Development and Clinical Validation of a Skin Test for In Vivo Assessment of SARS-CoV-2 Specific T-Cell Immunity
Abstract
1. Introduction
2. Materials and Methods
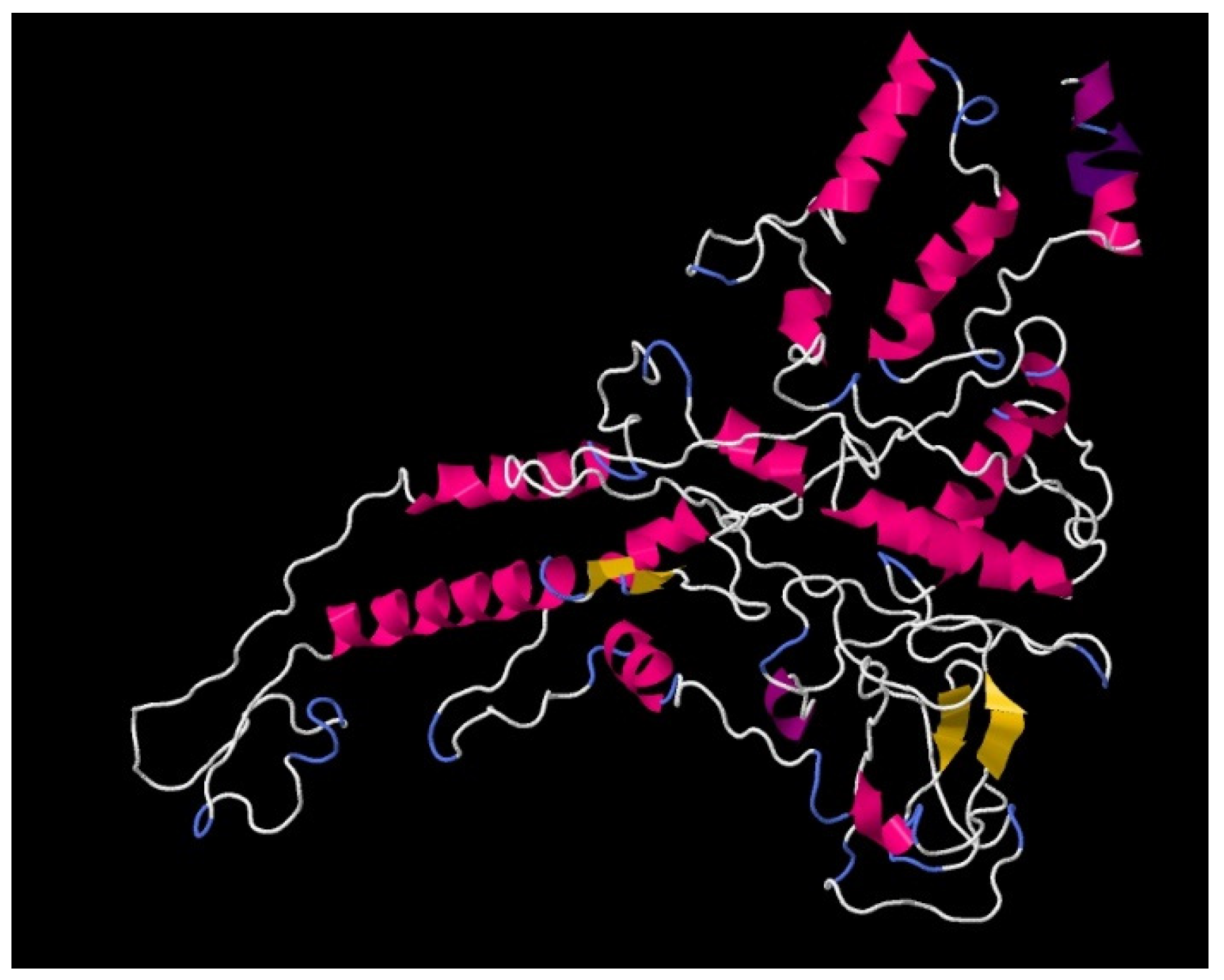
2.1. Development of the Recombinant Antigen Preparation CoronaDerm-PS
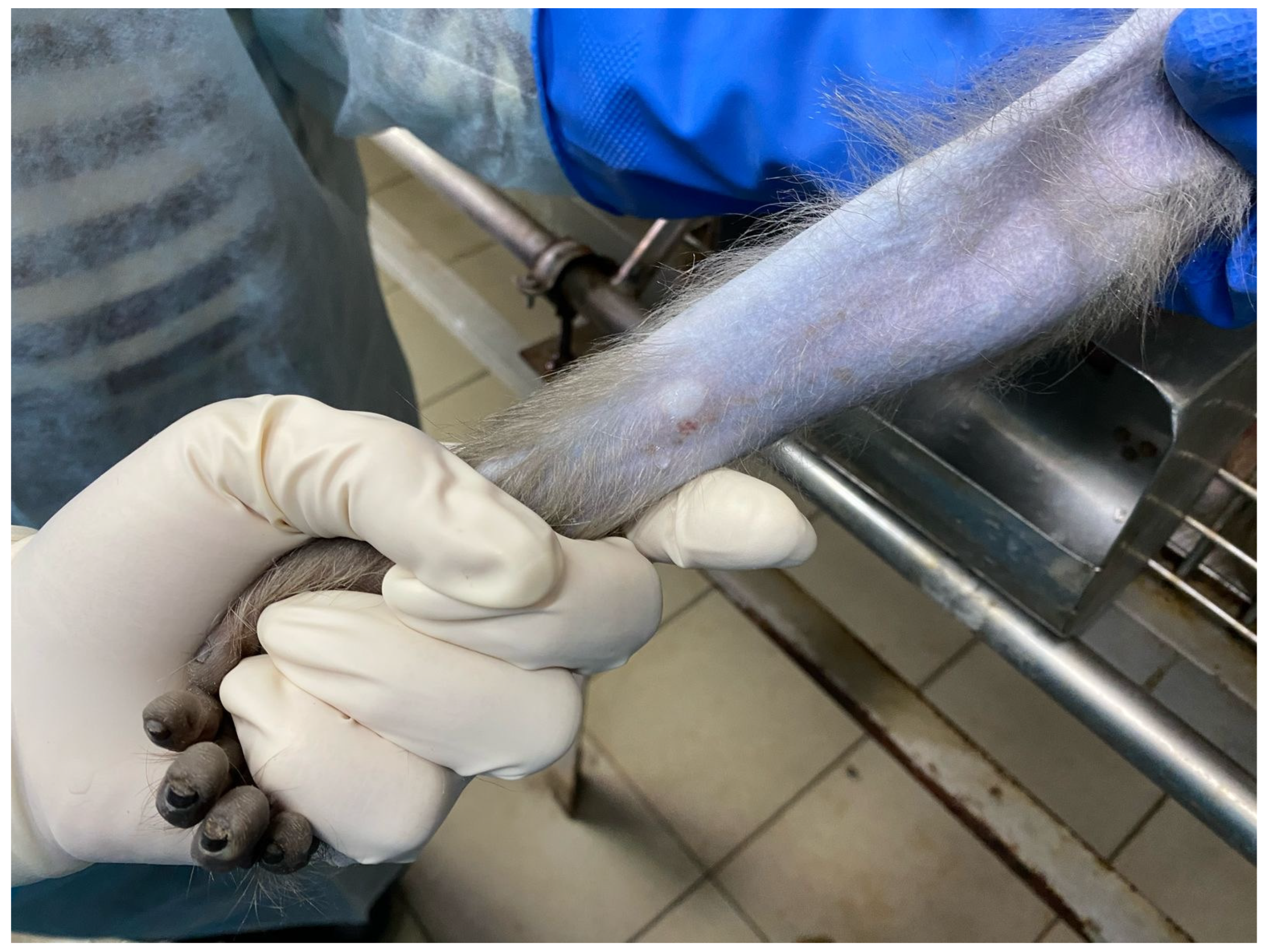
2.2. Preclinical Study Phase in Animals
2.3. Evaluation of Safety and Specific Activity of CoronaDerm-PS in Volunteer Groups
2.3.1. Demographic and Clinical Characteristics of Volunteer Groups
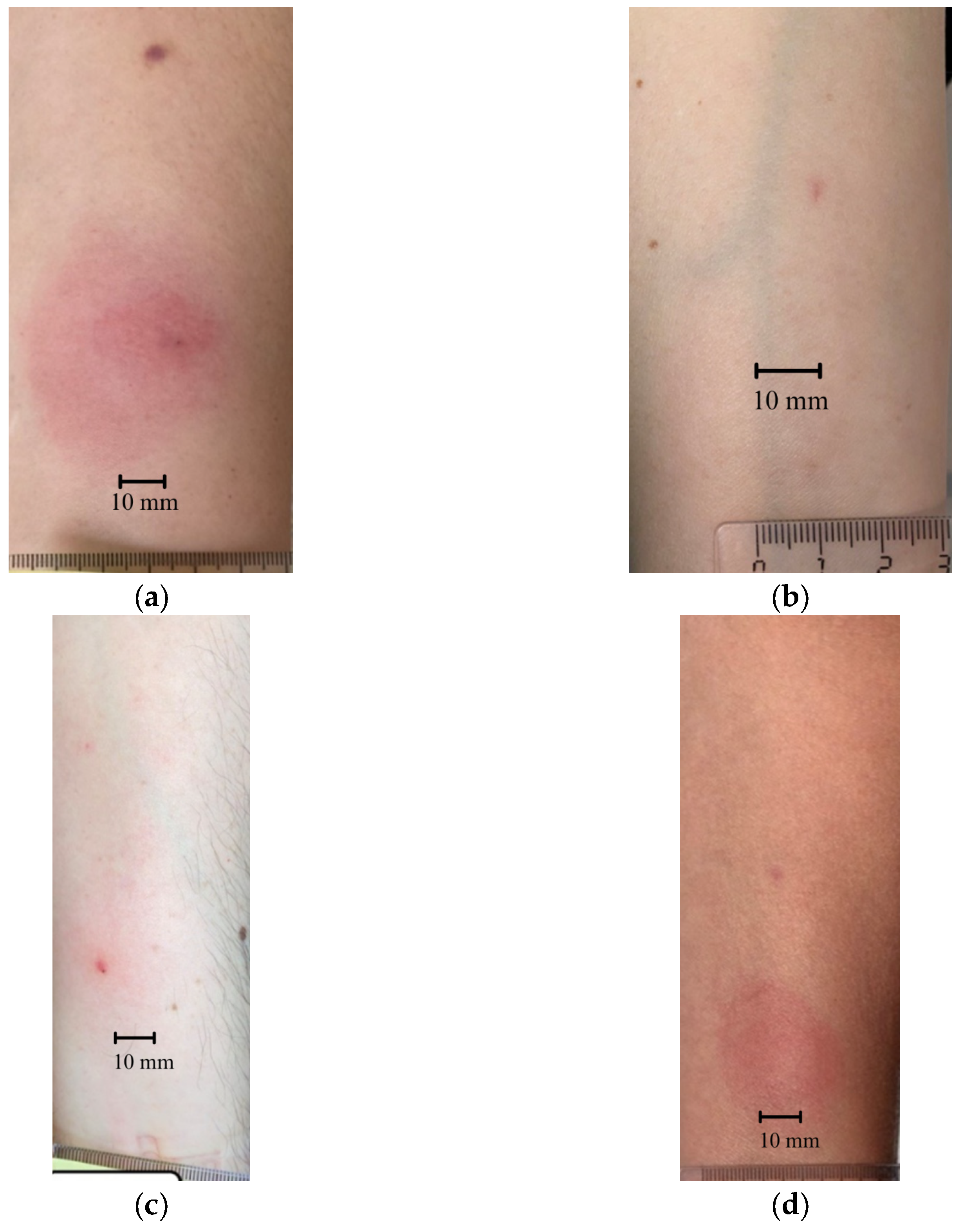
2.3.2. Skin Test Assessment and Interpretation Criteria
2.3.3. Clinical and Laboratory Investigations
3. Results
3.1. Finalized Recombinant Antigen
3.2. Preclinical Study Results with CoronaDerm-PS in Animals
3.3. Integrated Analysis Results of CoronaDerm-PS Safety and Specific Activity in Volunteer Groups
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix B
| Adverse Effect | n | Proportion of AEs | % of Administrations with AE |
|---|---|---|---|
| Itching at injection site | 87 | 18.20% | 30.10% |
| Pain at injection site | 63 | 13.18% | 21.80% |
| Weakness | 52 | 10.88% | 17.99% |
| Headache | 45 | 9.41% | 15.57% |
| Increased body temperature | 27 | 5.65% | 9.34% |
| Swelling at injection site | 25 | 5.23% | 8.65% |
| Sweating | 25 | 5.23% | 8.65% |
| Dizziness | 24 | 5.02% | 8.30% |
| Insomnia | 18 | 3.77% | 6.23% |
| Increased blood pressure | 8 | 1.67% | 2.77% |
| Abdominal pain | 8 | 1.67% | 2.77% |
| Chest pain | 7 | 1.46% | 2.42% |
| Arthralgia | 6 | 1.26% | 2.08% |
| Lymph node enlargement | 5 | 1.05% | 1.73% |
| Palpitations | 5 | 1.05% | 1.73% |
| Urobilinuria | 4 | 0.84% | 1.38% |
| Elevated ESR | 4 | 0.84% | 1.38% |
| Shortness of breath | 4 | 0.84% | 1.38% |
| Decreased appetite | 4 | 0.84% | 1.38% |
| Sleepiness | 4 | 0.84% | 1.38% |
| Bruising at injection site | 3 | 0.63% | 1.04% |
| Elevated C-reactive protein | 3 | 0.63% | 1.04% |
| Tenderness at injection site | 2 | 0.42% | 0.69% |
| Burning at injection site | 2 | 0.42% | 0.69% |
| Itching at placebo injection site | 2 | 0.42% | 0.69% |
| Decreased blood pressure | 2 | 0.42% | 0.69% |
| Rhinitis | 2 | 0.42% | 0.69% |
| Acute respiratory viral infection | 2 | 0.42% | 0.69% |
| Painful menstruation | 1 | 0.21% | 0.35% |
| Eye pain | 1 | 0.21% | 0.35% |
| Sore throat | 1 | 0.21% | 0.35% |
| Back pain | 1 | 0.21% | 0.35% |
| Bradycardia | 1 | 0.21% | 0.35% |
| Ear congestion | 1 | 0.21% | 0.35% |
| Stool changes | 1 | 0.21% | 0.35% |
| Upper respiratory tract catarrhal symptoms | 1 | 0.21% | 0.35% |
| Conjunctivitis | 1 | 0.21% | 0.35% |
| Urticaria | 1 | 0.21% | 0.35% |
| Body aches | 1 | 0.21% | 0.35% |
| Flatulence | 1 | 0.21% | 0.35% |
| Migraine | 1 | 0.21% | 0.35% |
| Nosebleed | 1 | 0.21% | 0.35% |
| Exertional dyspnea | 1 | 0.21% | 0.35% |
| Localized acute urticaria | 1 | 0.21% | 0.35% |
| Feeling of “heat” in the body | 1 | 0.21% | 0.35% |
| Warmth at injection site | 1 | 0.21% | 0.35% |
| Heaviness in occipital region | 1 | 0.21% | 0.35% |
| Elevated immunoglobulin E (IgE) | 1 | 0.21% | 0.35% |
| Increased salivation | 1 | 0.21% | 0.35% |
| Lymphangitis | 1 | 0.21% | 0.35% |
| Eye tearing | 1 | 0.21% | 0.35% |
