Spike Gene Analysis and Prevalence of Porcine Epidemic Diarrhea Virus from Pigs in South Korea: 2013–2022
Abstract
:1. Introduction
2. Materials and Methods
2.1. Detection of PEDV-Positive Samples from Suckling Piglets and Sows
2.2. RT-PCR for PEDV
2.3. Phylogenetic Analysis of PEDV
2.4. Changes in Population Size, Substitution Rates, and Recombination of PEDV
2.5. Serum Neutralization Antibody Tests
2.6. Statistical Analysis
3. Results
3.1. PEDV Incidence over 10 Years
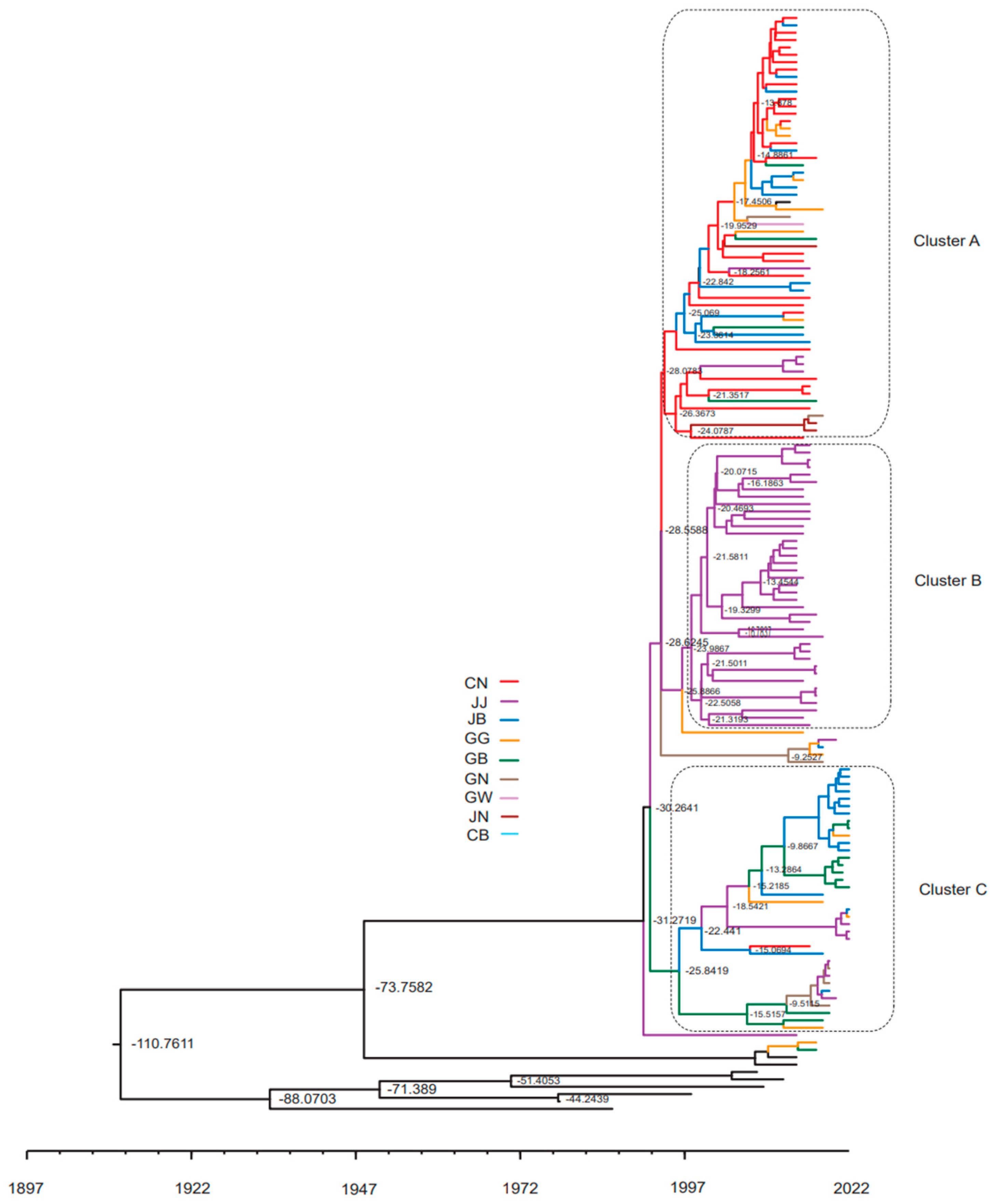
3.2. ML Trees for the Complete S Gene Sequences
3.3. Changes in the Genotype of South Korean PEDVs by Year
3.4. Nucleotide Deletions and Insertion in the S Gene
3.5. PEDV Antibody Titers by Year
3.6. Genotype of PEDV According to Region
3.7. Substitution Rate and Recombination of Spike Gene
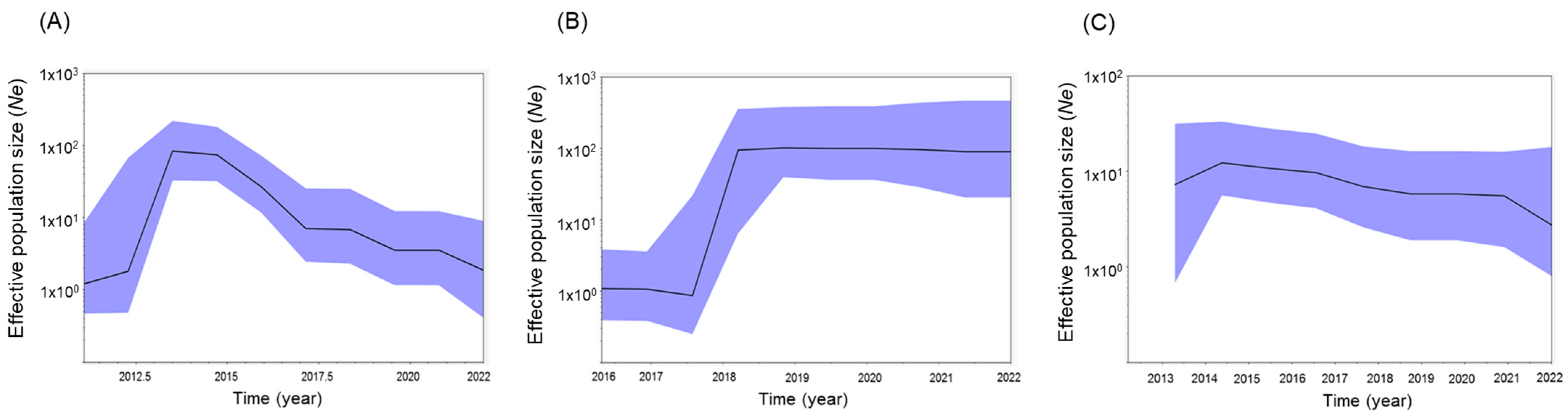
3.8. Effective Population Size by Region
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pensaert, M.B.; de Bouck, P. A new coronavirus-like particle associated with diarrhea in swine. Arch. Virol. 1978, 58, 243–247. [Google Scholar] [CrossRef]
- Kocherhans, R.; Bridgen, A.; Ackermann, M.; Tobler, K. Completion of the porcine epidemic diarrhoea coronavirus (PEDV) genome sequence. Virus Genes 2001, 23, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.W.; Dickerman, A.W.; Pineyro, P.; Li, L.; Fang, L.; Kiehne, R.; Opriessnig, T.; Meng, X.J. Origin, evolution, and genotyping of emergent porcine epidemic diarrhea virus strains in the United States. mBio 2013, 4, e00737-13. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Park, B. Porcine epidemic diarrhoea virus: A comprehensive review of molecular epidemiology, diagnosis, and vaccines. Virus Genes 2012, 44, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Li, Z.; Zou, Y.; Wicht, O.; Kuppeveld, F.J.M.V.; Rottier, P.J.M.; Bosch, B.J. Manipulation of the Porcine Epidemic Diarrhea Virus Genome Using Targeted RNA Recombination. PLoS ONE 2013, 8, e69997. