A Hypothesis for Bacteriophage DNA Packaging Motors
Abstract
:1. Introduction
2. Examination of Some Past Assumptions
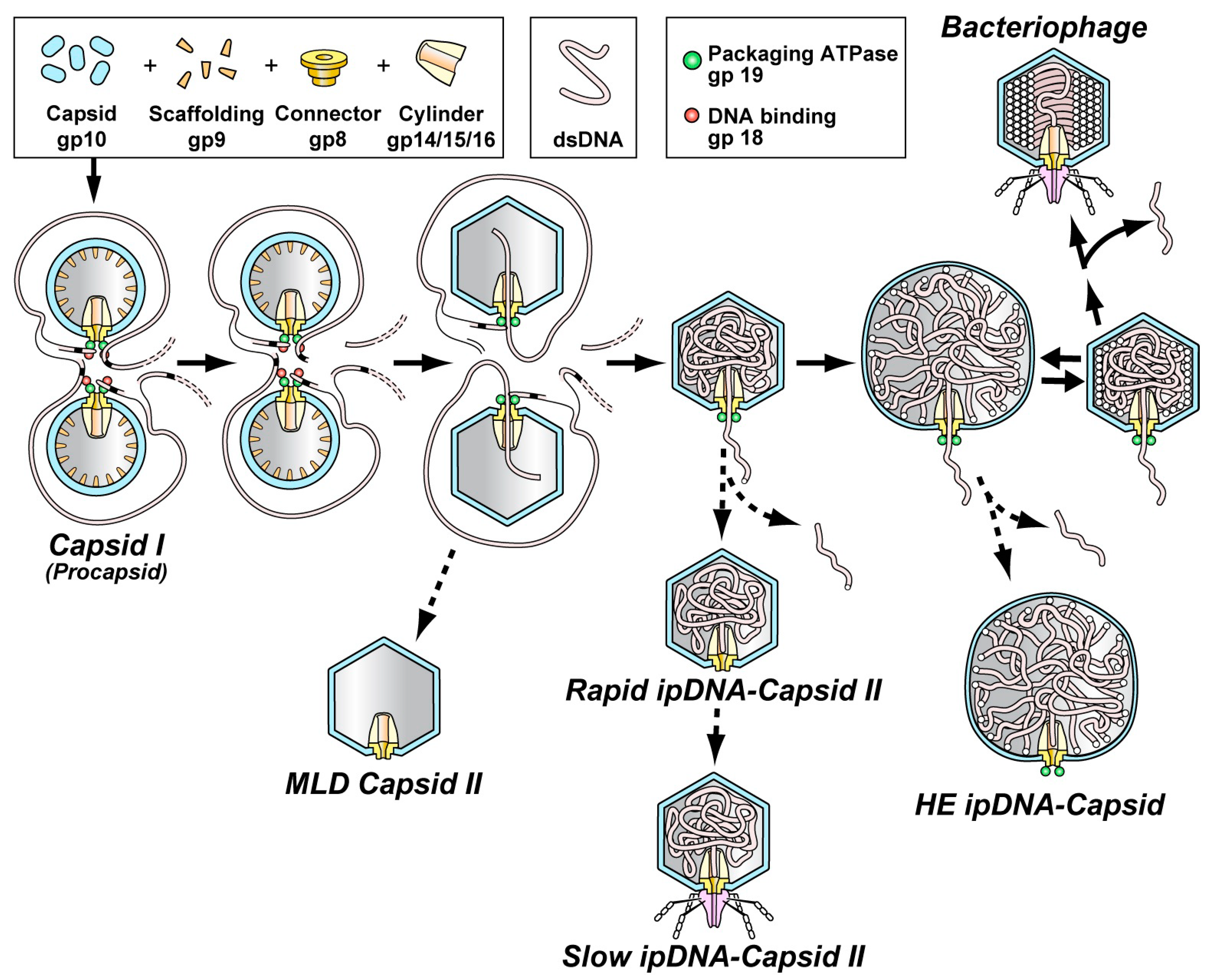
3. Key Data from Previous Studies of the Type 1 Cycle
3.1. Thermal Ratcheting and Its Limitations
3.2. Connector Dynamics in the Type 1 Cycle
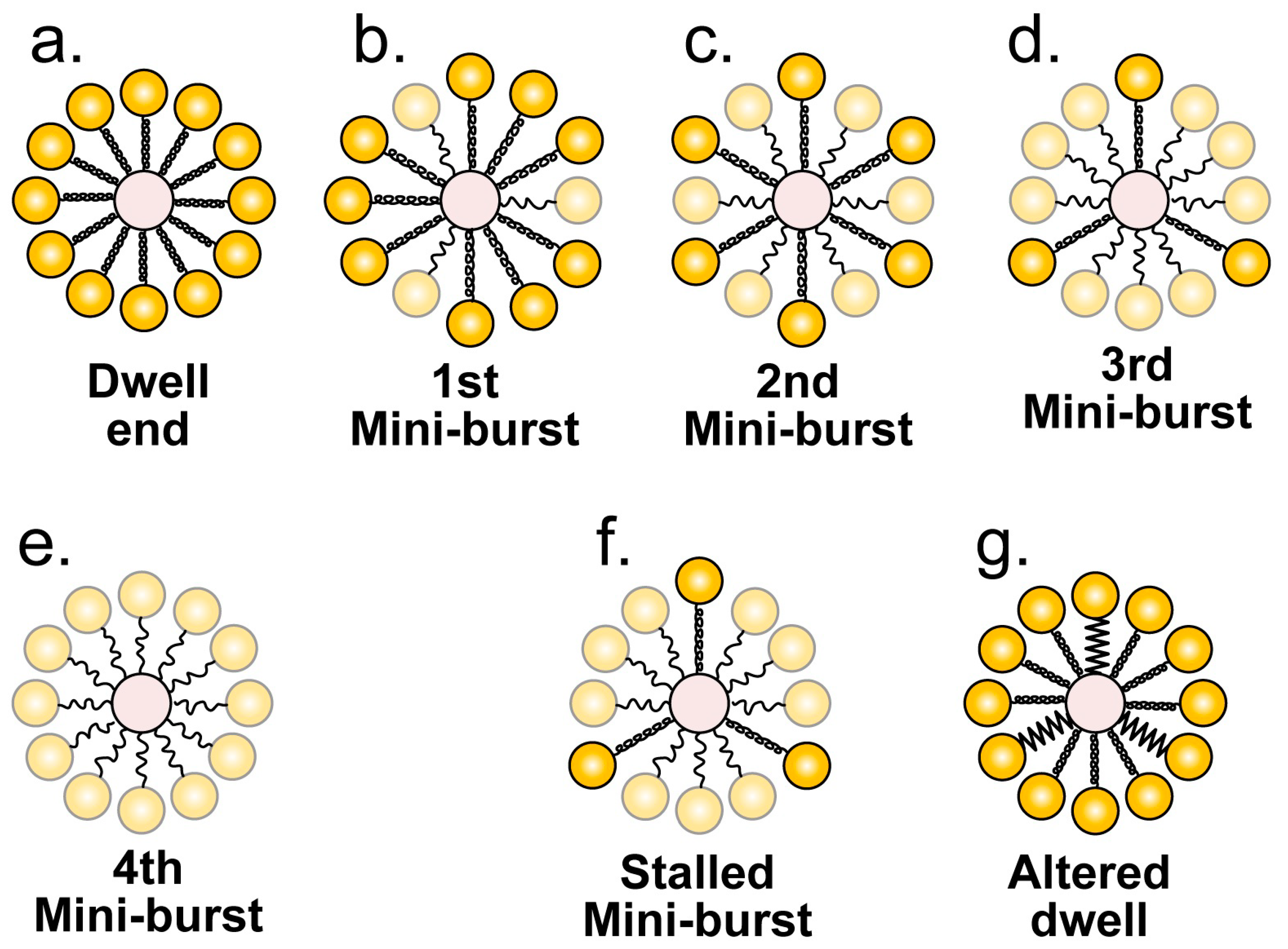
3.3. Fine Structure of the Single-Molecule Nanometry
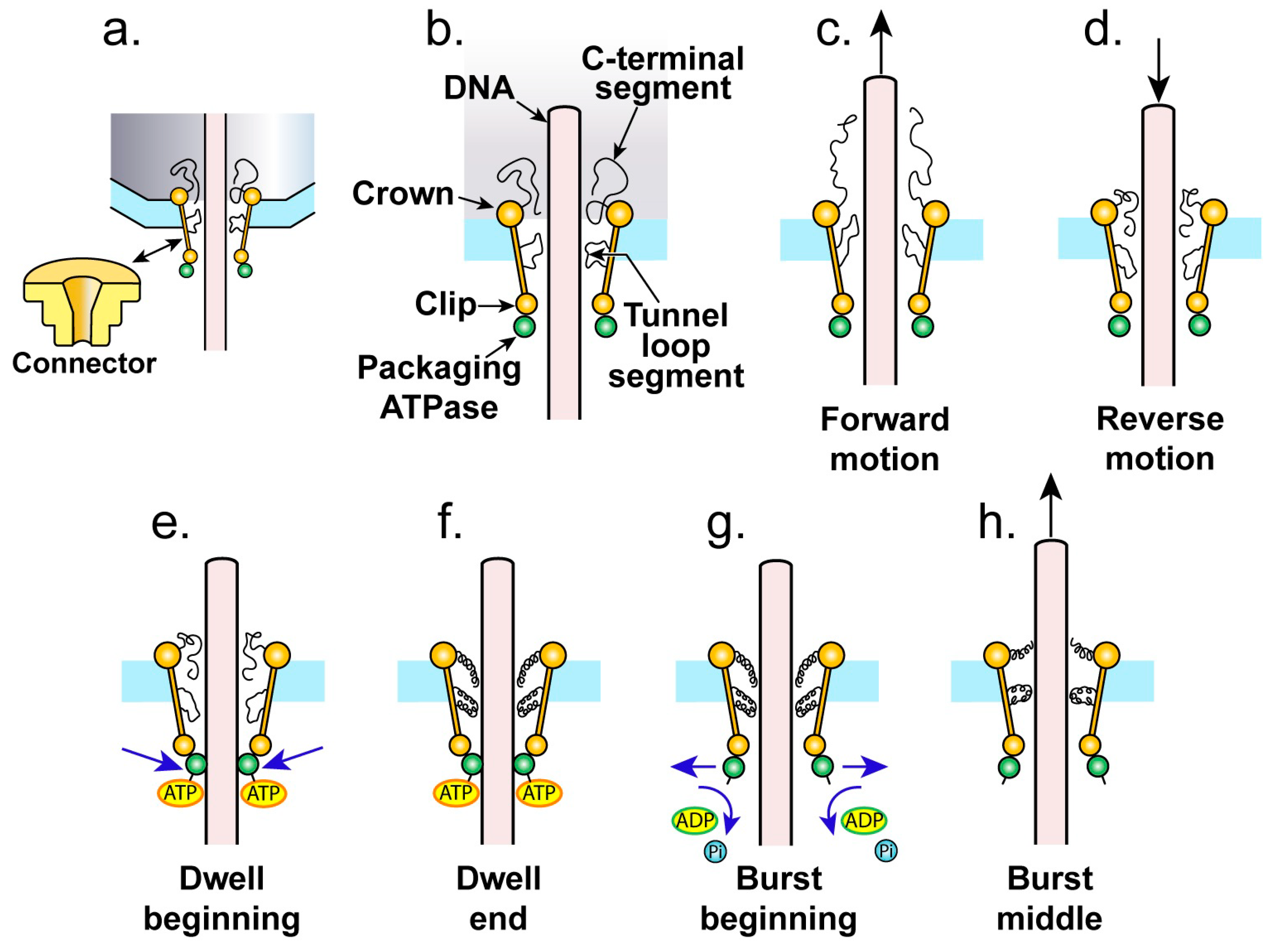
4. The Proposed Type 1 Cycle
4.1. The Ratcheting Component
4.2. The Non-Ratcheting Component: ATP-Driven Mobile Peptide Segment Activation
5. The Type 2 Cycle and Terminase Cleavage of a Concatemer
6. Comparison with Thermal Ratcheting in Other Systems
7. Symmetry Mismatches, Evolution and Non-Viral Eukaryotic Motor Proteins
8. Other, Recently Proposed Type 1 Cycles: Predictions of the Cycle of Figure 2 and Figure 3
Acknowledgments
References
- Pajunen, M.I.; Elizondo, M.R.; Skurnik, M.; Kieleczawa, J.; Molineux, I.J. Complete nucleotide sequence and likely recombinatorial origin of bacteriophage T3. J. Mol. Biol. 2002, 319, 1115–1132. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, H.; Kimura, M.; Hashimoto, C. In vitro cleavage of the concatemer joint of bacteriophage T3 DNA. Virology 1990, 174, 26–34. [Google Scholar] [CrossRef]
- Catalano, C.E. The terminase enzyme from bacteriophage lambda: a DNA-packaging machine. Cell Mol. Life Sci. 2000, 57, 128–148. [Google Scholar] [CrossRef] [PubMed]
- Petrov, A.S.; Harvey, S.C. Packaging double-helical DNA into viral capsids: structures, forces, and energetics. Biophys. J. 2008, 95, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Rao, V.B.; Feiss, M. The bacteriophage DNA packaging motor. Annu. Rev. Genet. 2008, 42, 647–681. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.J.; Schwartz, C.; Guo, P. Construction of bacteriophage phi29 DNA packaging motor and its applications in nanotechnology and therapy. Ann. Biomed. Engineering 2009, 37, 2064–2081. [Google Scholar] [CrossRef]
- Ponchon, L.; Boulanger, P.; Labesse, G.; Letellier, L. The endonuclease domain of bacteriophage terminases belongs to the resolvase/integrase/ribonuclease H superfamily: a bioinformatics analysis validated by a functional study on bacteriophage T5. J. Biol. Chem. 2006, 281, 5829–5836. [Google Scholar] [CrossRef]
- Al-Zahrani, A.S.; Kondabagil, K.; Gao, S.; Kelly, N.; Ghosh-Kumar, M.; Rao, V.B. The small terminase, gp16, of bacteriophage T4 is a regulator of the DNA packaging motor. J. Biol. Chem. 2009, 284, 24490–24500. [Google Scholar] [CrossRef]