| Spontaneous urticaria of unclear origin | 1 | 0.21% | 0.35% |
| Rash on shoulders and forearms | 1 | 0.21% | 0.35% |
| Prolonged menstruation | 1 | 0.21% | 0.35% |
| Chronic recurrent urticaria | 1 | 0.21% | 0.35% |
| Toothache | 1 | 0.21% | 0.35% |
| Dry mouth | 1 | 0.21% | 0.35% |
| Pain in right wrist joint | 1 | 0.21% | 0.35% |
| Tonsillitis | 1 | 0.21% | 0.35% |
| Hemorrhoidal thrombosis | 1 | 0.21% | 0.35% |
| Numbness at injection site | 1 | 0.21% | 0.35% |
| Tenderness on palpation at injection site | 1 | 0.21% | 0.35% |
| Sneezing | 1 | 0.21% | 0.35% |
References
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020, 395, 565–574. [Google Scholar] [CrossRef]
- Yang, L.; Liu, S.; Liu, J.; Zhang, Z.; Wan, X.; Huang, B.; Chen, Y.; Zhang, Y. COVID-19: Immunopathogenesis and Immunotherapeutics. Signal Transduct. Target. Ther. 2020, 5, 128. [Google Scholar] [CrossRef]
- Xu, X.; Chen, P.; Wang, J.; Feng, J.; Zhou, H.; Li, X.; Zhong, W.; Hao, P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020, 63, 457–460. [Google Scholar] [CrossRef]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef]
- Gordon, D.E.; Jang, G.M.; Bouhaddou, M.; Xu, J.; Obernier, K.; White, K.M.; O’Meara, M.J.; Rezelj, V.V.; Guo, J.Z.; Swaney, D.L.; et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature 2020, 583, 459–468. [Google Scholar] [CrossRef]
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2022, 23, 3–20. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Zhou, R.; To, K.K.; Wong, Y.C.; Liu, L.; Zhou, B.; Li, X.; Huang, H.; Mo, Y.; Luk, T.Y.; Lau, T.T.; et al. Acute SARS-CoV-2 Infection Impairs Dendritic Cell and T Cell Responses. Immunity 2020, 53, 864–877.e5. [Google Scholar] [CrossRef]
- Domingo, P.; Mur, I.; Pomar, V.; Corominas, H.; Casademont, J.; de Benito, N. The four horsemen of a viral Apocalypse: The pathogenesis of SARS-CoV-2 infection (COVID-19). EBioMedicine 2020, 58, 102887. [Google Scholar] [CrossRef]
- Marsh, S.G.E. The HLA FactsBook; Academic Press: Amsterdam, The Netherlands, 2000; 564p. [Google Scholar]
- Grifoni, A.; Weiskopf, D.; Ramirez, S.I.; Mateus, J.; Dan, J.M.; Moderbacher, C.R.; Rawlings, S.A.; Sutherland, A.; Premkumar, L.; Jadi, R.S.; et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell 2020, 181, 1489–1501.e15. [Google Scholar] [CrossRef]
- Le Bert, N.; Tan, A.T.; Kunasegaran, K.; Tham, C.Y.L.; Hafezi, M.; Chia, A.; Chng, M.H.Y.; Lin, M.; Tan, N.; Linster, M.; et al. SARS-CoV-2-specific T cell immunity in cases of COVID-19 and SARS, and uninfected controls. Nature 2020, 584, 457–462. [Google Scholar] [CrossRef]
- Peng, Y.; Mentzer, A.J.; Liu, G.; Yao, X.; Yin, Z.; Dong, D.; Dejnirattisai, W.; Rostron, T.; Supasa, P.; Liu, C.; et al. Broad and strong memory CD4+ and CD8+ T cells induced by SARS-CoV-2 in UK convalescent individuals following COVID-19. Nat. Immunol. 2020, 21, 1336–1345. [Google Scholar] [CrossRef]
- Tan, A.T.; Linster, M.; Tan, C.W.; Le Bert, N.; Chia, W.N.; Kunasegaran, K.; Zhuang, Y.; Tham, C.Y.L.; Chia, A.; Smith, G.J.D.; et al. Early induction of functional SARS-CoV-2-specific T cells associates with rapid viral clearance and mild disease in COVID-19 patients. Cell Rep. 2021, 34, 108728. [Google Scholar] [CrossRef]
