Zoonotic Animal Influenza Virus and Potential Mixing Vessel Hosts
Abstract
1. Influenza Viruses
1.1. Classification and Structure
1.2. Virus Evolution (Shift and Drift)
2. Host Range of (Zoonotic) Influenza A Virus
2.1. Birds
2.1.1. Wild Birds
2.1.2. Domestic Birds
2.2. Mammals
2.2.1. Humans
2.2.2. Pigs
2.2.3. Other Mammals
3. Zoonotic Influenza Viruses
3.1. Animal Influenza A Viruses with High Zoonotic Potentials
3.1.1. Zoonotic Avian Influenza A Virus
Confirmed Human Infections
| Subtype | Year of First Human Identification | Year of Last Human Identification | Number (Fatal Cases) | Country | References |
|---|---|---|---|---|---|
| H3N8 | 2022 | 2022 | 2 | China | [80,81] |
| H5N1 | 1997 | 1997 | 18 (6) | Hong Kong | [82] |
| 2003 | 2023 | 868 (457) | Many * | [80,83] | |
| 2022 | 2023 | 5 | UK, USA, Spain, Ecuador | [84,85,86] | |
| H5N6 | 2014 | 2021 | 83 (33) | China | [83] |
| H5N8 | 2020 | 2020 | 7 | Russia | [87] |
| H6N1 | 2013 | 2013 | 1 | Taiwan | [88] |
| H7N2 | 2002 | 2016 | 8 | UK, USA | [88] |
| H7N3 | 2004 | 2012 | 5 | Canada, Mexico, UK | [88,89] |
| H7N4 | 2018 | 2018 | 1 | China | [83] |
| H7N7 | 1959 | 2013 | 96 (1) | USA, Australia, Netherlands, Italy, UK | [88,90] |
| H7N9 | 2013 | 2017 | 1568 (616) | China, Taiwan | [83] |
| H9N2 | 1998 | 2014 | 19 | China, Bangladesh, Hong Kong | [65] |
| 2015 | 2022 | 85 (2) | China, Cambodia, Egypt | [80,83] | |
| H10N3 | 2021 | 2022 | 2 | China | [80] |
| H10N7 | 2004 | 2010 | 4 | Egypt, Australia | [88] |
| H10N8 | 2013 | 2014 | 3 (2) | China | [88] |
| Total | From 1959 to 2023 | 2775 (1117) | |||
Limited Human-to-Human Transmission of AIV
Subclinical Infection with AIV Is More Prevalent Than Laboratory Confirmed Infections
3.1.2. Zoonotic Swine Influenza A Virus (swIAV)
3.2. Animal Influenza A Viruses with Low Zoonotic Potential
3.2.1. Bovine Influenza D Virus
3.2.2. Equine Influenza A Virus (EIV)
3.2.3. Canine Influenza A Virus
3.2.4. Bat Influenza A Virus
4. Potential “Mixing Vessel” Hosts
4.1. Humans
4.2. Pigs
4.3. Ferrets
4.4. Minks
4.5. Seals
4.6. Dogs
4.7. Cats
4.8. Non-Human Primates (NHPs)
4.9. Raccoons (Procyon Lotor)
4.10. Camels
4.11. Plateau Pika (Ochotona Curzoniae)
4.12. Foxes
4.13. Bats
4.14. Horses
4.15. Other Mammals
4.16. Birds
4.16.1. Chickens
4.16.2. Turkeys
4.16.3. Guinea Fowls
4.16.4. Quails
4.16.5. Pheasants
4.16.6. Ducks and Geese
4.16.7. Pigeons
4.16.8. Emus
4.16.9. Partridges
4.16.10. Wild Birds
5. Summary and Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Kuhn, J.H.; Adkins, S.; Alioto, D.; Alkhovsky, S.V.; Amarasinghe, G.K.; Anthony, S.J.; Avšič-Županc, T.; Ayllón, M.A.; Bahl, J.; Balkema-Buschmann, A.; et al. 2020 taxonomic update for phylum Negarnaviricota (Riboviria: Orthornavirae), including the large orders Bunyavirales and Mononegavirales. Arch. Virology 2020, 165, 3023–3072. [Google Scholar] [CrossRef]
- Suarez, D.L. Influenza A virus. In Animal Influenza; John Wiley & Sons, Inc.: New York, NY, USA, 2016; pp. 1–30. [Google Scholar]
- Gamblin, S.J.; Skehel, J.J. Influenza hemagglutinin and neuraminidase membrane glycoproteins. J. Biol. Chem. 2010, 285, 28403–28409. [Google Scholar] [CrossRef]
- Ferhadian, D.; Contrant, M.; Printz-Schweigert, A.; Smyth, R.P.; Paillart, J.C.; Marquet, R. Structural and Functional Motifs in Influenza Virus RNAs. Front. Microbiol. 2018, 9, 559. [Google Scholar] [CrossRef]
- Morgan, C.; Rose, H.M.; Moore, D.H. Structure and development of viruses observed in the electron microscope. III. Influenza virus. J. Exp. Med. 1956, 104, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Vijayakrishnan, S.; Loney, C.; Jackson, D.; Suphamungmee, W.; Rixon, F.J.; Bhella, D. Cryotomography of budding influenza A virus reveals filaments with diverse morphologies that mostly do not bear a genome at their distal end. PLoS Pathog. 2013, 9, e1003413. [Google Scholar] [CrossRef]
- Bouvier, N.M.; Palese, P. The biology of influenza viruses. Vaccine 2008, 26 (Suppl. 4), D49–D53. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Fu, X.L.; Li, G.R.; Kerlin, F.; Veit, M. Novel Influenza D virus: Epidemiology, pathology, evolution and biological characteristics. Virulence 2017, 8, 1580–1591. [Google Scholar] [CrossRef]
- Dou, D.; Revol, R.; Ostbye, H.; Wang, H.; Daniels, R. Influenza A Virus Cell Entry, Replication, Virion Assembly and Movement. Front. Immunol. 2018, 9, 1581. [Google Scholar] [CrossRef]
- Froggatt, H.M.; Burke, K.N.; Chaparian, R.R.; Miranda, H.A.; Zhu, X.; Chambers, B.S.; Heaton, N.S. Influenza A virus segments five and six can harbor artificial introns allowing expanded coding capacity. PLoS Pathog. 2021, 17, e1009951. [Google Scholar] [CrossRef] [PubMed]
- Dubois, J.; Terrier, O.; Rosa-Calatrava, M. Influenza viruses and mRNA splicing: Doing more with less. mBio 2014, 5, e00070-14. [Google Scholar] [CrossRef]
- Lowen, A.C. Constraints, Drivers, and Implications of Influenza A Virus Reassortment. Annu. Rev. Virol. 2017, 4, 105–121. [Google Scholar] [CrossRef]
- Herfst, S.; Imai, M.; Kawaoka, Y.; Fouchier, R.A. Avian influenza virus transmission to mammals. Curr. Top Microbiol. Immunol. 2014, 385, 137–155. [Google Scholar] [PubMed]
- Petrova, V.N.; Russell, C.A. The evolution of seasonal influenza viruses. Nat. Rev. Microbiol. 2018, 16, 47–60. [Google Scholar] [CrossRef]
- Stallknecht, D.E.; Brown, J.D. Ecology of Avian Influenza in Wild Birds. In Avian Influenza; John Wiley & Sons, Inc.: New York, NY, USA, 2008; pp. 43–58. [Google Scholar]
- Stallknecht, D.E.; Brown, J.D. Wild bird infections and the ecology of avian influenza viruses. In Animal Influenza; John Wiley & Sons, Inc.: New York, NY, USA; pp. 153–176.
- Olsen, B.; Munster, V.J.; Wallensten, A.; Waldenstrom, J.; Osterhaus, A.D.; Fouchier, R.A. Global patterns of influenza a virus in wild birds. Science 2006, 312, 384–388. [Google Scholar] [CrossRef]
- Bodewes, R.; Kuiken, T. Chapter Twelve—Changing Role of Wild Birds in the Epidemiology of Avian Influenza A Viruses. In Advances in Virus Research; Kielian, M., Mettenleiter, T.C., Roossinck, M.J., Eds.; Academic Press: Cambridge, MA, USA, 2018; Volume 100, pp. 279–307. [Google Scholar]
- Webster, R.G.; Yakhno, M.; Hinshaw, V.S.; Bean, W.J.; Murti, K.G. Intestinal influenza: Replication and characterization of influenza viruses in ducks. Virology 1978, 84, 268–278. [Google Scholar] [CrossRef]
- Venkatesh, D.; Poen, M.J.; Bestebroer, T.M.; Scheuer, R.D.; Vuong, O.; Chkhaidze, M.; Machablishvili, A.; Mamuchadze, J.; Ninua, L.; Fedorova, N.B.; et al. Avian Influenza Viruses in Wild Birds: Virus Evolution in a Multihost Ecosystem. J. Virol. 2018, 92, e00433-18. [Google Scholar] [CrossRef] [PubMed]
- Henaux, V.; Samuel, M.D.; Dusek, R.J.; Fleskes, J.P.; Ip, H.S. Presence of avian influenza viruses in waterfowl and wetlands during summer 2010 in California: Are resident birds a potential reservoir? PLoS ONE 2012, 7, e31471. [Google Scholar] [CrossRef]
- Wille, M.; Latorre-Margalef, N.; Tolf, C.; Halpin, R.; Wentworth, D.; Fouchier, R.A.M.; Raghwani, J.; Pybus, O.G.; Olsen, B.; Waldenstrom, J. Where do all the subtypes go? Temporal dynamics of H8-H12 influenza A viruses in waterfowl. Virus Evol. 2018, 4, vey025. [Google Scholar] [CrossRef]
- Ramey, A.M.; Hill, N.J.; DeLiberto, T.J.; Gibbs, S.E.J.; Hopkins, M.C.; Lang, A.S.; Poulson, R.L.; Prosser, D.J.; Sleeman, J.M.; Stallknecht, D.E.; et al. Highly pathogenic avian influenza is an emerging disease threat to wild birds in North America. J. Wildl. Manag. 2022, 86, e22171. [Google Scholar] [CrossRef]
- Gass, J.D., Jr.; Kellogg, H.K.; Hill, N.J.; Puryear, W.B.; Nutter, F.B.; Runstadler, J.A. Epidemiology and Ecology of Influenza A Viruses among Wildlife in the Arctic. Viruses 2022, 14, 1531. [Google Scholar] [CrossRef] [PubMed]
- Pohlmann, A.; King, J.; Fusaro, A.; Zecchin, B.; Banyard, A.C.; Brown, I.H.; Byrne, A.M.P.; Beerens, N.; Liang, Y.; Heutink, R.; et al. Has Epizootic Become Enzootic? Evidence for a Fundamental Change in the Infection Dynamics of Highly Pathogenic Avian Influenza in Europe, 2021. mBio 2022, 13, e0060922. [Google Scholar] [CrossRef]
- USDA, 2022–2023 Detections of Highly Pathogenic Avian Influenza in Mammals. 2023. Available online: https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/avian/avian-influenza/hpai-2022/2022-hpai-mammals (accessed on 8 February 2023).
- Alexander, D.J. A review of avian influenza in different bird species. Vet. Microbiol. 2000, 74, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.H.; Li, J.; Li, S.Q.; Fu, G.H.; Jin, T.; Zhang, C.; Yang, Y.C.; Ma, Z.H.; Tian, W.X.; Li, J.D.; et al. Dominant subtype switch in avian influenza viruses during 2016–2019 in China. Nat. Commun. 2020, 11, 5909. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.H.; Chen, Q.; Tan, M.; Liu, J.; Li, X.Y.; Yang, L.; Shu, Y.L.; Wang, D.Y.; Zhu, W.F. Epidemiology, evolution, and biological characteristics of H6 avian influenza viruses in China. Emerg. Microbes Infect. 2023, 12, 2151380. [Google Scholar] [CrossRef]
- Swayne, D.E.; Sims, L.D. Influenza. In Diseases of Poultry, 4th ed.; Swayne, D.E.B.M., Logue, C., McDougald, L.R., Nair, V., Suarez, D.L., Eds.; Wiley Publishing: Ames, IA, USA, 2020; pp. 210–256. [Google Scholar]
- Verhagen, J.H.; Fouchier, R.A.M.; Lewis, N. Highly Pathogenic Avian Influenza Viruses at the Wild-Domestic Bird Interface in Europe: Future Directions for Research and Surveillance. Viruses 2021, 13, 212. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Feng, H.; Xu, J.; Zhao, D.; Shi, J.; Li, Y.; Deng, G.; Jiang, Y.; Li, X.; Zhu, P.; et al. The PA protein directly contributes to the virulence of H5N1 avian influenza viruses in domestic ducks. J. Virol. 2011, 85, 2180–2188. [Google Scholar] [CrossRef]
- Scheibner, D.; Blaurock, C.; Mettenleiter, T.C.; Abdelwhab, E.M. Virulence of three European highly pathogenic H7N1 and H7N7 avian influenza viruses in Pekin and Muscovy ducks. BMC Vet. Res. 2019, 15, 142. [Google Scholar] [CrossRef]
- Grund, C.; Hoffmann, D.; Ulrich, R.; Naguib, M.; Schinkothe, J.; Hoffmann, B.; Harder, T.; Saenger, S.; Zscheppang, K.; Tonnies, M.; et al. A novel European H5N8 influenza A virus has increased virulence in ducks but low zoonotic potential. Emerg. Microbes Infect. 2018, 7, 132. [Google Scholar] [CrossRef]
- Pantin-Jackwood, M.; Swayne, D.E.; Smith, D.; Shepherd, E. Effect of species, breed and route of virus inoculation on the pathogenicity of H5N1 highly pathogenic influenza (HPAI) viruses in domestic ducks. Vet. Res. 2013, 44, 62. [Google Scholar] [CrossRef]
- Tang, Y.; Wu, P.; Peng, D.; Wang, X.; Wan, H.; Zhang, P.; Long, J.; Zhang, W.; Li, Y.; Wang, W.; et al. Characterization of duck H5N1 influenza viruses with differing pathogenicity in mallard (Anas platyrhynchos) ducks. Avian Pathol. 2009, 38, 457–467. [Google Scholar] [CrossRef]
- Cagle, C.; To, T.L.; Nguyen, T.; Wasilenko, J.; Adams, S.C.; Cardona, C.J.; Spackman, E.; Suarez, D.L.; Pantin-Jackwood, M.J. Pekin and Muscovy ducks respond differently to vaccination with a H5N1 highly pathogenic avian influenza (HPAI) commercial inactivated vaccine. Vaccine 2011, 29, 6549–6557. [Google Scholar] [CrossRef]
- Cagle, C.; Wasilenko, J.; Adams, S.C.; Cardona, C.J.; To, T.L.; Nguyen, T.; Spackman, E.; Suarez, D.L.; Smith, D.; Shepherd, E.; et al. Differences in pathogenicity, response to vaccination, and innate immune responses in different types of ducks infected with a virulent H5N1 highly pathogenic avian influenza virus from Vietnam. Avian Dis. 2012, 56, 479–487. [Google Scholar] [CrossRef] [PubMed]
- Pantin-Jackwood, M.J.; Smith, D.M.; Wasilenko, J.L.; Cagle, C.; Shepherd, E.; Sarmento, L.; Kapczynski, D.R.; Afonso, C.L. Effect of age on the pathogenesis and innate immune responses in Pekin ducks infected with different H5N1 highly pathogenic avian influenza viruses. Virus Res. 2012, 167, 196–206. [Google Scholar] [CrossRef]
- Blaurock, C.; Pfaff, F.; Scheibner, D.; Hoffmann, B.; Fusaro, A.; Monne, I.; Mettenleiter, T.C.; Breithaupt, A.; Abdelwhab, E.M. Evidence for Different Virulence Determinants and Host Response after Infection of Turkeys and Chickens with Highly Pathogenic H7N1 Avian Influenza Virus. J. Virol. 2022, 96, e0099422. [Google Scholar] [CrossRef] [PubMed]
- Scheibner, D.; Ulrich, R.; Fatola, O.I.; Graaf, A.; Gischke, M.; Salaheldin, A.H.; Harder, T.C.; Veits, J.; Mettenleiter, T.C.; Abdelwhab, E.M. Variable impact of the hemagglutinin polybasic cleavage site on virulence and pathogenesis of avian influenza H7N7 virus in chickens, turkeys and ducks. Sci. Rep. 2019, 9, 11556. [Google Scholar] [CrossRef]
- Swayne, D.E. Impact of vaccines and vaccination on global control of avian influenza. Avian Dis. 2012, 56 (Suppl. 4), 818–828. [Google Scholar] [CrossRef]
- Krammer, F.; Smith, G.J.D.; Fouchier, R.A.M.; Peiris, M.; Kedzierska, K.; Doherty, P.C.; Palese, P.; Shaw, M.L.; Treanor, J.; Webster, R.G.; et al. Influenza. Nat. Rev. Dis. Prim. 2018, 4, 3. [Google Scholar] [CrossRef] [PubMed]
- Dunning, J.; Thwaites, R.S.; Openshaw, P.J.M. Seasonal and pandemic influenza: 100 years of progress, still much to learn. Mucosal. Immunol. 2020, 13, 566–573. [Google Scholar] [CrossRef]
- Uyeki, T.M.; Hui, D.S.; Zambon, M.; Wentworth, D.E.; Monto, A.S. Influenza. Lancet 2022, 400, 693–706. [Google Scholar] [CrossRef]
- Gu, Y.; Zuo, X.; Zhang, S.; Ouyang, Z.; Jiang, S.; Wang, F.; Wang, G. The Mechanism behind Influenza Virus Cytokine Storm. Viruses 2021, 13, 1362. [Google Scholar] [CrossRef]
- Fiore, A.E.; Uyeki, T.M.; Broder, K.; Finelli, L.; Euler, G.L.; Singleton, J.A.; Iskander, J.K.; Wortley, P.M.; Shay, D.K.; Bresee, J.S.; et al. Prevention and control of influenza with vaccines: Recommendations of the Advisory Committee on Immunization Practices (ACIP), 2010. MMWR Recomm. Rep. 2010, 59, 1–62. [Google Scholar] [PubMed]
- Jones, J.C.; Yen, H.L.; Adams, P.; Armstrong, K.; Govorkova, E.A. Influenza antivirals and their role in pandemic preparedness. Antivir. Res. 2023, 210, 105499. [Google Scholar] [CrossRef]
- Van Reeth, K. Avian and swine influenza viruses: Our current understanding of the zoonotic risk. Vet. Res. 2007, 38, 243–260. [Google Scholar] [CrossRef]
- Bourret, V. Avian influenza viruses in pigs: An overview. Vet. J. 2018, 239, 7–14. [Google Scholar] [CrossRef]
- Hennig, C.; Graaf, A.; Petric, P.P.; Graf, L.; Schwemmle, M.; Beer, M.; Harder, T. Are pigs overestimated as a source of zoonotic influenza viruses? Porc. Health Manag. 2022, 8, 30. [Google Scholar] [CrossRef]
- Zhang, X.; Outlaw, C.; Olivier, A.K.; Woolums, A.; Epperson, W.; Wan, X.F. Pathogenesis of co-infections of influenza D virus and Mannheimia haemolytica in cattle. Vet. Microbiol. 2019, 231, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Dhama, K.; Karthik, K.; Khandia, R.; Munjal, A.; Khurana, S.K.; Chakraborty, S.; Malik, Y.S.; Virmani, N.; Singh, R.; et al. A Comprehensive Review on Equine Influenza Virus: Etiology, Epidemiology, Pathobiology, Advances in Developing Diagnostics, Vaccines, and Control Strategies. Front. Microbiol. 2018, 9, 1941. [Google Scholar] [CrossRef]
- Parrish, C.R.; Murcia, P.R.; Holmes, E.C. Influenza virus reservoirs and intermediate hosts: Dogs, horses, and new possibilities for influenza virus exposure of humans. J. Virol. 2015, 89, 2990–2994. [Google Scholar] [CrossRef]