- Bogner, E. Human cytomegalovirus terminase as a target for antiviral chemotherapy. Rev. Med. Virol. 2002, 12, 115–127. [Google Scholar] [CrossRef]
- Cardone, G.; Winkler, D.C.; Trus, B.L.; Cheng, N.; Heuser, J.E.; Newcomb, W.W.; Brown, J.C.; Steven, A.C. Visualization of the herpes simplex virus portal in situ by cryo-electron tomography. Virology 2007, 361, 426–434. [Google Scholar] [CrossRef]
- Sheaffer, A.K.; Newcomb, W.W.; Gao, M.; Yu, D.; Weller, S.K.; Brown, J.C.; Tenney, D.J. Herpes simplex virus DNA cleavage and packaging proteins associate with the procapsid prior to its maturation. J. Virol. 2001, 75, 687–698. [Google Scholar] [CrossRef]
- Serwer, P.; Hayes, S.J.; Zaman, S.; Lieman, K.; Rolando, M.; Hardies, S.C. Improved isolation of undersampled bacteriophages: finding of distant terminase genes. Virology 2004, 329, 412–424. [Google Scholar] [CrossRef] [PubMed]
- Baker, M.L.; Jiang, W.; Rixon, F.J.; Chiu, W. Common ancestry of herpesviruses and tailed DNA bacteriophages. J. Virol. 2005, 79, 14967–14970. [Google Scholar] [CrossRef] [PubMed]
- Kutschera, U.; Niklas, K.J. The modern theory of biological evolution: an expanded synthesis. Naturwissenschaften 2004, 91, 255–276. [Google Scholar] [CrossRef]
- Moll, W.D.; Guo, P. Translocation of nicked but not gapped DNA by the packaging motor of bacteriophage phi29. J. Mol. Biol. 2005, 351, 100–107. [Google Scholar] [CrossRef]
- Aathavan, K.; Politzer, A.T.; Kaplan, A.; Moffitt, J.R.; Chemla, Y.R.; Grimes, S.; Jardine, P.J.; Anderson, D.L.; Bustamante, C. Substrate interactions and promiscuity in a viral DNA packaging motor. Nature 2009, 461, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Oram, M.; Sabanayagam, C.; Black, L.W. Modulation of the packaging reaction of bacteriophage t4 terminase by DNA structure. J. Mol. Biol. 2008, 381, 61–72. [Google Scholar] [CrossRef]
- Hendrix, R.W. Symmetry mismatch and DNA packaging in large bacteriophages. Proc. Natl. Acad. Sci. U. S. A. 1978, 75, 4779–4783. [Google Scholar] [CrossRef]
- Chen, C.; Guo, P. Sequential action of six virus-encoded DNA-packaging RNAs during phage phi29 genomic DNA translocation. J. Virol. 1997, 71, 3864–3871. [Google Scholar] [CrossRef]
- Grimes, S.; D. Anderson, D. The bacteriophage phi29 packaging proteins supercoil the DNA ends. J. Mol. Biol. 1997, 266, 901–914. [Google Scholar] [CrossRef]
- Simpson, A.A.; Tao, Y.; Leiman, P.G.; Badasso, M.O.; He, Y.; Jardine, P.J.; Olson, N.H.; Morais, M.C.; Grimes, S.; Anderson, D.L.; et al. Structure of the bacteriophage phi29 DNA packaging motor. Nature 2000, 408, 745–750. [Google Scholar] [CrossRef]