- Lucas, C.; Klein, J.; Sundaram, M.E.; Liu, F.; Wong, P.; Silva, J.; Mao, T.; Oh, J.E.; Mohanty, S.; Huang, J.; et al. Delayed production of neutralizing antibodies correlates with fatal COVID-19. Nat. Med. 2021, 27, 1178–1186. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef]
- Tavakolpour, S.; Rakhshandehroo, T.; Wei, E.X.; Rashidian, M. Lymphopenia during the COVID-19 infection: What it shows and what can be learned. Immunol. Lett. 2020, 225, 31–32. [Google Scholar] [CrossRef]
- Diao, B.; Wang, C.; Tan, Y.; Chen, X.; Liu, Y.; Ning, L.; Chen, L.; Li, M.; Liu, Y.; Wang, G.; et al. Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19). Front. Immunol. 2020, 11, 827. [Google Scholar] [CrossRef]
- Shouman, S.; El-Kholy, N.; Hussien, A.E.; El-Derby, A.M.; Magdy, S.; Abou-Shanab, A.M.; Elmehrath, A.O.; Abdelwaly, A.; Helal, M.; El-Badri, N. SARS-CoV-2-associated lymphopenia: Possible mechanisms and the role of CD147. Cell Commun. Signal. 2024, 22, 349. [Google Scholar] [CrossRef]
- MacLeod, M.K.; Clambey, E.T.; Kappler, J.W.; Marrack, P. CD4 memory T cells: What are they and what can they do? Semin. Immunol. 2009, 21, 53–61. [Google Scholar] [CrossRef]
- Iyer, S.S.; Latner, D.R.; Zilliox, M.J.; McCausland, M.; Akondy, R.S.; Penaloza-Macmaster, P.; Hale, J.S.; Ye, L.; Mohammed, A.U.; Yamaguchi, T.; et al. Identification of novel markers for mouse CD4(+) T follicular helper cells. Eur. J. Immunol. 2013, 43, 3219–3232. [Google Scholar] [CrossRef]
- Hale, J.S.; Ahmed, R. Memory T follicular helper CD4 T cells. Front. Immunol. 2015, 6, 16. [Google Scholar] [CrossRef]
- Baumjohann, D.; Okada, T.; Ansel, K.M. Cutting Edge: Distinct waves of BCL6 expression during T follicular helper cell development. J. Immunol. 2011, 187, 2089–2092. [Google Scholar] [CrossRef]
- Schulien, I.; Kemming, J.; Oberhardt, V.; Wild, K.; Seidel, L.M.; Killmer, S.; Sagar; Daul, F.; Salvat Lago, M.; Decker, A.; et al. Characterization of pre-existing and induced SARS-CoV-2-specific CD8+ T cells. Nat. Med. 2021, 27, 78–85. [Google Scholar] [CrossRef]
- Zuo, J.; Dowell, A.C.; Pearce, H.; Verma, K.; Long, H.M.; Begum, J.; Aiano, F.; Amin-Chowdhury, Z.; Hoschler, K.; Brooks, T.; et al. Robust SARS-CoV-2-specific T cell immunity is maintained at 6 months following primary infection. Nat. Immunol. 2021, 22, 620–626. [Google Scholar] [CrossRef]
- Nelde, A.; Bilich, T.; Heitmann, J.S.; Maringer, Y.; Salih, H.R.; Roerden, M.; Lübke, M.; Bauer, J.; Rieth, J.; Wacker, M.; et al. SARS-CoV-2-derived peptides define heterologous and COVID-19-induced T cell recognition. Nat. Immunol. 2021, 22, 74–85. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Faliti, C.E.; Ramirez, S.I.; Frazier, A.; Yu, E.D.; Grifoni, A.; Rawlings, S.A.; et al. Immunological memory to SARS-CoV-2 assessed for up to eight months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Rydyznski Moderbacher, C.; Ramirez, S.I.; Dan, J.M.; Grifoni, A.; Hastie, K.M.; Weiskopf, D.; Belanger, S.; Abbott, R.K.; Kim, C.; Choi, J.; et al. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell 2020, 183, 996–1012.e19. [Google Scholar] [CrossRef]
