Role of Cellular Lipids in Positive-Sense RNA Virus Replication Complex Assembly and Function
Abstract
:1. Introduction
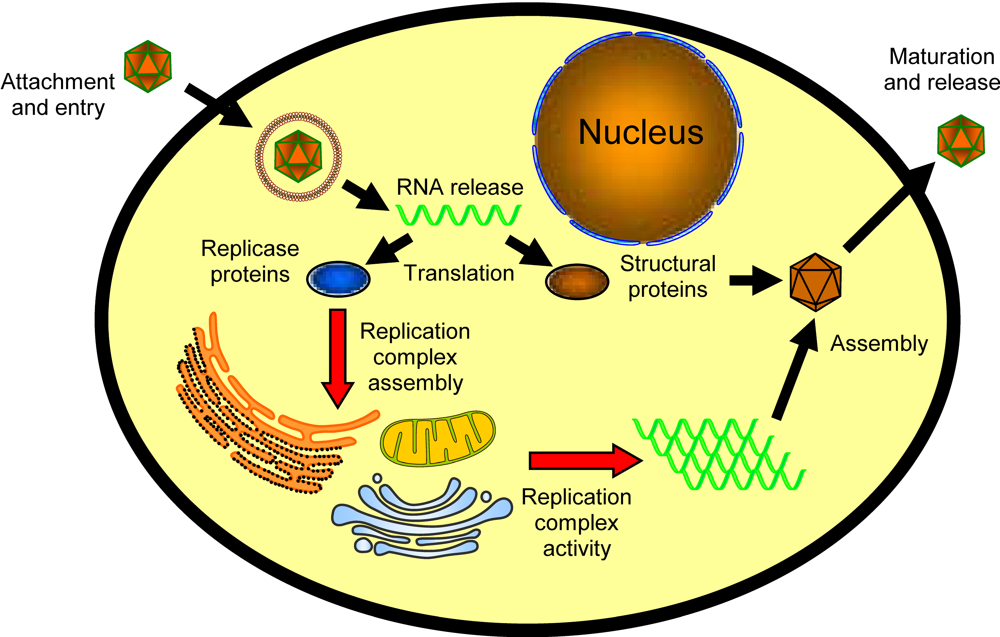
1.1. Lipids and replication complex targeting and assembly
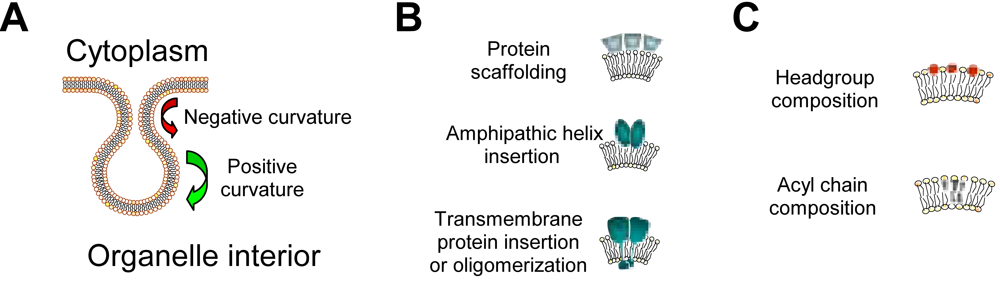
1.2. Lipids and membrane structure alterations
| Family | Virus | Membrane(s) | References |
|---|---|---|---|
| Arteriviridae | Equine arteritis virus | Endoplasmic reticulum | [15,16] |
| Bromoviridae | Alfalfa mosaic virusBrome mosaic virus | VacuoleEndoplasmic reticulum | [17,18] |
| Coronaviridae | SARS-coronavirus | Endoplasmic reticulum/Golgi | [19-21] |
| Flaviviridae | Hepatitis C virusDengue virusWest Nile virus | Endoplasmic reticulumEndoplasmic reticulumEndoplasmic reticulum/Golgi | [22-24] |
| Nodaviridae | Flock House virus | Mitochondria | [25,26] |
| Picornaviridae | Poliovirus | Endoplasmic reticulum/Golgi | [27-29] |
| Togaviridae | Rubella virusSemliki Forest virus | LysosomesEndosomes/lysosomes | [30-32] |
| Tombusviridae | Carnation Italian ringspot virusCucumber necrosis virusTomato bushy stunt virus | MitochondriaEndoplasmic reticulumPeroxisome | [33-36] |
| Tymoviridae | Turnip yellow mosaic virus | Chloroplast | [37] |
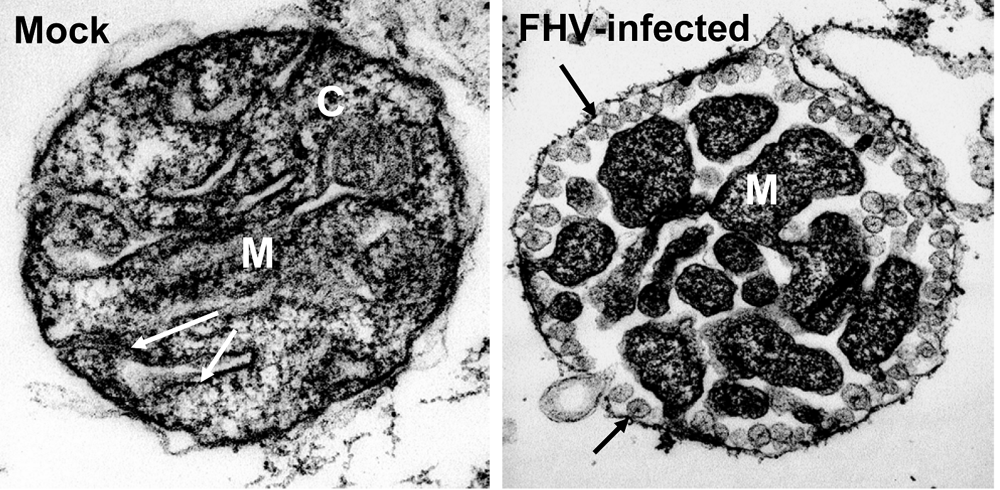
1.3. Lipids and replication complex function

2. Conclusions
References
- Morse, S.S. The public health threat of emerging viral diseases. J. Nutrition 1997, 127 (Suppl 5) , 951S–957S. [Google Scholar]
- Gubler, D.J. The global emergence/resurgence of arboviral diseases as public health problems. Arch. Med. Res. 2002, 33, 330–342. [Google Scholar] [CrossRef] [PubMed]
- Drake, J.W.; Holland, J.J. Mutation rates among RNA viruses. Proc. Natl. Acad. Sci. USA 1999, 96, 13910–13913. [Google Scholar] [CrossRef]
- Pillay, D.; Zambon, M. Antiviral drug resistance. BMJ 1998, 317, 660–662. [Google Scholar] [PubMed]
- Fox, J. L. Antivirals become a broader enterprise. Nat. Biotechnol. 2007, 25, 1395–1402. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wang, C.; Sumpter Jr. , R.; Brown, M.S.; Goldstein, J.L.; Gale Jr., M. Disruption of hepatitis C virus RNA replication through inhibition of host protein geranylgeranylation. Proc. Natl. Acad. Sci. USA 2003, 100, 15865–15870. [Google Scholar] [CrossRef]
- Ye, J. Reliance of host cholesterol metabolic pathways for the life cycle of hepatitis C virus. PLoS Pathog. 2007, 3, e108. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, M.; Kato, N. Modulation of host metabolism as a target of new antivirals. Adv. Drug Deliv. Rev. 2007, 59, 1277–1289. [Google Scholar] [CrossRef] [PubMed]
- Amemiya, F.; Maekawa, S.; Itakura, Y.; Kanayama, A.; Matsui, A.; Takano, S.; Yamaguchi, T.; Itakura, J.; Kitamura, T.; Inoue, T.; Sakamoto, M.; Yamauchi, K.; Okada, S.; Yamashita, A.; Sakamoto, N.; Itoh, M.; Enomoto, N. Targeting lipid metabolism in the treatment of hepatitis C virus infection. J. Infect. Dis. 2008, 197, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Dreher, T.W.; Miller, W.A. Translational control in positive strand RNA plant viruses. Virology 2006, 344, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Bushell, M.; Sarnow, P. Hijacking the translation apparatus by RNA viruses. J. Cell Biol. 2002, 158, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.; Krijnse-Locker, J. Modification of intracellular membrane structures for virus replication. Nat. Rev. Microbiol. 2008, 6, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Denison, M.R. Seeking membranes: positive-strand RNA virus replication complexes. PLoS Biol. 2008, 6, e270. [Google Scholar] [CrossRef] [PubMed]
