Chikungunya Virus–Vector Interactions
Abstract
:1. Introduction
2. Chikungunya Infection and Vector Life History Traits
2.1. Chikungunya Virus Transmission Cycles and Genetic Origins
2.2. Chikungunya Distribution, Emergence and Spread
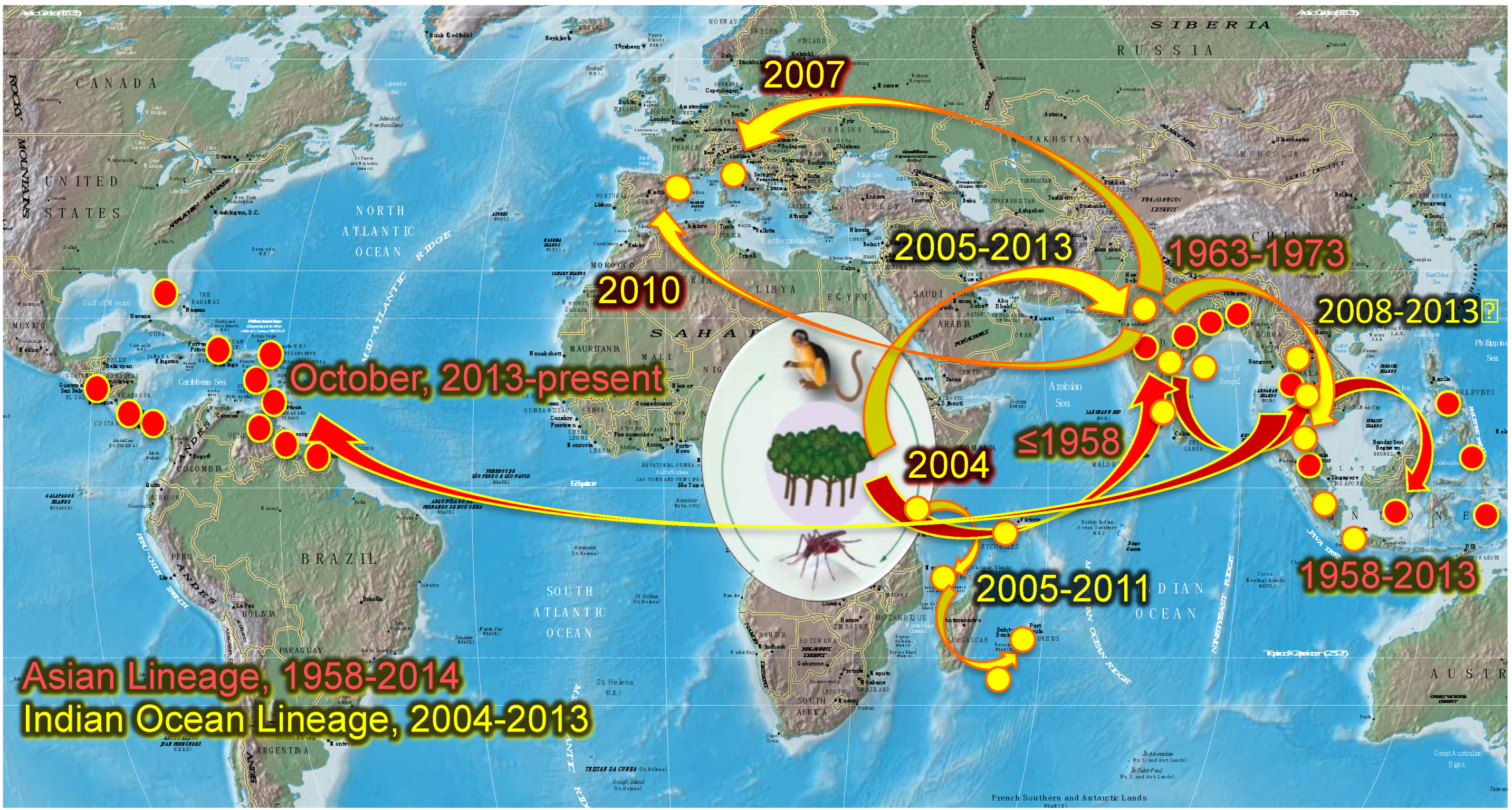
3. Vectors
3.1. Chikungunya Vector Distribution
3.2. Vector Dispersal and Genetics
3.3. Vector Competence
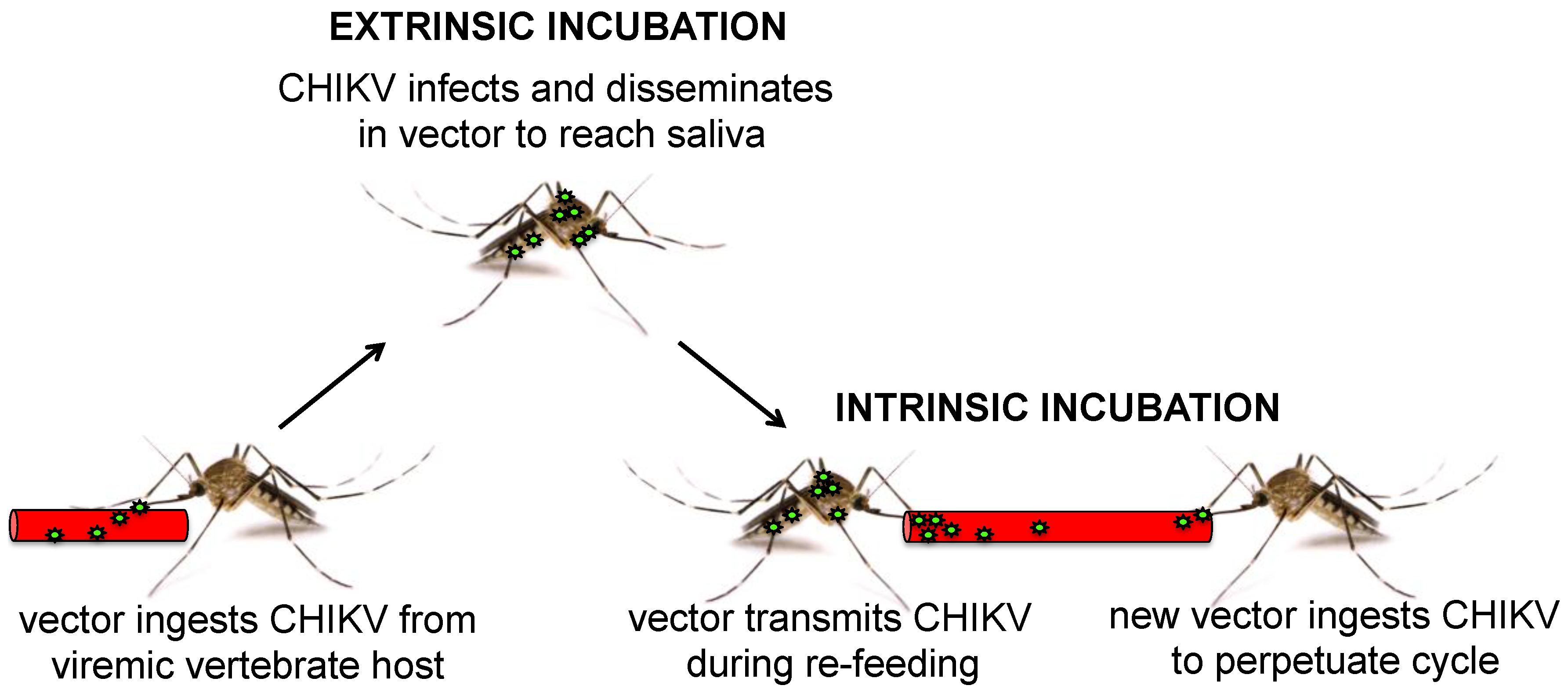
3.4. Chikungunya Virus Vector Immunity and Microbial Interactions
3.5. Vector co-Infection by Chikungunya and Other Arboviruses
4. Chikungunya Virus
4.1. Genetics of Vector Susceptibility and Host Range Changes
4.2. Population Heterogeneity & Selection for Fittest Genomes
4.3. Dual Host Cycling & Chikungunya Virus Adaptation
4.4. Viral Bottlenecks and Intrahost Diversity
4.5. Adaptive Constraints on Chikungunya Virus Evolution
5. Future Prospects
5.1. Potential for Re-Emergence and Expansion into New Areas
5.2. Prospects for Prevention via Vector Control
| Mosquito Species | Source | Generation Number | Source of CHIKV, Isolation Year | Strain Name | Bloodmeal Titer | Incubation Period (Days) | % Infected (n) | % Disseminated (n) | % Transmitted (n) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Aedes aegypti | Queensland, Australia | 1 | patient in Melbourne ex. Mauritius, March 2006 | not stated | 4 | 14-15 | 92 (23/25) | 92 (23/25) | 64 (16/25) | van den Hurk et al., 2010 [128] |
| New Caledonia | 1 | patient in New Caledonia ex. Indonesia, February 2011 | NC 2011-568 | 7 | 14 | n.d. | n.d. | 27 (10/37) | Dupont-Rouzeyrol et al., 2012 [41] | |
| patient in Reunion Island, 2006 | 6.21 | n.d. | n.d. | 75 (27/36) | ||||||
| Mayotte | 3 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 87 (54/62) | n.d. | Martin et al., 2010 [94] | |
| patient in Reunion Island, 2005 (E1226A) | 6.115 | n.d. | 78 (43/55) | n.d. | ||||||
| Cameroon | 1 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 89 (333/376) | n.d. | Paupy et al., 2010 [185] | |
| patient in Reunion Island, 2005 (E1226A) | 06.115 | n.d. | 97 (36/37) | n.d. | Vazeille et al., 2007 [10] | |||||
| patient in Reunion Island, 2005 (E1226V) | 06.21 | n.d | 65 (70/107) | n.d. | ||||||
| patient in Mayotte, 2006 | 06.111 | n.d. | 82 (68/82) | n.d. | ||||||
| patient in Democratic Republic of Congo, 2000 | 06.117 | n.d | 84 (56/66) | n.d. | ||||||
| Dakar, Senegal | 4 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 2 (1/45) | 0 (0/45) | n.d. | Turell et al., 1992 [186] | |
| Lagos, Nigeria | colony | patient in Calcutta, India, 1963 | 63-266 | 8 | 14 | 10 (2/20) | n.d. | 50 (1/2) | Shah et al., 1964 [187] | |
| French West Indies | 1-2 | patient in Reunion Island, 2006 | 06.21 | 7.5 | 14 | 98 (900/918) | n.d. | n.d. | Girod et al., 2011 [188] | |
| patient in Reunion Island, 2006 | 06.21 | 6 | 7 | 47 (301/634) | n.d. | n.d. | ||||
| Trinidad, West Indies | colony | patient in Calcutta, India, 1963 | 63-266 | 8 | 14 | 42 (3/7) | n.d. | 100 (1/1) | Shah et al., 1964 | |
| Aedes aegypti | Guadeloupe | 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 96 (346/358) | n.d. | Girod et al., 2011 |
| Martinique | 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 98 (285/290) | n.d. | Girod et al., 2011 | |
| French Guyana | 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 99 (269/270) | n.d. | Girod et al., 2011 | |
| Florida, USA | 1 | patient in France ex. Reunion Island, 2006 | LR2006-OPY1 | 6.1 | 6 | 58 (15/26) | 73 (11/15) | n.d. | Pesko et al., 2009 [93] | |
| patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 100 (48/48) | n.d. | Vega-Rua et al., 2014 [43] | |||
| Louisiana, USA | 4-5 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 5 (3/60) | 2 (1/60) | n.d. | Turell et al., 1992 | |
| Indiana, USA | 7 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 6 (2/35) | 6 (2/35) | n.d. | Turell et al., 1992 | |
| Puerto Rico, USA | 3 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 55 (9/55) | 11 (6/55) | n.d. | Turell et al., 1992 | |
| 5 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 33 (10/30) | 10 (3/30) | n.d. | |||
| Mexico | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 97 (58/60) | n.d. | Vega-Rua et al., 2014 | |
| Panama | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 97 (58/60) | n.d. | Vega-Rua et al., 2014 | |
| patient in New Caledonia, 2011 | NC/2011-568 | n.d. | 100 (30/30) | n.d. | ||||||
| Venezuela | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 100 (51/51) | n.d. | Vega-Rua et al., 2014 | |
| Peru | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 100 (89/89) | n.d. | Vega-Rua et al., 2014 | |
| Brazil | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 98 (128/130) | n.d. | Vega-Rua et al., 2014 | |
| patient in New Caledonia, 2011 | NC/2011-568 | n.d. | 95 (57/60) | n.d. | Vega-Rua et al., 2014 | |||||
| Bolivia | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 100 (60/60) | n.d. | Vega-Rua et al., 2014 | |
| Aedes aegypti | Paraguay | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 99 (89/90) | n.d. | Vega-Rua et al., 2014 |
| Uruguay | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 100 (60/60) | n.d. | Vega-Rua et al., 2014 | |
| Argentina | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 99 (119/120) | n.d. | Vega-Rua et al., 2014 | |
| Bangkok, Thailand | 2 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 66 (20/30) | 66 (20/30) | n.d. | Turell et al., 1992 | |
| Ho Chi Minh City, Vietnam | colony | patient in Reunion Island, 2005 (E1226A) | 06.115 | 7 | 14 | n.d. | 66 (135/206) | n.d. | Vazeille et al., 2007 | |
| patient in Reunion Island, 2005 (E1226V) | 06.21 | n.d | 97 (227/234) | n.d. | ||||||
| patient in Mayotte, 2006 | 06.111 | n.d. | 92 (126/137) | n.d. | ||||||
| patient in Democratic Republic of Congo, 2000 | 06.117 | n.d | 78 (108/138) | n.d. | ||||||
| Higgs variant, Rexville D | colony | clone derived from patient in France ex. Reunion Island, 2006 E1226A | LR2006-OPY1 E1A226 | 5 | 7 | 20 (not stated) | n.d. | n.d. | Tsetsarkin et al., 2007 [9] | |
| clone derived from patient in France ex. Reunion Island, 2006 E1226V | LR2006-OPY1 E1V226 | 5 (not stated) | n.d. | n.d. | ||||||
| infectious clone from patient in West Africa, E1226A | 37997 E1A226 | 20 (not stated) | n.d. | n.d. | ||||||
| infectious clone from patient in West Africa, E1226V | 37997 E1V226 | 10 (not stated) | n.d. | n.d. | ||||||
| Aedes furcifer, Kadougou, Senegal, 1983 | 37997 | 8 | 14 | 100 (7/7) | 63 (5/8) | n.d. | Vanlandingham et al., 2005 [189] | |||
| not stated | colony | patient in Africa, not stated | not stated | 8.6-9.2 | 14 | 53 (24/45) | n.d. | 44 (20/45) | Mangiafico, 1971 [190] | |
| Rockefeller | colony | patient in Thailand, 1962 | 15561 | 4.2-4.6 | 7 | 18 (9/50) | 12 (6/50) | n.d. | Turell et al., 1992 | |
