Bacteriophage–Host Interactions and the Therapeutic Potential of Bacteriophages
Abstract
1. Introduction
2. Classification of Bacteriophages
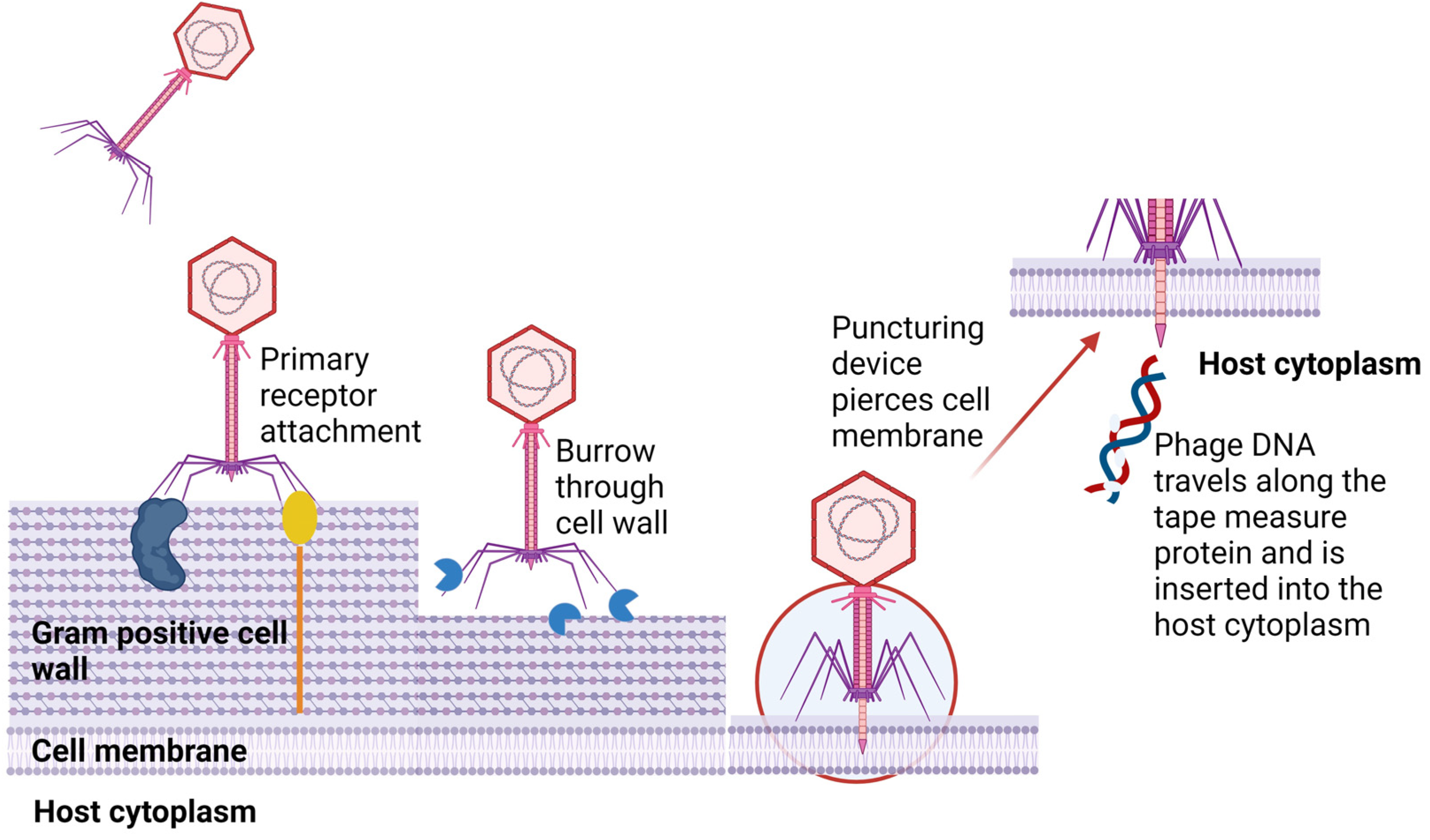
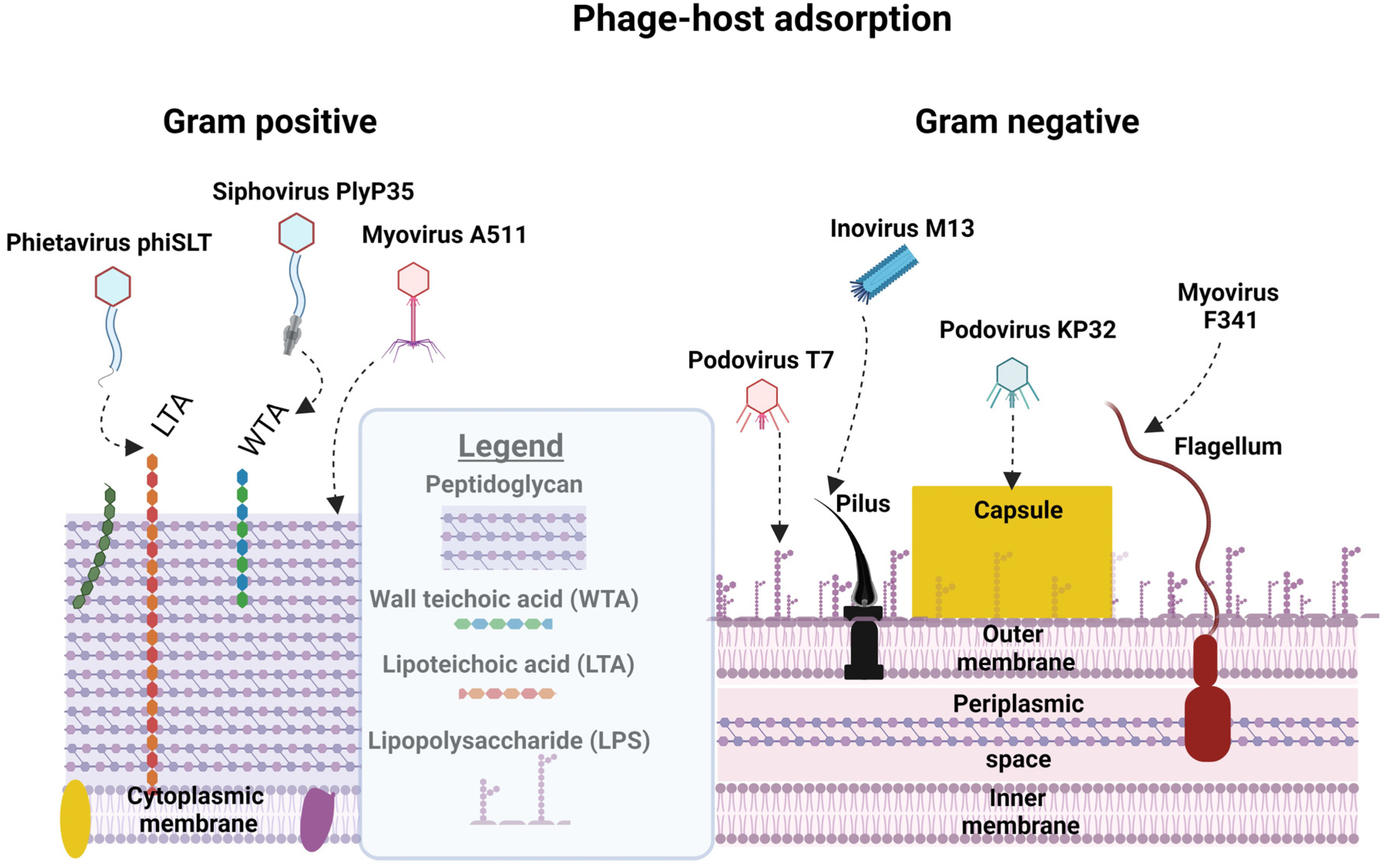
3. Phage–Host Adsorption and Cell Entry Strategies
4. Resistance to Phage Attack
5. Bacterial Immunity to Phage Infections
6. Bacteriophage-Based Therapeutics
6.1. Phage Therapy
| Infection(s)/Phage Trial Interest | Causative Agent(s)/ Agents of Interest | Outcomes/Comments | Reference/ Clinical Trial Identifier |
|---|---|---|---|
| Suppurative skin infections * | Pseudomonas, Staphylococcus, Klebsiella, Proteus, and E. coli | Thirty-one patients were treated orally and locally for chronically infected skin ulcers with a 74% success rate | [107,108,109] |
| Acute postoperative empyema in chronic suppurative lung diseases * | Staphylococcus, Streptococcus, E. coli, Proteus, Pseudomonas aeruginosa, Burkholderia dolosa | Phage–antibiotic combinations were used in the successful treatment of 45 patients | [110,111,112] |
| Complications due to bacterial infections in cancer patients * | Staphylococcus and Pseudomonas | 82% (65) successful treatment with phages compared to 61% (66) of patients treated with antibiotics | [113] |
| Recurrent subphrenic abscess * | Antibiotic-resistant E. coli | A single patient was successfully treated with phages after 33 days | [114] |
| Urinary tract infections (UTIs) * | Staphylococcus, E. coli, and Proteus | Forty-six UTI patients were treated with phages with 92% making clinical improvements and 84% achieving bacterial clearance | [115] |
| Rhinitis, pharyngitis, dermatitis, and conjunctivitis * | Staphylococcus, Streptococcus, E. coli, Proteus, enterococci, and P. aeruginosa | Patients were treated with phages (360), antibiotics (404), and phage–antibiotic combinations (576). Clinical improvements of 86%, 48%, and 83% across the treatment regimes, respectively | [116] |
| Cerebrospinal meningitis * | K. pneumoniae | Successful treatment with orally administered phages in a newborn. | [117] |
| Bacterial diarrhea | E. coli | Orally administered coliphages showed no improvement in clinical outcome; some dysbiosis with streptococci was observed | [118] |
| Complicated or recurrent UTI patients with transurethral resection of the prostate | Enterococcus, E. coli, streptococci, P. mirabilis, P. aeruginosa, staphylococci | Patients with intravesical-administered pyophage cocktail, orally administered antibiotics, and a placebo bladder irrigation. Success rates of 18%, 28% and 37% were observed, respectively | [85] |
| Burn wounds | P. aeruginosa | Phages PP1131 showed no significant difference to standard of care antibiotics—patients treated with PP1131 were found to have phage-resistant P. aeruginosa | [119] |
| Prosthetic joint infections | S. aureus, S. epidermidis, S. lugdunensis, Streptococcus sp., E. faecium, E. faecalis, E. coli, P. aeruginosa, and/or K. pneumoniae | Phage treatment, with intraoperative and intravenous PhageBank™ bacteriophages, in conjunction with standard-of-care antibiotics/Debridement, Antibiotics, and Implant Retention (DAIR) procedures. Completion is predicted in 2024 | [87,91,120] |
| Diabetic foot ulcers (DFUs) | Staphylococcus spp., wound microbiome | Use of anti-staphylococcal phage gel (Intralytix Inc., Baltimore, MA, USA). Effect on bacterial microbiome of DFU wounds and patient outcomes. Trial was abandoned for funding reasons | [121] |
| Probiotic application for overall gut health | Bifidobacterium animalis subsp. lactis BL04 | The use of bacteriophages (PreforPro) increased the survival and efficacy of probiotic bacteria administered vs. probiotics only vs. placebo | [122] |
| Phages preventing the acquisition of multidrug-resistant enterobacteria (PHAGE-BMR) | E. coli or K. pneumoniae containing ESBL or carbapenemases | Collection of multidrug-resistant bacteria from patients in intensive care, subsequent search for presence and absence of phages in carriers/non-carriers. Currently active but of unknown status | [123] |
| Phage dynamics and influences during human gut microbiome establishment (METAKIDS) | A broad range of bacteriophage and bacterial hosts | Characterize phage and bacterial genomes, abundance, and variations during infant gut development. Terminated | [124] |
| Bacterial infection in cystic fibrosis patients | P. aeruginosa | A cocktail of 10 bacteriophages was used to reduce Pseudomonas presence after 6 and 24 h including sensitivity of isolates. Completed with no recorded outcomes | [125] |
| Prebiotic | Escherichia coli and microbiota | Commercial coliphage cocktail effects on the microbiota and systemic inflammation. No disruption to microbiota and no effect on inflammatory markers | [126] |
| Venous leg ulcers | P. aeruginosa, S. aureus, and E. coli | Polyvalent phage preparation of 8 bacteriophages was assessed for their safety and efficacy. No available outcomes but the trial was completed | [127] |
| Lower urinary tract colonization | E. coli | Assess the safety, tolerability, pharmacokinetics, and pharmacodynamics of phage cocktail LBP-EC01 | [128,129] |
| Safety of topical phage solution intended for wound infections | S. aureus | Evaluating the safety and skin reactions to ascending doses of phages compared to the placebo | [130,131] |
| Infection(s) | Bacterial Species | Outcome/Comment | Reference |
|---|---|---|---|
| Complicated necrotizing pancreatitis | Acinetobacter baumannii | Clearance of A. baumannii and return to health using intravenously (IV) and percutaneously administered (9) phages screened from a phage bank | [86] |
| Bacteremia | P. aeruginosa | An IV-administered bacteriophage cocktail comprising two phages cleared the bacteremia, but the patient succumbed to other complications | [132] |
| Lung infection and transplant recipient | P. aeruginosa | An IV- and nebulizer-administered bacteriophage cocktail, AB-PA01 and Navy, with the patient recovering from pneumonia | [111] |
| Infection of left ventricular assist device | P. aeruginosa | Six-week IV-administered (3) phage cocktail; the patient was clear and then relapsed but a change in antibiotics led to recovery | [133] |
| Osteomyelitis | A. baumannii and K. pneumoniae | The patient developed postoperative infection with multidrug-resistant isolates. IV bacteriophage–antibiotic combination led to the patient’s full recovery without the need for amputation | [133] |
| UTI | ESBL E. coli | Phage treatment with two phages over 23 days in conjunction with antibiotic treatment led to negative urine cultures and full recovery of the patient | [132] |
| CNS infection of a recovering trauma patient | A. baumannii | IV treatment with an A. baumannii phage for 8 days led to CSF cultures coming back negative for A. baumannii but positive for K. pneumoniae and S. aureus. The patient was declared brain dead and later announced deceased | [132] |
| Lung infection of cystic fibrosis patient | Achromobacter xylosoxidans | Cefiderocol and phage treatments were performed for 5 days followed by continuous phage therapy. The patient recovered and was discharged | [132,134] |
