Thrombophilia and Immune-Related Genetic Markers in Long COVID
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethic Aspects
2.2. Sample Processing and Genotyping
2.3. Statistical Analyses
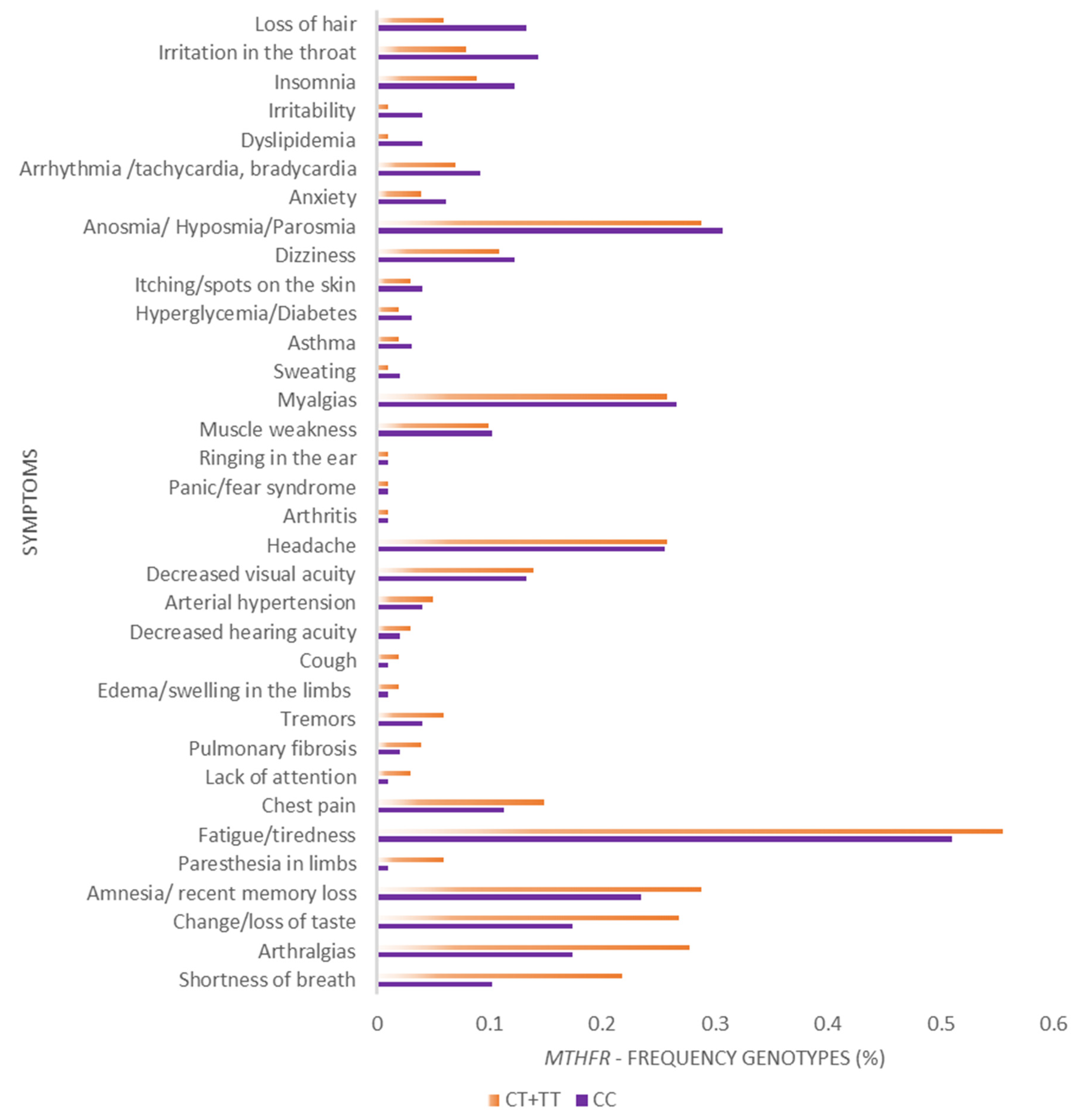
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marco Cascella, A.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; di Napoli, R. Italy Uniformed Services Un of the Health Sc Istituto Nazionale Tumori-IRCCS-Fondazione Pascale. 2020. Available online: https://pubmed.ncbi.nlm.nih.gov/32150360/ (accessed on 18 February 2023).