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Tortorici, M.A.; Bosch, B.J.; Frenz, B.; Rottier, P.J.M.; DiMaio, F.; Rey, F.A.; Veesler, D. Cryo-electron microscopy structure of a coronavirus spike glycoprotein trimer. Nature 2016, 531, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Li, B.X.; Ge, J.W.; Li, Y.J. Porcine aminopeptidase N is a functional receptor for the PEDV coronavirus. Virology 2007, 365, 166–172. [Google Scholar] [CrossRef]
- Liu, C.; Tang, J.; Ma, Y.; Liang, X.; Yang, Y.; Peng, G.; Qi, Q.; Jiang, S.; Li, J.; Du, L.; et al. Receptor usage and cell entry of porcine epidemic diarrhea coronavirus. J. Virol. 2015, 89, 6121–6125. [Google Scholar] [CrossRef]
- Li, F. Structure, Function, and Evolution of Coronavirus Spike Proteins. Annu. Rev. Virol. 2016, 3, 237–261. [Google Scholar] [CrossRef]
- Hulswit, R.J.; de Haan, C.A.; Bosch, B.J. Coronavirus Spike Protein and Tropism Changes. Adv. Virus Res. 2016, 96, 29–57. [Google Scholar]
- Sun, R.Q.; Cai, R.J.; Chen, Y.Q.; Liang, P.S.; Chen, D.K.; Song, C.X. Outbreak of porcine epidemic diarrhea in suckling piglets, China. Emerg. Infect. Dis. 2012, 18, 161–163. [Google Scholar] [CrossRef]
- Chen, Q.; Gauger, P.C.; Stafne, M.R.; Thomas, J.T.; Madson, D.M.; Huang, H.; Zheng, Y.; Li, G.; Zhang, J. Pathogenesis comparison between the United States porcine epidemic diarrhoea virus prototype and S-INDEL-variant strains in conventional neonatal piglets. J. Gen. Virol. 2016, 97, 1107–1121. [Google Scholar] [CrossRef] [PubMed]
- Wood, E.N. An apparently new syndrome of porcine epidemic diarrhoea. Vet. Rec. 1977, 100, 243–244. [Google Scholar] [CrossRef] [PubMed]
- Kusanagi, K.; Kuwahara, H.; Katoh, T.; Nunoya, T.; Ishikawa, Y.; Samejima, T.; Tajima, M. Isolation and serial propagation of porcine epidemic diarrhea virus in cell cultures and partial characterization of the isolate. J. Vet. Med. Sci. 1992, 54, 313–318. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Okada, K.; Ohshima, K. An outbreak of swine diarrhea of a new-type associated with coronavirus-like particles in Japan. Jpn. J. Vet. Sci. 1983, 45, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Yang, J.; Zixiang Zhu, Z.; Zheng, H. Porcine Epidemic Diarrhea Virus and the Host Innate Immune Response. Pathogens 2020, 9, 367. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, G.W.; Hoang, H.; Schwartz, K.J.; Burrough, E.R.; Sun, D.; Madson, D.; Cooper, V.L.; Pillatzki, A.; Gauger, P.; Schmitt, B.J.; et al. Emergence of Porcine epidemic diarrhea virus in the United States: Clinical signs, lesions, and viral genomic sequences. J. Vet. Diagn. Investig. 2013, 25, 649–654. [Google Scholar] [CrossRef]
- Li, W.; Li, H.; Liu, Y.; Pan, Y.; Deng, F.; Song, Y.; Tang, X.; He, Q. New variants of porcine epidemic diarrhea virus, China, 2011. Emerg. Infect. Dis. 2012, 18, 1350–1353. [Google Scholar] [CrossRef]
- Sun, M.; Ma, J.; Wang, Y.; Wang, M.; Song, W.; Zhang, W.; Lu, C.; Yao, H. Genomic and epidemiological characteristics provide new insights into the phylogeographical and spatiotemporal spread of porcine epidemic diarrhea virus in Asia. J. Clin. Microbiol. 2015, 53, 1484–1492. [Google Scholar] [CrossRef]
- Lee, S.; Lee, C. Outbreak-related porcine epidemic diarrhea virus strains similar to US strains, South Korea, 2013. Emerg. Infect. Dis. 2014, 20, 1223–1226. [Google Scholar] [CrossRef]
- Cho, Y.Y.; Lim, S.I.; Kim, Y.K.; Song, J.Y.; Lee, J.B.; An, D.J. Complete Genome Sequence of K14JB01, a Novel Variant Strain of Porcine Epidemic Diarrhea Virus in South Korea. Genome Announc. 2014, 2, e00505-14. [Google Scholar] [CrossRef] [PubMed]
- Jang, G.; Lee, D.; Shin, S.; Lim, J.; Won, H.; Eo, Y.; Kim, C.H.; Lee, C. Porcine epidemic diarrhea virus: An update overview of virus epidemiology, vaccines, and control strategies in South Korea. J. Vet. Sci. 2023, 24, e58. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Linda, J.; Saif, J.; Wang, Q. Porcine epidemic diarrhea virus (PEDV): An update on etiology, transmission, pathogenesis, and prevention and control. Virus Res. 2020, 286, 198045. [Google Scholar] [CrossRef]
- Lee, S.; Lee, C. Complete genome sequence of a novel S-insertion variant of porcine epidemic diarrhea virus from South Korea. Arch. Virol. 2017, 162, 2919–2922. [Google Scholar] [CrossRef] [PubMed]