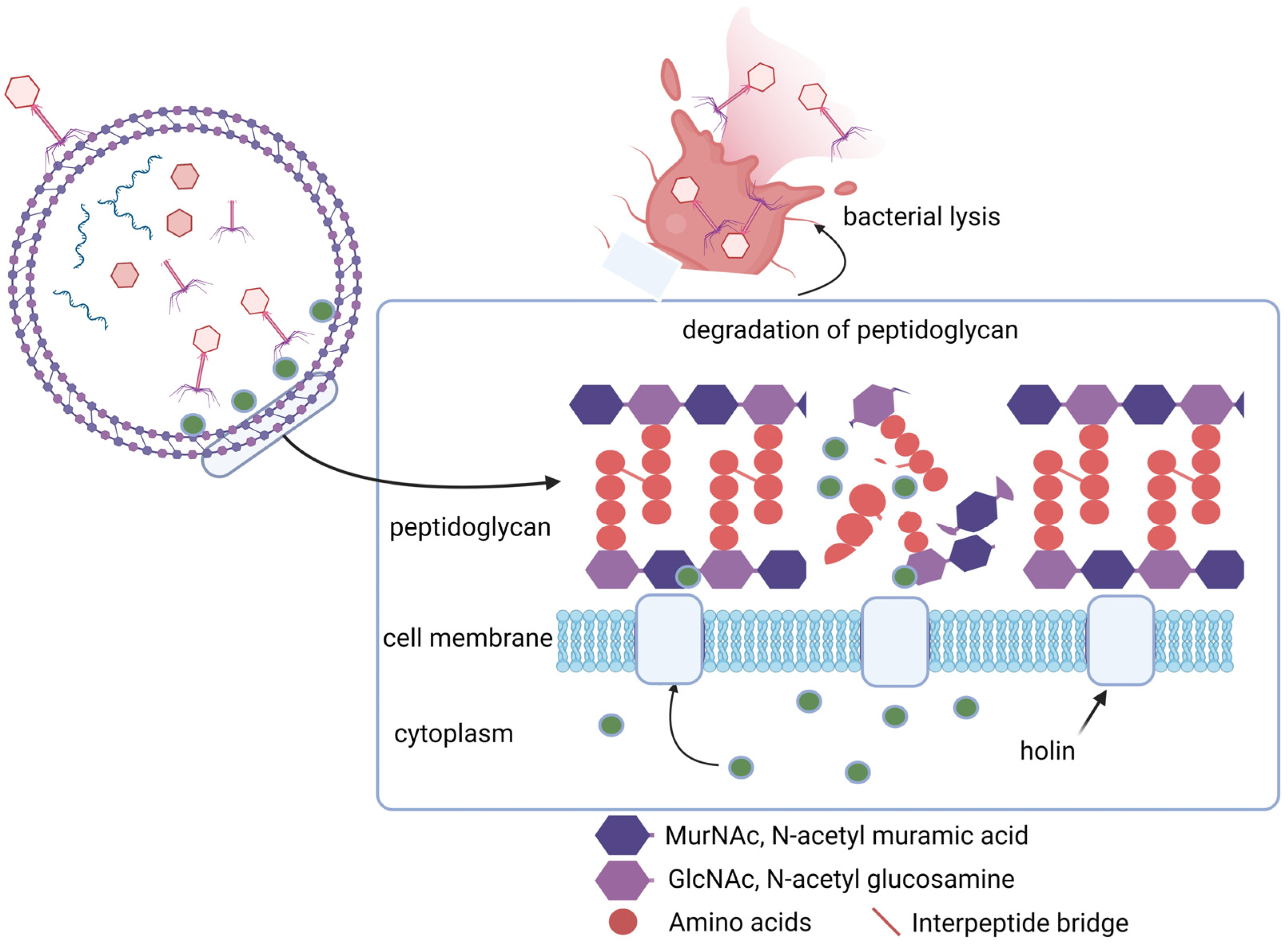
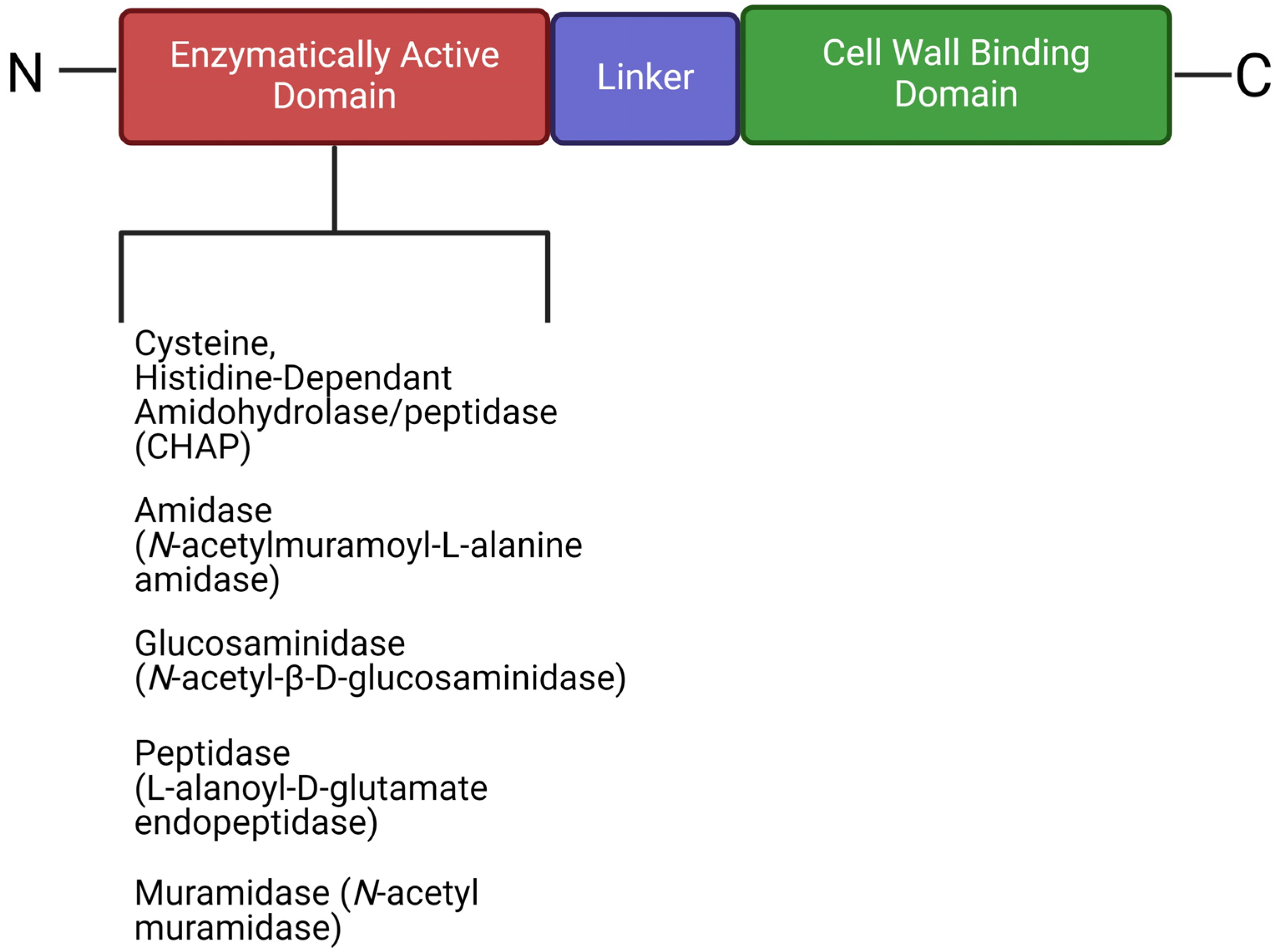
6.2. Therapeutic Potential of Phage-Derived Proteins
| Enzyme Class | Phage/Enzyme | Polymer Substrates | Targeted Genera | References |
|---|---|---|---|---|
| Hydrolases | ||||
| Sialidases | Phi92 | Polysialic acid | E. coli K1 and K92 | [151] |
| K1E | E. coli K5 | [162] | ||
| K1F | E. coli K1 | [163] | ||
| Levanase | SP10 | Levan | Bacillus spp. | [155] |
| SPG24 | ||||
| Rhamnosidase | Sf6 | O-antigen LPS | Shigella flexneri | [156,164] |
| P22 | Rhamnogalacturonan | Salmonella (ser.) Typhimurium | ||
| Cellulases | S6 | Cellulose | Erwinia amylovora | [165] |
| Peptidases | CHAPK | Pentaglycine cross-bridge peptidoglycan | Staphylococcus aureus | [166,167] |
| phiNIT1 | Poly-γ-glutamate | Bacillus spp. | ||
| Lyases | ||||
| Hyaluronidases | Prophages | Hyaluronan | Streptococcus equi | [168,169] |
| H4489A | Streptococcus pyogenes | |||
| Alginate lyases | PT 6 | Alginic acid | P. aeruginosa | [14,65] |
| AF | P. putida | |||
| Pectin/pectate lyases | ΦIPLA7 | Pectin * | Staphylococcal spp. | [170] |
| Others | ||||
| Colanidase | Phi92 | Colanic acid | E. coli | [63] |
| Lipases/triacylglycerol hydrolases | Phi3ST:2 | Carboxyl ester bonds * | Cellulophaga spp. | [171] |
| Tf | Pseudomonas spp. | |||
7. Limitations of Phage Therapy
8. Genomic Engineering of Phages
9. Diagnostic Potential of Phages and Phage-Derived Proteins
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wall, S. Prevention of Antibiotic Resistance—An Epidemiological Scoping Review to Identify Research Categories and Knowledge Gaps. Glob. Health Action 2019, 12, 1756191. [Google Scholar] [CrossRef] [PubMed]
- Forde, A.; Hill, C. Phages of Life—The Path to Pharma. Br. J. Pharmacol. 2018, 175, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Koskella, B.; Meaden, S. Understanding bacteriophage specificity in natural microbial communities. Viruses 2013, 5, 806–823. [Google Scholar] [CrossRef] [PubMed]
- Seed, K.D. Battling Phages: How Bacteria Defend against Viral Attack. PLoS Pathog. 2015, 11, e1004847. [Google Scholar] [CrossRef] [PubMed]
- Riede, I.; Eschbach, M.-L. Evidence That TraT Interacts with OmpA of Escherichia coli. FEBS Lett. 1986, 205, 241–245. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Deora, R.; Doulatov, S.R.; Gingery, M.; Eiserling, F.A.; Preston, A.; Maskell, D.J.; Simons, R.W.; Cotter, P.A.; Parkhill, J.; et al. Reverse Transcriptase-Mediated Tropism Switching in Bordetella Bacteriophage. Science (1979) 2002, 295, 2091–2094. [Google Scholar] [CrossRef] [PubMed]
- Fischetti, V.A. Bacteriophage Lysins as Effective Antibacterials. Curr. Opin. Microbiol. 2008, 11, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.-N.; Smith, D.L.; Young, R. Holins: The Protein Clocks of Bacteriophage Infections. Annu. Rev. Microbiol. 2000, 54, 799–825. [Google Scholar] [CrossRef]
- Young, R. Phage Lysis: Three Steps, Three Choices, One Outcome. J. Microbiol. 2014, 52, 243–258. [Google Scholar] [CrossRef]
- Cahill, J.; Young, R. Phage Lysis: Multiple Genes for Multiple Barriers. Adv. Virus Res. 2019, 103, 33–70. [Google Scholar] [CrossRef]
- Oliveira, H.; Azeredo, J.; Lavigne, R.; Kluskens, L.D. Bacteriophage Endolysins as a Response to Emerging Foodborne Pathogens. Trends Food Sci. Technol. 2012, 28, 103–115. [Google Scholar] [CrossRef]
- Akoh, C.C.; Lee, G.-C.; Liaw, Y.-C.; Huang, T.-H.; Shaw, J.-F. GDSL Family of Serine Esterases/Lipases. Prog. Lipid Res. 2004, 43, 534–552. [Google Scholar] [CrossRef]
- Gupta, R.; Gupta, N.; Rathi, P. Bacterial Lipases: An Overview of Production, Purification and Biochemical Properties. Appl. Microbiol. Biotechnol. 2004, 64, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Glonti, T.; Chanishvili, N.; Taylor, P.W. Bacteriophage-Derived Enzyme That Depolymerizes the Alginic Acid Capsule Associated with Cystic Fibrosis Isolates of Pseudomonas aeruginosa. J. Appl. Microbiol. 2010, 108, 695–702. [Google Scholar] [CrossRef]
- Ackermann, H.W. Phage Classification and Characterization. Meth Mol. Biol. 2009, 501, 127–140. [Google Scholar]
- Dion, M.B.; Oechslin, F.; Moineau, S. Phage Diversity, Genomics and Phylogeny. Nat. Rev. Microbiol. 2020, 18, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Zerbini, F.M.; Siddell, S.G.; Lefkowitz, E.J.; Mushegian, A.R.; Adriaenssens, E.M.; Alfenas-Zerbini, P.; Dempsey, D.M.; Dutilh, B.E.; García, M.L.; Hendrickson, R.C.; et al. Changes to virus taxonomy and the ICTV Statutes ratified by the International Committee on Taxonomy of Viruses. Arch. Virol. 2023, 168, 175. [Google Scholar] [CrossRef]
- Hatfull, G.F.; Hendrix, R.W. Bacteriophages and Their Genomes. Curr. Opin. Virol. 2011, 1, 298–303. [Google Scholar] [CrossRef]
- Liu, Y.; Demina, T.A.; Roux, S.; Aiewsakun, P.; Kazlauskas, D.; Simmonds, P.; Prangishvili, D.; Oksanen, H.M.; Krupovic, M. Diversity, Taxonomy, and Evolution of Archaeal Viruses of the Class Caudoviricetes. PLoS Biol. 2021, 19, e3001442. [Google Scholar] [CrossRef]
- Brüssow, H.; Hendrix, R.W. Phage Genomics. Cell 2002, 108, 13–16. [Google Scholar] [CrossRef]
- Santamaria, R.M.; Innes, N.P.T.; Machiulskiene, V.; Evans, D.J.P.; Splieth, C.H. Caries Management Strategies for Primary Molars: 1-Yr Randomized Control Trial Results. J. Dent. Res. 2014, 93, 1062–1069. [Google Scholar] [CrossRef]
- Pantůcek, R.; Rosypalová, A.; Doskar, J.; Kailerová, J.; Růzicková, V.; Borecká, P.; Snopková, S.; Horváth, R.; Götz, F.; Rosypal, S. The Polyvalent Staphylococcal Phage Phi 812: Its Host-Range Mutants and Related Phages. Virology 1998, 246, 241–252. [Google Scholar] [CrossRef][Green Version]
- Cazares, D.; Cazares, A.; Figueroa, W.; Guarneros, G.; Edwards, R.A.; Vinuesa, P. A Novel Group of Promiscuous Podophages Infecting Diverse Gammaproteobacteria from River Communities Exhibits Dynamic Intergenus Host Adaptation. mSystems 2021, 6, e00773-20. [Google Scholar] [CrossRef]
- Figueroa-Bossi, N.; Uzzau, S.; Maloriol, D.; Bossi, L. Variable Assortment of Prophages Provides a Transferable Repertoire of Pathogenic Determinants in Salmonella. Mol. Microbiol. 2001, 39, 260–272. [Google Scholar] [CrossRef]
- McAllister, W.T.; Barrett, C.L. Roles of the Early Genes of Bacteriophage T7 in Shutoff of Host Macromolecular Synthesis. J. Virol. 1977, 23, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Labrie, S.J.; Samson, J.E.; Moineau, S. Bacteriophage Resistance Mechanisms. Nat. Rev. Microbiol. 2010, 8, 317–327. [Google Scholar] [CrossRef]
- Scanlan, P.D.; Buckling, A.; Hall, A.R. Experimental Evolution and Bacterial Resistance: (Co)Evolutionary Costs and Trade-Offs as Opportunities in Phage Therapy Research. Bacteriophage 2015, 5, e1050153. [Google Scholar] [CrossRef]
- Bertozzi Silva, J.; Storms, Z.; Sauvageau, D. Host Receptors for Bacteriophage Adsorption. FEMS Microbiol. Lett. 2016, 363, fnw002. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, S.; Tsurumi, S.; Imai, N. Crossover Behavior for Brownian Motion. J. Chem. Phys. 1986, 84, 539–540. [Google Scholar] [CrossRef]
