Genetic Characterization and Pathogenesis of Avian Influenza Virus H3N8 Isolated from Chinese pond heron in China in 2021
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples Collection and Virus Isolation
2.2. RNA Extraction and RT-PCR
2.3. DNA Cloning and Gene Sequencing
2.4. Genetic and Phylogenetic Analysis
2.5. Determination of 50% Egg Infectious Dose (EID50) and 50% Tissue Culture Infectious Dose (TCID50)
2.6. Animal Experiment
3. Results
3.1. Virus Isolation and Homology Comparison
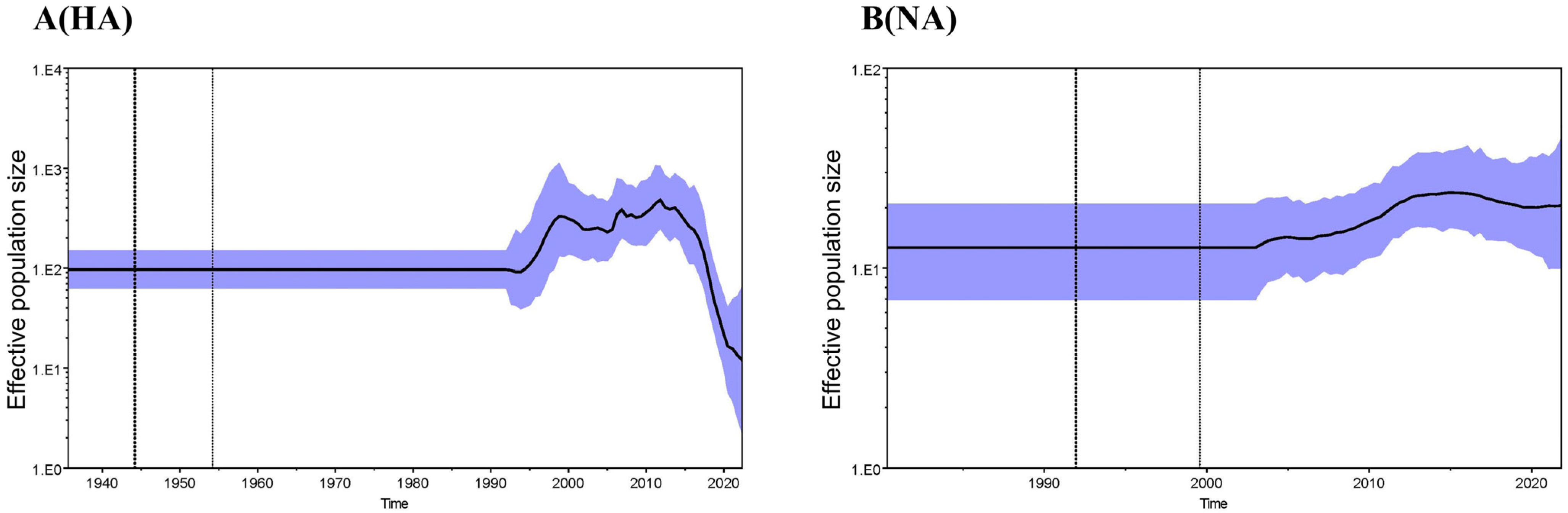
3.2. Phylogenetic Analysis and Hypothesis for Reassortment Event of Each Gene Segment
3.3. Molecular Analysis
| Viral Protein | Amino Acid | JX 5-1 | HN-410 | South Korea2020 (H3N8) | Zhejiang2013 (H3N8) | Xuyi2014 (H3N8) | Amur Region2020 (H3N8) | Comments | Reference |
|---|---|---|---|---|---|---|---|---|---|
| PB2 | L89V | V | V | V | V | V | V | Increased polymerase activity and virulence in mammals | [41] |
| G309D | D | D | D | D | D | N | Increased polymerase activity and virulence in mammals | [41] | |
| T339K | K | K | K | K | K | T | Increased polymerase activity and virulence in mammals | [41] | |
| E627K | E | K | E | E | E | E | Mammalian host adaptation | [42,43] | |
| PB1 | H436Y | Y | Y | Y | Y | Y | Y | Increased polymerase activity and virulence | [44] |
| L473V | V | V | V | V | V | V | Increased polymerase activity and replication efficiency | [45] | |
| PA | K356R | K | R | K | K | K | K | Enhanced virulence and mammalian adaptation | [46] |
| N383D | D | D | D | D | D | D | Increased polymerase activity and mammalian adaptation. | [37] | |
| N409S | S | N | S | S | S | S | Increased polymerase activity, viral replication and virulence to mammalian | [47] | |
| M1 | V15I | V | I | V | V | V | V | Increased virulence in mammals | [48] |
| N30D | D | D | D | D | D | D | Increase pathogenicity and transmission in mammals | [49] | |
| A166V | V | A | V | V | V | V | Increased polymerase activity and virulence in mammals | [50] | |
| T215A | A | A | A | A | A | A | Increased virulence in mammals | [35] | |
| M2 | V27I | V | V | V | V | I | V | Reduce the sensitivity of Adamantane | [51] |
| S31N | S | N | S | S | S | S | Reduce the sensitivity of Adamantane | [52,53] | |
| L55F | L | F | L | L | L | L | Increased transmission | [54] | |
| NS1 | P42S | S | S | S | S | - | S | Enhanced virulence in mice | [36] |
3.4. Pathogenicity in Mice
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Russell, C.A.; Kasson, P.M.; Donis, R.O.; Riley, S.; Dunbar, J.; Rambaut, A.; Asher, J.; Burke, S.; Davis, C.T.; Garten, R.J.; et al. Improving pandemic influenza risk assessment. elife 2014, 3, e03883. [Google Scholar] [CrossRef] [PubMed]
- Glezen, W.P. Emerging infections: Pandemic influenza. Epidemiol. Rev. 1996, 18, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Novel Swine-Origin Influenza, A.V.I.T.; Dawood, F.S.; Jain, S.; Finelli, L.; Shaw, M.W.; Lindstrom, S.; Garten, R.J.; Gubareva, L.V.; Xu, X.; Bridges, C.B.; et al. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N. Engl. J. Med. 2009, 360, 2605–2615. [Google Scholar] [CrossRef]