- Jang, G.; Park, J.; Lee, C. Complete genome sequences of novel S-deletion variants of porcine epidemic diarrhea virus identified from a recurrent outbreak on Jeju Island, South Korea. Arch. Virol. 2019, 164, 2621–2625. [Google Scholar] [CrossRef]
- Park, J.; Lee, C. Emergence and evolution of novel G2b-like porcine epidemic diarrhea virus inter-subgroup G1b recombinants. Arch. Virol. 2020, 165, 2471–2478. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Lee, J.M.; Jung, J.; Kim, I.J.; Hyun, B.H.; Kim, H.I.; Park, C.K.; Oem, J.K.; Kim, Y.H.; Lee, M.H.; et al. Genetic characterization of porcine epidemic diarrhoea virus in Korea from 1998 to 2013. Arch. Virol. 2015, 160, 1055–1064. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Bouckaert, R.; Heledl, J.; Kühnert, D.; Vaughan, T.; Wu, C.H.; Xie, D.; Suchard, M.A.; Rambaut, A.; Drummond, A.J. BEAST 2: A software platform for Bayesian evolutionary analysis. PLoS Comput. Biol. 2014, 10, e1003537. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A. Tracer, 1.6; University of Edinburgh: Edinburgh, Scotland, 2013.
- Rambaut, A.; Drummond, A. TreeAnnotator, v1.7.5; University of Edinburgh: Edinburgh, Scotland, 2013.
- Rambaut, A. FigTree Version 1.4.0. 2012. Available online: http://tree.bio.ed.ac.uk/software/figtree (accessed on 6 May 2023).
- Drummond, A.J.; Rambaut, A.; Shapiro, B.; Pybus, O.G. Bayesian coalescent inference of past population dynamics from molecular sequences. Mol. Biol. Evol. 2005, 22, 1185–1192. [Google Scholar] [CrossRef]
- Martin, D.P.; Murrell, B.; Golden, M.; Khoosal, A.; Muhire, B. RDP4: Detection and analysis of recombination patterns in virus genomes. Virus Evol. 2015, 1, vev003. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.N.; Chung, W.B.; Chang, S.W.; Wen, C.C.; Liu, H.; Chien, C.H.; Chiou, M.T. US-like strain of porcine epidemic diarrhoea virus outbreaks in Taiwan, 2013–2014. J. Vet. Med. Sci. 2014, 76, 1297–1299. [Google Scholar] [CrossRef] [PubMed]