| Madeira Island, Spain | 1 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 100 (27/27) | 40 (4/10) | Vazeille et al., 2012 [191] | |
| Aedes albopictus | Queensland, Australia | 7 | patient in Melbourne ex. Mauritius, March 2006 | not stated | 3.9 | 14-15 | 92 (23/25) | 92 (23/25) | 32 (8/25) | van den Hurk et al., 2010 |
| Torres Strait, Australia | 7 | patient in Melbourne ex. Mauritius, March 2006 | not stated | 8 | 14 | 80 (4/5) | 80 (4/5) | 60 (3/5) | Nicholson et al., 2014 [192] | |
| Mauritius | 1 | patient in India, 1973 | Barsi, P0-731460 | 5.8 | 8-9 | 32 (13/41) | n.d. | n.d. | Tesh et al., 1976 [193] | |
| patient in Tanzania, 1953 | Ross, S-27 | 6.8 | 8-9 | 19 (13/67) | n.d. | n.d. | Tesh et al., 1976 | |||
| Mayotte | 1 | patient in Reunion Island, 2005 (E1226A) | 6.115 | 7 | 14 | n.d | 25 (115/462) | n.d. | Vazeille et al., 2007 | |
| 6 | patient in Reunion Island, 2005 (E1226A) | 6.115 | 7.5 | 14 | n.d. | 79 (45/57) | n.d. | Martin et al., 2010 [94] | ||
| 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7 | 14 | n.d | 91 (296/325) | n.d. | Vazeille et al., 2007 | ||
| 6 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 99 (64/65) | n.d. | Martin et al., 2010 | ||
| 1 | patient in Mayotte, 2006 | 06.111 | 7 | 14 | n.d. | 98 (48/49) | n.d. | Vazeille et al., 2007 | ||
| 1 | patient in Democratic Republic of Congo, 2000 | 06.117 | 7 | 14 | n.d | 73 (41/56) | n.d. | Vazeille et al., 2007 | ||
| Reunion Island | 2 | patient in Reunion Island, 2005 (E1226A) | 6.115 | 7.5 | 14 | n.d. | 90 (26/29) | n.d. | Martin et al., 2010 | |
| 1 | patient in Reunion Island, 2005 (E1226A) | 6.115 | 7 | 14 | n.d. | 25 (114/462) | n.d. | Vazeille et al., 2007 | ||
| 2 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 98 (55/56) | n.d. | Martin et al., 2010 | ||
| 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7 | 14 | n.d | 96 (391/409) | n.d. | Vazeille et al., 2007 | ||
| 2 | patient in Mayotte, 2006 | 6.111 | 7 | 14 | n.d. | 97 (91/94) | n.d. | Vazeille et al., 2007 | ||
| 2 | patient in Democratic Republic of Congo, 2000 | 6.117 | 7 | 14 | n.d | 80 (25/31) | n.d. | Vazeille et al., 2007 | ||
| Madagascar | 7 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 95 (19/20) | 35 (7/20) | n.d. | Turell et al., 1992 | |
| 1 | patient in India, 1973 | Barsi, P0-731460 | 6.2 | 8–9 | 87 (33/38) | n.d. | n.d. | Tesh et al., 1976 | ||
| 1 | patient in Tanzania, 1953 | Ross, S-27 | 6.8 | 8–9 | 39 (19/49) | n.d. | n.d. | Tesh et al., 1976 | ||
| 2-5 | patient in Reunion Island, 2006 | not stated | 7.5 | 14 | n.d. | 98 (497/503) | n.d. | Raharimalala et al., 2012 [76] | ||
| Aedes albopictus | Cameroon | 1 | patient in Reunion Island, 2006 | 06.21 | 7 | 14 | n.d. | 85 (187/218) | n.d. | Paupy et al., 2010 |
| patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d | 68 (41/60) | n.d. | Vazeille et al., 2007 | |||
| patient in Mayotte, 2006 | 06.111 | 7 | 14 | n.d. | 44 (34/44) | n.d. | Vazeille et al., 2007 | |||
| patient in Democratic Republic of Congo, 2000 | 06.117 | 7 | 14 | n.d | 56 (22/39) | n.d. | Vazeille et al., 2007 | |||
| patient in Reunion Island, 2005 (E1226A) | 06.115 | 7 | 14 | n.d | 12 (11/90) | n.d. | Vazeille et al., 2007 | |||
| Virginia and Georgia, USA | 1 | mosquito pool, Comoros, 2005 strain COM125 | COM125 | 4.9 | 7 | 73 (83/114) | n.d. | 40 (33/83) | McTighe & Vaidyanathan, 2012 | |
| Hawaii, USA | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7-7.5 | 8-9 | 69 (76/110) | n.d. | n.d. | Tesh et al., 1976 | |
| 1 | patient in India, 1973 | Barsi, P0-731460 | 5.8 | 8-9 | 97 (120/124) | n.d. | n.d. | Tesh et al., 1976 | ||
| colony | patient in Calcutta, India, 1963 | 63-266 | 8 | 14 | 100 (32/32) | n.d. | 34 (8/22) | Shah et al., 1964 | ||
| colony | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 93 (28/30) | 60 (18/30) | n.d. | Turell et al., 1992 | ||
| Florida, USA | 1 | patient in France ex. Reunion Island, 2006 | LR2006-OPY1 | 6.1 | 6 | 100 (22/22) | 91 (20/22) | n.d. | Pesko et al., 2009 | |
| patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 83 (50/60) | n.d. | Vega-Rua et al., 2014 | |||
| 2 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 97 (29/30) | 37 (11/30) | n.d. | Turell et al., 1992 | ||
| Missouri, USA | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 90 (54/60) | n.d. | Vega-Rua et al., 2014 | |
| Aedes albopictus | Texas, USA | colony | clone derived from patient in France ex. Reunion Island, 2006 E1226A | LR2006-OPY1 E1A226 | 5 | 7 | 31 (61/194) | 30 (not stated) | Tsetsarkin et al., 2007 | |
| clone derived from patient in France ex. Reunion Island, 2006 E1226V | LR2006-OPY1 E1V226 | 5 | 7 | 90 (241/269) | 65 (not stated) | Tsetsarkin et al., 2007 | ||||
| infectious clone from patient in West Africa, E1226A | 37997 E1A226 | 5 | 7 | 37 (97/226) | n.d. | n.d. | Tsetsarkin et al., 2007 | |||
| infectious clone from patient in West Africa, E1226V | 37997 E1V226 | 5 | 7 | 92 (253/274) | n.d. | n.d. | Tsetsarkin et al., 2007 | |||
| 9-10 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 75 (15/20) | 35 (7/20) | n.d. | Turell et al., 1992 | ||
| Louisiana, USA | 4-5 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 97 (29/30) | 80 (24/30) | n.d. | Turell et al., 1992 | |
| Mexico | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 70 (42/60) | n.d. | Vega-Rua et al., 2014 | |
| Panama | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 95 (57/60) | n.d. | Vega-Rua et al., 2014 | |
| patient in New Caledonia, 2011 | NC/2011-568 | 7.5 | 10 | n.d. | 97 (29/30) | n.d. | Vega-Rua et al., 2014 | |||
| Brazil | 6-7 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 73 (22/30) | 50 (15/30) | n.d. | Turell et al., 1992 | |
| 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 94 (301/320) | n.d. | Vega-Rua et al., 2014 | ||
| Argentina | 1 | patients in Reunion Island, 2005 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 63 (35/56) | n.d. | Vega-Rua et al., 2014 | |
| patient in New Caledonia, 2011 | 06.21 and/or 06.115 | 7.5 | 10 | n.d. | 93 (28/30) | n.d. | Vega-Rua et al., 2014 | |||
| Israel | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7.2 | 8-9 | 30 (14/47) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1973 | Barsi, P0-731460 | 5.8 | 8-9 | 67 (37/55) | n.d. | n.d. | Tesh et al., 1976 | |||
| Lebanon | 1 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 8 | 14 | n.d. | 29 (12/42) | n.d. | Haddad et al., 2012 [194] | |
| Aedes albopictus | Italy | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 83 (52/63) | n.d. | Talbalaghi et al., 2010 [195] |
| France | 0 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7 | 14 | n.d. | 77.1 (27/35) | n.d. | Vazeille et al., 2008 [196] | |
| 13 | patient in France, 2010 (E1226A) | 1909 | 7.3 | 14 | n.d. | 96 (21/22) | 14 (3/21) | Vega-Rua et al., 2013 [36] | ||
| 13 | patient in France, 2010 (E1226A) | 1630 | 7.3 | 14 | n.d. | 90 (17/19) | 12 (2/17) | Vega-Rua et al., 2013 | ||