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-Month Consequences of COVID-19 in Patients Discharged from Hospital: A Cohort Study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Doykov, I.; Hällqvist, J.; Gilmour, K.C.; Grandjean, L.; Mills, K.; Heywood, W.E. “The Long Tail of COVID-19”—The Detection of a Prolonged Inflammatory Response after a SARS-CoV-2 Infection in Asymptomatic and Mildly Affected Patients. F1000Research 2020, 9, 1349. [Google Scholar] [CrossRef] [PubMed]
- del Rio, C.; Collins, L.F.; Malani, P. Long-Term Health Consequences of COVID-19. JAMA—J. Am. Med. Assoc. 2020, 324, 1723–1724. [Google Scholar] [CrossRef] [PubMed]
- Mehandru, S.; Merad, M. Pathological Sequelae of Long-Haul COVID. Nat. Immunol. 2022, 23, 194–202. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Knight, M.; A’Court, C.; Buxton, M.; Husain, L. Management of Post-Acute COVID-19 in Primary Care. BMJ 2020, 370, m3026. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence (NICE) COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19. 2020. Available online: https://www.nice.org.uk/guidance/ng188/resources/covid19-rapid-guideline-managing-the-longterm-effects-of-covid19-pdf-51035515742 (accessed on 4 February 2023).
- Chen, C.; Haupert, S.R.; Zimmermann, L.; Shi, X.; Fritsche, L.G.; Mukherjee, B. Global Prevalence of Post-Coronavirus Disease 2019 (COVID-19) Condition or Long COVID: A Meta-Analysis and Systematic Review. J. Infect. Dis. 2022, 226, 1593–1607. [Google Scholar] [CrossRef]
- Mahase, E. Long Covid Could Be Four Different Syndromes, Review Suggests. BMJ 2020, 371, m3981. [Google Scholar] [CrossRef]
- Buonsenso, D.; Piazza, M.; Boner, A.L.; Bellanti, J.A. Long COVID: A Proposed Hypothesis-Driven Model of Viral Persistence for the Pathophysiology of the Syndrome. Allergy Asthma Proc. 2022, 43, 187–193. [Google Scholar] [CrossRef]
- Queiroz, M.A.F.; Neves, P.F.M.D.; Lima, S.S.; Lopes, J.D.C.; Torres, M.K.D.S.; Vallinoto, I.M.V.C.; Bichara, C.D.A.; Santos, E.F.D.; de Brito, M.T.F.M.; da Silva, A.L.S.; et al. Cytokine Profiles Associated with Acute COVID-19 and Long COVID-19 Syndrome. Front Cell Infect. Microbiol. 2022, 12. [Google Scholar] [CrossRef]
- Fernández-de-las-Peñas, C.; Arendt-Nielsen, L.; Díaz-Gil, G.; Gil-Crujera, A.; Gómez-Sánchez, S.M.; Ambite-Quesada, S.; Palomar-Gallego, M.A.; Pellicer-Valero, O.J.; Giordano, R. ACE1 Rs1799752 Polymorphism Is Not Associated with Long-COVID Symptomatology in Previously Hospitalized COVID-19 Survivors. J. Infect. 2022, 86, e67–e69. [Google Scholar] [CrossRef]
- Health Organization, W. Guideline Clinical Management of COVID-19 Patients: Living Guideline, 18 November 2021. 2021. Available online: https://www.who.int/publications/i/item/WHO-2019-nCoV-clinical-2021-2 (accessed on 20 February 2023).
