Vector-Virus Interactions and Transmission Dynamics of West Nile Virus
Abstract
:1. Introduction
2. Vector
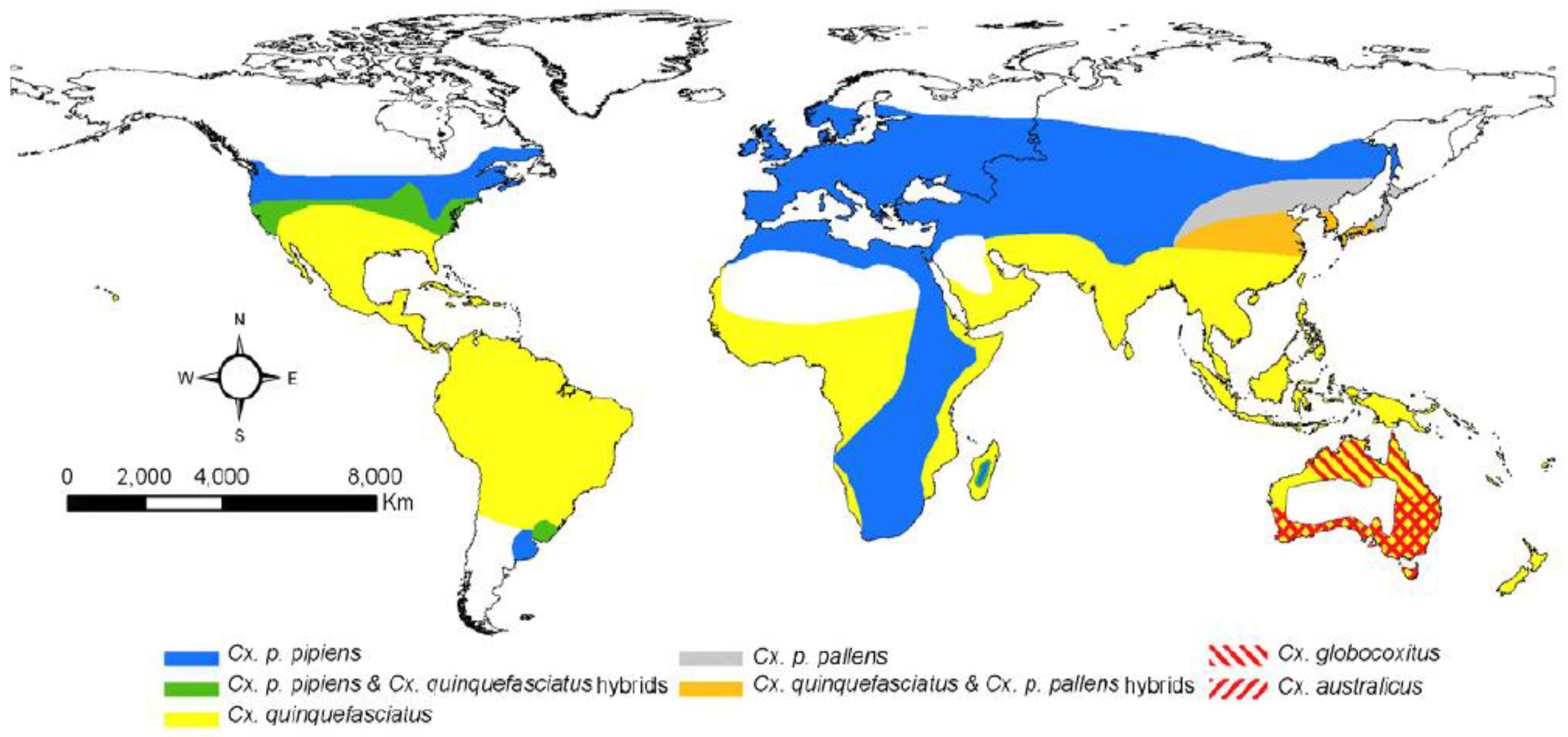
2.1. Vector Distribution and Genetics
2.2. Vectorial Capacity
2.2.1. Vector Competence (b) and Extrinsic Incubation Period (n)
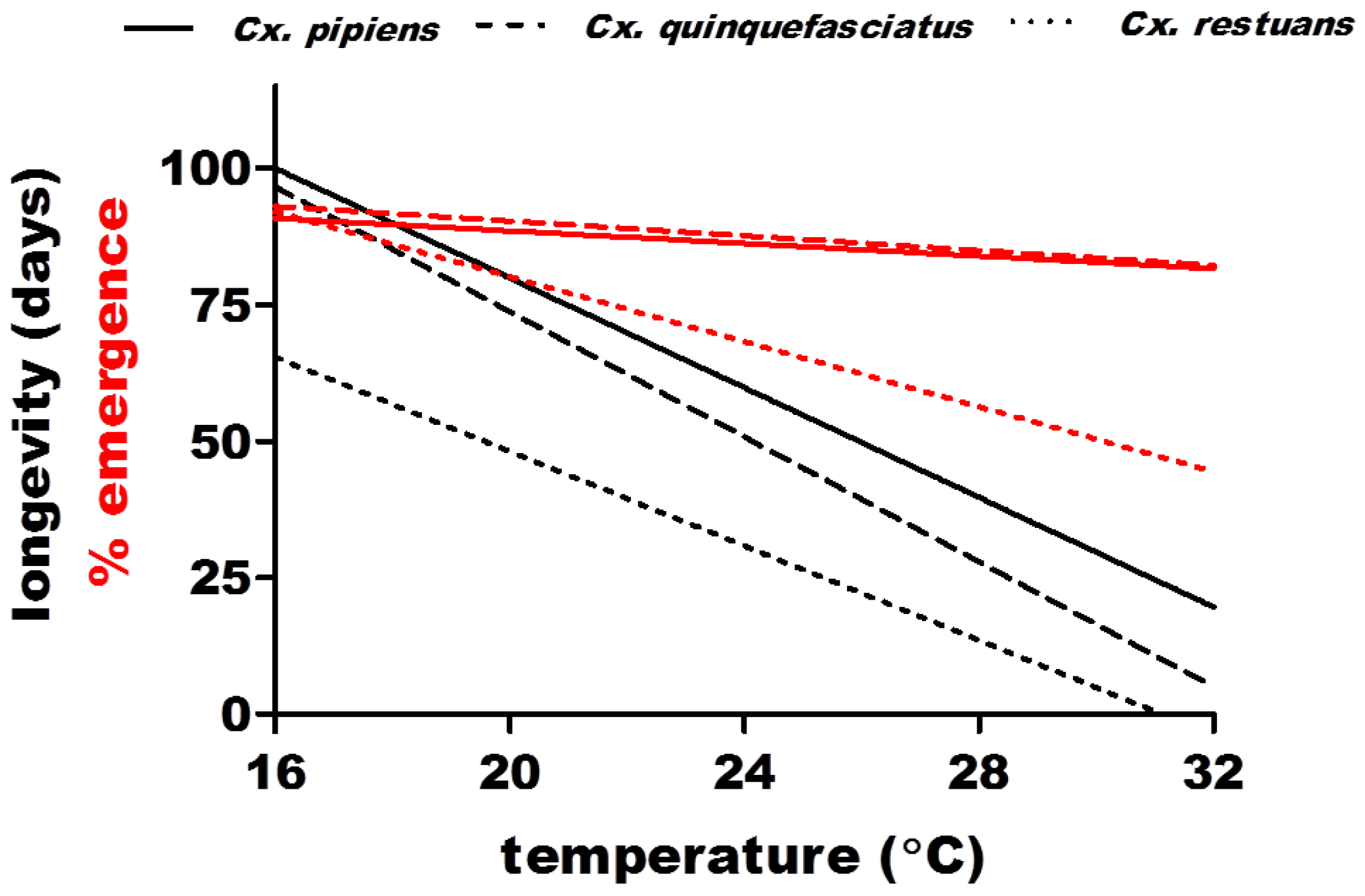
2.2.2. Population Density (m) and Daily Survival (p)
2.2.3. Blood Feeding Behavior (a)
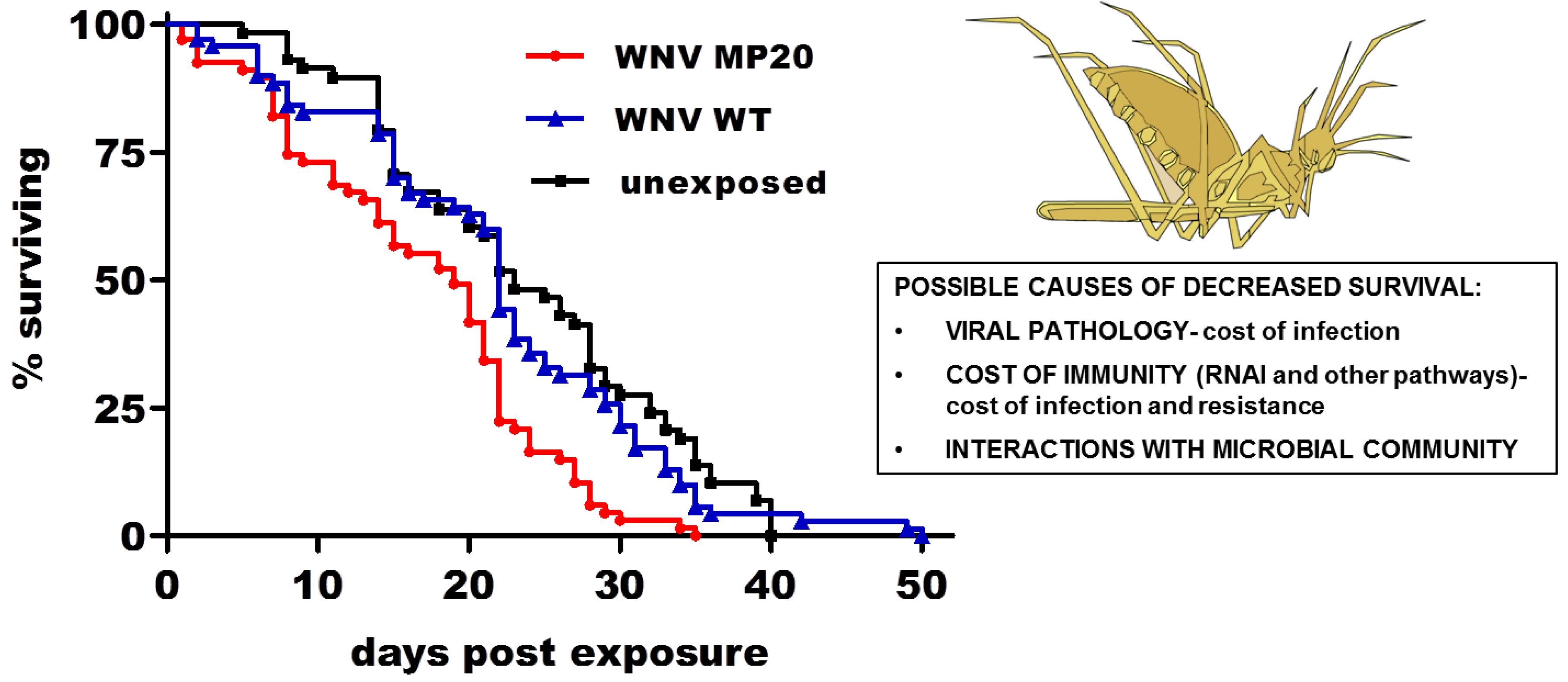
2.3. WNV Infection and Mosquito Life History Traits
2.4. Mosquito Immunity and Microbial Interactions
3. Vertebrate
3.1. Avian Host Competence
3.2. Other Vertebrates
4. Virus
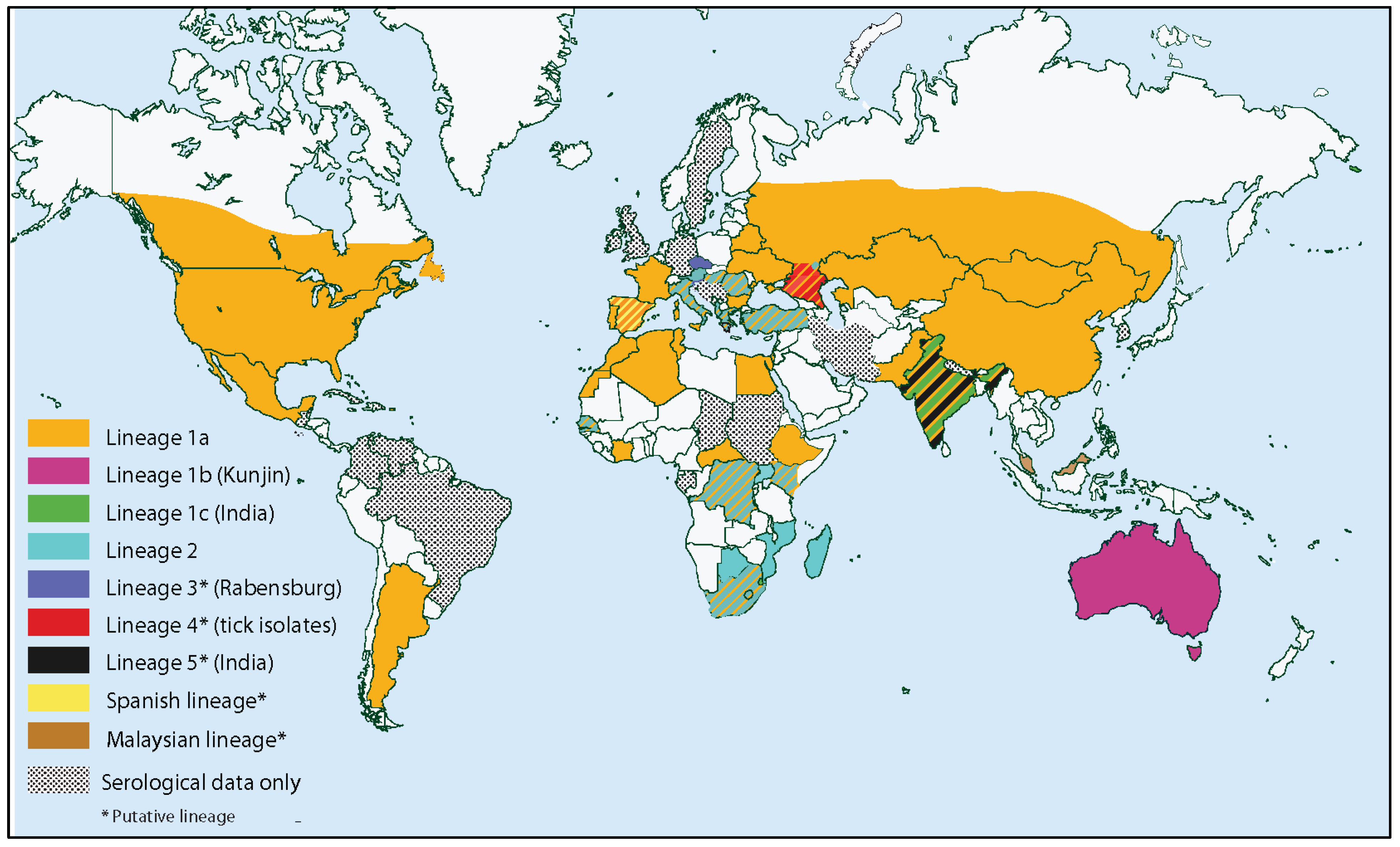
4.1. Population Genetics and Molecular Epidemiology
4.2. Bottlenecks and Intrahost Diversity
4.3. Adaptive Constraint
5. Concluding Remarks
Acknowledgments
Conflicts of Interest
References
- Ebel, G.D.; Dupuis, A.P., II; Ngo, K.A.; Nicholas, D.C.; Kauffman, E.B.; Jones, S.A.; Young, D.M.; Maffei, J.G.; Shi, P.-Y.; Bernard, K.A.; et al. Partial genetic characterization of West Nile virus strains, New York State. Emerg. Infect. Dis. 2001, 7, 650–653. [Google Scholar]
- Lanciotti, R.S.; Roehrig, J.T.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef]
- Bosch, I.; Herrera, F.; Navarro, J.C.; Lentino, M.; Dupuis, A.P.; Maffei, J.; Jones, M.J.; Fernandez, E.; Perez, N.; Perez-Eman, J.; et al. West Nile viurs, Venezuela. Emerg. Infect. Dis. 2007, 4, 651–653. [Google Scholar]
- Hayes, E.B.; Sejvar, J.J.; Zaki, S.R.; Lanciotti, R.S.; Bode, A.V.; Campbell, G.L. Virology, pathology, and clinical manifestations of West Nile virus disease. Emerg. Infect. Dis. 2005, 11, 1174–1179. [Google Scholar] [CrossRef]
- Komar, N.; Clark, G.G. West Nile virus activity in Latin America and the Caribbean. Rev. Panam. Salud Publica 2006, 19, 112–117. [Google Scholar] [CrossRef]
- Morales, M.A.; Barrandeguy, M.; Fabbri, C.; Garcia, J.B.; Vissani, A.; Trono, K.; Gutierrez, G.; Pigretti, S.; Menchaca, H.; Garrido, N.; et al. West Nile virus isolation from equines in Argentina, 2006. Emerg. Infect. Dis. 2006, 12, 1559–1561. [Google Scholar] [CrossRef]
- Chamberlain, R.W.; Sudia, W.D. Mechanism of transmission of viruses by mosquitoes. Annu. Rev. Entomol. 1961, 6, 371–390. [Google Scholar] [CrossRef]
- Hardy, J.L.; Houk, E.J.; Kramer, L.D.; Reeves, W.C. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Annu. Rev. Entomol. 1983, 28, 229–262. [Google Scholar] [CrossRef]
- Macdonald, G. Epidemiologic models in studies of vector-borne diseases. Public Health Rep. 1961, 76, 753–764. [Google Scholar] [CrossRef]
- Kramer, L.D.; Ebel, G.D. Dynamics of flavivirus infection in mosquitoes. Adv. Virus Res. 2003, 60, 187–232. [Google Scholar]
- Reiner, R.C., Jr.; Perkins, T.A.; Barker, C.M.; Niu, T.; Chaves, L.F.; Ellis, A.M.; George, D.B.; Le, M.A.; Pulliam, J.R.; Bisanzio, D.; et al. A systematic review of mathematical models of mosquito-borne pathogen transmission: 1970–2010. J. R. Soc. Interface 2013, 10. [Google Scholar] [CrossRef] [Green Version]
- Kilpatrick, A.M.; Kramer, L.D.; Campbell, S.R.; Alleyne, E.O.; Dobson, A.P.; Daszak, P. West Nile virus risk assessment and the bridge vector paradigm. Emerg. Infect. Dis. 2005, 11, 425–429. [Google Scholar] [CrossRef]
- Ebel, G.D.; Rochlin, I.; Longacker, J.; Kramer, L.D. Culex restuans (Diptera: Culicidae) relative abundance and vector competence for West Nile virus. J. Med. Entomol. 2005, 42, 838–843. [Google Scholar]
- Geery, P.R.; Holub, R.E. Seasonal abundance and control of Culex spp. in catch basins in Illinois. J. Am. Mosq. Control Assoc. 1989, 5, 537–540. [Google Scholar]
- Savage, H.M.; Aggarwal, D.; Apperson, C.S.; Katholi, C.R.; Gordon, E.; Hassan, H.K.; Anderson, M.; Charnetzky, D.; McMillen, L.; Unnasch, E.A.; et al. Host choice and West Nile virus infection rates in blood-fed mosquitoes, including members of the Culex pipiens complex, from Memphis and Shelby County, Tennessee, 2002–2003. Vector Borne Zoonotic Dis. 2007, 7, 365–386. [Google Scholar] [CrossRef]
- Andreadis, T.G.; Anderson, J.F.; Vossbrinck, C.R. Mosquito surveillance for West Nile virus in Connecticut, 2000: Isolation from Culex pipiens, Cx. restuans, Cx. salinarius, and Culiseta melanura. Emerg. Infect. Dis. 2001, 7, 670–674. [Google Scholar]
- Fonseca, D.M.; Okada, K.; Kramer, L.D. Microsatellite loci for the white-dotted mosquito (Culex restuans), a principal vector of West Nile virus in North America. Mol. Ecol. Resour. 2009, 9, 958–960. [Google Scholar] [CrossRef]
- Collins, F.H.; Paskewitz, S.M. A review of the use of ribosomal DNA (rDNA) to differentiate among cryptic Anopheles species. Insect Mol. Biol. 1996, 5, 1–9. [Google Scholar] [CrossRef]