- Ahlquist, P.; Noueiry, A.O.; Lee, W.M.; Kushner, D.B.; Dye, B.T. Host factors in positive-strand RNA virus genome replication. J. Virol. 2003, 77, 8181–8186. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, Y.; van Tol, H.; Locker, J.K.; Snijder, E.J. ORF1a-encoded replicase subunits are involved in the membrane association of the arterivirus replication complex. J. Virol. 1998, 72, 6689–6698. [Google Scholar] [PubMed]
- Posthuma, C.C.; Pedersen, K.W.; Lu, Z.; Joosten, R.G.; Roos, N.; Zevenhoven-Dobbe, J.C.; Snijder, E.J. Formation of the arterivirus replication/transcription complex: a key role for nonstructural protein 3 in the remodeling of intracellular membranes. J. Virol. 2008, 82, 4480–4491. [Google Scholar] [CrossRef] [PubMed]
- van der Heijden, M.W.; Carette, J.E.; Reinhoud, P.J.; Haegi, A.; Bol, J.F. Alfalfa mosaic virus replicase proteins P1 and P2 interact and colocalize at the vacuolar membrane. J. Virol. 2001, 75, 1879–1887. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, M.; Chen, J.; Janda, M.; Sullivan, M.; den Boon, J.; Ahlquist, P. A positive-strand RNA virus replication complex parallels form and function of retrovirus capsids. Mol. Cell 2002, 9, 505–514. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, Y.; Snijder, E.J.; Dobbe, J.C.; Schleich, S.; Denison, M.R.; Spaan, W.J.; Locker, J.K. Localization of mouse hepatitis virus nonstructural proteins and RNA synthesis indicates a role for late endosomes in viral replication. J. Virol. 1999, 73, 7641–7657. [Google Scholar] [PubMed]
- Knoops, K.; Kikkert, M.; Worm, S.H.; Zevenhoven-Dobbe, J.C.; van der Meer, Y.; Koster, A.J.; Mommaas, A.M.; Snijder, E.J. SARS-coronavirus replication is supported by a reticulovesicular network of modified endoplasmic reticulum. PLoS Biol. 2008, 6, e226. [Google Scholar] [CrossRef] [PubMed]
- Ulasli, M.; Verheije, M.H.; de Haan, C.A.; Reggiori, F. Qualitative and quantitative ultrastructural analysis of the membrane rearrangements induced by coronavirus. Cell Microbiol. 2010. [Google Scholar]
- Westaway, E.G.; Mackenzie, J.M.; Kenney, M.T.; Jones, M.K.; Khromykh, A.A. Ultrastructure of Kunjin virus-infected cells: colocalization of NS1 and NS3 with double-stranded RNA, and of NS2B with NS3, in virus- induced membrane structures. J. Virol. 1997, 71, 6650–6661. [Google Scholar] [PubMed]