| Corsica, France | 0 | patient in Reunion Island, 2005 (E1226V) | 6.21 | 7.5 | 14 | n.d. | 94 (377/401) | n.d. | Moutailler et al., 2009 [197] | |
| Indonesia | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7.1 | 8–9 | 64 34/53) | n.d. | n.d. | Tesh et al., 1976 | |
| Philippines | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7.2 | 8–9 | 55 (21/38) | n.d. | n.d. | Tesh et al., 1976 | |
| India | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7–7.2 | 8–9 | 38 (30/79) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1973 | Barsi, P0-731460 | 5.7–5.9 | 8–9 | 71 (74/104) | n.d. | n.d. | Tesh et al., 1976 | |||
| Vietnam | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7.7 | 8–9 | 44 (29/66) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1978 | Barsi, P0-731460 | 5.7 | 8–9 | 49 (37/94) | n.d. | n.d. | Tesh et al., 1976 | |||
| Thailand | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7.4 | 8–9 | 38 (12/32) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1973 | Barsi, P0-731460 | 6 | 8–9 | 73 (24/33) | n.d. | n.d. | Tesh et al., 1976 | |||
| Malaysia | 1 | patient in Tanzania, 1953 | Ross, S-27 | 6.9 | 8–9 | 29 (15/51) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1973 | Barsi, P0-731460 | 6 | 8–9 | 42 (27/64) | n.d. | n.d. | Tesh et al., 1976 | |||
| Taipei | 1 | patient in Tanzania, 1953 | Ross, S-27 | 7 | 8–9 | 25 (12/48) | n.d. | n.d. | Tesh et al., 1976 | |
| patient in India, 1973 | Barsi, P0-731460 | 5.8 | 8–9 | 28 (14/50) | n.d. | n.d. | Tesh et al., 1976 | |||
| Taiwan | 2 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 90 (27/30) | 20 (6/30) | n.d. | Turell et al., 1992 | |
| Okinawa, Japan | 5 | patient in Thailand, 1962 | 15561 | 5.3 | 7 | 72 (18/25) | 28 (7/25) | n.d. | Turell et al., 1992 | |
| Tokyo, Japan | 5 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 50 (14/28) | 14 (7/50 | n.d. | Turell et al., 1992 | |
| Jakarta | 1 | patient in India, 1975 | Barsi, P0-731460 | 5.8 | 8–9 | 70 (23/33) | n.d. | n.d. | Tesh et al., 1976 | |
| Philippines | 1 | patient in India, 1976 | Barsi, P0-731460 | 6 | 8–9 | 87 (33/38) | n.d. | n.d. | Tesh et al., 1976 | |
| Sabah, Malaysia | 5 | patient in Thailand, 1962 | 15561 | 4.2–4.6 | 7 | 43 (15/35) | 6 (2/43) | n.d. | Turell et al., 1992 | |
| Aedes albopictus | Hanoi, Vietnam | 3 | patient in Reunion Island, 2005 (E1226A) | 6.115 | 7 | 14 | n.d | 30 (16/54) | n.d. | Vazeille et al., 2007 |
| patient in Reunion Island, 2005 (E1226V) | 6.21 | 7 | 14 | n.d | 84 (105/126) | n.d. | Vazeille et al., 2007 | |||
| patient in Mayotte, 2006 | 6.111 | 7 | 14 | n.d. | 84 (105/126) | n.d. | Vazeille et al., 2007 | |||
| patient in Democratic Republic of Congo, 2000 | 6.117 | 7 | 14 | n.d | 47 (56/119) | n.d. | Vazeille et al., 2007 | |||
| not stated | colony | patient in Africa, not stated | not stated | 8.6–9.2 | 14 | 100 (50/50) | n.d. | 80 (40/50) | Mangiafico, 1971 | |
| Aedes antipodeus | North Auckland, New Zealand | 1 | patient in India | 91064A | 7.8 | 21** | 100 (15/15) | 73 (11/15) | 0 (0/15) | Kramer et al., 2011 [198] |
| Aedes caspius | France | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 25 (4/16) | n.d. | Vazeille et al., 2008 |
| Aedes detritus | France | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 67.3 (33/49) | n.d. | Vazeille et al., 2008 |
| Aedes fulgens | South Africa | 1 | not stated | H817 | 5.7 | 9–12 | 88 (29/33) | n.d. | 10 (3/29) | Jupp et al., 1981 [199] |
| Aedes furcifer | South Africa | colony or 1-4 | not stated | H817 | 5.7–6.9 | 8–29 | 71 (192/271) | n.d. | 30 (8/27) | Jupp et al., 1981 |
| Aedes hensilli | Micronesia | 12-15 | Mosquito in Comoros, 2005 | COM 125 | 5.7 | 8 | 63 (20/32) | 80 (16/20) | n.d. | Ledermann et al., 2014 [200] |
| Aedes notoscriptus | Auckland, New Zealand | 1 | patient in India | 91064A | 10.5 | 14* | 36 (8/32) | 75 (6/8) | 0 (0/8) | Kramer et al., 2011 |
| Aedes polynesiensis | Samoa | colony | patient in Calcutta, India, 1963 | 63-266 | 8 | 14 | 40 (4/10) | n.d. | n.d. | Shah et al., 1964 |
| Aedes togoi | not stated | colony | patient in Africa, not stated | not stated | 8.6–9.2 | 14 | 97 (42/43) | n.d. | 12 (5/43) | Mangiafico, 1971 |
| Aedes triseriatus | not stated | colony | patient in Africa, not stated | not stated | 8.6–9.2 | 14 | 100 (50/50) | n.d. | 84 (42/50) | Mangiafico, 1971 |
| Aedes vexans | Italy | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 8 (2/26) | n.d. | Talbalaghi et al., 2010 |
| France | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 0 (0/13) | n.d. | Vazeille et al., 2008 | ||
| Aedes vittatus | Senegal | 1 | mosquitoes, bats or humans in Senegal, 1962, '79 and '05 | ArD30237, CS13-288 or HD 180738 | 6 | 10 | 89 (41/46) | 54 (22/41) | 18 (4/22) | Diagne et al., 2014 [201] |
| 6-7 | 10 | 19 (19/98) | 37 (7/19) | 43 (3/7) | Diagne et al., 2014 | |||||
| Anopheles gambiae | G3 | colony | Aedes furcifer, Kadoug ou, Senegal, 1983 | 37997 | 8 | 14 | 0 (0/8) | 0 (0/8) | n.d. | Vanlandingham et al., 2005 |
| Anopheles maculipennis | Italy | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 0 (0/10) | n.d. | Talbalaghi et al., 2010 |
| Culex fatigans | Philippines | colony | patient in Calcutta, India, 1963 | 63-266 | 8 | 14 | 0 (0/10) | n.d. | n.d. | Shah et al., 1964 |
| Culex horridus | South Africa | 1 | not stated | H817 | 4.6–5.4 | 14–25 | 6 (1/17) | n.d. | n.d. | Jupp et al., 1981 |
| Culex pipiens | Italy | 0 | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 0 (0/45) | n.d. | Talbalaghi et al., 2010 |
| France | patient in Reunion Island, 2005 (E1226V) | 06.21 | 7 | 14 | n.d. | 0 (0/11) | n.d. | Vazeille et al., 2008 | ||
| Culex quinquefasciatus | Zimbabwe | 1 | not stated | H817 | 5.3 | 20–22 | 0 (0/19) | n.d. | 0 (0/19) | Jupp et al., 1981 |
| Eretmapodites chrysogaster | not stated | colony | patient in Africa, not stated | not stated | 8.6–9.2 | 14 | 80 (40/50) | n.d. | 36 (18/50) | Mangiafico, 1971 |
| Mansonia africana | Mozambique | 0 | not stated | H817 | 4.7–5.2 | 8–15 | 34 (23/67) | n.d. | n.d. | Jupp et al., 1981 |
| Opifex fuscus | Wellington, New Zealand | 1 | patient in India | 91064A | 6.2 | 11* | 98 (46/47) | 100 (46/46) | 100 (46/46) | Kramer et al., 2011 |
| Ornithodoros savignyi | South Africa | 0 | not stated | H817 | 6.6 | 50-61 | 0 (0/11) | n.d. | n.d. | Jupp et al., 1981 |
6. Conclusions
Conflicts of Interest
References and Notes
- Powers, A.M.; Logue, C.H. Changing patterns of chikungunya virus: Re-emergence of a zoonotic arbovirus. J. Gen. Virol. 2007, 88, 2363–2377. [Google Scholar] [PubMed]
- Kuehn, B.M. Chikungunya virus transmission found in the United States: US health authorities brace for wider spread. JAMA 2014, 312, 776–777. [Google Scholar] [PubMed]
- CDC. Chikungunya virus in the United States. Available online: http://www.cdc.gov/chikungunya/geo/united-states.html (accessed on 8 November 2014).