- Heller, K.J.; Schwarz, H.; Tubingen, U.; Mikrobiologie, L.I. Irreversible Binding to the Receptor of Bacteriophages T5 and BF23 Does Not Occur with the Tip of the Tail. J. Bacteriol. 1985, 162, 621–625. [Google Scholar] [CrossRef] [PubMed]
- Dunne, M.; Hupfeld, M.; Klumpp, J.; Loessner, M.J. Molecular Basis of Bacterial Host Interactions by Gram-Positive Targeting Bacteriophages. Viruses 2018, 10, 397. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Kim, S.; Park, B.; Ryu, S. Core Lipopolysaccharide-Specific Phage SSU5 as an Auxiliary Component of a Phage Cocktail for Salmonella Biocontrol. Appl. Environ. Microbiol. 2014, 80, 1026. [Google Scholar] [CrossRef] [PubMed]
- Sandulache, R.; Prehm, P.; Kamp, D. Cell Wall Receptor for Bacteriophage Mu G(+). J. Bacteriol. 1984, 160, 299–303. [Google Scholar] [CrossRef] [PubMed]
- Munsch-Alatossava, P.; Alatossava, T. The Extracellular Phage-Host Interactions Involved in the Bacteriophage LL-H Infection of Lactobacillus delbrueckii ssp. Lactis ATCC 15808. Front. Microbiol. 2013, 4, 408. [Google Scholar] [CrossRef] [PubMed]
- Wolin, M.J.; Douglas, L.J. Cell Wall Polymers and Phage Lysis of Lactobacillus plantarum. Biochemistry 1971, 10, 1551–1555. [Google Scholar] [CrossRef] [PubMed]
- Wendlinger, G.; Loessner, M.J.; Scherer, S. Bacteriophage Receptors on Listeria Monocytogenes Cells Are the N-Acetylglucosamine and Rhamnose Substituents of Teichoic Acids or the Peptidoglycan Itself. Microbiology 1996, 142, 985–992. [Google Scholar] [CrossRef]
- Moak, M.; Molineux, I.J. Peptidoglycan Hydrolytic Activities Associated with Bacteriophage Virions. Mol. Microbiol. 2004, 51, 1169–1183. [Google Scholar] [CrossRef]
- Barbirz, S.; Müller, J.J.; Uetrecht, C.; Clark, A.J.; Heinemann, U.; Seckler, R. Crystal Structure of Escherichia coli Phage HK620 Tailspike: Podoviral Tailspike Endoglycosidase Modules Are Evolutionarily Related. Mol. Microbiol. 2008, 69, 303–316. [Google Scholar] [CrossRef]
- Baptista, C.; Santos, M.A.; São-José, C. Phage SPP1 Reversible Adsorption to Bacillus subtilis Cell Wall Teichoic Acids Accelerates Virus Recognition of Membrane Receptor YueB. J. Bacteriol. 2008, 190, 4989–4996. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xiang, Y. Membrane Penetration by Bacterial Viruses. J. Virol. 2017, 91, e00162-17. [Google Scholar] [CrossRef] [PubMed]
- Olsen, R.H.; Siak, J.-S.; Gray, R.H. Characteristics of PRD1, a Plasmid-Dependent Broad Host Range DNA Bacteriophage. J. Virol. 1974, 14, 689–699. [Google Scholar] [CrossRef]
- Ojala, V.; Laitalainen, J.; Jalasvuori, M. Fight Evolution with Evolution: Plasmid-dependent Phages with a Wide Host Range Prevent the Spread of Antibiotic Resistance. Evol. Appl. 2013, 6, 925–932. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, R.A.; Pickard, D.; Dougan, S.; Goulding, D.; Cormie, C.; Hardy, J.; Li, F.; Grinter, R.; Harcourt, K.; Yu, L.; et al. The Flagellotropic Bacteriophage YSD1 Targets Salmonella Typhi with a Chi-like Protein Tail Fibre. Mol. Microbiol. 2019, 112, 1831–1846. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.M.; Dunstan, R.A.; Grinter, R.; Belousoff, M.J.; Wang, J.; Pickard, D.; Venugopal, H.; Dougan, G.; Lithgow, T.; Coulibaly, F. The Architecture and Stabilisation of Flagellotropic Tailed Bacteriophages. Nat. Commun. 2020, 11, 3748. [Google Scholar] [CrossRef] [PubMed]
- Majkowska-Skrobek, G.; Łątka, A.; Berisio, R.; Maciejewska, B.; Squeglia, F.; Romano, M.; Lavigne, R.; Struve, C.; Drulis-Kawa, Z. Capsule-Targeting Depolymerase, Derived from Klebsiella KP36 Phage, as a Tool for the Development of Anti-Virulent Strategy. Viruses 2016, 8, 324. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Yang, X.; Huang, J.; Zhu, X.; Han, G.; Wan, Y.; Xu, Y.; Luan, G.; Jia, X. Phage Selective Pressure Reduces Virulence of Hypervirulent Klebsiella pneumoniae Through Mutation of the Wzc Gene. Front. Microbiol. 2021, 12, 739319. [Google Scholar] [CrossRef] [PubMed]
- Tzagoloff, H.; Pratt, D. The Initial Steps in Infection with Coliphage M13. Virology 1964, 24, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Roncero, C.; Darzins, A.; Casadaban, M.J. Pseudomonas aeruginosa Transposable Bacteriophages D3112 and B3 Require Pili and Surface Growth for Adsorption. J. Bacteriol. 1990, 172, 1899–1904. [Google Scholar] [CrossRef]
- Manchak, J.; Anthony, G.; Frost, L.S. Mutational Analysis of F-pilin Reveals Domains for Pilus Assembly, Phage Infection and DNA Transfer. Mol. Microbiol. 2002, 43, 195–205. [Google Scholar] [CrossRef]
- Jalasvuori, M.; Friman, V.-P.; Nieminen, A.; Bamford, J.K.H.; Buckling, A. Bacteriophage Selection against a Plasmid-Encoded Sex Apparatus Leads to the Loss of Antibiotic-Resistance Plasmids. Biol. Lett. 2011, 7, 902–905. [Google Scholar] [CrossRef]
- Nováček, J.; Šiborová, M.; Benešík, M.; Pantůček, R.; Doškař, J.; Plevka, P. Structure and genome release of Twort-like Myoviridae Phage with a Double-layered Baseplate. Proc. Natl. Acad. Sci. USA 2016, 113, 9351–9356. [Google Scholar] [CrossRef] [PubMed]
- Mahony, J.; Alqarni, M.; Stockdale, S.; Spinelli, S.; Feyereisen, M.; Cambillau, C.; van Sinderen, D. Functional and Structural Dissection of the Tape Measure Protein of Lactococcal Phage TP901-1. Sci. Rep. 2016, 6, 36667. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, H.; São-José, C.; Azeredo, J. Phage-Derived Peptidoglycan Degrading Enzymes: Challenges and Future Prospects for In Vivo Therapy. Viruses 2018, 10, 292. [Google Scholar] [CrossRef] [PubMed]
- Boulanger, P.; Jacquot, P.; Plançon, L.; Chami, M.; Engel, A.; Parquet, C.; Herbeuval, C.; Letellier, L. Phage T5 Straight Tail Fiber Is a Multifunctional Protein Acting as a Tape Measure and Carrying Fusogenic and Muralytic Activities. J. Biol. Chem. 2008, 283, 13556–13564. [Google Scholar] [CrossRef] [PubMed]
- Fokine, A.; Rossmann, M.G. Molecular Architecture of Tailed Double-Stranded DNA Phages. Bacteriophage 2014, 4, e28281. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, R.; Bradley, R.; Paranchych, W. The Effect of M13 Phage Infection upon the F Pili of E. coli. Virology 1973, 54, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Abrescia, N.; Cockburn, J.; Grimes, J.; Sutton, G.C.; Diprose, J.M.; Butcher, S.J.; Fuller, S.D.; San Martín, C.; Burnett, R.M.; Stuart, D.I.; et al. Insights into Assembly from Structural Analysis of Bacteriophage PRD1. Nature 2004, 432, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Grahn, A.M.; Daugelavičius, R.; Bamford, D.H. Sequential Model of Phage PRD1 DNA Delivery: Active Involvement of the Viral Membrane. Mol. Microbiol. 2002, 46, 1199–1209. [Google Scholar] [CrossRef]
- Daugelavicius, R.; Cvirkaite, V.; Gaidelytė, A.; Bakiene, E.; Gabrenaite-Verkhovskaya, R.; Bamford, D.H. Penetration of Enveloped Double-Stranded RNA Bacteriophages Φ13 and Φ6 into Pseudomonas syringae Cells. J. Virol. 2005, 79, 5017–5026. [Google Scholar] [CrossRef]
- Cvirkaitė-Krupovič, V.; Poranen, M.M.; Bamford, D.H. Phospholipids Act as Secondary Receptor during the Entry of the Enveloped, Double-Stranded RNA Bacteriophage Φ6. J. General. Virol. 2010, 91, 2116–2120. [Google Scholar] [CrossRef]