- Welsch, S.; Miller, S.; Romero-Brey, I.; Merz, A.; Bleck, C.K.; Walther, P.; Fuller, S.D.; Antony, C.; Krijnse-Locker, J.; Bartenschlager, R. Composition and three-dimensional architecture of the dengue virus replication and assembly sites. Cell Host Microbe 2009, 5, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Egger, D.; Wolk, B.; Gosert, R.; Bianchi, L.; Blum, H.E.; Moradpour, D.; Bienz, K. Expression of hepatitis C virus proteins induces distinct membrane alterations including a candidate viral replication complex. J. Virol. 2002, 76, 5974–5984. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.J.; Schwartz, M.D.; Ahlquist, P. Flock House virus RNA replicates on outer mitochondrial membranes in Drosophila cells. J. Virol. 2001, 75, 11664–11676. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.J.; Schwartz, M.D.; Dye, B.T.; Ahlquist, P. Engineered retargeting of viral RNA replication complexes to an alternative intracellular membrane. J. Virol. 2003, 77, 12193–12202. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, A.; Giddings, T.H J.; Ladinsky, M.S.; Kirkegaard, K. Cellular origin and ultrastructure of membranes induced during poliovirus infection. J. Virol. 1996, 70, 6576–6588. [Google Scholar] [PubMed]
- Bienz, K.; Egger, D.; Pfister, T.; Troxler, M. Structural and functional characterization of the poliovirus replication complex. J. Virol. 1992, 66, 2740–2747. [Google Scholar] [PubMed]
- Suhy, D.A.; Giddings, T.H. J.; Kirkegaard, K. Remodeling the endoplasmic reticulum by poliovirus infection and by individual viral proteins: an autophagy-like origin for virus-induced vesicles. J. Virol. 2000, 74, 8953–8965. [Google Scholar] [CrossRef] [PubMed]
- Magliano, D.; Marshall, J.A.; Bowden, D.S.; Vardaxis, N.; Meanger, J.; Lee, J. Y. Rubella virus replication complexes are virus-modified lysosomes. Virology 1998, 240, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Kujala, P.; Ikaheimonen, A.; Ehsani, N.; Vihinen, H.; Auvinen, P.; Kaariainen, L. Biogenesis of the Semliki Forest virus RNA replication complex. J. Virol. 2001, 75, 3873–3884. [Google Scholar] [CrossRef] [PubMed]
- Froshauer, S.; Kartenbeck, J.; Helenius, A. Alphavirus RNA replicase is located on the cytoplasmic surface of endosomes and lysosomes. J. Cell Biol. 1988, 107, 2075–2086. [Google Scholar] [CrossRef] [PubMed]
- Weber-Lotfi, F.; Dietrich, A.; Russo, M.; Rubino, L. Mitochondrial targeting and membrane anchoring of a viral replicase in plant and yeast cells. J. Virol. 2002, 76, 10485–10496. [Google Scholar] [CrossRef] [PubMed]
- Turner, K. A.; Sit, T. L.; Callaway, A. S.; Allen, N. S.; Lommel, S. A. Red clover necrotic mosaic virus replication proteins accumulate at the endoplasmic reticulum. Virology 2004, 320, 276–290. [Google Scholar] [CrossRef] [PubMed]
- Burgyan, J.; Rubino, L.; Russo, M. The 5'-terminal region of a tombusvirus genome determines the origin of multivesicular bodies. J. Gen. Virol. 1996, 77, 1967–1974. [Google Scholar] [CrossRef] [PubMed]
- Rubino, L.; Weber-Lotfi, F.; Dietrich, A.; Stussi-Garaud, C.; Russo, M. The open reading frame 1-encoded ('36K') protein of Carnation Italian ringspot virus localizes to mitochondria. J. Gen. Virol. 2001, 82, 29–34. [Google Scholar] [PubMed]
- Prod'homme, D.; Le Panse, S.; Drugeon, G.; Jupin, I. Detection and subcellular localization of the turnip yellow mosaic virus 66K replication protein in infected cells. Virology 2001, 281, 88–101. [Google Scholar] [CrossRef] [PubMed]