- Jupp, P.G.; McIntosh, B.M. Aedes furcifer and other mosquitoes as vectors of chikungunya virus at Mica, Northeastern Transvaal, South Africa. J. Am. Mosq. Control. Assoc. 1990, 6, 415–420. [Google Scholar] [PubMed]
- Volk, S.M.; Chen, R.; Tsetsarkin, K.A.; Adams, A.P.; Garcia, T.I.; Sall, A.A.; Nasar, F.; Schuh, A.J.; Holmes, E.C.; Higgs, S.; et al. Genome-scale phylogenetic analyses of chikungunya virus reveal independent emergences of recent epidemics and various evolutionary rates. J. Virol. 2010, 84, 6497–6504. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Chen, R.; Sherman, M.B.; Weaver, S.C. Chikungunya virus: Evolution and genetic determinants of emergence. Curr. Opin. Virol. 2011, 1, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Kariuki Njenga, M.; Nderitu, L.; Ledermann, J.P.; Ndirangu, A.; Logue, C.H.; Kelly, C.H.; Sang, R.; Sergon, K.; Breiman, R.; Powers, A.M. Tracking epidemic chikungunya virus into the Indian Ocean from East Africa. J. Gen. Virol. 2008, 89, 2754–2760. [Google Scholar]
- Schuffenecker, I.; Iteman, I.; Michault, A.; Murri, S.; Frangeul, L.; Vaney, M.C.; Lavenir, R.; Pardigon, N.; Reynes, J.M.; Pettinelli, F.; et al. Genome microevolution of chikungunya viruses causing the Indian Ocean outbreak. PLoS Med. 2006, 3, e263. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Vanlandingham, D.L.; McGee, C.E.; Higgs, S. A single mutation in chikungunya virus affects vector specificity and epidemic potential. PLoS Pathog. 2007, 3, e201. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Moutailler, S.; Coudrier, D.; Rousseaux, C.; Khun, H.; Huerre, M.; Thiria, J.; Dehecq, J.S.; Fontenille, D.; Schuffenecker, I.; et al. Two chikungunya isolates from the outbreak of La Reunion (Indian Ocean) exhibit different patterns of infection in the mosquito, Aedes albopictus. PLoS One 2007, 2, e1168. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; McGee, C.E.; Volk, S.M.; Vanlandingham, D.L.; Weaver, S.C.; Higgs, S. Epistatic roles of E2 glycoprotein mutations in adaption of chikungunya virus to Aedes albopictus and Ae. aegypti mosquitoes. PLoS One 2009, 4, e6835. [Google Scholar] [CrossRef] [PubMed]
- Arias-Goeta, C.; Mousson, L.; Rougeon, F.; Failloux, A.B. Dissemination and transmission of the E1–226V variant of chikungunya virus in Aedes albopictus are controlled at the midgut barrier level. PLoS One 2013, 8, e57548. [Google Scholar] [CrossRef] [PubMed]
- Tsetsarkin, K.A.; Chen, R.; Yun, R.; Rossi, S.L.; Plante, K.S.; Guerbois, M.; Forrester, N.; Perng, G.C.; Sreekumar, E.; Leal, G.; et al. Multi-peaked adaptive landscape for chikungunya virus evolution predicts continued fitness optimization in Aedes albopictus mosquitoes. Nat. Commun. 2014, 5, 4084. [Google Scholar] [CrossRef] [PubMed]
- Ratsitorahina, M.; Harisoa, J.; Ratovonjato, J.; Biacabe, S.; Reynes, J.M.; Zeller, H.; Raoelina, Y.; Talarmin, A.; Richard, V.; Louis Soares, J. Outbreak of dengue and chikungunya fevers, toamasina, Madagascar, 2006. Emerg. Infect. Dis. 2008, 14, 1135–1137. [Google Scholar] [CrossRef] [PubMed]
- De Lamballerie, X.; Leroy, E.; Charrel, R.N.; Ttsetsarkin, K.; Higgs, S.; Gould, E.A. Chikungunya virus adapts to tiger mosquito via evolutionary convergence: A sign of things to come? Virol. J. 2008, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Bagny, L.; Delatte, H.; Quilici, S.; Fontenille, D. Progressive decrease in Aedes aegypti distribution in reunion island since the 1900s. J. Med. Entomol. 2009, 46, 1541–1545. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.P.; Joseph, R.; Kamaraj, T.; Jambulingam, P. A226V mutation in virus during the 2007 chikungunya outbreak in Kerala, India. J. Gen. Virol. 2008, 89, 1945–1948. [Google Scholar] [CrossRef] [PubMed]
- Yergolkar, P.N.; Tandale, B.V.; Arankalle, V.A.; Sathe, P.S.; Sudeep, A.B.; Gandhe, S.S.; Gokhle, M.D.; Jacob, G.P.; Hundekar, S.L.; Mishra, A.C. Chikungunya outbreaks caused by African genotype, India. Emerg. Infect. Dis. 2006, 12, 1580–1583. [Google Scholar] [CrossRef] [PubMed]
- Kularatne, S.A.; Gihan, M.C.; Weerasinghe, S.C.; Gunasena, S. Concurrent outbreaks of chikungunya and dengue fever in Kandy, Sri Lanka, 2006–07: A comparative analysis of clinical and laboratory features. Postgrad. Med. J. 2009, 85, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Peyrefitte, C.N.; Rousset, D.; Pastorino, B.A.; Pouillot, R.; Bessaud, M.; Tock, F.; Mansaray, H.; Merle, O.L.; Pascual, A.M.; Paupy, C.; et al. Chikungunya virus, Cameroon, 2006. Emerg. Infect. Dis. 2007, 13, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Peyrefitte, C.N.; Bessaud, M.; Pastorino, B.A.; Gravier, P.; Plumet, S.; Merle, O.L.; Moltini, I.; Coppin, E.; Tock, F.; Daries, W.; et al. Circulation of chikungunya virus in Gabon, 2006–2007. J. Med. Virol. 2008, 80, 430–433. [Google Scholar] [CrossRef] [PubMed]
- Paupy, C.; Kassa Kassa, F.; Caron, M.; Nkoghe, D.; Leroy, E.M. A chikungunya outbreak associated with the vector Aedes albopictus in remote villages of Gabon. Vector Borne Zoonotic Dis. 2012, 12, 167–169. [Google Scholar] [CrossRef] [PubMed]
- Mombouli, J.V.; Bitsindou, P.; Elion, D.O.; Grolla, A.; Feldmann, H.; Niama, F.R.; Parra, H.J.; Munster, V.J. Chikungunya virus infection, Brazzaville, Republic of Congo, 2011. Emerg. Infect. Dis. 2013, 19, 1542–1543. [Google Scholar] [CrossRef] [PubMed]
- Rezza, G.; Nicoletti, L.; Angelini, R.; Romi, R.; Finarelli, A.C.; Panning, M.; Cordioli, P.; Fortuna, C.; Boros, S.; Magurano, F.; et al. Infection with chikungunya virus in Italy: An outbreak in a temperate region. Lancet 2007, 370, 1840–1846. [Google Scholar] [CrossRef] [PubMed]
- Sam, I.C.; Chan, Y.F.; Chan, S.Y.; Loong, S.K.; Chin, H.K.; Hooi, P.S.; Ganeswrie, R.; Abubakar, S. Chikungunya virus of Asian and Central/East African genotypes in Malaysia. J. Clin. Virol. 2009, 46, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Ng, L.C.; Tan, L.K.; Tan, C.H.; Tan, S.S.; Hapuarachchi, H.C.; Pok, K.Y.; Lai, Y.L.; Lam-Phua, S.G.; Bucht, G.; Lin, R.T.; et al. Entomologic and virologic investigation of chikungunya, Singapore. Emerg. Infect. Dis. 2009, 15, 1243–1249. [Google Scholar] [CrossRef]
- Theamboonlers, A.; Rianthavorn, P.; Praianantathavorn, K.; Wuttirattanakowit, N.; Poovorawan, Y. Clinical and molecular characterization of chikungunya virus in South Thailand. Jpn. J. Infect. Dis. 2009, 62, 303–305. [Google Scholar] [PubMed]
- Wu, D.; Wu, J.; Zhang, Q.; Zhong, H.; Ke, C.; Deng, X.; Guan, D.; Li, H.; Zhang, Y.; Zhou, H.; et al. Chikungunya outbreak in Guangdong Province, China, 2010. Emerg. Infect. Dis. 2012, 18, 493–495. [Google Scholar] [CrossRef] [PubMed]
- Duong, V.; Andries, A.C.; Ngan, C.; Sok, T.; Richner, B.; Asgari-Jirhandeh, N.; Bjorge, S.; Huy, R.; Ly, S.; Laurent, D.; et al. Reemergence of chikungunya virus in Cambodia. Emerg. Infect. Dis. 2012, 18, 2066–2069. [Google Scholar] [CrossRef] [PubMed]
- Wangchuk, S.; Chinnawirotpisan, P.; Dorji, T.; Tobgay, T.; Dorji, T.; Yoon, I.K.; Fernandez, S. Chikungunya fever outbreak, Bhutan, 2012. Emerg. Infect. Dis. 2013, 19, 1681–1684. [Google Scholar] [CrossRef] [PubMed]
- Hapuarachchi, H.C.; Bandara, K.B.; Sumanadasa, S.D.; Hapugoda, M.D.; Lai, Y.L.; Lee, K.S.; Tan, L.K.; Lin, R.T.; Ng, L.F.; Bucht, G.; et al. Re-emergence of chikungunya virus in south-east Asia: Virological evidence from Sri Lanka and Singapore. J. Gen. Virol. 2010, 91, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Rianthavorn, P.; Prianantathavorn, K.; Wuttirattanakowit, N.; Theamboonlers, A.; Poovorawan, Y. An outbreak of chikungunya in Southern Thailand from 2008 to 2009 caused by African strains with A226V mutation. Int. J. Infect. Dis. 2010, 14, e161–e165. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.A.; Gallian, P.; De Lamballerie, X.; Charrel, R.N. First cases of autochthonous dengue fever and chikungunya fever in France: From bad dream to reality! Clin. Microbiol. Infect. 2010, 16, 1702–1704. [Google Scholar] [CrossRef] [PubMed]
- Grandadam, M.; Caro, V.; Plumet, S.; Thiberge, J.M.; Souares, Y.; Failloux, A.B.; Tolou, H.J.; Budelot, M.; Cosserat, D.; Leparc-Goffart, I.; et al. Chikungunya virus, Southeastern France. Emerg. Infect. Dis. 2011, 17, 910–913. [Google Scholar] [CrossRef] [PubMed]
- Delaunay, P.; Jeannin, C.; Schaffner, F.; Marty, P. News on the presence of the tiger mosquito Aedes albopictus in metropolitan France. Arch. Pediatr. 2009, 16, S66–S71. [Google Scholar] [CrossRef] [PubMed]
- Vega-Rua, A.; Zouache, K.; Caro, V.; Diancourt, L.; Delaunay, P.; Grandadam, M.; Failloux, A.B. High efficiency of temperate Aedes albopictus to transmit chikungunya and dengue viruses in the Southeast of France. PLoS One 2013, 8, e59716. [Google Scholar] [CrossRef] [PubMed]
- Paty, M.; Six, C.; Charlet, F.; Heuze, G.; Cochet, A.; Wiegandt, A.; Chappert, J.; Dejour-Salamanca, D.; Guinard, A.; Soler, P.; et al. Large number of imported chikungunya cases in mainland France, 2014: A challenge for surveillance and response. Euro Surveill. 2014, 19, 20856. [Google Scholar] [PubMed]
- Tomasello, D.; Schlagenhauf, P. Chikungunya and dengue autochthonous cases in Europe, 2007–2012. Travel Med. Infect. Dis. 2013, 11, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Napoli, C.; Salcuni, P.; Pompa, M.G.; Declich, S.; Rizzo, C. Estimated imported infections of chikungunya and dengue in Italy, 2008 to 2011. J. Travel Med. 2012, 19, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Leparc-Goffart, I.; Nougairede, A.; Cassadou, S.; Prat, C.; de Lamballerie, X. Chikungunya in the Americas. Lancet 2014, 383, 514. [Google Scholar] [CrossRef] [PubMed]
- Dupont-Rouzeyrol, M.; Caro, V.; Guillaumot, L.; Vazeille, M.; DʼOrtenzio, E.; Thiberge, J.M.; Baroux, N.; Gourinat, A.C.; Grandadam, M.; Failloux, A.B. Chikungunya virus and the mosquito vector Aedes aegypti in New Caledonia (South Pacific region). Vector borne and Zoonotic Dis. 2012, 12, 1036–1041. [Google Scholar] [CrossRef]
- Weaver, S.C. Arrival of chikungunya virus in the new world: Prospects for spread and impact on public health. PLoS Negl. Trop. Dis. 2014, 8, e2921. [Google Scholar] [CrossRef] [PubMed]
- Vega-Rua, A.; Zouache, K.; Girod, R.; Failloux, A.B.; Lourenco-de-Oliveira, R. High level of vector competence of Aedes aegypti and Aedes albopictus from ten American countries as a crucial factor in the spread of chikungunya virus. J. Virol. 2014, 88, 6294–6306. [Google Scholar] [CrossRef] [PubMed]
- ARBOnet. Chikungunya Virus Disease—United States, 2014 Provisional Data Reported to Arbonet; Centers for Disease Control Arboviral Diseases Branch: Fort Collins, CO, USA, 2014.
