M.; Gilbert, A.T.; Pulliam, J.R.C.; Mills, J.N.; Timonin, M.E.; Willis, C.K.R.; Cunningham, A.A.; et al. A comparison of bats and rodents as reservoirs of zoonotic viruses: Are bats special? Proc. R. Soc. B-Biol. Sci. 2013, 280. [Google Scholar] [CrossRef]
- Newman, S.H.; Field, H.E.; Jong, C.E.D.; Epstein, J.H. Investigating the Role of Bats in Emerging Zoonoses: Balancing Ecology, Conservcation and Public Health Interests; Food and Agriculture Organisation of the United Nations: Rome, Italy, 2011; Volume 12. [Google Scholar]
- Smith, C.S.; De Jong, C.; Field, H. Sampling small quantities of blood from microbats. Acta Chiropterol. 2010, 12, 255–258. [Google Scholar] [CrossRef]
- Baker, M.L.; Schountz, T.; Wang, L.F. Antiviral Immune Responses of Bats: A Review. Zoonoses Public Health 2013, 60, 104–116. [Google Scholar] [CrossRef]
- Bossart, K.N.; McEachern, J.A.; Hickey, A.C.; Choudhry, V.; Dimitrov, D.S.; Eaton, B.T.; Wang, L.F. Neutralization assays for differential henipavirus serology using Bio-Plex protein array systems. J. Virol. Methods 2007, 142, 29–40. [Google Scholar] [CrossRef]
- Wright, E.; Hayman, D.T.; Vaughan, A.; Temperton, N.J.; Wood, J.L.; Cunningham, A.A.; Suu-Ire, R.; Weiss, R.A.; Fooks, A.R. Virus neutralising activity of African fruit bat (Eidolon helvum) sera against emerging lyssaviruses. Virology 2010, 408, 183–189. [Google Scholar] [CrossRef]
- Baker, K.S.; Suu-Ire, R.; Barr, J.; Hayman, D.T.; Broder, C.C.; Horton, D.L.; Durrant, C.; Murcia, P.R.; Cunningham, A.A.; Wood, J.L. Viral antibody dynamics in a chiropteran host. J. Anim. Ecol. 2013. [Google Scholar] [CrossRef]
- Peel, A.J.; McKinley, T.J.; Baker, K.S.; Barr, J.A.; Crameri, G.; Hayman, D.T.; Feng, Y.R.; Broder, C.C.; Wang, L.F.; Cunningham, A.A.; et al. Use of cross-reactive serological assays for detecting novel pathogens in wildlife: Assessing an appropriate cutoff for henipavirus assays in African bats. J. Virol. Methods 2013, 193, 295–303. [Google Scholar] [CrossRef]
- Zhang, G.; Cowled, C.; Shi, Z.; Huang, Z.; Bishop-Lilly, K.A.; Fang, X.; Wynne, J.W.; Xiong, Z.; Baker, M.L.; Zhao, W.; et al. Comparative analysis of bat genomes provides insight into the evolution of flight and immunity. Science 2013, 339, 456–460. [Google Scholar] [CrossRef]
- Katzourakis, A.; Gifford, R.J. Endogenous Viral Elements in Animal Genomes. PLoS Genet. 2010, 6, e1001191. [Google Scholar] [CrossRef]
- Belyi, V.A.; Levine, A.J.; Skalka, A.M. Unexpected Inheritance: Multiple Integrations of Ancient Bornavirus and Ebolavirus/Marburgvirus Sequences in Vertebrate Genomes. PLoS Pathog. 2010, 6, e1001030. [Google Scholar] [CrossRef]
- Timen, A.; Koopmans, M.P.; Vossen, A.C.; van Doornum, G.J.; Gunther, S.; van den Berkmortel, F.; Verduin, K.M.; Dittrich, S.; Emmerich, P.; Osterhaus, A.D.; et al. Response to imported case of Marburg hemorrhagic fever, the Netherland. Emerg. Infect. Dis. 2009, 15, 1171–1175. [Google Scholar] [CrossRef]
- W.H.O. Marburg haemorrhagic fever, Uganda. Wkly Epidemiol. Rec. 2007, 82, 297–298. [Google Scholar]
- W.H.O. Case of Marburg Haemorrhagic Fever Imported into the Netherlands from Uganda. Available online: http://www.who.int/csr/don/2008_07_10/en/ (accessed on 15 March 2014).