- Gudbjartsson, D.F.; Norddahl, G.L.; Melsted, P.; Gunnarsdottir, K.; Holm, H.; Eythorsson, E.; Arnthorsson, A.O.; Helgason, D.; Bjarnadottir, K.; Ingvarsson, R.F.; et al. Humoral Immune Response to SARS-CoV-2 in Iceland. N. Engl. J. Med. 2020, 383, 1724–1734. [Google Scholar] [CrossRef]
- Lipsitch, M.; Grad, Y.H.; Sette, A.; Crotty, S. Cross-reactive memory T cells and herd immunity to SARS-CoV-2. Nat. Rev. Immunol. 2020, 20, 709–713. [Google Scholar] [CrossRef]
- Villemonteix, J.; Cohen, L.; Guihot, A.; Guérin, V.; Moulin, C.; Caseris, M.; Carol, A.; Bonacorsi, S.; Carcelain, G. Comparison between enzyme-linked immunospot assay and intracellular cytokine flow cytometry assays for the evaluation of T cell response to SARS-CoV-2 after symptomatic COVID-19. Immun. Inflamm. Dis. 2022, 10, e617. [Google Scholar] [CrossRef]
- Aleksova, M.; Todorova, Y.; Emilova, R.; Baymakova, M.; Yancheva, N.; Andonova, R.; Zasheva, A.; Grifoni, A.; Weiskopf, D.; Sette, A.; et al. Virus-Specific Stem Cell Memory CD8+ T Cells May Indicate a Long-Term Protection against Evolving SARS-CoV-2. Diagnostics 2023, 13, 1280. [Google Scholar] [CrossRef]
- Tormo, N.; Giménez, E.; Martínez-Navarro, M.; Albert, E.; Navalpotro, D.; Torres, I.; Gimeno, C.; Navarro, D. Performance comparison of a flow cytometry immunoassay for intracellular cytokine staining and the QuantiFERON SARS-CoV-2 test for detection and quantification of SARS-CoV-2-Spike-reactive-IFN-γ-producing T cells after COVID-19 vaccination. Eur. J. Clin. Microbiol. Infect. Dis. 2022, 41, 657–662. [Google Scholar] [CrossRef]
- Ning, B.; Chandra, S.; Rosen, J.; Multala, E.; Argrave, M.; Pierson, L.; Trinh, I.; Simone, B.; Escarra, M.D.; Drury, S.; et al. Evaluation of SARS-CoV-2-Specific T-Cell Activation with a Rapid On-Chip IGRA. ACS Nano 2023, 17, 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Black, C.A. Delayed type hypersensitivity: Current theories with an historic perspective. Dermatol. Online J. 1999, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Kalish, R.S.; Askenase, P.W. Molecular mechanisms of CD8+ T cell-mediated delayed hypersensitivity: Implications for allergies, asthma, and autoimmunity. J. Allergy Clin. Immunol. 1999, 103, 192–199. [Google Scholar] [CrossRef]
- Huebner, R.E.; Schein, M.F.; Bass, J.B. The tuberculin skin test. Clin. Infect. Dis. 1993, 17, 968–975. [Google Scholar] [CrossRef]
- Barrios, Y.; Franco, A.; Sánchez-Machín, I.; Poza-Guedes, P.; González-Pérez, R.; Matheu, V. The Beauty of Simplicity: Delayed-Type Hypersensitivity Reaction to Measure Cellular Immune Responses in RNA-SARS-Cov-2 Vaccinated Individuals. Vaccines 2021, 9, 575. [Google Scholar] [CrossRef]
- Barrios, Y.; Alava-Cruz, C.; Franco, A.; Matheu, V. Long Term Cell Immune Response to COVID-19 Vaccines Assessment Using a Delayed-Type Hypersensitivity (DTH) Cutaneous Test. Diagnostics 2022, 12, 1421. [Google Scholar] [CrossRef]
- Barrios, Y.; Alava-Cruz, C.; Marrero-Miranda, D.; Matheu, V. Early riser specific immune cell response by delayed-type hypersensitivity in a kidney transplant patient vaccinated against COVID-19. BMJ Case Rep. 2022, 15, e250509. [Google Scholar] [CrossRef]
- Barrios, Y.; Franco, A.; Sanchez-Machin, I.; Poza-Guedes, P.; Gonzalez-Perez, R.; Matheu, V. A novel application of delayed-type hypersensitivity reaction to measure cellular immune response in SARS-CoV-2 exposed individuals. Clin. Immunol. 2021, 226, 108730. [Google Scholar] [CrossRef]