- Chambers, T.M. A brief introduction to equine influenza and equine influenza viruses. Methods Mol. Biol. 2014, 1161, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.W.; Kim, Y.I.; Lim, G.J.; Kwon, H.I.; Si, Y.J.; Park, S.J.; Kim, E.H.; Kim, S.M.; Nguyen, H.D.; Song, M.S.; et al. Comparison of the virulence and transmissibility of canine H3N2 influenza viruses and characterization of their canine adaptation factors. Emerg. Microbes. Infect. 2018, 7, 17. [Google Scholar] [CrossRef]
- Lee, C.T.; Slavinski, S.; Schiff, C.; Merlino, M.; Daskalakis, D.; Liu, D.; Rakeman, J.L.; Misener, M.; Thompson, C.; Leung, Y.L.; et al. Outbreak of Influenza A(H7N2) among Cats in an Animal Shelter with Cat-to-Human Transmission-New York City, 2016. Clin. Infect. Dis. 2017, 65, 1927–1929. [Google Scholar] [CrossRef]
- He, S.; Shi, J.; Qi, X.; Huang, G.; Chen, H.; Lu, C. Lethal infection by a novel reassortant H5N1 avian influenza A virus in a zoo-housed tiger. Microbes. Infect. 2015, 17, 54–61. [Google Scholar] [CrossRef]
- Bodewes, R.; Rubio Garcia, A.; Brasseur, S.M.; Sanchez Conteras, G.J.; van de Bildt, M.W.; Koopmans, M.P.; Osterhaus, A.D.; Kuiken, T. Seroprevalence of Antibodies against Seal Influenza A(H10N7) Virus in Harbor Seals and Gray Seals from the Netherlands. PLoS ONE 2015, 10, e0144899. [Google Scholar] [CrossRef]
- Hinshaw, V.S.; Bean, W.J.; Geraci, J.; Fiorelli, P.; Early, G.; Webster, R.G. Characterization of two influenza A viruses from a pilot whale. J. Virol. 1986, 58, 655–656. [Google Scholar] [CrossRef]
- Taubenberger, J.K.; Kash, J.C. Influenza virus evolution, host adaptation, and pandemic formation. Cell Host Microbe 2010, 7, 440–451. [Google Scholar] [CrossRef]
- Olsen, B.; Munster, V.J.; Wallensten, A.; Waldenstrom, J.; Osterhaus, A.D.; Fouchier, R.A. Global patterns of influenza a virus in wild birds. Science 2006, 312, 384–388. [Google Scholar] [CrossRef]
- Hensley, S.E.; Das, S.R.; Bailey, A.L.; Schmidt, L.M.; Hickman, H.D.; Jayaraman, A.; Viswanathan, K.; Raman, R.; Sasisekharan, R.; Bennink, J.R.; et al. Hemagglutinin receptor binding avidity drives influenza A virus antigenic drift. Science 2009, 326, 734–736. [Google Scholar] [CrossRef] [PubMed]
- Ghebrehewet, S.; MacPherson, P.; Ho, A. Influenza. BMJ 2016, 355, i6258. [Google Scholar] [CrossRef] [PubMed]
- Saunders-Hastings, P.R.; Krewski, D. Reviewing the History of Pandemic Influenza: Understanding Patterns of Emergence and Transmission. Pathogens 2016, 5, 66. [Google Scholar] [CrossRef] [PubMed]
- Webster, R.G.; Laver, W.G.; Air, G.M.; Schild, G.C. Molecular mechanisms of variation in influenza viruses. Nature 1982, 296, 115–121. [Google Scholar] [CrossRef]
- Krammer, F.; Smith, G.J.D.; Fouchier, R.A.M.; Peiris, M.; Kedzierska, K.; Doherty, P.C.; Palese, P.; Shaw, M.L.; Treanor, J.; Webster, R.G.; et al. Influenza. Nat. Rev. Dis. Primers 2018, 4, 3. [Google Scholar] [CrossRef]