- Harbach, R.E.; Dahl, C.; White, G.B. Culex (Culex) pipiens Linnaeus (Diptera: Culicidae): Concepts, type designations, and description. Proc. Entomol. Soc. Wash. 1985, 87, 1–24. [Google Scholar]
- Fonseca, D.M.; Smith, J.L.; Kim, H.C.; Mogi, M. Population genetics of the mosquito Culex pipiens pallens reveals sex-linked asymmetric introgression by Culex quinquefasciatus. Infect. Genet. Evol. 2009, 9, 1197–1203. [Google Scholar] [CrossRef]
- Fonseca, D.M.; Keyghobadi, N.; Malcolm, C.A.; Mehmet, C.; Schaffner, F.; Mogi, M.; Fleischer, R.C.; Wilkerson, R.C. Emerging vectors in the Culex pipiens complex. Science 2004, 303, 1535–1538. [Google Scholar] [CrossRef]
- Farajollahi, A.; Fonseca, D.M.; Kramer, L.D.; Marm, K.A. “Bird biting” mosquitoes and human disease: A review of the role of Culex pipiens complex mosquitoes in epidemiology. Infect. Genet. Evol. 2011, 11, 1577–1585. [Google Scholar] [CrossRef]
- Andreadis, T.G. The contribution of Culex pipiens complex mosquitoes to transmission and persistence of West Nile virus in North America. J. Am. Mosq. Control Assoc. 2012, 28, 137–151. [Google Scholar] [CrossRef]
- Kramer, L.D.; Styer, L.M.; Ebel, G.D. A global perspective on the epidemiology of West Nile virus. Annu. Rev. Entomol. 2007, 53, 61–81. [Google Scholar] [CrossRef]
- Huang, S.; Molaei, G.; Andreadis, T.G. Reexamination of Culex pipiens hybridization zone in the Eastern United States by ribosomal DNA-based single nucleotide polymorphism markers. Am. J. Trop. Med. Hyg. 2011, 85, 434–441. [Google Scholar] [CrossRef]
- Kothera, L.; Zimmerman, E.M.; Richards, C.M.; Savage, H.M. Microsatellite characterization of subspecies and their hybrids in Culex pipiens complex (Diptera: Culicidae) mosquitoes along a north-south transect in the central United States. J. Med. Entomol. 2009, 46, 236–248. [Google Scholar] [CrossRef]
- Sanogo, Y.O.; Kim, C.H.; Lampman, R.; Halvorsen, J.G.; Gad, A.M.; Novak, R.J. Identification of male specimens of the Culex pipiens complex (Diptera: Culicidae) in the hybrid zone using morphology and molecular techniques. J. Med. Entomol. 2008, 45, 203–209. [Google Scholar] [CrossRef]
- Ciota, A.T.; Chin, P.A.; Kramer, L.D. The effect of hybridization of Culex pipiens complex mosquitoes on transmission of West Nile virus. Parasites Vectors 2013, 6. [Google Scholar] [CrossRef]
- Brown, H.E.; Childs, J.E.; Diuk-Wasser, M.A.; Fish, D. Ecological factors associated with West Nile virus transmission, northeastern United States. Emerg. Infect. Dis. 2008, 14, 1539–1545. [Google Scholar]
- Kilpatrick, A.M. Globalization, land use, and the invasion of West Nile virus. Science 2011, 334, 323–327. [Google Scholar] [CrossRef]
- Kent, R.; Harrington, L.; Norris, D. Genetic differences between Culex pipiens f. molestus and Culex pipiens pipiens (Diptera: Culicidea) in New York. J. Med. Entomol. 2007, 44, 50–59. [Google Scholar] [CrossRef]
- Harbach, R.E. Culex pipiens: Species versus species complex taxonomic history and perspective. J. Am. Mosq. Control Assoc. 2012, 28, 10–23. [Google Scholar] [CrossRef]
- Bolling, B.G.; Moore, C.G.; Anderson, S.L.; Blair, C.D.; Beaty, B.J. Entomological studies along the Colorado Front Range during a period of intense West Nile virus activity. J. Am. Mosq. Control Assoc. 2007, 23, 37–46. [Google Scholar] [CrossRef]
- Reisen, W.K.; Reeves, W.C. Bionomics and Ecology of Culex tarsalis and Other Potential Mosquito Vector Species. In Epidemiology and Control of Mosquito-borne Arboviruses in California, 1943–1987; Reeves, W.C., Ed.; California Mosquito and Vector Control Association, Inc.: Sacramento, C.A., USA, 1990; pp. 254–329. [Google Scholar]
- Goldberg, T.L.; Anderson, T.K.; Hamer, G.L. West Nile virus may have hitched a ride across the Western United States on Culex tarsalis mosquitoes. Mol. Ecol. 2010, 19, 1518–1519. [Google Scholar] [CrossRef]
- Bolling, B.G.; Barker, C.M.; Moore, C.G.; Pape, W.J.; Eisen, L. Seasonal patterns for entomological measures of risk for exposure to Culex vectors and West Nile virus in relation to human disease cases in northeastern Colorado. J. Med. Entomol. 2009, 46, 1519–1531. [Google Scholar] [CrossRef]
- Bowden, S.E.; Magori, K.; Drake, J.M. Regional differences in the association between land cover and West Nile virus disease incidence in humans in the United States. Am. J. Trop. Med. Hyg. 2011, 84, 234–238. [Google Scholar] [CrossRef]
- Venkatesan, M.; Rasgon, J.L. Population genetic data suggest a role for mosquito-mediated dispersal of West Nile virus across the western United States. Mol. Ecol. 2010, 19, 1573–1584. [Google Scholar] [CrossRef]
