In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks
Abstract
1. Introduction
2. Materials and Methods
2.1. Virus Isolation, Sequencing and Phylogenetic Analysis
2.2. IVPI in Chickens and Ducks
2.3. Chicken and Duck In Ovo Model
2.4. Statistics
3. Results
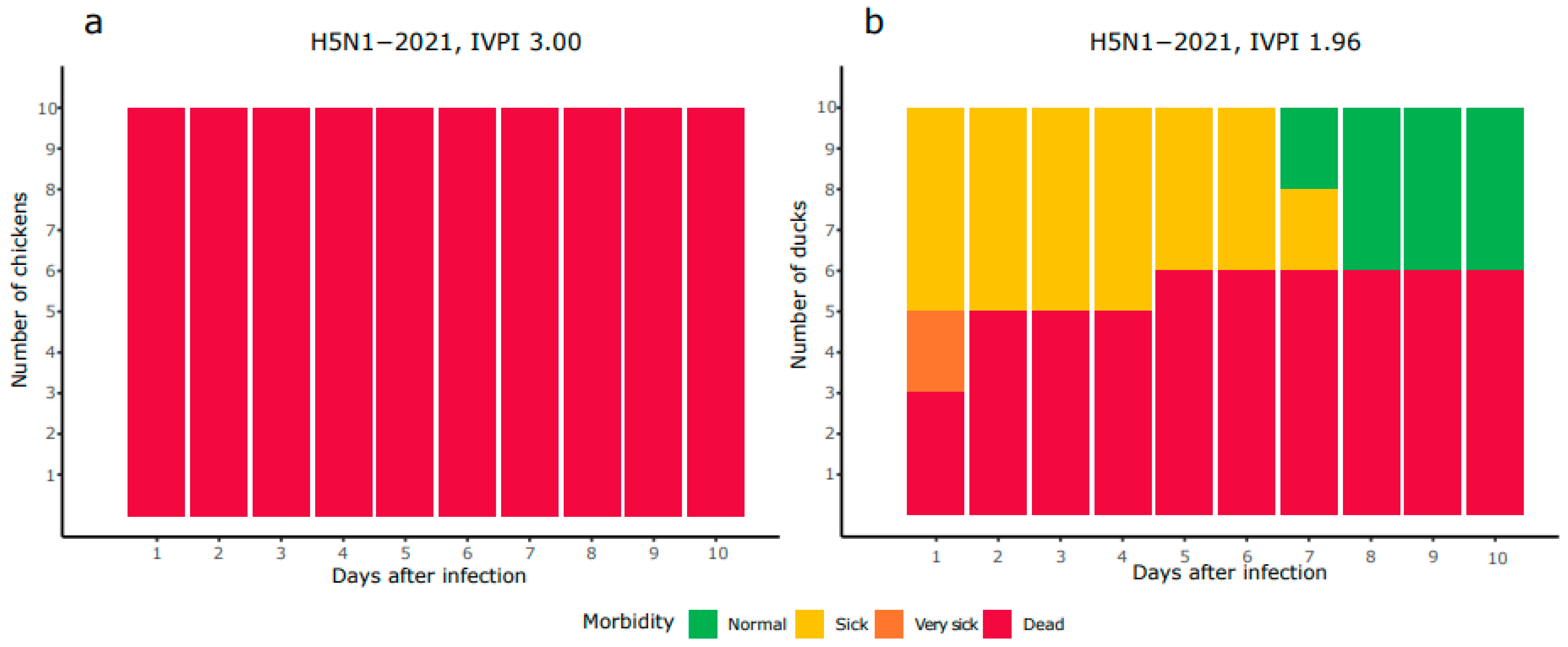
3.1. Mild Virulence of HPAI H5N1-2021 Virus in Ducks Compared to Chickens
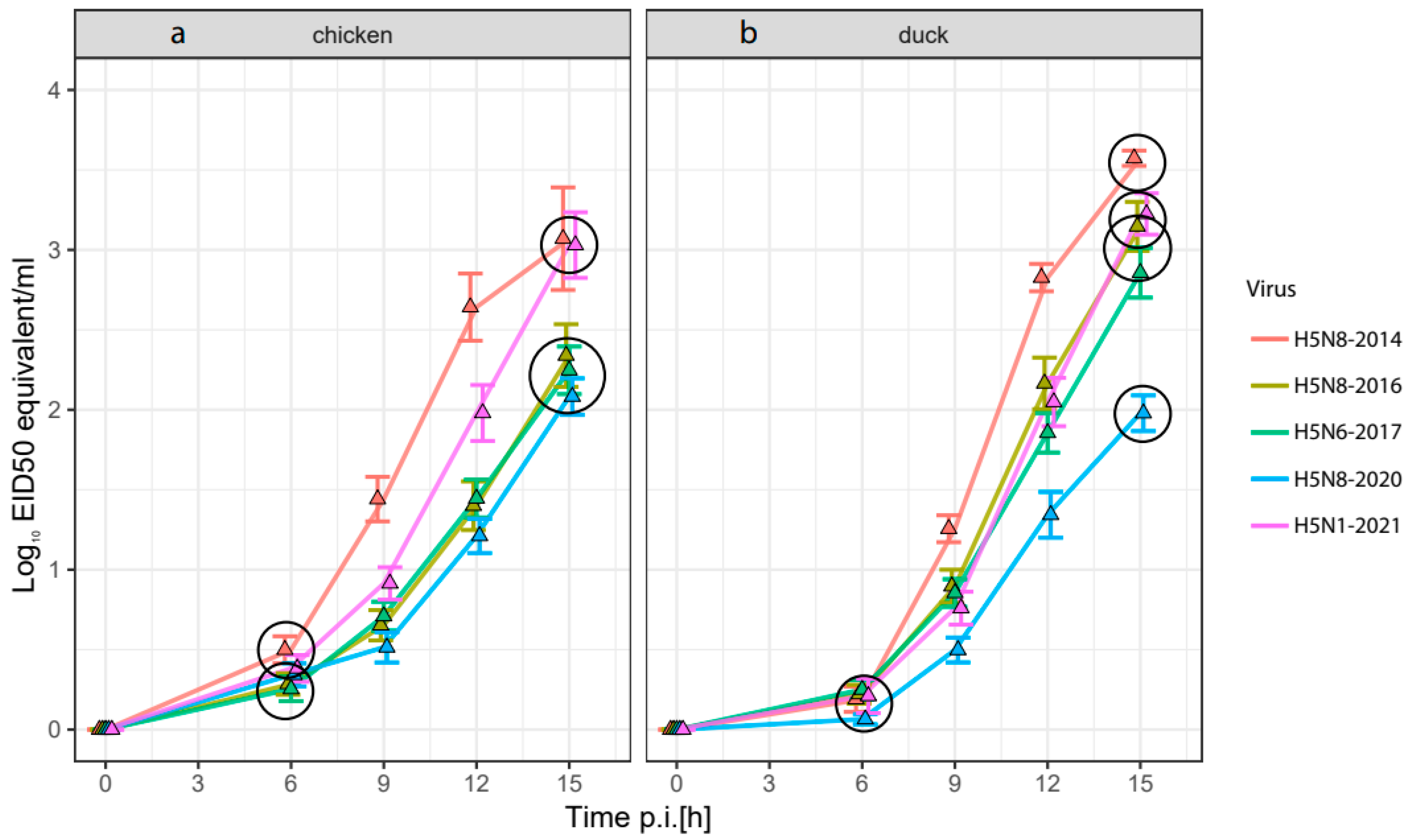
3.2. Replication Rate of HPAI H5-Viruses in Embryonated Chicken and Duck Eggs
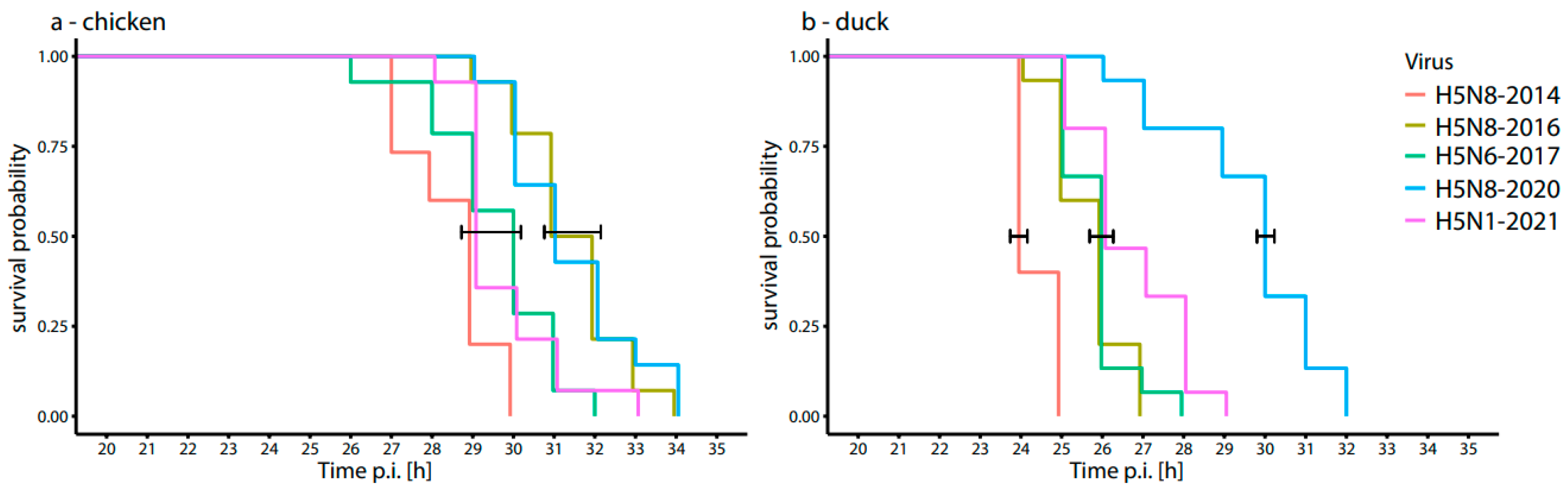
3.3. Time to Death of Chicken and Duck Embryos after Inoculation with HPAI H5-Viruses
3.4. Systemic Virus Spread in Chicken and Duck Embryos after Infection with HPAI-H5 Viruses
3.5. Tissue-Specific Tropism of HPAI H5-Viruses in Chicken and Duck Embryos
3.6. Comparative Analysis of the Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Adlhoch, C.; Gossner, C.; Koch, G.; Brown, I.; Bouwstra, R.; Verdonck, F.; Penttinen, P.; Harder, T. Comparing introduction to Europe of highly pathogenic avian influenza viruses A (H5N8) in 2014 and A (H5N1) in 2005. Eurosurveillance 2014, 19, 20996. [Google Scholar] [CrossRef]