- Guo, Y.; Wang, M.; Kawaoka, Y.; Gorman, O.; Ito, T.; Saito, T.; Webster, R.G. Characterization of a new avian-like influenza A virus from horses in China. Virology 1992, 188, 245–255. [Google Scholar] [CrossRef]
- Zhu, H.; Hughes, J.; Murcia, P.R. Origins and Evolutionary Dynamics of H3N2 Canine Influenza Virus. J. Virol. 2015, 89, 5406–5418. [Google Scholar] [CrossRef] [PubMed]
- Na, W.; Yeom, M.; Yuk, H.; Moon, H.; Kang, B.; Song, D. Influenza virus vaccine for neglected hosts: Horses and dogs. Clin. Exp. Vaccine Res. 2016, 5, 117–124. [Google Scholar] [CrossRef]
- de Vries, R.D.; Herfst, S.; Richard, M. Avian Influenza A Virus Pandemic Preparedness and Vaccine Development. Vaccines 2018, 6, 46. [Google Scholar] [CrossRef]
- Wallensten, A. Influenza virus in wild birds and mammals other than man. Microb. Ecol. Health Dis. 2009, 19, 122–139. [Google Scholar]
- Reperant, L.A.; Rimmelzwaan, G.F.; Kuiken, T. Avian influenza viruses in mammals. Rev. Sci. Tech. 2009, 28, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Root, J.; Shriner, S. Avian Influenza A Virus Associations in Wild, Terrestrial Mammals: A Review of Potential Synanthropic Vectors to Poultry Facilities. Viruses 2020, 12, 1352. [Google Scholar] [CrossRef]
- Kaplan, B.S.; Webby, R.J. The avian and mammalian host range of highly pathogenic avian H5N1 influenza. Virus Res. 2013, 178, 3–11. [Google Scholar] [CrossRef]
- Carnaccini, S.; Perez, D. H9 Influenza Viruses: An Emerging Challenge. Cold Spring Harb. Perspect. Med. 2019, 10, a038588. [Google Scholar] [CrossRef]
- He, W.T.; Hou, X.; Zhao, J.; Sun, J.; He, H.; Si, W.; Wang, J.; Jiang, Z.; Yan, Z.; Xing, G.; et al. Virome characterization of game animals in China reveals a spectrum of emerging pathogens. Cell 2022, 185, 1117–1129 e8. [Google Scholar] [CrossRef]
- Peacock, T.H.P.; James, J.; Sealy, J.E.; Iqbal, M. A Global Perspective on H9N2 Avian Influenza Virus. Viruses 2019, 11, 620. [Google Scholar] [CrossRef] [PubMed]
- Offeddu, V.; Cowling, B.J.; Malik Peiris, J.S. Interventions in live poultry markets for the control of avian influenza: A systematic review. One Health 2016, 2, 55–64. [Google Scholar] [CrossRef]
- Bos, M.E.H.; Beest, D.E.T.; van Boven, M.; Holle, M.R.D.R.V.; Meijer, A.; Bosman, A.; Mulder, Y.M.; Koopmans, M.P.G.; Stegeman, A. High Probability of Avian Influenza Virus (H7N7) Transmission from Poultry to Humans Active in Disease Control on Infected Farms. J. Infect. Dis. 2010, 201, 1390–1396. [Google Scholar] [CrossRef]
- Gill, J.S.; Webby, R.; Gilchrist, M.J.; Gray, G.C. Avian influenza among waterfowl hunters and wildlife professionals. Emerg. Infect. Dis. 2006, 12, 1284–1286. [Google Scholar] [CrossRef] [PubMed]
- Shafir, S.C.; Fuller, T.; Smith, T.B.; Rimoin, A.W. A national study of individuals who handle migratory birds for evidence of avian and swine-origin influenza virus infections. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2012, 54, 364–367. [Google Scholar] [CrossRef]
- Fasina, F.O.; Ifende, V.I.; Ajibade, A.A. Avian influenza A(H5N1) in humans: Lessons from Egypt. Euro Surveill 2010, 15, 19473. [Google Scholar] [CrossRef] [PubMed]
- Braga, E.M.; Van Kerkhove, M.D.; Mumford, E.; Mounts, A.W.; Bresee, J.; Ly, S.; Bridges, C.B.; Otte, J. Highly Pathogenic Avian Influenza (H5N1): Pathways of Exposure at the Animal-Human Interface, a Systematic Review. PLoS ONE 2011, 6, e14582. [Google Scholar]
- Uiprasertkul, M.; Puthavathana, P.; Sangsiriwut, K.; Pooruk, P.; Srisook, K.; Peiris, M.; Nicholls, J.M.; Chokephaibulkit, K.; Vanprapar, N.; Auewarakul, P. Influenza A H5N1 replication sites in humans. Emerg. Infect. Dis. 2005, 11, 1036–1041. [Google Scholar] [CrossRef]
- Korteweg, C.; Gu, J. Pathology, molecular biology, and pathogenesis of avian influenza A (H5N1) infection in humans. Am. J. Pathol. 2008, 172, 1155–1170. [Google Scholar] [CrossRef]
- Poovorawan, Y.; Pyungporn, S.; Prachayangprecha, S.; Makkoch, J. Global alert to avian influenza virus infection: From H5N1 to H7N9. Pathog. Glob Health 2013, 107, 217–223. [Google Scholar] [CrossRef]
- Li, Y.T.; Linster, M.; Mendenhall, I.H.; Su, Y.C.F.; Smith, G.J.D. Avian influenza viruses in humans: Lessons from past outbreaks. Br. Med. Bull 2019, 132, 81–95. [Google Scholar] [CrossRef]
- Liu, W.J.; Xiao, H.; Dai, L.; Liu, D.; Chen, J.; Qi, X.; Bi, Y.; Shi, Y.; Gao, G.F.; Liu, Y. Avian influenza A (H7N9) virus: From low pathogenic to highly pathogenic. Front. Med. 2021, 15, 507–527. [Google Scholar] [CrossRef] [PubMed]
- WHO. Avian Influenza Weekly Update Number 883. 2023. Available online: https://www.who.int/docs/default-source/wpro---documents/emergency/surveillance/avian-influenza/ai_20230217.pdf?sfvrsn=22ea0816_24 (accessed on 21 February 2023).
- WHO. Cumulative Number of Confirmed Human Cases for Avian Influenza A(H5N1) Reported to WHO, 2003–2023. 2023. Available online: https://cdn.who.int/media/docs/default-source/influenza/human-animal-interface-risk-assessments/cumulative-number-of-confirmed-human-cases-for-avian-influenza-a(h5n1)-reported-to-who--2003-2023.pdf?sfvrsn=a11e93cf_1&download=true (accessed on 21 February 2023).
- Watanabe, Y.; Ibrahim, M.S.; Suzuki, Y.; Ikuta, K. The changing nature of avian influenza A virus (H5N1). Trends Microbiol. 2012, 20, 11–20. [Google Scholar] [CrossRef]
- WHO. Human Infection with Avian Influenza A(H5) Viruses. Avian Influenza Weekly Update Number 884 from 24 February 2023. 2023. Available online: https://www.who.int/docs/default-source/wpro---documents/emergency/surveillance/avian-influenza/ai_20230224.pdf?sfvrsn=5f006f99_111 (accessed on 7 March 2023).
- Yang, R.; Sun, H.; Gao, F.; Luo, K.; Huang, Z.; Tong, Q.; Song, H.; Han, Q.; Liu, J.; Lan, Y.; et al. Human infection of avian influenza A H3N8 virus and the viral origins: A descriptive study. Lancet Microbe 2022, 3, e824–e834. [Google Scholar] [CrossRef] [PubMed]
- Chan, P.K. Outbreak of avian influenza A(H5N1) virus infection in Hong Kong in 1997. Clin. Infect. Dis. 2002, 34 (Suppl. 2), S58–S64. [Google Scholar] [CrossRef] [PubMed]
- WHO Avian Influenza Weekly Update Number 828. Available online: https://www.who.int/docs/default-source/wpro---documents/emergency/surveillance/avian-influenza/ai-20220121.pdf (accessed on 7 February 2022).
- Oliver, I.; Roberts, J.; Brown, C.S.; Byrne, A.M.; Mellon, D.; Hansen, R.; Banyard, A.C.; James, J.; Donati, M.; Porter, R.; et al. A case of avian influenza A(H5N1) in England, January 2022. Euro Surveill 2022, 27, 2200061. [Google Scholar] [CrossRef] [PubMed]
- CDC, U.S. Case of Human Avian Influenza A(H5) Virus Reported. CDC. 2022. Available online: https://www.cdc.gov/media/releases/2022/s0428-avian-flu.html (accessed on 25 August 2022).
- CDC, Highlights in the History of Avian Influenza (Bird Flu) Timeline—2020–2023. 2023. Available online: https://www.cdc.gov/flu/avianflu/timeline/avian-timeline-2020s.htm (accessed on 7 March 2023).
- Pyankova, O.G.; Susloparov, I.M.; Moiseeva, A.A.; Kolosova, N.P.; Onkhonova, G.S.; Danilenko, A.V.; Vakalova, E.V.; Shendo, G.L.; Nekeshina, N.N.; Noskova, L.N.; et al. Isolation of clade 2.3.4.4b A(H5N8), a highly pathogenic avian influenza virus, from a worker during an outbreak on a poultry farm, Russia, December 2020. Euro Surveill 2021, 26, 2100439. [Google Scholar] [CrossRef] [PubMed]
- Philippon, D.A.M.; Wu, P.; Cowling, B.J.; Lau, E.H.Y. Avian Influenza Human Infections at the Human-Animal Interface. J. Infect. Dis. 2020, 222, 528–537. [Google Scholar] [CrossRef]
- Freidl, G.S.; Meijer, A.; de Bruin, E.; de Nardi, M.; Munoz, O.; Capua, I.; Breed, A.C.; Harris, K.; Hill, A.; Kosmider, R.; et al. Influenza at the animal-human interface: A review of the literature for virological evidence of human infection with swine or avian influenza viruses other than A(H5N1). Euro Surveill 2014, 19, 20793. [Google Scholar] [CrossRef]
- Kalthoff, D.; Globig, A.; Beer, M. (Highly pathogenic) avian influenza as a zoonotic agent. Vet. Microbiol. 2010, 140, 237–245. [Google Scholar] [CrossRef]
- Katz, J.M.; Lim, W.; Bridges, C.B.; Rowe, T.; Hu-Primmer, J.; Lu, X.; Abernathy, R.A.; Clarke, M.; Conn, L.; Kwong, H.; et al. Antibody response in individuals infected with avian influenza A (H5N1) viruses and detection of anti-H5 antibody among household and social contacts. J. Infect. Dis. 1999, 180, 1763–1770. [Google Scholar] [CrossRef]
- Tran, T.H.; Nguyen, T.L.; Nguyen, T.D.; Luong, T.S.; Pham, P.M.; Nguyen, V.; Pham, T.S.; Vo, C.D.; Le, T.Q.; Ngo, T.T.; et al. Avian influenza A (H5N1) in 10 patients in Vietnam. N. Engl. J. Med. 2004, 350, 1179–1188. [Google Scholar]
- Ungchusak, K.; Auewarakul, P.; Dowell, S.F.; Kitphati, R.; Auwanit, W.; Puthavathana, P.; Uiprasertkul, M.; Boonnak, K.; Pittayawonganon, C.; Cox, N.J.; et al. Probable person-to-person transmission of avian influenza A (H5N1). N. Engl. J. Med. 2005, 352, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wu, P.; Pei, Y.; Tsang, T.K.; Gu, D.; Wang, W.; Zhang, J.; Horby, P.W.; Uyeki, T.M.; Cowling, B.J.; et al. Assessment of Human-to-Human Transmissibility of Avian Influenza A(H7N9) Virus Across 5 Waves by Analyzing Clusters of Case Patients in Mainland China, 2013–2017. Clin. Infect. Dis. 2019, 68, 623–631. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.J.; Ma, G.Y.; Yang, X.X.; Chen, S.H.; Gray, G.C.; Zhao, T.; Bao, J.; Zhou, J.J.; Qian, Y.H.; Lu, B.; et al. Avian Influenza A(H7N9) virus antibodies in close contacts of infected persons, China, 2013-2014. Emerg. Infect. Dis. 2015, 21, 709–711. [Google Scholar] [CrossRef] [PubMed]
- Buxton Bridges, C.; Katz, J.M.; Seto, W.H.; Chan, P.K.; Tsang, D.; Ho, W.; Mak, K.H.; Lim, W.; Tam, J.S.; Clarke, M.; et al. Risk of influenza A (H5N1) infection among health care workers exposed to patients with influenza A (H5N1), Hong Kong. J. Infect. Dis. 2000, 181, 344–348. [Google Scholar] [CrossRef]
- Du Ry van Beest Holle, M.; Meijer, A.; Koopmans, M.; de Jager, C.M. Human-to-human transmission of avian influenza A/H7N7, The Netherlands, 2003. Euro Surveill 2005, 10, 264–268. [Google Scholar] [CrossRef]
- Koopmans, M.; Wilbrink, B.; Conyn, M.; Natrop, G.; van der Nat, H.; Vennema, H.; Meijer, A.; van Steenbergen, J.; Fouchier, R.; Osterhaus, A.; et al. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet 2004, 363, 587–593. [Google Scholar] [CrossRef]
- Qi, Y.P.; Ni, H.B.; Chen, X.L.; Li, S.H. Seroprevalence of highly pathogenic avian influenza (H5N1) virus infection among humans in mainland China: A systematic review and meta-analysis. Transbound. Emerg. Dis. 2020, 67, 1861–1871. [Google Scholar] [CrossRef]
- Wang, W.; Chen, X.; Wang, Y.; Lai, S.; Yang, J.; Cowling, B.J.; Horby, P.W.; Uyeki, T.M.; Yu, H. Serological Evidence of Human Infection With Avian Influenza A(H7N9) Virus: A Systematic Review and Meta-analysis. J. Infect. Dis. 2020, 226, 70–82. [Google Scholar] [CrossRef]
- APPENDIX 1: Evidence Review on Occupational Exposure of Swine and Poultry Workers: An Advisory Committee Statement (ACS) National Advisory Committee on Immunization (NACI). Can Commun. Dis. Rep. 2013, 39, 1–47. [CrossRef] [PubMed]
- Khan, S.U.; Anderson, B.D.; Heil, G.L.; Liang, S.; Gray, G.C. A Systematic Review and Meta-Analysis of the Seroprevalence of Influenza A(H9N2) Infection Among Humans. J. Infect. Dis. 2015, 212, 562–569. [Google Scholar] [CrossRef]
- Sikkema, R.S.; Freidl, G.S.; de Bruin, E.; Koopmans, M. Weighing serological evidence of human exposure to animal influenza viruses—a literature review. Euro Surveill 2016, 21, 30388. [Google Scholar] [CrossRef]
- Qi, Y.; Guo, W.; Liu, C.; Li, W.; Gu, Y.; Li, S.; Chen, X. Seroprevalence of influenza A (H9N2) virus infection among humans in China: A meta-analysis. Microb. Pathog. 2021, 155, 104881. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Wang, W.; Wang, Y.; Lai, S.; Yang, J.; Cowling, B.J.; Horby, P.W.; Uyeki, T.M.; Yu, H. Serological evidence of human infections with highly pathogenic avian influenza A(H5N1) virus: A systematic review and meta-analysis. BMC Med. 2020, 18, 377. [Google Scholar] [CrossRef] [PubMed]
- Toner, E.S.; Adalja, A.A.; Nuzzo, J.B.; Inglesby, T.V.; Henderson, D.A.; Burke, D.S. Assessment of serosurveys for H5N1. Clin. Infect. Dis. 2013, 56, 1206–1212. [Google Scholar] [CrossRef]
- Yang, S.; Chen, Y.; Cui, D.; Yao, H.; Lou, J.; Huo, Z.; Xie, G.; Yu, F.; Zheng, S.; Yang, Y.; et al. Avian-Origin Influenza A(H7N9) Infection in Influenza A(H7N9)–Affected Areas of China: A Serological Study. J. Infect. Dis. 2013, 209, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.C.; Krueger, W.S.; Chum, C.; Putnam, S.D.; Wierzba, T.F.; Heil, G.L.; Anderson, B.D.; Yasuda, C.Y.; Williams, M.; Kasper, M.R.; et al. Little evidence of subclinical avian influenza virus infections among rural villagers in Cambodia. PLoS ONE 2014, 9, e97097. [Google Scholar] [CrossRef] [PubMed]
- Khuntirat, B.P.; Yoon, I.K.; Blair, P.J.; Krueger, W.S.; Chittaganpitch, M.; Putnam, S.D.; Supawat, K.; Gibbons, R.V.; Pattamadilok, S.; Sawanpanyalert, P.; et al. Evidence for subclinical avian influenza virus infections among rural Thai villagers. Clin. Infect. Dis. 2011, 53, e107–e116. [Google Scholar] [CrossRef] [PubMed]
- Okoye, J.O.; Eze, D.C.; Krueger, W.S.; Heil, G.L.; White, S.K.; Merrill, H.R.; Gray, G.C. Evidence for Subclinical H5N1 Avian Influenza Infections Among Nigerian Poultry Workers. J. Med. Virol. 2014, 86, 2070–2075. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Wulandari, L.; Poetranto, E.D.; Setyoningrum, R.A.; Yudhawati, R.; Sholikhah, A.; Nastri, A.M.; Poetranto, A.L.; Candra, A.Y.; Puruhito, E.F.; et al. Seroevidence for a High Prevalence of Subclinical Infection With Avian Influenza A(H5N1) Virus Among Workers in a Live-Poultry Market in Indonesia. J. Infect. Dis. 2016, 214, 1929–1936. [Google Scholar] [CrossRef] [PubMed]
- Quan, C.; Wang, Q.; Zhang, J.; Zhao, M.; Dai, Q.; Huang, T.; Zhang, Z.; Mao, S.; Nie, Y.; Liu, J.; et al. Avian Influenza A Viruses among Occupationally Exposed Populations, China, 2014–2016. Emerg. Infect. Dis. 2019, 25, 2215–2225. [Google Scholar] [CrossRef]
- Gray, G.C.; McCarthy, T.; Capuano, A.W.; Setterquist, S.F.; Alavanja, M.C.; Lynch, C.F. Evidence for avian influenza A infections among Iowa’s agricultural workers. Influenza Other Respir Viruses 2008, 2, 61–69. [Google Scholar] [CrossRef] [PubMed]
- De Marco, M.A.; Delogu, M.; Facchini, M.; Di Trani, L.; Boni, A.; Cotti, C.; Graziosi, G.; Venturini, D.; Regazzi, D.; Ravaioli, V.; et al. Serologic Evidence of Occupational Exposure to Avian Influenza Viruses at the Wildfowl/Poultry/Human Interface. Microorganisms 2021, 9, 2153. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.B.; Su, S.; Xiao, C.C.; Zhou, P.; Li, H.N.; Ke, C.W.; Gray, G.C.; Zhang, G.H.; Liao, M. Antibodies against H10N8 avian influenza virus among animal workers in Guangdong Province before November 30, 2013, when the first human H10N8 case was recognized. BMC Med. 2014, 12, 205. [Google Scholar] [CrossRef] [PubMed]
- Kayali, G.; Ortiz, E.J.; Chorazy, M.L.; Gray, G.C. Evidence of previous avian influenza infection among US turkey workers. Zoonoses Public Health 2010, 57, 265–272. [Google Scholar] [CrossRef] [PubMed]