- Weinbren, M.P.; Haddow, A.J.; Williams, M.C. The occurrence of chikungunya virus in Uganda. I. Isolation from mosquitoes. Trans. R. Soc. Trop. Med. Hygiene 1958, 52, 253–257. [Google Scholar] [CrossRef]
- McCarthy, M.C.; Haberberger, R.L.; Salib, A.W.; Soliman, B.A.; El-Tigani, A.; Khalid, I.O.; Watts, D.M. Evaluation of arthropod-borne viruses and other infectious disease pathogens as the causes of febrile illnesses in the Khartoum Province of Sudan. J. Med. Virol. 1996, 48, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Diallo, M.; Thonnon, J.; Traore-Lamizana, M.; Fontenille, D. Vectors of chikungunya virus in Senegal: Current data and transmission cycles. The American journal of tropical medicine and hygiene 1999, 60, 281–286. [Google Scholar] [PubMed]
- Jupp, P.G.; Kemp, A. What is the potential for future outbreaks of chikungunya, dengue and yellow fever in Southern Africa? South. Afr. Med. J. 1996, 86, 35–37. [Google Scholar]
- Gubler, D.J. Epidemic dengue/dengue hemorrhagic fever as a public health, social and economic problem in the 21st century. Trends Microbiol. 2002, 10, 100–103. [Google Scholar] [CrossRef] [PubMed]
- McClelland, G.A.H. A worldwide survey of variation in scale pattern of the abdominal tergum of Aedes aegypti (l.) (diptera: Culicidae). Trans. R. Entomol. Soc. Lond. 1974, 126, 239–259. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Powell, J.R. A world-wide survey of genetic variation in the yellow fever mosquito, Aedes Aegypti. Genet. Res. 1979, 34, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Failloux, A.B.; Vazeille, M.; Rodhain, F. Geographic genetic variation in populations of the dengue virus vector Aedes aegypti. J. Mol. Evolut. 2002, 55, 653–663. [Google Scholar] [CrossRef]
- Powell, J.R.; Tabachnick, W.J. History of domestication and spread of Aedes aegypti—A review. Memorias do Instituto Oswaldo Cruz 2013, 108, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Juliano, S.A.; Lounibos, L.P.; OʼMeara, G.F. A field test for competitive effects of Aedes albopictus on Aedes aegypti in South Florida: Differences between sites of coexistence and exclusion? Oecologia 2004, 139, 583–593. [Google Scholar] [CrossRef] [PubMed]
- Braks, M.A.; Honorio, N.A.; Lourencqo-De-Oliveira, R.; Juliano, S.A.; Lounibos, L.P. Convergent habitat segregation of Aedes aegypti and Aedes albopictus (diptera: Culicidae) in Southeastern Brazil and Florida. J. Med. Entomol. 2003, 40, 785–794. [Google Scholar] [CrossRef] [PubMed]
- Simard, F.; Nchoutpouen, E.; Toto, J.C.; Fontenille, D. Geographic distribution and breeding site preference of Aedes albopictus and Aedes aegypti (diptera: Culicidae) in Cameroon, Central Africa. J. Med. Entomol. 2005, 42, 726–731. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.E. The history of dengue in tropical Asia and its probable relationship to the mosquito Aedes aegypti. J. Trop. Med. Hygiene 1956, 59, 243–251. [Google Scholar]
- Medlock, J.M.; Hansford, K.M.; Schaffner, F.; Versteirt, V.; Hendrickx, G.; Zeller, H.; Van Bortel, W. A review of the invasive mosquitoes in Europe: Ecology, public health risks, and control options. Vector borne Zoonotic Dis. 2012, 12, 435–447. [Google Scholar] [CrossRef] [PubMed]
- Lounibos, L.P. Invasions by insect vectors of human disease. Annu. Rev. Entomol. 2002, 47, 233–266. [Google Scholar] [CrossRef] [PubMed]
- Rai, K.S. Aedes albopictus in the Americas. Annu. Rev. Entomol. 1991, 36, 459–484. [Google Scholar] [CrossRef] [PubMed]
- Hawley, W.A.; Reiter, P.; Copeland, R.S.; Pumpuni, C.B.; Craig, G.B., Jr. Aedes albopictus in North America: Probable introduction in used tires from Northern Asia. Science 1987, 236, 1114–1116. [Google Scholar] [CrossRef] [PubMed]
- Sprenger, D.; Wuithiranyagool, T. The discovery and distribution of Aedes albopictus in Harris County, Texas. J. Am. Mosq. Control Assoc. 1986, 2, 217–219. [Google Scholar] [PubMed]
- Adhami, J.; Reiter, P. Introduction and establishment of Aedes (Stegomyia) albopictus skuse (diptera: Culicidae) in Albania. J. Am. Mosq. Control Assoc. 1998, 14, 340–343. [Google Scholar] [PubMed]
- Sabatini, A.; Raineri, V.; Trovato, G.; Coluzzi, M. Aedes albopictus in Italy and possible diffusion of the species into the Mediterranean area. Parassitologia 1990, 32, 301–304. [Google Scholar] [PubMed]
- Hawley, W.A. The biology of Aedes albopictus. J. Am. Mosq. Control Association. Suppl. 1988, 1, 1–39. [Google Scholar]
- Paupy, C.; Delatte, H.; Bagny, L.; Corbel, V.; Fontenille, D. Aedes albopictus, an arbovirus vector: From the darkness to the light. Microbes Infect. 2009, 11, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Metselaar, D.; Grainger, C.R.; Oei, K.G.; Reynolds, D.G.; Pudney, M.; Leake, C.J.; Tukei, P.M.; D’Offay, R.M.; Simpson, D.I. An outbreak of type 2 dengue fever in the Seychelles, probably transmitted by Aedes albopictus (skuse). Bull. World Health Organ. 1980, 58, 937–943. [Google Scholar] [PubMed]
- Delatte, H.; Dehecq, J.S.; Thiria, J.; Domerg, C.; Paupy, C.; Fontenille, D. Geographic distribution and developmental sites of Aedes albopictus (diptera: Culicidae) during a chikungunya epidemic event. Vector Borne Zoonotic Dis. 2008, 8, 25–34. [Google Scholar] [CrossRef] [PubMed]
- Gjenero-Margan, I.; Aleraj, B.; Krajcar, D.; Lesnikar, V.; Klobucar, A.; Pem-Novosel, I.; Kurecic-Filipovic, S.; Komparak, S.; Martic, R.; Duricic, S.; et al. Autochthonous dengue fever in Croatia, August-September 2010. Euro Surveillance 2011, 16, 9. [Google Scholar]
- La Ruche, G.; Souares, Y.; Armengaud, A.; Peloux-Petiot, F.; Delaunay, P.; Despres, P.; Lenglet, A.; Jourdain, F.; Leparc-Goffart, I.; Charlet, F.; et al. First two autochthonous dengue virus infections in metropolitan France, September 2010. Euro Surveill. 2010, 15, 19676. [Google Scholar] [PubMed]
- Juliano, S.A.; O'Meara, G.F.; Morrill, J.R.; Cutwa, M.M. Desiccation and thermal tolerance of eggs and the coexistence of competing mosquitoes. Oecologia 2002, 130, 458–469. [Google Scholar] [CrossRef] [PubMed]
- O’Meara, G.F.; Evans, L.F., Jr.; Gettman, A.D.; Cuda, J.P. Spread of Aedes albopictus and decline of Ae. aegypti (diptera: Culicidae) in Florida. J. Med. Entomol. 1995, 32, 554–562. [Google Scholar] [PubMed]
- Juliano, S.A.; Lounibos, L.P. Ecology of invasive mosquitoes: Effects on resident species and on human health. Ecol. Lett. 2005, 8, 558–574. [Google Scholar] [PubMed]
- Kamgang, B.; Brengues, C.; Fontenille, D.; Njiokou, F.; Simard, F.; Paupy, C. Genetic structure of the tiger mosquito, Aedes albopictus, in Cameroon (Central Africa). PLoS One 2011, 6, e20257. [Google Scholar] [CrossRef] [PubMed]
- Kamgang, B.; Ngoagouni, C.; Manirakiza, A.; Nakoune, E.; Paupy, C.; Kazanji, M. Temporal patterns of abundance of Aedes aegypti and Aedes albopictus (diptera: Culicidae) and mitochondrial DNA analysis of Ae. albopictus in the Central African Republic. PLoS Negl. Trop. Dis. 2013, 7, e2590. [Google Scholar] [CrossRef] [PubMed]
- Raharimalala, F.N.; Ravaomanarivo, L.H.; Ravelonandro, P.; Rafarasoa, L.S.; Zouache, K.; Tran-Van, V.; Mousson, L.; Failloux, A.B.; Hellard, E.; Moro, C.V.; et al. Biogeography of the two major arbovirus mosquito vectors, Aedes aegypti and Aedes albopictus (diptera, culicidae), in Madagascar. Parasites Vectors 2012, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Reiter, P.; Sprenger, D. The used tire trade: A mechanism for the worldwide dispersal of container breeding mosquitoes. J. Am. Mosq. Control Assoc. 1987, 3, 494–501. [Google Scholar] [PubMed]
- Nene, V.; Wortman, J.R.; Lawson, D.; Haas, B.; Kodira, C.; Tu, Z.J.; Loftus, B.; Xi, Z.; Megy, K.; Grabherr, M.; et al. Genome sequence of Aedes aegypti, a major arbovirus vector. Science 2007, 316, 1718–1723. [Google Scholar] [PubMed]
- Bonizzoni, M.; Gasperi, G.; Chen, X.; James, A.A. The invasive mosquito species Aedes albopictus: Current knowledge and future perspectives. Trends Parasitol. 2013, 29, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Urdaneta-Marquez, L.; Failloux, A.B. Population genetic structure of Aedes aegypti, the principal vector of dengue viruses. Infect. Genetics Evol. 2011, 11, 253–261. [Google Scholar] [CrossRef]
- Birungi, J.; Munstermann, L.E. Genetic structure of Aedes albopictus (diptera: Culicidae) populations based on mitochondrial nd5 sequences: Evidence for an independent invasion into Brazil and United States. Ann. Entomol. Soc. Am. 2002, 95, 125–132. [Google Scholar] [CrossRef]
- Maia, R.T.; Scarpassa, V.M.; Maciel-Litaiff, L.H.; Tadei, W.P. Reduced levels of genetic variation in Aedes albopictus (diptera: Culicidae) from Manaus, Amazonas State, Brazil, based on analysis of the mitochondrial DNA nd5 gene. Genetics Mol. Res. 2009, 8, 998–1007. [Google Scholar] [CrossRef]
- Mousson, L.; Dauga, C.; Garrigues, T.; Schaffner, F.; Vazeille, M.; Failloux, A.B. Phylogeography of Aedes (stegomyia) aegypti (l.) and Aedes (stegomyia) albopictus (skuse) (diptera: Culicidae) based on mitochondrial DNA variations. Genet. Res. 2005, 86, 1–11. [Google Scholar] [PubMed]
- Usmani-Brown, S.; Cohnstaedt, L.; Munstermann, L.E. Population genetics of Aedes albopictus (diptera: Culicidae) invading populations, using mitochondrial nicotinamide adenine dinucleotide dehydrogenase subunit 5 sequences. Ann. Entomol. Soc. Am. 2009, 102, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, R.G.; Roderick, G.K. Arthropods on islands: Colonization, speciation, and conservation. Annu. Rev. Entomol. 2002, 47, 595–632. [Google Scholar] [CrossRef] [PubMed]
- Beebe, N.W.; Ambrose, L.; Hill, L.A.; Davis, J.B.; Hapgood, G.; Cooper, R.D.; Russell, R.C.; Ritchie, S.A.; Reimer, L.J.; Lobo, N.F.; et al. Tracing the tiger: Population genetics provides valuable insights into the Aedes (stegomyia) albopictus invasion of the Australasian region. PLoS Negl. Trop. Dis. 2013, 7, e2361. [Google Scholar] [CrossRef] [PubMed]