- Guasch, A.; Pous, J.; Ibarra, B.; Gomis-Ruth, F.X.; Valpuesta, J.M.; Sousa, N.; Carrascosa, J.L.; Coll, M. Detailed architecture of a DNA translocating machine: the high-resolution structure of the bacteriophage phi29 connector particle. J. Mol. Biol. 2002, 315, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Hügel, T.; Michaelis, J.; Hetherington, C.L.; Jardine, P.J.; Grimes, S.; Walter, J.M.; Falk, W.; Anderson, D.L.; Bustamante, C. Experimental test of connector rotation during DNA packaging into bacteriophage phi29 capsids. PLoS Biol. 2007, 5, e59. [Google Scholar] [CrossRef] [PubMed]
- Baumann, R.G.; Mullaney, J.; Black, L.W. Portal fusion protein constraints on function in DNA packaging of bacteriophage T4. Mol. Microbiol. 2006, 61, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Serwer, P. The source of energy for bacteriophage DNA packaging: an osmotic pump explains the data. Biopolymers 1988, 27, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Serwer, P. Models of bacteriophage DNA packaging motors. J. Struct. Biol. 2003, 141, 179–188. [Google Scholar] [CrossRef]
- Alberts, B.; Miake-Lye, R. Unscrambling the puzzle of biological machines: The importance of the details. Cell 1992, 68, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Howard, J. Motor proteins as nanomachines: The roles of thermal fluctuations in generating force and motion. Séminaire Poincaré 2009, 12, 33–44. [Google Scholar]
- Mickler, M.; Schleiff, E.; Hügel, T. From biological towards artificial molecular motors. Chemphyschem. 2008, 9, 1503–1509. [Google Scholar] [CrossRef]
- Serwer, P.; Wright, E.T.; Hakala, K.; Weintraub, S.T.; Su, M.; Jiang, W. DNA packaging-associated hyper-capsid expansion of bacteriophage T3. J. Mol. Biol. 2010, 397, 361–374. [Google Scholar]
- Fuller, D.N.; Raymer, D.M.; Rickgauer, J.P.; Robertson, R.M.; Catalano, C.E.; Anderson, D.L.; Grimes, S.; Smith, D.E. Measurements of single DNA molecule packaging dynamics in bacteriophage lambda reveal high forces, high motor processivity, and capsid transformations. J. Mol. Biol. 2007, 373, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Maluf, N.K.; Catalano, C.E. Packaging of a unit-length viral genome: the role of nucleotides and the gpD decoration protein in stable nucleocapsid assembly in bacteriophage lambda. J. Mol. Biol. 2008, 383, 1037–1048. [Google Scholar] [CrossRef] [PubMed]
- JConway, J.F.; Cheng, N.; Ross, P.D.; Hendrix, R.W.; Duda, R.L.; Steven, A.C. A thermally induced phase transition in a viral capsid transforms the hexamers, leaving the pentamers unchanged. J. Struct. Biol. 2007, 158, 224–232. [Google Scholar]