- Barrios, Y.; Franco, A.; Alava-Cruz, C.; Cuesta-Martin, R.; Camara, C.; Matheu, V. Easy approach to detect cell immunity to COVID vaccines in common variable immunodeficiency patients. Allergol. Immunopathol. 2022, 50, 101–105. [Google Scholar] [CrossRef]
- Kopat, V.V.; Riabchenkova, A.A.; Chirak, E.L.; Chirak, E.R.; Saenko, A.I.; Kudryavtsev, I.V.; Trulioff, A.S.; Savin, T.V.; Zueva, E.V.; Simbirtsev, A.S.; et al. Purification technology design, biochemical and immunological characteristics of the recombinant chimeric antigen for evaluation of T cell immunity against coronavirus infection. Med. Immunol. 2024, 26, 591–606. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.L.; Wang, X.G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.R.; Zhu, Y.; Li, B.; Huang, C.L.; et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Sidney, J.; Sette, A.; Peters, B. TepiTool: A Pipeline for Computational Prediction of T Cell Epitope Candidates. Curr. Protoc. Immunol. 2016, 114, 18.19.1–18.19.24. [Google Scholar] [CrossRef] [PubMed]
- Reynisson, B.; Barra, C.; Kaabinejadian, S.; Hildebrand, W.H.; Peters, B.; Nielsen, M. Improved Prediction of MHC II Antigen Presentation through Integration and Motif Deconvolution of Mass Spectrometry MHC Eluted Ligand Data. J. Proteome Res. 2020, 19, 2304–2315. [Google Scholar] [CrossRef]
- Sette, A.; Sidney, J. Nine major HLA class I supertypes account for the vast preponderance of HLA-A and -B polymorphism. Immunogenetics 1999, 50, 201–212. [Google Scholar] [CrossRef]
- Kopat, V.V.; Riabchenkova, A.A.; Chirak, E.L.; Chirak, E.R.; Saenko, A.I.; Kolmakov, N.N.; Simbirtsev, A.S.; Dukhovlinov, I.V.; Totolian, A.A. Designing structure and E. coli strain-producer bearing SARS-CoV-2 N, S, M, E protein-related sequence antigen. Russ. J. Infect. Immun. 2023, 13, 653–662. [Google Scholar] [CrossRef]
- Springer, I.; Besser, H.; Tickotsky-Moskovitz, N.; Dvorkin, S.; Louzoun, Y. Prediction of Specific TCR-Peptide Binding From Large Dictionaries of TCR-Peptide Pairs. Front. Immunol. 2020, 11, 1803. [Google Scholar] [CrossRef]
- Ramachandran, G.N.; Ramakrishnan, C.; Sasisekharan, V. Stereochemistry of polypeptide chain configurations. J. Mol. Biol. 1963, 7, 95–99. [Google Scholar] [CrossRef]
- Walker, J.M. (Ed.) The Proteomics Protocols Handbook; Humana Press: New York, NY, USA, 2005. [Google Scholar]
- Kozlowski, L.P. IPC—Isoelectric Point Calculator. Biol. Direct 2016, 11, 55. [Google Scholar] [CrossRef]
- Boratyn, G.M.; Thierry-Mieg, J.; Thierry-Mieg, D.; Busby, B.; Madden, T.L. Magic-BLAST, an accurate RNA-seq aligner for long and short reads. BMC Bioinform. 2019, 20, 405. [Google Scholar] [CrossRef]
- Chang, C.K.; Hou, M.H.; Chang, C.F.; Hsiao, C.D.; Huang, T.H. The SARS coronavirus nucleocapsid protein—Forms and functions. Antivir. Res. 2014, 103, 39–50. [Google Scholar] [CrossRef]
- Savin, T.V.; Kopat, V.V.; Riabchenkova, A.A.; Chirak, E.L.; Chirak, E.R.; Saenko, A.I.; Dukhovlinov, I.V.; Sysoeva, G.M.; Gamaley, S.G.; Shimina, G.G.; et al. Experimentally investigated “CoronaDerm-PS”-driven SARS-CoV-2-specific cellular immunity and safety. Russ. J. Infect. Immun. 2024, 14, 238–250. [Google Scholar] [CrossRef]