- Vlasova, A.N.; Marthaler, D.; Wang, Q.; Culhane, M.R.; Rossow, K.D.; Rovira, A.; Collins, J.; Saif, L.J. Distinct characteristics and complex evolution of PEDV strains, North America, May 2013–February 2014. Emerg. Infect. Dis. 2014, 20, 1620–1628. [Google Scholar] [CrossRef] [PubMed]
- Cheun-Arom, T.; Temeeyasen, G.; Srijangwad, A.; Tripipat, T.; Sangmalee, S.; Vui, D.T.; Chuanasa, T.; Tantituvanont, A.; Nilubol, D. Complete genome sequences of two genetically distinct variants of porcine epidemic diarrhoea virus in the eastern region of Thailand. Genome Announc. 2015, 3, e00634-15. [Google Scholar] [CrossRef]
- Diep, N.V.; Norimine, J.; Sueyoshi, M.; Lan, N.T.; Hirai, T.; Yamaguchi, R. US-like isolates of porcine epidemic diarrhoea virus from Japanese outbreaks between 2013 and 2014. Springerplus 2015, 4, 756. [Google Scholar] [CrossRef]
- Kim, Y.K.; Cho, Y.Y.; An, B.H.; Lim, S.I.; Lim, J.A.; Cho, I.S.; Le, V.P.; An, D.J. Molecular characterization of the spike and ORF3 genes of porcine epidemic diarrhoea virus in the Philippines. Arch. Virol. 2016, 161, 1323–1328. [Google Scholar] [CrossRef]
- Stadler, J.; Zoels, S.; Fux, R.; Hanke, D.; Pohlmann, A.; Blome, S.; Weissenböck, H.; Weissenbacher-Lang, C.; Ritzmann, M.; Ladinig, A. Emergence of porcine epidemic diarrhea virus in southern Germany. BMC Vet. Res. 2015, 11, 142. [Google Scholar] [CrossRef]
- Theuns, S.; Conceic¸ão-Neto, N.; Christiaens, I.; Zeller, M.; Desmarets, L.M.; Roukaerts, I.D.; Acar, D.D.; Heylen, E.; Matthijnssens, J.; Nauwynck, H.J. Complete genome sequence of a porcine epidemic diarrhea virus from a novel outbreak in Belgium, January 2015. Genome Announc. 2015, 3, e00506-15. [Google Scholar] [CrossRef]
- Grasland, B.; Bigault, L.; Bernard, C.; Quenault, H.; Toulouse, O.; Fablet, C.; Rose, N.; Touzain, F.; Blanchard, Y. Complete genome sequence of a porcine epidemic diarrhea S gene indel strain isolated in France in December 2014. Genome Announc. 2015, 3, e00535-15. [Google Scholar] [CrossRef]
- Bartolo, I.D.; Tofani, S.; Angeloni, G.; Ponterio, E.; Ostanello, F.; Ruggeri, F.M. Detection and characterization of porcine caliciviruses in Italy. Arch. Virol. 2014, 159, 2479–2484. [Google Scholar] [CrossRef]