- Gertsman, I.; Komives, E.A.; Johnson, J.E. HK97 maturation studied by crystallography and H/2H exchange reveals the structural basis for exothermic particle transitions J. Mol. Biol. 2010, 397, 560–574. [Google Scholar] [CrossRef] [PubMed]
- Tuma, R.; Prevelige, P.E., Jr.; Thomas, G.J., Jr. Mechanism of capsid maturation in a double-stranded DNA virus. Proc. Natl. Acad. Sci. U. S. A. 1998, 95, 9885–9890. [Google Scholar] [CrossRef]
- Lander, G.C.; Evilevitch, A.; Jeembaeva, M.; Potter, C.S.; Carragher, B.; Johnson, J.E. Bacteriophage lambda stabilization by auxiliary protein gpD: timing, location, and mechanism of attachment determined by cryo-EM. Structure 2008, 16, 1399–1406. [Google Scholar] [CrossRef]
- Agirrezabala, X.; Martin-Benito, J.; Valle, M.; Gonzalez, J.M.; Valencia, A.; J. Valpuesta, J.M.; Carrascosa, J.L. Structure of the connector of bacteriophage T7 at 8A resolution: structural homologies of a basic component of a DNA translocating machinery. J. Mol. Biol. 2005, 347, 895–902. [Google Scholar] [CrossRef]
- Lebedev, A.A.; Krause, M.H.; Isidro, A.L.; Vagin, A.A.; Orlova, E.V.; Turner, J.; Dodson, E.J.; Tavares, P.; Antson, A.A. Structural framework for DNA translocation via the viral portal protein. EMBO J. 2007, 26, 1984–1994. [Google Scholar] [CrossRef]
- Hardies, S.C.; Serwer, P. Alignment and structural modeling of DNA packaging ATPases: Utilization of a steric clamp to hold the DNA. In Proceeding of the XXth Biennial Conference on Phage/Virus Assembly, Barrie, ON, Canada, 25–27 May 2007. [Google Scholar]
- Grimes, S.; Jardine, P.J.; Anderson, D. Bacteriophage phi 29 DNA packaging. Adv. Virus Res. 2002, 58, 255–294. [Google Scholar]
- Guo, P.; Peterson, C.; Anderson, D. Prohead and DNA-gp3-dependent ATPase activity of the DNA packaging protein gp16 of bacteriophage phi 29. J. Mol. Biol. 1987, 197, 229–236. [Google Scholar] [CrossRef]
- Hamada, K.; Fujisawa, H.; Minagawa, T. Characterization of ATPase activity of a defined in vitro system for packaging of bacteriophage T3 DNA. Virology 1987, 159, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Yanagida, T. Fluctuation as a tool of biological molecular machines. Biosystems 2008, 93, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Verbrugge, S.; Lechner, B.; Woehlke, G.; Peterman, E.J. Alternating-site mechanism of kinesin-1 characterized by single-molecule FRET using fluorescent ATP analogues. Biophys. J. 2009, 97, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Petrov, A.S.; Lim-Hing, K.; Harvey, S.C. Packaging of DNA by bacteriophage epsilon15: structure, forces, and thermodynamics. Structure 2007, 15, 807–812. [Google Scholar] [CrossRef]