- Kopek, B.G.; Perkins, G.; Miller, D.J.; Ellisman, M.H.; Ahlquist, P. Three-dimensional analysis of a viral RNA replication complex reveals a virus-induced mini-organelle. PLoS Biol. 2007, 5, e220. [Google Scholar] [CrossRef] [PubMed]
- Mas, P.; Beachy, R.N. Replication of tobacco mosaic virus on endoplasmic reticulum and role of the cytoskeleton and virus movement protein in intracellular distribution of viral RNA. J. Cell Biol. 1999, 147, 945–958. [Google Scholar] [CrossRef] [PubMed]
- Barton, D.J.; Sawicki, S.G.; Sawicki, D.L. Solubilization and immunoprecipitation of alphavirus replication complexes. J. Virol. 1991, 65, 1496–1506. [Google Scholar] [PubMed]
- Chu, P.W.G.; Westaway, E G. Molecular and ultrastructural analysis of heavy membrane fractions associated with the replication of Kunjin virus RNA. Arch. Virol. 1992, 125, 177–191. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.X.; Kaesberg, P. Synthesis of template-sense, single-strand Flock House virus RNA in a cell-free replication system. Virology 1991, 183, 392–396. [Google Scholar] [CrossRef] [PubMed]
- van Hemert, M.J.; van den Worm, S.H.; Knoops, K.; Mommaas, A.M.; Gorbalenya, A.E.; Snijder, E.J. SARS-coronavirus replication/transcription complexes are membrane-protected and need a host factor for activity in vitro. PLoS Pathog. 2008, 4, e1000054. [Google Scholar] [CrossRef] [PubMed]
- Ahola, T.; Lampio, A.; Auvinen, P.; Kaariainen, L. Semliki Forest virus mRNA capping enzyme requires association with anionic membrane phospholipids for activity. EMBO J. 1999, 18, 3164–3172. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.X.; Ahlquist, P.; Kaesberg, P. Active complete in vitro replication of nodavirus RNA requires glycerophospholipid. Proc. Natl. Acad. Sci. USA 1992, 89, 11136–11140. [Google Scholar] [CrossRef]
- Guinea, R.; Carrasco, L. Phospholipid biosynthesis and poliovirus genome replication, two coupled phenomena. EMBO J. 1990, 9, 2011–2016. [Google Scholar] [PubMed]
- Maynell, L.A.; Kirkegaard, K.; Klymkowsky, M.K. Inhibition of poliovirus RNA synthesis by brefeldin A. J. Virol. 1992, 66, 1985–1994. [Google Scholar] [PubMed]
- Perez, L.; Guinea, R.; Carrasco, L. Synthesis of Semliki Forest virus RNA requires continuous lipid synthesis. Virology 1991, 183, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.M.; Ishikawa, M.; Ahlquist, P. Mutation of host Δ9 fatty acid desaturase inhibits brome mosaic virus RNA replication between template recognition and RNA synthesis. J. Virol. 2001, 75, 2097–2106. [Google Scholar] [CrossRef] [PubMed]
- Kampmueller, K.M.; Miller, D.J. The cellular chaperone heat shock protein 90 facilitates Flock House virus RNA replication in Drosophila cells. J. Virol. 2005, 79, 6827–6837. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hood, B.L.; Chadwick, S.L.; Liu, S.; Watkins, S.C.; Luo, G.; Conrads, T.P.; Wang, T. Fatty acid synthase is up-regulated during hepatitis C virus infection and regulates hepatitis C virus entry and production. Hepatology 2008, 48, 1396–1403. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, S.B.; Chisari, F.V. Hepatitis C virus RNA replication is regulated by host geranylgeranylation and fatty acids. Proc. Natl. Acad. Sci. USA 2005, 102, 2561–2566. [Google Scholar] [CrossRef]
