Seroprevalence of Neutralizing Antibodies in Healthy Adults, in Mexico, Against Human and Simian Adenovirus Types
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Serum Samples
2.2. Cells and Viruses
2.3. Anti-AdV IgG Enzyme-Linked Immunosorbent Assay
2.4. Adenovirus Neutralization Assay
2.5. Data Analysis
2.6. Phylogenetic Analysis
3. Results
3.1. Evaluation of Binding Antibodies Against HAdVs and SAdVs
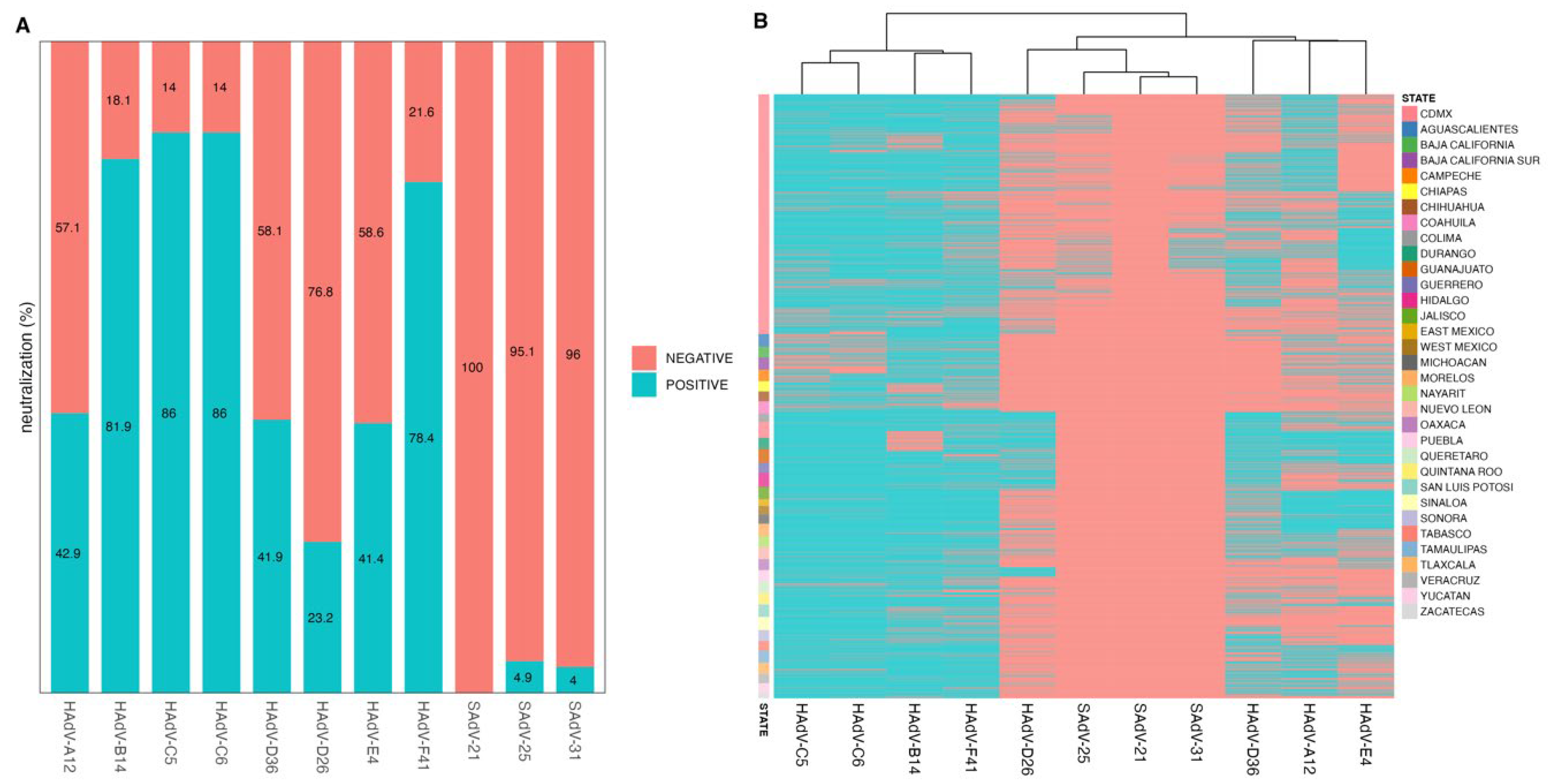
3.2. Evaluation of Neutralizing Antibodies Against HAdVs and SAdVs
4. Discussion
4.1. Seroprevalence of AdV Antibodies Across Different Populations
4.2. Considerations on Study Population and Generalizability
4.3. Implications of Seroprevalence Data for Vaccine Design
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khanal, S.; Ghimire, P.; Dhamoon, A.S. The Repertoire of Adenovirus in Human Disease: The Innocuous to the Deadly. Biomedicines 2018, 6, 30. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.N.; Knipe, D.M.; Howley, P.M. Fields Virology; Wolters Kluwer Health/Lippincott Williams & Wilkins: Waltham, MA, USA, 2007. [Google Scholar]
- Wold, W.S.; Toth, K. Adenovirus vectors for gene therapy, vaccination and cancer gene therapy. Curr. Gene Ther. 2013, 13, 421–433. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tatsis, N.; Ertl, H.C.J. Adenoviruses as vaccine vectors. Mol. Ther. 2004, 10, 616–629. [Google Scholar] [CrossRef]
- Fields, P.A.; Kowalczyk, D.W.; Arruda, V.R.; Armstrong, E.; McCleland, M.L.; Hagstrom, J.N.; Pasi, K.J.; Ertl, H.C.J.; Herzog, R.W.; High, K.A. Role of Vector in Activation of T Cell Subsets in Immune Responses against the Secreted Transgene Product Factor IX. Mol. Ther. 2000, 1, 225–235. [Google Scholar] [CrossRef]
- Saito, I.; Oya, Y.; Yamamoto, K.; Yuasa, T.; Shimojo, H. Construction of nondefective adenovirus type 5 bearing a 2.8-kilobase hepatitis B virus DNA near the right end of its genome. J. Virol. 1985, 54, 711–719. [Google Scholar] [CrossRef]
- Yamada, M.; Lewis, J.A.; Grodzicker, T. Overproduction of the protein product of a nonselected foreign gene carried by an adenovirus vector. Proc. Natl. Acad. Sci. USA 1985, 82, 3567–3571. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Haj-Ahmad, Y.; Graham, F.L. Development of a helper-independent human adenovirus vector and its use in the transfer of the herpes simplex virus thymidine kinase gene. J. Virol. 1986, 57, 267–274. [Google Scholar] [CrossRef]
- Massie, B.; Gluzman, Y.; Hassell, J.A. Construction of a Helper-Free Recombinant Adenovirus That Expresses Polyomavirus Large T Antigen. Mol. Cell. Biol. 1986, 6, 2872–2883. [Google Scholar] [CrossRef]