- Fang, P.A.; Wright, E.T.; Weintraub, S.T.; Hakala, K.; Wu, W.; Serwer, P.; Jiang, W. Visualization of bacteriophage T3 capsids with DNA incompletely packaged in vivo. J. Mol. Biol. 2008, 384, 1384–1399. [Google Scholar] [CrossRef]
- Smith, D.E.; Tans, S.J.; Smith, S.B.; Grimes, S.; Anderson, D.L.; Bustamante, C. The bacteriophage straight phi29 portal motor can package DNA against a large internal force. Nature 2001, 413, 748–752. [Google Scholar] [CrossRef]
- Rickgauer, J.P.; Fuller, D.N.; Grimes, S.; Jardine, P.J.; Anderson, D.L.; Smith, D.E. Portal motor velocity and internal force resisting viral DNA packaging in bacteriophage phi29. Biophys. J. 2008, 94, 159–167. [Google Scholar] [CrossRef]
- Fuller, D.N.; Raymer, D.M.; Kottadiel, V.I.; Rao, V.B.; Smith, D.E. Single phage T4 DNA packaging motors exhibit large force generation, high velocity, and dynamic variability. Proc. Natl. Acad. Sci. U. S. A. 2007, 104, 16868–16873. [Google Scholar] [CrossRef]
- Koch, A.L. Shrinkage of growing Escherichia coli cells by osmotic challenge. J. Bact. 1984, 159, 919–924. [Google Scholar] [CrossRef]
- Ray, K.; Sabanayagam, C.R.; Lakowicz, J.R.; Black, L.W. DNA crunching by a viral packaging motor: Compression of a procapsid-portal stalled Y-DNA substrate. Virology 2010, 398, 224–232. [Google Scholar] [CrossRef]
- Moffitt, J.R.; Chemla, Y.R.; Aathavan, K.; Grimes, S.; Jardine, P.J.; Anderson, D.L.; Bustamante, C. Intersubunit coordination in a homomeric ring ATPase. Nature 2009, 457, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Chemla, Y.R.; Aathavan, K.; Michaelis, J.; Grimes, S.; Jardine, P.J.; Anderson, D.L.; Bustamante, C. Mechanism of force generation of a viral DNA packaging motor. Cell 2005, 122, 683–692. [Google Scholar] [CrossRef]
- Gope, R.; Serwer, P. Bacteriophage P22 in vitro DNA packaging monitored by agarose gel electrophoresis: rate of DNA entry into capsids. J. Virol. 1983, 47, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Son, M.; Hayes, S.J.; Serwer, P. Optimization of the in vitro packaging efficiency of bacteriophage T7 DNA: effects of neutral polymers. Gene 1989, 82, 321–325. [Google Scholar] [CrossRef] [PubMed]
- Shibata, H.; Fujisawa, H.; Minagawa, T. Characterization of the bacteriophage T3 DNA packaging reaction in vitro in a defined system. J. Mol. Biol. 1987, 196, 845–851. [Google Scholar] [CrossRef]
