Elimination of Solanum nigrum ilarvirus 1 and Apple Hammerhead Viroid from Apple Cultivars Using Antivirals Ribavirin, Rimantadine, and Zidovudine
Abstract
1. Introduction
2. Materials and Methods
2.1. In Vitro Cultures
2.2. Antivirals
2.3. Application of Antivirals
2.4. Plant Material & RNA Extraction
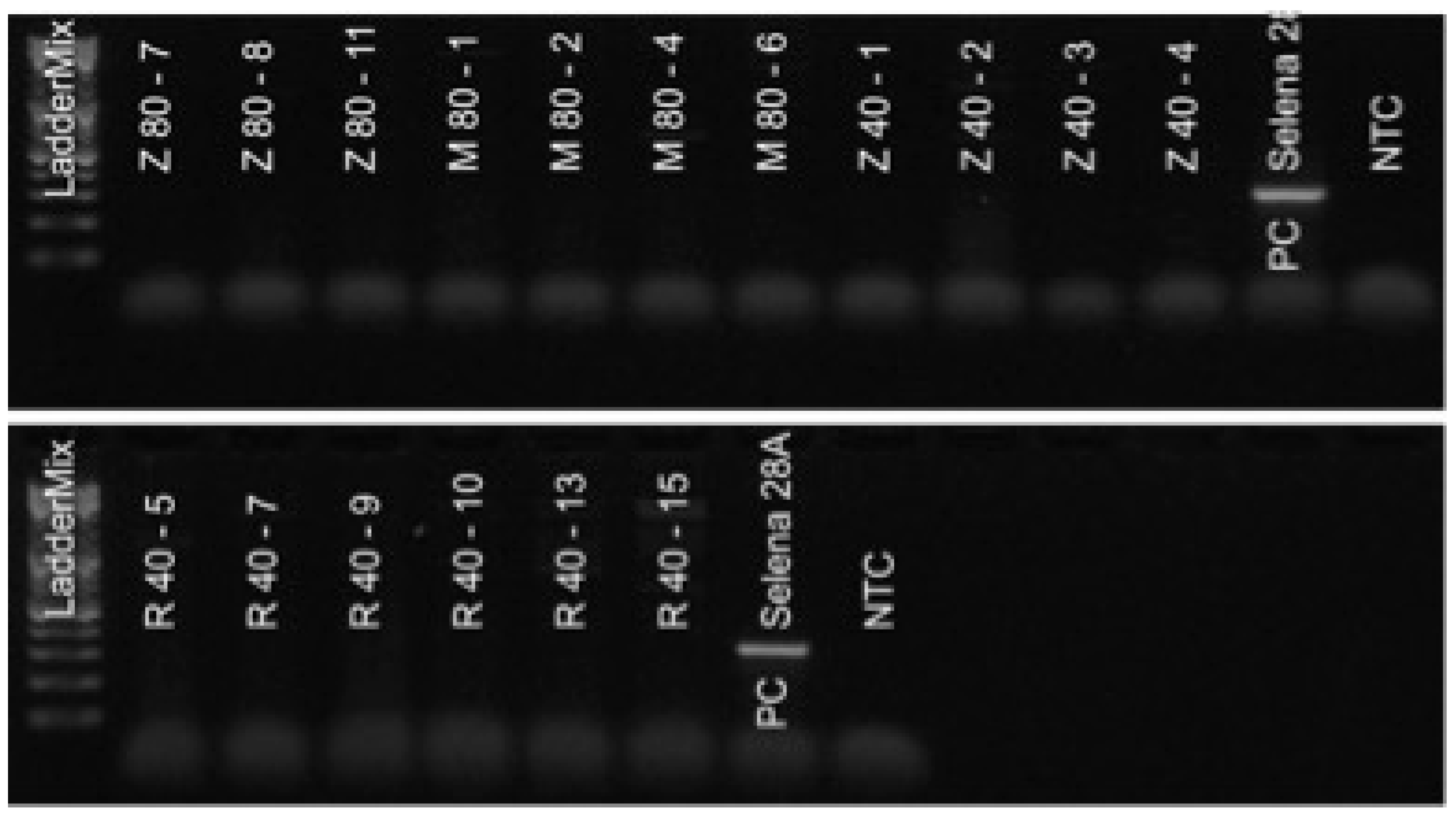
2.5. Diagnostics of SnIV-1 and AHVd by RT-PCR
2.6. Virus and Viroid Diagnostics by RT-qPCR
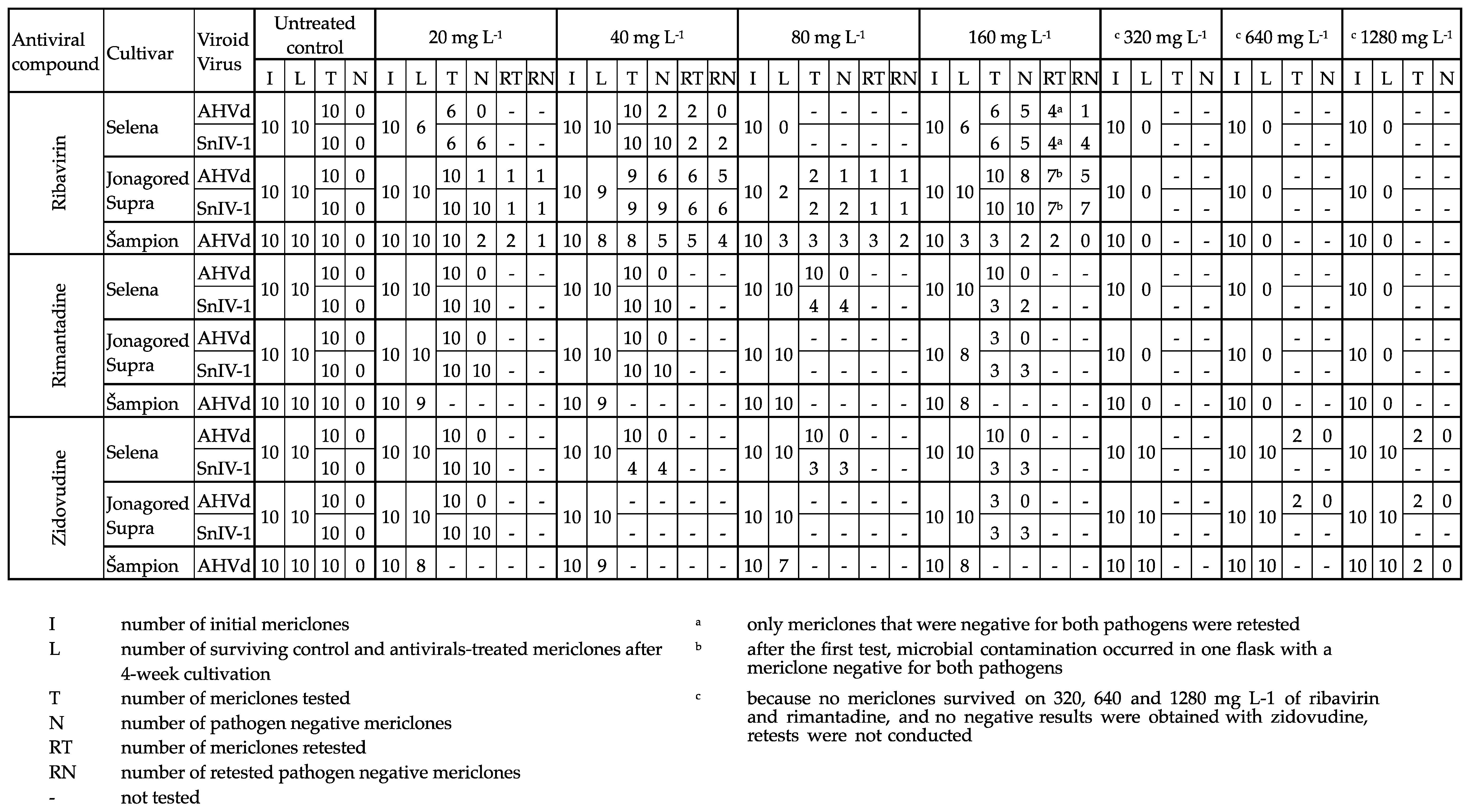
3. Results
3.1. Elimination of SnIV-1
3.2. Elimination of AHVd
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loebenstein, G. Plant virus diseases: Economic aspects. In Desk Encyclopedia of Plant and Fungal Virology, 1st ed.; van Regenmortel, M., Mahy, B., Eds.; Academic Press: Oxford, UK, 2009; pp. 426–430. [Google Scholar]
- Jones, R.A.C.; Naidu, R.A. Global Dimensions of Plant Virus Diseases: Current Status and Future Perspectives. Annu. Rev. Virol. 2019, 6, 387–409. [Google Scholar] [CrossRef] [PubMed]
- Hilaire, J.; Tindale, S.; Jones, G.; Pingarron-Cardenas, G.; Bačnik, K.; Ojo, M.; Frewer, L.J. Risk perception associated with an emerging agri-food risk in Europe: Plant viruses in agriculture. Agric. Food Secur. 2022, 11, 21. [Google Scholar] [CrossRef] [PubMed]
- Pautasso, M.; Petter, F.; Rortais, A.; Roy, A.-S. Emerging risks to plant health: A European perspective. CAB Rev. 2015, 10, 1–16. [Google Scholar] [CrossRef]
- Bettoni, J.C.; Fazio, G.; Costa, L.C.; Hurtado-Gonzales, O.P.P.; Al Rwahnih, M.; Nedrow, A.; Volk, G.M.M. Thermotherapy Followed by Shoot Tip Cryotherapy Eradicates Latent Viruses and Apple Hammerhead Viroid from In Vitro Apple Rootstocks. Plants 2022, 11, 18. [Google Scholar] [CrossRef]
- Xiao, H.; Hao, W.; Storoschuk, G.; MacDonald, J.L.; Sanfaçon, H. Characterizing the Virome of Apple Orchards Affected by Rapid Decline in the Okanagan and Similkameen Valleys of British Columbia (Canada). Pathogens 2022, 11, 1231. [Google Scholar] [CrossRef]
- Boonham, N.; Kreuze, J.; Winter, S.; van der Vlugt, R.; Bergervoet, J.; Tomlinson, J.; Mumford, R. Methods in virus diagnostics: From ELISA to next generation sequencing. Virus Res. 2014, 186, 20–31. [Google Scholar] [CrossRef]