- Chan, P.K. A review on human influenza A H5N1 infections in Hong Kong. Sci. China C Life Sci. 2009, 52, 412–418. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, T.; Ou, X.; Liu, R.; Yang, Y.; Ye, W.; Chen, J.; Yao, D.; Sun, B.; Zhang, X.; et al. Clinical, epidemiological and virological characteristics of the first detected human case of avian influenza A (H5N6) virus. Infect. Genet. Evol. 2016, 40, 236–242. [Google Scholar] [CrossRef]
- Gao, R.; Cao, B.; Hu, Y.; Feng, Z.; Wang, D.; Hu, W.; Chen, J.; Jie, Z.; Qiu, H.; Xu, K.; et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N. Engl. J. Med. 2013, 368, 1888–1897. [Google Scholar] [CrossRef]
- Sovinova, O.; Tumova, B.; Pouska, F.; Nemec, J. Isolation of a virus causing respiratory disease in horses. Acta Virol. 1958, 2, 52–61. [Google Scholar]
- Payungporn, S.; Crawford, P.C.; Kouo, T.S.; Chen, L.M.; Pompey, J.; Castleman, W.L.; Dubovi, E.J.; Katz, J.M.; Donis, R.O. Influenza A virus (H3N8) in dogs with respiratory disease, Florida. Emerg. Infect. Dis. 2008, 14, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Zhou, H.; Jiang, T.; Li, C.; Zhang, A.; Guo, X.; Zou, W.; Chen, H.; Jin, M. Isolation and molecular characterization of equine H3N8 influenza viruses from pigs in China. Arch. Virol. 2009, 154, 887–890. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Wang, L.; Fu, X.; He, S.; Hong, M.; Zhou, P.; Lai, A.; Gray, G.; Li, S. Equine influenza A (H3N8) virus infection in cats. Emerg. Infect. Dis. 2014, 20, 2096–2099. [Google Scholar] [CrossRef]
- Anthony, S.J.; St Leger, J.A.; Pugliares, K.; Ip, H.S.; Chan, J.M.; Carpenter, Z.W.; Navarrete-Macias, I.; Sanchez-Leon, M.; Saliki, J.T.; Pedersen, J.; et al. Emergence of fatal avian influenza in New England harbor seals. mBio 2012, 3, e00166-12. [Google Scholar] [CrossRef]
- Yondon, M.; Zayat, B.; Nelson, M.I.; Heil, G.L.; Anderson, B.D.; Lin, X.; Halpin, R.A.; McKenzie, P.P.; White, S.K.; Wentworth, D.E.; et al. Equine influenza A (H3N8) virus isolated from Bactrian camel, Mongolia. Emerg. Infect. Dis. 2014, 20, 2144–2147. [Google Scholar] [CrossRef]
- Qi, T.; Guo, W.; Huang, W.; Dai, L.; Zhao, L.; Li, H.; Li, X.; Zhang, X.; Wang, Y.; Yan, Y.; et al. Isolation and genetic characterization of H3N8 equine influenza virus from donkeys in China. Vet. Microbiol. 2010, 144, 455–460. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Dong, Y.; Wen, S.; Shi, C. A child with acute respiratory distress syndrome caused by avian influenza H3N8 virus. J. Infect. 2022, 85, 174–211. [Google Scholar] [CrossRef]
- Yang, R.; Sun, H.; Gao, F.; Luo, K.; Huang, Z.; Tong, Q.; Song, H.; Han, Q.; Liu, J.; Lan, Y.; et al. Human infection of avian influenza A H3N8 virus and the viral origins: A descriptive study. Lancet Microbe 2022, 3, E824–E834. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: More models, new heuristics and parallel computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef]
- Suchard, M.A.; Lemey, P.; Baele, G.; Ayres, D.L.; Drummond, A.J.; Rambaut, A. Bayesian phylogenetic and phylodynamic data integration using BEAST 1.10. Virus Evol. 2018, 4, vey016. [Google Scholar] [CrossRef]