- Bertasio, C.; Giacomini, E.; Lazzaro, M.; Perulli, S.; Papetti, A.; Lavazza, A.; Lelli, D.; Alborali, G.; Boniotti, M.B. Porcine epidemic diarrhea virus shedding and antibody response in swine farms: A longitudinal study. Front. Microbiol. 2016, 7, 2009. [Google Scholar] [CrossRef] [PubMed]
- Steinrigl, A.; Ferna´ndez, S.R.; Stoiber, F.; Pikalo, J.; Sattler, T.; Schmoll, F. First detection, clinical presentation and phylogenetic characterization of Porcine epidemic diarrhea virus in Austria. BMC Vet. Res. 2015, 11, 310. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, J.R.; Hakze-van der Honing, R.; Almeida, A.; Lourenco, M.; van der Poel, W.H.M.; Nascimento, M.S.J. Outbreak of Porcine Epidemic Diarrhea Virus in Portugal, 2015. Transbound. Emerg. Dis. 2015, 62, 586–588. [Google Scholar] [CrossRef] [PubMed]
- Toplak, I.; Ipavec, M.; Kuhar, U.; Kušar, D.; Papić, B.P.; Koren, S.; Toplak, N. Complete Genome Sequence of the Porcine Epidemic Diarrhea Virus Strain SLO/JH-11/2015. Genome Announc. 2016, 4, e01725-15. [Google Scholar] [CrossRef]
- Valkó, A.; Biksi, I.; Cságola, A.; Tuboly, T.; Kiss, K.; Ursu, K.; Dán, Á. Porcine epidemic diarrhoea virus with a recombinant S gene detected in Hungary, 2016. Acta Vet. Hung. 2017, 65, 253–261. [Google Scholar] [CrossRef]
- Dastjerdi, A.; Carr, J.; Ellis, R.J.; Steinbach, F.; Williamson, S. Porcine epidemic diarrhea virus among farmed pigs, Ukraine. Emerg. Infect. Dis. 2015, 21, 2235–2237. [Google Scholar] [CrossRef]
- Wang, L.; Byrum, B.; Zhang, Y. New variant of porcine epidemic diarrhea virus, United States, 2014. Emerg. Infect. Dis. 2014, 20, 917–919. [Google Scholar] [CrossRef]
- Park, S.; Kim, S.; Song, D.; Park, B. Novel Porcine Epidemic Diarrhea Virus Variant with Large Genomic Deletion, South Korea. Emerg. Infect. Dis. 2014, 20, 2089–2092. [Google Scholar] [CrossRef]
- Masuda, T.; Murakami, S.; Takahashi, O.; Miyazaki, A.; Ohashi, S.; Yamasato, H.; Suzuki, T. New porcine epidemic diarrhoea virus variant with a large deletion in the spike gene identified in domestic pigs. Arch. Virol. 2015, 160, 2565–2568. [Google Scholar] [CrossRef]