- Rassmann, A.; Henke, A.; Jarasch, N.; Lottspeich, F.; Saluz, H.P.; Munder, T. The human fatty acid synthase: a new therapeutic target for coxsackievirus B3-induced diseases? Antiviral Res. 2007, 76, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Castorena, K.M.; Stapleford, K.A.; Miller, D.J. Complementary transcriptomic, lipidomic, and targeted functional genetic analyses in cultured Drosophila cells highlight the role of glycerophospholipid metabolism in Flock House virus RNA replication. BMC Genomics 2010, 11, 183. [Google Scholar] [CrossRef] [PubMed]
- Cherry, S.; Kunte, A.; Wang, H.; Coyne, C.; Rawson, R.B.; Perrimon, N. COPI activity coupled with fatty acid biosynthesis is required for viral replication. PLoS Pathog. 2006, 2, e102. [Google Scholar] [CrossRef] [PubMed]
- Mackenzie, J.M.; Khromykh, A.A.; Parton, R.G. Cholesterol manipulation by West Nile virus perturbs the cellular immune response. Cell Host Microbe 2007, 2, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Panavas, T.; Serviene, E.; Brasher, J.; Nagy, P.D. Yeast genome-wide screen reveals dissimilar sets of host genes affecting replication of RNA viruses. Proc. Natl. Acad. Sci.U SA 2005, 102, 7326–7331. [Google Scholar] [CrossRef]
- Kushner, D.B.; Lindenbach, B.D.; Grdzelishvili, V. Z.; Noueiry, A.O.; Paul, S.M.; Ahlquist, P. Systematic, genome-wide identification of host genes affecting replication of a positive-strand RNA virus. Proc. Natl. Acad. Sci. USA 2003, 100, 15764–15769. [Google Scholar] [CrossRef]
- Jiang, Y.; Serviene, E.; Gal, J.; Panavas, T.; Nagy, P.D. Identification of essential host factors affecting tombusvirus RNA replication based on the yeast Tet promoters Hughes Collection. J. Virol. 2006, 80, 7394–7404. [Google Scholar] [CrossRef] [PubMed]
- Berger, K.L.; Cooper, J.D.; Heaton, N.S.; Yoon, R.; Oakland, T.E.; Jordan, T. X.; Mateu, G.; Grakoui, A.; Randall, G. Roles for endocytic trafficking and phosphatidylinositol 4-kinase III alpha in hepatitis C virus replication. Proc. Natl. Acad. Sci. USA 2009, 106, 7577–7582. [Google Scholar] [CrossRef]
- Borawski, J.; Troke, P.; Puyang, X.; Gibaja, V.; Zhao, S.; Mickanin, C.; Leighton-Davies, J.; Wilson, C.J.; Myer, V.; Cornellataracido, I.; Baryza, J.; Tallarico, J.; Joberty, G.; Bantscheff, M.; Schirle, M.; Bouwmeester, T.; Mathy, J.E.; Lin, K.; Compton, T.; Labow, M.; Wiedmann, B.; Gaither, L.A. Class III phosphatidylinositol 4-kinase α and β are novel host factor regulators of hepatitis C virus replication. J. Virol. 2009, 83, 10058–10074. [Google Scholar] [CrossRef] [PubMed]
- Tai, A.W.; Benita, Y.; Peng, L.F.; Kim, S.S.; Sakamoto, N.; Xavier, R.J.; Chung, R.T. A functional genomic screen identifies cellular cofactors of hepatitis C virus replication. Cell Host Microbe 2009, 5, 298–307. [Google Scholar] [CrossRef]