- Meyer, J.R.; Dobias, D.T.; Weitz, J.S.; Barrick, J.E.; Quick, R.T.; Lenski, R.E. Repeatability and Contingency in the Evolution of a Key Innovation in Phage Lambda. Science 2012, 335, 428–432. [Google Scholar] [CrossRef]
- Scholl, D.; Adhya, S.; Merril, C. Escherichia coli K1’s Capsule Is a Barrier to Bacteriophage T7. Appl. Environ. Microbiol. 2005, 71, 4872–4874. [Google Scholar] [CrossRef]
- Schwarzer, D.; Buettner, F.F.R.; Browning, C.; Nazarov, S.; Rabsch, W.; Bethe, A.; Oberbeck, A.; Bowman, V.D.; Stummeyer, K.; Mühlenhoff, M.; et al. A Multivalent Adsorption Apparatus Explains the Broad Host Range of Phage Phi92: A Comprehensive Genomic and Structural Analysis. J. Virol. 2012, 86, 10384. [Google Scholar] [CrossRef]
- Patro, L.P.P.; Rathinavelan, T. Targeting the Sugary Armor of Klebsiella Species. Front. Cell Infect. Microbiol. 2019, 9. [Google Scholar] [CrossRef]
- Cornelissen, A.; Ceyssens, P.-J.; Krylov, V.N.; Noben, J.-P.; Volckaert, G.; Lavigne, R. Identification of EPS-Degrading Activity within the Tail Spikes of the Novel Pseudomonas putida Phage AF. Virology 2012, 434, 251–256. [Google Scholar] [CrossRef]
- Yan, G.; Yang, R.; Fan, K.; Dong, H.; Gao, C.; Wang, S.; Yu, L.; Cheng, Z.; Lei, L. External Lysis of Escherichia coli by a Bacteriophage Endolysin Modified with Hydrophobic Amino Acids. AMB Express 2019, 9, 106. [Google Scholar] [CrossRef]
- Samson, J.E.; Magadán, A.H.; Sabri, M.; Moineau, S. Revenge of the Phages: Defeating Bacterial Defences. Nat. Rev. Microbiol. 2013, 11, 675–687. [Google Scholar] [CrossRef]
- Holst Sørensen, M.C.; van Alphen, L.B.; Fodor, C.; Crowley, S.M.; Christensen, B.B.; Szymanski, C.M.; Brøndsted, L. Phase Variable Expression of Capsular Polysaccharide Modifications Allows Campylobacter jejuni to Avoid Bacteriophage Infection in Chickens. Front. Cell Infect. Microbiol. 2012, 2, 11. [Google Scholar] [CrossRef]
- Manning, A.J.; Kuehn, M.J. Contribution of Bacterial Outer Membrane Vesicles to Innate Bacterial Defense. BMC Microbiol. 2011, 11, 258. [Google Scholar] [CrossRef]
- Bernheim, A.; Sorek, R. The Pan-Immune System of Bacteria: Antiviral Defence as a Community Resource. Nat. Rev. Microbiol. 2020, 18, 113–119. [Google Scholar] [CrossRef]
- Murray, N.E. Immigration Control of DNA in Bacteria: Self versus Non-Self. Microbiology 2002, 148, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Auer, B.; Schweiger, M. Evidence That Escherichia coli Virus T1 Induces a DNA Methyltransferase. J. Virol. 1984, 49, 588–590. [Google Scholar] [CrossRef] [PubMed]
- Walkinshaw, M.D.; Taylor, P.; Sturrock, S.S.; Atanasiu, C.; Berge, T.; Henderson, R.M.; Edwardson, J.M.; Dryden, D.T.F. Structure of Ocr from Bacteriophage T7, a Protein That Mimics B-Form DNA. Mol. Cell 2002, 9, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Dillingham, M.S.; Kowalczykowski, S.C. RecBCD Enzyme and the Repair of Double-Stranded DNA Breaks. Microbiol. Mol. Biol. Rev. 2008, 72, 642–671. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.C. The λ Gam Protein Inhibits RecBCD Binding to DsDNA Ends. J. Mol. Biol. 2007, 371, 19–24. [Google Scholar] [CrossRef]
- d’Adda di Fagagna, F.; Weller, G.R.; Doherty, A.J.; Jackson, S.P. The Gam Protein of Bacteriophage Mu Is an Orthologue of Eukaryotic Ku. EMBO Rep. 2003, 4, 47–52. [Google Scholar] [CrossRef]
- Barrangou, R.; Marraffini, L.A. CRISPR-Cas Systems: Prokaryotes Upgrade to Adaptive Immunity. Mol. Cell 2014, 54, 234–244. [Google Scholar] [CrossRef]
- Deveau, H.; Barrangou, R.; Garneau, J.E.; Labonté, J.; Fremaux, C.; Boyaval, P.; Romero, D.A.; Horvath, P.; Moineau, S. Phage Response to CRISPR-Encoded Resistance in Streptococcus thermophilus. J. Bacteriol. 2008, 190, 1390–1400. [Google Scholar] [CrossRef]
- Seed, K.D.; Lazinski, D.W.; Calderwood, S.B.; Camilli, A. A Bacteriophage Encodes Its Own CRISPR/Cas Adaptive Response to Evade Host Innate Immunity. Nature 2013, 494, 489–491. [Google Scholar] [CrossRef]
- Pawluk, A.; Bondy-Denomy, J.; Cheung, V.H.W.; Maxwell, K.L.; Davidson, A.R. A New Group of Phage Anti-CRISPR Genes Inhibits the Type I-E CRISPR-Cas System of Pseudomonas aeruginosa. mBio 2014, 5, e00896. [Google Scholar] [CrossRef]
- Bondy-Denomy, J.; Pawluk, A.; Maxwell, K.L.; Davidson, A.R. Bacteriophage Genes That Inactivate the CRISPR/Cas Bacterial Immune System. Nature 2013, 493, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Durmaz, E.; Klaenhammer, T.R. Abortive Phage Resistance Mechanism AbiZ Speeds the Lysis Clock To Cause Premature Lysis of Phage-Infected Lactococcus lactis. J. Bacteriol. 2007, 189, 1417–1425. [Google Scholar] [CrossRef]
- Shinedling, S.; Parma, D.; Gold, L. Wild-Type Bacteriophage T4 Is Restricted by the Lambda Rex Genes. J. Virol. 1987, 61, 3790–3794. [Google Scholar] [CrossRef]
- Bingham, R.; Ekunwe, S.I.N.; Falk, S.; Snyder, L.; Kleanthous, C. The Major Head Protein of Bacteriophage T4 Binds Specifically to Elongation Factor Tu. J. Biol. Chem. 2000, 275, 23219–23226. [Google Scholar] [CrossRef]
- Leitner, L.; Ujmajuridze, A.; Chanishvili, N.; Goderdzishvili, M.; Chkonia, I.; Rigvava, S.; Chkhotua, A.; Changashvili, G.; McCallin, S.; Schneider, M.P.; et al. Intravesical Bacteriophages for Treating Urinary Tract Infections in Patients Undergoing Transurethral Resection of the Prostate: A Randomised, Placebo-Controlled, Double-Blind Clinical Trial. Lancet Infect. Dis. 2021, 21, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Schooley, R.T.; Biswas, B.; Gill, J.J.; Hernandez-Morales, A.; Lancaster, J.; Lessor, L.; Barr, J.J.; Reed, S.L.; Rohwer, F.; Benler, S.; et al. Development and Use of Personalized Bacteriophage-Based Therapeutic Cocktails To Treat a Patient with a Disseminated Resistant Acinetobacter baumannii Infection. Antimicrob. Agents Chemother. 2017, 61, e00954-17. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.; Turner, P.E.; Kim, S.; Mojibian, H.R.; Elefteriades, J.A.; Narayan, D. Phage Treatment of an Aortic Graft Infected with Pseudomonas aeruginosa. Evol. Med. Public. Health 2018, 2018, 60–66. [Google Scholar] [CrossRef]
- Chan, B.K.; Sistrom, M.; Wertz, J.E.; Kortright, K.E.; Narayan, D.; Turner, P.E. Phage Selection Restores Antibiotic Sensitivity in MDR Pseudomonas aeruginosa. Sci. Rep. 2016, 6, 26717. [Google Scholar] [CrossRef]
- Dedrick, R.M.; Guerrero-Bustamante, C.A.; Garlena, R.A.; Russell, D.A.; Ford, K.; Harris, K.; Gilmour, K.C.; Soothill, J.; Jacobs-Sera, D.; Schooley, R.T.; et al. Engineered Bacteriophages for Treatment of a Patient with a Disseminated Drug-Resistant Mycobacterium abscessus. Nat. Med. 2019, 25, 730–733. [Google Scholar] [CrossRef]
- Uyttebroek, S.; Chen, B.; Onsea, J.; Ruythooren, F.; Debaveye, Y.; Devolder, D.; Spriet, I.; Depypere, M.; Wagemans, J.; Lavigne, R.; et al. Safety and Efficacy of Phage Therapy in Difficult-to-Treat Infections: A Systematic Review. Lancet Infect. Dis. 2022, 22, e208–e220. [Google Scholar] [CrossRef]
- Hibstu, Z.; Belew, H.; Akelew, Y.; Mengist, H.M. Phage Therapy: A Different Approach to Fight Bacterial Infections. Biologics 2022, 16, 173–186. [Google Scholar] [CrossRef]