- Drummond, A.J.; Bouckaert, R.R. Bayesian Evolutionary Analysis with BEAST; Cambridge University Press: Cambridge, UK, 2015; p. xii. 249p. [Google Scholar]
- Kang, Y.; Shen, X.; Yuan, R.; Xiang, B.; Fang, Z.; Murphy, R.W.; Liao, M.; Shen, Y.; Ren, T. Pathogenicity and transmissibility of three avian influenza A (H5N6) viruses isolated from wild birds. J. Infect. 2018, 76, 286–294. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Manual for the Laboratory Diagnosis and Virological Surveillance of Influenza; World Health Organization: Geneva, Switzerland, 2011; p. xii. 139p. [Google Scholar]
- Steel, J.; Lowen, A.C.; Mubareka, S.; Palese, P. Transmission of influenza virus in a mammalian host is increased by PB2 amino acids 627K or 627E/701N. PLoS Pathog. 2009, 5, e1000252. [Google Scholar] [CrossRef] [PubMed]
- Fan, S.; Deng, G.; Song, J.; Tian, G.; Suo, Y.; Jiang, Y.; Guan, Y.; Bu, Z.; Kawaoka, Y.; Chen, H. Two amino acid residues in the matrix protein M1 contribute to the virulence difference of H5N1 avian influenza viruses in mice. Virology 2009, 384, 28–32. [Google Scholar] [CrossRef] [PubMed]
- Jiao, P.; Tian, G.; Li, Y.; Deng, G.; Jiang, Y.; Liu, C.; Liu, W.; Bu, Z.; Kawaoka, Y.; Chen, H. A single-amino-acid substitution in the NS1 protein changes the pathogenicity of H5N1 avian influenza viruses in mice. J. Virol. 2008, 82, 1146–1154. [Google Scholar] [CrossRef]
- Xu, C.; Hu, W.B.; Xu, K.; He, Y.X.; Wang, T.Y.; Chen, Z.; Li, T.X.; Liu, J.H.; Buchy, P.; Sun, B. Amino acids 473V and 598P of PB1 from an avian-origin influenza A virus contribute to polymerase activity, especially in mammalian cells. J. Gen. Virol. 2012, 93, 531–540. [Google Scholar] [CrossRef]
- Song, J.; Xu, J.; Shi, J.; Li, Y.; Chen, H. Synergistic Effect of S224P and N383D Substitutions in the PA of H5N1 Avian Influenza Virus Contributes to Mammalian Adaptation. Sci. Rep. 2015, 5, 10510. [Google Scholar] [CrossRef]
- Mehle, A.; Dugan, V.G.; Taubenberger, J.K.; Doudna, J.A. Reassortment and mutation of the avian influenza virus polymerase PA subunit overcome species barriers. J. Virol. 2012, 86, 1750–1757. [Google Scholar] [CrossRef]
- Ilyushina, N.A.; Khalenkov, A.M.; Seiler, J.P.; Forrest, H.L.; Bovin, N.V.; Marjuki, H.; Barman, S.; Webster, R.G.; Webby, R.J. Adaptation of Pandemic H1N1 Influenza Viruses in Mice. J. Virol. 2010, 84, 8607–8616. [Google Scholar] [CrossRef]
- Li, J.W.; Ishaq, M.; Prudence, M.; Xi, X.; Hu, T.; Liu, Q.Z.; Guo, D.Y. Single mutation at the amino acid position 627 of PB2 that leads to increased virulence of an H5N1 avian influenza virus during adaptation in mice can be compensated by multiple mutations at other sites of PB2. Virus Res. 2009, 144, 123–129. [Google Scholar] [CrossRef]
- Hatta, M.; Gao, P.; Halfmann, P.; Kawaoka, Y. Molecular basis for high virulence of Hong Kong H5N1 influenza A viruses. Science 2001, 293, 1840–1842. [Google Scholar] [CrossRef]
- Subbarao, E.K.; London, W.; Murphy, B.R. A single amino acid in the PB2 gene of influenza A virus is a determinant of host range. J. Virol. 1993, 67, 1761–1764. [Google Scholar] [CrossRef] [PubMed]