- To, K.K.; Hung, I.F.; Lui, Y.M.; Mok, F.K.; Chan, A.S.; Li, P.T.; Wong, T.L.; Ho, D.T.; Chan, J.F.; Chan, K.H.; et al. Ongoing transmission of avian influenza A viruses in Hong Kong despite very comprehensive poultry control measures: A prospective seroepidemiology study. J. Infect. 2016, 72, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Di, B.; Zhou, D.H.; Zheng, B.J.; Jing, H.; Lin, Y.P.; Liu, Y.F.; Wu, X.W.; Qin, P.Z.; Wang, Y.L.; et al. Food markets with live birds as source of avian influenza. Emerg. Infect. Dis. 2006, 12, 1773–1775. [Google Scholar] [CrossRef]
- Liao, Q.; Bai, T.; Zhou, L.; Vong, S.; Guo, J.; Lv, W.; Dong, L.; Xiang, N.; Li, Z.; Huai, Y.; et al. Seroprevalence of antibodies to highly pathogenic avian influenza A (H5N1) virus among close contacts exposed to H5N1 cases, China, 2005–2008. PLoS ONE 2013, 8, e71765. [Google Scholar] [CrossRef]
- Huo, X.; Zu, R.; Qi, X.; Qin, Y.; Li, L.; Tang, F.; Hu, Z.; Zhu, F. Seroprevalence of avian influenza A (H5N1) virus among poultry workers in Jiangsu Province, China: An observational study. BMC Infect. Dis. 2012, 12, 93. [Google Scholar] [CrossRef]
- Wang, Y.; Kong, W.H.; Zhu, H.H.; Luo, T.Y.; Lin, X.M.; Yu, B.; Chen, B.H.; Hu, Q.; Peng, J.S. Investigation on the distribution of avian influenza virus in external environment and the level of H5N1 antibody in poultry-exposed population in Wuhan. Zhonghua Yu Fang Yi Xue Za Zhi 2011, 45, 1082–1085. [Google Scholar]
- Wang, X.; Fang, S.; Lu, X.; Xu, C.; Cowling, B.J.; Tang, X.; Peng, B.; Wu, W.; He, J.; Tang, Y.; et al. Seroprevalence to avian influenza A(H7N9) virus among poultry workers and the general population in southern China: A longitudinal study. Clin. Infect. Dis. 2014, 59, e76–e83. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Ma, C.; Cui, S.; Zhang, D.; Shi, W.; Pan, Y.; Sun, Y.; Lu, G.; Peng, X.; Zhao, J.; et al. Avian influenza A(H7N9) and (H5N1) infections among poultry and swine workers and the general population in Beijing, China, 2013–2015. Sci. Rep. 2016, 6, 33877. [Google Scholar] [CrossRef]
- Ma, M.J.; Zhao, T.; Chen, S.H.; Xia, X.; Yang, X.X.; Wang, G.L.; Fang, L.Q.; Ma, G.Y.; Wu, M.N.; Qian, Y.H.; et al. Avian Influenza A Virus Infection among Workers at Live Poultry Markets, China, 2013-2016. Emerg. Infect. Dis. 2018, 24, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Kwon, D.; Lee, J.Y.; Choi, W.; Choi, J.H.; Chung, Y.S.; Lee, N.J.; Cheong, H.M.; Katz, J.M.; Oh, H.B.; Cho, H.; et al. Avian influenza a (H5N1) virus antibodies in poultry cullers, South Korea, 2003–2004. Emerg. Infect. Dis. 2012, 18, 986–988. [Google Scholar] [CrossRef] [PubMed]
- Uyeki, T.M.; Nguyen, D.C.; Rowe, T.; Lu, X.H.; Hu-Primmer, J.; Huynh, L.P.; Hang, N.L.K.; Katz, J.M. Seroprevalence of Antibodies to Avian Influenza A (H5) and A (H9) Viruses among Market Poultry Workers, Hanoi, Vietnam, 2001. PLoS ONE 2012, 7, e43948. [Google Scholar] [CrossRef]
- Powell, T.J.; Fox, A.; Peng, Y.; Quynh Mai le, T.; Lien, V.T.; Hang, N.L.; Wang, L.; Lee, L.Y.; Simmons, C.P.; McMichael, A.J.; et al. Identification of H5N1-specific T-cell responses in a high-risk cohort in vietnam indicates the existence of potential asymptomatic infections. J. Infect. Dis. 2012, 205, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Dung, T.C.; Dinh, P.N.; Nam, V.S.; Tan, L.M.; Hang Nle, K.; Thanh le, T.; Mai le, Q. Seroprevalence survey of avian influenza A(H5N1) among live poultry market workers in northern Viet Nam, 2011. West. Pac Surveill Respons. J. 2014, 5, 21–26. [Google Scholar]
- Horm, S.V.; Tarantola, A.; Rith, S.; Ly, S.; Gambaretti, J.; Duong, V.; Y, P.; Sorn, S.; Holl, D.; Allal, L.; et al. Intense circulation of A/H5N1 and other avian influenza viruses in Cambodian live-bird markets with serological evidence of sub-clinical human infections. Emerg. Microbes Infect. 2016, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ly, S.; Horwood, P.; Chan, M.; Rith, S.; Sorn, S.; Oeung, K.; Nguon, K.; Chan, S.; Y, P.; Parry, A.; et al. Seroprevalence and Transmission of Human Influenza A(H5N1) Virus before and after Virus Reassortment, Cambodia, 2006-2014. Emerg. Infect. Dis. 2017, 23, 300–303. [Google Scholar] [CrossRef]
- Cavailler, P.; Chu, S.; Ly, S.; Garcia, J.M.; Do, Q.H.; Bergeri, I.; Som, L.; Ly, S.; Sok, T.; Vong, S.; et al. Seroprevalence of anti-H5 antibody in rural Cambodia, 2007. J. Clin. Virol. 2010, 48, 123–126. [Google Scholar] [CrossRef]
- Vong, S.; Coghlan, B.; Mardy, S.; Holl, D.; Seng, H.; Ly, S.; Miller, M.J.; Buchy, P.; Froehlich, Y.; Dufourcq, J.B.; et al. Low frequency of poultry-to-human H5NI virus transmission, southern Cambodia, 2005. Emerg. Infect. Dis. 2006, 12, 1542–1547. [Google Scholar] [CrossRef] [PubMed]
- Vong, S.; Ly, S.; Van Kerkhove, M.D.; Achenbach, J.; Holl, D.; Buchy, P.; Sorn, S.; Seng, H.; Uyeki, T.M.; Sok, T.; et al. Risk factors associated with subclinical human infection with avian influenza A (H5N1) virus–Cambodia, 2006. J. Infect. Dis. 2009, 199, 1744–1752. [Google Scholar] [CrossRef]
- Ceyhan, M.; Yildirim, I.; Ferraris, O.; Bouscambert-Duchamp, M.; Frobert, E.; Uyar, N.; Tezer, H.; Oner, A.F.; Buzgan, T.; Torunoglu, M.A.; et al. Serosurveillance study on transmission of H5N1 virus during a 2006 avian influenza epidemic. Epidemiol. Infect. 2010, 138, 1274–1280. [Google Scholar] [CrossRef]
- Okoye, J.; Eze, D.; Krueger, W.S.; Heil, G.L.; Friary, J.A.; Gray, G.C. Serologic evidence of avian influenza virus infections among Nigerian agricultural workers. J. Med. Virol. 2013, 85, 670–676. [Google Scholar] [CrossRef]
- Gomaa, M.R.; Kayed, A.S.; Elabd, M.A.; Zeid, D.A.; Zaki, S.A.; El Rifay, A.S.; Sherif, L.S.; McKenzie, P.P.; Webster, R.G.; Webby, R.J.; et al. Avian influenza A(H5N1) and A(H9N2) seroprevalence and risk factors for infection among Egyptians: A prospective, controlled seroepidemiological study. J. Infect. Dis. 2015, 211, 1399–1407. [Google Scholar] [CrossRef]
- Gomaa, M.R.; El Rifay, A.S.; Abu Zeid, D.; Elabd, M.A.; Elabd, E.; Kandeil, A.; Shama, N.M.A.; Kamel, M.N.; Marouf, M.A.; Barakat, A.; et al. Incidence and Seroprevalence of Avian Influenza in a Cohort of Backyard Poultry Growers, Egypt, August 2015-March 2019. Emerg. Infect. Dis. 2020, 26, 2129–2136. [Google Scholar] [CrossRef]
- Huang, S.Y.; Yang, J.R.; Lin, Y.J.; Yang, C.H.; Cheng, M.C.; Liu, M.T.; Wu, H.S.; Chang, F.Y. Serological comparison of antibodies to avian influenza viruses, subtypes H5N2, H6N1, H7N3 and H7N9 between poultry workers and non-poultry workers in Taiwan in 2012. Epidemiol. Infect. 2015, 143, 2965–2974. [Google Scholar] [CrossRef] [PubMed]
- Venter, M.; Treurnicht, F.K.; Buys, A.; Tempia, S.; Samudzi, R.; McAnerney, J.; Jacobs, C.A.; Thomas, J.; Blumberg, L. Risk of Human Infections With Highly Pathogenic H5N2 and Low Pathogenic H7N1 Avian Influenza Strains During Outbreaks in Ostriches in South Africa. J. Infect. Dis. 2017, 216 (Suppl. 4), S512–S519. [Google Scholar] [CrossRef]
- Myers, K.P.; Setterquist, S.F.; Capuano, A.W.; Gray, G.C. Infection due to 3 avian influenza subtypes in United States veterinarians. Clin. Infect. Dis. 2007, 45, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Terebuh, P.; Adija, A.; Edwards, L.; Rowe, T.; Jenkins, S.; Kleene, J.; Fukuda, K.; Katz, J.M.; Bridges, C.B. Human infection with avian influenza A(H7N2) virus-Virginia, 2002. Influenza Other Respir Viruses 2018, 12, 529–532. [Google Scholar] [CrossRef] [PubMed]
- Puzelli, S.; Di Trani, L.; Fabiani, C.; Campitelli, L.; De Marco, M.A.; Capua, I.; Aguilera, J.F.; Zambon, M.; Donatelli, I. Serological analysis of serum samples from humans exposed to avian H7 influenza viruses in Italy between 1999 and 2003. J. Infect. Dis. 2005, 192, 1318–1322. [Google Scholar] [CrossRef]
- Di Trani, L.; Porru, S.; Bonfanti, L.; Cordioli, P.; Cesana, B.M.; Boni, A.; Di Carlo, A.S.; Arici, C.; Donatelli, I.; Tomao, P.; et al. Serosurvey Against H5 and H7 Avian Influenza Viruses in Italian Poultry Workers. Avian Dis. 2012, 56, 1068–1071. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.C.; Ferguson, D.D.; Lowther, P.E.; Heil, G.L.; Friary, J.A. A national study of US bird banders for evidence of avian influenza virus infections. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2011, 51, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.Y.; Lu, J.H.; Chen, W.Q.; Jiang, L.F.; Tan, B.Y.; Ling, W.H.; Zheng, B.J.; Sui, H.Y. Potential infections of H5N1 and H9N2 avian influenza do exist in Guangdong populations of China. Chin. Med. J. (Engl.) 2008, 121, 2050–2053. [Google Scholar] [CrossRef] [PubMed]
- Ahad, A.; Thornton, R.N.; Rabbani, M.; Yaqub, T.; Younus, M.; Muhammad, K.; Mahmood, A.; Shabbir, M.Z.; Kashem, M.A.; Islam, M.Z.; et al. Risk factors for H7 and H9 infection in commercial poultry farm workers in provinces within Pakistan. Prev. Vet. Med. 2014, 117, 610–614. [Google Scholar] [CrossRef]
- Ahad, A.; Rabbani, M.; Yaqub, T.; Younus, M.; Mahmood, A.; Shabbir, M.Z.; Fatima, Z.; Khalid, R.K.; Rasheed, M. Sero-surveillance to H9 and H7 avian influenza virus among poultry workers in Punjab Province, Pakistan. Pak. Vet. J. 2013, 33, 107–112. [Google Scholar]
- Gomaa, M.R.; Kandeil, A.; Kayed, A.S.; Elabd, M.A.; Zaki, S.A.; Abu Zeid, D.; El Rifay, A.S.; Mousa, A.A.; Farag, M.M.; McKenzie, P.P.; et al. Serological Evidence of Human Infection with Avian Influenza A H7virus in Egyptian Poultry Growers. PLoS ONE 2016, 11, e0155294. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Zhu, Y.; Zhao, B.; Li, J.; Liu, L.; Gu, K.; Zhang, W.; Su, H.; Teng, Z.; Tang, S.; et al. Limited human-to-human transmission of avian influenza A(H7N9) virus, Shanghai, China, March to April 2013. Euro Surveill 2014, 19, 20838. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, H.; Lu, J.; Luo, L.; Li, K.; Liu, Y.; Lau, E.H.; Di, B.; Wang, H.; Yang, Z.; et al. Asymptomatic, mild, and severe influenza A(H7N9) virus infection in humans, Guangzhou, China. Emerg. Infect. Dis. 2014, 20, 1535–1540. [Google Scholar] [CrossRef]
- de Bruin, E.; Zhang, X.; Ke, C.; Sikkema, R.; Koopmans, M. Serological evidence for exposure to avian influenza viruses within poultry workers in southern China. Zoonoses Public Health 2017, 64, e51–e59. [Google Scholar] [CrossRef]
- Blair, P.J.; Putnam, S.D.; Krueger, W.S.; Chum, C.; Wierzba, T.F.; Heil, G.L.; Yasuda, C.Y.; Williams, M.; Kasper, M.R.; Friary, J.A.; et al. Evidence for avian H9N2 influenza virus infections among rural villagers in Cambodia. J. Infect. Public Health 2013, 6, 69–79. [Google Scholar] [CrossRef]
- Krueger, W.S.; Khuntirat, B.; Yoon, I.K.; Blair, P.J.; Chittagarnpitch, M.; Putnam, S.D.; Supawat, K.; Gibbons, R.V.; Bhuddari, D.; Pattamadilok, S.; et al. Prospective study of avian influenza virus infections among rural Thai villagers. PLoS ONE 2013, 8, e72196. [Google Scholar] [CrossRef] [PubMed]
- Pawar, S.D.; Tandale, B.V.; Raut, C.G.; Parkhi, S.S.; Barde, T.D.; Gurav, Y.K.; Kode, S.S.; Mishra, A.C. Avian influenza H9N2 seroprevalence among poultry workers in Pune, India, 2010. PLoS ONE 2012, 7, e36374. [Google Scholar] [CrossRef]
- Khurelbaatar, N.; Krueger, W.S.; Heil, G.L.; Darmaa, B.; Ulziimaa, D.; Tserennorov, D.; Baterdene, A.; Anderson, B.D.; Gray, G.C. Little evidence of avian or equine influenza virus infection among a cohort of Mongolian adults with animal exposures, 2010–2011. PLoS ONE 2014, 9, e85616. [Google Scholar] [CrossRef]
- Tahir, M.F.; Abbas, M.A.; Ghafoor, T.; Dil, S.; Shahid, M.A.; Bullo, M.M.H.; Ain, Q.U.; Abbas Ranjha, M.; Khan, M.A.; Naseem, M.T. Seroprevalence and risk factors of avian influenza H9 virus among poultry professionals in Rawalpindi, Pakistan. J. Infect. Public Health 2020, 13, 414–417. [Google Scholar] [CrossRef]
- Rasheed, M.; Rehmani, S.F.; Iqbal, M.; Ahmad, A.; Akhtar, F.; Akhtar, R.; Bibi, T.; Aslam, H.B.; Afridi, S.Q.; Mukhtar, N. Seropositivity to Avian Influenza Virus Subtype H9N2 among Human Population of Selected Districts of Punjab, Pakistan. J. Inf. Mol. Biol. 2013, 1, 32–34. [Google Scholar]
- Chaudhry, M.; Webby, R.; Swayne, D.; Bin Rashid, H.; DeBeauchamp, J.; Killmaster, L.; Criado, M.F.; Lee, D.H.; Webb, A.; Yousaf, S.; et al. Avian influenza at animal-human interface: One-health challenge in live poultry retail stalls of Chakwal, Pakistan. Influenza Other Resp 2020, 14, 257–265. [Google Scholar] [CrossRef]
- Alizadeh, E.; Kheiri, M.T.; Bashar, R.; Tabatabaeian, M.; Seyed Masoud, H.; Mazaheri, V. Avian Influenza (H9N2) among poultry workers in Iran. Iran. J. Microbiol. 2009, 1, 3–6. [Google Scholar]
- Anvar, E.; Hosseini, S.M.; Kheiri, M.T.; Mazaheri, V.; Fazaei, K.; Shabani, M.; Alizadeh, E.; Tabatabaiean, M.; Torabi, A. Serological Survey of Avian Influenza (H9N2) Among Different Occupational Groups in Tehran and Qazvin Provinces in IR Iran. Jundishapur J. Microbiol. 2013, 6. [Google Scholar] [CrossRef]
- Heidari, A.; Mancin, M.; Nili, H.; Pourghanbari, G.H.; Lankarani, K.B.; Leardini, S.; Cattoli, G.; Monne, I.; Piccirillo, A. Serological evidence of H9N2 avian influenza virus exposure among poultry workers from Fars province of Iran. Virol. J. 2016, 13, 16. [Google Scholar] [CrossRef] [PubMed]
- Barbour, E.K.; Sagherian, V.K.; Sagherian, N.K.; Dankar, S.K.; Jaber, L.S.; Usayran, N.N.; Farran, M.T. Avian influenza outbreak in poultry in the Lebanon and transmission to neighbouring farmers and swine. Vet. Ital. 2006, 42, 77–85. [Google Scholar]
- Coman, A.; Maftei, D.N.; Krueger, W.S.; Heil, G.L.; Friary, J.A.; Chereches, R.M.; Sirlincan, E.; Bria, P.; Dragnea, C.; Kasler, I.; et al. Serological evidence for avian H9N2 influenza virus infections among Romanian agriculture workers. J. Infect. Public Health 2013, 6, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Coman, A.; Maftei, D.N.; Krueger, W.S.; Heil, G.L.; Chereches, R.M.; Sirlincan, E.; Bria, P.; Dragnea, C.; Kasler, I.; Valentine, M.A.; et al. A prospective study of Romanian agriculture workers for zoonotic influenza infections. PLoS ONE 2014, 9, e98248. [Google Scholar] [CrossRef] [PubMed]
- Myers, K.P.; Olsen, C.W.; Gray, G.C. Cases of swine influenza in humans: A review of the literature. Clin. Infect. Dis. 2007, 44, 1084–1088. [Google Scholar] [CrossRef]
- Smith, G.J.; Bahl, J.; Vijaykrishna, D.; Zhang, J.; Poon, L.L.; Chen, H.; Webster, R.G.; Peiris, J.S.; Guan, Y. Dating the emergence of pandemic influenza viruses. Proc. Natl. Acad. Sci. USA 2009, 106, 11709–11712. [Google Scholar] [CrossRef]
- Guan, Y.; Vijaykrishna, D.; Bahl, J.; Zhu, H.; Wang, J.; Smith, G.J. The emergence of pandemic influenza viruses. Protein Cell 2010, 1, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Kessler, S.; Harder, T.C.; Schwemmle, M.; Ciminski, K. Influenza A Viruses and Zoonotic Events-Are We Creating Our Own Reservoirs? Viruses 2021, 13, 2250. [Google Scholar] [CrossRef] [PubMed]
- Glud, H.A.; George, S.; Skovgaard, K.; Larsen, L.E. Zoonotic and reverse zoonotic transmission of viruses between humans and pigs. Apmis 2021, 129, 675–693. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Lee, J.H.; Erickson, G.; Goyal, S.M.; Joo, H.S.; Webster, R.G.; Webby, R.J. H3N2 influenza virus transmission from swine to turkeys, United States. Emerg. Infect. Dis. 2004, 10, 2156–2160. [Google Scholar] [CrossRef] [PubMed]