- O’Shea, Y.; Boyd, E.F. Mobilization of the Vibrio Pathogenicity Island between Vibrio Cholerae Isolates Mediated by CP-T1 Generalized Transduction. FEMS Microbiol. Lett. 2002, 214, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Brabban, A.D.; Hite, E.; Callaway, T.R. Evolution of Foodborne Pathogens via Temperate Bacteriophage-Mediated Gene Transfer. Foodborne Pathog. Dis. 2005, 2, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Maiques, E.; Úbeda, C.; Tormo, M.A.; Ferrer, M.D.; Lasa, I.; Novick, R.P.; Penadès, J.R. Role of Staphylococcal Phage and SaPI Integrase in Intra- and Interspecies SaPI Transfer. J. Bacteriol. 2007, 189, 5608–5616. [Google Scholar] [CrossRef] [PubMed]
- Parsley, L.C.; Consuegra, E.J.; Kakirde, K.S.; Land, A.M.; Harper, W.F.; Liles, M.R. Identification of Diverse Antimicrobial Resistance Determinants Carried on Bacterial, Plasmid, or Viral Metagenomes from an Activated Sludge Microbial Assemblage. Appl. Environ. Microbiol. 2010, 76, 3753–3757. [Google Scholar] [CrossRef] [PubMed]
- Marti, E.; Variatza, E.; Balcázar, J.L. Bacteriophages as a Reservoir of Extended-Spectrum β-Lactamase and Fluoroquinolone Resistance Genes in the Environment. Clin. Microbiol. Infect. 2014, 20, O456–O459. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mozaz, S.; Chamorro, S.; Marti, E.; Huerta, B.; Gros, M.; Sànchez-Melsió, A.; Borrego, C.M.; Barceló, D.; Balcázar, J.L. Occurrence of Antibiotics and Antibiotic Resistance Genes in Hospital and Urban Wastewaters and Their Impact on the Receiving River. Water Res. 2015, 69, 234–242. [Google Scholar] [CrossRef] [PubMed]
- Subirats, J.; Sànchez-Melsió, A.; Borrego, C.M.; Balcázar, J.L.; Simonet, P. Metagenomic Analysis Reveals That Bacteriophages Are Reservoirs of Antibiotic Resistance Genes. Int. J. Antimicrob. Agents 2016, 48, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Marti, R.; Zurfluh, K.; Hagens, S.; Pianezzi, J.; Klumpp, J.; Loessner, M.J. Long Tail Fibres of the Novel Broad-Host-Range T-Even Bacteriophage S16 Specifically Recognize Salmonella OmpC. Mol. Microbiol. 2013, 87, 818–834. [Google Scholar] [CrossRef]
- Quirós, P.; Colomer-Lluch, M.; Martínez-Castillo, A.; Miró, E.; Argente, M.; Jofre, J.; Navarro, F.; Muniesa, M. Antibiotic Resistance Genes in the Bacteriophage DNA Fraction of Human Fecal Samples. Antimicrob. Agents Chemother. 2014, 58, 606–609. [Google Scholar] [CrossRef]
- Torres-Barceló, C. The Disparate Effects of Bacteriophages on Antibiotic-Resistant Bacteria. Emerg. Microbes Infect. 2018, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Colomer-Lluch, M.; Jofre, J.; Muniesa, M. Quinolone Resistance Genes (QnrA and QnrS) in Bacteriophage Particles from Wastewater Samples and the Effect of Inducing Agents on Packaged Antibiotic Resistance Genes. J. Antimicrob. Chemother. 2014, 69, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Fancello, L.; Desnues, C.; Raoult, D.; Rolain, J.M. Bacteriophages and Diffusion of Genes Encoding Antimicrobial Resistance in Cystic Fibrosis Sputum Microbiota. J. Antimicrob. Chemother. 2011, 66, 2448–2454. [Google Scholar] [CrossRef] [PubMed]
- Enault, F.; Briet, A.; Bouteille, L.; Roux, S.; Sullivan, M.B.; Petit, M.-A. Phages Rarely Encode Antibiotic Resistance Genes: A Cautionary Tale for Virome Analyses. ISME J. 2017, 11, 237–247. [Google Scholar] [CrossRef] [PubMed]
- Essoh, C.; Blouin, Y.; Loukou, G.; Cablanmian, A.; Lathro, S.; Kutter, E.; Thien, H.V.; Vergnaud, G.; Pourcel, C. The Susceptibility of Pseudomonas aeruginosa Strains from Cystic Fibrosis Patients to Bacteriophages. PLoS ONE 2013, 8, e60575. [Google Scholar] [CrossRef]
- Abedon, S.T.; Kuhl, S.J.; Blasdel, B.G.; Kutter, E.M. Phage Treatment of Human Infections. Bacteriophage 2011, 1, 66–85. [Google Scholar] [CrossRef]
- Cislo, M.; Dabrowski, M.; Weber-Dabrowska, B.; Woyton, A. Bacteriophage Treatment of Suppurative Skin Infections. Arch. Immunol. Ther. Exp. (Warsz.) 1987, 35, 175–183. [Google Scholar]
- Gupta, P.; Singh, H.S.; Shukla, V.K.; Nath, G.; Bhartiya, S.K. Bacteriophage Therapy of Chronic Nonhealing Wound: Clinical Study. Int. J. Low. Extrem. Wounds 2019, 18, 171–175. [Google Scholar] [CrossRef]
- Patel, D.R.; Bhartiya, S.K.; Kumar, R.; Shukla, V.K.; Nath, G. Use of Customized Bacteriophages in the Treatment of Chronic Nonhealing Wounds: A prospective study. Int. J. Low. Extrem. Wounds 2021, 20, 37–46. [Google Scholar]
- Ioseliani, G.D.; Meladze, G.D.; Chkhetiia, N.S.; Mebuke, M.G.; Kiknadze, N.I. Use of Bacteriophage and Antibiotics for Prevention of Acute Postoperative Empyema in Chronic Suppurative Lung Diseases. Grud. Khir 1980, 63–67. [Google Scholar]
- Aslam, S.; Courtwright, A.M.; Koval, C.; Lehman, S.M.; Morales, S.; Furr, C.L.L.; Rosas, F.; Brownstein, M.J.; Fackler, J.R.; Sisson, B.M.; et al. Early Clinical Experience of Bacteriophage Therapy in 3 Lung Transplant Recipients. Am. J. Transplant. 2019, 19, 2631–2639. [Google Scholar]
- Aslam, S.; Lampley, E.; Wooten, D.; Karris, M.; Benson, C.; Strathdee, S.; Schooley, R.T. Lessons Learned from the First 10 Consecutive Cases of Intravenous Bacteriophage Therapy to Treat Multidrug-resistant Bacterial Infections at a Single Center in the United States. Open Forum Infect. Dis. 2020, 7, ofaa389. [Google Scholar]
- Kochetkova, V.A.; Mamontov, A.S.; Moskovtseva, R.L.; Erastova, E.I.; Trofimov, E.I.; Popov, M.I.; Dzhubalieva, S.K. Phagotherapy of Postoperative Suppurative-Inflammatory Complications in Patients with Neoplasms. Sov. Med. 1989, 23–26. [Google Scholar]
- Kwarciński, W.; Lazarkiewicz, B.; Weber-Dabrowska, B.; Rudnicki, J.; Kamiński, K.; Sciebura, M. Bacteriophage Therapy in the Treatment of Recurrent Subphrenic and Subhepatic Abscess with Jejunal Fistula after Stomach Resection. Pol. Tyg. Lek. 1994, 49, 535. [Google Scholar] [PubMed]
- Perepanova, T.S.; Darbeeva, O.S.; Kotliarova, G.A.; Kondrat’eva, E.M.; Maiskaia, L.M.; Malysheva, V.F.; Baiguzina, F.A.; Grishkova, N.V. The Efficacy of Bacteriophage Preparations in Treating Inflammatory Urologic Diseases. Urol. Nefrol. (Mosk.) 1995, 5, 14–17. [Google Scholar]
- Sakandelidze, V.M. The Combined Use of Specific Phages and Antibiotics in Different Infectious Allergoses. Vrach. Delo 1991, 3, 60–63. [Google Scholar]
- Strój, L.; Weber-Dabrowska, B.; Partyka, K.; Mulczyk, M.; Wójcik, M. Successful Treatment with Bacteriophage in Purulent Cerebrospinal Meningitis in a Newborn. Neurol. Neurochir. Pol. 1999, 33, 693–698. [Google Scholar] [PubMed]
- Sarker, S.A.; Sultana, S.; Reuteler, G.; Moine, D.; Descombes, P.; Charton, F.; Bourdin, G.; McCallin, S.; Ngom-Bru, C.; Neville, T.; et al. Oral Phage Therapy of Acute Bacterial Diarrhea with Two Coliphage Preparations: A Randomized Trial in Children From Bangladesh. EBioMedicine 2016, 4, 124–137. [Google Scholar] [CrossRef] [PubMed]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; le Floch, R.; et al. Efficacy and Tolerability of a Cocktail of Bacteriophages to Treat Burn Wounds Infected by Pseudomonas aeruginosa (PhagoBurn): A Randomised, Controlled, Double-Blind Phase 1/2 Trial. Lancet Infect. Dis. 2019, 19, 35–45. [Google Scholar] [CrossRef]
- NIH, U.S. National Library of Medicine. Bacteriophage Therapy in First Time Chronic Prosthetic Joint Infections. Available online: https://clinicaltrials.gov/study/NCT05269121 (accessed on 28 October 2023).