- Savin, T.V.; Milichkina, A.M.; Krasnov, A.A.; Kuznetsova, R.N.; Shchederkina, E.E.; Svarval, A.V.; Sharova, A.A.; Reingardt, D.E.; Ostankova, Y.V.; Gubanova, A.V.; et al. Safety and specific activity of the recombinant SARS-CoV-2 allergen (“CoronaDerm-PS”) based on phase I–II clinical trial results. Russ. J. Infect. Immun. 2024, 14, 900–916. [Google Scholar] [CrossRef]
- Ding, X.; Du, W.; Liu, Q.; Tao, L.; Shao, Y.; Lu, P.; Yang, H.; Teng, X.; Chen, C.; Li, Z.; et al. Accuracy of ESAT6-CFP10 skin test compared with tuberculin skin test in a healthy population: A randomized, blind, parallel controlled phase III clinical study. BMC Infect. Dis. 2024, 24, 1479. [Google Scholar] [CrossRef]
- Dukhovlinov, I.V.; Simbirtsev, A.S.; Totolian Areg, A. Method for Assessing Cellular Immune Response Against Coronavirus Infection. Eurasian Patent No. 047119, 3 June 2024. [Google Scholar]

| Experiment | Animals |
|---|---|
| Evaluation of specific activity, CoronaDerm-PS | guinea pigs (*), 46 males and 46 females, weighing 350–400 g, aged 6–7 weeks (n = 92) white-sided guinea pigs (**), 14 males and 15 females, weighing 350–400 g, aged 7–8 weeks (n = 29) |
| Assessment of “acute” toxicity, CoronaDerm-PS | outbred CD-1 mice (***), 40 males and 40 females weighing 25–30 g, aged 8 weeks (n = 80) shorthaired guinea pigs of motley color (***),40 males and 40 females, weighing 650–750 g, aged 12–14 weeks (n = 80) |
| Assessment of chronic toxicity, CoronaDerm-PS | outbred CD-1 mice (***), 40 males and 40 females, weighing 25–30 g, aged 8 weeks (n = 80) shorthaired guinea pigs of motley color (***),20 males and 20 females, weighing 650–750 g, aged 12–14 weeks (n = 40) |
| Assessment of local irritant action, CoronaDerm-PS | outbred CD-1 mice (***), 80 males and 80 females, weighing 25–30 g, aged 8 weeks (n = 160) shorthaired guinea pigs of motley color (***),60 males and 60 females, weighing 650–750 g, aged 12–14 weeks (n = 120) |
| Assessment of immunological safety, CoronaDerm-PS | BALB/c mice (****), female, weighing 18–20 g, aged 8 weeks (n = 130) |
| Assessment of allergenic properties, CoronaDerm-PS | guinea pigs (****), 16 males and 16 females, weighing 350–400 g, aged 6-7 weeks (n = 32) |
| Evaluation of mutagenic potential by chromosomal aberration test, CoronaDerm-PS | C57Bl/6 mice (****), males, weighing 22–24 g, aged 9–10 weeks (n = 35) |
| Evaluation of reproductive system effects, CoronaDerm-PS | conducted as part of the study of acute and chronic toxicity in guinea pigs |
| Component | Concentration |
|---|---|
| Active Ingredient | |
| Recombinant fusion protein containing SARS-CoV-2 structural proteins (partial) | 10.0 ug |
| Inactive Ingredients | |
| polysorbate 20 | 2 uL (1%) |
| Tris-HCl | 0.605 mg (25 mM) |
| sodium chloride | 1.8 mg (0.9%) |
| phenol | 0.3 mg (0.15%) |
| water for injection | to 0.2 mL |
| pH | 7.4 |
| Analyses | Group 1 | Group 2 | Group 3 | Group 4 | ||||
|---|---|---|---|---|---|---|---|---|
| Group 2a | Group 2b | Group 2c | Group 3a | Group 3b | Group 3c | |||
| receipt of written informed consent | X | X | X | X | X | X | X | X |