- Tortorici, M.A.; Veesler, D. Structural insights into coronavirus entry. Adv. Virus Res. 2019, 105, 93–116. [Google Scholar]
- Sato, T.; Oroku, K.; Ohshima, Y.; Furuya, Y.; Sasakawa, C. Efficacy of genogroup 1 based porcine epidemic diarrhea live vaccine against genogroup 2 field strain in Japan. Virol. J. 2018, 15, 28. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Moon, H.; Kang, B. Porcine epidemic diarrhea: A review of current epidemiology and available vaccines. Clin. Exp. Vaccine Res. 2015, 4, 166. [Google Scholar] [CrossRef] [PubMed]
- Lara-Romero, R.; Gómez-Núñez, L.; Cerriteño-Sánchez, J.L.; Márquez-Valdelamar, L.; Mendoza-Elvira, S.; Ramírez-Mendoza, H.; Rivera-Benítez, J.F. Molecular characterization of the spike gene of the porcine epidemic diarrhea virus in Mexico, 2013–2016. Virus Genes 2018, 54, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.H.; Bae, J.L.; Kang, T.J.; Kim, J.; Chung, G.H.; Lim, C.W.; Laude, H.; Yang, M.S.; Jang, Y.S. Identification of the epitope region capable of inducing neutralizing antibodies against the porcine epidemic diarrhea virus. Mol. Cells 2002, 14, 295–299. [Google Scholar]
- Cruz, D.J.M.; Kim, C.J.; Shin, H.J. The GPRLQPY motif located at the carboxy-terminal of the spike protein induces antibodies that neutralize Porcine epidemic diarrhea virus. Virus Res. 2008, 132, 192–196. [Google Scholar] [CrossRef]
- Sun, D.; Feng, L.; Shi, H.; Chen, J.; Cui, X.; Chen, H.; Liu, S.; Tong, Y.; Wang, Y.; Tong, G. Identification of two novel B cell epitopes on porcine epidemic diarrhea virus spike protein. Vet. Microbiol. 2008, 131, 73–81. [Google Scholar] [CrossRef]
- Jang, J.; Yoon, S.H.; Lee, W.; Yu, J.; Yoon, J.; Shim, S.; Kim, H. Time-calibrated phylogenomics of the porcine epidemic diarrhea virus: Genome-wide insights into the spatio-temporal dynamics. Genes Genom. 2018, 40, 825–834. [Google Scholar] [CrossRef]
- Chung, H.C.; Lee, J.H.; Nguyen, V.G.; Huynh, T.M.L.; Lee, G.E.; Moon, H.J.; Park, S.J.; Kim, H.K.; Park, B.K. New emergence pattern with variant porcine epidemic diarrhea viruses, South Korea, 2012-2015. Virus Res. 2016, 226, 14–19. [Google Scholar] [CrossRef]