- Reid, S.M.; Cox, W.J.; Ceeraz, V.; Sutton, D.; Essen, S.C.; Howard, W.A.; Slomka, M.J.; Irvine, R.M.; Brown, I.H. First reported detection of influenza A (H1N1)pdm09 in turkeys in the United Kingdom. Avian Dis. 2012, 56 (Suppl. 4), 1062–1067. [Google Scholar] [CrossRef]
- Berhane, Y.; Ojkic, D.; Pople, N.; Lung, O.; Pasick, J. Reoccurrence of Suspected Human-to-Turkey Transmission of H1N1 Pandemic 2009 Virus in Turkey Breeder Flocks in Ontario and Manitoba, 2016. Transbound. Emerg. Dis. 2016, 63, 590–594. [Google Scholar] [CrossRef]
- Hause, B.M.; Ducatez, M.; Collin, E.A.; Ran, Z.; Liu, R.; Sheng, Z.; Armien, A.; Kaplan, B.; Chakravarty, S.; Hoppe, A.D.; et al. Isolation of a novel swine influenza virus from Oklahoma in 2011 which is distantly related to human influenza C viruses. PLoS Pathog. 2013, 9, e1003176. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.; Puig, A.; Bassols, M.; Fraile, L.; Armengol, R. Influenza D Virus: A Review and Update of Its Role in Bovine Respiratory Syndrome. Viruses 2022, 14, 2717. [Google Scholar] [CrossRef] [PubMed]
- Gaudino, M.; Chiapponi, C.; Moreno, A.; Zohari, S.; O’Donovan, T.; Quinless, E.; Sausy, A.; Oliva, J.; Salem, E.; Fusade-Boyer, M.; et al. Evolutionary and temporal dynamics of emerging influenza D virus in Europe (2009–2022). Virus Evol. 2022, 8, veac081. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Li, F.; Wang, D. The first decade of research advances in influenza D virus. J. Gen. Virol. 2021, 102, jgv001529. [Google Scholar] [CrossRef]
- Borkenhagen, L.K.; Mallinson, K.A.; Tsao, R.W.; Ha, S.J.; Lim, W.H.; Toh, T.H.; Anderson, B.D.; Fieldhouse, J.K.; Philo, S.E.; Chong, K.S.; et al. Surveillance for respiratory and diarrheal pathogens at the human-pig interface in Sarawak, Malaysia. PLoS ONE 2018, 13, e0201295. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, C.M.; Marchi, S.; Manini, I.; Kistner, O.; Li, F.; Piu, P.; Manenti, A.; Biuso, F.; Sreenivasan, C.; Druce, J.; et al. Influenza D Virus: Serological Evidence in the Italian Population from 2005 to 2017. Viruses 2019, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- White, S.K.; Ma, W.; McDaniel, C.J.; Gray, G.C.; Lednicky, J.A. Serologic evidence of exposure to influenza D virus among persons with occupational contact with cattle. J. Clin. Virol. Off. Publ. Pan Am. Soc. Clin. Virol. 2016, 81, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Qi, J.; Khedri, Z.; Diaz, S.; Yu, H.; Chen, X.; Varki, A.; Shi, Y.; Gao, G.F. An Open Receptor-Binding Cavity of Hemagglutinin-Esterase-Fusion Glycoprotein from Newly-Identified Influenza D Virus: Basis for Its Broad Cell Tropism. PLoS Pathog. 2016, 12, e1005411. [Google Scholar]
- Morens, D.M.; Taubenberger, J.K. An avian outbreak associated with panzootic equine influenza in 1872: An early example of highly pathogenic avian influenza? Influenza Other Respir Viruses 2010, 4, 373–377. [Google Scholar] [CrossRef]
- Xie, T.; Anderson, B.D.; Daramragchaa, U.; Chuluunbaatar, M.; Gray, G.C. A Review of Evidence that Equine Influenza Viruses Are Zoonotic. Pathogens 2016, 5, 50. [Google Scholar] [CrossRef]
- Chambers, T.M. Equine Influenza. Cold Spring Harb. Perspect Med. 2022, 12, a038331. [Google Scholar] [CrossRef] [PubMed]
- Crawford, P.C.; Dubovi, E.J.; Castleman, W.L.; Stephenson, I.; Gibbs, E.P.; Chen, L.; Smith, C.; Hill, R.C.; Ferro, P.; Pompey, J.; et al. Transmission of equine influenza virus to dogs. Science 2005, 310, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Yondon, M.; Zayat, B.; Nelson, M.I.; Heil, G.L.; Anderson, B.D.; Lin, X.; Halpin, R.A.; McKenzie, P.P.; White, S.K.; Wentworth, D.E.; et al. Equine influenza A(H3N8) virus isolated from Bactrian camel, Mongolia. Emerg. Infect. Dis. 2014, 20, 2144–2147. [Google Scholar] [CrossRef]
- Su, S.; Wang, L.; Fu, X.; He, S.; Hong, M.; Zhou, P.; Lai, A.; Gray, G.; Li, S. Equine influenza A(H3N8) virus infection in cats. Emerg. Infect. Dis. 2014, 20, 2096–2099. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Zhou, H.; Jiang, T.; Li, C.; Zhang, A.; Guo, X.; Zou, W.; Chen, H.; Jin, M. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in China. Arch. Virol. 2009, 154, 887–890. [Google Scholar] [CrossRef]
- Kasel, J.A.; Alford, R.H.; Knight, V.; Waddell, G.H.; Sigel, M.M. Experimental infection of human volunteers with equine influenza virus. Nature 1965, 206, 41–43. [Google Scholar] [CrossRef]
- Alford, R.H.; Kasel, J.A.; Lehrich, J.R.; Knight, V. Human responses to experimental infection with influenza A/Equi 2 virus. Am. J. Epidemiol. 1967, 86, 185–192. [Google Scholar] [CrossRef]
- Couch, R.B.; Douglas, R.G.; Kasel, J.A.; Riggs, S.; Knight, V. Production of the Influenza Syndrome in Man with Equine Influenza Virus. Nature 1969, 224, 512–514. [Google Scholar] [CrossRef]
- Daly, J.M.; Blunden, A.S.; Macrae, S.; Miller, J.; Bowman, S.J.; Kolodziejek, J.; Nowotny, N.; Smith, K.C. Transmission of equine influenza virus to English foxhounds. Emerg. Infect. Dis. 2008, 14, 461–464. [Google Scholar] [CrossRef]
- Frymus, T.; Belák, S.; Egberink, H.; Hofmann-Lehmann, R.; Marsilio, F.; Addie, D.D.; Boucraut-Baralon, C.; Hartmann, K.; Lloret, A.; Lutz, H.; et al. Influenza Virus Infections in Cats. Viruses 2021, 13, 1435. [Google Scholar] [CrossRef]
- Wasik, B.R.; Voorhees, I.E.H.; Parrish, C.R. Canine and Feline Influenza. Cold Spring Harb. Perspect Med. 2021, 11, a038562. [Google Scholar] [CrossRef] [PubMed]
- Voorhees, I.E.H.; Glaser, A.L.; Toohey-Kurth, K.; Newbury, S.; Dalziel, B.D.; Dubovi, E.J.; Poulsen, K.; Leutenegger, C.; Willgert, K.J.E.; Brisbane-Cohen, L.; et al. Spread of Canine Influenza A(H3N2) Virus, United States. Emerg. Infect. Dis. 2017, 23, 1950–1957. [Google Scholar] [CrossRef] [PubMed]
- Jeoung, H.-Y.; Lim, S.-I.; Shin, B.-H.; Lim, J.-A.; Song, J.-Y.; Song, D.-S.; Kang, B.-K.; Moon, H.-J.; An, D.-J. A novel canine influenza H3N2 virus isolated from cats in an animal shelter. Vet. Microbiol. 2013, 165, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Lyoo, K.S.; Kim, J.K.; Kang, B.; Moon, H.; Kim, J.; Song, M.; Park, B.; Kim, S.H.; Webster, R.G.; Song, D. Comparative analysis of virulence of a novel, avian-origin H3N2 canine influenza virus in various host species. Virus Res. 2015, 195, 135–140. [Google Scholar] [CrossRef]
- Lee, Y.N.; Lee, D.H.; Park, J.K.; Yuk, S.S.; Kwon, J.H.; Nahm, S.S.; Lee, J.B.; Park, S.Y.; Choi, I.S.; Song, C.S. Experimental infection and natural contact exposure of ferrets with canine influenza virus (H3N2). J. Gen. Virol. 2013, 94 (Pt 2), 293–297. [Google Scholar] [CrossRef]
- Martinez-Sobrido, L.; Blanco-Lobo, P.; Rodriguez, L.; Fitzgerald, T.; Zhang, H.; Nguyen, P.; Anderson, C.S.; Holden-Wiltse, J.; Bandyopadhyay, S.; Nogales, A.; et al. Characterizing Emerging Canine H3 Influenza Viruses. PLoS Pathog. 2020, 16, e1008409. [Google Scholar] [CrossRef]
- Ciminski, K.; Schwemmle, M. Bat-Borne Influenza A Viruses: An Awakening. Cold Spring Harb. Perspect Med. 2021, 11, a038612. [Google Scholar] [CrossRef]
- Ciminski, K.; Pfaff, F.; Beer, M.; Schwemmle, M. Bats reveal the true power of influenza A virus adaptability. PLoS Pathog. 2020, 16, e1008384. [Google Scholar] [CrossRef]
- Zhong, G.; Fan, S.; Hatta, M.; Nakatsu, S.; Walters, K.B.; Lopes, T.J.S.; Wang, J.I.; Ozawa, M.; Karasin, A.; Li, Y.; et al. Mutations in the Neuraminidase-Like Protein of Bat Influenza H18N11 Virus Enhance Virus Replication in Mammalian Cells, Mice, and Ferrets. J. Virol. 2020, 94, e01416–e01419. [Google Scholar] [CrossRef]
- Juozapaitis, M.; Aguiar Moreira, E.; Mena, I.; Giese, S.; Riegger, D.; Pohlmann, A.; Hoper, D.; Zimmer, G.; Beer, M.; Garcia-Sastre, A.; et al. An infectious bat-derived chimeric influenza virus harbouring the entry machinery of an influenza A virus. Nat. Commun. 2014, 5, 4448. [Google Scholar] [CrossRef]
- Ciminski, K.; Ran, W.; Gorka, M.; Lee, J.; Malmlov, A.; Schinköthe, J.; Eckley, M.; Murrieta, R.A.; Aboellail, T.A.; Campbell, C.L.; et al. Bat influenza viruses transmit among bats but are poorly adapted to non-bat species. Nat. Microbiol. 2019, 4, 2298–2309. [Google Scholar] [CrossRef] [PubMed]
- Giotis, E.S.; Carnell, G.; Young, E.F.; Ghanny, S.; Soteropoulos, P.; Wang, L.F.; Barclay, W.S.; Skinner, M.A.; Temperton, N. Entry of the bat influenza H17N10 virus into mammalian cells is enabled by the MHC class II HLA-DR receptor. Nat. Microbiol. 2019, 4, 2035–2038. [Google Scholar] [CrossRef] [PubMed]
- Karakus, U.; Thamamongood, T.; Ciminski, K.; Ran, W.; Günther, S.C.; Pohl, M.O.; Eletto, D.; Jeney, C.; Hoffmann, D.; Reiche, S.; et al. MHC class II proteins mediate cross-species entry of bat influenza viruses. Nature 2019, 567, 109–112. [Google Scholar] [CrossRef] [PubMed]
- Rogers, G.N.; D’Souza, B.L. Receptor binding properties of human and animal H1 influenza virus isolates. Virology 1989, 173, 317–322. [Google Scholar] [CrossRef]
- Beare, A.S.; Webster, R.G. Replication of Avian Influenza-Viruses in Humans. Arch. Virol. 1991, 119, 37–42. [Google Scholar] [CrossRef]
- Li, X.; Shi, J.; Guo, J.; Deng, G.; Zhang, Q.; Wang, J.; He, X.; Wang, K.; Chen, J.; Li, Y.; et al. Genetics, receptor binding property, and transmissibility in mammals of naturally isolated H9N2 Avian Influenza viruses. PLoS Pathog. 2014, 10, e1004508. [Google Scholar] [CrossRef]
- Liu, Y.; Li, S.; Sun, H.; Pan, L.; Cui, X.; Zhu, X.; Feng, Y.; Li, M.; Yu, Y.; Wu, M.; et al. Variation and Molecular Basis for Enhancement of Receptor Binding of H9N2 Avian Influenza Viruses in China Isolates. Front. Microbiol. 2020, 11, 602124. [Google Scholar] [CrossRef]
- Guo, J.; Wang, Y.; Zhao, C.; Gao, X.; Zhang, Y.; Li, J.; Wang, M.; Zhang, H.; Liu, W.; Wang, C.; et al. Molecular characterization, receptor binding property, and replication in chickens and mice of H9N2 avian influenza viruses isolated from chickens, peafowls, and wild birds in eastern China. Emerg. Microbes Infect. 2021, 10, 2098–2112. [Google Scholar] [CrossRef]
- Yang, J.; Yan, J.; Zhang, C.; Li, S.; Yuan, M.; Zhang, C.; Shen, C.; Yang, Y.; Fu, L.; Xu, G.; et al. Genetic, biological and epidemiological study on a cluster of H9N2 avian influenza virus infections among chickens, a pet cat, and humans at a backyard farm in Guangxi, China. Emerg. Microbes Infect. 2023, 12, 2143282. [Google Scholar] [CrossRef]
- Messenger, A.M.; Barnes, A.N.; Gray, G.C. Reverse zoonotic disease transmission (zooanthroponosis): A systematic review of seldom-documented human biological threats to animals. PLoS ONE 2014, 9, e89055. [Google Scholar] [CrossRef]
- Walther, T.; Karamanska, R.; Chan, R.W.; Chan, M.C.; Jia, N.; Air, G.; Hopton, C.; Wong, M.P.; Dell, A.; Malik Peiris, J.S.; et al. Glycomic analysis of human respiratory tract tissues and correlation with influenza virus infection. PLoS Pathog. 2013, 9, e1003223. [Google Scholar] [CrossRef] [PubMed]
- Nicholls, J.M.; Bourne, A.J.; Chen, H.; Guan, Y.; Peiris, J.S. Sialic acid receptor detection in the human respiratory tract: Evidence for widespread distribution of potential binding sites for human and avian influenza viruses. Respir. Res. 2007, 8, 73. [Google Scholar] [CrossRef] [PubMed]
- Sriwilaijaroen, N.; Nakakita, S.I.; Kondo, S.; Yagi, H.; Kato, K.; Murata, T.; Hiramatsu, H.; Kawahara, T.; Watanabe, Y.; Kanai, Y.; et al. N-glycan structures of human alveoli provide insight into influenza A virus infection and pathogenesis. Febs J. 2018, 285, 1611–1634. [Google Scholar] [CrossRef]
- Shinya, K.; Ebina, M.; Yamada, S.; Ono, M.; Kasai, N.; Kawaoka, Y. Avian flu: Influenza virus receptors in the human airway. Nature 2006, 440, 435–436. [Google Scholar] [CrossRef]
- Ibricevic, A.; Pekosz, A.; Walter, M.J.; Newby, C.; Battaile, J.T.; Brown, E.G.; Holtzman, M.J.; Brody, S.L. Influenza virus receptor specificity and cell tropism in mouse and human airway epithelial cells. J. Virol. 2006, 80, 7469–7480. [Google Scholar] [CrossRef]
- Kuchipudi, S.V.; Nelli, R.K.; Gontu, A.; Satyakumar, R.; Nair, M.S.; Subbiah, M. Sialic Acid Receptors: The Key to Solving the Enigma of Zoonotic Virus Spillover. Viruses-Basel 2021, 13, 262. [Google Scholar] [CrossRef]
- Institute of Medicine Forum on Microbial, T. The National Academies Collection: Reports funded by National Institutes of Health. In The Threat of Pandemic Influenza: Are We Ready? Workshop Summary; Knobler, S.L., Mack, A., Mahmoud, A., Lemon, S.M., Eds.; National Academies Press (US); National Academy of Sciences: Washington, DC, USA, 2005. [Google Scholar]
- Nelli, R.K.; Kuchipudi, S.V.; White, G.A.; Perez, B.B.; Dunham, S.P.; Chang, K.C. Comparative distribution of human and avian type sialic acid influenza receptors in the pig. BMC Vet. Res. 2010, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Trebbien, R.; Larsen, L.E.; Viuff, B.M. Distribution of sialic acid receptors and influenza A virus of avian and swine origin in experimentally infected pigs. Virol. J. 2011, 8, 434. [Google Scholar] [CrossRef]
- Sriwilaijaroen, N.; Kondo, S.; Yagi, H.; Takemae, N.; Saito, T.; Hiramatsu, H.; Kato, K.; Suzuki, Y. N-glycans from porcine trachea and lung: Predominant NeuAcalpha2-6Gal could be a selective pressure for influenza variants in favor of human-type receptor. PLoS ONE 2011, 6, e16302. [Google Scholar] [CrossRef]
- Patterson, A.R.; Cooper, V.L.; Yoon, K.J.; Janke, B.H.; Gauger, P.C. Naturally occurring influenza infection in a ferret (Mustela putorius furo) colony. J. Vet. Diagn. Investig. 2009, 21, 527–530. [Google Scholar] [CrossRef]
- Skowronski, D.M.; De Serres, G.; Janjua, N.Z.; Gardy, J.L.; Gilca, V.; Dionne, M.; Hamelin, M.E.; Rhéaume, C.; Boivin, G. Cross-reactive antibody to swine influenza A(H3N2) subtype virus in children and adults before and after immunisation with 2010/11 trivalent inactivated influenza vaccine in Canada, August to November 2010. Euro Surveill 2012, 17, 20066. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lin, H.T.; Wang, C.H.; Wu, W.L.; Chi, C.H.; Wang, L.C. Natural A(H1N1)pdm09 influenza virus infection case in a pet ferret in Taiwan. Jpn. J. Vet. Res. 2014, 62, 181–185. [Google Scholar] [PubMed]
- Campagnolo, E.R.; Moll, M.E.; Tuhacek, K.; Simeone, A.J.; Miller, W.S.; Waller, K.O.; Simwale, O.; Rankin, J.T.; Ostroff, S.M. Concurrent 2009 Pandemic Influenza A (H1N1) Virus Infection in Ferrets and in a Community in Pennsylvania. Zoonoses Public Health 2013, 60, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Swenson, S.L.; Koster, L.G.; Jenkins-Moore, M.; Killian, M.L.; DeBess, E.E.; Baker, R.J.; Mulrooney, D.; Weiss, R.; Galeota, J.; Bredthauer, A. Natural cases of 2009 pandemic H1N1 Influenza A virus in pet ferrets. J. Vet. Diagn. Investig. 2010, 22, 784–788. [Google Scholar] [CrossRef]
- Sooksawasdi Na Ayudhya, S.; Kuiken, T. Reverse Zoonosis of COVID-19: Lessons From the 2009 Influenza Pandemic. Vet. Pathol. 2021, 58, 234–242. [Google Scholar] [CrossRef]