- Napp, S.; Majó, N.; Sánchez-Gónzalez, R.; Vergara-Alert, J. Emergence and spread of highly pathogenic avian influenza A (H5N8) in Europe in 2016–2017. Transbound. Emerg. Dis. 2018, 65, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Poen, M.J.; Venkatesh, D.; Bestebroer, T.M.; Vuong, O.; Scheuer, R.D.; Oude Munnink, B.B.; de Meulder, D.; Richard, M.; Kuiken, T.; Koopmans, M.P. Co-circulation of genetically distinct highly pathogenic avian influenza A clade 2.3. 4.4 (H5N6) viruses in wild waterfowl and poultry in Europe and East Asia, 2017–2018. Virus Evol. 2019, 5, vez004. [Google Scholar] [CrossRef] [PubMed]
- King, J.; Schulze, C.; Engelhardt, A.; Hlinak, A.; Lennermann, S.-L.; Rigbers, K.; Skuballa, J.; Staubach, C.; Mettenleiter, T.C.; Harder, T. Novel HPAIV H5N8 reassortant (clade 2.3. 4.4 b) detected in Germany. Viruses 2020, 12, 281. [Google Scholar] [CrossRef] [PubMed]
- EFSA; ECDC; EURL; Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, É.; Staubach, C.; et al. Scientific report: Avian influenza overview December 2021–March 2022. EFSA J. 2022, 20, e07289. [Google Scholar] [PubMed]