| collection and registration of medical history | X | X | X | X | X | X | X | X |
| physical examination | X | X | X | X | X | X | X | X |
| vital sign assessment (BP, HR, RR, temperature) | X | X | X | X | X | X | X | X |
| blood analysis (biochemical, clinical, coagulogram, total IgE) | X | X | X | X | X | X | ||
| serological analysis (HIV, hepatitis B/C, syphilis) | X | X | X | X | X | X | ||
| general urine analysis | X | X | X | X | X | X | ||
| anti-SARS-CoV-2 IgG determination (ELISA) | X | X | X | X | X | X | X | X |
| analysis of nasopharyngeal, oropharyngeal swabs for SARS-CoV-2 RNA (PCR) | X | X | X | X | X | X | ||
| evaluation of T-cell immunity by flow cytometry (ex vivo) | X | X | X | X | X | X | ||
| lung fluorography | X | X | X | X | X | X | ||
| pregnancy test | X | X | X | X | X | X | X | X |
| ECG | X | X | X | X | X | X | ||
| CoronaDerm-PS injection | X | X | X | X | X | X | X | X |
| adverse event assessment | X | X | X | X | X | X | X | X |
| Indicator | Experimental Animal Group | |
|---|---|---|
| Control Saline Solution (n = 8) | CoronaDerm-PS 10 μg/0.2 mL/Mouse (n = 8) | |
| One day after administration | ||
| mouse mass, g | 19.4 ± 0.3 | 20.2 ± 0.4 |
| splenic mass index, ×10, mg/g | 100.6 ± 3.7 | 105.1 ± 4.9 |
| bsolute number of nucleated splenocytes, ×106/organ | 180.6 ± 18.0 | 178.8 ± 10.0 |
| thymic weight index, ×10, mg/g | 30.3 ± 1.9 | 31.5 ± 1.5 |
| absolute thymocyte count, ×106/organ | 80.7 ± 7.0 | 75.1 ± 6.8 |
| Ab producing cells/spleen | 15,081.6 ± 774.0 | 14,145.9 ± 1795.2 |
| hemagglutinin titer, log2 | 8.0 ± 0 | 8.0 ± 0 |
| Twenty-one days after administration | ||
| mouse mass, g | 22.0 ± 0.4 | 23.2 ± 0.3 * |
| splenic mass index, ×10, mg/g | 88.3 ± 3.0 | 98.7 ± 3.7 * |
| absolute number of nucleated splenocytes, ×106/organ | 333.6 ± 19.7 | 333.0 ± 16.3 * |
| thymic weight index, ×10, mg/g | 27.5 ± 1.9 | 28.6 ± 2.0 |
| absolute thymocyte count, ×106/organ | 85.4 ± 7.6 | 97.0 ± 3.0 * |
| Ab producing cells/spleen | 22,128.5 ± 1957.8 | 22,847.3 ± 1688.9 |
| hemagglutinin titer, log2 | 8.0 ± 0 | 8.0 ± 0 |
| Experimental Conditions (Frequency of Administration, Preparation, Dose) | Cells Examined | Number of Aberrations | Damaged Cells, % | |||
|---|---|---|---|---|---|---|
| Single Fragments, % | Paired Fragments, % | Exchanges, % | Multiple Aberrations, % | |||
| CoronaDerm-PS 10 ug protein/0.2 mL/mouse, once, males | 500 | 0.4 | 0 | 0 | 0 | 0.4 |
| 0.9% NaCl 0.2 mL/mouse, once, males | 500 | 1.0 | 0 | 0 | 0 | 1.0 |
| CoronaDerm-PS 10 ug protein/0.2 mL/mouse, 4 times, males | 500 | 0.8 | 0.2 | 0 | 0 | 1.0 |
| 0.9% NaCl 0.2 mL/mouse, 4 times, males | 500 | 1.2 | 0 | 0 | 0 | 1.2 |
| CoronaDerm-PS 10 ug protein/0.2 mL/mouse, 4 times, females | 500 | 0.8 | 0 | 0 | 0 | 0.8 |
| 0.9% NaCl 0.2 mL/mouse, 4 times, females | 500 | 1.4 | 0 | 0 | 0 | 1.4 |
| Cyclophosphamide 20 mg/kg, 0.2 mL/mouse, once, males | 500 | 13.8 | 3 | 0.2 | 8.0 | 25.0 |
| Group | Animal Mass (g) | Ovarian Mass (g) | Mass Coefficient (%) | ||
|---|---|---|---|---|---|
| Day 1 | control (n = 5) | 658.20 ± 53.88 | 0.11 ±0.01 | 0.02 ± 0.00 | |
| 1.0 mL/animal (n = 5) | 621.60 ± 19.76 | 0.11 ± 0.00 | 0.02 ± 0.00 | ||
| Day 7 | control (n = 5) | 635.00 ± 20.02 | 0.07 ± 0.01 | 0.01 ± 0.00 | |