- Little, J.; Higgins, J.P.T.; Ioannidis, J.P.A.; Moher, D.; Gagnon, F.; von Elm, E.; Khoury, M.J.; Cohen, B.; Davey-Smith, G.; Grimshaw, J.; et al. STrengthening the REporting of Genetic Association Studies (STREGA)-an Extension of the Strobe Statement. PLoS Med. 2009, 6, 0151–0163. [Google Scholar] [CrossRef] [PubMed]
- Amraei, R.; Yin, W.; Napoleon, M.A.; Suder, E.L.; Berrigan, J.; Zhao, Q.; Olejnik, J.; Chandler, K.B.; Xia, C.; Feldman, J.; et al. CD209L/L-SIGN and CD209/DC-SIGN Act as Receptors for SARS-CoV-2. ACS Cent. Sci. 2021, 7, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Tso, H.W.; Ip, W.K.; Chong, W.P.; Tam, C.M.; Chiang, A.K.S.; Lau, Y.L. Association of Interferon Gamma and Interleukin 10 Genes with Tuberculosis in Hong Kong Chinese. Genes Immun. 2005, 6, 358–363. [Google Scholar] [CrossRef]
- Conde, S.R.S.; Feitosa, R.N.M.; Freitas, F.B.; Hermes, R.B.; Demachki, S.; Araújo, M.T.F.; Soares, M.C.P.; Ishak, R.; Vallinoto, A.C.R. Association of Cytokine Gene Polymorphisms and Serum Concentrations with the Outcome of Chronic Hepatitis, B. Cytokine 2013, 61, 940–944. [Google Scholar] [CrossRef]
- Medina, T.S.; Costa, S.P.; Oliveira, M.D.; Ventura, A.M.; Souza, J.M.; Gomes, T.F.; Vallinoto, A.C.R.; Póvoa, M.M.; Silva, J.S.; Cunha, M.G. Increased Interleukin-10 and Interferon-Levels in Plasmodium Vivax Malaria Suggest a Reciprocal Regulation Which Is Not Altered by IL-10 Gene Promoter Polymorphism. Malar. J. 2011, 10, 264. [Google Scholar] [CrossRef]
- Santiago, A.M.; da Silva Graça Amoras, E.; Queiroz, M.A.F.; da Silva Conde, S.R.S.; Cayres-Vallinoto, I.M.V.; Ishak, R.; Vallinoto, A.C.R. TNFA -308G>A and IL10 -1082A>G Polymorphisms Seem to Be Predictive Biomarkers of Chronic HCV Infection. BMC Infect. Dis. 2021, 21, s12879–s021. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, H.P.M.; Martins, L.C.; dos Santos, S.E.B.; Demachki, S.; Assumpção, M.B.; Aragão, C.D.; de Oliveira Corvelo, T.C. Interleukin-1 and TNF-α Polymorphisms and Helicobacter Pylori in a Brazilian Amazon Population. World J. Gastroenterol. 2009, 15, 1465–1471. [Google Scholar] [CrossRef] [PubMed]
- de Brito, W.B.; Queiroz, M.A.F.; da Silva Graça Amoras, E.; Lima, S.S.; da Silva Conde, S.R.S.; dos Santos, E.J.M.; Cayres-Vallinoto, I.M.V.; Ishak, R.; Vallinoto, A.C.R. The TGFB1 -509C/T Polymorphism and Elevated TGF-Β1 Levels Are Associated with Chronic Hepatitis C and Cirrhosis. Immunobiology 2020, 225, 152002. [Google Scholar] [CrossRef]
- Keiko Nascimento Yoshioka, F.; Góes Araújo, A.; Haydee Tavella, M.; Guerreiro Hamoy, I.; Farias Guerreiro, J. Prevalence of Hereditary Risk Factors for Thrombophilia in Belém, Brazilian Amazon. Genet. Mol. Biol. 2006, 29, 38–40. [Google Scholar] [CrossRef]
- Queiroz, M.A.F.; Santiago, A.M.; Moura, T.C.F.; Amoras, E.D.S.G.; Conde, S.R.S.D.S.; Cayres-Vallinoto, I.M.V.; Ishak, R.; Vallinoto, A.C.R. The IL6-174G/C Polymorphism Associated with High Levels of IL-6 Contributes to HCV Infection, but Is Not Related to HBV Infection, in the Amazon Region of Brazil. Viruses 2022, 14, 507. [Google Scholar] [CrossRef]