- Taubenberger, J.K.; Reid, A.H.; Lourens, R.M.; Wang, R.; Jin, G.; Fanning, T.G. Characterization of the 1918 influenza virus polymerase genes. Nature 2005, 437, 889–893. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Sun, H.; Sun, Z.; Sun, Y.; Kong, W.; Pu, J.; Ma, G.; Yin, Y.; Yang, H.; Guo, X.; et al. Influenza A virus acquires enhanced pathogenicity and transmissibility after serial passages in swine. J. Virol. 2014, 88, 11981–11994. [Google Scholar] [CrossRef] [PubMed]
- Xu, G.; Zhang, X.; Gao, W.; Wang, C.; Wang, J.; Sun, H.; Sun, Y.; Guo, L.; Zhang, R.; Chang, K.C.; et al. Prevailing PA Mutation K356R in Avian Influenza H9N2 Virus Increases Mammalian Replication and Pathogenicity. J. Virol. 2016, 90, 8105–8114. [Google Scholar] [CrossRef]
- Yamayoshi, S.; Yamada, S.; Fukuyama, S.; Murakami, S.; Zhao, D.; Uraki, R.; Watanabe, T.; Tomita, Y.; Macken, C.; Neumann, G.; et al. Virulence-affecting amino acid changes in the PA protein of H7N9 influenza A viruses. J. Virol. 2014, 88, 3127–3134. [Google Scholar] [CrossRef]
- Sun, Y.; Hu, Z.; Zhang, X.; Chen, M.; Wang, Z.; Xu, G.; Bi, Y.; Tong, Q.; Wang, M.; Sun, H.; et al. An R195K Mutation in the PA-X Protein Increases the Virulence and Transmission of Influenza A Virus in Mammalian Hosts. J. Virol. 2020, 94, 1817–1819. [Google Scholar] [CrossRef]
- Katz, J.M.; Lu, X.; Tumpey, T.M.; Smith, C.B.; Shaw, M.W.; Subbarao, K. Molecular correlates of influenza A H5N1 virus pathogenesis in mice. J. Virol. 2000, 74, 10807–10810. [Google Scholar] [CrossRef]
- Zhu, W.; Zou, X.; Zhou, J.; Tang, J.; Shu, Y. Residues 41V and/or 210D in the NP protein enhance polymerase activities and potential replication of novel influenza (H7N9) viruses at low temperature. Virol. J. 2015, 12, 71. [Google Scholar] [CrossRef]
- Pinto, L.H.; Holsinger, L.J.; Lamb, R.A. Influenza-Virus M2 Protein Has Ion Channel Activity. Cell 1992, 69, 517–528. [Google Scholar] [CrossRef]
- Cappelle, J.; Zhao, D.L.; Gilbert, M.; Nelson, M.I.; Newman, S.H.; Takekawa, J.Y.; Gaidet, N.; Prosser, D.J.; Liu, Y.; Li, P.; et al. Risks of Avian Influenza Transmission in Areas of Intensive Free-Ranging Duck Production with Wild Waterfowl. EcoHealth 2014, 11, 109–119. [Google Scholar] [CrossRef]
- Andreas, L.B.; Eddy, M.T.; Chou, J.J.; Griffin, R.G. Magic-Angle-Spinning NMR of the Drug Resistant S31N M2 Proton Transporter from Influenza A. J. Am. Chem. Soc. 2012, 134, 7215–7218. [Google Scholar] [CrossRef] [PubMed]
- Mei, K.; Liu, G.; Chen, Z.; Gao, Z.; Zhao, L.; Jin, T.; Yu, X.; Chen, Q. Deep sequencing reveals the viral adaptation process of environment-derived H10N8 in mice. Infect. Genet. Evol. 2016, 37, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, D.; Safi, K.; Avril, A.; Fiedler, W.; Wikelski, M.; Gunnarsson, G.; Elmberg, J.; Tolf, C.; Olsen, B.; Waldenstrom, J. Does influenza A virus infection affect movement behaviour during stopover in its wild reservoir host? R. Soc. Open Sci. 2016, 3, 150633. [Google Scholar] [CrossRef]