- Fontenille, D.; Rodhain, F. Biology and distribution of Aedes albopictus and Aedes aegypti in Madagascar. J. Am. Mosq. Control Assoc. 1989, 5, 219–225. [Google Scholar] [PubMed]
- Bagny, L.; Arnoux, S.; Delatte, H.; Lajoie, G.; Fontenille, D. Spread of invasive Aedes albopictus and decline of resident Aedes aegypti in urban areas of Mayotte 2007–2010. Biol. Invasions 2012, 14, 1623–1633. [Google Scholar] [CrossRef]
- Diallo, M.; Laganier, R.; Nangouma, A. First record of Ae. albopictus (skuse 1894), in Central African Republic. Trop. Med. Int. Health 2010, 15, 1185–1189. [Google Scholar] [CrossRef] [PubMed]
- Fontenille, D.; Toto, J.C. Aedes (stegomyia) albopictus (skuse), a potential new dengue vector in Southern Cameroon. Emerg. Infect. Dis. 2001, 7, 1066–1067. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Dash, P.K.; Singh, A.K.; Sharma, S.; Gopalan, N.; Rao, P.V.; Parida, M.M.; Reiter, P. Evidence of experimental vertical transmission of emerging novel ECSA genotype of chikungunya virus in Aedes aegypti. PLoS Negl. Trop. Dis. 2014, 8, e2990. [Google Scholar] [CrossRef] [PubMed]
- Thiberville, S.D.; Boisson, V.; Gaudart, J.; Simon, F.; Flahault, A.; de Lamballerie, X. Chikungunya fever: A clinical and virological investigation of outpatients on Reunion Island, South-West Indian Ocean. PLoS Negl. Trop. Dis. 2013, 7, e2004. [Google Scholar] [CrossRef] [PubMed]
- Pesko, K.; Westbrook, C.J.; Mores, C.N.; Lounibos, L.P.; Reiskind, M.H. Effects of infectious virus dose and bloodmeal delivery method on susceptibility of Aedes aegypti and Aedes albopictus to chikungunya virus. J. Med. Entomol. 2009, 46, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.; Moutailler, S.; Madec, Y.; Failloux, A.B. Differential responses of the mosquito Aedes albopictus from the Indian Ocean region to two chikungunya isolates. BMC Ecol. 2010, 10, 8. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vargas, I.; Travanty, E.A.; Keene, K.M.; Franz, A.W.; Beaty, B.J.; Blair, C.D.; Olson, K.E. RNA interference, arthropod-borne viruses, and mosquitoes. Virus Res. 2004, 102, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008, 4, e1000098. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Dimopoulos, G. The toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev. Compar. Immunol. 2010, 34, 625–629. [Google Scholar] [CrossRef]
- Souza-Neto, J.A.; Sim, S.; Dimopoulos, G. An evolutionary conserved function of the jak-stat pathway in anti-dengue defense. Proc. Natl. Acad. Sci. USA 2009, 106, 17841–17846. [Google Scholar] [CrossRef] [PubMed]
- Sanders, H.R.; Foy, B.D.; Evans, A.M.; Ross, L.S.; Beaty, B.J.; Olson, K.E.; Gill, S.S. Sindbis virus induces transport processes and alters expression of innate immunity pathway genes in the midgut of the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 2005, 35, 1293–1307. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Vargas, I.; Scott, J.C.; Poole-Smith, B.K.; Franz, A.W.; Barbosa-Solomieu, V.; Wilusz, J.; Olson, K.E.; Blair, C.D. Dengue virus type 2 infections of Aedes aegypti are modulated by the mosquito's RNA interference pathway. PLoS Pathog. 2009, 5, e1000299. [Google Scholar] [CrossRef] [PubMed]
- Keene, K.M.; Foy, B.D.; Sanchez-Vargas, I.; Beaty, B.J.; Blair, C.D.; Olson, K.E. RNA interference acts as a natural antiviral response to o’nyong-nyong virus (alphavirus; Togaviridae) infection of Anopheles gambiae. Proc. Natl. Acad. Sci. USA 2004, 101, 17240–17245. [Google Scholar] [CrossRef] [PubMed]
- Campbell, C.L.; Keene, K.M.; Brackney, D.E.; Olson, K.E.; Blair, C.D.; Wilusz, J.; Foy, B.D. Aedes aegypti uses RNA interference in defense against sindbis virus infection. BMC Microbiol. 2008, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Myles, K.M.; Wiley, M.R.; Morazzani, E.M.; Adelman, Z.N. Alphavirus-derived small RNAs modulate pathogenesis in disease vector mosquitoes. Proc. Natl. Acad. Sci. USA 2008, 105, 19938–19943. [Google Scholar] [CrossRef] [PubMed]
- Cirimotich, C.M.; Scott, J.C.; Phillips, A.T.; Geiss, B.J.; Olson, K.E. Suppression of RNA interference increases alphavirus replication and virus-associated mortality in Aedes aegypti mosquitoes. BMC Microbiol. 2009, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Khoo, C.C.; Piper, J.; Sanchez-Vargas, I.; Olson, K.E.; Franz, A.W. The RNA interference pathway affects midgut infection- and escape barriers for sindbis virus in Aedes aegypti. BMC Microbiol. 2010, 10, 130. [Google Scholar] [CrossRef] [PubMed]
- Donald, C.L.; Kohl, A.; Schnettler, E. New insights into control of arbovirus replication and spread by insect RNA interference pathways. Insects 2012, 3, 511–531. [Google Scholar]
- McFarlane, M.; Arias-Goeta, C.; Martin, E.; OʼHara, Z.; Lulla, A.; Mousson, L.; Rainey, S.M.; Misbah, S.; Schnettler, E.; Donald, C.L.; et al. Characterization of Aedes aegypti innate-immune pathways that limit chikungunya virus replication. PLoS Negl. Trop. Dis. 2014, 8, e2994. [Google Scholar] [CrossRef] [PubMed]
- Schnettler, E.; Donald, C.L.; Human, S.; Watson, M.; Siu, R.W.; McFarlane, M.; Fazakerley, J.K.; Kohl, A.; Fragkoudis, R. Knockdown of piRNA pathway proteins results in enhanced semliki forest virus production in mosquito cells. J. Gen. Virol. 2013, 94, 1680–1689. [Google Scholar] [CrossRef] [PubMed]
- McGraw, E.A.; OʼNeill, S.L. Beyond insecticides: New thinking on an ancient problem. Nat. Rev. Microbiol. 2013, 11, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Franz, A.W.; Sanchez-Vargas, I.; Adelman, Z.N.; Blair, C.D.; Beaty, B.J.; James, A.A.; Olson, K.E. Engineering RNA interference-based resistance to dengue virus type 2 in genetically modified Aedes aegypti. Proc. Natl. Acad. Sci. USA 2006, 103, 4198–4203. [Google Scholar] [CrossRef] [PubMed]
- Mathur, G.; Sanchez-Vargas, I.; Alvarez, D.; Olson, K.E.; Marinotti, O.; James, A.A. Transgene-mediated suppression of dengue viruses in the salivary glands of the yellow fever mosquito, Aedes aegypti. Insect Mol. Biol. 2010, 19, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Travanty, E.A.; Adelman, Z.N.; Franz, A.W.; Keene, K.M.; Beaty, B.J.; Blair, C.D.; James, A.A.; Olson, K.E. Using RNA interference to develop dengue virus resistance in genetically modified Aedes aegypti. Insect Biochem. Mol. Biol. 2004, 34, 607–613. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L.; McKemey, A.; Nimmo, D.; Neira Oviedo, M.; Lacroix, R.; Matzen, K.; Beech, C. Genetic control of Aedes mosquitoes. Pathog. Glob. Health 2013, 107, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Alphey, L. Natural and engineered mosquito immunity. J. Biol. 2009, 8, 40. [Google Scholar] [CrossRef] [PubMed]
- Fraser, M.J., Jr. Insect transgenesis: Current applications and future prospects. Annu. Rev. Entomol. 2012, 57, 267–289. [Google Scholar] [CrossRef] [PubMed]
- Walker, T.; Johnson, P.H.; Moreira, L.A.; Iturbe-Ormaetxe, I.; Frentiu, F.D.; McMeniman, C.J.; Leong, Y.S.; Dong, Y.; Axford, J.; Kriesner, P.; et al. The wMel wolbachia strain blocks dengue and invades caged Aedes aegypti populations. Nature 2011, 476, 450–453. [Google Scholar] [CrossRef] [PubMed]
- Sinkins, S.P.; Gould, F. Gene drive systems for insect disease vectors. Nat. Rev. Genet. 2006, 7, 427–435. [Google Scholar] [CrossRef]
- Chen, C.H.; Huang, H.; Ward, C.M.; Su, J.T.; Schaeffer, L.V.; Guo, M.; Hay, B.A. A synthetic maternal-effect selfish genetic element drives population replacement in Drosophila. Science 2007, 316, 597–600. [Google Scholar] [CrossRef] [PubMed]
- McMeniman, C.J.; Lane, R.V.; Cass, B.N.; Fong, A.W.; Sidhu, M.; Wang, Y.F.; OʼNeill, S.L. Stable introduction of a life-shortening wolbachia infection into the mosquito Aedes aegypti. Science 2009, 323, 141–144. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.M.; Brownlie, J.C.; O’Neill, S.L.; Johnson, K.N. Wolbachia and virus protection in insects. Science 2008, 322, 702. [Google Scholar] [CrossRef] [PubMed]
- Moreira, L.A.; Iturbe-Ormaetxe, I.; Jeffery, J.A.; Lu, G.; Pyke, A.T.; Hedges, L.M.; Rocha, B.C.; Hall-Mendelin, S.; Day, A.; Riegler, M.; et al. A wolbachia symbiont in Aedes aegypti limits infection with dengue, chikungunya, and plasmodium. Cell 2009, 139, 1268–1278. [Google Scholar] [CrossRef] [PubMed]
- Hancock, P.A.; Sinkins, S.P.; Godfray, H.C. Strategies for introducing wolbachia to reduce transmission of mosquito-borne diseases. PLoS Negl. Trop. Dis. 2011, 5, e1024. [Google Scholar] [CrossRef] [PubMed]
- Zouache, K.; Voronin, D.; Tran-Van, V.; Mousson, L.; Failloux, A.B.; Mavingui, P. Persistent wolbachia and cultivable bacteria infection in the reproductive and somatic tissues of the mosquito vector Aedes albopictus. PLoS One 2009, 4, e6388. [Google Scholar] [CrossRef] [PubMed]
- Mousson, L.; Martin, E.; Zouache, K.; Madec, Y.; Mavingui, P.; Failloux, A.B. Wolbachia modulates chikungunya replication in Aedes albopictus. Mol. Ecol. 2010, 19, 1953–1964. [Google Scholar] [PubMed]
- Mousson, L.; Zouache, K.; Arias-Goeta, C.; Raquin, V.; Mavingui, P.; Failloux, A.B. The native wolbachia symbionts limit transmission of dengue virus in Aedes albopictus. PLoS Negl. Trop. Dis. 2012, 6, e1989. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Bian, G.; Pan, X.; Xi, Z. Wolbachia induces density-dependent inhibition to dengue virus in mosquito cells. PLoS Negl. Trop. Dis. 2012, 6, e1754. [Google Scholar] [PubMed]
- Blagrove, M.S.; Arias-Goeta, C.; Di Genua, C.; Failloux, A.B.; Sinkins, S.P. A wolbachia wMel transinfection in Aedes albopictus is not detrimental to host fitness and inhibits chikungunya virus. PLoS Negl. Trop. Dis. 2013, 7, e2152. [Google Scholar] [CrossRef] [PubMed]