- Oliveira, L.F.D.; Lima, C.P.S.D.; Azevedo, R.D.S.S.; Mendonça, D.S.F.D.; Rodrigues, S.G.; Carvalho, V.L.; Pinto, E.V.; Maia, A.L.; Maia, M.H.T.; Vasconcelos, J.M.; et al. Polymorphism of DC-SIGN (CD209) Promoter in Association with Clinical Symptoms of Dengue Fever. Viral Immunol. 2014, 27, 245–249. [Google Scholar] [CrossRef]
- Bai, F.; Tomasoni, D.; Falcinella, C.; Barbanotti, D.; Castoldi, R.; Mulè, G.; Augello, M.; Mondatore, D.; Allegrini, M.; Cona, A.; et al. Female Gender Is Associated with Long COVID Syndrome: A Prospective Cohort Study. Clin. Microbiol. Infect. 2022, 28, 611.e9–611.e16. [Google Scholar] [CrossRef] [PubMed]
- Falchi, A.; Fernández-De-Las-Peñas, C.; Martín-Guerrero, J.D.; Pellicer-Valero, Ó.J.; Navarro-Pardo, E.; Gómez-Mayordomo, V.; Cuadrado, M.L.; Arias-Navalón, J.A.; Cigarán-Méndez, M.; Hernández-Barrera, V.; et al. Female Sex Is a Risk Factor Associated with Long-Term Post-COVID Related-Symptoms but Not with COVID-19 Symptoms: The LONG-COVID-EXP-CM Multicenter Study. J. Clin. Med. 2022, 11, 413. [Google Scholar] [CrossRef]
- Riccardo, M.; Inciardi; Alvin, C. Editorial Commentary: Long COVID-19: A Tangled Web of Lungs, Heart, Mind, and Gender. Trends Cardiovasc. Med. 2022, 32, 18–19. [Google Scholar]
- Sylvester, S.V.; Rusu, R.; Chan, B.; Bellows, M.; O’Keefe, C.; Nicholson, S. Sex Differences in Sequelae from COVID-19 Infection and in Long COVID Syndrome: A Review. Curr. Med. Res. Opin. 2022, 38, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Nirantharakumar, K.; Hughes, S.; Myles, P.; Williams, T.; Gokhale, K.M.; Taverner, T.; Chandan, J.S.; Brown, K.; Simms-Williams, N.; et al. Symptoms and Risk Factors for Long COVID in Non-Hospitalized Adults. Nat. Med. 2022, 28, 1706–1714. [Google Scholar] [CrossRef] [PubMed]
- Torjesen, I. COVID-19: Middle Aged Women Face Greater Risk of Debilitating Long Term Symptoms. BMJ 2021, 372, n829. [Google Scholar] [CrossRef]
- Xie, Y.; Xu, E.; Bowe, B.; Al-Aly, Z. Long-Term Cardiovascular Outcomes of COVID-19. Nat. Med. 2022, 28, 583–590. [Google Scholar] [CrossRef]
- Liew, S.C.; Gupta, E.D. Methylenetetrahydrofolate Reductase (MTHFR) C677T Polymorphism: Epidemiology, Metabolism and the Associated Diseases. Eur J. Med. Genet. 2015, 58, 1–10. [Google Scholar] [CrossRef]
- Meadows, D.N.; Pyzik, M.; Wu, Q.; Torre, S.; Gros, P.; Vidal, S.M.; Rozen, R. Increased Resistance to Malaria in Mice with Methylenetetrahydrofolate Reductase (Mthfr) Deficiency Suggests a Mechanism for Selection of the MTHFR 677C>T (c.665C>T) Variant. Hum. Mutat. 2014, 35, 594–600. [Google Scholar] [CrossRef]
- Vickers, T.J.; Orsomando, G.; de La Garza, R.D.; Scott, D.A.; Kang, S.O.; Hanson, A.D.; Beverley, S.M. Biochemical and Genetic Analysis of Methylenetetrahydrofolate Reductase in Leishmania Metabolism and Virulence. J. Biol. Chem. 2006, 281, 38150–38158. [Google Scholar] [CrossRef] [PubMed]