- Li, Y.; Li, P.; Xi, J.; Yang, J.; Wu, H.; Zhang, Y.; Cao, M.; Chen, M.; Li, Y.; Xiao, C. Wild bird-origin H3N8 avian influenza virus exhibit well adaptation in mammalian host. J. Infect. 2022, 84, 579–613. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yang, L.; Zhu, W.; Wang, D.; Shu, Y. Epidemiological and Genetic Characteristics of the H3 Subtype Avian Influenza Viruses in China. China CDC Wkly. 2021, 3, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Li, Q.; Cai, L.; Yuan, Q.; Chen, L.; Lin, Q.; Xiao, C.; Xiang, B.; Ren, T. Adaptation of Two Wild Bird-Origin H3N8 Avian Influenza Viruses to Mammalian Hosts. Viruses 2022, 14, 1097. [Google Scholar] [CrossRef]
- Chen, H.; Yuan, H.; Gao, R.; Zhang, J.; Wang, D.; Xiong, Y.; Fan, G.; Yang, F.; Li, X.; Zhou, J.; et al. Clinical and epidemiological characteristics of a fatal case of avian influenza A H10N8 virus infection: A descriptive study. Lancet 2014, 383, 714–721. [Google Scholar] [CrossRef]
- Venkatesh, D.; Poen, M.J.; Bestebroer, T.M.; Scheuer, R.D.; Vuong, O.; Chkhaidze, M.; Machablishvili, A.; Mamuchadze, J.; Ninua, L.; Fedorova, N.B.; et al. Avian Influenza Viruses in Wild Birds: Virus Evolution in a Multihost Ecosystem. J. Virol. 2018, 92, e00433-18. [Google Scholar] [CrossRef]
- Ramey, A.M.; Reeves, A.B.; Donnelly, T.; Poulson, R.L.; Stallknecht, D.E. Introduction of Eurasian-Origin Influenza A(H8N4) Virus into North America by Migratory Birds. Emerg. Infect. Dis. 2018, 24, 1950–1953. [Google Scholar] [CrossRef]
- Jeong, S.; Lee, D.H.; Kim, Y.J.; Lee, S.H.; Cho, A.Y.; Noh, J.Y.; Tseren-Ochir, E.O.; Jeong, J.H.; Song, C.S. Introduction of Avian Influenza A(H6N5) Virus into Asia from North America by Wild Birds. Emerg. Infect. Dis. 2019, 25, 2138–2140. [Google Scholar] [CrossRef]
- Sun, J.; Zhao, L.; Li, X.; Meng, W.; Chu, D.; Yang, X.; Peng, P.; Zhi, M.; Qin, S.; Fu, T.; et al. Novel H5N6 avian influenza virus reassortants with European H5N8 isolated in migratory birds, China. Transbound Emerg. Dis. 2020, 67, 648–660. [Google Scholar] [CrossRef] [PubMed]
- THE global Consortium for H5N8 and Related Influenza Viruses. Role for migratory wild birds in the global spread of avian influenza H5N8. Science 2016, 354, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Gu, M.; Liu, D.; Liu, B.; Jiang, K.; Zhong, L.; Liu, K.; Sun, W.; Hu, J.; Wang, X.; et al. Phylogenetic and biological characterization of three K1203 (H5N8)-like avian influenza A virus reassortants in China in 2014. Arch. Virol. 2016, 161, 289–302. [Google Scholar] [CrossRef]
- Casalegno, J.S.; Ferraris, O.; Escuret, V.; Bouscambert, M.; Bergeron, C.; Lines, L.; Excoffier, T.; Valette, M.; Frobert, E.; Pillet, S.; et al. Functional balance between the hemagglutinin and neuraminidase of influenza A(H1N1)pdm09 HA D222 variants. PLoS ONE 2014, 9, e104009. [Google Scholar] [CrossRef] [PubMed]
- Ha, Y.; Stevens, D.J.; Skehel, J.J.; Wiley, D.C. X-ray structure of the hemagglutinin of a potential H3 avian progenitor of the 1968 Hong Kong pandemic influenza virus. Virology 2003, 309, 209–218. [Google Scholar] [CrossRef]