- Venkatesan, M.; Westbrook, C.J.; Hauer, M.C.; Rasgon, J.L. Evidence for a population expansion in the West Nile virus vector Culex tarsalis. Mol. Biol. Evol. 2007, 24, 1208–1218. [Google Scholar] [CrossRef]
- Kramer, L.D.; Hardy, J.L.; Presser, S.B.; Houk, E.J. Dissemination barriers for western equine encephalomyelitis virus in Culex tarsalis infected after ingestion of low viral doses. Am. J. Trop. Med. Hyg. 1981, 30, 190–197. [Google Scholar]
- Grimstad, P.R.; Paulson, S.L.; Craig, G.B., Jr. Vector competence of Aedes hendersoni (Diptera: Culicidae) for La Crosse virus and evidence of a salivary-gland escape barrier. J. Med. Entomol. 1985, 22, 447–453. [Google Scholar]
- Miller, B.R.; Monath, T.P.; Tabachnick, W.J.; Ezike, V.I. Epidemic yellow fever caused by an incompetent mosquito vector. Trop. Med. Parasitol. 1989, 40, 396–399. [Google Scholar]
- Garrett-Jones, C. Prognosis for interruption of malaria transmission through assessment of the mosquito’s vectorial capacity. Nature 1964, 204, 1173–1175. [Google Scholar] [CrossRef]
- Macdonald, G. The Epidemiology and Control of Malaria; Oxford Univ. Press: London, UK, 1957. [Google Scholar]
- Turell, M.J.; Dohm, D.J.; Sardelis, M.R.; Oguinn, M.L.; Andreadis, T.G.; Blow, J.A. An update on the potential of north American mosquitoes (Diptera: Culicidae) to transmit West Nile Virus. J. Med. Entomol. 2005, 42, 57–62. [Google Scholar] [CrossRef]
- Goddard, L.B.; Roth, A.E.; Reisen, W.K.; Scott, T.W. Vector competence of California mosquitoes for West Nile virus. Emerg. Infect. Dis. 2002, 8, 1385–1391. [Google Scholar] [CrossRef]
- Turell, M.J.; O’Guinn, M.L.; Dohm, D.J.; Jones, J.W. Vector competence of North American mosquitoes (Diptera: Culicidae) for West Nile virus. J. Med. Entomol. 2001, 38, 130–134. [Google Scholar] [CrossRef]
- Turell, M.J.; O’Guinn, M.; Oliver, J. Potential for New York mosquitoes to transmit West Nile virus. Am. J. Trop. Med. Hyg. 2000, 62, 413–414. [Google Scholar]
- Sardelis, M.R.; Turell, M.J.; Dohm, D.J.; O’Guinn, M.L. Vector competence of selected North American Culex and Coquillettidia mosquitoes for West Nile virus. Emerg. Infect. Dis. 2001, 7, 1018–1022. [Google Scholar] [CrossRef]
- Moudy, R.M.; Meola, M.A.; Morin, L.L.; Ebel, G.D.; Kramer, L.D. A newly emergent genotype of west nile virus is transmitted earlier and more efficiently by culex mosquitoes. Am. J. Trop. Med. Hyg. 2007, 77, 365–370. [Google Scholar]
- Kilpatrick, A.M.; Meola, M.A.; Moudy, R.M.; Kramer, L.D. Temperature, viral genetics, and the transmission of West Nile virus by Culex pipiens mosquitoes. PLoS Pathog. 2008, 4, e1000092. [Google Scholar] [CrossRef]
- Ebel, G.D.; Carricaburu, J.; Young, D.; Bernard, K.A.; Kramer, L.D. Genetic and phenotypic variation of West Nile virus in New York, 2000–2003. Am. J. Trop. Med. Hyg. 2004, 71, 493–500. [Google Scholar]
- Dohm, D.J.; O’Guinn, M.L.; Turell, M.J. Effect of environmental temperature on the ability of Culex pipiens (Diptera: Culicidae) to transmit West Nile virus. J. Med. Entomol. 2002, 39, 221–225. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Martinez, V.M. Effects of temperature on the transmission of west nile virus by Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2006, 43, 309–317. [Google Scholar] [CrossRef]
- Reisen, W.K.; Barker, C.M.; Fang, Y.; Martinez, V.M. Does variation in Culex (Diptera: Culicidae) vector competence enable outbreaks of West Nile virus in California? J. Med. Entomol. 2008, 45, 1126–1138. [Google Scholar] [CrossRef]
- Sardelis, M.R.; Turell, M.J.; O’Guinn, M.L.; Andre, R.G.; Roberts, D.R. Vector competence of three North American strains of Aedes albopictus for West Nile virus. J. Am. Mosq. Control Assoc. 2002, 18, 284–289. [Google Scholar]
- Vaidyanathan, R.; Scott, T.W. Geographic variation in vector competence for West Nile virus in the Culex pipiens (Diptera: Culicidae) complex in California. Vector Borne Zoonotic Dis. 2007, 7, 193–198. [Google Scholar] [CrossRef]
- Hayes, C.G.; Baker, R.H.; Baqar, S.; Ahmed, T. Genetic variation for West Nile virus susceptibility in Culex tritaeniorhynchus. Am. J. Trop. Med. Hyg. 1984, 33, 715–724. [Google Scholar]
- Vaidyanathan, V.; Scott, T.W. Seasonal variation in susceptibility to West Nile virus infection in Culex pipiens pipiens (L.) (Diptera: Culicidea) from San Joaquin County, California. J. Vector Ecol. 2006, 31, 423–425. [Google Scholar] [CrossRef]
- Kilpatrick, A.M.; Fonseca, D.M.; Ebel, G.D.; Reddy, M.R.; Kramer, L.D. Spatial and temporal variation in vector competence of Culex pipiens and Cx. restuans mosquitoes for West Nile virus. Am. J. Trop. Med. Hyg. 2010, 83, 607–613. [Google Scholar] [CrossRef]