- Patel, R.; Mitra, B.; Vinchurkar, M.; Adami, A.; Patkar, R.; Giacomozzi, F.; Lorenzelli, L.; Baghini, M.S. A review of recent advances in plant-pathogen detection systems. Heliyon 2022, 8, 12. [Google Scholar] [CrossRef]
- Massart, S.; Candresse, T.; Gil, J.; Lacomme, C.; Predajna, L.; Ravnikar, M.; Reynard, J.S.; Rumbou, A.; Saldarelli, P.; Skoric, D.; et al. A Framework for the Evaluation of Biosecurity, Commercial, Regulatory, and Scientific Impacts of Plant Viruses and Viroids Identified by NGS Technologies. Front. Microbiol. 2017, 8, 7. [Google Scholar] [CrossRef]
- Varallyay, E.; Pribylova, J.; Galbacs, Z.N.; Jahan, A.; Varga, T.; Spak, J.; Lenz, O.; Franova, J.; Sedlak, J.; Koloniuk, I. Detection of Apple Hammerhead Viroid, Apple Luteovirus 1 and Citrus Concave Gum-Associated Virus in Apple Propagation Materials and Orchards in the Czech Republic and Hungary. Viruses 2022, 14, 20. [Google Scholar] [CrossRef]
- Serra, P.; Messmer, A.; Sanderson, D.; James, D.; Flores, R. Apple hammerhead viroid-like RNA is a bona fide viroid: Autonomous replication and structural features support its inclusion as a new member in the genus Pelamoviroid. Virus Res. 2018, 249, 8–15. [Google Scholar] [CrossRef]
- Ma, Y.X.; Marais, A.; Lefebvre, M.; Faure, C.; Candresse, T. Metagenomic analysis of virome cross-talk between cultivated Solanum lycopersicum and wild Solanum nigrum. Virology 2020, 540, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Messmer, A.; Sanderson, D.; Braun, G.; Serra, P.; Flores, R.; James, D. Molecular and phylogenetic identification of unique isolates of hammerhead viroid-like RNA from ‘Pacific Gala’ apple (Malus domestica) in Canada. Can. J. Plant Pathol. 2017, 39, 342–353. [Google Scholar] [CrossRef]
- Graci, J.D.; Cameron, C.E. Mechanisms of action of ribavirin against distinct viruses. Rev. Med. Virol. 2006, 16, 37–48. [Google Scholar] [CrossRef]
- Hansen, A.J. Antiviral chemicals for plant-disease control. Crit. Rev. Plant Sci. 1989, 8, 45–88. [Google Scholar] [CrossRef]
- Hu, G.J.; Dong, Y.F.; Zhang, Z.P.; Fan, X.D.; Ren, F.; Zhou, J. Virus elimination from in vitro apple by thermotherapy combined with chemotherapy. Plant Cell Tissue Organ Cult. 2015, 121, 435–443. [Google Scholar] [CrossRef]
- Kudelkova, M.; Pavelkova, R.; Ondrusikova, E. Virus elimination in peach using chemotherapy. Acta Hortic. 2017, 1155, 431–437. [Google Scholar] [CrossRef]