- Driskell, E.A.; Pickens, J.A.; Humberd-Smith, J.; Gordy, J.T.; Bradley, K.C.; Steinhauer, D.A.; Berghaus, R.D.; Stallknecht, D.E.; Howerth, E.W.; Tompkins, S.M. Low pathogenic avian influenza isolates from wild birds replicate and transmit via contact in ferrets without prior adaptation. PLoS ONE 2012, 7, e38067. [Google Scholar] [CrossRef] [PubMed]
- Guan, M.; Hall, J.S.; Zhang, X.; Dusek, R.J.; Olivier, A.K.; Liu, L.; Li, L.; Krauss, S.; Danner, A.; Li, T.; et al. Aerosol Transmission of Gull-Origin Iceland Subtype H10N7 Influenza A Virus in Ferrets. J. Virol. 2019, 93, e00282-19. [Google Scholar] [CrossRef]
- Song, H.; Wan, H.; Araya, Y.; Perez, D.R. Partial direct contact transmission in ferrets of a mallard H7N3 influenza virus with typical avian-like receptor specificity. Virol. J. 2009, 6, 126. [Google Scholar] [CrossRef]
- Belser, J.A.; Blixt, O.; Chen, L.M.; Pappas, C.; Maines, T.R.; Van Hoeven, N.; Donis, R.; Busch, J.; McBride, R.; Paulson, J.C.; et al. Contemporary North American influenza H7 viruses possess human receptor specificity: Implications for virus transmissibility. Proc. Natl. Acad. Sci. USA 2008, 105, 7558–7563. [Google Scholar] [CrossRef]
- Sun, X.; Belser, J.A.; Pulit-Penaloza, J.A.; Zeng, H.; Lewis, A.; Shieh, W.J.; Tumpey, T.M.; Maines, T.R. Pathogenesis and Transmission Assessments of Two H7N8 Influenza A Viruses Recently Isolated from Turkey Farms in Indiana Using Mouse and Ferret Models. J. Virol. 2016, 90, 10936–10944. [Google Scholar] [CrossRef]
- Richard, M.; Herfst, S.; Tao, H.; Jacobs, N.T.; Lowen, A.C. Influenza A Virus Reassortment Is Limited by Anatomical Compartmentalization following Coinfection via Distinct Routes. J. Virol. 2018, 92, e02063-17. [Google Scholar] [CrossRef]
- Phipps, K.L.; Marshall, N.; Tao, H.; Danzy, S.; Onuoha, N.; Steel, J.; Lowen, A.C. Seasonal H3N2 and 2009 Pandemic H1N1 Influenza A Viruses Reassort Efficiently but Produce Attenuated Progeny. J. Virol. 2017, 91, e00830-17. [Google Scholar] [CrossRef] [PubMed]
- Ganti, K.; Bagga, A.; Carnaccini, S.; Ferreri, L.M.; Geiger, G.; Joaquin Caceres, C.; Seibert, B.; Li, Y.; Wang, L.; Kwon, T.; et al. Influenza A virus reassortment in mammals gives rise to genetically distinct within-host subpopulations. Nat. Commun. 2022, 13, 6846. [Google Scholar] [CrossRef] [PubMed]
- Jackson, S.; Van Hoeven, N.; Chen, L.M.; Maines, T.R.; Cox, N.J.; Katz, J.M.; Donis, R.O. Reassortment between avian H5N1 and human H3N2 influenza viruses in ferrets: A public health risk assessment. J. Virol. 2009, 83, 8131–8140. [Google Scholar] [CrossRef] [PubMed]
- Schrauwen, E.J.; Bestebroer, T.M.; Rimmelzwaan, G.F.; Osterhaus, A.D.; Fouchier, R.A.; Herfst, S. Reassortment between Avian H5N1 and human influenza viruses is mainly restricted to the matrix and neuraminidase gene segments. PLoS ONE 2013, 8, e59889. [Google Scholar] [CrossRef]
- Jayaraman, A.; Chandrasekaran, A.; Viswanathan, K.; Raman, R.; Fox, J.G.; Sasisekharan, R. Decoding the distribution of glycan receptors for human-adapted influenza A viruses in ferret respiratory tract. PLoS ONE 2012, 7, e27517. [Google Scholar] [CrossRef]
- Ng, P.S.; Bohm, R.; Hartley-Tassell, L.E.; Steen, J.A.; Wang, H.; Lukowski, S.W.; Hawthorne, P.L.; Trezise, A.E.; Coloe, P.J.; Grimmond, S.M.; et al. Ferrets exclusively synthesize Neu5Ac and express naturally humanized influenza A virus receptors. Nat. Commun. 2014, 5, 5750. [Google Scholar] [CrossRef]
- Jia, N.; Barclay, W.S.; Roberts, K.; Yen, H.L.; Chan, R.W.; Lam, A.K.; Air, G.; Peiris, J.S.; Dell, A.; Nicholls, J.M.; et al. Glycomic characterization of respiratory tract tissues of ferrets: Implications for its use in influenza virus infection studies. J. Biol. Chem. 2014, 289, 28489–28504. [Google Scholar] [CrossRef]
- Fenollar, F.; Mediannikov, O.; Maurin, M.; Devaux, C.; Colson, P.; Levasseur, A.; Fournier, P.E.; Raoult, D. Mink, SARS-CoV-2, and the Human-Animal Interface. Front. Microbiol. 2021, 12, 663815. [Google Scholar] [CrossRef]
- Berg, M.; Englund, L.; Abusugra, I.A.; Klingeborn, B.; Linne, T. Close Relationship between Mink Influenza (H10n4) and Concomitantly Circulating Avian Influenza-Viruses. Arch. Virol. 1990, 113, 61–71. [Google Scholar] [CrossRef]
- Peng, L.; Chen, C.; Kai-yi, H.; Feng-xia, Z.; Yan-li, Z.; Zong-shuai, L.; Xing-xiao, Z.; Shi-jin, J.; Zhi-jing, X. Molecular characterization of H9N2 influenza virus isolated from mink and its pathogenesis in mink. Vet. Microbiol. 2015, 176, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.J.; Schwartz, K.; Sun, D.; Zhang, J.; Hildebrandt, H. Naturally occurring Influenza A virus subtype H1N2 infection in a Midwest United States mink (Mustela vison) ranch. J. Vet. Diagn. Investig. 2012, 24, 388–391. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, Z.; Cui, Y.; Yang, H.; Shan, H.; Zhang, C. Emergence of an Eurasian avian-like swine influenza A (H1N1) virus from mink in China. Vet. Microbiol. 2020, 240, 108509. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, C.A.; Spearman, G.; Hamel, A.; Godson, D.L.; Fortin, A.; Fontaine, G.; Tremblay, D. Characterization of a Canadian mink H3N2 influenza A virus isolate genetically related to triple reassortant swine influenza virus. J. Clin. Microbiol. 2009, 47, 796–799. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, S.; Zhang, C.; Li, J.; Hou, G.; Peng, C.; Chen, J.; Shan, H. Characterization of H5N1 highly pathogenic mink influenza viruses in eastern China. Vet. Microbiol. 2017, 201, 225–230. [Google Scholar] [CrossRef]
- Sun, H.; Li, F.; Liu, Q.; Du, J.; Liu, L.; Sun, H.; Li, C.; Liu, J.; Zhang, X.; Yang, J.; et al. Mink is a highly susceptible host species to circulating human and avian influenza viruses. Emerg. Microbes Infect. 2021, 10, 472–480. [Google Scholar] [CrossRef]
- Yagyu, K.; Yanagawa, R.; Matsuura, Y.; Noda, H. Contact infection of mink with influenza A viruses of avian and mammalian origin. Arch. Virol. 1981, 68, 143–145. [Google Scholar] [CrossRef]
- Okazaki, K.; Yanagawa, R.; Kida, H. Contact infection of mink with 5 subtypes of avian influenza virus. Brief report. Arch. Virol. 1983, 77, 265–269. [Google Scholar] [CrossRef]
- Matsuura, Y.; Yanagawa, R.; Noda, H. Experimental infection of mink with influenza A viruses. Brief report. Arch. Virol. 1979, 62, 71–76. [Google Scholar] [CrossRef]
- Zohari, S.; Metreveli, G.; Kiss, I.; Belak, S.; Berg, M. Full genome comparison and characterization of avian H10 viruses with different pathogenicity in Mink (Mustela vison) reveals genetic and functional differences in the non-structural gene. Virol. J. 2010, 7, 145. [Google Scholar] [CrossRef]
- Reinhardt, U.; Scholtissek, C. Comparison of the nucleoprotein genes of a chicken and a mink influenza A H 10 virus. Arch. Virol. 1988, 103, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Scholtissek, C.; Ludwig, S.; Fitch, W.M. Analysis of influenza A virus nucleoproteins for the assessment of molecular genetic mechanisms leading to new phylogenetic virus lineages. Arch. Virol. 1993, 131, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Englund, L.; Klingeborn, B.; Mejerland, T. Avian influenza A virus causing an outbreak of contagious interstitial pneumonia in mink. Acta Vet. Scand. 1986, 27, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Klingeborn, B.; Englund, L.; Rott, R.; Juntti, N.; Rockborn, G. An avian influenza A virus killing a mammalian species--the mink. Brief report. Arch. Virol. 1985, 86, 347–351. [Google Scholar] [CrossRef]
- Englund, L. Studies on influenza viruses H10N4 and H10N7 of avian origin in mink. Vet. Microbiol. 2000, 74, 101–107. [Google Scholar] [CrossRef]
- Feldmann, H.; Kretzschmar, E.; Klingeborn, B.; Rott, R.; Klenk, H.D.; Garten, W. The structure of serotype H10 hemagglutinin of influenza A virus: Comparison of an apathogenic avian and a mammalian strain pathogenic for mink. Virology 1988, 165, 428–437. [Google Scholar] [CrossRef]
- Zohari, S.; Munir, M.; Metreveli, G.; Belak, S.; Berg, M. Differences in the ability to suppress interferon beta production between allele A and allele B NS1 proteins from H10 influenza A viruses. Virol. J. 2010, 7, 376. [Google Scholar] [CrossRef]
- Yong-Feng, Z.; Fei-Fei, D.; Jia-Yu, Y.; Feng-Xia, Z.; Chang-Qing, J.; Jian-Li, W.; Shou-Yu, G.; Kai, C.; Chuan-Yi, L.; Xue-Hua, W.; et al. Intraspecies and interspecies transmission of mink H9N2 influenza virus. Sci. Rep. 2017, 7, 7429. [Google Scholar] [CrossRef]
- Zhang, C.; Xuan, Y.; Shan, H.; Yang, H.; Wang, J.; Wang, K.; Li, G.; Qiao, J. Avian influenza virus H9N2 infections in farmed minks. Virol. J. 2015, 12, 180. [Google Scholar] [CrossRef]
- Agüero, M.; Monne, I.; Sánchez, A.; Zecchin, B.; Fusaro, A.; Ruano, M.J.; del Valle Arrojo, M.; Fernández-Antonio, R.; Souto, A.M.; Tordable, P.; et al. Highly pathogenic avian influenza A(H5N1) virus infection in farmed minks, Spain, October 2022. Eurosurveillance 2023, 28, 2300001. [Google Scholar] [CrossRef]
- Kiss, I.; Gyarmati, P.; Zohari, S.; Ramsay, K.W.; Metreveli, G.; Weiss, E.; Brytting, M.; Stivers, M.; Lindstrom, S.; Lundkvist, A.; et al. Molecular characterization of highly pathogenic H5N1 avian influenza viruses isolated in Sweden in 2006. Virol. J. 2008, 5, 113. [Google Scholar] [CrossRef]
- Åkerstedt, J.; Valheim, M.; Germundsson, A.; Moldal, T.; Lie, K.I.; Falk, M.; Hungnes, O. Pneumonia caused by influenza A H1N1 2009 virus in farmed American mink (Neovison visonins). Vet. Rec. 2012, 170, 362. [Google Scholar] [CrossRef]
- Clayton, M.J.; Kelly, E.J.; Mainenti, M.; Wilhelm, A.; Torchetti, M.K.; Killian, M.L.; Van Wettere, A.J. Pandemic lineage 2009 H1N1 influenza A virus infection in farmed mink in Utah. J. Vet. Diagn. Investig. 2022, 34, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, D.; Allard, V.; Doyon, J.F.; Bellehumeur, C.; Spearman, J.G.; Harel, J.; Gagnon, C.A. Emergence of a new swine H3N2 and pandemic (H1N1) 2009 influenza A virus reassortant in two Canadian animal populations, mink and swine. J. Clin. Microbiol. 2011, 49, 4386–4390. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, T.; Mena, I.; Anthony, S.J.; Medina, R.; Robinson, P.W.; Greig, D.J.; Costa, D.P.; Lipkin, W.I.; Garcia-Sastre, A.; Boyce, W.M. Pandemic H1N1 influenza isolated from free-ranging Northern Elephant Seals in 2010 off the central California coast. PLoS ONE 2013, 8, e62259. [Google Scholar] [CrossRef] [PubMed]
- Harder, T.C.; Siebert, U.; Wohlsein, P.; Vahlenkamp, T. Influenza A virus infections in marine mammals and terrestrial carnivores. Berl Munch Tierarztl Wochenschr. 2013, 126, 500–508. [Google Scholar]
- Runstadler, J.A.; Puryear, W. A Brief Introduction to Influenza A Virus in Marine Mammals. Methods Mol. Biol. 2020, 2123, 429–450. [Google Scholar]
- Fereidouni, S.; Munoz, O.; Von Dobschuetz, S.; De Nardi, M. Influenza Virus Infection of Marine Mammals. Ecohealth 2016, 13, 161–170. [Google Scholar] [CrossRef]
- Bodewes, R.; Zohari, S.; Krog, J.S.; Hall, M.D.; Harder, T.C.; Bestebroer, T.M.; van de Bildt, M.W.G.; Spronken, M.I.; Larsen, L.E.; Siebert, U.; et al. Spatiotemporal Analysis of the Genetic Diversity of Seal Influenza A(H10N7) Virus, Northwestern Europe. J. Virol. 2016, 90, 4269–4277. [Google Scholar] [CrossRef]
- Hinshaw, V.S.; Bean, W.J.; Webster, R.G.; Rehg, J.E.; Fiorelli, P.; Early, G.; Geraci, J.R.; St Aubin, D.J. Are seals frequently infected with avian influenza viruses? J. Virol. 1984, 51, 863–865. [Google Scholar] [CrossRef]
- Berhane, Y.; Joseph, T.; Lung, O.; Embury-Hyatt, C.; Xu, W.H.; Cottrell, P.; Raverty, S. Isolation and Characterization of Novel Reassortant Influenza A(H10N7) Virus in a Harbor Seal, British Columbia, Canada. Emerg. Infect. Dis. 2022, 28, 1480–1484. [Google Scholar] [CrossRef] [PubMed]
- Puryear, W.; Sawatzki, K.; Hill, N.; Foss, A.; Stone, J.J.; Doughty, L.; Walk, D.; Gilbert, K.; Murray, M.; Cox, E.; et al. Outbreak of Highly Pathogenic Avian Influenza H5N1 in New England Seals. bioRxiv 2022. bioRxiv:2022.07.29.501155. [Google Scholar]
- Webster, R.G.; Geraci, J.; Petursson, G.; Skirnisson, K. Conjunctivitis in human beings caused by influenza A virus of seals. N. Engl. J. Med. 1981, 304, 911. [Google Scholar] [PubMed]
- Scheiblauer, H.; Kendal, A.P.; Rott, R. Pathogenicity of influenza A/Seal/Mass/1/80 virus mutants for mammalian species. Arch Virol. 1995, 140, 341–348. [Google Scholar] [CrossRef]
- Solorzano, A.; Foni, E.; Cordoba, L.; Baratelli, M.; Razzuoli, E.; Bilato, D.; Martin del Burgo, M.A.; Perlin, D.S.; Martinez, J.; Martinez-Orellana, P.; et al. Cross-Species Infectivity of H3N8 Influenza Virus in an Experimental Infection in Swine. J. Virol. 2015, 89, 11190–11202. [Google Scholar] [CrossRef]
- Anthony, S.J.; St Leger, J.A.; Pugliares, K.; Ip, H.S.; Chan, J.M.; Carpenter, Z.W.; Navarrete-Macias, I.; Sanchez-Leon, M.; Saliki, J.T.; Pedersen, J.; et al. Emergence of fatal avian influenza in New England harbor seals. mBio 2012, 3, e00166-12. [Google Scholar] [CrossRef]
- Herfst, S.; Zhang, J.; Richard, M.; McBride, R.; Lexmond, P.; Bestebroer, T.M.; Spronken, M.I.J.; de Meulder, D.; van den Brand, J.M.; Rosu, M.E.; et al. Hemagglutinin Traits Determine Transmission of Avian A/H10N7 Influenza Virus between Mammals. Cell Host Microbe 2020, 28, 602–613 e7. [Google Scholar] [CrossRef]
- Dittrich, A.; Scheibner, D.; Salaheldin, A.H.; Veits, J.; Gischke, M.; Mettenleiter, T.C.; Abdelwhab, E.M. Impact of Mutations in the Hemagglutinin of H10N7 Viruses Isolated from Seals on Virus Replication in Avian and Human Cells. Viruses-Basel 2018, 10, 83. [Google Scholar] [CrossRef]
- Karlsson, E.A.; Ip, H.S.; Hall, J.S.; Yoon, S.W.; Johnson, J.; Beck, M.A.; Webby, R.J.; Schultz-Cherry, S. Respiratory transmission of an avian H3N8 influenza virus isolated from a harbour seal. Nat. Commun. 2014, 5, 4791. [Google Scholar] [CrossRef]
- Callan, R.J.; Early, G.; Kida, H.; Hinshaw, V.S. The appearance of H3 influenza viruses in seals. J. Gen. Virol. 1995, 76 Pt 1, 199–203. [Google Scholar] [CrossRef]
- Venkatesh, D.; Bianco, C.; Nunez, A.; Collins, R.; Thorpe, D.; Reid, S.M.; Brookes, S.M.; Essen, S.; McGinn, N.; Seekings, J.; et al. Detection of H3N8 influenza A virus with multiple mammalian-adaptive mutations in a rescued Grey seal (Halichoerus grypus) pup. Virus Evol. 2020, 6, veaa016. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, M.L.; Wise, H.M.; Nicol, M.Q.; Smith, N.; Dunfee, R.L.; Beard, P.M.; Jagger, B.W.; Ligertwood, Y.; Hardisty, G.R.; Xiao, H.; et al. Role of the B Allele of Influenza A Virus Segment 8 in Setting Mammalian Host Range and Pathogenicity. J. Virol. 2016, 90, 9263–9284. [Google Scholar] [CrossRef] [PubMed]
- Gulyaeva, M.; Sobolev, I.; Sharshov, K.; Kurskaya, O.; Alekseev, A.; Shestopalova, L.; Kovner, A.; Bi, Y.; Shi, W.; Shchelkanov, M.; et al. Characterization of Avian-like Influenza A (H4N6) Virus Isolated from Caspian Seal in 2012. Virol. Sin. 2018, 33, 449–452. [Google Scholar] [CrossRef]
- European Food Safety Authority; European Centre for Disease Prevention and Control; European Union Reference Laboratory for Avian Influenza; Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, É.; Staubach, C.; et al. Avian influenza overview September—December 2022. EFSA J. 2023, 21, e07786. [Google Scholar]
- ProMed. Archive Number: 20230210.8708269. Available online: https://promedmail.org/promed-posts/ (accessed on 5 March 2023).