- Rijks, J.M.; Leopold, M.F.; Kühn, S.; in ‘t Veld, R.; Schenk, F.; Brenninkmeijer, A.; Lilipaly, S.J.; Ballmann, M.Z.; Kelder, L.; de Jong, J.W. Mass Mortality Caused by Highly Pathogenic Influenza A (H5N1) Virus in Sandwich Terns, the Netherlands, 2022. Emerg. Infect. Dis. 2022, 28, 2538–2542. [Google Scholar] [CrossRef] [PubMed]
- Bordes, L.; Vreman, S.; Heutink, R.; Roose, M.; Venema, S.; Pritz-Verschuren, S.B.; Rijks, J.M.; Gonzales, J.L.; Germeraad, E.A.; Engelsma, M. Highly pathogenic avian influenza H5N1 virus infections in wild red foxes (Vulpes vulpes) show neurotropism and adaptive virus mutations. Microbiol. Spectr. 2023, 11, e02867-22. [Google Scholar] [CrossRef]
- Vreman, S.; Kik, M.; Germeraad, E.; Heutink, R.; Harders, F.; Spierenburg, M.; Engelsma, M.; Rijks, J.; van den Brand, J.; Beerens, N. Zoonotic Mutation of Highly Pathogenic Avian Influenza H5N1 Virus Identified in the Brain of Multiple Wild Carnivore Species. Pathogens 2023, 12, 168. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; Organisation mondiale de la Santé. Antigenic and genetic characteristics of zoonotic influenza A viruses and development of candidate vaccine viruses for pandemic preparedness—Caractéristiques génétiques et antigéniques des virus grippaux A zoonotiques et mise au point de virus vaccinaux candidats pour se préparer à une pandémie. Wkly. Epidemiol. Rec. Relev. Épidémiologique Hebd. 2022, 97, 550–565. [Google Scholar]