- Tomita, K.; Sakurai, F.; Tachibana, M.; Mizuguchi, H. Correlation between adenovirus-neutralizing antibody titer and adenovirus vector-mediated transduction efficiency following intratumoral injection. Anticancer Res. 2012, 32, 1145–1152. [Google Scholar] [PubMed]
- Fausther-Bovendo, H.; Kobinger, G.P. Pre-existing immunity against Ad vectors: Humoral, cellular, and innate response, what’s important? Hum. Vaccin. Immunother. 2014, 10, 2875–2884. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saxena, M.; Van, T.T.H.; Baird, F.J.; Coloe, P.J.; Smooker, P.M. Pre-existing immunity against vaccine vectors—friend or foe? Microbiology 2013, 159 Pt 1, 1–11. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Thacker, E.E.; Timares, L.; Matthews, Q.L. Strategies to overcome host immunity to adenovirus vectors in vaccine development. Expert. Rev. Vaccines 2009, 8, 761–777. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Milligan, I.D.; Gibani, M.M.; Sewell, R.; Clutterbuck, E.A.; Campbell, D.; Plested, E.; Nuthall, E.; Voysey, M.; Silva-Reyes, L.; McElrath, M.J.; et al. Safety and Immunogenicity of Novel Adenovirus Type 26- and Modified Vaccinia Ankara-Vectored Ebola Vaccines: A Randomized Clinical Trial. JAMA 2016, 315, 1610–1623. [Google Scholar] [CrossRef] [PubMed]
- Zhu, F.-C.; Wurie, A.H.; Hou, L.-H.; Liang, Q.; Li, Y.-H.; Russell, J.B.W.; Wu, S.-P.; Li, J.-X.; Hu, Y.-M.; Guo, Q.; et al. Safety and immunogenicity of a recombinant adenovirus type-5 vector-based Ebola vaccine in healthy adults in Sierra Leone: A single-centre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet 2017, 389, 621–628. [Google Scholar] [CrossRef]
- Bates, E.A.; Davies, J.A.; Váňová, J.; Nestić, D.; Meniel, V.S.; Koushyar, S.; Cunliffe, T.G.; Mundy, R.M.; Moses, E.; Uusi-Kerttula, H.K.; et al. Development of a low-seroprevalence, αvβ6 integrin-selective virotherapy based on human adenovirus type 10. Mol. Ther. Oncolytics 2022, 25, 43–56. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ballmann, M.Z.; Raus, S.; Engelhart, R.; Kaján, G.L.; Beqqali, A.; Hadoke, P.W.F.; van der Zalm, C.; Papp, T.; John, L.; Khan, S.; et al. Human AdV-20-42-42, a Promising Novel Adenoviral Vector for Gene Therapy and Vaccine Product Development. J. Virol. 2021, 95, e0038721. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mexican Government. Vacunas COVID-19 Autorizadas. April 2025. Available online: https://www.gob.mx/cofepris/acciones-y-programas/vacunas-covid-19-autorizadas (accessed on 3 February 2025).