- Oliveira, L.; Alonso, J.C.; Tavares, P. A defined in vitro system for DNA packaging by the bacteriophage SPP1: insights into the headful packaging mechanism. J. Mol. Biol. 2005, 353, 529–539. [Google Scholar] [CrossRef]
- Morais, M.C.; Koti, J.S.; Bowman, V.D.; Reyes-Aldrete, E.; Anderson, D.L.; Rossmann, M.G. Defining molecular and domain boundaries in the bacteriophage phi29 DNA packaging motor. Structure 2008, 16, 1267–1274. [Google Scholar] [CrossRef]
- Cuervo, A.; Vaney, M.C.; Antson, A.A.; Tavares, P.; Oliveira, L. Structural rearrangements between portal protein subunits are essential for viral DNA translocation. J. Biol. Chem. 2007, 282, 18907–18913. [Google Scholar] [CrossRef]
- Oliveira, L.; Henriques, A.O.; Tavares, P. Modulation of the viral ATPase activity by the portal protein correlates with DNA packaging efficiency. J. Biol. Chem. 2006, 281, 21914–21923. [Google Scholar] [CrossRef]
- Michaelis, J.; Muschielok, A.; Adrecka, J.; Kügel, W.; Moffitt, J.R. DNA based molecular motors. Phys. Life Rev. 2009, 6, 250–266. [Google Scholar] [CrossRef]
- Fink, T.M.; Ball, R.C. How many conformations can a protein remember? Phys. Rev. Lett. 2001, 87, 198103. [Google Scholar] [CrossRef] [PubMed]
- Atz, R.; Ma, S.; Aathavan, K.; Bustamante, C.; Gao, J.; Anderson, D.L.; Grimes, S. Role of ϕ29 connector channel domains in late phase DNA-gp3 packaging. In Proceeding of the XXth Biennial Conference on Phage/Virus Assembly, Barrie, ON, Canada, 25–27 May 2007. [Google Scholar]
- Pearson, R.K.; Fox, M.S. Effects of DNA heterologies on bacteriophage lambda packaging. Genetics 1988, 118, 5–12. [Google Scholar] [CrossRef]
- Louie, D.; Serwer, P. Quantification of the effect of excluded volume on double-stranded DNA. J. Mol. Biol. 1994, 242, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, C.F. Multiple molecular mechanisms for multidrug resistance transporters. Nature 2007, 446, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Linton, K.J. Structure and function of ABC transporters. Physiology 2007, 22, 122–130. [Google Scholar] [CrossRef]
- Lin, Z.; Madan, D.; Rye, H.S. GroEL stimulates protein folding through forced unfolding. Nature Struct. Mol. Biol. 2008, 15, 303–311. [Google Scholar] [CrossRef]
- Horwich, A.L.; Fenton, W.A. Chaperonin-mediated protein folding: using a central cavity to kinetically assist polypeptide chain folding. Q. Rev. Biophys. 2009, 42, 83–116. [Google Scholar] [CrossRef]
- Jewett, A.I.; Shea, J.E. Reconciling theories of chaperonin accelerated folding with experimental evidence. Cell Mol. Life Sci. 2010, 67, 255–276. [Google Scholar] [CrossRef]