- Vaillancourt, F.H.; Pilote, L.; Cartier, M.; Lippens, J.; Liuzzi, M.; Bethell, R. C.; Cordingley, M. G.; Kukolj, G. Identification of a lipid kinase as a host factor involved in hepatitis C virus RNA replication. Virology 2009, 387, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Annamalai, P.; Rofail, F.; Demason, D.A.; Rao, A.L. Replication-coupled packaging mechanism in positive-strand RNA viruses: synchronized coexpression of functional multigenome RNA components of an animal and a plant virus in Nicotiana benthamiana cells by agroinfiltration. J. Virol. 2008, 82, 1484–1495. [Google Scholar] [CrossRef] [PubMed]
- Venter, P.A.; Schneemann, A. Assembly of two independent populations of Flock House virus particles with distinct RNA packaging characteristics in the same cell. J. Virol. 2007, 81, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Nugent, C.I.; Johnson, K.L.; Sarnow, P.; Kirkegaard, K. Functional coupling between replication and packaging of poliovirus replicon RNA. J. Virol. 1999, 73, 427–435. [Google Scholar] [PubMed]
- Den Boon, J.A.; Chen, J.; Ahlquist, P. Identification of sequences in brome mosaic virus replicase protein 1a that mediate association with endoplasmic reticulum membranes. J. Virol. 2001, 75, 12370–12381. [Google Scholar] [CrossRef] [PubMed]
- Miller, D.J.; Ahlquist, P. Flock House virus RNA polymerase is a transmembrane protein with amino-terminal sequences sufficient for mitochondrial localization and membrane insertion. J. Virol. 2002, 76, 9856–9867. [Google Scholar] [CrossRef] [PubMed]
- Schaad, M.C.; Jensen, P.E.; Carrington, J.C. Formation of plant RNA virus replication complexes on membranes: role of an endoplasmic reticulum-targeted viral protein. EMBO J. 1997, 16, 4049–4059. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Mende, J.; Bieck, E.; Hügle, T.; Penin, F.; Rice, C.M.; Blum, H.E.; Moradpour, D. Determinants for membrane association of the hepatitis C virus RNA-dependent RNA polymerase. J. Biol. Chem. 2001, 276, 44052–44063. [Google Scholar] [CrossRef] [PubMed]
- Jonczyk, M.; Pathak, K.B.; Sharma, M.; Nagy, P.D. Exploiting alternative subcellular location for replication: tombusvirus replication switches to the endoplasmic reticulum in the absence of peroxisomes. Virology 2007, 362, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Nishikiori, M.; Dohi, K.; Mori, M.; Meshi, T.; Naito, S.; Ishikawa, M. Membrane-bound tomato mosaic virus replication proteins participate in RNA synthesis and are associated with host proteins in a pattern distinct from those that are not membrane bound. J. Virol. 2006, 80, 8459–8468. [Google Scholar] [CrossRef] [PubMed]
- Pathak, K.B.; Sasvari, Z.; Nagy, P.D. The host Pex19p plays a role in peroxisomal localization of tombusvirus replication proteins. Virology 2008, 379, 294–305. [Google Scholar] [CrossRef] [PubMed]
- Stapleford, K.A.; Rapaport, D.; Miller, D.J. Mitochondrion-enriched anionic phospholipids facilitate Flock House virus RNA polymerase membrane association. J. Virol. 2009, 83, 4498–4507. [Google Scholar] [CrossRef] [PubMed]
- Zinser, E.; Daum, G. Isolation and biochemical characterization of organelles from the yeast, Saccharomyces cerevisiae. Yeast 1995, 11, 493–536. [Google Scholar] [CrossRef] [PubMed]
- van Meer, G.; Voelker, D.R.; Feigenson, G.W. Membrane lipids: where they are and how they behave. Nat. Rev. Mol. Cell. Biol. 2008, 9, 112–124. [Google Scholar] [CrossRef] [PubMed]