- Sohayati, A.R.; Hassan, L.; Sharifah, S.H.; Lazarus, K.; Zaini, C.M.; Epstein, J.H.; Shamsyul Naim, N.; Field, H.E.; Arshad, S.S.; Abdul Aziz, J.; et al. Evidence for Nipah virus recrudescence and serological patterns of captive Pteropus vampyrus. Epidemiol. Infect. 2011, 139, 1570–1579. [Google Scholar] [CrossRef]
- Jahrling, P.B.; Geisbert, T.W.; Jaax, N.K.; Hanes, M.A.; Ksiazek, T.G.; Peters, C.J. Experimental infection of cynomolgus macaques with Ebola-Reston filoviruses from the 1989–1990 U.S. epizootic. Arch. Virol. Suppl. 1996, 11, 115–134. [Google Scholar]
- Jahrling, P.B.; Geisbert, J.B.; Swearengen, J.R.; Larsen, T.; Geisbert, T.W. Ebola hemorrhagic fever: Evaluation of passive immunotherapy in nonhuman primates. J. Infect. Dis. 2007, 196 (Suppl. 2), S400–S403. [Google Scholar]
- Williamson, M.M.; Hooper, P.T.; Selleck, P.W.; Gleeson, L.J.; Daniels, P.W.; Westbury, H.A.; Murray, P.K. Transmission studies of Hendra virus (equine morbillivirus) in fruit bats, horses and cats. Aust. Vet. J. 1998, 76, 813–818. [Google Scholar] [CrossRef]
- Middleton, D.J.; Westbury, H.A.; Morrissy, C.J.; van der Heide, B.M.; Russell, G.M.; Braun, M.A.; Hyatt, A.D. Experimental Nipah virus infection in pigs and cats. J. Comp. Pathol. 2002, 126, 124–136. [Google Scholar] [CrossRef]
- Marsh, G.A.; Haining, J.; Robinson, R.; Foord, A.; Yamada, M.; Barr, J.A.; Payne, J.; White, J.; Yu, M.; Bingham, J.; et al. Ebola Reston Virus Infection of Pigs: Clinical Significance and Transmission Potential. J. Infect. Dis. 2011, 204, S804–S809. [Google Scholar] [CrossRef]
- Kobinger, G.P.; Leung, A.; Neufeld, J.; Richardson, J.S.; Falzarano, D.; Smith, G.; Tierney, K.; Patel, A.; Weingartl, H.M. Replication, pathogenicity, shedding, and transmission of Zaire ebolavirus in pigs. J. Infect. Dis. 2011, 204, 200–208. [Google Scholar] [CrossRef]
- Weingartl, H.M.; Embury-Hyatt, C.; Nfon, C.; Leung, A.; Smith, G.; Kobinger, G. Transmission of Ebola virus from pigs to non-human primates. Sci. Rep. 2012, 2. [Google Scholar] [CrossRef]
- Becquart, P.; Wauquier, N.; Mahlakoiv, T.; Nkoghe, D.; Padilla, C.; Souris, M.; Ollomo, B.; Gonzalez, J.P.; de Lamballerie, X.; Kazanji, M.; et al. High Prevalence of Both Humoral and Cellular Immunity to Zaire ebolavirus among Rural Populations in Gabon. PLoS One 2010, 5, e9126. [Google Scholar] [CrossRef]
- Chua, K.B.; Koh, C.L.; Hooi, P.S.; Wee, K.F.; Khong, J.H.; Chua, B.H.; Chan, Y.P.; Lim, M.E.; Lam, S.K. Isolation of Nipah virus from Malaysian Island flying-foxes. Microbes Infect./Inst. Pasteur 2002, 4, 145–151. [Google Scholar] [CrossRef]
- Gonzalez, J.P.; Pourrut, X.; Leroy, E. Ebolavirus and other filoviruses. Curr. Top. Microbiol. Immunol. 2007, 315, 363–387. [Google Scholar]
- Fogarty, R.; Halpin, K.; Hyatt, A.D.; Daszak, P.; Mungall, B.A. Henipavirus susceptibility to environmental variables. Virus Res. 2008, 132, 140–144. [Google Scholar] [CrossRef]
- Lekone, P.E.; Finkenstadt, B.F. Statistical inference in a stochastic epidemic SEIR model with control intervention: Ebola as a case study. Biometrics 2006, 62, 1170–1177. [Google Scholar] [CrossRef]
- Legrand, J.; Grais, R.F.; Boelle, P.Y.; Valleron, A.J.; Flahault, A. Understanding the dynamics of Ebola epidemics. Epidemiol. Infect. 2007, 135, 610–621. [Google Scholar] [CrossRef]
- Chowell, G.; Hengartner, N.W.; Castillo-Chavez, C.; Fenimore, P.W.; Hyman, J.M. The basic reproductive number of Ebola and the effects of public health measures: The cases of Congo and Uganda. J. Theor. Biol. 2004, 229, 119–126. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Olival, K.J.; Hayman, D.T.S. Filoviruses in Bats: Current Knowledge and Future Directions. Viruses 2014, 6, 1759-1788. https://doi.org/10.3390/v6041759
Olival KJ, Hayman DTS. Filoviruses in Bats: Current Knowledge and Future Directions. Viruses. 2014; 6(4):1759-1788. https://doi.org/10.3390/v6041759
Chicago/Turabian StyleOlival, Kevin J., and David T. S. Hayman. 2014. "Filoviruses in Bats: Current Knowledge and Future Directions" Viruses 6, no. 4: 1759-1788. https://doi.org/10.3390/v6041759
APA StyleOlival, K. J., & Hayman, D. T. S. (2014). Filoviruses in Bats: Current Knowledge and Future Directions. Viruses, 6(4), 1759-1788. https://doi.org/10.3390/v6041759