- Delatte, H.; Gimonneau, G.; Triboire, A.; Fontenille, D. Influence of temperature on immature development, survival, longevity, fecundity, and gonotrophic cycles of Aedes albopictus, vector of chikungunya and dengue in the Indian Ocean. J. Med. Entomol. 2009, 46, 33–41. [Google Scholar] [CrossRef]
- Rueda, L.M.; Patel, K.J.; Axtell, R.C.; Stinner, R.E. Temperature-dependent development and survival rates of Culex quinquefasciatus and Aedes aegypti (Diptera: Culicidae). J. Med. Entomol. 1990, 27, 892–898. [Google Scholar]
- Dye, C. The analysis of parasite transmission by bloodsucking insects. Annu. Rev. Entomol. 1992, 37, 1–19. [Google Scholar] [CrossRef]
- Su, T.; Mulla, M.S. Effects of temperature on development, mortality, mating and blood feeding behavior of Culiseta incidens (Diptera: Culicidae). J. Vector Ecol. 2001, 26, 83–92. [Google Scholar]
- Kilpatrick, A.M.; Randolph, S.E. Drivers, dynamics, and control of emerging vector-borne zoonotic diseases. Lancet 2012, 380, 1946–1955. [Google Scholar] [CrossRef]
- Ciota, A.T.; Matacchiero, A.C.; Kilpatrick, A.M.; Kramer, L.D. The effect of temperature on life history traits of Culex mosquitoes. J. Med. Entomol. in press.
- Reisen, W.K.; Thiemann, T.; Barker, C.M.; Lu, H.; Carroll, B.; Fang, Y.; Lothrop, H.D. Effects of warm winter temperature on the abundance and gonotrophic activity of Culex (Diptera: Culicidae) in California. J. Med. Entomol. 2010, 47, 230–237. [Google Scholar] [CrossRef]
- Ciota, A.T.; Drummond, C.L.; Drobnack, J.; Ruby, M.A.; Kramer, L.D.; Ebel, G.D. Emergence of Culex pipiens from overwintering hibernacula. J. Am. Mosq. Control Assoc. 2011, 27, 21–29. [Google Scholar] [CrossRef]
- Dodson, B.L.; Kramer, L.D.; Rasgon, J.L. Larval nutritional stress does not affect vector competence for West Nile virus (WNV) in Culex tarsalis. Vector Borne Zoonotic Dis. 2011, 11, 1493–1497. [Google Scholar] [CrossRef]
- Jennings, C.D.; Kay, B.H. Dissemination barriers to Ross River virus in Aedes vigilax and the effects of larval nutrition on their expression. Med. Vet. Entomol. 1999, 13, 431–438. [Google Scholar] [CrossRef]
- Grimstad, P.R.; Walker, E.D. Aedes triseriatus (Diptera: Culicidae) and La Crosse virus. IV. Nutritional deprivation of larvae affects the adult barriers to infection and transmission. J. Med. Entomol. 1991, 28, 378–386. [Google Scholar]
- Vaidyanathan, R.; Fleisher, A.E.; Minnick, S.L.; Simmons, K.A.; Scott, T.W. Nutritional stress affects mosquito survival and vector competence for West Nile virus. Vector Borne Zoonotic Dis. 2008, 8, 727–732. [Google Scholar] [CrossRef]
- Shaman, J.; Day, J.F.; Stieglitz, M. Drought-induced amplification and epidemic transmission of West Nile virus in southern Florida. J. Med. Entomol. 2005, 42, 134–141. [Google Scholar] [CrossRef]
- Epstein, P.R. West Nile virus and the climate. J. Urban Health 2001, 78, 367–371. [Google Scholar] [CrossRef]
- Johnson, B.J.; Sukhdeo, M.V. Drought-induced amplification of local and regional West Nile virus infection rates in New Jersey. J. Med. Entomol. 2013, 50, 195–204. [Google Scholar] [CrossRef]
- Crowder, D.W.; Dykstra, E.A.; Brauner, J.M.; Duffy, A.; Reed, C.; Martin, E.; Peterson, W.; Carriere, Y.; Dutilleul, P.; Owen, J.P. West nile virus prevalence across landscapes is mediated by local effects of agriculture on vector and host communities. PLoS One 2013, 8, e55006. [Google Scholar]
- Kilpatrick, A.M.; Daszak, P.; Jones, M.J.; Marra, P.P.; Kramer, L.D. Host heterogeneity dominates West Nile virus transmission. Proc. Biol. Sci. 2006, 273, 2327–2333. [Google Scholar] [CrossRef]
- Kent, R.; Juliusson, L.; Weissmann, M.; Evans, S.; Komar, N. Seasonal blood-feeding behavior of Culex tarsalis (Diptera: Culicidae) in Weld County, Colorado, 2007. J. Med. Entomol. 2009, 46, 380–390. [Google Scholar] [CrossRef]
- Thiemann, T.C.; Lemenager, D.A.; Kluh, S.; Carroll, B.D.; Lothrop, H.D.; Reisen, W.K. Spatial variation in host feeding patterns of Culex tarsalis and the Culex pipiens complex (Diptera: Culicidae) in California. J. Med. Entomol. 2012, 49, 903–916. [Google Scholar] [CrossRef]
- Reisen, W.K.; Hardy, J.L.; Reeves, W.C.; Presser, S.B.; Milby, M.M.; Meyer, R.P. Persistence of mosquito-borne viruses in Kern County, California, 1983–1988. Am. J. Trop. Med. Hyg. 1990, 43, 419–437. [Google Scholar]
- Hamer, G.L.; Kitron, U.D.; Goldberg, T.L.; Brawn, J.D.; Loss, S.R.; Ruiz, M.O.; Hayes, D.B.; Walker, E.D. Host selection by Culex pipiens mosquitoes and West Nile virus amplification. Am. J. Trop. Med. Hyg. 2009, 80, 268–278. [Google Scholar]