- Long Island University, Clinical Drug Experience Knowledgebase. Assessing the Efficacy of Anti-Staphylococcal Phages in the Management of Infected Foot Ulcers in Diabetes. NCT04289948. Available online: https://www.cdek.liu.edu/trial/NCT04289948/ (accessed on 28 October 2023).
- National Library of Medicine. BacterioPHAGE for Gastrointestinal Health 2 Study. NCT04511221. Available online: https://clinicaltrials.gov/study/NCT04511221 (accessed on 28 October 2023).
- National Library of Medicine. Existence in the Human Digestive Flora of Phages Able to Prevent the Acquisition of Multiresistant Enterobacteria (PHAGO-BMR). NCT03231267. Available online: https://clinicaltrials.gov/study/NCT03231267 (accessed on 28 October 2023).
- Sybesma, W.; Rohde, C.; Bardy, P.; Pirnay, J.-P.; Cooper, I.; Caplin, J.; Chanishvili, N.; Coffey, A.; De Vos, D.; Scholz, A.; et al. Silk Route to the Acceptance and Re-Implementation of Bacteriophage Therapy—Part II. Antibiotics 2018, 7, 35. [Google Scholar] [CrossRef]
- NIH U.S. National Library of Medicine. Bacteriophage Effects on Pseudomonas aeruginosa (MUCOPHAGES). NCT01818206. Available online: https://classic.clinicaltrials.gov/ct2/show/NCT01818206 (accessed on 28 October 2023).
- NIH U.S. National Library of Medicine. PHAGE Study: Bacteriophages as Novel Prebiotics. NCT03269617. Available online: https://clinicaltrials.gov/study/NCT03269617 (accessed on 28 October 2023).
- NIH U.S. National Library of Medicine. A Prospective, Randomized, Double-Blind Controlled Study of WPP-201 for the Safety and Efficacy of Treatment of Venous Leg Ulcers (WPP-201). NCT00663091. Available online: https://clinicaltrials.gov/study/NCT00663091 (accessed on 28 October 2023).
- WCG. Center Watch Safety Tolerability and PK of LBP-EC01 in Patients with Lower Urinary Tract Colonization Caused by E. coli. Available online: https://www.centerwatch.com/clinical-trials/listings/234324/safety-tolerability-and-pk-of-lbp-ec01-in-patients-with-lower-urinary-tract-colonization-caused-by-e-coli/ (accessed on 28 October 2023).
- Lenneman, B.R.; Fernbach, J.; Loessner, M.J.; Lu, T.K.; Kilcher, S. Enhancing Phage Therapy through Synthetic Biology and Genome Engineering. Curr. Opin. Biotechnol. 2021, 68, 151–159. [Google Scholar] [CrossRef] [PubMed]
- NIH U.S. National Library of Medicine. Ascending Dose Study of the Safety of AB-SA01 When Topically Applied to Intact Skin of Healthy Adults. NCT02757755. Available online: https://clinicaltrials.gov/study/NCT02757755 (accessed on 28 October 2023).
- Fadlallah, A.; Chelala, E.; Legeais, J.M. Corneal Infection Therapy with Topical Bacteriophage Administration. Open J. Ophthalmol. 2015, 9, 167. [Google Scholar]
- Duplessis, C.A.; Stockelman, M.; Hamilton, T.; Merril, G.; Brownstein, M.; Bishop-Lilly, K.; Schooley, R.; Henry, M.; Horne, B.; Sisson, B.M.; et al. A Case Series of Emergency Investigational New Drug Applications for Bacteriophages Treating Recalcitrant Multi-Drug Resistant Bacterial Infections: Confirmed Safety and a Signal of Efficacy. J. Intensive Crit. Care 2019, 5. [Google Scholar]
- Nir-Paz, R.; Gelman, D.; Khouri, A.; Sisson, B.M.; Fackler, J.; Alkalay-Oren, S.; Khalifa, L.; Rimon, A.; Yerushalmy, O.; Bader, R.; et al. Successful Treatment of Antibiotic-Resistant, Poly-Microbial Bone Infection with Bacteriophages and Antibiotics Combination. Clin. Infect. Dis. 2019, 69, 2015–2018. [Google Scholar] [CrossRef]
- Gainey, A.B.; Burch, A.K.; Brownstein, M.J.; Brown, D.E.; Fackler, J.; Horne, B.; Biswas, B.; Bivens, B.N.; Malagon, F.; Daniels, R. Combining Bacteriophages with Cefiderocol and Meropenem/Vaborbactam to Treat a Pan-Drug Resistant Achromobacter Species Infection in a Pediatric Cystic Fibrosis Patient. Pediatr. Pulmonol. 2020, 55, 2990–2994. [Google Scholar] [CrossRef]
- Oechslin, F. Resistance Development to Bacteriophages Occurring during Bacteriophage Therapy. Viruses 2018, 10, 351. [Google Scholar] [CrossRef]
- LeBlanc, L.; Nezami, S.; Yost, D.; Tsourkas, P.; Amy, P.S. Isolation and Characterization of a Novel Phage Lysin Active against Paenibacillus larvae, a Honeybee Pathogen. Bacteriophage 2015, 5, e1080787. [Google Scholar] [CrossRef]
- Bateman, A.; Rawlings, N.D. The CHAP Domain: A Large Family of Amidases Including GSP Amidase and Peptidoglycan Hydrolases. Trends Biochem. Sci. 2003, 28, 234–237. [Google Scholar] [CrossRef]
- Oliveira, H.; Boas, D.V.; Mesnage, S.; Kluskens, L.D.; Lavigne, R.; Sillankorva, S.; Secundo, F.; Azeredo, J. Structural and Enzymatic Characterization of ABgp46, a Novel Phage Endolysin with Broad Anti-Gram-Negative Bacterial Activity. Front. Microbiol. 2016, 7, 184317. [Google Scholar] [CrossRef]
- Lood, R.; Winer, B.Y.; Pelzek, A.J.; Diez-Martinez, R.; Thandar, M.; Euler, C.W.; Schuch, R.; Fischetti, V.A. Novel Phage Lysin Capable of Killing the Multidrug-Resistant Gram-Negative Bacterium Acinetobacter baumannii in a Mouse Bacteremia Model. Antimicrob. Agents Chemother. 2015, 59, 1983–1991. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.A.; Shin, H.; Heu, S.; Ryu, S. Exogenous Lytic Activity of SPN9CC Endolysin against Gram-Negative Bacteria. J. Microbiol. Biotechnol. 2014, 24, 803–811. [Google Scholar] [CrossRef]
- Djurkovic, N.; McCormack, D.; Casimir, G. The Behavioral Reactions of Victims to Different Types of Workplace Bullying. Int. J. Organ. Theory Behav. 2005, 8, 439–460. [Google Scholar] [CrossRef]
- Fowler, V.G.; Das, A.F.; Lipka-Diamond, J.; Schuch, R.; Pomerantz, R.; Jáuregui-Peredo, L.; Bressler, A.; Evans, D.; Moran, G.J.; Rupp, M.E.; et al. Exebacase for Patients with Staphylococcus aureus Bloodstream Infection and Endocarditis. J. Clin. Investig. 2020, 130, 3750–3760. [Google Scholar] [CrossRef]
- Hurdle, J.G.; O’Neill, A.J.; Chopra, I.; Lee, R.E. Targeting Bacterial Membrane Function: An Underexploited Mechanism for Treating Persistent Infections. Nat. Rev. Microbiol. 2011, 9, 62–75. [Google Scholar] [CrossRef]
- Carratalá, J.V.; Arís, A.; Garcia-Fruitós, E.; Ferrer-Miralles, N. Design Strategies for Positively Charged Endolysins: Insights into Artilysin Development. Biotechnol. Adv. 2023, 69, 108250. [Google Scholar] [CrossRef] [PubMed]
- Briers, Y.; Walmagh, M.; Van Puyenbroeck, V.; Cornelissen, A.; Cenens, W.; Aertsen, A.; Oliveira, H.; Azeredo, J.; Verween, G.; Pirnay, J.P.; et al. Engineered Endolysin-based “Artilysins” to Combat Multidrug Resistant Gram-negative Pathogens. mBio 2014, 5, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.F.; Kong, L.C.; Liu, Y.; Fu, Q.; Cui, Z.L.; Wang, J.; Ma, J.J.; Wang, H.A.; Yan, Y.X.; Sun, J.H. A Phage Lysin Fused to a Cell-penetrating Peptide Kills Intracellular Methicillin-resistant Staphylococcus aureus in Keratinocytes and Has Potential as a Treatment for Skin Infections in Mice. Appl. Environ. Microbiol. 2018, 84, e00380-18. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, M.; Zhang, P.; Leung, S.S.Y.; Xia, J. Membrane-Permeable Antibacterial Enzyme Against Multidrug-resistant Acinetobacter baumannii. ACS Infect. Dis. 2021, 7, 2192–2204. [Google Scholar] [CrossRef] [PubMed]
- Davies, G.; Henrissat, B. Structures and Mechanisms of Glycosyl Hydrolases. Structure 1995, 3, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Severi, E.; Hood, D.W.; Thomas, G.H. Sialic Acid Utilization by Bacterial Pathogens. Microbiology 2007, 153, 2817–2822. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, S.; Taylor, P.W. Neuraminidase Associated with Coliphage E That Specifically Depolymerizes the Escherichia coli K1 Capsular Polysaccharide. J. Virol. 1985, 55, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, D.; Browning, C.; Stummeyer, K.; Oberbeck, A.; Mühlenhoff, M.; Gerardy-Schahn, R.; Leiman, P.G. Structure and Biochemical Characterization of Bacteriophage Phi92 Endosialidase. Virology 2015, 477, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Murakami, H.; Kuramoto, T.; Mizutani, K.; Nakano, H.; Kitahata, S. Purification and Some Properties of a New Levanase from Bacillus Sp. No. 71. Biosci. Biotechnol. Biochem. 1992, 56, 608–613. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jathore, N.R.; Bule, M.V.; Tilay, A.V.; Annapure, U.S. Microbial Levan from Pseudomonas Fluorescens: Characterization and Medium Optimization for Enhanced Production. Food Sci. Biotechnol. 2012, 21, 1045–1053. [Google Scholar] [CrossRef]
- Dogsa, I.; Brloznik, M.; Stopar, D.; Mandic-Mulec, I. Exopolymer Diversity and the Role of Levan in Bacillus subtilis Biofilms. PLoS ONE 2013, 8, e62044. [Google Scholar] [CrossRef] [PubMed]
- Maaroufi, H.; Levesque, R.C. Glycoside Hydrolase Family 32 Is Present in Bacillus subtilis Phages. Virol. J. 2015, 12, 157. [Google Scholar] [CrossRef] [PubMed]