- Hajiesmaeil, M.; Tafvizi, F.; Sarmadi, S. The Effect of Methylenetetrahydrofolate Reductase Polymorphisms on Susceptibility to Human Papilloma Virus Infection and Cervical Cancer. Infect. Genet. Evol. 2016, 46, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fodil-Cornu, N.; Kozij, N.; Wu, Q.; Rozen, R.; Vidal, S.M. Methylenetetrahydrofolate Reductase (MTHFR) Deficiency Enhances Resistance against Cytomegalovirus Infection. Genes Immun. 2009, 10, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Baba, H.; Bouqdayr, M.; Saih, A.; Bensghir, R.; Ouladlahsen, A.; Sodqi, M.; Marih, L.; Zaidane, I.; Kettani, A.; Abidi, O.; et al. Association between Methylene-Tetrahydrofolate Reductase C677T Polymorphism and Human Immunodeficiency Virus Type 1 Infection in Morocco. Lab. Med. 2023, 54, 23–29. [Google Scholar] [CrossRef]
- Karakus, N.; Duygu, F.; Rustemoglu, A.; Yigit, S. Methylene-Tetrahydrofolate Reductase Gene C677T and A1298C Polymorphisms as a Risk Factor for Crimean-Congo Hemorrhagic Fever. Nucleosides Nucleotides Nucleic Acids 2022, 41, 878–890. [Google Scholar] [CrossRef]
- Bronowicki, J.P.; Abdelmouttaleb, I.; Peyrin-Biroulet, L.; Venard, V.; Khiri, H.; Chabi, N.; Amouzou, E.K.; Barraud, H.; Halfon, P.; Sanni, A.; et al. Methylenetetrahydrofolate Reductase 677 T Allele Protects against Persistent HBV Infection in West Africa. J. Hepatol. 2008, 48, 532–539. [Google Scholar] [CrossRef]
- Ponti, G.; Pastorino, L.; Manfredini, M.; Ozben, T.; Oliva, G.; Kaleci, S.; Iannella, R.; Tomasi, A. COVID-19 Spreading across World Correlates with C677T Allele of the Methylenetetrahydrofolate Reductase (MTHFR) Gene Prevalence. J. Clin. Lab. Anal. 2021, 35, e23798. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Pérez, O.; Merino, E.; Leon-Ramirez, J.M.; Andres, M.; Ramos, J.M.; Arenas-Jiménez, J.; Asensio, S.; Sanchez, R.; Ruiz-Torregrosa, P.; Galan, I.; et al. Post-Acute COVID-19 Syndrome. Incidence and Risk Factors: A Mediterranean Cohort Study. J. Infect. 2021, 82, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Ding, X.; Deng, J.; Lu, Y.; Matsuda, Z.; Thiel, A.; Chen, J.; Deng, H.; Qin, Z. Interferon-Gamma Negatively Regulates Th17-Mediated Immunopathology during Mouse Hepatitis Virus Infection. J. Mol. Med. 2011, 89, 399–409. [Google Scholar] [CrossRef]
- Zielinski, C.E.; Mele, F.; Aschenbrenner, D.; Jarrossay, D.; Ronchi, F.; Gattorno, M.; Monticelli, S.; Lanzavecchia, A.; Sallusto, F. Pathogen-Induced Human T H17 Cells Produce IFN-γ or IL-10 and Are Regulated by IL-1β. Nature 2012, 484, 514–518. [Google Scholar] [CrossRef]
- Kak, G.; Raza, M.; Tiwari, B.K. Interferon-Gamma (IFN-γ): Exploring Its Implications in Infectious Diseases. Biomol. Concepts 2018, 9, 64–79. [Google Scholar] [CrossRef] [PubMed]
- Pirault, J.; Polyzos, K.A.; Petri, M.H.; Ketelhuth, D.F.J.; Bäck, M.; Hansson, G.K. The Inflammatory Cytokine Interferon-Gamma Inhibits Sortilin-1 Expression in Hepatocytes via the JAK/STAT Pathway. Eur. J. Immunol. 2017, 47, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- Mitok, K.A.; Keller, M.P.; Attie, A.D. Sorting through the Extensive and Confusing Roles of Sortilin in Metabolic Disease. J. Lipid. Res. 2022, 63, 100243. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, M.A.F.; Azevedo, V.N.; Amoras, E.D.S.G.; Moura, T.C.F.; Guimarães Ishak, M.D.O.; Ishak, R.; Vallinoto, A.C.R.; Martins Feitosa, R.N. IFNG +874A/T Polymorphism among Asymptomatic HTLV-1-Infected Individuals Is Potentially Related to a Worse Prognosis. Front. Microbiol. 