- Kilpatrick, A.M.; Kramer, L.D.; Jones, M.J.; Marra, P.P.; Daszak, P. West Nile virus epidemics in North America are driven by shifts in mosquito feeding behavior. PLoS Biol. 2006, 4, e82. [Google Scholar] [CrossRef]
- Thiemann, T.C.; Wheeler, S.S.; Barker, C.M.; Reisen, W.K. Mosquito host selection varies seasonally with host availability and mosquito density. PLoS Negl. Trop. Dis. 2011, 5, e1452. [Google Scholar] [CrossRef]
- Huang, S.; Hamer, G.L.; Molaei, G.; Walker, E.D.; Goldberg, T.L.; Kitron, U.D.; Andreadis, T.G. Genetic variation associated with mammalian feeding in Culex pipiens from a West Nile virus epidemic region in Chicago, Illinois. Vector Borne Zoonotic Dis. 2009, 9, 637–642. [Google Scholar] [CrossRef]
- Schwartz, A.; Koella, J.C. Trade-offs, conflicts of interest and manipulation in Plasmodium-mosquito interactions. Trends Parasitol. 2001, 17, 189–194. [Google Scholar] [CrossRef]
- Jackson, B.T.; Brewster, C.C.; Paulson, S.L. La Crosse virus infection alters blood feeding behavior in Aedes triseriatus and Aedes albopictus (Diptera: Culicidae). J. Med. Entomol. 2012, 49, 1424–1429. [Google Scholar] [CrossRef]
- Styer, L.M.; Meola, M.A.; Kramer, L.D. West Nile virus infection decreases fecundity of Culex tarsalis females. J. Med. Entomol. 2007, 44, 1074–1085. [Google Scholar] [CrossRef]
- Ciota, A.T.; Styer, L.M.; Meola, M.A.; Kramer, L.D. The costs of infection and resistance as determinants of West Nile virus susceptibility in Culex mosquitoes. BMC Ecol. 2011, 11. [Google Scholar] [CrossRef]
- Ciota, A.T.; Ehrbar, D.J.; Matacchiero, A.C.; van Slyke, G.A.; Kramer, L.D. The evolution of virulence of West Nile virus in a mosquito vector: Implications for arbovirus adaptation and evolution. BMC Evol. Biol. 2013, 13. [Google Scholar] [CrossRef]
- Maciel-de-Freitas, R.; Koella, J.C.; Lourenco-de-Oliveira, R. Lower survival rate, longevity and fecundity of Aedes aegypti (Diptera: Culicidae) females orally challenged with dengue virus serotype 2. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 452–458. [Google Scholar] [CrossRef]
- Mahmood, F.; Reisen, W.K.; Chiles, R.E.; Fang, Y. Western equine encephalomyelitis virus infection affects the life table characteristics of Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2004, 41, 982–986. [Google Scholar] [CrossRef]
- Moncayo, A.C.; Edman, J.D.; Turell, M.J. Effect of eastern equine encephalomyelitis virus on the survival of Aedes albopictus, Anopheles quadrimaculatus, and Coquillettidia perturbans (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 701–706. [Google Scholar] [CrossRef]
- Scott, T.W.; Lorenz, L.H. Reduction of Culiseta melanura fitness by eastern equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1998, 59, 341–346. [Google Scholar]
- Girard, Y.A.; Popov, V.; Wen, J.; Han, V.; Higgs, S. Ultrastructural study of West Nile virus pathogenesis in Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2005, 42, 429–444. [Google Scholar] [CrossRef]
- Vaidyanathan, R.; Scott, T.W. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 2006, 11, 1643–1651. [Google Scholar] [CrossRef]
- Fragkoudis, R.; Attarzadeh-Yazdi, G.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Advances in dissecting mosquito innate immune responses to arbovirus infection. J. Gen. Virol. 2009, 90, 2061–2072. [Google Scholar] [CrossRef]
- Blair, C.D. Mosquito RNAi is the major innate immune pathway controlling arbovirus infection and transmission. Future Microbiol. 2011, 6, 265–277. [Google Scholar] [CrossRef]
- Wang, X.H.; Aliyari, R.; Li, W.X.; Li, H.W.; Kim, K.; Carthew, R.; Atkinson, P.; Ding, S.W. RNA interference directs innate immunity against viruses in adult Drosophila. Science 2006, 312, 452–454. [Google Scholar] [CrossRef]
- Chotkowski, H.L.; Ciota, A.T.; Jia, Y.; Puig-Basagoiti, F.; Kramer, L.D.; Shi, P.Y.; Glaser, R.L. West Nile virus infection of Drosophila melanogaster induces a protective RNAi response. Virology 2008, 377, 197–206. [Google Scholar] [CrossRef]
- Arjona, A.; Wang, P.; Montgomery, R.R.; Fikrig, E. Innate immune control of West Nile virus infection. Cell. Microbiol. 2011, 13, 1648–1658. [Google Scholar] [CrossRef]
- Paradkar, P.N.; Trinidad, L.; Voysey, R.; Duchemin, J.B.; Walker, P.J. Secreted Vago restricts West Nile virus infection in Culex mosquito cells by activating the Jak-STAT pathway. Proc. Natl. Acad. Sci. USA 2012, 109, 18915–18920. [Google Scholar]
- Prasad, A.N.; Brackney, D.E.; Ebel, G.D. The role of innate immunity in conditioning mosquito susceptibility to West Nile virus. Viruses. in press.