- McMahon, H.T.; Gallop, J.L. Membrane curvature and mechanisms of dynamic cell membrane remodeling. Nature 2005, 438, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Elazar, M.; Cheong, K.H.; Liu, P.; Greenberg, H.B.; Rice, C.M.; Glenn, J.S. Amphipathic helix-dependent localization of NS5A mediates hepatitis C virus RNA replication. J. Virol. 2003, 77, 6055–6061. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Westler, W.M.; den Boon, J.A.; Wang, X.; Diaz, A.; Steinberg, H.A.; Ahlquist, P. An amphipathic α-helix controls multiple roles of brome mosaic virus protein 1a in RNA replication complex assembly and function. PLoS Pathog. 2009, 5, e1000351. [Google Scholar] [CrossRef] [PubMed]
- Fuller, N.; Rand, R.P. The influence of lysolipids on the spontaneous curvature and bending elasticity of phospholipid membranes. Biophys. J. 2001, 81, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Szule, J.A.; Fuller, N.L.; Rand, R.P. The effects of acyl chain length and saturation of diacylglycerols and phosphatidylcholines on membrane monolayer curvature. Biophys. J. 2002, 83, 977–984. [Google Scholar] [CrossRef] [PubMed]
- Pichlmair, A.; Reis e Sousa, C. Innate recognition of viruses. Immunity 2007, 27, 370–383. [Google Scholar] [CrossRef] [PubMed]
- Suthar, M.S.; Gale Jr., M.; Owen, D.M. Evasion and disruption of innate immune signalling by hepatitis C and West Nile viruses. Cell Microbiol. 2009, 11, 880–888. [Google Scholar] [CrossRef] [PubMed]
- Bowie, A.G.; Unterholzner, L. Viral evasion and subversion of pattern-recognition receptor signalling. Nat. Rev. Immunol. 2008, 8, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Molla, A.; Paul, A. V.; Wimmer, E. Cell-free, de novo synthesis of poliovirus. Science 1991, 254, 1647–1651. [Google Scholar] [PubMed]
- Komoda, K.; Naito, S.; Ishikawa, M. Replication of plant RNA virus genomes in a cell-free extract of evacuolated plant protoplasts. Proc. Natl. Acad. Sci. USA 2004, 101, 1863–1867. [Google Scholar] [CrossRef]
- Saito, K.; Nishijima, M.; Kuge, O. Phosphatidylserine is involved in gene expression from Sindbis virus subgenomic promoter. Biochem. Biophys. Res. Comm. 2006, 345, 878–885. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Share and Cite
Stapleford, K.A.; Miller, D.J. Role of Cellular Lipids in Positive-Sense RNA Virus Replication Complex Assembly and Function. Viruses 2010, 2, 1055-1068. https://doi.org/10.3390/v2051055
Stapleford KA, Miller DJ. Role of Cellular Lipids in Positive-Sense RNA Virus Replication Complex Assembly and Function. Viruses. 2010; 2(5):1055-1068. https://doi.org/10.3390/v2051055
Chicago/Turabian StyleStapleford, Kenneth A., and David J. Miller. 2010. "Role of Cellular Lipids in Positive-Sense RNA Virus Replication Complex Assembly and Function" Viruses 2, no. 5: 1055-1068. https://doi.org/10.3390/v2051055
APA StyleStapleford, K. A., & Miller, D. J. (2010). Role of Cellular Lipids in Positive-Sense RNA Virus Replication Complex Assembly and Function. Viruses, 2(5), 1055-1068. https://doi.org/10.3390/v2051055