2018, 9, 795. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, P.A.S.; Assone, T.; Prates, G.; Tedeschi, M.R.M.; Fonseca, L.A.M.; Casseb, J. The Role of IFN-γ Production during Retroviral Infections: An Important Cytokine Involved in Chronic Inflammation and Pathogenesis. Rev. Inst. Med. Trop. Sao Paulo 2022, 64. [Google Scholar] [CrossRef] [PubMed]
- Sun, P.; García, J.; Comach, G.; Vahey, M.T.; Wang, Z.; Forshey, B.M.; Morrison, A.C.; Sierra, G.; Bazan, I.; Rocha, C.; et al. Sequential Waves of Gene Expression in Patients with Clinically Defined Dengue Illnesses Reveal Subtle Disease Phases and Predict Disease Severity. PLoS Negl. Trop. Dis. 2013, 7, e2298. [Google Scholar] [CrossRef]


| Variables | NLC * n = 79 (28%) | LC * n = 199 (72%) | Total n = 278 (100%) |
|---|---|---|---|
| Sex (n, %) | |||
| Female | 40 (51%) | 140 (70%) | 180 (65%) |
| Male | 39 (49%) | 59 (30%) | 98 (35%) |
| Age range (years) (n, %) | |||
| ≤59 | 76 (96%) | 169 (85%) | 245 (88%) |
| ≥60 | 3 (4%) | 30 (15%) | 33 (12%) |
| Age median (years) | 43 ± 14 | 48 ± 13 | 46 ± 14 |
| Smoker or ex-smoker | |||
| Yes | 4 (5%) | 16 (8%) | 20 (7%) |
| No | 75 (95%) | 183 (92%) | 258 (93%) |
| Major Comorbidities | |||
| Heart Diseas. | 6 (7%) a | 48 (24%) a | 54 (19%) |
| Obesity | 6 (7%) | 12 (6%) | 18 (6.5%) |
| Asthma | 0 | 17 (8.5%) | 17 (6.1%) |
| Diabetes Mellitus | 0 | 7 (3.5%) | 7 (2.5%) |
| Arterial hypertension | 2 (2%) | 4 (2%) | 6 (2%) |
| Liver Disease | 0 | 4 (2%) | 4 (1.4%) |
| Immunosuppression, autoimmune disease | 0 | 3 (1.5%) | 3 (1.0%) |
| Chronic kidney disease | 1 (1.3%) | 1 (0.5%) | 2 (0.7%) |
| Hypothyroidism | 1 (1.3%) | 1 (0.5%) | 2 (0.7%) |
| Major symptoms—acute phase COVID-19 (n, %) | |||
| Myalgias | 41 (52%) | 155 (78%) | 196 (70%) |
| Headache | 49 (63%) | 140 (70%) | 189 (68%) |
| Loss of taste | 46 (58%) | 137 (69%) | 183 (66%) |
| Fatigue/tiredness | 36 (45%) | 146 (73%) | 182 (65%) |
| Anosmia | 48 (61%) | 127 (64%) | 175 (63%) |
| Fever | 49 (62%) | 125 (63%) | 174 (62%) |
| Cough | 40 (51%) | 117 (59%) | 157 (56%) |
| Shortness of breath | 23 (29%) | 123 (62%) | 146 (52%) |
| Muscle weakness | 37 (47%) | 109 (55%) | 146 (52%) |
| Chest pain | 26 (33%) | 113 (57%) | 139 (50%) |
| Sore throat | 32 (40%) | 106 (53%) | 138 (49%) |
| Diarrhea | 27 (34%) | 76 (38%) | 103 (37%) |
| Coryza | 29 (37%) | 71 (35%) | 100 (36%) |
| Abdominal pain | 14 (17%) | 65 (33%) | 79 (28%) |
| Nausea | 11 (14%) | 64 (32%) | 75 (27%) |
| Retro-orbital pain | 24 (30%) | 36 (18%) | 60 (22%) |
| Vomit | 6 (7%) | 42 (21%) | 48 (17%) |
| Disease severity—acute phase COVID-19 (n, %) ** | |||
| Asymptomatic | 3 (4%) | 0 | 3 (1%) |
| Mild | 76 (96%) | 199 (100%) | 275 (99%) |
| Number of symptoms presented in long COVID (n, %) | |||
| Up to 3 symptoms | - | 121 (61%) | - |
| More than 3 to 9 symptoms | - | 67 (34%) | - |
| More than 9 to 15 symptoms | - | 11 (5%) | - |
| Major symptoms in long COVID (n, %) | |||
| Fatigue/tiredness | - | 106 (53%) | - |