- Mennechet, F.J.D.; Paris, O.; Ouoba, A.R.; Salazar Arenas, S.; Sirima, S.B.; Takoudjou Dzomo, G.R.; Diarra, A.; Traore, I.T.; Kania, D.; Eichholz, K.; et al. A review of 65 years of human adenovirus seroprevalence. Expert. Rev. Vaccines 2019, 18, 597–613. [Google Scholar] [CrossRef] [PubMed]
- Mast, T.C.; Kierstead, L.; Gupta, S.B.; Nikas, A.A.; Kallas, E.G.; Novitsky, V.; Mbewe, B.; Pitisuttithum, P.; Schechter, M.; Vardas, E.; et al. International epidemiology of human pre-existing adenovirus (Ad) type-5, type-6, type-26 and type-36 neutralizing antibodies: Correlates of high Ad5 titers and implications for potential HIV vaccine trials. Vaccine 2010, 28, 950–957. [Google Scholar] [CrossRef] [PubMed]
- Piedra, P.A.; Poveda, G.A.; Ramsey, B.; McCoy, K.; Hiatt, P.W. Incidence and Prevalence of Neutralizing Antibodies to the Common Adenoviruses in Children with Cystic Fibrosis: Implication for Gene Therapy with Adenovirus Vectors. Pediatrics 1998, 101, 1013–1019. [Google Scholar] [CrossRef]
- Duffy, M.R.; Alonso-Padilla, J.; John, L.; Chandra, N.; Khan, S.; Ballmann, M.Z.; Lipiec, A.; Heemskerk, E.; Custers, J.; Arnberg, N.; et al. Generation and characterization of a novel candidate gene therapy and vaccination vector based on human species D adenovirus type 56. J. Gen. Virol. 2018, 99, 135–147. [Google Scholar] [CrossRef] [PubMed]
- Rosenecker, J.; Harms, K.H.; Bertele, R.M.; Pohl-Koppe, A.; v Mutius, E.; Adam, D.; Nicolai, T. Adenovirus infection in cystic fibrosis patients: Implications for the use of adenoviral vectors for gene transfer. Infection 1996, 24, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Lemckert, A.A.C.; Grimbergen, J.; Smits, S.; Hartkoorn, E.; Holterman, L.; Berkhout, B.; Barouch, D.H.; Vogels, R.; Quax, P.; Goudsmit, J.; et al. Generation of a novel replication-incompetent adenoviral vector derived from human adenovirus type 49: Manufacture on PER.C6 cells, tropism and immunogenicity. J. Gen. Virol. 2006, 87, 2891–2899. [Google Scholar] [CrossRef]
- Vogels, R.; Zuijdgeest, D.; Rijnsoever, R.V.; Hartkoorn, E.; Damen, I.; de Béthune, M.-P.; Kostense, S.; Penders, G.; Helmus, N.; Koudstaal, W.; et al. Replication-Deficient Human Adenovirus Type 35 Vectors for Gene Transfer and Vaccination: Efficient Human Cell Infection and Bypass of Preexisting Adenovirus Immunity. J. Virol. 2003, 77, 8263–8271. [Google Scholar] [CrossRef]
- Barouch, D.H.; Kik, S.V.; Weverling, G.J.; Dilan, R.; King, S.L.; Maxfield, L.F.; Clark, S.; Ng’ang’a, D.; Brandariz, K.L.; Abbink, P.; et al. International seroepidemiology of adenovirus serotypes 5, 26, 35, and 48 in pediatric and adult populations. Vaccine 2011, 29, 5203–5209. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Abbink, P.; Lemckert, A.A.; Ewald, B.A.; Lynch, D.M.; Denholtz, M.; Smits, S.; Holterman, L.; Damen, I.; Vogels, R.; Thorner, A.R.; et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. J. Virol. 2007, 81, 4654–4663. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sumida, S.M.; Truitt, D.M.; Lemckert, A.A.; Vogels, R.; Custers, J.H.; Addo, M.M.; Lockman, S.; Peter, T.; Peyerl, F.W.; Kishko, M.G.; et al. Neutralizing antibodies to adenovirus serotype 5 vaccine vectors are directed primarily against the adenovirus hexon protein. J. Immunol. 2005, 174, 7179–7185. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liang, B.; Wang, W.; Li, L.; Feng, N.; Zhao, Y.; Wang, T.; Yan, F.; Yang, S.; Xia, X. Viral vectored vaccines: Design, development, preventive and therapeutic applications in human diseases. Signal Transduct. Target. Ther. 2023, 8, 149. [Google Scholar] [CrossRef]