- Cook, S.; Bennett, S.N.; Holmes, E.C.; Chesse, R.; Moureau, G.; Lamballeria, X. Isolation of a new strain of the flavivirus cell fusing agent virus in a natural mosquito population from Puerto Rico. J. Gen. Virol. 2006, 87, 735–748. [Google Scholar] [CrossRef]
- Crabtree, M.B.; Sang, R.C.; Stollar, V.; Dunster, L.M.; Miller, B.R. Genetic and phenotypic characterization of the newly described insect flavivirus, Kamiti River virus. Arch. Virol. 2003, 148, 1095–1118. [Google Scholar] [CrossRef]
- Hoshino, K.; Isawa, H.; Tsuda, Y.; Yano, K.; Sasaki, T.; Yuda, M.; Takasaki, T.; Kobayashi, M.; Sawabe, K. Genetic characterization of a new insect flavivirus isolated from Culex pipiens mosquito in Japan. Virology 2007, 359, 405–414. [Google Scholar]
- Huhtamo, E.; Putkuri, N.; Kurkela, S.; Manni, T.; Vaheri, A.; Vapalahti, O.; Uzcategui, N.Y. Characterization of a novel flavivirus from mosquitoes in northern europe that is related to mosquito-borne flaviviruses of the tropics. J. Virol. 2009, 83, 9532–9540. [Google Scholar]
- Bolling, B.G.; Eisen, L.; Moore, C.G.; Blair, C.D. Insect-specific flaviviruses from Culex mosquitoes in Colorado, with evidence of vertical transmission. Am. J. Trop. Med. Hyg. 2011, 85, 169–177. [Google Scholar] [CrossRef]
- Hobson-Peters, J.; Yam, A.W.; Lu, J.W.; Setoh, Y.X.; May, F.J.; Kurucz, N.; Walsh, S.; Prow, N.A.; Davis, S.S.; Weir, R.; et al. A new insect-specific flavivirus from northern Australia suppresses replication of West Nile virus and Murray Valley encephalitis virus in co-infected mosquito cells. PLoS One 2013, 8, e56534. [Google Scholar]
- Calzolari, M.; Bonilauri, P.; Bellini, R.; Albieri, A.; Defilippo, F.; Maioli, G.; Galletti, G.; Gelati, A.; Barbieri, I.; Tamba, M.; et al. Evidence of simultaneous circulation of West Nile and Usutu viruses in mosquitoes sampled in Emilia-Romagna region (Italy) in 2009. PLoS One 2010, 5, e14324. [Google Scholar] [CrossRef]
- Newman, C.M.; Cerutti, F.; Anderson, T.K.; Hamer, G.L.; Walker, E.D.; Kitron, U.D.; Ruiz, M.O.; Brawn, J.D.; Goldberg, T.L. Culex flavivirus and West Nile virus mosquito coinfection and positive ecological association in Chicago, United States. Vector Borne Zoonotic Dis. 2011, 11, 1099–1105. [Google Scholar] [CrossRef]
- Bolling, B.G.; Olea-Popelka, F.J.; Eisen, L.; Moore, C.G.; Blair, C.D. Transmission dynamics of an insect-specific flavivirus in a naturally infected Culex pipiens laboratory colony and effects of co-infection on vector competence for West Nile virus. Virology 2012, 427, 90–97. [Google Scholar] [CrossRef]
- Kent, R.J.; Crabtree, M.B.; Miller, B.R. Transmission of West Nile virus by Culex quinquefasciatus say infected with Culex Flavivirus Izabal. PLoS Negl. Trop. Dis. 2010, 4, e671. [Google Scholar] [CrossRef]
- Zou, G.; Zhang, B.; Lim, P.Y.; Yuan, Z.; Bernard, K.A.; Shi, P.Y. Exclusion of West Nile virus superinfection through RNA replication. J. Virol. 2009, 83, 11765–11776. [Google Scholar] [CrossRef]
- Weiss, B.; Aksoy, S. Microbiome influences on insect host vector competence. Trends Parasitol. 2011, 27, 514–522. [Google Scholar] [CrossRef]
- Hoffmann, A.A.; Montgomery, B.L.; Popovici, J.; Iturbe-Ormaetxe, I.; Johnson, P.H.; Muzzi, F.; Greenfield, M.; Durkan, M.; Leong, Y.S.; Dong, Y.; et al. Successful establishment of Wolbachia in Aedes populations to suppress dengue transmission. Nature 2011, 476, 454–457. [Google Scholar] [CrossRef]
- Pan, X.; Zhou, G.; Wu, J.; Bian, G.; Lu, P.; Raikhel, A.S.; Xi, Z. Wolbachia induces reactive oxygen species (ROS)-dependent activation of the Toll pathway to control dengue virus in the mosquito Aedes aegypti. Proc. Natl. Acad. Sci. USA 2012, 109, E23–E31. [Google Scholar]
- Glaser, R.L.; Meola, M.A. The native Wolbachia endosymbionts of Drosophila melanogaster and Culex quinquefasciatus increase host resistance to West Nile virus infection. PLoS One 2010, 5, e11977. [Google Scholar] [CrossRef]
- Hussain, M.; Lu, G.; Torres, S.; Edmonds, J.H.; Kay, B.H.; Khromykh, A.A.; Asgari, S. Effect of wolbachia on replication of west nile virus in a mosquito cell line and adult mosquitoes. J. Virol. 2013, 87, 851–858. [Google Scholar] [CrossRef]
- Bian, G.; Zhou, G.; Lu, P.; Xi, Z. Replacing a native Wolbachia with a novel strain results in an increase in endosymbiont load and resistance to dengue virus in a mosquito vector. PLoS Negl. Trop. Dis. 2013, 7, e2250. [Google Scholar] [CrossRef]
- Pidiyar, V.J.; Jangid, K.; Patole, M.S.; Shouche, Y.S. Studies on cultured and uncultured microbiota of wild culex quinquefasciatus mosquito midgut based on 16s ribosomal RNA gene analysis. Am. J. Trop. Med. Hyg. 2004, 70, 597–603. [Google Scholar]
- Ramirez, J.L.; Souza-Neto, J.; Torres, C.R.; Rovira, J.; Ortiz, A.; Pascale, J.M.; Dimopoulos, G. Reciprocal tripartite interactions between the Aedes aegypti midgut microbiota, innate immune system and dengue virus influences vector competence. PLoS Negl. Trop. Dis. 2012, 6, e1561. [Google Scholar] [CrossRef]
- Ricci, I.; Damiani, C.; Capone, A.; DeFreece, C.; Rossi, P.; Favia, G. Mosquito/microbiota interactions: From complex relationships to biotechnological perspectives. Curr. Opin. Microbiol. 2012, 15, 278–284. [Google Scholar] [CrossRef]
- Ignatova, E.A.; Nagornaia, S.S.; Povazhnaia, T.N.; Ianishevskaia, G.S. The yeast flora of blood-sucking mosquitoes. Mikrobiol. Z. 1996, 58, 12–15. [Google Scholar]
- Ricci, I.; Mosca, M.; Valzano, M.; Damiani, C.; Scuppa, P.; Rossi, P.; Crotti, E.; Cappelli, A.; Ulissi, U.; Capone, A.; et al. Different mosquito species host Wickerhamomyces anomalus (Pichia anomala): Perspectives on vector-borne diseases symbiotic control. Antonie Van Leeuwenhoek 2011, 99, 43–50. [Google Scholar] [CrossRef] [Green Version]
- Garza-Hernandez, J.A.; Rodriguez-Perez, M.A.; Salazar, M.I.; Russell, T.L.; Adeleke, M.A.; de Luna-Santillana, E.J.; Reyes-Villanueva, F. Vectorial capacity of Aedes aegypti for dengue virus type 2 is reduced with co-infection of Metarhizium anisopliae. PLoS Negl. Trop. Dis. 2013, 7, e2013. [Google Scholar] [CrossRef]
- Komar, N.; Dohm, D.J.; Turell, M.J.; Spielman, A. Eastern equine encephalitis virus in birds: Relative competence of European starlings (Sturnus vulgaris). Am. J. Trop. Med. Hyg. 1999, 60, 387–391. [Google Scholar]
- Komar, N.; Langevin, S.; Hinten, S.; Nemeth, N.; Edwards, E.; Hettler, D.; Davis, B.; Bowen, R.; Bunning, M. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 2003, 9, 311–322. [Google Scholar] [CrossRef]
- Dusek, R.J.; McLean, R.G.; Kramer, L.D.; Ubico, S.R.; Dupuis, A.P., II; Ebel, G.D.; Guptill, S.C. Prevalence of West Nile virus in migratory birds during spring and fall migration. Am. J. Trop. Med. Hyg. 2009, 81, 1151–1158. [Google Scholar] [CrossRef]
- Kramer, L.D.; Bernard, K.A. West Nile virus in the western hemisphere. Curr. Opin. Infect. Dis. 2001, 14, 519–525. [Google Scholar] [CrossRef]
- Bernard, K.A.; Kramer, L.D. West Nile virus activity in the United States, 2001. Viral Immunol. 2001, 14, 319–338. [Google Scholar] [CrossRef]
- Ladeau, S.L.; Kilpatrick, A.M.; Marra, P.P. West Nile virus emergence and large-scale declines of North American bird populations. Nature 2007, 447, 710–713. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Species of dead birds in which West Nile virus has been detected, United States, 1999–2012. Available online: http://www.cdc.gov/westnile/resources/pdfs/Bird%20Species%201999-2012.pdf (accessed 1 December 2013).