- Zaikin, A.; Garcia-Ojalvo, J.; Bascones, R.; Ullner, E.; Kurths, J. Doubly stochastic coherence via noise-induced symmetry in bistable neural models. Phys. Rev. Lett. 2003, 90, 030601. [Google Scholar] [CrossRef]
- Ray, K.; Oram, M.; Ma, J.; Black, L.W. Portal control of viral prohead expansion and DNA packaging. Virology 2009, 391, 44–50. [Google Scholar] [CrossRef]
- Fujisawa, H.; Kimura, M.; Hashimoto, C. In vitro cleavage of the concatemer joint of bacteriophage T3 DNA. Virology 1990, 174, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.; Wyckoff, E.; Hayden, M.; Sampson, L.; Eppler, K.; Randall, S.; Moreno, E.T.; Serwer, P. Bacteriophage P22 portal protein is part of the gauge that regulates packing density of intravirion DNA. J. Mol. Biol. 1992, 224, 1055–1074. [Google Scholar] [CrossRef] [PubMed]
- Nemecek, D.; Lander, G.C.; Johnson, J.E.; Casjens, S.R.; Thomas, G.J., Jr. Assembly architecture and DNA binding of the bacteriophage P22 terminase small subunit. J. Mol. Biol. 2008, 383, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Morais, M.C.; Tao, Y.; Olson, N.H.; Grimes, S.; Jardine, P.J.; Anderson, D.L.; Baker, T.S.; Rossmann, M.G. Cryoelectron-microscopy image reconstruction of symmetry mismatches in bacteriophage phi29. J. Struct. Biol. 2001, 135, 38–46. [Google Scholar] [CrossRef]
- Feynmann, R.P.; Leighton, R.B.; Sands, M.L. The Feynman Lectures on Physics, 2nd ed.; Addison-Wesley: Reading, MA, USA, 1963; Volume I, Chapter 46. [Google Scholar]
- Fujisawa, H.; Morita, M. Phage DNA packaging. Genes Cells 1997, 2, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Huxley, A.F. Muscle structure and theories of contraction. Prog. Biophys. Biophys. Chem. 1957, 7, 255–318. [Google Scholar] [CrossRef] [PubMed]
- Donnan, F.G. The mystery of Life. In Smithsonian Report for 1929; 1929; pp. 309–321. [Google Scholar]
- Ishii, Y.; Taniguchi, Y.; Iwaki, M.; Yanagida, T. Thermal fluctuations biased for directional motion in molecular motors. BioSystems 2008, 93, 34–38. [Google Scholar] [CrossRef]
- Iwaki, M.; Iwane, A.H.; Shimokawa, T.; Cooke, R.; Yanagida, T. Brownian search-and-catch mechanism for myosin-VI steps. Nat. Chem. Biol. 2009, 5, 403–405. [Google Scholar] [CrossRef]
- Sun, S.; Kondabagil, K.; Draper, B.; Alam, T.I.; Bowman, V.D.; Zhang, Z.; Hegde, S.; Fokine, A.; Rossmann, M.G.; Rao, V.B. The structure of the phage T4 DNA packaging motor suggests a mechanism dependent on electrostatic forces. Cell 2008, 135, 1251–1262. [Google Scholar] [CrossRef]