- van den Hurk, A.F.; Hall-Mendelin, S.; Pyke, A.T.; Frentiu, F.D.; McElroy, K.; Day, A.; Higgs, S.; OʼNeill, S.L. Impact of wolbachia on infection with chikungunya and yellow fever viruses in the mosquito vector Aedes aegypti. PLoS Negl. Trop. Dis. 2012, 6, e1892. [Google Scholar] [CrossRef] [PubMed]
- Rances, E.; Ye, Y.H.; Woolfit, M.; McGraw, E.A.; OʼNeill, S.L. The relative importance of innate immune priming in wolbachia-mediated dengue interference. PLoS Pathog. 2012, 8, e1002548. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive oxygen species (ros)-dependent activation of the toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar] [CrossRef] [PubMed]
- Caragata, E.P.; Rances, E.; Hedges, L.M.; Gofton, A.W.; Johnson, K.N.; OʼNeill, S.L.; McGraw, E.A. Dietary cholesterol modulates pathogen blocking by wolbachia. PLoS Pathog. 2013, 9, e1003459. [Google Scholar] [CrossRef] [PubMed]
- Zouache, K.; Raharimalala, F.N.; Raquin, V.; Tran-Van, V.; Raveloson, L.H.; Ravelonandro, P.; Mavingui, P. Bacterial diversity of field-caught mosquitoes, Aedes albopictus and Aedes aegypti, from different geographic regions of Madagascar. FEMS Microbiol. Ecol. 2011, 75, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Minard, G.; Tran, F.H.; Raharimalala, F.N.; Hellard, E.; Ravelonandro, P.; Mavingui, P.; Valiente Moro, C. Prevalence, genomic and metabolic profiles of acinetobacter and asaia associated with field-caught Aedes albopictus from Madagascar. FEMS Microbiol. Ecol. 2013, 83, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Souza-Neto, J.; Torres Cosme, R.; Rovira, J.; Ortiz, A.; Pascale, J.M.; Dimopoulos, G. Reciprocal tripartite interactions between the Aedes aegypti midgut microbiota, innate immune system and dengue virus influences vector competence. PLoS Negl. Trop. Dis. 2012, 6, e1561. [Google Scholar] [CrossRef] [PubMed]
- Apte-Deshpande, A.D.; Paingankar, M.S.; Gokhale, M.D.; Deobagkar, D.N. Serratia odorifera mediated enhancement in susceptibility of Aedes aegypti for chikungunya virus. Indian J. Med. Res. 2014, 139, 762–768. [Google Scholar] [PubMed]
- Black, W.C.; Bennett, K.E.; Gorrochotegui-Escalante, N.; Barillas-Mury, C.V.; Fernandez-Salas, I.; de Lourdes Munoz, M.; Farfan-Ale, J.A.; Olson, K.E.; Beaty, B.J. Flavivirus susceptibility in Aedes aegypti. Arch. Med. Res. 2002, 33, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.E.; Flick, D.; Fleming, K.H.; Jochim, R.; Beaty, B.J.; Black, W.C.T. Quantitative trait loci that control dengue-2 virus dissemination in the mosquito Aedes aegypti. Genetics 2005, 170, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Stapleford, K.A.; Coffey, L.L.; Lay, S.; Borderia, A.V.; Duong, V.; Isakov, O.; Rozen-Gagnon, K.; Arias-Goeta, C.; Blanc, H.; Beaucourt, S.; et al. Emergence and transmission of arbovirus evolutionary intermediates with epidemic potential. Cell. Host Microbe 2014, 15, 706–716. [Google Scholar] [CrossRef] [PubMed]
- Myers, R.M.; Carey, D.E. Concurrent isolation from patient of two arboviruses, chikungunya and dengue type 2. Science 1967, 157, 1307–1308. [Google Scholar] [CrossRef] [PubMed]
- Hapuarachchi, H.A.; Bandara, K.B.; Hapugoda, M.D.; Williams, S.; Abeyewickreme, W. Laboratory confirmation of dengue and chikungunya co-infection. Ceylon Med. J. 2008, 53, 104–105. [Google Scholar] [PubMed]
- Ezzedine, K.; Cazanave, C.; Pistone, T.; Receveur, M.C.; Neau, D.; Ragnaud, J.M.; Malvy, D. Dual infection by chikungunya virus and other imported infectious agent in a traveller returning from India. Travel Med. Infect. Dis. 2008, 6, 152–154. [Google Scholar] [CrossRef] [PubMed]
- Chahar, H.S.; Bharaj, P.; Dar, L.; Guleria, R.; Kabra, S.K.; Broor, S. Co-infections with chikungunya virus and dengue virus in Delhi, India. Emerg. Infect. Dis. 2009, 15, 1077–1080. [Google Scholar] [CrossRef] [PubMed]
- Schilling, S.; Emmerich, P.; Gunther, S.; Schmidt-Chanasit, J. Dengue and chikungunya virus co-infection in a German traveller. J. Clin. Virol. 2009, 45, 163–164. [Google Scholar] [CrossRef] [PubMed]
- Nayar, S.K.; Noridah, O.; Paranthaman, V.; Ranjit, K.; Norizah, I.; Chem, Y.K.; Mustafa, B.; Chua, K.B. Co-infection of dengue virus and chikungunya virus in two patients with acute febrile illness. Med. J. Malays. 2007, 62, 335–336. [Google Scholar]
- Leroy, E.M.; Nkoghe, D.; Ollomo, B.; Nze-Nkogue, C.; Becquart, P.; Grard, G.; Pourrut, X.; Charrel, R.; Moureau, G.; Ndjoyi-Mbiguino, A.; et al. Concurrent chikungunya and dengue virus infections during simultaneous outbreaks, Gabon, 2007. Emerg. Infect. Dis. 2009, 15, 591–593. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.F.; Su, C.L.; Shu, P.Y.; Yang, C.F.; Liao, T.L.; Cheng, C.H.; Hu, H.C.; Huang, J.H. Concurrent isolation of chikungunya virus and dengue virus from a patient with coinfection resulting from a trip to Singapore. J. Clin. Microbiol. 2010, 48, 4586–4589. [Google Scholar] [CrossRef] [PubMed]
- Parreira, R.; Centeno-Lima, S.; Lopes, A.; Portugal-Calisto, D.; Constantino, A.; Nina, J. Dengue virus serotype 4 and chikungunya virus coinfection in a traveller returning from Luanda, Angola, January 2014. Euro Surveill. 2014, 19, 10. [Google Scholar]
- Caron, M.; Paupy, C.; Grard, G.; Becquart, P.; Mombo, I.; Nso, B.B.; Kassa Kassa, F.; Nkoghe, D.; Leroy, E.M. Recent introduction and rapid dissemination of chikungunya virus and dengue virus serotype 2 associated with human and mosquito coinfections in Gabon, Central Africa. Clin. Infect. Dis. 2012, 55, e45–e53. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Mousson, L.; Martin, E.; Failloux, A.B. Orally co-infected Aedes albopictus from La Reunion Island, Indian Ocean, can deliver both dengue and chikungunya infectious viral particles in their saliva. PLoS Negl. Trop. Dis. 2010, 4, e706. [Google Scholar] [CrossRef] [PubMed]
- Courtois, G.; Osterrieth, P.; Blanes Ridaura, G. Isolation of the yellow fever virus in the Belgian Congo. Ann. Soc. Belge Med. Tropicale 1960, 40, 29–60. [Google Scholar]
- Osterrieth, P.; Rathe, E.; Deleplanque-Liegeois, P. Simultaneous isolation of the yellow fever and chikungunya viruses at Bili (Haut-Uele, Belgian Congo). Ann. Soc. Belge Med. Tropicale 1961, 41, 207–212. [Google Scholar]
- Filipe, A.F.; Pinto, M.R. Arbovirus studies in Luanda, Angola. 2. Virological and serological studies during an outbreak of dengue-like disease caused by the chikungunya virus. Bull. World Health Organ. 1973, 49, 37–40. [Google Scholar] [PubMed]
- Gould, L.H.; Osman, M.S.; Farnon, E.C.; Griffith, K.S.; Godsey, M.S.; Karch, S.; Mulenda, B.; El Kholy, A.; Grandesso, F.; de Radigues, X.; et al. An outbreak of yellow fever with concurrent chikungunya virus transmission in South Kordofan, Sudan, 2005. Trans. R. Soc. Trop. Med. Hygiene 2008, 102, 1247–1254. [Google Scholar] [CrossRef]
- Grard, G.; Caron, M.; Mombo, I.M.; Nkoghe, D.; Mboui Ondo, S.; Jiolle, D.; Fontenille, D.; Paupy, C.; Leroy, E.M. Zika virus in Gabon (Central Africa)—2007: A new threat from Aedes albopictus? PLoS Negl. Trop. Dis. 2014, 8, e2681. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, P.M.; Rico-Hesse, R. Differential susceptibility of Aedes aegypti to infection by the American and Southeast Asian genotypes of dengue type 2 virus. Vector Borne Zoonotic Dis. 2001, 1, 159–168. [Google Scholar] [CrossRef] [PubMed]
- Cologna, R.; Armstrong, P.M.; Rico-Hesse, R. Selection for virulent dengue viruses occurs in humans and mosquitoes. J. Virol. 2005, 79, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Brault, A.C.; Powers, A.M.; Ortiz, D.; Estrada-Franco, J.G.; Navarro-Lopez, R.; Weaver, S.C. Venezuelan equine encephalitis emergence: Enhanced vector infection from a single amino acid substitution in the envelope glycoprotein. Proc. Natl. Acad. Sci. USA 2004, 101, 11344–11349. [Google Scholar] [CrossRef] [PubMed]
- Deardorff, E.R.; Weaver, S.C. Vector competence of Culex (Melanoconion) taeniopus for equine-virulent subtype ie strains of venezuelan equine encephalitis virus. Am. J. Trop. Med. Hygiene 2010, 82, 1047–1052. [Google Scholar] [CrossRef]
- Tsetsarkin, K.A.; Weaver, S.C. Sequential adaptive mutations enhance efficient vector switching by chikungunya virus and its epidemic emergence. PLoS Pathog. 2011, 7, e1002412. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C. The Evolution and Emergence of RNA Viruses; Oxford University Press: New York, NY, USA, 2009. [Google Scholar]
- Rozen-Gagnon, K.; Stapleford, K.A.; Mongelli, V.; Blanc, H.; Failloux, A.B.; Saleh, M.C.; Vignuzzi, M. Alphavirus mutator variants present host-specific defects and attenuation in mammalian and insect models. PLoS Pathog. 2014, 10, e1003877. [Google Scholar] [CrossRef] [PubMed]
- Coffey, L.L.; Beeharry, Y.; Borderia, A.V.; Blanc, H.; Vignuzzi, M. Arbovirus high fidelity variant loses fitness in mosquitoes and mice. Proc. Natl. Acad. Sci. USA 2011, 108, 16038–16043. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.T.; Ebel, G.D.; Lanciotti, R.S.; Brault, A.C.; Guzman, H.; Siirin, M.; Lambert, A.; Parsons, R.E.; Beasley, D.W.; Novak, R.J.; et al. Phylogenetic analysis of North American West Nile virus isolates, 2001–2004: Evidence for the emergence of a dominant genotype. Virology 2005, 342, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Ebel, G.D.; Carricaburu, J.; Young, D.; Bernard, K.A.; Kramer, L.D. Genetic and phenotypic variation of West Nile virus in New York, 2000–2003. Am. J. Trop. Med. Hygiene 2004, 71, 493–500. [Google Scholar]