- Beerens, N.; Germeraad, E.A.; Venema, S.; Verheij, E.; Pritz-Verschuren, S.B.; Gonzales, J.L. Comparative pathogenicity and environmental transmission of recent highly pathogenic avian influenza H5 viruses. Emerg. Microbes Infect. 2021, 10, 97–108. [Google Scholar] [CrossRef]
- Pantin-Jackwood, M.J.; Costa-Hurtado, M.; Shepherd, E.; DeJesus, E.; Smith, D.; Spackman, E.; Kapczynski, D.R.; Suarez, D.L.; Stallknecht, D.E.; Swayne, D.E. Pathogenicity and transmission of H5 and H7 highly pathogenic avian influenza viruses in mallards. J. Virol. 2016, 90, 9967–9982. [Google Scholar] [CrossRef] [PubMed]
- Pantin-Jackwood, M.J.; Costa-Hurtado, M.; Bertran, K.; DeJesus, E.; Smith, D.; Swayne, D.E. Infectivity, transmission and pathogenicity of H5 highly pathogenic avian influenza clade 2.3. 4.4 (H5N8 and H5N2) United States index viruses in Pekin ducks and Chinese geese. Vet. Res. 2017, 48, 33. [Google Scholar] [CrossRef] [PubMed]
- WOAH, Manual of Diagnostic Tests and Vaccines for Terrestrial Animals. Available online: https://www.woah.org/en/what-we-do/standards/codes-and-manuals/terrestrial-manual-online-access/ (accessed on 28 June 2023).