- Ludert, J.E.; Pujol, F.H.; Arbiza, J. Human Virology in Latin America: From Biology to Control; Springer International Publishing: Berlin/Heidelberg, Germany, 2017. [Google Scholar]
- Groitl, P.; Dobner, T. Construction of adenovirus type 5 early region 1 and 4 virus mutants. Methods Mol. Med. 2007, 130, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, U.; Weigert, M.; Broaddus, C.; Myers, G. (Eds.) Cell Detection with Star-Convex Polygons 2018; Springer International Publishing: Cham, Switzerland, 2018. [Google Scholar]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer International Publishing: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Kolde, R. Pheatmap: Pretty Heatmaps. R Package Version. 2019, Volume 1, p. 726. Available online: https://cran.r-project.org/web/packages/pheatmap/pheatmap.pdf (accessed on 3 February 2025).
- Bodenhofer, U.; Bonatesta, E.; Horejš-Kainrath, C.; Hochreiter, S. Msa: An R package for multiple sequence alignment. Bioinformatics 2015, 31, 3997–3999. [Google Scholar] [CrossRef] [PubMed]
- Charif, D.; Lobry, J.R. SeqinR 1.0-2: A Contributed Package to the R Project for Statistical Computing Devoted to Biological Sequences Retrieval and Analysis. In Structural Approaches to Sequence Evolution: Molecules, Networks, Populations; Bastolla, U., Porto, M., Roman, H.E., Vendruscolo, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2007; pp. 207–232. [Google Scholar]
- Paradis, E.; Claude, J.; Strimmer, K. APE: Analyses of Phylogenetics and Evolution in R language. Bioinformatics 2004, 20, 289–290. [Google Scholar] [CrossRef]
- Yu, G.; Smith, D.K.; Zhu, H.; Guan, Y.; Lam, T.T.-Y. Ggtree: An R package for visualization and annotation of phylogenetic trees with their covariates and other associated data. Methods Ecol. Evol. 2017, 8, 28–36. [Google Scholar] [CrossRef]
- Dai, Y.; Dong, H.; Gleason, C.; Mora, J.; Kolaitis, G.; Balasubramanian, N.; Surapaneni, S.; Kozhich, A.; Jawa, V. Comparison of Pre-existing Anti-AAV8 Total Antibody Screening and Confirmatory Assays with a Cell-Based Neutralizing Assay in Normal Human Serum. AAPS J. 2023, 25, 35. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Fan, Y.; Li, X.; Gu, S.; Zhou, Z.; Xu, D.; Qiu, S.; Li, C.; Zhou, R.; Tian, X. Identification of adenovirus neutralizing antigens using capsid chimeric viruses. Virus Research 2018, 256, 100–106. [Google Scholar] [CrossRef]
- Yuan, X.; Qu, Z.; Wu, X.; Wang, Y.; Liu, L.; Wei, F.; Gao, H.; Shang, L.; Zhang, H.; Cui, H.; et al. Molecular modeling and epitopes mapping of human adenovirus type 3 hexon protein. Vaccine 2009, 27, 5103–5110. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q.; Tian, X.; Jiang, Z.; Huang, J.; Liu, Q.; Lu, X.; Luo, Q.; Zhou, R. Neutralizing epitopes mapping of human adenovirus type 14 hexon. Vaccine 2015, 33, 6659–6665. [Google Scholar] [CrossRef] [PubMed]