- Reed, L.M.; Johansson, M.A.; Panella, N.; McLean, R.; Creekmore, T.; Puelle, R.; Komar, N. Declining mortality in American crow (Corvus brachyrhynchos) following natural West Nile virus infection. Avian Dis. 2009, 53, 458–461. [Google Scholar] [CrossRef]
- Komar, N.; Panella, N.A.; Burns, J.E.; Dusza, S.W.; Mascarenhas, T.M.; Talbot, T.O. Serologic evidence for West Nile virus infection in birds in the New York City vicinity during an outbreak in 1999. Emerg. Infect. Dis. 2001, 7, 621–625. [Google Scholar]
- Malkinson, M.; Banet, C.; Weisman, Y.; Pokamunski, S.; King, R.; Drouet, M.T.; Deubel, V. Introduction of West Nile virus in the Middle East by migrating white storks. Emerg. Infect. Dis. 2002, 8, 392–397. [Google Scholar] [CrossRef]
- Tempelis, C.H.; Reeves, W.C.; Bellamy, R.E.; Lofy, M.F. A three-year study of the feeding habits of Culex tarsalis in Kern County, California. Am. J. Trop. Med. Hyg. 1965, 14, 170–177. [Google Scholar]
- Tempelis, C.H.; Francy, D.B.; Hayes, R.O.; Lofy, M.F. Variations in feeding patterns of seven culicine mosquitoes on vertebrate hosts in Weld and Larimer counties, Colorado. Am. J. Trop. Med. Hyg. 1967, 111–119. [Google Scholar]
- Root, J.J.; Oesterle, P.T.; Nemeth, N.M.; Klenk, K.; Gould, D.H.; McLean, R.G.; Clark, L.; Hall, J.S. Experimental infection of fox squirrels (Sciurus niger) with West Nile virus. Am. J. Trop. Med. Hyg. 2006, 75, 697–701. [Google Scholar]
- Gomez, A.; Kramer, L.D.; Dupuis, A.P.; Kilpatrick, A.M.; Davis, L.J.; Jones, M.J.; Daszak, P.; Aguirre, A.A. Experimental infection of eastern gray squirrels (Sciurus carolinensis) with West Nile virus. Am. J. Trop. Med. Hyg. 2008, 79, 447–451. [Google Scholar]
- Gomez, A.; Kilpatrick, A.M.; Kramer, L.D.; Dupuis, A.P.; Maffei, J.G.; Goetz, S.J.; Marra, P.P.; Daszak, P.; Aguirre, A.A. Land use and west nile virus seroprevalence in wild mammals. Emerg. Infect. Dis. 2008, 14, 962–965. [Google Scholar] [CrossRef]
- Root, J.J. West Nile virus associations in wild mammals: A synthesis. Arch. Virol. 2013, 158, 735–752. [Google Scholar] [CrossRef]
- Smithburn, K.C.; Hughes, T.P.; Burke, A.W.; Paul, J.H. A neurotropic virus isolated from the blood of a native of Uganda. Am. J. Trop. Med. Hyg. 1940, 20, 471–473. [Google Scholar]
- May, F.J.; Davis, C.T.; Tesh, R.B.; Barrett, A.D. Phylogeography of West Nile virus: From the cradle of evolution in Africa to Eurasia, Australia, and the Americas. J. Virol. 2011, 85, 2964–2974. [Google Scholar] [CrossRef]
- Papa, A.; Bakonyi, T.; Xanthopoulou, K.; Vazquez, A.; Tenorio, A.; Nowotny, N. Genetic characterization of West Nile virus lineage 2, Greece, 2010. Emerg. Infect. Dis. 2011, 17, 920–922. [Google Scholar] [CrossRef]
- Ciccozzi, M.; Peletto, S.; Cella, E.; Giovanetti, M.; Lai, A.; Gabanelli, E.; Acutis, P.L.; Modesto, P.; Rezza, G.; Platonov, A.E.; et al. Epidemiological history and phylogeography of West Nile virus lineage 2. Infect. Genet. Evol. 2013, 17, 46–50. [Google Scholar] [CrossRef]
- Beasley, D.W.; Li, L.; Suderman, M.T.; Barrett, A.D. Mouse neuroinvasive phenotype of West Nile virus strains varies depending upon virus genotype. Virology 2002, 296, 17–23. [Google Scholar] [CrossRef]
- Bakonyi, T.; Hubalek, Z.; Rudolf, I.; Nowotny, N. Novel flavivirus or new lineage of West Nile virus, central Europe. Emerg. Infect. Dis. 2005, 11, 225–231. [Google Scholar] [CrossRef]
- Hubalek, Z.; Rudolf, I.; Bakonyi, T.; Kazdova, K.; Halouzka, J.; Sebesta, O.; Sikutova, S.; Juricova, Z.; Nowotny, N. Mosquito (Diptera: Culicidae) surveillance for arboviruses in an area endemic for West Nile (Lineage Rabensburg) and Tahyna viruses in Central Europe. J. Med. Entomol. 2010, 47, 466–472. [Google Scholar] [CrossRef]
- Aliota, M.T.; Jones, S.A.; Dupuis, A.P.; Ciota, A.T.; Hubalek, Z.; Kramer, L.D. Characterization of Rabensburg virus, a flavivirus closely related to West Nile virus of the Japanese encephalitis antigenic group. PLoS One 2012, 7, e39387. [Google Scholar]
- Prilipov, A.G.; Kinney, R.M.; Samokhvalov, E.I.; Savage, H.M.; Al’khovskii, S.V.; Tsuchiya, K.R.; Gromashevskii, V.L.; Sadykova, G.K.; Shatalov, A.G.; Vyshemirskii, O.I.; et al. Analysis of new variants of West Nile fever virus. Vopr. Virusol. 2002, 47, 36–41. [Google Scholar]
- Vazquez, A.; Sanchez-Seco, M.P.; Ruiz, S.; Molero, F.; Hernandez, L.; Moreno, J.; Magallanes, A.; Tejedor, C.G.; Tenorio, A. Putative new lineage of west nile virus, Spain. Emerg. Infect. Dis. 2010, 16, 549–552. [Google Scholar] [CrossRef]
- Bondre, V.P.; Jadi, R.S.; Mishra, A.C.; Yergolkar, P.N.; Arankalle, V.A. West Nile virus isolates from India: Evidence for a distinct genetic lineage. J. Gen. Virol. 2007, 88, 875–884. [Google Scholar] [CrossRef]
- Scherret, J.H.; Poidinger, M.; Mackenzie, J.S.; Broom, A.K.; Deubel, V.; Lipkin, W.I.; Briese, T.; Gould, E.A.; Hall, R.A. The relationships between West Nile and Kunjin viruses. Emerg. Infect. Dis. 2001, 4, 697–705. [Google Scholar]
- Charrel, R.N.; Brault, A.C.; Gallian, P.; Lemasson, J.J.; Murgue, B.; Murri, S.; Pastorino, B.; Zeller, H.; de Chesse, R.; de Micco, P.; et al. Evolutionary relationship between Old World West Nile virus strains. Evidence for viral gene flow between Africa, the Middle East, and Europe. Virology 2003, 315, 381–388. [Google Scholar] [CrossRef]
- Malkinson, M.; Banet, C.; Weisman, Y.; Pokamonski, S.; King, R.; Deubel, V. Intercontinental transmission of West Nile virus by migrating white storks. Emerg. Infect. Dis. 2001, 7. [Google Scholar]
- McMullen, A.R.; May, F.J.; Li, L.; Guzman, H.; Bueno, R., Jr.; Dennett, J.A.; Tesh, R.B.; Barrett, A.D. Evolution of new genotype of West Nile virus in North America. Emerg. Infect. Dis. 2011, 17, 785–793. [Google Scholar] [CrossRef]
- Armstrong, P.M.; Vossbrinck, C.R.; Andreadis, T.G.; Anderson, J.F.; Pesko, K.N.; Newman, R.M.; Lennon, N.J.; Birren, B.W.; Ebel, G.D.; Henn, M.R. Molecular evolution of West Nile virus in a northern temperate region: Connecticut, USA 1999–2008. Virology 2011, 417, 203–210. [Google Scholar] [CrossRef]
- Brault, A.C.; Huang, C.Y.; Langevin, S.A.; Kinney, R.M.; Bowen, R.A.; Ramey, W.N.; Panella, N.A.; Holmes, E.C.; Powers, A.M.; Miller, B.R. A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat. Genet. 2007, 39, 1162–1166. [Google Scholar] [CrossRef]
- Pesko, K.N.; Ebel, G.D. West Nile virus population genetics and evolution. Infect. Genet. Evol. 2012, 12, 181–190. [Google Scholar] [CrossRef]
- Scholle, F.; Girard, Y.A.; Zhao, Q.Z.; Higgs, S.; Mason, P.W. trans-Packaged West Nile virus-like particles: Infectious properties in vitro and in infected mosquito vectors. J. Virol. 2004, 78, 11605–11614. [Google Scholar] [CrossRef]
- Ciota, A.T.; Ehrbar, D.J.; van Slyke, G.A.; Payne, A.F.; Willsey, G.G.; Viscio, R.E.; Kramer, L.D. Quantification of intrahost bottlenecks of West Nile virus in Culex pipiens mosquitoes using an artificial mutant swarm. Infect. Genet. Evol. 2012, 12, 557–564. [Google Scholar] [CrossRef]
- Brackney, D.E.; Pesko, K.N.; Brown, I.K.; Deardorff, E.R.; Kawatachi, J.; Ebel, G.D. West Nile virus genetic diversity is maintained during transmission by Culex pipiens quinquefasciatus Mosquitoes. PLoS One 2011, 6, e24466. [Google Scholar]
- Drake, J.W. Rates of Spontaneous Mutation Among RNA Viruses. Proc. Natl. Acad. Sci. USA 1993, 90, 4171–4175. [Google Scholar] [CrossRef]
- Jerzak, G.V.; Bernard, K.; Kramer, L.D.; Shi, P.Y.; Ebel, G.D. The West Nile virus mutant spectrum is host-dependant and a determinant of mortality in mice. Virology 2007, 360, 469–476. [Google Scholar] [CrossRef]