- Pavelkova, R.; Kudelkova, M.; Ondrusikova, E.; Eichmeier, A. Virus Elimination in Peach cv. ‘Red Haven’ by Chemotherapy. Agric. Commun. 2015, 3, 16–20. [Google Scholar]
- Murashige, T.; Skoog, F. A Revised Medium for Rapid Growth and Bio Assays with Tobacco Tissue Cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Sidwell, R.W.; Witkowski, J.T.; Allen, L.B.; Robins, R.K.; Khare, G.P.; Huffman, J.H. Broad-spectrum antiviral activity of Virazole: 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide. Science 1972, 177, 705–706. [Google Scholar] [CrossRef]
- Huffman, J.H.; Sidwell, R.W.; Khare, G.P.; Witkowski, J.T.; Allen, L.B.; Robins, R.K. In vitro effect of 1-beta-D-ribofuranosyl-1,2,4-triazole-3-carboxamide (virazole, ICN 1229) on deoxyribonucleic acid and ribonucleic-acid viruses. Antimicrob. Agents Chemother. 1973, 3, 235–241. [Google Scholar] [CrossRef]
- James, D. Long term assessment of the effects of in vitro chemotherapy as a tool for apple stem grooving virus elimination. Acta Hortic. 2001, 550, 459–462. [Google Scholar] [CrossRef]
- Wright, K. AIDS therapy. First tentative signs of therapeutic promise. Nature 1986, 323, 283. [Google Scholar] [CrossRef][Green Version]
- Tochikura, T.S.; Nakashima, H.; Yamamoto, N. Antiviral agents with activity against human retroviruses. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1989, 2, 441–447. [Google Scholar]
- Hannoun. Rimantadine in the prevention and treatment of influenza A. Rev. Médecine Interne 1988, 9, 554–558. [Google Scholar]
- Thomaston, J.L.; Samways, M.L.; Konstantinidi, A.; Ma, C.; Hu, Y.; Macdonald, H.E.B.; Wang, J.; Essex, J.W.; DeGrado, W.F.; Kolocouris, A. Rimantadine binds to and inhibits the influenza A M2 proton channel without enantiomeric specificity. Biochemistry 2021, 60, 2471–2482. [Google Scholar] [CrossRef]
- Sasaki-Tanaka, R.; Shibata, T.; Moriyama, M.; Okamoto, H.; Kogure, H.; Kanda, T. Amantadine and Rimantadine Inhibit Hepatitis A Virus Replication through the Induction of Autophagy. J. Virol. 2022, 96, e00646-22. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.X.; Qi, S.S.; Tang, N.; Zhang, X.X.; Chen, S.S.; Zhu, P.F.; Ma, L.; Cheng, J.P.; Xu, Y.; Lu, M.G.; et al. Discovery of Replicating Circular RNAs by RNA-Seq and Computational Algorithms. PLoS Pathog. 2014, 10, 14. [Google Scholar] [CrossRef]