- Schrauwen, E.J.; Fouchier, R.A. Host adaptation and transmission of influenza A viruses in mammals. Emerg. Microbes. Infect. 2014, 3, e9. [Google Scholar] [CrossRef]
- Yamada, S.; Hatta, M.; Staker, B.L.; Watanabe, S.; Imai, M.; Shinya, K.; Sakai-Tagawa, Y.; Ito, M.; Ozawa, M.; Watanabe, T.; et al. Biological and structural characterization of a host-adapting amino acid in influenza virus. PLoS Pathog. 2010, 6, e1001034. [Google Scholar] [CrossRef]
- Mok, C.K.; Yen, H.L.; Yu, M.Y.; Yuen, K.M.; Sia, S.F.; Chan, M.C.; Qin, G.; Tu, W.W.; Peiris, J.S. Amino acid residues 253 and 591 of the PB2 protein of avian influenza virus A H9N2 contribute to mammalian pathogenesis. J. Virol. 2011, 85, 9641–9645. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Jin, S.; Zhang, Y.; Sun, L.; Hu, X.; Zhao, M.; Li, F.; Wang, T.; Sun, W.; et al. PB1 S524G mutation of wild bird-origin H3N8 influenza A virus enhances virulence and fitness for transmission in mammals. Emerg. Microbes. Infect. 2021, 10, 1038–1051. [Google Scholar] [CrossRef]
- Dong, B.B.; Xu, C.L.; Dong, L.B.; Cheng, H.J.; Yang, L.; Zou, S.M.; Chen, M.; Bai, T.; Zhang, Y.; Gao, R.B.; et al. A novel reassortant H3N8 influenza virus isolated from drinking water for duck in a domestic duck farm in Poyang Lake area. Biomed. Environ. Sci. 2013, 26, 546–551. [Google Scholar] [CrossRef]
- Zou, Z.; Chen, S.; Liu, Z.; Jin, M. Identification and genetic analysis of H3N8 subtype influenza viruses isolated from domestic pigeons in Central China. Virus Genes 2016, 52, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Xie, S.; Jiang, X.; Li, Z.; Xu, L.; Wen, K.; Zhang, M.; Liao, M.; Jia, W. Emergence of one novel reassortment H3N8 avian influenza virus in China, originating from North America and Eurasia. Infect. Genet. Evol. 2021, 91, 104782. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Xie, Z.; Song, D.; Luo, S.; Xie, L.; Li, M.; Xie, Z.; Deng, X. Genetic Characterization of a Natural Reassortant H3N8 Avian Influenza Virus Isolated from Domestic Geese in Guangxi, Southern China. Genome Announc. 2014, 2, e00747-14. [Google Scholar] [CrossRef] [PubMed]
- Pu, J.; Liu, Q.F.; Xia, Y.J.; Fan, Y.L.; Brown, E.G.; Tian, F.L.; Liu, J.H. Genetic analysis of H3 subtype influenza viruses isolated from domestic ducks in northern China during 2004–2005. Virus Genes 2009, 38, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Wang, M.; Kawaoka, Y.; Gorman, O.; Ito, T.; Saito, T.; Webster, R.G. Characterization of a new avian-like influenza A virus from horses in China. Virology 1992, 188, 245–255. [Google Scholar] [CrossRef]
- Karlsson, E.A.; Ip, H.S.; Hall, J.S.; Yoon, S.W.; Johnson, J.; Beck, M.A.; Webby, R.J.; Schultz-Cherry, S. Respiratory transmission of an avian H3N8 influenza virus isolated from a harbour seal. Nat. Commun. 2014, 5, 4791. [Google Scholar] [CrossRef]
- Hoffmann, E.; Stech, J.; Guan, Y.; Webster, R.G.; Perez, D.R. Universal primer set for the full-length amplification of all influenza A viruses. Arch. Virol. 2001, 146, 2275–2289. [Google Scholar] [CrossRef]