| 1.0 mL/animal (n = 5) | 652.80 ± 21.70 | 0.09 ± 0.01 | 0.01 ± 0.00 | ||
| Group | Positive (Volunteers) | Inconclusive (Volunteers) | Negative (Volunteers) |
|---|---|---|---|
| Group 1 (no history of illness or vaccination), phase I | 0 | 0 † | 20 |
| Group 2a (EpiVacCorona) | 61 | 0 † | 18 |
| Group 2b (Gam-COVID-Vac) | 67 | 0 † | 15 |
| Group 2c (CoviVac) | 21 | 0 † | 4 |
| Group 3a (Wuhan strain and Alpha variant) | 27 | 5 | 1 |
| Group 3b (Delta variant) | 63 | 0 † | 17 |
| Group 3c (Omicron subvariants) | 40 | 7 | 5 |
| Group 4 (no history of illness or vaccination), phases I, II | 3 | 0 † | 20 |
| Group | AUC (95% CI) | SE | p | Sensitivity (95% CI) | Specificity (95% CI) |
|---|---|---|---|---|---|
| Group 2a (EpiVacCorona) | 0.782 (0.678–0.887) | 0.053 | <0.001 | 76.60% (95% CI: 67.36–83.85%) | 80.00% (95% CI: 71.07–86.69%) |
| Group 2b (Gam-COVID-Vac) | 0.843 (0.751–0.935) | 0.047 | <0.001 | 81.70% (95%CI: 73.09–88.01%) | 87.00% (95% CI: 79.11–92.20%) |
| Group 2c (CoviVac) | 0.87 (0.764–0.975) | 0.054 | <0.001 | 84.00% (95% CI: 70.70–91.95%) | 87.00% (95% CI: 74.22–93.96%) |
| Group 3a (Wuhan strain and Alpha genetic variant) | 0.844 (0.733–0.955) | 0.057 | <0.001 | 81.80% (95% CI: 69.39–89.91%) | 86.95% (95% CI: 75.35–93.56%) |
| Group 3b (Delta variant) | 0.844 (0.760–0.928) | 0.043 | <0.001 | 79.70% (95% CI: 70.79–86.42%) | 87.50% (95% CI: 79.60–92.62%) |
| Group 3c (Omicron subvariants) | 0.799 (0.689–0.909) | 0.056 | <0.001 | 76.47% (95% CI: 65.49–84.77%) | 86.95% (95% CI: 77.27–92.89%) |
| DeLong test, Group 2 | 2a vs 2b | Z = 0.861 | p = 0.39 | ||
| 2a vs 2c | Z = 1.162 | p = 0.246 | |||
| 2b vs 2c | Z = 0.377 | p = 0.706 | |||
| DeLong test, Group 3 | 3a vs 3b | Z = 0 | p = 1 | ||
| 3a vs 3c | Z = 0.563 | p = 0.574 | |||
| 3b vs 3c | Z = 0.637 | p = 0.524 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Savin, T.V.; Kopat, V.V.; Danilenko, E.D.; Churin, A.A.; Milichkina, A.M.; Ramsay, E.S.; Dukhovlinov, I.V.; Simbirtsev, A.S.; Totolian, A.A. Development and Clinical Validation of a Skin Test for In Vivo Assessment of SARS-CoV-2 Specific T-Cell Immunity. Viruses 2025, 17, 1186. https://doi.org/10.3390/v17091186
Savin TV, Kopat VV, Danilenko ED, Churin AA, Milichkina AM, Ramsay ES, Dukhovlinov IV, Simbirtsev AS, Totolian AA. Development and Clinical Validation of a Skin Test for In Vivo Assessment of SARS-CoV-2 Specific T-Cell Immunity. Viruses. 2025; 17(9):1186. https://doi.org/10.3390/v17091186
Chicago/Turabian StyleSavin, Tikhon V., Vladimir V. Kopat, Elena D. Danilenko, Alexey A. Churin, Anzhelika M. Milichkina, Edward S. Ramsay, Ilya V. Dukhovlinov, Andrey S. Simbirtsev, and Areg A. Totolian. 2025. "Development and Clinical Validation of a Skin Test for In Vivo Assessment of SARS-CoV-2 Specific T-Cell Immunity" Viruses 17, no. 9: 1186. https://doi.org/10.3390/v17091186
APA StyleSavin, T. V., Kopat, V. V., Danilenko, E. D., Churin, A. A., Milichkina, A. M., Ramsay, E. S., Dukhovlinov, I. V., Simbirtsev, A. S., & Totolian, A. A. (2025). Development and Clinical Validation of a Skin Test for In Vivo Assessment of SARS-CoV-2 Specific T-Cell Immunity. Viruses, 17(9), 1186. https://doi.org/10.3390/v17091186