- Singh, K.P.; Ropars, G.; Brunel, M.; Le Floch, A. Lever-assisted two-noise stochastic resonance. Phys. Rev. Lett. 2003, 90, 073901. [Google Scholar] [CrossRef]
- Ropars, G.; Singh, K.P.; Brunel, M.; Dore, F.; Le Floch, A. The dual stochastic response of nonlinear systems. Europhys. Lett. 2004, 68, 755–761. [Google Scholar] [CrossRef]
- Beuron, F.; Maurizi, M.R.; Belnap, D.M.; Kocsis, E.; Booy, F.P.; Kessel, M.; Steven, A.C. At sixes and sevens: characterization of the symmetry mismatch of the ClpAP chaperone-assisted protease. J. Struct. Biol. 1998, 123, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Bewley, M.C.; Graziano, V.; Griffin, K.; Flanagan, J.M. The asymmetry in the mature amino-terminus of ClpP facilitates a local symmetry match in ClpAP and ClpXP complexes. J. Struct. Biol. 2006, 153, 113–128. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Sheng, S.; Shao, Z.; Guo, P. A dimer as a building block in assembling RNA. A hexamer that gears bacterial virus phi29 DNA-translocating machinery. J. Biol. Chem. 2000, 275, 17510–17516. [Google Scholar] [CrossRef] [PubMed]
- Klug, A. Architectural design of spherical viruses. Nature 1983, 303, 378–379. [Google Scholar] [CrossRef]
- Jiang, W.; Chang, J.; Jakana, P.; Weigele, J.; King, J.A.; Chiu, W. Structure of epsilon15 bacteriophage reveals genome organization and DNA packaging/injection apparatus. Nature 2006, 439, 612–616. [Google Scholar] [CrossRef]
- Johnson, J.E.; Chiu, W. DNA packaging and delivery machines in tailed bacteriophages. Curr. Opin. Struct. Biol. 2007, 17, 237–243. [Google Scholar] [CrossRef]
- Yu, J.; Moffitt, J.; Hetherington, C.L.; Bustamante, C.; Oster, G. Mechanochemistry of a viral DNA packging motor. J. Mol. Biol. 2010, 400, 186–203. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Louie, D.; Serwer, P. Single-event analysis of the packaging of bacteriophage T7 DNA concatemers in vitro. Biophys. J. 1999, 77, 1627–1637. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2010 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serwer, P. A Hypothesis for Bacteriophage DNA Packaging Motors. Viruses 2010, 2, 1821-1843. https://doi.org/10.3390/v2091821
Serwer P. A Hypothesis for Bacteriophage DNA Packaging Motors. Viruses. 2010; 2(9):1821-1843. https://doi.org/10.3390/v2091821
Chicago/Turabian StyleSerwer, Philip. 2010. "A Hypothesis for Bacteriophage DNA Packaging Motors" Viruses 2, no. 9: 1821-1843. https://doi.org/10.3390/v2091821
APA StyleSerwer, P. (2010). A Hypothesis for Bacteriophage DNA Packaging Motors. Viruses, 2(9), 1821-1843. https://doi.org/10.3390/v2091821