- APHA, Confirmed Findings of Influenza of Avian Origin in Non-Avian Wildlife. 2023. Available online: https://www.gov.uk/government/publications/bird-flu-avian-influenza-findings-in-non-avian-wildlife/confirmed-findings-of-influenza-of-avian-origin-in-non-avian-wildlife (accessed on 7 March 2023).
- Shin, D.L.; Siebert, U.; Lakemeyer, J.; Grilo, M.; Pawliczka, I.; Wu, N.H.; Valentin-Weigand, P.; Haas, L.; Herrler, G. Highly Pathogenic Avian Influenza A(H5N8) Virus in Gray Seals, Baltic Sea. Emerg. Infect. Dis. 2019, 25, 2295–2298. [Google Scholar] [CrossRef]
- Floyd, T.; Banyard, A.C.; Lean, F.Z.X.; Byrne, A.M.P.; Fullick, E.; Whittard, E.; Mollett, B.C.; Bexton, S.; Swinson, V.; Macrelli, M.; et al. Encephalitis and Death in Wild Mammals at a Rehabilitation Center after Infection with Highly Pathogenic Avian Influenza A(H5N8) Virus, United Kingdom. Emerg. Infect. Dis. 2021, 27, 2856–2863. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority; Adlhoch, C.; Fusaro, A.; Gonzales, J.L.; Kuiken, T.; Marangon, S.; Niqueux, E.; Staubach, C.; Terregino, C.; Aznar, I.; et al. Avian influenza overview September—December 2021. EFSA J. 2021, 19, e07108. [Google Scholar] [CrossRef]
- Lang, G.; Gagnon, A.; Geraci, J.R. Isolation of an influenza A virus from seals. Arch. Virol. 1981, 68, 189–195. [Google Scholar] [CrossRef]
- Lin, H.T.; Wang, C.H.; Chueh, L.L.; Su, B.L.; Wang, L.C. Influenza A(H6N1) Virus in Dogs, Taiwan. Emerg. Infect. Dis. 2015, 21, 2154–2157. [Google Scholar] [CrossRef]
- Klivleyeva, N.G.; Glebova, T.I.; Shamenova, M.G.; Saktaganov, N.T. Influenza A viruses circulating in dogs: A review of the scientific literature. Open Vet. J. 2022, 12, 676–687. [Google Scholar] [CrossRef]
- Lin, D.; Sun, S.; Du, L.; Ma, J.; Fan, L.; Pu, J.; Sun, Y.; Zhao, J.; Sun, H.; Liu, J. Natural and experimental infection of dogs with pandemic H1N1/2009 influenza virus. J. Gen. Virol. 2012, 93 Pt 1, 119–123. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Moon, H.J.; An, D.J.; Jeoung, H.Y.; Kim, H.; Yeom, M.J.; Hong, M.; Nam, J.H.; Park, S.J.; Park, B.K.; et al. A novel reassortant canine H3N1 influenza virus between pandemic H1N1 and canine H3N2 influenza viruses in Korea. J. Gen. Virol. 2012, 93 Pt 3, 551–554. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Hong, M.; Kim, J.K.; Seon, B.; Na, W.; Park, S.J.; An, D.J.; Jeoung, H.Y.; Kim, D.J.; Kim, J.M.; et al. H3N2 canine influenza virus with the matrix gene from the pandemic A/H1N1 virus: Infection dynamics in dogs and ferrets. Epidemiol. Infect. 2015, 143, 772–780. [Google Scholar] [CrossRef]
- Bao, P.T.; Liu, Y.; Zhang, X.A.; Fan, H.; Zhao, J.; Mu, M.; Li, H.Y.; Wang, Y.H.; Ge, H.H.; Li, S.; et al. Human infection with a reassortment avian influenza A H3N8 virus: An epidemiological investigation study. Nat. Commun. 2022, 13, 6817. [Google Scholar] [CrossRef] [PubMed]
- Paniker, C.K.; Nair, C.M. Experimental infection of animals with influenzavirus types A and B. Bull World Health Organ. 1972, 47, 461–463. [Google Scholar] [PubMed]
- Kovalenko, G.; Galat, M.; Ishchenko, L.; Halka, I. Serological Evidence for Influenza A Viruses Among Domestic Dogs and Cats in Kyiv, Ukraine. Vector Borne Zoonotic Dis. 2021, 21, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Bluhm, P.; Sepulveda, A.; Baumberger, C.; Di Pillo, F.; Ruiz, S.; Salazar, C.; Marambio, V.; Berrios, F.; Galdames, P.; Amaro, A.; et al. Evidence of influenza infection in dogs and cats in central Chile. Prev. Vet. Med. 2021, 191, 105349. [Google Scholar] [CrossRef]
- Su, S.; Zhou, P.; Fu, X.; Wang, L.; Hong, M.; Lu, G.; Sun, L.; Qi, W.; Ning, Z.; Jia, K.; et al. Virological and epidemiological evidence of avian influenza virus infections among feral dogs in live poultry markets, china: A threat to human health? Clin. Infect. Dis. 2014, 58, 1644–1646. [Google Scholar] [CrossRef]
- Ning, Z.Y.; Wu, X.T.; Cheng, Y.F.; Qi, W.B.; An, Y.F.; Wang, H.; Zhang, G.H.; Li, S.J. Tissue distribution of sialic acid-linked influenza virus receptors in beagle dogs. J. Vet. Sci. 2012, 13, 219–222. [Google Scholar] [CrossRef]
- Zhao, S.; Schuurman, N.; Tieke, M.; Quist, B.; Zwinkels, S.; van Kuppeveld, F.J.M.; de Haan, C.A.M.; Egberink, H. Serological Screening of Influenza A Virus Antibodies in Cats and Dogs Indicates Frequent Infection with Different Subtypes. J. Clin. Microbiol. 2020, 58, e01689-20. [Google Scholar] [CrossRef]
- Hatta, M.; Zhong, G.; Gao, Y.; Nakajima, N.; Fan, S.; Chiba, S.; Deering, K.M.; Ito, M.; Imai, M.; Kiso, M.; et al. Characterization of a Feline Influenza A(H7N2) Virus. Emerg. Infect. Dis. 2018, 24, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Kuiken, T.; Rimmelzwaan, G.; van Riel, D.; van Amerongen, G.; Baars, M.; Fouchier, R.; Osterhaus, A. Avian H5N1 influenza in cats. Science 2004, 306, 241. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Gao, X.; Wang, T.; Li, Y.; Li, Y.; Xu, Y.; Chu, D.; Sun, H.; Wu, C.; Li, S.; et al. Fatal H5N6 Avian Influenza Virus Infection in a Domestic Cat and Wild Birds in China. Sci. Rep. 2015, 5, 10704. [Google Scholar] [CrossRef]
- Belser, J.A.; Pulit-Penaloza, J.A.; Sun, X.; Brock, N.; Pappas, C.; Creager, H.M.; Zeng, H.; Tumpey, T.M.; Maines, T.R. A Novel A(H7N2) Influenza Virus Isolated from a Veterinarian Caring for Cats in a New York City Animal Shelter Causes Mild Disease and Transmits Poorly in the Ferret Model. J. Virol. 2017, 91. [Google Scholar] [CrossRef]
- Poirot, E.; Levine, M.Z.; Russell, K.; Stewart, R.J.; Pompey, J.M.; Chiu, S.; Fry, A.M.; Gross, L.; Havers, F.P.; Li, Z.N.; et al. Detection of Avian Influenza A(H7N2) Virus Infection Among Animal Shelter Workers Using a Novel Serological Approach-New York City, 2016–2017. J. Infect. Dis. 2019, 219, 1688–1696. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wu, X.; Cheng, Y.; An, Y.; Ning, Z. Tissue distribution of human and avian type sialic acid influenza virus receptors in domestic cat. Acta Vet. Hung. 2013, 61, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Thongratsakul, S.; Suzuki, Y.; Hiramatsu, H.; Sakpuaram, T.; Sirinarumitr, T.; Poolkhet, C.; Moonjit, P.; Yodsheewan, R.; Songserm, T. Avian and human influenza A virus receptors in trachea and lung of animals. Asian Pac. J. Allergy Immunol. 2010, 28, 294–301. [Google Scholar] [PubMed]
- Kroeze, E.J.; Kuiken, T.; Osterhaus, A.D. Animal models. Methods Mol. Biol. 2012, 865, 127–146. [Google Scholar] [PubMed]
- Davis, A.S.; Taubenberger, J.K.; Bray, M. The use of nonhuman primates in research on seasonal, pandemic and avian influenza, 1893–2014. Antivir. Res. 2015, 117, 75–98. [Google Scholar] [CrossRef] [PubMed]
- Paungpin, W.; Thongdee, M.; Ketchim, N.; Chaiwattanarungruengpaisan, S.; Saechin, A.; Sariya, L.; Kaewchot, S.; Puthavathana, P.; Wiriyarat, W. Evidence of Influenza A Virus Infection in Cynomolgus Macaques, Thailand. Vet. Sci. 2022, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, E.A.; Engel, G.A.; Feeroz, M.M.; San, S.; Rompis, A.; Lee, B.P.; Shaw, E.; Oh, G.; Schillaci, M.A.; Grant, R.; et al. Influenza virus infection in nonhuman primates. Emerg. Infect. Dis. 2012, 18, 1672–1675. [Google Scholar] [CrossRef] [PubMed]
- Wachtman, L.; Mansfield, K. Viral Diseases of Nonhuman Primates. Nonhum. Primates Biomed. Res. 2012, 1–104. [Google Scholar] [CrossRef]
- Bunuma, E.K.; Ochola, L.; Nyerere, A.K. A survey of influenza subtypes in olive baboons in selected areas in Kenya. bioRxiv 2018. bioRxiv:380345. [Google Scholar]
- O’Brien, T.C.; Tauraso, N.M. Antibodies to type A influenza viruses in sera from nonhuman primates. Arch. Virol. 1973, 40, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Buitendijk, H.; Fagrouch, Z.; Niphuis, H.; Bogers, W.M.; Warren, K.S.; Verschoor, E.J. Retrospective Serology Study of Respiratory Virus Infections in Captive Great Apes. Viruses-Basel 2014, 6, 1442–1453. [Google Scholar] [CrossRef]
- Nakayama, M.; Itoh, Y. Lectin Staining to Detect Human and Avian Influenza Virus Receptors in the Airway of Nonhuman Primates. Methods Mol. Biol. 2022, 2556, 37–43. [Google Scholar]
- Matsuoka, Y.; Suguitan, A., Jr.; Orandle, M.; Paskel, M.; Boonnak, K.; Gardner, D.J.; Feldmann, F.; Feldmann, H.; Marino, M.; Jin, H.; et al. African green monkeys recapitulate the clinical experience with replication of live attenuated pandemic influenza virus vaccine candidates. J. Virol. 2014, 88, 8139–8152. [Google Scholar] [CrossRef]
- Horimoto, T.; Maeda, K.; Murakami, S.; Kiso, M.; Iwatsuki-Horimoto, K.; Sashika, M.; Ito, T.; Suzuki, K.; Yokoyama, M.; Kawaoka, Y. Highly pathogenic avian influenza virus infection in feral raccoons, Japan. Emerg. Infect. Dis. 2011, 17, 714–717. [Google Scholar] [CrossRef]
- Yamaguchi, E.; Sashika, M.; Fujii, K.; Kobayashi, K.; Bui, V.N.; Ogawa, H.; Imai, K. Prevalence of multiple subtypes of influenza A virus in Japanese wild raccoons. Virus Res. 2014, 189, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Roberts, N.M.; Henzler, D.J.; Clark, L. Serologic evidence of avian influenza (H4N6) exposure in a wild-caught raccoon. Avian Dis. 2009, 53, 455–457. [Google Scholar] [CrossRef]
- Sullivan, H.J.; Blitvich, B.J.; VanDalen, K.; Bentler, K.T.; Franklin, A.B.; Root, J.J. Evaluation of an epitope-blocking enzyme-linked immunosorbent assay for the detection of antibodies to influenza A virus in domestic and wild avian and mammalian species. J. Virol. Methods 2009, 161, 141–146. [Google Scholar] [CrossRef][Green Version]
- Root, J.J.; Bentler, K.T.; Sullivan, H.J.; Blitvich, B.J.; McLean, R.G.; Franklin, A.B. Antibody responses of raccoons naturally exposed to influenza A virus. Vector Borne Zoonotic Dis. 2010, 10, 821–823. [Google Scholar] [CrossRef]
- Hall, J.S.; Bentler, K.T.; Landolt, G.; Elmore, S.A.; Minnis, R.B.; Campbell, T.A.; Barras, S.C.; Root, J.J.; Pilon, J.; Pabilonia, K.; et al. Influenza infection in wild raccoons. Emerg. Infect. Dis. 2008, 14, 1842–1848. [Google Scholar] [CrossRef]
- Yamaguchi, E.; Fujii, K.; Ogawa, H.; Imai, K. First detection of influenza A virus genes from wild raccoons in Japan. Virus Genes 2018, 54, 591–595. [Google Scholar] [CrossRef]
- Yamnikova, S.S.; Mandler, J.; Bekh-Ochir, Z.H.; Dachtzeren, P.; Ludwig, S.; Lvov, D.K.; Scholtissek, C. A reassortant H1N1 influenza A virus caused fatal epizootics among camels in Mongolia. Virology 1993, 197, 558–563. [Google Scholar] [CrossRef]
- Anchlan, D.; Ludwig, S.; Nymadawa, P.; Mendsaikhan, J.; Scholtissek, C. Previous H1N1 influenza A viruses circulating in the Mongolian population. Arch. Virol. 1996, 141, 1553–1569. [Google Scholar] [CrossRef]
- Chu, D.K.W.; Perera, R.; Ali, A.; Oladipo, J.O.; Mamo, G.; So, R.T.Y.; Zhou, Z.; Chor, Y.Y.; Chan, C.K.; Belay, D.; et al. Influenza A Virus Infections in Dromedary Camels, Nigeria and Ethiopia, 2015-2017. Emerg. Infect. Dis. 2020, 26, 173–176. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Liu, Y.; Fen, J.; Liu, K.; Qin, T.; Chen, S.; Peng, D.; Liu, X. Characterization of an H7N9 Influenza Virus Isolated from Camels in Inner Mongolia, China. Microbiol. Spectr. 2023, e0179822. [Google Scholar] [CrossRef] [PubMed]
- Alghamdi, A.; Hassan, A.M.; Tolah, A.M.; Alamari, S.S.; Alzahrani, A.A.; Alsaaidi, G.A.; Abujamel, T.S.; Azhar, E.I.; Hashem, A.M. Molecular Evidence of Influenza A Virus Circulation in African Dromedary Camels Imported to Saudi Arabia, 2017–2018. Open Forum Infect. Dis. 2019, 6, ofz370. [Google Scholar] [CrossRef]
- Chaiwattanarungruengpaisan, S.; Ketchim, N.; Surarith, W.; Thongdee, M.; Prompiram, P.; Tonchiangsai, K.; Tipkantha, W.; Wiriyarat, W.; Paungpin, W. Serologic evidence of pandemic (H1N1) 2009 virus infection in camel and Eld’s deer, Thailand. Vet. World 2021, 14, 2596–2601. [Google Scholar] [CrossRef] [PubMed]
- Caffar Elamin, M.A.; Kheir, S.A. Detection of influenza antibody in animal sera from Kassala region, Sudan, by agar gel diffusion test. Rev. Elev. Med. Vet. Pays Trop. 1985, 38, 127–129. [Google Scholar] [PubMed]
- Olaleye, O.D.; Baba, S.S.; Omolabu, S.A. Preliminary survey for antibodies against respiratory viruses among slaughter camels (Camelus Dromedarius) in north-eastern nigeria. Rev. Sci. Tech. 1989, 8, 779–783. [Google Scholar] [CrossRef] [PubMed]
- Salem, E.; Cook, E.A.J.; Lbacha, H.A.; Oliva, J.; Awoume, F.; Aplogan, G.L.; Hymann, E.C.; Muloi, D.; Deem, S.L.; Alali, S.; et al. Serologic Evidence for Influenza C and D Virus among Ruminants and Camelids, Africa, 1991–2015. Emerg. Infect. Dis. 2017, 23, 1556–1559. [Google Scholar] [CrossRef]