| Anosmia/hyposmia/parosmia | - | 59 (29%) | - |
| Myalgias | - | 52 (26%) | - |
| Amnesia/ recent memory loss | - | 52 (26%) | - |
| Headache | - | 51 (25%) | - |
| Arthralgias | - | 45 (22%) | - |
| Change/loss of taste | - | 44 (22%) | - |
| Shortness of breath | - | 32 (16%) | - |
| Decreased visual acuity | - | 27 (13%) | - |
| Chest pain | - | 26 (13%) | - |
| Dizziness | - | 23 (11%) | - |
| Irritation in the throat/throat | - | 22 (11%) | - |
| Insomnia | - | 21 (10%) | - |
| Muscle weakness | - | 20 (10%) | - |
| Loss of hair | - | 19 (9%) | - |
| Arrhythmia | - | 16 (8%) | - |
| Anxiety | - | 10 (5%) | - |
| Tremors | - | 10 (5%) | - |
| Arterial hypertension | - | 9 (4%) | - |
| Itching/spots on the skin | - | 7 (3%) | - |
| Paresthesia in limbs | - | 7 (3%) | - |
| Pulmonary fibrosis | - | 6 (3%) | - |
| Asthma | - | 5 (2%) | - |
| Decreased hearing acuity | - | 5 (2%) | - |
| Dyslipidemia | - | 5 (2%) | - |
| Hyperglycemia/Diabetes | - | 5 (2%) | - |
| Irritability | - | 5 (2%) | - |
| Facial paralysis | - | 4 (2%) | - |
| Lack of attention | - | 4 (2%) | - |
| Edema/swelling in the limbs | - | 3 (1.5%) | - |
| Sweating | - | 3 (1.5%) | - |
| Arthritis | - | 2 (1%) | - |
| Depression | - | 2 (1%) | - |
| Nausea | - | 2 (1%) | - |
| Panic/fear syndrome | - | 2 (1%) | - |
| Ringing in the ear | - | 2 (1%) | - |
| Weight gain/loss | - | 2 (1%) | - |
| Urinary incontinence/dysuria | - | 1 (0.5%) | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, R.; de Sarges, K.M.L.; Cantanhede, M.H.D.; da Costa, F.P.; dos Santos, E.F.; Rodrigues, F.B.B.; de Nazaré do Socorro de Almeida Viana, M.; de Meira Leite, M.; da Silva, A.L.S.; de Brito, M.T.M.; et al. Thrombophilia and Immune-Related Genetic Markers in Long COVID. Viruses 2023, 15, 885. https://doi.org/10.3390/v15040885
da Silva R, de Sarges KML, Cantanhede MHD, da Costa FP, dos Santos EF, Rodrigues FBB, de Nazaré do Socorro de Almeida Viana M, de Meira Leite M, da Silva ALS, de Brito MTM, et al. Thrombophilia and Immune-Related Genetic Markers in Long COVID. Viruses. 2023; 15(4):885. https://doi.org/10.3390/v15040885
Chicago/Turabian Styleda Silva, Rosilene, Kevin Matheus Lima de Sarges, Marcos Henrique Damasceno Cantanhede, Flávia Póvoa da Costa, Erika Ferreira dos Santos, Fabíola Brasil Barbosa Rodrigues, Maria de Nazaré do Socorro de Almeida Viana, Mauro de Meira Leite, Andréa Luciana Soares da Silva, Mioni Thieli Magalhães de Brito, and et al. 2023. "Thrombophilia and Immune-Related Genetic Markers in Long COVID" Viruses 15, no. 4: 885. https://doi.org/10.3390/v15040885
APA Styleda Silva, R., de Sarges, K. M. L., Cantanhede, M. H. D., da Costa, F. P., dos Santos, E. F., Rodrigues, F. B. B., de Nazaré do Socorro de Almeida Viana, M., de Meira Leite, M., da Silva, A. L. S., de Brito, M. T. M., da Silva Torres, M. K., Queiroz, M. A. F., Vallinoto, I. M. V. C., Henriques, D. F., dos Santos, C. P., Viana, G. M. R., Quaresma, J. A. S., Falcão, L. F. M., Vallinoto, A. C. R., & dos Santos, E. J. M. (2023). Thrombophilia and Immune-Related Genetic Markers in Long COVID. Viruses, 15(4), 885. https://doi.org/10.3390/v15040885