- Kreibich, A.; Stech, J.; Mettenleiter, T.C.; Stech, O. Simultaneous one-tube full-length amplification of the NA, NP, M, and NS genes of influenza A viruses for reverse genetics. J. Virol. Methods. 2009, 159, 308–310. [Google Scholar] [CrossRef]
- Dong, C.Y.; Sun, X.N.; Guan, Z.H.; Zhang, M.L.; Duan, M. Modulation of influenza A virus replication by microRNA-9 through targeting MCPIP1. J. Med. Virol. 2017, 89, 41–48. [Google Scholar] [CrossRef]
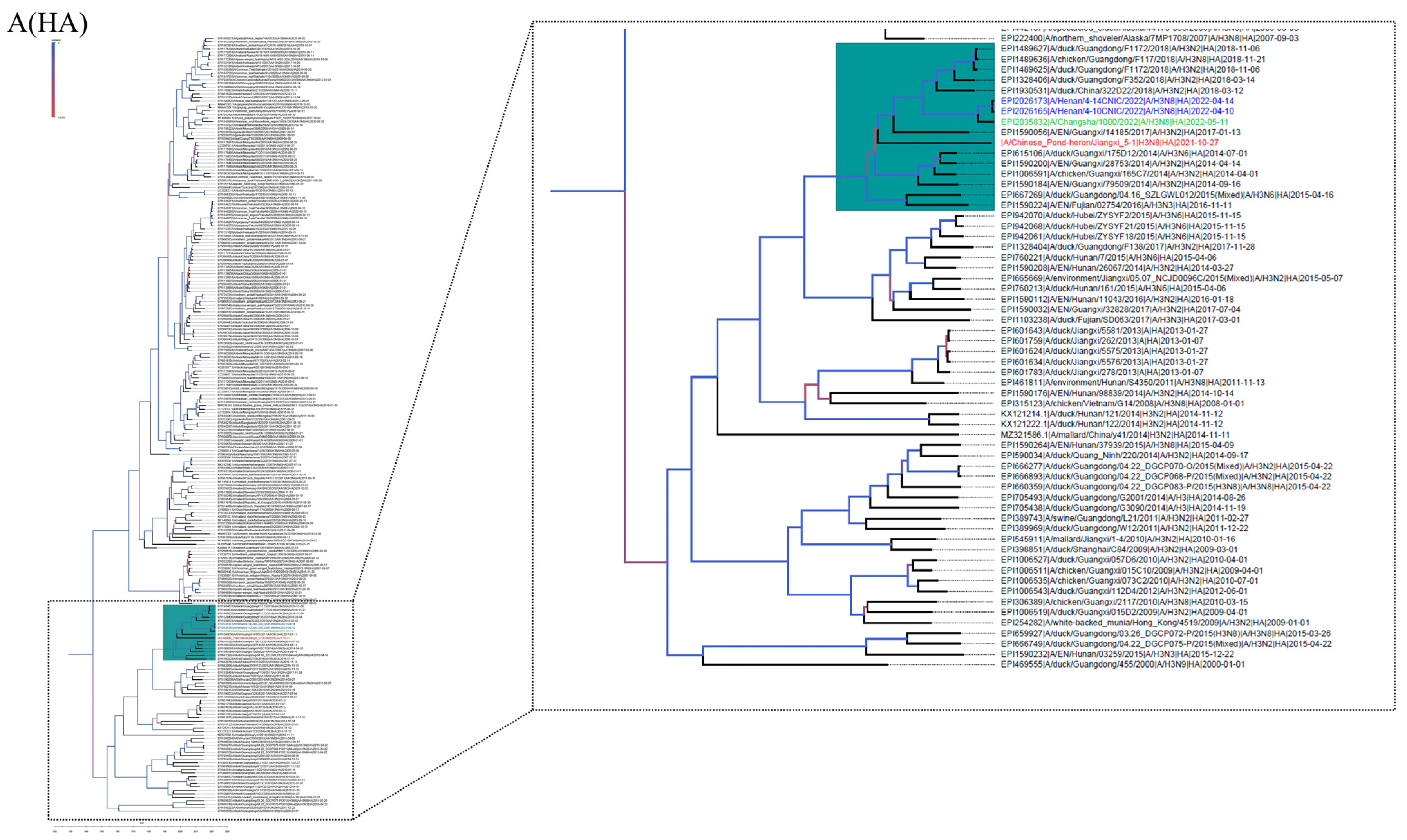
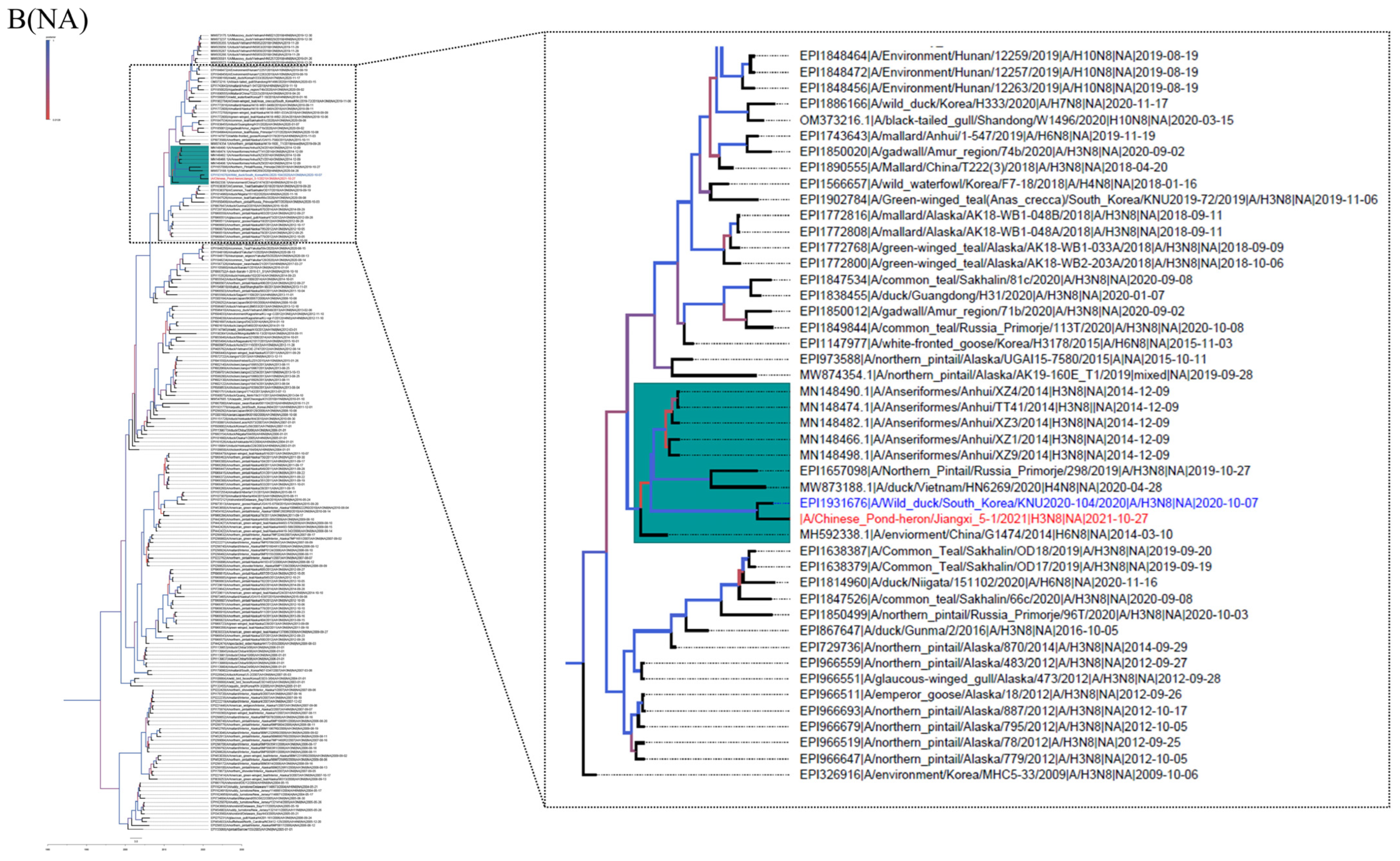
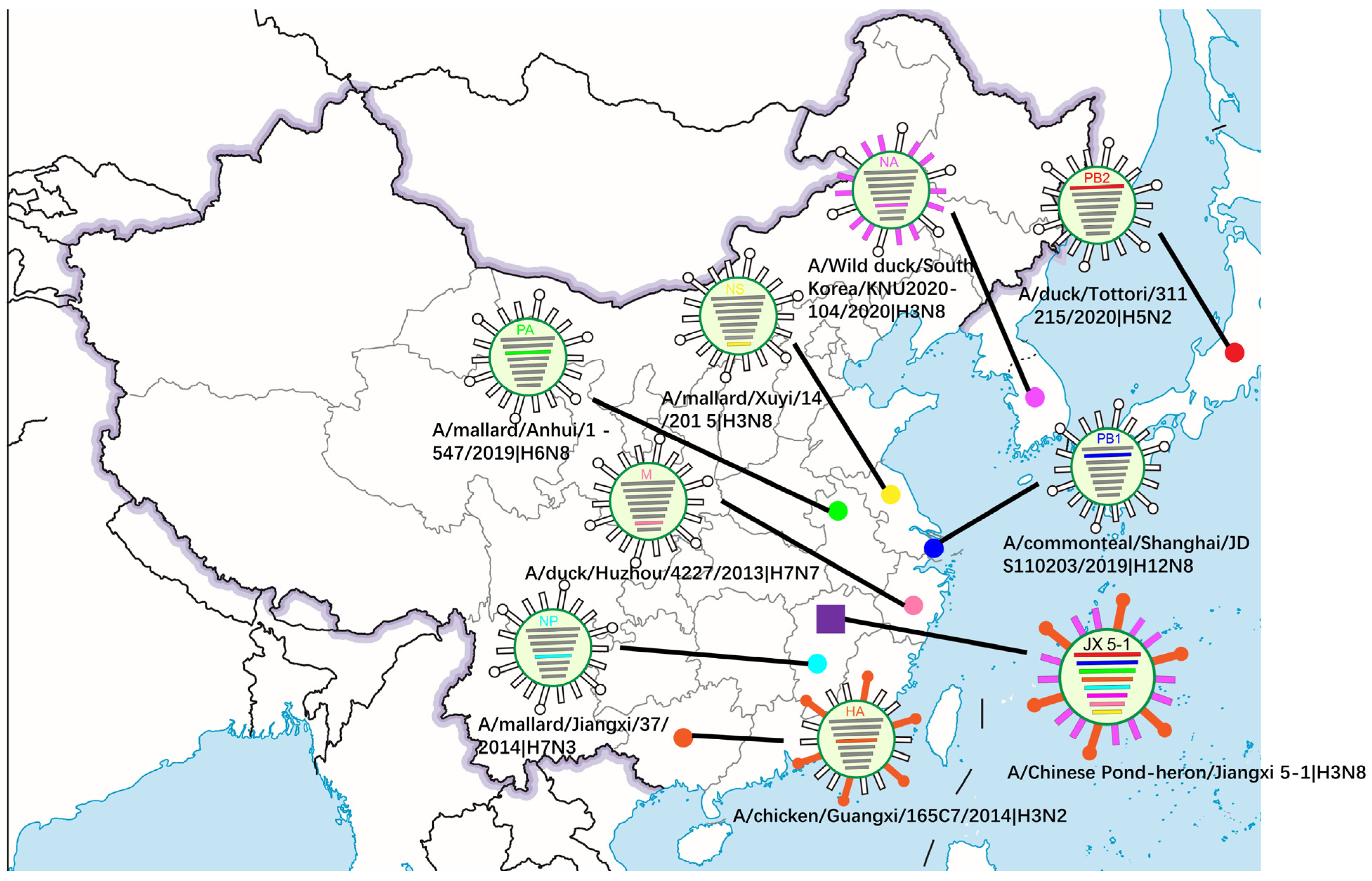
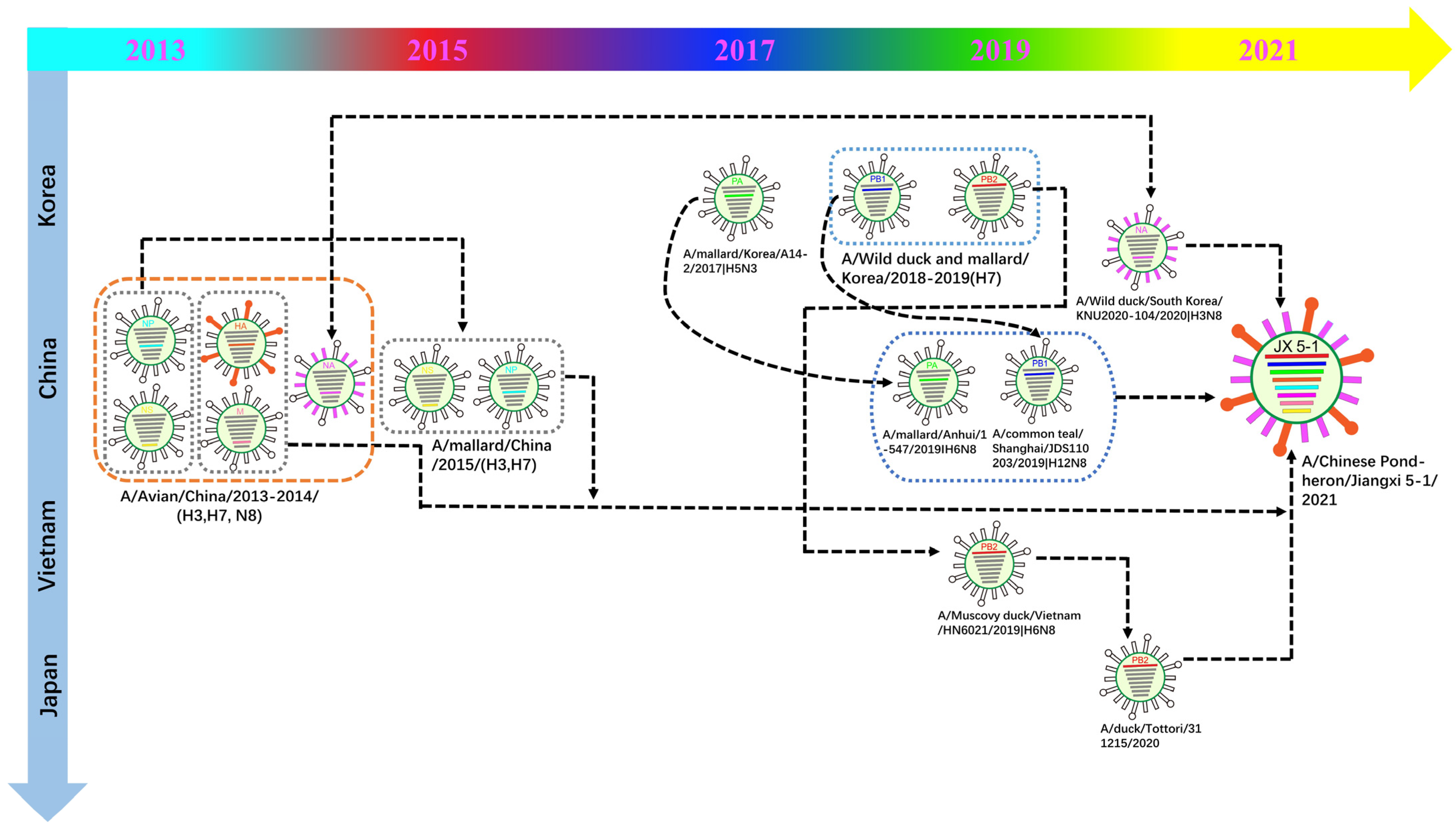

| Gene | Viruses with Greatest Homology | Accession | Identity (%) |
|---|---|---|---|
| PB2 | A/duck/Tottori/311215/2020 (H5N2) | LC656330.1 | 98.96% |
| PB1 | A/common teal/Shanghai/JDS110203/ 2019 (H12N8) | MN795765.1 | 99.43% |
| PA | A/wild goose/dongting lake/121/ 2018 (H6N2) | MH727479.1 | 98.68% |
| HA | A/chicken/Guangxi/165C7/2014 (H3N2) | KT022317.1 | 95.36% |
| NP | A/canine/Zhejiang/S34/2015 (H3N8) | MH018583.1 | 97.33% |
| NA | A/Wild duck/South Korea/KNU2020-104/2020 (H3N8) | OK236005.1 | 99.30% |
| M | A/duck/Huzhou/4227/2013 (H7N7) | KP413918.1 | 98.37% |
| NS | A/duck/China/F1473-2/2016 (H6N2) | MT828327.1 | 98.57% |
| Virus Strain | HA Receptor-Binding Residues (H3 Numbering) | NA | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Cleavage Sites | 135 | 138 | 160 | 186 | 192 | 226 | 228 | Stalk Region Deletion | |
| JX 5-1 | PEKQTR↓GLF | E | T | A | N | R | Q | T | No deletion |
| HN-410 | PEKQTR↓GLF | D | T | A | N | K | Q | T | No deletion |
| South Korea2020 (H3N8) | PEKQTR↓GLF | E | T | A | N | K | Q | T | No deletion |
| Zhejiang2013 (H3N8) | PEKQTR↓GLF | E | T | A | N | K | Q | T | No deletion |
| Xuyi2014 (H3N8) | PEKQTR↓GLF | E | T | D | N | K | Q | T | No deletion |
| Amur region2020 (H3N8) | PEKQTR↓GLF | E | T | A | N | K | Q | T | No deletion |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, H.; Han, S.; Wang, B.; Xing, Y.; Yuan, G.; Wang, Y.; Zhao, Z.; Li, G.; Li, Q.; Pan, J.; et al. Genetic Characterization and Pathogenesis of Avian Influenza Virus H3N8 Isolated from Chinese pond heron in China in 2021. Viruses 2023, 15, 383. https://doi.org/10.3390/v15020383
Zhang H, Han S, Wang B, Xing Y, Yuan G, Wang Y, Zhao Z, Li G, Li Q, Pan J, et al. Genetic Characterization and Pathogenesis of Avian Influenza Virus H3N8 Isolated from Chinese pond heron in China in 2021. Viruses. 2023; 15(2):383. https://doi.org/10.3390/v15020383
Chicago/Turabian StyleZhang, Heng, Shuyi Han, Bo Wang, Yanan Xing, Guohui Yuan, Ye Wang, Zhilei Zhao, Gaojian Li, Qiaoqiao Li, Jinchao Pan, and et al. 2023. "Genetic Characterization and Pathogenesis of Avian Influenza Virus H3N8 Isolated from Chinese pond heron in China in 2021" Viruses 15, no. 2: 383. https://doi.org/10.3390/v15020383
APA StyleZhang, H., Han, S., Wang, B., Xing, Y., Yuan, G., Wang, Y., Zhao, Z., Li, G., Li, Q., Pan, J., Li, W., & He, H. (2023). Genetic Characterization and Pathogenesis of Avian Influenza Virus H3N8 Isolated from Chinese pond heron in China in 2021. Viruses, 15(2), 383. https://doi.org/10.3390/v15020383