- Adamu, A.M.; Furlong, M.; Ogunlade, S.; Adikwu, A.A.; Anyang, A.S.; Malgwi, A.; Abdulrahman, A.M.; Bida, N.A.; Owolodun, O.A.; Adegboye, O.A. Seroprevalence of Influenza A Virus in Dromedaries in North-Western Nigeria. Pathogens 2022, 11, 1476. [Google Scholar] [CrossRef]
- Murakami, S.; Odagiri, T.; Melaku, S.K.; Bazartseren, B.; Ishida, H.; Takenaka-Uema, A.; Muraki, Y.; Sentsui, H.; Horimoto, T. Influenza D Virus Infection in Dromedary Camels, Ethiopia. Emerg. Infect. Dis. 2019, 25, 1224–1226. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Hulswit, R.J.G.; Widjaja, I.; Raj, V.S.; McBride, R.; Peng, W.; Widagdo, W.; Tortorici, M.A.; van Dieren, B.; Lang, Y.; et al. Identification of sialic acid-binding function for the Middle East respiratory syndrome coronavirus spike glycoprotein. Proc. Natl. Acad. Sci. USA 2017, 114, e8508–e8517. [Google Scholar] [CrossRef] [PubMed]
- Esievo, K.A.; Saror, D.I.; Tulpule, S.S. High sialic acid content of camels’ erythrocytes. Vet. Rec. 1981, 109, 414. [Google Scholar] [CrossRef] [PubMed]
- Adamczyk, B.; Albrecht, S.; Stockmann, H.; Ghoneim, I.M.; Al-Eknah, M.; Al-Busadah, K.A.S.; Karlsson, N.G.; Carrington, S.D.; Rudd, P.M. Pregnancy-Associated Changes of IgG and Serum N-Glycosylation in Camel (Camelus dromedarius). J. Proteome Res. 2016, 15, 3255–3265. [Google Scholar] [CrossRef]
- Accogli, G.; Monaco, D.; El Bahrawy, K.A.; El-Sayed, A.A.; Ciannarella, F.; Beneult, B.; Lacalandra, G.M.; Desantis, S. Morphological and glycan features of the camel oviduct epithelium. Ann. Anat.-Anat. Anz. 2014, 196, 197–205. [Google Scholar] [CrossRef]
- Desantis, S.; Lacalandra, G.M.; Batista, M.; Amann, O.; Antonelli, D.; Monaco, D. Seminal plasma Alters surface Glycoprofile of dromedary camel cryopreserved epididymal spermatozoa. Theriogenology 2021, 167, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Xing, G.; Wang, J.; Li, Z.; Gu, J.; Yan, L.; Lei, J.; Ji, S.; Hu, B.; Gray, G.C.; et al. Characterization of H7N2 Avian Influenza Virus in Wild Birds and Pikas in Qinghai-Tibet Plateau Area. Sci. Rep. 2016, 6, 30974. [Google Scholar] [CrossRef]
- Yan, Y.; Gu, J.Y.; Yuan, Z.C.; Chen, X.Y.; Li, Z.K.; Lei, J.; Hu, B.L.; Yan, L.P.; Xing, G.; Liao, M.; et al. Genetic characterization of H9N2 avian influenza virus in plateau pikas in the Qinghai Lake region of China. Arch. Virol. 2017, 162, 1025–1029. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Cheng, K.; Sun, W.; Xin, Y.; Cai, J.; Ma, R.; Zhao, Q.; Li, L.; Huang, J.; Sang, X.; et al. Lowly pathogenic avian influenza (H9N2) infection in Plateau pika (Ochotona curzoniae), Qinghai Lake, China. Vet. Microbiol. 2014, 173, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Sun, W.; Wang, J.; Guo, J.; Yin, W.; Wu, N.; Li, L.; Yan, Y.; Liao, M.; Huang, Y.; et al. Characterization of the H5N1 highly pathogenic avian influenza virus derived from wild pikas in China. J. Virol. 2009, 83, 8957–8964. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiao, H.; Huang, C.; Sun, H.; Li, L.; Su, J.; Ma, J.; Liu, D.; Wang, H.; Liu, W.; et al. Distribution of sialic acid receptors and experimental infections with different subtypes of influenza A viruses in Qinghai-Tibet plateau wild pika. Virol. J. 2015, 12, 63. [Google Scholar] [CrossRef]
- Cha, S.Y.; Seo, H.S.; Kang, M.; Jang, H.K. Serologic Survey for Antibodies to Canine Parvovirus and Influenza Virus in Wild Raccoon Dogs (Nyctereutes procyonoides) in South Korea. J. Wildl. Dis. 2013, 49, 200–202. [Google Scholar] [CrossRef][Green Version]
- Rijks, J.M.; Hesselink, H.; Lollinga, P.; Wesselman, R.; Prins, P.; Weesendorp, E.; Engelsma, M.; Heutink, R.; Harders, F.; Kik, M.; et al. Highly Pathogenic Avian Influenza A(H5N1) Virus in Wild Red Foxes, the Netherlands, 2021. Emerg. Infect. Dis. 2021, 27, 2960–2962. [Google Scholar] [CrossRef]
- Bordes, L.; Vreman, S.; Heutink, R.; Roose, M.; Venema, S.; Pritz-Verschuren, S.B.E.; Rijks, J.M.; Gonzales, J.L.; Germeraad, E.A.; Engelsma, M.; et al. Highly Pathogenic Avian Influenza H5N1 Virus Infections in Wild Red Foxes (Vulpes vulpes) Show Neurotropism and Adaptive Virus Mutations. Microbiol. Spectr. 2023, 11, e0286722. [Google Scholar] [CrossRef]
- Qi, X.; Li, X.; Rider, P.; Fan, W.X.; Gu, H.W.; Xu, L.T.; Yang, Y.H.; Lu, S.W.; Wang, H.; Liu, F.Y. Molecular Characterization of Highly Pathogenic H5N1 Avian Influenza A Viruses Isolated from Raccoon Dogs in China. PLoS ONE 2009, 4, e4682. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.; Shou-yu, G.; Feng-xia, Z.; Peng, Y.; Wen-jian, S.; Jian-liang, L.; Jiang, S.; Zhi-jing, X. Molecular characteristics of H9N2 influenza viruses isolated from farmed raccoon dogs and arctic foxes in China. Res. Vet. Sci. 2021, 135, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Hiono, T.; Kobayashi, D.; Kobayashi, A.; Suzuki, T.; Satake, Y.; Harada, R.; Matsuno, K.; Sashika, M.; Ban, H.; Kobayashi, M.; et al. Virological, pathological, and glycovirological investigations of an Ezo red fox and a tanuki naturally infected with H5N1 high pathogenicity avian influenza viruses in Hokkaido, Japan. Virology 2023, 578, 35–44. [Google Scholar] [CrossRef]
- Plumer, L.; Davison, J.; Saarma, U. Rapid Urbanization of Red Foxes in Estonia: Distribution, Behaviour, Attacks on Domestic Animals, and Health-Risks Related to Zoonotic Diseases. PLoS ONE 2014, 9, e115124. [Google Scholar] [CrossRef] [PubMed]
- Stankov, S.; Lalošević, D.; Fooks, A.R. History of Rabies Incidence and Rabies Control in Serbia in Support of the Zero by 2030 Campaign to Eliminate Dog-Mediated Human Rabies. Viruses 2021, 14, 75. [Google Scholar] [CrossRef]
- Kandeil, A.; Gomaa, M.R.; Shehata, M.M.; El Taweel, A.N.; Mahmoud, S.H.; Bagato, O.; Moatasim, Y.; Kutkat, O.; Kayed, A.S.; Dawson, P.; et al. Isolation and Characterization of a Distinct Influenza A Virus from Egyptian Bats. J. Virol. 2019, 93, e01059-18. [Google Scholar] [CrossRef]
- Freidl, G.S.; Binger, T.; Muller, M.A.; de Bruin, E.; van Beek, J.; Corman, V.M.; Rasche, A.; Drexler, J.F.; Sylverken, A.; Oppong, S.K.; et al. Serological evidence of influenza A viruses in frugivorous bats from Africa. PLoS ONE 2015, 10, e0127035. [Google Scholar] [CrossRef] [PubMed]
- Chothe, S.K.; Bhushan, G.; Nissly, R.H.; Yeh, Y.T.; Brown, J.; Turner, G.; Fisher, J.; Sewall, B.J.; Reeder, D.M.; Terrones, M.; et al. Avian and human influenza virus compatible sialic acid receptors in little brown bats. Sci. Rep. 2017, 7, 660. [Google Scholar] [CrossRef]
- FAOSTAT, Compare Data, Production—Crops and Livestock Products in 2020. 2023. Available online: https://www.fao.org/faostat/en/#compare (accessed on 22 February 2022).
- Muranaka, M.; Yamanaka, T.; Katayama, Y.; Hidari, K.; Kanazawa, H.; Suzuki, T.; Oku, K.; Oyamada, T. Distribution of influenza virus sialoreceptors on upper and lower respiratory tract in horses and dogs. J. Vet. Med. Sci. 2011, 73, 125–127. [Google Scholar] [CrossRef][Green Version]
- Scocco, P.; Pedini, V. Localization of influenza virus sialoreceptors in equine respiratory tract. Histol. Histopathol. 2008, 23, 973–978. [Google Scholar]
- Suzuki, Y.; Ito, T.; Suzuki, T.; Holland, R.E., Jr.; Chambers, T.M.; Kiso, M.; Ishida, H.; Kawaoka, Y. Sialic acid species as a determinant of the host range of influenza A viruses. J. Virol. 2000, 74, 11825–11831. [Google Scholar] [CrossRef] [PubMed]
- Wen, F.; Blackmon, S.; Olivier, A.K.; Li, L.; Guan, M.; Sun, H.; Wang, P.G.; Wan, X.F. Mutation W222L at the Receptor Binding Site of Hemagglutinin Could Facilitate Viral Adaption from Equine Influenza A(H3N8) Virus to Dogs. J. Virol. 2018, 92, e01115-18. [Google Scholar] [CrossRef]
- Spruit, C.M.; Zhu, X.; Tomris, I.; Rios-Carrasco, M.; Han, A.X.; Broszeit, F.; van der Woude, R.; Bouwman, K.M.; Luu, M.M.T.; Matsuno, K.; et al. N-Glycolylneuraminic Acid Binding of Avian and Equine H7 Influenza A Viruses. J. Virol. 2022, 96, e0212021. [Google Scholar] [CrossRef]
- Sangkachai, N.; Thongdee, M.; Chaiwattanarungruengpaisan, S.; Buddhirongawatr, R.; Chamsai, T.; Poltep, K.; Wiriyarat, W.; Paungpin, W. Serological evidence of influenza virus infection in captive wild felids, Thailand. J. Vet. Med. Sci. 2019, 81, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Iwatsuki-Horimoto, K.; Nakajima, N.; Ichiko, Y.; Sakai-Tagawa, Y.; Noda, T.; Hasegawa, H.; Kawaoka, Y. Syrian Hamster as an Animal Model for the Study of Human Influenza Virus Infection. J. Virol. 2018, 92, e01693-17. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Bi, Y.; Pu, J.; Hu, Y.; Wang, J.; Gao, H.; Liu, L.; Xu, Q.; Tan, Y.; Liu, M.; et al. Guinea pig model for evaluating the potential public health risk of swine and avian influenza viruses. PLoS ONE 2010, 5, e15537. [Google Scholar] [CrossRef]
- Newby, C.M.; Rowe, R.K.; Pekosz, A. Influenza A virus infection of primary differentiated airway epithelial cell cultures derived from Syrian golden hamsters. Virology 2006, 354, 80–90. [Google Scholar] [CrossRef]
- Lowen, A.C.; Mubareka, S.; Tumpey, T.M.; García-Sastre, A.; Palese, P. The guinea pig as a transmission model for human influenza viruses. Proc. Natl. Acad. Sci. USA 2006, 103, 9988–9992. [Google Scholar] [CrossRef]
- Ganti, K.; Bagga, A.; DaSilva, J.; Shepard, S.S.; Barnes, J.R.; Shriner, S.; Koelle, K.; Lowen, A.C. Avian Influenza A Viruses Reassort and Diversify Differently in Mallards and Mammals. Viruses 2021, 13, 509. [Google Scholar] [CrossRef]
- Ning, Z.Y.; Luo, M.Y.; Qi, W.B.; Yu, B.; Jiao, P.R.; Liao, M. Detection of expression of influenza virus receptors in tissues of BALB/c mice by histochemistry. Vet. Res. Commun. 2009, 33, 895–903. [Google Scholar] [CrossRef]
- Leyva-Grado, V.H.; Mubareka, S.; Krammer, F.; Cárdenas, W.B.; Palese, P. Influenza virus infection in guinea pigs raised as livestock, Ecuador. Emerg. Infect. Dis. 2012, 18, 1135–1138. [Google Scholar] [CrossRef]
- Lvov, D.K.; Zdanov, V.M.; Sazonov, A.A.; Braude, N.A.; Vladimirtceva, E.A.; Agafonova, L.V.; Skljanskaja, E.I.; Kaverin, N.V.; Reznik, V.I.; Pysina, T.V.; et al. Comparison of influenza viruses isolated from man and from whales. Bull World Health Organ. 1978, 56, 923–930. [Google Scholar]
- Mandler, J.; Gorman, O.T.; Ludwig, S.; Schroeder, E.; Fitch, W.M.; Webster, R.G.; Scholtissek, C. Derivation of the Nucleoproteins (Np) of Influenza-a Viruses Isolated from Marine Mammals. Virology 1990, 176, 255–261. [Google Scholar] [CrossRef]
- Nymo, I.H.; Siebert, U.; Baechlein, C.; Postel, A.; Breines, E.M.; Lydersen, C.; Kovacs, K.M.; Tryland, M. Pathogen Exposure in White Whales (Delphinapterus leucas) in Svalbard, Norway. Pathogens 2023, 12, 58. [Google Scholar] [CrossRef]
- Hinshaw, V.S.; Bean, W.J.; Geraci, J.; Fiorelli, P.; Early, G.; Webster, R.G. Characterization of two influenza A viruses from a pilot whale. J. Virol. 1986, 58, 655–656. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-J. Transmission of Avian Influenza Virus by Humpback Whale and Its Stranding along the Atlantic Coast with CO2 Emissions. Adv. Asp. Eng. Res. 2021, 3, 59–81. [Google Scholar]
- Costa, T.; Chaves, A.J.; Valle, R.; Darji, A.; van Riel, D.; Kuiken, T.; Majo, N.; Ramis, A. Distribution patterns of influenza virus receptors and viral attachment patterns in the respiratory and intestinal tracts of seven avian species. Vet. Res. 2012, 43, 28. [Google Scholar] [CrossRef]
- Gambaryan, A.S.; Tuzikov, A.B.; Bovin, N.V.; Yamnikova, S.S.; Lvov, D.K.; Webster, R.G.; Matrosovich, M.N. Differences between influenza virus receptors on target cells of duck and chicken and receptor specificity of the 1997 H5N1 chicken and human influenza viruses from Hong Kong. Avian Dis. 2003, 47 (Suppl. 3), 1154–1160. [Google Scholar] [CrossRef]
- Gambaryan, A.S.; Tuzikov, A.B.; Pazynina, G.V.; Webster, R.G.; Matrosovich, M.N.; Bovin, N.V. H5N1 chicken influenza viruses display a high binding affinity for Neu5Acalpha2-3Galbeta1-4(6-HSO3)GlcNAc-containing receptors. Virology 2004, 326, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Kuchipudi, S.V.; Nelli, R.; White, G.A.; Bain, M.; Chang, K.C.; Dunham, S. Differences in influenza virus receptors in chickens and ducks: Implications for interspecies transmission. J. Mol. Genet. Med. Int. J. Biomed. Res. 2009, 3, 143–151. [Google Scholar] [CrossRef]