- Webster, R.G.; Rott, R. Influenza virus A pathogenicity: The pivotal role of hemagglutinin. Cell 1987, 50, 665–666. [Google Scholar] [CrossRef] [PubMed]
- Böttcher-Friebertshäuser, E.; Garten, W.; Matrosovich, M.; Klenk, H.D. The hemagglutinin: A determinant of pathogenicity. Influenza Pathog. Control 2014, 1, 3–34. [Google Scholar]
- Abdelwhab, E.-S.M.; Veits, J.; Mettenleiter, T.C. Genetic changes that accompanied shifts of low pathogenic avian influenza viruses toward higher pathogenicity in poultry. Virulence 2013, 4, 441–452. [Google Scholar] [CrossRef] [PubMed]
- EMC; IZSVe; in ‘t Veld, R.; Richard, M.; Fouchier, R.; Monne, I.; Kuiken, T. Mechanisms and risk factors for mutation from low to highly pathogenic avian influenza virus. EFSA Support. Publ. 2017, 14, 1287E. [Google Scholar]
- Lee, D.-H.; Criado, M.F.; Swayne, D.E. Pathobiological origins and evolutionary history of highly pathogenic avian influenza viruses. Cold Spring Harb. Perspect. Med. 2021, 11, a038679. [Google Scholar] [CrossRef] [PubMed]
- Beerens, N.; Heutink, R.; Pritz-Verschuren, S.; Germeraad, E.; Bergervoet, S.; Harders, F.; Bossers, A.; Koch, G. Genetic relationship between poultry and wild bird viruses during the highly pathogenic avian influenza H5N6 epidemic in the Netherlands, 2017–2018. Transbound. Emerg. Dis. 2019, 66, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Engelsma, M.; Heutink, R.; Harders, F.; Germeraad, E.A.; Beerens, N. Multiple Introductions of Reassorted Highly Pathogenic Avian Influenza H5Nx Viruses Clade 2.3. 4.4 b Causing Outbreaks in Wild Birds and Poultry in The Netherlands, 2020–2021. Microbiol. Spectr. 2022, 10, e02499-21. [Google Scholar] [CrossRef]
- EFSA; ECDC; EURL; Brown, I.; Mulatti, P.; Smietanka, K.; Staubach, C.; Willeberg, P.; Adlhoch, C.; Candiani, D. Scientific report on the avian influenza overview October 2016–August 2017. EFSA J. 2017, 15, e05018. [Google Scholar]
- EFSA; ECDC; EURL; Adlhoch, C.; Brouwer, A.; Kuiken, T.; Mulatti, P.; Smietanka, K.; Staubach, C.; Muñoz Guajardo, I. Scientific report: Avian influenza overview February–May 2018. EFSA J. 2018, 16, e05358. [Google Scholar] [PubMed]
- EFSA. Highly pathogenic avian influenza A subtype H5N8. EFSA J. 2014, 12, 3941. [Google Scholar]
- EFSA; ECDC; EURL; Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, É.; Staubach, C.; et al. Scientific report: Avian influenza overview May–September 2021. EFSA J. 2022, 20, e07122. [Google Scholar] [PubMed]
- EFSA; ECDC; EURL; Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, É.; Staubach, C. Scientific report: Avian influenza overview June–September 2022. EFSA J. 2022, 20, e07597. [Google Scholar] [PubMed]
- Vigeveno, R.M.; Poen, M.J.; Parker, E.; Holwerda, M.; de Haan, K.; van Montfort, T.; Lewis, N.S.; Russell, C.A.; Fouchier, R.A.; de Jong, M.D. Outbreak severity of highly pathogenic avian influenza A (H5N8) viruses is inversely correlated to polymerase complex activity and interferon induction. J. Virol. 2020, 94, e00375-20. [Google Scholar] [CrossRef] [PubMed]
- Youk, S.-S.; Leyson, C.M.; Seibert, B.A.; Jadhao, S.; Perez, D.R.; Suarez, D.L.; Pantin-Jackwood, M.J. Mutations in PB1, NP, HA, and NA contribute to increased virus fitness of H5N2 highly pathogenic avian influenza virus clade 2.3. 4.4 in chickens. J. Virol. 2021, 95, e01675-20. [Google Scholar] [CrossRef] [PubMed]