- Iwashita, S.; Kanegasaki, S. Smooth Specific Phage Adsorption: Endorhamnosidase Activity of Tail Parts of P22. Biochem. Biophys. Res. Commun. 1973, 55, 403–409. [Google Scholar] [CrossRef] [PubMed]
- León, M.; Bastías, R. Virulence Reduction in Bacteriophage Resistant Bacteria. Front. Microbiol. 2015, 06. [Google Scholar] [CrossRef]
- Thurow, H.; Niemann, H.; Rudolph, C.; Stirm, S. Host Capsule Depolymerase Activity of Bacteriophage Particles Active on Klebsiella K20 and K24 Strains. Virology 1974, 58, 306–309. [Google Scholar] [CrossRef]
- Hanna, A.; Berg, M.; Stout, V.; Razatos, A. Role of Capsular Colanic Acid in Adhesion of Uropathogenic Escherichia coli. Appl. Environ. Microbiol. 2003, 69, 4474. [Google Scholar] [CrossRef]
- Danese, P.N.; Pratt, L.A.; Kolter, R. Exopolysaccharide Production Is Required for Development of Escherichia coli K-12 Biofilm Architecture. J. Bacteriol. 2000, 182, 3593–3596. [Google Scholar] [CrossRef]
- Knirel, Y.A.; Prokhorov, N.S.; Shashkov, A.S.; Ovchinnikova, O.G.; Zdorovenko, E.L.; Liu, B.; Kostryukova, E.S.; Larin, A.K.; Golomidova, A.K.; Letarov, A.V. Variations in O-Antigen Biosynthesis and O-Acetylation Associated with Altered Phage Sensitivity in Escherichia coli 4s. J. Bacteriol. 2015, 197, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.E.; Pourhossein, M.; Waterhouse, A.; Hudson, T.; Goldrick, M.; Derrick, J.P.; Roberts, I.S. The K5 Lyase KflA Combines a Viral Tail Spike Structure with a Bacterial Polysaccharide Lyase Mechanism. J. Biol. Chem. 2010, 285, 23963–23969. [Google Scholar] [CrossRef]
- Hallenbeck, P.C.; Vimr, E.R.; Yu, F.; Bassler, B.; Troy, F.A. Purification and Properties of a Bacteriophage-Induced Endo-N-Acetylneuraminidase Specific for Poly-Alpha-2,8-Sialosyl Carbohydrate Units. J. Biol. Chem. 1987, 262, 3553–3561. [Google Scholar] [CrossRef]
- Freiberg, A.; Morona, R.; Van Den Bosch, L.; Jung, C.; Behlke, J.; Carlin, N.; Seckler, R.; Baxa, U. The Tailspike Protein of Shigella Phage Sf6. J. Biol. Chem. 2003, 278, 1542–1548. [Google Scholar] [CrossRef]
- Knecht, L.E.; Heinrich, N.; Born, Y.; Felder, K.; Pelludat, C.; Loessner, M.J.; Fieseler, L. Bacteriophage S6 Requires Bacterial Cellulose for Erwinia amylovora Infection. Environ. Microbiol. 2022. [Google Scholar] [CrossRef]
- Kimura, K.; Itoh, Y. Characterization of Poly-γ-Glutamate Hydrolase Encoded by a Bacteriophage Genome: Possible Role in Phage Infection of Bacillus subtilis Encapsulated with Poly-γ-Glutamate. Appl. Environ. Microbiol. 2003, 69, 2491–2497. [Google Scholar] [CrossRef]
- Fenton, M.; Keary, R.; McAuliffe, O.; Ross, R.P.; O’Mahony, J.; Coffey, A. Bacteriophage-Derived Peptidase Eliminates and Prevents Staphylococcal Biofilms. Int. J. Microbiol. 2013, 2013, 625341. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K.; Bharati, A.P.; Singh, N.; Pandey, P.; Joshi, P.; Singh, K.; Mitra, K.; Gayen, J.R.; Sarkar, J.; Akhtar, M.d.S. The Prophage-Encoded Hyaluronate Lyase Has Broad Substrate Specificity and Is Regulated by the N-Terminal Domain. J. Biol. Chem. 2014, 289, 35225–35236. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.R.; Dong, S.; Pritchard, D.G. The Hyaluronan Lyase of Streptococcus Pyogenes Bacteriophage H4489A. Biochem. J. 2002, 365, 317–322. [Google Scholar] [CrossRef]
- Gutiérrez, D.; Briers, Y.; Rodríguez-Rubio, L.; Martínez, B.; Rodríguez, A.; Lavigne, R.; García, P. Role of the Pre-Neck Appendage Protein (Dpo7) from Phage VB_SepiS-PhiIPLA7 as an Anti-Biofilm Agent in Staphylococcal Species. Front. Microbiol. 2015, 6, 1315. [Google Scholar] [CrossRef]
- Pires, D.P.; Oliveira, H.; Melo, L.D.R.; Sillankorva, S.; Azeredo, J. Bacteriophage-Encoded Depolymerases: Their Diversity and Biotechnological Applications. Appl. Microbiol. Biotechnol. 2016, 100, 2141–2151. [Google Scholar] [CrossRef] [PubMed]
- Alkawash, M.A.; Soothill, J.S.; Schiller, N.L. Alginate Lyase Enhances Antibiotic Killing of Mucoid Pseudomonas aeruginosa in Biofilms. APMIS 2006, 114, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Colvin, K.M.; Gordon, V.D.; Murakami, K.; Borlee, B.R.; Wozniak, D.J.; Wong, G.C.L.; Parsek, M.R. The Pel Polysaccharide Can Serve a Structural and Protective Role in the Biofilm Matrix of Pseudomonas aeruginosa. PLoS Pathog. 2011, 7, e1001264. [Google Scholar] [CrossRef] [PubMed]
- Bansal, S.; Harjai, K.; Chhibber, S. Depolymerase Improves Gentamicin Efficacy during Klebsiella pneumoniae Induced Murine Infection. BMC Infect. Dis. 2014, 14, 456. [Google Scholar] [CrossRef] [PubMed]
- Domenech, M.; Ramos-Sevillano, E.; García, E.; Moscoso, M.; Yuste, J. Biofilm Formation Avoids Complement Immunity and Phagocytosis of Streptococcus pneumoniae. Infect. Immun. 2013, 81, 2606–2615. [Google Scholar] [CrossRef] [PubMed]
- Eftekhar, F.; Speert, D.P. Alginase Treatment of Mucoid Pseudomonas aeruginosa Enhances Phagocytosis by Human Monocyte-Derived Macrophages. Infect. Immun. 1988, 56, 2788–2793. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Leung, S.S.Y.; Huang, Y.; Guo, Y.; Jiang, N.; Li, P.; Chen, J.; Wang, R.; Bai, C.; Mi, Z.; et al. Identification of Two Depolymerases From Phage IME205 and Their Antivirulent Functions on K47 Capsule of Klebsiella pneumoniae. Front. Microbiol. 2020, 11, 218. [Google Scholar] [CrossRef]
- Chen, X.; Liu, M.; Zhang, P.; Xu, M.; Yuan, W.; Bian, L.; Liu, Y.; Xia, J.; Leung, S.S.Y. Phage-Derived Depolymerase as an Antibiotic Adjuvant Against Multidrug-Resistant Acinetobacter baumannii. Front. Microbiol. 2022, 13. [Google Scholar] [CrossRef]
- Rice, C.J.; Kelly, S.A.; O’Brien, S.C.; Melaugh, E.M.; Ganacias, J.C.B.; Chai, Z.H.; Gilmore, B.F.; Skvortsov, T. Novel Phage-Derived Depolymerase with Activity against Proteus Mirabilis Biofilms. Microorganisms 2021, 9, 2172. [Google Scholar] [CrossRef]
- Melo, L.D.R.; Veiga, P.; Cerca, N.; Kropinski, A.M.; Almeida, C.; Azeredo, J.; Sillankorva, S. Development of a Phage Cocktail to Control Proteus Mirabilis Catheter-Associated Urinary Tract Infections. Front. Microbiol. 2016, 0, 1024. [Google Scholar] [CrossRef]
- Shahed-Al-Mahmud, M.; Roy, R.; Sugiokto, F.G.; Islam, M.N.; Lin, M.-D.; Lin, L.-C.; Lin, N.-T. Phage ΦAB6-Borne Depolymerase Combats Acinetobacter baumannii Biofilm Formation and Infection. Antibiotics 2021, 10, 279. [Google Scholar] [CrossRef] [PubMed]
- Kaźmierczak, Z.; Piotrowicz, A.; Owczarek, B.; Hodyra, K.; Miernikiewicz, P.; Lecion, D.; Harhala, M.; Górski, A.; Dąbrowska, K. Molecular Imaging of T4 Phage in Mammalian Tissues and Cells. Bacteriophage 2014, 4, e28364. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.S.; Kaido, T.; Carrier, E. Immunological Factors That Affect the in Vivo Fate of T7 Phage in the Mouse. J. Virol. Methods 2004, 115, 99–104. [Google Scholar] [CrossRef]
- Park, K.; Cha, K.E.; Myung, H. Observation of Inflammatory Responses in Mice Orally Fed with Bacteriophage T7. J. Appl. Microbiol. 2014, 117, 627–633. [Google Scholar] [CrossRef]
- Miernikiewicz, P.; Dąbrowska, K.; Piotrowicz, A.; Owczarek, B.; Wojas-Turek, J.; Kicielińska, J.; Rossowska, J.; Pajtasz-Piasecka, E.; Hodyra, K.; Macegoniuk, K.; et al. T4 Phage and Its Head Surface Proteins Do Not Stimulate Inflammatory Mediator Production. PLoS ONE 2013, 8, e71036. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.-Y.; Kim, J.-E.; Song, Y.-J.; Park, J.-H. Safety of Using Escherichia coli Bacteriophages as a Sanitizing Agent Based on Inflammatory Responses in Rats. Food Sci. Biotechnol. 2016, 25, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Carmody, L.A.; Gill, J.J.; Summer, E.J.; Sajjan, U.S.; Gonzalez, C.F.; Young, R.F.; LiPuma, J.J. Efficacy of Bacteriophage Therapy in a Model of Burkholderia cenocepacia Pulmonary Infection. J. Infect. Dis. 2010, 201, 264–271. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska, K.; Miernikiewicz, P.; Piotrowicz, A.; Hodyra, K.; Owczarek, B.; Lecion, D.; Kaźmierczak, Z.; Letarov, A.; Górski, A. Immunogenicity Studies of Proteins Forming the T4 Phage Head Surface. J. Virol. 2014, 88, 12551–12557. [Google Scholar] [CrossRef]
- Hussain, W.; Yang, X.; Ullah, M.; Wang, H.; Aziz, A.; Xu, F.; Asif, M.; Ullah, M.W.; Wang, S. Genetic Engineering of Bacteriophages: Key Concepts, Strategies, and Applications. Biotechnol. Adv. 2023, 64, 108116. [Google Scholar] [CrossRef]
- Edgar, R.; Friedman, N.; Molshanski-Mor, S.; Qimron, U. Reversing Bacterial Resistance to Antibiotics by Phage-Mediated Delivery of Dominant Sensitive Genes. Appl. Environ. Microbiol. 2012, 78, 744–751. [Google Scholar] [CrossRef]