- Szostek, S.A.; Wright, A.A.; Harper, S.J. First Report of Apple Hammerhead Viroid in the United States, Japan, Italy, Spain, and New Zealand. Plant Dis. 2018, 102, 2670. [Google Scholar] [CrossRef]
- Wright, A.A.; Cross, A.R.; Harper, S.J. A bushel of viruses: Identification of seventeen novel putative viruses by RNA-seq in six apple trees. PLoS ONE 2020, 15, e0227669. [Google Scholar] [CrossRef]
- Lim, S.; Moon, J.S.; Cho, I.S.; Kim, H.R.; Lee, S.H. First Report of Apple Hammerhead Viroid Infecting Apple Trees in South Korea. Plant Dis. 2019, 103, 2700. [Google Scholar] [CrossRef]
- Nabi, S.U.; Baranwal, V.K. First Report of Apple Hammerhead Viroid Infecting Apple Cultivars in India. Plant Dis. 2020, 104, 3086–3087. [Google Scholar] [CrossRef]
- Hamdi, I.; Soltani, R.; Baraket, G.; Varsani, A.; Najar, A. First report of apple hammerhead viroid infecting ‘Richared Delicious’ apple (Malus domestica) in Tunisia. J. Plant Pathol. 2022, 104, 811–812. [Google Scholar] [CrossRef]
- Barba, M.; Hosakawa, M.; Wang, Q.-C.; Taglienti, A.; Hamborg, Z. Viroid Elimination by Thermotherapy, Cold Therapy, Tissue Culture, In Vitro Micrografting, or Cryotherapy. In Viroids and Satellites, 1st ed.; Hadidi, A., Flores, R., Randles, J.W., Palukaitis, P., Eds.; Academic Press: Oxford, UK, 2017; pp. 425–435. [Google Scholar]
- Desvignes, J.C.; Grasseau, N.; Boye, R.; Cornaggia, D.; Aparicio, F.; Di Serio, F.; Flores, R. Biological properties of apple scar skin viroid: Isolates, host range, different sensitivity of apple cultivars, elimination, and natural transmission. Plant Dis. 1999, 83, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Hu, G.J.; Zhang, Z.P.; Dong, Y.F.; Fan, X.D.; Ren, F.; Zhu, H.J. Efficiency of virus elimination from potted apple plants by thermotherapy coupled with shoot-tip grafting. Austral. Plant Pathol. 2015, 44, 167–173. [Google Scholar] [CrossRef]
- Wang, M.-R.; Bi, W.-L.; Bettoni, J.C.; Zhang, D.; Volk, G.M.; Wang, Q.-C. Shoot tip cryotherapy for plant pathogen eradication. Plant Pathol. 2022, 71, 1241–1254. [Google Scholar] [CrossRef]
- Hu, G.J.; Dong, Y.F.; Zhang, Z.P.; Fan, X.D.; Ren, F. Inefficiency of ribavirin to eliminate apple scar skin viroid from apple plants. Plant Cell Tissue Organ Cult. 2022, 151, 189–197. [Google Scholar] [CrossRef]