- Bortz, E.; Westera, L.; Maamary, J.; Steel, J.; Albrecht, R.A.; Manicassamy, B.; Chase, G.; Martínez-Sobrido, L.; Schwemmle, M.; García-Sastre, A. Host-and strain-specific regulation of influenza virus polymerase activity by interacting cellular proteins. MBio 2011, 2, e00151-11. [Google Scholar] [CrossRef]
- Seekings, A.H.; Howard, W.A.; Nuñéz, A.; Slomka, M.J.; Banyard, A.C.; Hicks, D.; Ellis, R.J.; Nuñéz-García, J.; Hartgroves, L.C.; Barclay, W.S. The emergence of H7N7 highly pathogenic avian influenza virus from low pathogenicity avian influenza virus using an in ovo embryo culture model. Viruses 2020, 12, 920. [Google Scholar] [CrossRef] [PubMed]
- Parvin, R.; Schinkoethe, J.; Grund, C.; Ulrich, R.; Bönte, F.; Behr, K.P.; Voss, M.; Samad, M.A.; Hassan, K.E.; Luttermann, C. Comparison of pathogenicity of subtype H9 avian influenza wild-type viruses from a wide geographic origin expressing mono-, di-, or tri-basic hemagglutinin cleavage sites. Vet. Res. 2020, 51, 48. [Google Scholar] [CrossRef]
- de Bruin, A.; Spronken, M.I.; Bestebroer, T.M.; Fouchier, R.A.; Richard, M. Reduced replication of highly pathogenic avian influenza virus in duck endothelial cells compared to chicken endothelial cells is associated with stronger antiviral responses. Viruses 2022, 14, 165. [Google Scholar] [CrossRef]
- Bouwstra, R.; Koch, G.; Heutink, R.; Harders, F.; Van Der Spek, A.; Elbers, A.; Bossers, A. Phylogenetic analysis of highly pathogenic avian influenza A (H5N8) virus outbreak strains provides evidence for four separate introductions and one between-poultry farm transmission in the Netherlands, November 2014. Eurosurveillance 2015, 20, 21174. [Google Scholar] [CrossRef] [PubMed]
- Reed, L.J.; Muench, H. A simple method of estimating fifty per cent endpoints. Am. J. Epidemiol. 1938, 27, 493–497. [Google Scholar] [CrossRef]
- Katoh, K.; Toh, H. Parallelization of the MAFFT multiple sequence alignment program. Bioinformatics 2010, 26, 1899–1900. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.-T.; Schmidt, H.A.; Von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Smith, D.K.; Zhu, H.; Guan, Y.; Lam, T.T.Y. ggtree: An R package for visualization and annotation of phylogenetic trees with their covariates and other associated data. Methods Ecol. Evol. 2017, 8, 28–36. [Google Scholar] [CrossRef]
- Vreman, S.; Bergervoet, S.A.; Zwart, R.; Stockhofe-Zurwieden, N.; Beerens, N. Tissue tropism and pathology of highly pathogenic avian influenza H5N6 virus in chickens and Pekin ducks. Res. Vet. Sci. 2022, 146, 1–4. [Google Scholar] [CrossRef]
- Therneau, T. Mixed effects Cox models. CRAN Repos. 2015, 1–14. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Available online: http://www.R-project.org/ (accessed on 28 June 2023).
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 48. [Google Scholar] [CrossRef]
- Therneau, T. A Package for Survival Analysis in R (R Package Version 3.5-0). CRAN Repos. 2023, 1–188. [Google Scholar]
- Harrell, F.E., Jr.; Harrell, M.F.E., Jr. Package ‘hmisc’. CRAN Repos. 2019, 235–236. [Google Scholar]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Emmeans: Estimated marginal means, aka least-squares means, R package version 1. 5. 1. CRAN Repos. 2020, 1–101. [Google Scholar]