- Parameswaran, P.; Charlebois, P.; Tellez, Y.; Nunez, A.; Ryan, E.M.; Malboeuf, C.M.; Levin, J.Z.; Lennon, N.J.; Balmaseda, A.; Harris, E.; et al. Genome-wide patterns of intrahuman dengue virus diversity reveal associations with viral phylogenetic clade and interhost diversity. J. Virol. 2012, 86, 8546–8558. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.E.; Morales, N.M.; Alto, B.W.; Remold, S.K. Role of evolved host breadth in the initial emergence of an RNA virus. Evolution 2010, 64, 3273–3286. [Google Scholar] [CrossRef] [PubMed]
- Coffey, L.L.; Forrester, N.; Tsetsarkin, K.; Vasilakis, N.; Weaver, S.C. Factors shaping the adaptive landscape for arboviruses: Implications for the emergence of disease. Future Microbiol. 2013, 8, 155–176. [Google Scholar] [CrossRef] [PubMed]
- Coffey, L.L.; Vignuzzi, M. Host alternation of chikungunya virus increases fitness while restricting population diversity and adaptability to novel selective pressures. J. Virol. 2011, 85, 1025–1035. [Google Scholar] [CrossRef] [PubMed]
- Muller, H.J. The relation of recombination to mutational advance. Mutat. Res. 1964, 1, 2–9. [Google Scholar]
- Weaver, S.C.; Brault, A.C.; Kang, W.; Holland, J.J. Genetic and fitness changes accompanying adaptation of an arbovirus to vertebrate and invertebrate cells. J. Virol. 1999, 73, 4316–4326. [Google Scholar] [PubMed]
- Duarte, E.A.; Clarke, D.K.; Moya, A.; Elena, S.F.; Domingo, E.; Holland, J. Many-trillionfold amplification of single RNA virus particles fails to overcome the Mullerʼs ratchet effect. J. Virol. 1993, 67, 3620–3623. [Google Scholar] [PubMed]
- Smith, D.R.; Adams, A.P.; Kenney, J.L.; Wang, E.; Weaver, S.C. Venezuelan equine encephalitis virus in the mosquito vector Aedes taeniorhynchus: Infection initiated by a small number of susceptible epithelial cells and a population bottleneck. Virology 2008, 372, 176–186. [Google Scholar] [CrossRef]
- Kenney, J.L.; Adams, A.P.; Gorchakov, R.; Leal, G.; Weaver, S.C. Genetic and anatomic determinants of enzootic venezuelan equine encephalitis virus infection of Culex (Melanoconion) taeniopus. PLoS Negl. Trop. Dis. 2012, 6, e1606. [Google Scholar] [CrossRef] [PubMed]
- Forrester, N.L.; Guerbois, M.; Seymour, R.L.; Spratt, H.; Weaver, S.C. Vector-borne transmission imposes a severe bottleneck on an RNA virus population. PLoS Pathog. 2012, 8, e1002897. [Google Scholar] [CrossRef] [PubMed]
- Brackney, D.E.; Pesko, K.N.; Brown, I.K.; Deardorff, E.R.; Kawatachi, J.; Ebel, G.D. West Nile virus genetic diversity is maintained during transmission by Culex pipiens quinquefasciatus mosquitoes. PLoS One 2011, 6, e24466. [Google Scholar] [CrossRef] [PubMed]
- Coffey, L.L.; Vasilakis, N.; Brault, A.C.; Powers, A.M.; Tripet, F.; Weaver, S.C. Arbovirus evolution in vivo is constrained by host alternation. Proc. Natl. Acad. Sci. USA 2008, 105, 6970–6975. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Aguilar, P.V.; Coffey, L.L.; Gromowski, G.D.; Wang, E.; Weaver, S.C. Venezuelan equine encephalitis virus transmission and effect on pathogenesis. Emerg. Infect. Dis. 2006, 12, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Moreno, D.; Vargas, I.S.; Olson, K.E.; Harrington, L.C. Modeling dynamic introduction of chikungunya virus in the United States. PLoS Negl. Trop. Dis. 2012, 6, e1918. [Google Scholar] [CrossRef] [PubMed]
- Eisen, L.; Monaghan, A.J.; Lozano-Fuentes, S.; Steinhoff, D.F.; Hayden, M.H.; Bieringer, P.E. The impact of temperature on the bionomics of Aedes (stegomyia) aegypti, with special reference to the cool geographic range margins. J. Med. Entomol. 2014, 51, 496–516. [Google Scholar] [CrossRef] [PubMed]
- Zouache, K.; Fontaine, A.; Vega-Rua, A.; Mousson, L.; Thiberge, J.M.; Lourenco-De-Oliveira, R.; Caro, V.; Lambrechts, L.; Failloux, A.B. Three-way interactions between mosquito population, viral strain and temperature underlying chikungunya virus transmission potential. Proc. Biol. Sci. 2014, 281, 1–8. [Google Scholar] [CrossRef]
- Tsetsarkin, K.A.; Chen, R.; Leal, G.; Forrester, N.; Higgs, S.; Huang, J.; Weaver, S.C. Chikungunya virus emergence is constrained in Asia by lineage-specific adaptive landscapes. Proc. Natl. Acad. Sci. USA 2011, 108, 7872–7877. [Google Scholar] [CrossRef] [PubMed]
- Lanciotti, R.S.; Kosoy, O.L.; Laven, J.J.; Panella, A.J.; Velez, J.O.; Lambert, A.J.; Campbell, G.L. Chikungunya virus in US travelers returning from India, 2006. Emerg. Infect. Dis. 2007, 13, 764–767. [Google Scholar] [CrossRef] [PubMed]
- Wilke, A.B.; Marrelli, M.T. Genetic control of mosquitoes: Population suppression strategies. Rev. Inst. Med. Trop. Sao Paulo 2012, 54, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Weaver, S.C.; Osorio, J.E.; Livengood, J.A.; Chen, R.; Stinchcomb, D.T. Chikungunya virus and prospects for a vaccine. Expert Rev. Vaccines 2012, 11, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Paupy, C.; Ollomo, B.; Kamgang, B.; Moutailler, S.; Rousset, D.; Demanou, M.; Herve, J.P.; Leroy, E.; Simard, F. Comparative role of Aedes albopictus and Aedes aegypti in the emergence of dengue and chikungunya in Central Africa. Vector Borne Zoonotic Dis. 2010, 10, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Turell, M.J.; Beaman, J.R.; Tammariello, R.F. Susceptibility of selected strains of Aedes aegypti and Aedes albopictus (diptera: Culicidae) to chikungunya virus. J. Med. Entomol. 1992, 29, 49–53. [Google Scholar] [PubMed]
- Shah, K.V.; Gilotra, S.K.; Gibbs, C.J., Jr.; Rozeboom, L.E. Laboratory studies of transmission of chikungunya virus by mosquitoes: A preliminary report. Indian J. Med. Res. 1964, 52, 703–709. [Google Scholar] [PubMed]
- Girod, R.; Gaborit, P.; Marrama, L.; Etienne, M.; Ramdini, C.; Rakotoarivony, I.; Dollin, C.; Carinci, R.; Issaly, J.; Dusfour, I.; et al. High susceptibility to chikungunya virus of Aedes aegypti from the French West Indies and French Guiana. Trop. Med. Int. Health 2011, 16, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Vanlandingham, D.L.; Hong, C.; Klingler, K.; Tsetsarkin, K.; McElroy, K.L.; Powers, A.M.; Lehane, M.J.; Higgs, S. Differential infectivities of oʼnyong-nyong and chikungunya virus isolates in Anopheles gambiae and Aedes aegypti mosquitoes. Am. J. Trop. Med. Hygiene 2005, 72, 616–621. [Google Scholar]
- Mangiafico, J.A. Chikungunya virus infection and transmission in five species of mosquito. Am. J. Trop. Med. Hygiene 1971, 20, 642–645. [Google Scholar]
- Vazeille, M.; Yebakima, A.; Lourenco-de-Oliveira, R.; Andriamahefazafy, B.; Correira, A.; Rodrigues, J.M.; Veiga, A.; Moreira, A.; Leparc-Goffart, I.; Grandadam, M.; et al. Oral receptivity of Aedes aegypti from Cape Verde for yellow fever, dengue, and chikungunya viruses. Vector Borne Zoonotic Dis. 2013, 13, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, J.; Ritchie, S.A.; van den Hurk, A.F. Aedes albopictus (diptera: Culicidae) as a potential vector of endemic and exotic arboviruses in Australia. J. Med. Entomol. 2014, 51, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Tesh, R.B.; Gubler, D.J.; Rosen, L. Variation among geographic strains of Aedes albopictus in susceptibility to infection with chikungunya virus. Am. J. Trop. Med. Hygiene 1976, 25, 326–335. [Google Scholar]
- Haddad, N.; Mousson, L.; Vazeille, M.; Chamat, S.; Tayeh, J.; Osta, M.A.; Failloux, A.B. Aedes albopictus in Lebanon, a potential risk of arboviruses outbreak. BMC Infect. Dis. 2012, 12, 300. [Google Scholar] [CrossRef] [PubMed]
- Talbalaghi, A.; Moutailler, S.; Vazeille, M.; Failloux, A.B. Are Aedes albopictus or other mosquito species from Northern Italy competent to sustain new arboviral outbreaks? Med. Vet. Entomol. 2010, 24, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Vazeille, M.; Jeannin, C.; Martin, E.; Schaffner, F.; Failloux, A.B. Chikungunya: A risk for Mediterranean countries? Acta Trop. 2008, 105, 200–202. [Google Scholar] [CrossRef] [PubMed]
- Moutailler, S.; Barre, H.; Vazeille, M.; Failloux, A.B. Recently introduced Aedes albopictus in Corsica is competent to chikungunya virus and in a lesser extent to dengue virus. Trop. Med. Int. Health 2009, 14, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.D.; Chin, P.; Cane, R.P.; Kauffman, E.B.; Mackereth, G. Vector competence of New Zealand mosquitoes for selected arboviruses. Am. J. Trop. Med. Hygiene 2011, 85, 182–189. [Google Scholar] [CrossRef]
- Jupp, P.G.; McIntosh, B.M.; Dos Santos, I.; DeMoor, P. Laboratory vector studies on six mosquito and one tick species with chikungunya virus. Trans. R. Soc. Trop. Med. Hygiene 1981, 75, 15–19. [Google Scholar] [CrossRef]
- Ledermann, J.P.; Guillaumot, L.; Yug, L.; Saweyog, S.C.; Tided, M.; Machieng, P.; Pretrick, M.; Marfel, M.; Griggs, A.; Bel, M.; et al. Aedes hensilli as a potential vector of chikungunya and zika viruses. PLoS Negl. Trop. Dis. 2014, 8, e3188. [Google Scholar] [CrossRef] [PubMed]
- Diagne, C.T.; Faye, O.; Guerbois, M.; Knight, R.; Diallo, D.; Faye, O.; Ba, Y.; Dia, I.; Faye, O.; Weaver, S.C.; et al. Vector competence of Aedes aegypti and Aedes vittatus (diptera: Culicidae) from Senegal and Cape Verde archipelago for West African lineages of chikungunya virus. Am. J. Trop. Med. Hygiene 2014, 91, 635–641. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Coffey, L.L.; Failloux, A.-B.; Weaver, S.C. Chikungunya Virus–Vector Interactions. Viruses 2014, 6, 4628-4663. https://doi.org/10.3390/v6114628
Coffey LL, Failloux A-B, Weaver SC. Chikungunya Virus–Vector Interactions. Viruses. 2014; 6(11):4628-4663. https://doi.org/10.3390/v6114628
Chicago/Turabian StyleCoffey, Lark L., Anna-Bella Failloux, and Scott C. Weaver. 2014. "Chikungunya Virus–Vector Interactions" Viruses 6, no. 11: 4628-4663. https://doi.org/10.3390/v6114628
APA StyleCoffey, L. L., Failloux, A.-B., & Weaver, S. C. (2014). Chikungunya Virus–Vector Interactions. Viruses, 6(11), 4628-4663. https://doi.org/10.3390/v6114628