- Bertolotti, L.; Kitron, U.; Goldberg, T.L. Diversity and evolution of West Nile virus in Illinois and the United States, 2002–2005. Virology 2007, 360, 143–149. [Google Scholar] [CrossRef]
- Amore, G.; Bertolotti, L.; Hamer, G.L.; Kitron, U.D.; Walker, E.D.; Ruiz, M.O.; Brawn, J.D.; Goldberg, T.L. Multi-year evolutionary dynamics of West Nile virus in suburban Chicago, USA, 2005–2007. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2010, 365, 1871–1878. [Google Scholar] [CrossRef]
- Jerzak, G.; Bernard, K.A.; Kramer, L.D.; Ebel, G.D. Genetic variation in West Nile virus from naturally infected mosquitoes and birds suggests quasispecies structure and strong purifying selection. J. Gen. Virol. 2005, 86, 2175–2183. [Google Scholar]
- Ciota, A.T.; Jia, Y.; Payne, A.F.; Jerzak, G.; Davis, L.J.; Young, D.S.; Ehrbar, D.; Kramer, L.D. Experimental passage of St. Louis encephalitis virus in vivo in mosquitoes and chickens reveals evolutionarily significant virus characteristics. PLoS One 2009, 4, e7876. [Google Scholar]
- Ciota, A.T.; Ehrbar, D.J.; van Slyke, G.A.; Willsey, G.G.; Kramer, L.D. Cooperative interactions in the West Nile virus mutant swarm. BMC Evol. Biol. 2012, 12. [Google Scholar] [CrossRef]
- Ciota, A.T.; Lovelace, A.O.; Jones, S.A.; Payne, A.; Kramer, L.D. Adaptation of two flaviviruses results in differences in genetic heterogeneity and virus adaptability. J. Gen. Virol. 2007, 88, 2398–2406. [Google Scholar] [CrossRef]
- Brackney, D.E.; Beane, J.E.; Ebel, G.D. RNAi targeting of West Nile virus in mosquito midguts promotes virus diversification. PLoS Pathog. 2009, 5, e1000502. [Google Scholar] [CrossRef]
- Fitzpatrick, K.A.; Deardorff, E.R.; Pesko, K.; Brackney, D.E.; Zhang, B.; Bedrick, E.; Shi, P.Y.; Ebel, G.D. Population variation of West Nile virus confers a host-specific fitness benefit in mosquitoes. Virology 2010, 404, 89–95. [Google Scholar] [CrossRef]
- Ciota, A.T.; Lovelace, A.O.; Jia, Y.; Davis, L.J.; Young, D.S.; Kramer, L.D. Characterization of mosquito-adapted West Nile virus. J. Gen. Virol. 2008, 89, 1633–1642. [Google Scholar] [CrossRef]
- Bugbee, L.M.; Forte, L.R. The discovery of West Nile virus in overwintering Culex pipiens (Diptera: Culicidae) mosquitoes in Lehigh County, Pennsylvania. J. Am. Mosq. Control Assoc. 2004, 20, 326–327. [Google Scholar]
- Farajollahi, A.; Crans, W.J.; Nickerson, D.; Bryant, P.; Wolf, B.; Glaser, A.; Andreadis, T.G. Detection of West Nile virus RNA from the louse fly Icosta Americana (Diptera: Hippoboscidae). J. Am. Mosq. Control Assoc. 2005, 21, 474–476. [Google Scholar] [CrossRef]
- Reisen, W.K.; Fang, Y.; Lothrop, H.D.; Martinez, V.M.; Wilson, J.; O’Connor, P.; Carney, R.; Cahoon-Young, B.; Shafii, M.; Brault, A.C. Overwintering of West Nile Virus in Southern California. J. Med. Entomol. 2006, 43, 344–355. [Google Scholar] [CrossRef]
- Dohm, D.J.; Sardelis, M.R.; Turell, M.J. Experimental vertical transmission of West Nile virus by Culex pipiens (Diptera: Culicidae). J. Med. Entomol. 2002, 39, 640–644. [Google Scholar] [CrossRef]
- Nelms, B.M.; Fechter-Leggett, E.; Carroll, B.D.; Macedo, P.; Kluh, S.; Reisen, W.K. Experimental and natural vertical transmission of West Nile virus by California Culex (Diptera: Culicidae) mosquitoes. J. Med. Entomol. 2013, 50, 371–378. [Google Scholar]
- Ciota, A.T.; Koch, E.M.; Willsey, G.G.; Davis, L.J.; Jerzak, G.V.; Ehrbar, D.J.; Wilke, C.O.; Kramer, L.D. Temporal and spatial alterations in mutant swarm size of St. Louis encephalitis virus in mosquito hosts. Infect. Genet. Evol. 2011, 11, 460–468. [Google Scholar] [CrossRef]
- Davis, C.T.; Ebel, G.D.; Lanciotti, R.S.; Brault, A.C.; Guzman, H.; Siirin, M.; Lambert, A.; Parsons, R.E.; Beasley, D.W.; Novak, R.J.; et al. Phylogenetic analysis of North American West Nile virus isolates, 2001–2004: Evidence for the emergence of a dominant genotype. Virology 2005, 342, 252–265. [Google Scholar] [CrossRef]
- Twiddy, S.S.; Pybus, O.G.; Holmes, E.C. Comparative population dynamics of mosquito-borne flaviviruses. Infect. Genet. Evol. 2003, 3, 87–95. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Taylor, L.H.; Haydon, D.T. Population biology of multihost pathogens. Science 2001, 292, 1109–1112. [Google Scholar]
- Scott, T.W.; Weaver, S.C.; Mallampalli, V.L. Evolution of Mosquito-Borne Viruses. In The Evolutionary Biology of Viruses; Morse, S.S., Ed.; Raven Press, Ltd.: New York, NY, USA, 1994; pp. 293–324. [Google Scholar]
- Levins, R. Evolution in Changing Environments; Princeton University Press: Princeton, NJ, USA, 1968. [Google Scholar]
- Ciota, A.T.; Lovelace, A.O.; Ngo, K.A.; Le, A.N.; Maffei, J.G.; Franke, M.A.; Payne, A.F.; Jones, S.A.; Kauffman, E.B.; Kramer, L.D. Cell-specific adaptation of two flaviviruses following serial passage in mosquito cell culture. Virology 2007, 357, 165–174. [Google Scholar] [CrossRef]
- Deardorff, E.R.; Fitzpatrick, K.A.; Jerzak, G.V.; Shi, P.Y.; Kramer, L.D.; Ebel, G.D. West Nile virus experimental evolution in vivo and the trade-off hypothesis. PLoS Pathog. 2011, 7, e1002335. [Google Scholar] [CrossRef]
- Wilke, C.O.; Wang, J.L.; Ofria, C.; Lenski, R.E.; Adami, C. Evolution of digital organisms at high mutation rates leads to survival of the flattest. Nature 2001, 412, 331–333. [Google Scholar] [CrossRef]
- Tsetsarkin, K.A.; McGee, C.E.; Volk, S.M.; Vanlandingham, D.L.; Weaver, S.C.; Higgs, S. Epistatic roles of E2 glycoprotein mutations in adaption of chikungunya virus to Aedes albopictus and Ae. Aegypti mosquitoes. PLoS One 2009, 4, e6835. [Google Scholar]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef]
- Aaskov, J.; Buzacott, K.; Thu, H.M.; Lowry, K.; Holmes, E.C. Long-term transmission of defective RNA viruses in humans and Aedes mosquitoes. Science 2006, 311, 236–238. [Google Scholar] [CrossRef]
- Lambrechts, L. Dissecting the genetic architecture of host-pathogen specificity. PLoS Pathog. 2010, 6, e1001019. [Google Scholar] [CrossRef]
- Lambrechts, L.; Quillery, E.; Noel, V.; Richardson, J.H.; Jarman, R.G.; Scott, T.W.; Chevillon, C. Specificity of resistance to dengue virus isolates is associated with genotypes of the mosquito antiviral gene Dicer-2. Proc. Biol. Sci. 2013, 280. [Google Scholar] [CrossRef]
- Snappin, K.W.; Holmes, E.C.; Young, D.S.; Bernard, K.A.; Kramer, L.D.; Ebel, G.D. Declining growth rate of West Nile virus in North America. J. Virol. 2007, 81, 2531–2534. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. West Nile virus disease cases and deaths reported to CDC by year and clinical presentation, 1999–2012. Available online: http://www.cdc.gov/westnile/resources/pdfs/cummulative/99_2012_CasesAndDeathsClinicalPresentationHumanCases.pdf (accessed 1 December 2013).
- Duggal, N.K.; D’Anton, M.; Xiang, J.; Seiferth, R.; Day, J.; Nasci, R.; Brault, A.C. Sequence analyses of 2012 west nile virus isolates from Texas fail to associate viral genetic factors with outbreak magnitude. Am. J. Trop. Med. Hyg. 2013, 89, 205–210. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ciota, A.T.; Kramer, L.D. Vector-Virus Interactions and Transmission Dynamics of West Nile Virus. Viruses 2013, 5, 3021-3047. https://doi.org/10.3390/v5123021
Ciota AT, Kramer LD. Vector-Virus Interactions and Transmission Dynamics of West Nile Virus. Viruses. 2013; 5(12):3021-3047. https://doi.org/10.3390/v5123021
Chicago/Turabian StyleCiota, Alexander T., and Laura D. Kramer. 2013. "Vector-Virus Interactions and Transmission Dynamics of West Nile Virus" Viruses 5, no. 12: 3021-3047. https://doi.org/10.3390/v5123021
APA StyleCiota, A. T., & Kramer, L. D. (2013). Vector-Virus Interactions and Transmission Dynamics of West Nile Virus. Viruses, 5(12), 3021-3047. https://doi.org/10.3390/v5123021