- Międzybrodzki, R.; Fortuna, W.; Weber-Dąbrowska, B.; Górski, A. Phage Therapy of Staphylococcal Infections (Including MRSA) May Be Less Expensive than Antibiotic Treatment Word Count: Coresponding. Adv. Hyg. Exp. Med. 2007, 61. [Google Scholar]
- Morozova, V.V.; Vlassov, V.V.; Tikunova, N.V. Applications of Bacteriophages in the Treatment of Localized Infections in Humans. Front. Microbiol. 2018, 9, 390010. [Google Scholar] [CrossRef] [PubMed]
- Rose, T.; Verbeken, G.; De Vos, D.; Merabishvili, M.; Vaneechoutte, M.; Lavigne, R.; Jennes, S.; Zizi, M.; Pirnay, J.-P. Experimental Phage Therapy of Burn Wound Infection: Difficult First Steps. Int. J. Burn. Trauma. 2014, 4, 66–73. [Google Scholar]
- Rhoads, D.D.; Wolcott, R.D.; Kuskowski, M.A.; Wolcott, B.M.; Ward, L.S.; Sulakvelidze, A. Bacteriophage Therapy of Venous Leg Ulcers in Humans: Results of a Phase I Safety Trial. J. Wound Care 2009, 18, 237–243. [Google Scholar] [CrossRef]
- Markoishvili, K.; Tsitlanadze, G.; Katsarava, R.; Glenn, J.; Morris, M.D., Jr.; Sulakvelidze, A. A Novel Sustained-release Matrix Based on Biodegradable Poly(Ester Amide)s and Impregnated with Bacteriophages and an Antibiotic Shows Promise in Management of Infected Venous Stasis Ulcers and Other Poorly Healing Wounds. Int. J. Dermatol. 2002, 41, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Fish, R.; Kutter, E.; Wheat, G.; Blasdel, B.; Kutateladze, M.; Kuhl, S. Compassionate Use of Bacteriophage Therapy for Foot Ulcer Treatment as an Effective Step for Moving Toward Clinical Trials. In Bacteriophage Therapy: From Lab to Clinical Practice; Humana Press: Totowa, NJ, USA, 2018; pp. 159–170. [Google Scholar]
- Abdul-Hassan, H.S.; El-Tahan, E.; Massoud, B.; Gomaa, R. Bacteriophage Therapy of Pseudomonas Burn Wound Sepsis. Ann. Medit. Burn. Club 1990, 3, 262–264. [Google Scholar]
- Sarkis, G.J.; Jacobs, W.R.; Hatfulll, G.F. L5 Luciferase Reporter Mycobacteriophages: A Sensitive Tool for the Detection and Assay of Live Mycobacteria. Mol. Microbiol. 1995, 15, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, W.R.; Tuckman, M.; Bloom, B.R. Introduction of Foreign DNA into Mycobacteria Using a Shuttle Phasmid. Nature 1987, 327, 532–535. [Google Scholar] [CrossRef]
- Chauthaiwale, V.M.; Therwath, A.; Deshpande, V.V. Bacteriophage Lambda as a Cloning Vector. Microbiol. Rev. 1992, 56, 577–591. [Google Scholar] [CrossRef]
- Marinelli, L.J.; Piuri, M.; Swigoňová, Z.; Balachandran, A.; Oldfield, L.M.; van Kessel, J.C.; Hatfull, G.F. BRED: A Simple and Powerful Tool for Constructing Mutant and Recombinant Bacteriophage Genomes. PLoS ONE 2008, 3, e3957. [Google Scholar] [CrossRef]
- Hatoum-Aslan, A. Phage Genetic Engineering Using CRISPR–Cas Systems. Viruses 2018, 10, 335. [Google Scholar] [CrossRef] [PubMed]
- Wetzel, K.S.; Guerrero-Bustamante, C.A.; Dedrick, R.M.; Ko, C.-C.; Freeman, K.G.; Aull, H.G.; Divens, A.M.; Rock, J.M.; Zack, K.M.; Hatfull, G.F. CRISPY-BRED and CRISPY-BRIP: Efficient Bacteriophage Engineering. Sci. Rep. 2021, 11, 6796. [Google Scholar] [CrossRef]
- Pires, D.P.; Cleto, S.; Sillankorva, S.; Azeredo, J.; Lu, T.K. Genetically Engineered Phages: A Review of Advances over the Last Decade. Microbiol. Mol. Biol. Rev. 2016, 80, 523–543. [Google Scholar] [CrossRef]
- Summers, W.C. Cholera and Plague in India: The Bacteriophage Inquiry of 1927–1936. J. Hist. Med. Allied Sci. 1993, 48, 275–301. [Google Scholar] [CrossRef] [PubMed]
- Sarker, S.A.; McCallin, S.; Barretto, C.; Berger, B.; Pittet, A.-C.; Sultana, S.; Krause, L.; Huq, S.; Bibiloni, R.; Bruttin, A.; et al. Oral T4-like Phage Cocktail Application to Healthy Adult Volunteers from Bangladesh. Virology 2012, 434, 222–232. [Google Scholar] [CrossRef] [PubMed]
- McCallin, S.; Alam Sarker, S.; Barretto, C.; Sultana, S.; Berger, B.; Huq, S.; Krause, L.; Bibiloni, R.; Schmitt, B.; Reuteler, G.; et al. Safety Analysis of a Russian Phage Cocktail: From MetaGenomic Analysis to Oral Application in Healthy Human Subjects. Virology 2013, 443, 187–196. [Google Scholar] [CrossRef]
- Bourdin, G.; Schmitt, B.; Marvin Guy, L.; Germond, J.-E.; Zuber, S.; Michot, L.; Reuteler, G.; Brüssow, H. Amplification and Purification of T4-Like Escherichia coli Phages for Phage Therapy: From Laboratory to Pilot Scale. Appl. Environ. Microbiol. 2014, 80, 1469–1476. [Google Scholar] [CrossRef]
- Lu, T.K.; Collins, J.J. Dispersing Biofilms with Engineered Enzymatic Bacteriophage. Proc. Natl. Acad. Sci. USA 2007, 104, 11197–11202. [Google Scholar] [CrossRef]
- Wu, J.W.; Wang, J.T.; Lin, T.L.; Liu, Y.Z.; Wu, L.T.; Pan, Y.J. Identification of Three Capsule Depolymerases in a Bacteriophage Infecting Klebsiella pneumoniae Capsular Types K7, K20, and K27 and Therapeutic Application. J. Biomed. Sci. 2023, 30. [Google Scholar] [CrossRef]
- Majkowska-Skrobek, G.; Latka, A.; Berisio, R.; Squeglia, F.; Maciejewska, B.; Briers, Y.; Drulis-Kawa, Z. Phage-Borne Depolymerases Decrease Klebsiella pneumoniae Resistance to Innate Defense Mechanisms. Front. Microbiol. 2018, 9, 2517. [Google Scholar] [CrossRef]
- Lin, H.; Paff, M.L.; Molineux, I.J.; Bull, J.J. Therapeutic Application of Phage Capsule Depolymerases against K1, K5, and K30 Capsulated E. coli in Mice. Front. Microbiol. 2017, 8, 292603. [Google Scholar] [CrossRef]
- Chen, Y.; Li, X.; Wang, S.; Guan, L.; Li, X.; Hu, D.; Gao, D.; Song, J.; Chen, H.; Qiana, P. A Novel Tail-Associated O91-Specific Polysaccharide Depolymerase from a Podophage Reveals Lytic Efficacy of Shiga Toxin-Producing Escherichia coli. Appl. Environ. Microbiol. 2020, 86, e00145-20. [Google Scholar] [CrossRef]
- Mi, L.; Liu, Y.; Wang, C.; He, T.; Gao, S.; Xing, S.; Huang, Y.; Fan, H.; Zhang, X.; Yu, W.; et al. Identification of a Lytic Pseudomonas aeruginosa Phage Depolymerase and Its Anti-Biofilm Effect and Bactericidal Contribution to Serum. Virus Genes. 2019, 55, 394–405. [Google Scholar] [CrossRef]
- Wyres, K.L.; Wick, R.R.; Gorrie, C.; Jenney, A.; Follador, R.; Thomson, N.R.; Holt, K.E. Identification of Klebsiella Capsule Synthesis Loci from Whole Genome Data. Microb. Genom. 2016, 2, e000102. [Google Scholar] [CrossRef]
- van der Graaf-van Bloois, L.; Chen, H.; Wagenaar, J.A.; Zomer, A.L. Development of Kaptive Databases for Vibrio parahaemolyticus O- and K-Antigen Genotyping. Microb. Genom. 2023, 9, 001007. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.-J.; Lin, T.-L.; Chen, C.-T.; Chen, Y.-Y.; Hsieh, P.-F.; Hsu, C.-R.; Wu, M.-C.; Wang, J.-T. Genetic Analysis of Capsular Polysaccharide Synthesis Gene Clusters in 79 Capsular Types of Klebsiella spp. Sci. Rep. 2015, 5, 15573. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.R.; Liao, C.H.; Lin, T.L.; Yang, H.R.; Yang, F.L.; Hsieh, P.F.; Wu, S.H.; Wang, J.T. Identification of a Capsular Variant and Characterization of Capsular Acetylation in Klebsiella pneumoniae PLA-associated Type K57. Sci. Rep. 2016, 6, 31946. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sheng, Y.; Ma, R.; Xu, M.; Liu, F.; Qin, R.; Zhu, M.; Zhu, X.; He, P. Identification of a Depolymerase Specific for K64-Serotype Klebsiella pneumoniae: Potential Applications in Capsular Typing and Treatment. Antibiotics 2021, 10, 144. [Google Scholar] [CrossRef] [PubMed]
- Park, D.-W.; Park, J.-H. Characterization of a Novel Phage Depolymerase Specific to Escherichia coli O157:H7 and Biofilm Control on Abiotic Surfaces. J. Microbiol. 2021, 59, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, R.A.; Bamert, R.S.; Belousoff, M.J.; Short, F.L.; Barlow, C.K.; Pickard, D.J.; Wilksch, J.J.; Schittenhelm, R.B.; Strugnell, R.A.; Dougan, G.; et al. Mechanistic Insights into the Capsule-Targeting Depolymerase from a Klebsiella pneumoniae Bacteriophage. Microbiol. Spectr. 2021, 9, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yang, H.; Luo, S.; Wang, L.; Lu, S.; Fu, Z. Engineering Phage Tail Fiber Protein as a Wide-Spectrum Probe for Acinetobacter baumannii Strains with a Recognition Rate of 100%. Anal. Chem. 2022, 94, 9610–9617. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dicks, L.M.T.; Vermeulen, W. Bacteriophage–Host Interactions and the Therapeutic Potential of Bacteriophages. Viruses 2024, 16, 478. https://doi.org/10.3390/v16030478
Dicks LMT, Vermeulen W. Bacteriophage–Host Interactions and the Therapeutic Potential of Bacteriophages. Viruses. 2024; 16(3):478. https://doi.org/10.3390/v16030478
Chicago/Turabian StyleDicks, Leon M. T., and Wian Vermeulen. 2024. "Bacteriophage–Host Interactions and the Therapeutic Potential of Bacteriophages" Viruses 16, no. 3: 478. https://doi.org/10.3390/v16030478
APA StyleDicks, L. M. T., & Vermeulen, W. (2024). Bacteriophage–Host Interactions and the Therapeutic Potential of Bacteriophages. Viruses, 16(3), 478. https://doi.org/10.3390/v16030478