- Hu, G.J.; Dong, Y.F.; Zhang, Z.P.; Fan, X.D.; Ren, F.; Li, Z.N. Efficacy of virus elimination from apple by thermotherapy coupled with in vivo shoot-tip grafting and in vitro meristem culture. J. Phytopathol. 2017, 165, 701–706. [Google Scholar] [CrossRef]
- Bhat, A.I.; Rao, G.P. Virus Elimination by Meristem-Tip Culture. In Characterization of Plant Viruses; Springer Protocols Handbooks; Humana: New York, NY, USA, 2020. [Google Scholar] [CrossRef]

| Target | Primer Name | Primer Sequence (5’-3’) | Usage; Region | Reference |
|---|---|---|---|---|
| Malus domestica, NADH a | 2277 | TGCTCCATGGATCTCATCGG | RT-qPCR, RT-PCR, RNA extraction and PCR amplification control; protein-coding region | [10] |
| 2278 | AATCGAGGGCTATGCGGATC | |||
| AHVd b | 2115 | CTGCCGAAACAGAGGTTGGA | RT-qPCR, RT-PCR; genome | |
| 2116 | GAGAAGTCGCTCTCTCTCGC | |||
| SnIV-1 c | 2696 | TTTGGGTTTGTAGCCGAATC | RT-qPCR; gene for coat protein | this work |
| 2697 | TTACCTCCGAGATCAACGTC | |||
| 2365 | GCCCTATCTCTACCCGAGGT | RT-PCR; gene for polymerase | ||
| 2363 | TGTCGAAGGACAGCCGAAAT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sedlák, J.; Přibylová, J.; Koloňuk, I.; Špak, J.; Lenz, O.; Semerák, M. Elimination of Solanum nigrum ilarvirus 1 and Apple Hammerhead Viroid from Apple Cultivars Using Antivirals Ribavirin, Rimantadine, and Zidovudine. Viruses 2023, 15, 1684. https://doi.org/10.3390/v15081684
Sedlák J, Přibylová J, Koloňuk I, Špak J, Lenz O, Semerák M. Elimination of Solanum nigrum ilarvirus 1 and Apple Hammerhead Viroid from Apple Cultivars Using Antivirals Ribavirin, Rimantadine, and Zidovudine. Viruses. 2023; 15(8):1684. https://doi.org/10.3390/v15081684
Chicago/Turabian StyleSedlák, Jiří, Jaroslava Přibylová, Igor Koloňuk, Josef Špak, Ondřej Lenz, and Matěj Semerák. 2023. "Elimination of Solanum nigrum ilarvirus 1 and Apple Hammerhead Viroid from Apple Cultivars Using Antivirals Ribavirin, Rimantadine, and Zidovudine" Viruses 15, no. 8: 1684. https://doi.org/10.3390/v15081684
APA StyleSedlák, J., Přibylová, J., Koloňuk, I., Špak, J., Lenz, O., & Semerák, M. (2023). Elimination of Solanum nigrum ilarvirus 1 and Apple Hammerhead Viroid from Apple Cultivars Using Antivirals Ribavirin, Rimantadine, and Zidovudine. Viruses, 15(8), 1684. https://doi.org/10.3390/v15081684