- Hossain, R.; Yasmin, T.; Hosen, M.I.; Nabi, A.H.M.N. In silico identification of potential epitopes present in human adenovirus proteins for vaccine design and of putative drugs for treatment against viral infection. J. Immunol. Methods 2018, 455, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Tufail, S.; Shah, M.A.; Zafar, M.; Asif, T.A.; Shehzad, A.; Shah, M.S.; Habib, M.; Saleemi, M.K.; Muddassar, M.; Mirza, O.; et al. Identification of potent epitopes on hexon capsid protein and their evaluation as vaccine candidates against infections caused by members of Adenoviridae family. Vaccine 2021, 39, 3560–3564. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Sun, X.; Ye, X.; Feng, Y.; Wang, J.; Zheng, X.; Liu, X.; Yi, C.; Hao, M.; Wang, Q.; et al. Hexon and fiber of adenovirus type 14 and 55 are major targets of neutralizing antibody but only fiber-specific antibody contributes to cross-neutralizing activity. Virology 2018, 518, 272–283. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.; Li, X.; Tian, X.; Zhou, Z.; Xing, K.; Li, H.; Tang, N.; Liu, W.; Bai, P.; Zhou, R. Serotype-specific neutralizing antibody epitopes of human adenovirus type 3 (HAdV-3) and HAdV-7 reside in multiple hexon hypervariable regions. J. Virol. 2012, 86, 7964–7975. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, W.X.; Zou, X.H.; Jiang, S.Y.; Lu, N.N.; Han, M.; Zhao, J.H.; Guo, X.J.; Zhao, S.C.; Lu, Z.Z. Prevalence of serum neutralizing antibodies to adenovirus type 5 (Ad5) and 41 (Ad41) in children is associated with age and sanitary conditions. Vaccine 2016, 34, 5579–5586. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ye, X.; Xiao, L.; Zheng, X.; Wang, J.; Shu, T.; Feng, Y.; Liu, X.; Su, W.; Wang, Q.; Li, C.; et al. Seroprevalence of Neutralizing Antibodies to Human Adenovirus Type 4 and 7 in Healthy Populations From Southern China. Front. Microbiol. 2018, 9, 3040. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tian, X.; Fan, Y.; Wang, C.; Liu, Z.; Liu, W.; Xu, Y.; Mo, C.; You, A.; Li, X.; Rong, X.; et al. Seroprevalence of Neutralizing Antibodies against Six Human Adenovirus Types Indicates the Low Level of Herd Immunity in Young Children from Guangzhou, China. Virol. Sin. 2021, 36, 373–381. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Appaiahgari, M.B.; Pandey, R.M.; Vrati, S. Seroprevalence of neutralizing antibodies to adenovirus type 5 among children in India: Implications for recombinant adenovirus-based vaccines. Clin. Vaccine Immunol. 2007, 14, 1053–1055. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Idris, O.O.; Kolawole, O.M. Seroprevalence and molecular characterization of human respiratory syncytial virus and human adenovirus among children in Ado-Ekiti, Nigeria. J. Med. Virol. 2022, 94, 2548–2557. [Google Scholar] [CrossRef] [PubMed]
- Ouoba, A.R.; Paris, O.; Adawaye, C.; Dzomo, G.T.; Fouda, A.A.; Kania, D.; Diarra, A.; Kallo, Z.A.; Traore, I.T.; Sirima, S.B.; et al. Prevalence of Neutralizing Antibodies against Adenoviruses types -C5, -D26 and -B35 used in vaccination platforms, in Healthy and HIV-Infected Adults and Children from Burkina Faso and Chad. medRxiv 2022. preprint. [Google Scholar] [CrossRef]
- Patel, E.U.; Bloch, E.M.; Grabowski, M.K.; Goel, R.; Lokhandwala, P.M.; Brunker, P.A.R.; White, J.L.; Shaz, B.; Ness, P.M.; Tobian, A.A.R. Sociodemographic and behavioral characteristics associated with blood donation in the United States: A population-based study. Transfusion 2019, 59, 2899–2907. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ma, M.; Yang, R.; Gu, J.; Ke, S.; Du, X.; Zheng, J. Factors associated with blood donation among college and university students in Wuhan, China: Structural equation model. BMC Public Health 2024, 24, 1847. [Google Scholar] [CrossRef]