- Kim, J.A.; Ryu, S.Y.; Seo, S.H. Cells in the respiratory and intestinal tracts of chickens have different proportions of both human and avian influenza virus receptors. J. Microbiol. 2005, 43, 366–369. [Google Scholar]
- Suzuki, N.; Abe, T.; Natsuka, S. Structural analysis of N-glycans in chicken trachea and lung reveals potential receptors of chicken influenza viruses. Sci. Rep. 2022, 12, 2081. [Google Scholar] [CrossRef]
- Wan, H.; Perez, D.R. Quail carry sialic acid receptors compatible with binding of avian and human influenza viruses. Virology 2006, 346, 278–286. [Google Scholar] [CrossRef]
- Pillai, S.P.; Lee, C.W. Species and age related differences in the type and distribution of influenza virus receptors in different tissues of chickens, ducks and turkeys. Virol. J. 2010, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.E.; Yoon, H.; Lee, H.J.; Lee, J.H.; Chang, B.J.; Song, C.S.; Nahm, S.S. Expression patterns of influenza virus receptors in the respiratory tracts of four species of poultry. J. Vet. Sci. 2011, 12, 7–13. [Google Scholar] [CrossRef][Green Version]
- Guo, C.T.; Takahashi, N.; Yagi, H.; Kato, K.; Takahashi, T.; Yi, S.Q.; Chen, Y.; Ito, T.; Otsuki, K.; Kida, H.; et al. The quail and chicken intestine have sialyl-galactose sugar chains responsible for the binding of influenza A viruses to human type receptors. Glycobiology 2007, 17, 713–724. [Google Scholar] [CrossRef]
- Han, D.P.; Hu, Y.X.; Teng, K.D.; Deng, X.M. Lower expression of sialic acid receptors in the cecum of silky fowl (Gallus gallus domesticus Brisson) compared to white leghorn. Poult. Sci. 2016, 95, 1290–1295. [Google Scholar] [CrossRef]
- Wang, J.Y.; Chen, Z.L.; Li, C.S.; Cao, X.L.; Wang, R.; Tang, C.; Huang, J.J.; Chang, C.D.; Liu, H.J. The distribution of sialic acid receptors of avian influenza virus in the reproductive tract of laying hens. Mol. Cell. Probes 2015, 29, 129–134. [Google Scholar] [CrossRef]
- Pillai, S.P.; Pantin-Jackwood, M.; Yassine, H.M.; Saif, Y.M.; Lee, C.W. The high susceptibility of turkeys to influenza viruses of different origins implies their importance as potential intermediate hosts. Avian Dis. 2010, 54 (Suppl. 1), 522–526. [Google Scholar] [CrossRef] [PubMed]
- Kimble, B.; Nieto, G.R.; Perez, D.R. Characterization of influenza virus sialic acid receptors in minor poultry species. Virol. J. 2010, 7, 365. [Google Scholar] [CrossRef]
- Bertran, K.; Dolz, R.; Majó, N. Pathobiology of avian influenza virus infection in minor gallinaceous species: A review. Avian Pathol. 2014, 43, 9–25. [Google Scholar] [CrossRef]
- Humberd, J.; Guan, Y.; Webster, R.G. Comparison of the replication of influenza A viruses in Chinese ring-necked pheasants and chukar partridges. J. Virol. 2006, 80, 2151–2161. [Google Scholar] [CrossRef]
- Kim, J.K.; Negovetich, N.J.; Forrest, H.L.; Webster, R.G. Ducks: The “Trojan horses” of H5N1 influenza. Influenza Other Respir. Viruses 2009, 3, 121–128. [Google Scholar] [CrossRef]
- Wan, X.F. Lessons from emergence of A/goose/Guangdong/1996-like H5N1 highly pathogenic avian influenza viruses and recent influenza surveillance efforts in southern China. Zoonoses Public Health 2012, 59 (Suppl. 2), 32–42. [Google Scholar] [CrossRef]
- Abolnik, C. A current review of avian influenza in pigeons and doves (Columbidae). Vet. Microbiol. 2014, 170, 181–196. [Google Scholar] [CrossRef]
- Liu, Y.; Han, C.; Wang, X.; Lin, J.; Ma, M.; Shu, Y.; Zhou, J.; Yang, H.; Liang, Q.; Guo, C.; et al. Influenza A virus receptors in the respiratory and intestinal tracts of pigeons. Avian Pathol. 2009, 38, 263–266. [Google Scholar] [CrossRef] [PubMed]
- Ellstrom, P.; Jourdain, E.; Gunnarsson, O.; Waldenstrom, J.; Olsen, B. The “human influenza receptor” Neu5Ac alpha2,6Gal is expressed among different taxa of wild birds. Arch. Virol. 2009, 154, 1533–1537. [Google Scholar] [CrossRef]
- Bisset, A.T.; Hoyne, G.F. An Outbreak of Highly Pathogenic Avian Influenza (H7N7) in Australia and the Potential for Novel Influenza A Viruses to Emerge. Microorganisms 2021, 9, 1639. [Google Scholar] [CrossRef] [PubMed]
- Gujjar, N.; Chothe, S.K.; Gawai, S.; Nissly, R.; Bhushan, G.; Kanagaraj, V.; Jayarao, B.M.; Kathaperumal, K.; Subbiah, M.; Kuchipudi, S.V. Co-expression of sialic acid receptors compatible with avian and human influenza virus binding in emus (Dromaius novaehollandiae). Virology 2017, 500, 114–121. [Google Scholar] [CrossRef]
- Nili, H.; Mohammadi, A.; Habibi, H.; Firouzi, S. Pathogenesis of H9N2 virus in Chukar partridges. Avian Pathol. 2013, 42, 230–234. [Google Scholar] [CrossRef]
- Callaghan, C.T.; Nakagawa, S.; Cornwell, W.K. Global abundance estimates for 9,700 bird species. Proc. Natl. Acad. Sci. USA 2021, 118, e2023170118. [Google Scholar] [CrossRef]
- Franca, M.; Stallknecht, D.E.; Howerth, E.W. Expression and distribution of sialic acid influenza virus receptors in wild birds. Avian Pathol. 2013, 42, 60–71. [Google Scholar] [CrossRef]
- Rahman, M.T.; Sobur, M.A.; Islam, M.S.; Ievy, S.; Hossain, M.J.; El Zowalaty, M.E.; Rahman, A.T.; Ashour, H.M. Zoonotic Diseases: Etiology, Impact, and Control. Microorganisms 2020, 8, 1405. [Google Scholar] [CrossRef] [PubMed]
- Piret, J.; Boivin, G. Pandemics Throughout History. Front. Microbiol. 2020, 11, 631736. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, R.; Saad, M.D.; Davis, C.T.; Swayne, D.E.; Wang, D.; Wong, F.Y.K.; McCauley, J.W.; Peiris, J.S.M.; Webby, R.J.; Fouchier, R.A.M.; et al. Pandemic potential of highly pathogenic avian influenza clade 2.3.4.4 A(H5) viruses. Rev. Med. Virol. 2020, 30, e2099. [Google Scholar] [CrossRef] [PubMed]
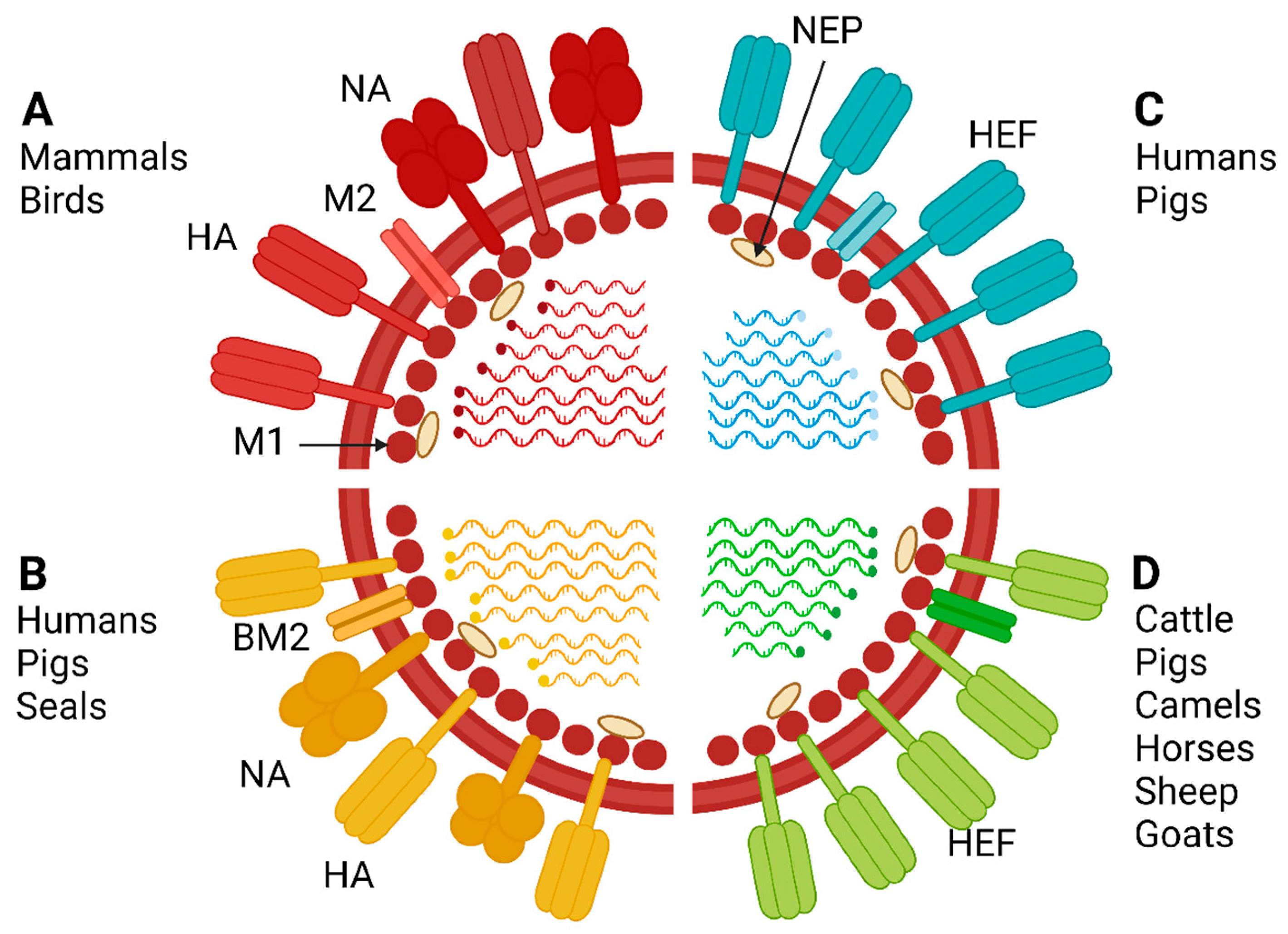


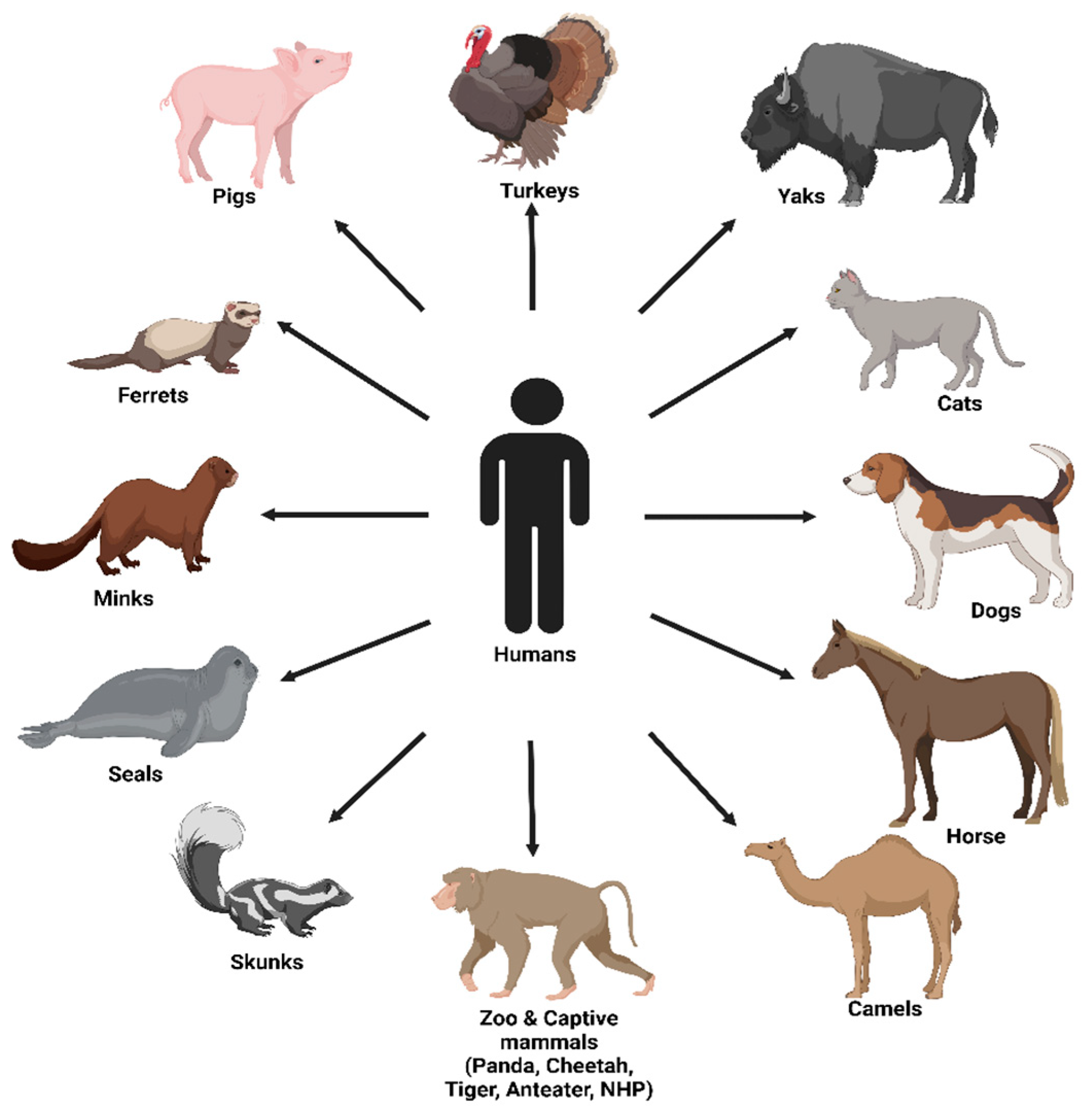
| Subtype | Virus/Strain | Total Number of Tested Individuals | Positive Individuals (%) | Country | Year | Reference |
|---|---|---|---|---|---|---|
| H5 | H5N1 | 22 | 7 (32%) | China | 2013–2014 | [117] |
| H5N1 | 110 | 1 (0.9) | China | 2006 | [118] | |
| H5N1 | 87 | 2 (2.3%) | China | 2005–2008 | [119] | |
| H5N1 | 306 | 8 (2.6%) | China | 2010 | [120] | |
| H5N1 | 249 | 5 (2.0%) | China | 2010 | [121] | |
| H5N1 | 501 | 4 (0.8%) | China | 2013 | [122] | |
| H5N1 | 652 | 6 (0.9) | China | 2014–2016 | [112] | |
| H5N1 | 2310 | 18 (0.8%) | China | 2014 | [123] | |
| H5N1 | 964 | 18 (1.9%) | China | 2013–2016 | [124] | |
| H5N1 | 2124 | 75 (3.5%) | China | 2014–2016 | [112] | |
| H5N1 | 35159 | 862 (2.45%) | China | 1997–2018 | [99] * | |
| H5N1 | 2512 | 9 (0.4%) | South Korea | 2003–2004 | [125] | |
| H5N1 | 200 | 6 (3%) | Vietnam | 2001 | [126] | |
| H5N1 | 747 | 37 (5%) | Vietnam | 2008–2009 | [127] | |
| H5N1 | 607 | 37 (6.1%) | Vietnam | 2011 | [128] | |
| H5N1 | 111 | 5 (4.5%) | Cambodia | 2013 | [129] | |
| H5N1 | 3594 | 37 (1.0%) | Cambodia | 2006–2014 | [130,131,132,133] | |
| H5N1 | 800 | 45 (5.6%) | Thailand | 2008 | [109] | |
| H5N1 | 101 | 63 (62%) | Indonesia | 2012–2014 | [111] | |
| H5N1 | 376 | 1 (0.3%) | Turkey | 2006 | [134] | |
| H5N1 | 316 | 3 (0.9%) | Nigeria | 2008–2011 | [110] | |
| H5N1 | 369 | 1 (0.3%) | Nigeria | 2009 | [135] | |
| H5N1 | 708 | 15 (2.1%) | Egypt | 2010–2012 | [136] | |
| H5N1 | 2397 | 9 (0.4%) | Egypt | 2015–2019 | [137] | |
| H5N2 | 1247 | 18 (1.4%) | Taiwan | 2012 | [138] | |
| H5N2 | 369 | 2 (0.5%) | Nigeria | 2009 | [135] | |
| H5N2 | 310 | 13 (4.2%) § | South Africa | 2011–2012 | [139] | |
| H5N2 | 42 | 5 (11.9%) | USA | 2002–2004 | [140] | |
| H7 | H7N1 | 310 | 6 (1.9%) § | South Africa | 2011–2012 | [139] |
| H7N2 | 787 | 34 (4.3%) | USA | 2004 | [113] | |
| H7N2 | 80 | 1 (1.3%) | USA | 2002 | [141] | |
| H7N2 | 42 | 6 (14.3%) | USA | 2002–2004 | [140] | |
| H7N3 | 1247 | 7 (0.6%) | Taiwan | 2012 | [138] | |
| H7N3 | 185 | 7 (3.8%) | Italy | 2003 | [142] | |
| H7N3/H7N1 | 188 | 6 (3.2%) | Italy | 2008–2010 | [143] | |
| H7N3 | 157 | 1 (0.6%) | USA | 2009–2010 | [144] | |
| H7N7 | 1214 | 1 (0.1) | China | 2004 | [145] | |
| H7N7 | 354 | 75 (21.2%) § | Pakistan | 2010–2011 | [146] | |
| H7N7 | 490 | Not clear | Pakistan | 2013 | [147] | |
| H7N7 | 56 | 33 (58.9%) | Netherlands | 2003 | [97] | |
| H7N7 | 649 | 14 (2.2%) | Egypt | 2012–2013 | [148] | |
| H7N9 | 27 | 1 (3.7%) | China | 2013 | [149] | |
| H7N9 | 96 | 52 (54.2%) | China | 2013 | [122] | |
| H7N9 | 396 | 25 (6.3%) | China | 2013 | [107] | |
| H7N9 | 361 | 3 (0.8%) | China | 2013–2014 | [150] | |
| H7N9 | 10 | 1 (10%) | China | 2013–2014 | [117] | |
| H7N9 | 12 | 1 (8.3%) | China | 2013–2014 | [117] | |
| H7N9 | 1056 | 4 (0.4%) | China | 2014 | [123] | |
| H7N9 | 35 | 5 (14.3%) | China | 2014 | [151] | |
| H7N9 | 964 | 9 (0.9%) | China | 2013–2016 | [124] | |
| H7N9 | 225 | 22 (9.8%) | China | 2013–2016 | [95] | |
| H7N9 | 2124 | 82 (3.9%) | China | 2014–2016 | [112] | |
| H9 | H9Nx | 400 | 7 (1.8%) | Vietnam | 2001 | [126] |
| H9N2 | 59590 | 3313 (5.6%) | China | 1990s–2018 | [104] * | |
| H9N2 | 111 | 2 (1.8%) | Cambodia | 2013 | [129] | |
| H9N2 | 777 | 21 (2.7%) | Cambodia | 2008 | [152] | |
| H9N2 | 768 | 21 (2.7%) | Thailand | 2008 | [153] | |
| H9N2 | 784 | 40 (5.1%) | Thailand | 2008 | [153] | |
| H9N2 | 338 | 21 (6.2%) | India | 2012 | [154] | |
| H9N2 | 347 | 4 (1.2%) | Mongolia | 2008–2011 | [155] | |
| H9N2 | 490 | 421 (86%) | Pakistan | 2013 | [147] | |
| H9N2 | 332 | 167 (50.3%) | Pakistan | 2016–2017 | [156] | |
| H9N2 | 354 | 169 (47.7%) § | Pakistan | 2010–2011 | [146] | |
| H9N2 | 435 § | 238 (54.7%) | Pakistan | n.a. | [157] | |
| H9N2 | 161 | 25 (15.5%) | Pakistan | 2015–2016 | [158] | |
| H9N2 | 127 | 48 (37.7%) | Iran | 2006 | [159] | |
| H9N2 | 182 | 21 (11.5%) | Iran | 2010–2011 | [160] | |
| H9N2 | 200 | 20 (10%) | Iran | 2012 | [161] | |
| H9N2 | 34 | 11 (32.3%) | Lebanon | 2005 | [162] | |
| H9N2 | 363 | 33 (9.1%) | Romania | 2009–2010 | [163] | |
| H9N2 | Not mentioned | 1 | Romania | 2010 | [164] | |
| H9N2 | 682 | 51 (7.5%) | Egypt | 2010–2012 | [136] | |
| H9N2 | 2397 | 266 (11.1%) | Egypt | 2015–2019 | [137] | |
| H9N2 | 369 | 4 (1.1%) | Nigeria | 2009 | [135] | |
| H9N2 | 42 | 4 (9.5%) | USA | 2002–2004 | [140] | |
| H9N2 | 91 | 4 (4.4%) | USA | 2007–2008 | [116] | |
| H9N2 | 157 | 1 (0.6%) | USA | 2009–2010 | [144] | |
| H9N2 | 787 | 15 (1.9%) | USA | 2004 | [113] | |
| H9N8 | 57 | 3 (5.3%) | Italy | 2005–2006 | [114] | |
| Total | ~138,730 | ~6639 |
| Subtype | Year | Example | Country | Reference |
|---|---|---|---|---|
| pdmH1N1 | 2010 | A/elephant seal/California/2/2010 | USA | [268] |
| H3N3 | 1992 | A/seal/Massachusetts/3911/1992 | USA | [283] |
| H3N8 | 2011 | A/harbor seal/New Hampshire/179629/2011 | USA | [282] |
| H3N8 | 2011 | A/harbor seal/Massachusetts/1/2011 | USA | [279] |
| H3N8 | 2017 | A/grey seal/England/027661/2017 | UK | [284] |
| H4N5 | 1982 | A/seal/Massachusetts/133/1982 | USA | [279] |
| H4N6 | 2002 | A/Caspian seal/Russia/1884/2002 | Russia | [285] |
| H4N6 | 2012 | A/Caspian seal/Russia/T1/2012 | Russia | [286] |
| H5N1 | 2022 | A/harbor seal/Maine/22-020455-001-original/2022 | USA | [275] |
| H5N1 | 2022 | Harbor seal | Canada | [287] |
| H5N1 | 2022–2023 | Grey and harbor seals | UK | [288,289] |
| H5N8 | 2016 | A/grey seal/361-10/BalticPL/2016 | Poland | [290] |
| H5N8 | 2020 | A/seal/England/AVP-031141/2020 | UK | [291] |
| H5N8 | 2021 | A/seal/Germany-SH/AI05373/2021 | Germany | [292] |
| H5N8 | 2021 | A/seal/Sweden/2021 | Sweden | [292] |
| H5N8 | 2021 | A/harbor seal/Denmark/521-2/2021 | Denmark | [287] |
| H7N7 | 1980 | A/seal/Massachusetts/1/1980 | USA | [276,293] |
| H10N7 | 2021 | A/harbor seal/British Colombia/OTH-52-1/2021 | Canada | [274] |
| H10N7 | 2014 | A/harbor seal/Germany/1/2014 | Europe * | [272] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdelwhab, E.M.; Mettenleiter, T.C. Zoonotic Animal Influenza Virus and Potential Mixing Vessel Hosts. Viruses 2023, 15, 980. https://doi.org/10.3390/v15040980
Abdelwhab EM, Mettenleiter TC. Zoonotic Animal Influenza Virus and Potential Mixing Vessel Hosts. Viruses. 2023; 15(4):980. https://doi.org/10.3390/v15040980
Chicago/Turabian StyleAbdelwhab, Elsayed M., and Thomas C. Mettenleiter. 2023. "Zoonotic Animal Influenza Virus and Potential Mixing Vessel Hosts" Viruses 15, no. 4: 980. https://doi.org/10.3390/v15040980
APA StyleAbdelwhab, E. M., & Mettenleiter, T. C. (2023). Zoonotic Animal Influenza Virus and Potential Mixing Vessel Hosts. Viruses, 15(4), 980. https://doi.org/10.3390/v15040980