- Hincke, M.T.; Da Silva, M.; Guyot, N.; Gautron, J.; McKee, M.D.; Guabiraba-Brito, R.; Réhault-Godbert, S. Dynamics of structural barriers and innate immune components during incubation of the avian egg: Critical interplay between autonomous embryonic development and maternal anticipation. J. Innate Immun. 2019, 11, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Caliendo, V.; Leijten, L.; Begeman, L.; Poen, M.J.; Fouchier, R.A.; Beerens, N.; Kuiken, T. Enterotropism of highly pathogenic avian influenza virus H5N8 from the 2016/2017 epidemic in some wild bird species. Vet. Res. 2020, 51, 117. [Google Scholar] [CrossRef]
- Caliendo, V.; Leijten, L.; van de Bildt, M.; Germeraad, E.; Fouchier, R.A.; Beerens, N.; Kuiken, T. Tropism of highly pathogenic avian influenza H5 viruses from the 2020/2021 epizootic in wild ducks and geese. Viruses 2022, 14, 280. [Google Scholar] [CrossRef] [PubMed]
| Virus Isolate | EPI Number | Clade 2.3.4.4 | Poultry Species Virus Was Isolated from | Differences between Swab Isolate and Second Egg Passage |
|---|---|---|---|---|
| A/chicken/Netherlands/14015531/2014 | EPI_ISL_18600237 | Group c | Chicken | PB2-R574K, PA-F451S, NA-A332V |
| A/duck/Netherlands/16014829-001005/2016 | EPI_ISL_529179 | Group b | Duck | PB2-V344A |
| A/Duck/Netherlands/17017236-001-005/2017 | EPI_ISL_18600770 | Group b | Duck | PB2-I30L |
| A/chicken/Netherlands/20016597-026030/2020 | EPI_ISL_8650948 | Group b | Chicken | - |
| A/chicken/Netherlands/21037287-006010/2021 | EPI_ISL_9856775 | Group b—genogroup AB | Chicken | - |
| Virus Isolate | Abbreviation | IVPI Chicken | IVPI Pekin Duck |
|---|---|---|---|
| A/chicken/Netherlands/14015531/2014 | H5N8-2014 [19] | 3.00 | 1.87 |
| A/duck/Netherlands/16014829-001005/2016 | H5N8-2016 [19] | 3.00 | 2.99 |
| A/Duck/Netherlands/17017236-001-005/2017 | H5N6-2017 [19] | 2.99 | 3.00 |
| A/chicken/Netherlands/20016597-026030/2020 | H5N8-2020 [20] | 2.98 | 1.74 |
| A/chicken/Netherlands/21037287-006010/2021 | H5N1-2021 | 3.00 | 1.96 |
| IVPI Chicken | Replication Rate Chicken | Time to Death Chicken | IHC Chicken | IVPI Pekin Duck | Replication Rate Pekin Duck | Time to Death Pekin Duck | IHC Pekin Duck | |
|---|---|---|---|---|---|---|---|---|
| H5N8-2014 | 1.00 | 1.06 | 0.96 | 2.14 (**) | 0.95 | 1.09 * | 0.92 * | 1.75 ** |
| H5N8-2016 | 1.00 | 0.78 * | 1.05 * | 1.49 * | 1.53 | 0.94 | 0.96 | 1.30 * |
| H5N6-2017 | 1.00 | 0.72 * | 1.00 | 1.49 * | 1.53 | 0.82 * | 0.97 | 1.00 |
| H5N8-2020 | 0.99 | 0.70 * | 1.05 * | 0.57 (*) | 0.89 | 0.60 ** | 1.11 * | 0.50 * |
| H5N1-2021 a | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bordes, L.; Gonzales, J.L.; Vreman, S.; Venema, S.; Portier, N.; Germeraad, E.A.; van der Poel, W.H.M.; Beerens, N. In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks. Viruses 2024, 16, 563. https://doi.org/10.3390/v16040563
Bordes L, Gonzales JL, Vreman S, Venema S, Portier N, Germeraad EA, van der Poel WHM, Beerens N. In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks. Viruses. 2024; 16(4):563. https://doi.org/10.3390/v16040563
Chicago/Turabian StyleBordes, Luca, José L. Gonzales, Sandra Vreman, Sandra Venema, Nadia Portier, Evelien A. Germeraad, Wim H. M. van der Poel, and Nancy Beerens. 2024. "In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks" Viruses 16, no. 4: 563. https://doi.org/10.3390/v16040563
APA StyleBordes, L., Gonzales, J. L., Vreman, S., Venema, S., Portier, N., Germeraad, E. A., van der Poel, W. H. M., & Beerens, N. (2024). In Ovo Models to Predict Virulence of Highly Pathogenic Avian Influenza H5-Viruses for Chickens and Ducks. Viruses, 16(4), 563. https://doi.org/10.3390/v16040563