- Feng, W.; Yun, W.; Le, W.; Zhi-Guo, X.; Hai-Ying, Y.; Shu-Fang, W.; Zhen-Yan, W.; Yi-Zhu, C.; Quan, S.; Jing-Xian, F. The influence of demographic and lifestyle factors on blood donation delay among student population: A retrospective study. Front. Public Health 2023, 11, 1297472. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shenga, N.; Thankappan, K.; Kartha, C.; Pal, R. Analyzing sociodemographic factors amongst blood donors. J. Emerg. Trauma Shock 2010, 3, 21–25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mazurkievz de Freitas, E.; Targa Pinto, R.; Forlin Robert, A.; Malta Purim, K.S. Sociodemographic Profile of Blood Donations and Ways to Encourage Them. Cureus 2024, 16, e60688. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhao, H.; Xu, C.; Luo, X.; Wei, F.; Wang, N.; Shi, H.; Ren, X. Seroprevalence of Neutralizing Antibodies against Human Adenovirus Type-5 and Chimpanzee Adenovirus Type-68 in Cancer Patients. Front. Immunol. 2018, 9, 335. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dehghan, S.; Seto, J.; Liu, E.B.; Ismail, A.M.; Madupu, R.; Heim, A.; Jones, M.S.; Dyer, D.W.; Chodosh, J.; Seto, D.A. Zoonotic Adenoviral Human Pathogen Emerged through Genomic Recombination among Human and Nonhuman Simian Hosts. J. Virol. 2019, 93, e00564-19. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, E.C.; Yagi, S.; Kelly, K.R.; Mendoza, S.P.; Maninger, N.; Rosenthal, A.; Spinner, A.; Bales, K.L.; Schnurr, D.P.; Lerche, N.W.; et al. Correction: Cross-Species Transmission of a Novel Adenovirus Associated with a Fulminant Pneumonia Outbreak in a New World Monkey Colony. PLOS Pathog. 2011, 7, e1002155. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López, R.E.; Valdés Alemán, M.; Torres-Flores, J.M.; Pérez-Llano, Y.; Cabrera Gaytán, D.A.; Santacruz Tinoco, C.E.; Alvarado Yaah, J.E.; Anguiano Hernández, Y.M.; Martínez Miguel, B.; Muñoz Medina, J.E.; et al. Seroprevalence of Neutralizing Antibodies in Healthy Adults, in Mexico, Against Human and Simian Adenovirus Types. Viruses 2025, 17, 1184. https://doi.org/10.3390/v17091184
López RE, Valdés Alemán M, Torres-Flores JM, Pérez-Llano Y, Cabrera Gaytán DA, Santacruz Tinoco CE, Alvarado Yaah JE, Anguiano Hernández YM, Martínez Miguel B, Muñoz Medina JE, et al. Seroprevalence of Neutralizing Antibodies in Healthy Adults, in Mexico, Against Human and Simian Adenovirus Types. Viruses. 2025; 17(9):1184. https://doi.org/10.3390/v17091184
Chicago/Turabian StyleLópez, Raúl E., Margarita Valdés Alemán, Jesús M. Torres-Flores, Yordanis Pérez-Llano, David Alejandro Cabrera Gaytán, Clara Esperanza Santacruz Tinoco, Julio Elias Alvarado Yaah, Yu Mei Anguiano Hernández, Bernardo Martínez Miguel, José Esteban Muñoz Medina, and et al. 2025. "Seroprevalence of Neutralizing Antibodies in Healthy Adults, in Mexico, Against Human and Simian Adenovirus Types" Viruses 17, no. 9: 1184. https://doi.org/10.3390/v17091184
APA StyleLópez, R. E., Valdés Alemán, M., Torres-Flores, J. M., Pérez-Llano, Y., Cabrera Gaytán, D. A., Santacruz Tinoco, C. E., Alvarado Yaah, J. E., Anguiano Hernández, Y. M., Martínez Miguel, B., Muñoz Medina, J. E., Sandoval Gutiérrez, N., Lagunes, I. R., Arroyo Pérez, J. A., & González, R. A. (2025). Seroprevalence of Neutralizing Antibodies in Healthy Adults, in Mexico, Against Human and Simian Adenovirus Types. Viruses, 17(9), 1184. https://doi.org/10.3390/v17091184