- Duffy, S.; Shackelton, L.A.; Holmes, E.C. Rates of evolutionary change in viruses: Patterns and determinants. Nat. Rev. Genet. 2008, 9, 267–276. [Google Scholar] [CrossRef]
- Elena, S.F.; Sanjuan, R. Adaptive value of high mutation rates of RNA viruses: Separating causes from consequences. J. Virol. 2005, 79, 11555–11558. [Google Scholar] [CrossRef]
- Lauring, A.S.; Andino, R. Quasispecies theory and the behavior of RNA viruses. PLoS Pathog. 2010, 6, e1001005. [Google Scholar] [CrossRef] [PubMed]
- Chattha, K.S.; Roth, J.A.; Saif, L.J. Strategies for design and application of enteric viral vaccines. Annu. Rev. Anim. Biosci. 2015, 3, 375–395. [Google Scholar] [CrossRef] [PubMed]
- de Arriba, M.L.; Carvajal, A.; Pozo, J.; Rubio, P. Mucosal and systemic isotype-specific antibody responses and protection in conventional pigs exposed to virulent or attenuated porcine epidemic diarrhoea virus. Vet. Immunol. Immunopathol. 2002, 85, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Langel, S.N.; Paim, F.C.; Alhamo, M.A.; Buckley, A.; Van Geelen, A.; Lager, K.M.; Vlasova, A.N.; Saif, L.J. Stage of Gestation at Porcine Epidemic Diarrhea Virus Infection of Pregnant Swine Impacts Maternal Immunity and Lactogenic Immune Protection of Neonatal Suckling Piglets. Front. Immunol. 2019, 10, 727. [Google Scholar] [CrossRef]
- He, W.T.; Bollen, N.; Xu, Y.; Zhao, J.; Dellicour, S.; Yan, Z.; Gong, W.; Zhang, C.; Zhang, L.; Lu, M.; et al. Phylogeography Reveals Association between Swine Trade and the Spread of Porcine Epidemic Diarrhea Virus in China and across the World. Mol. Biol. Evol. 2022, 39, msab364. [Google Scholar] [CrossRef]
- Lowe, J.; Gauger, P.; Harmon, K.; Zhang, J.; Connor, J.; Yeske, P.; Loula, T.; Levis, I.; Dufresne, L.; Main, R. Role of transportation in spread of porcine epidemic diarrhea virus infection, United States. Emerg. Infect. Dis. 2014, 20, 872–874. [Google Scholar] [CrossRef]
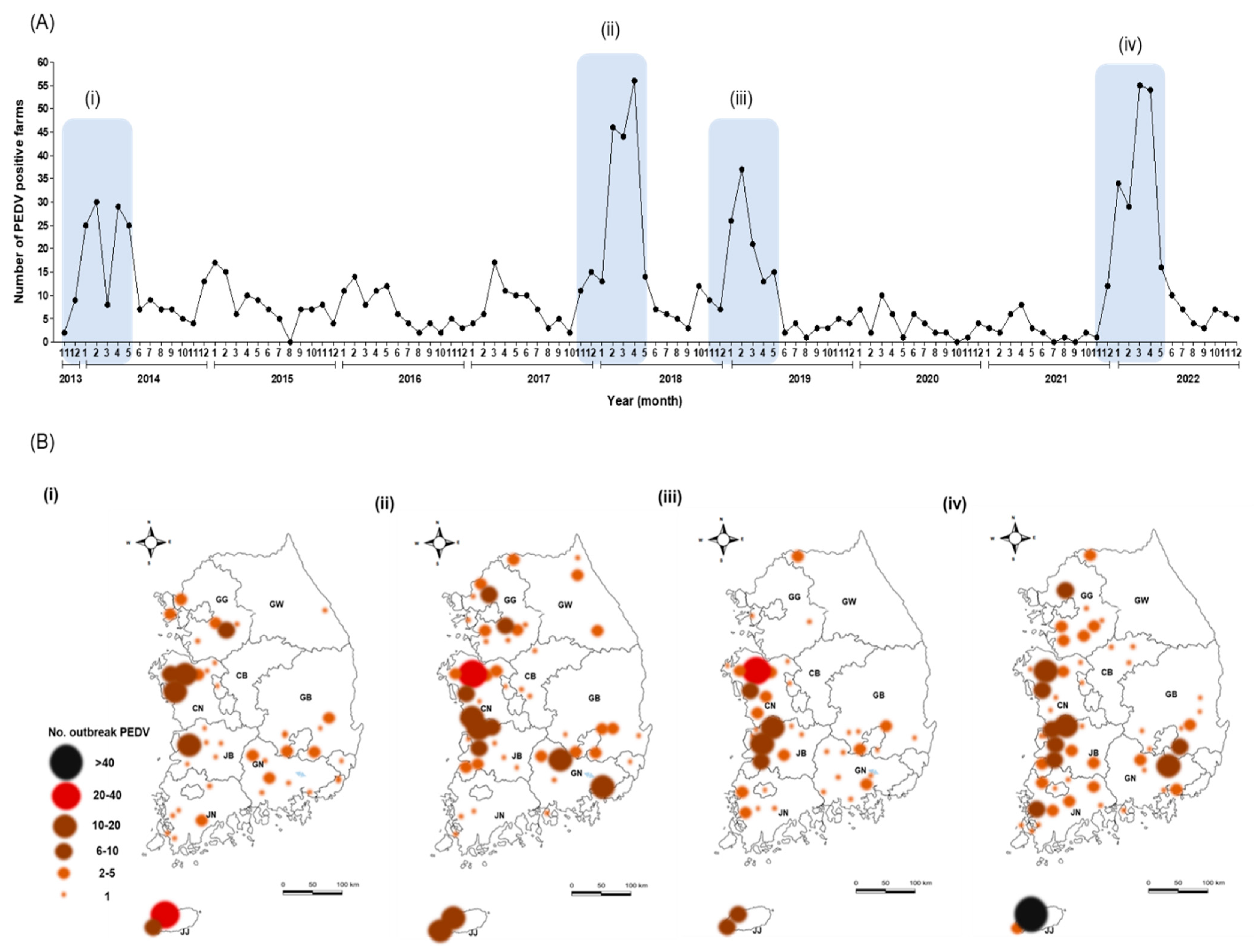
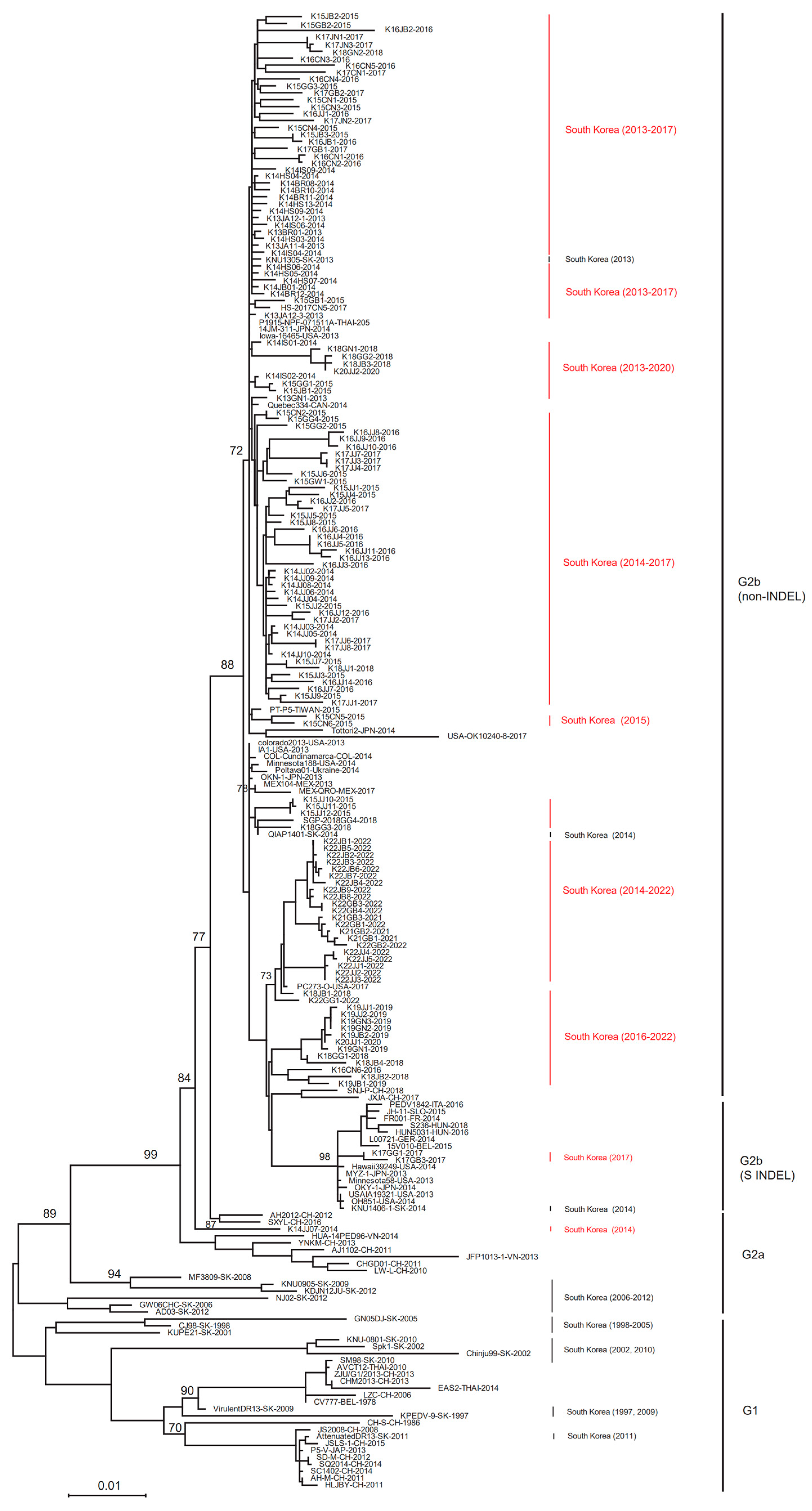
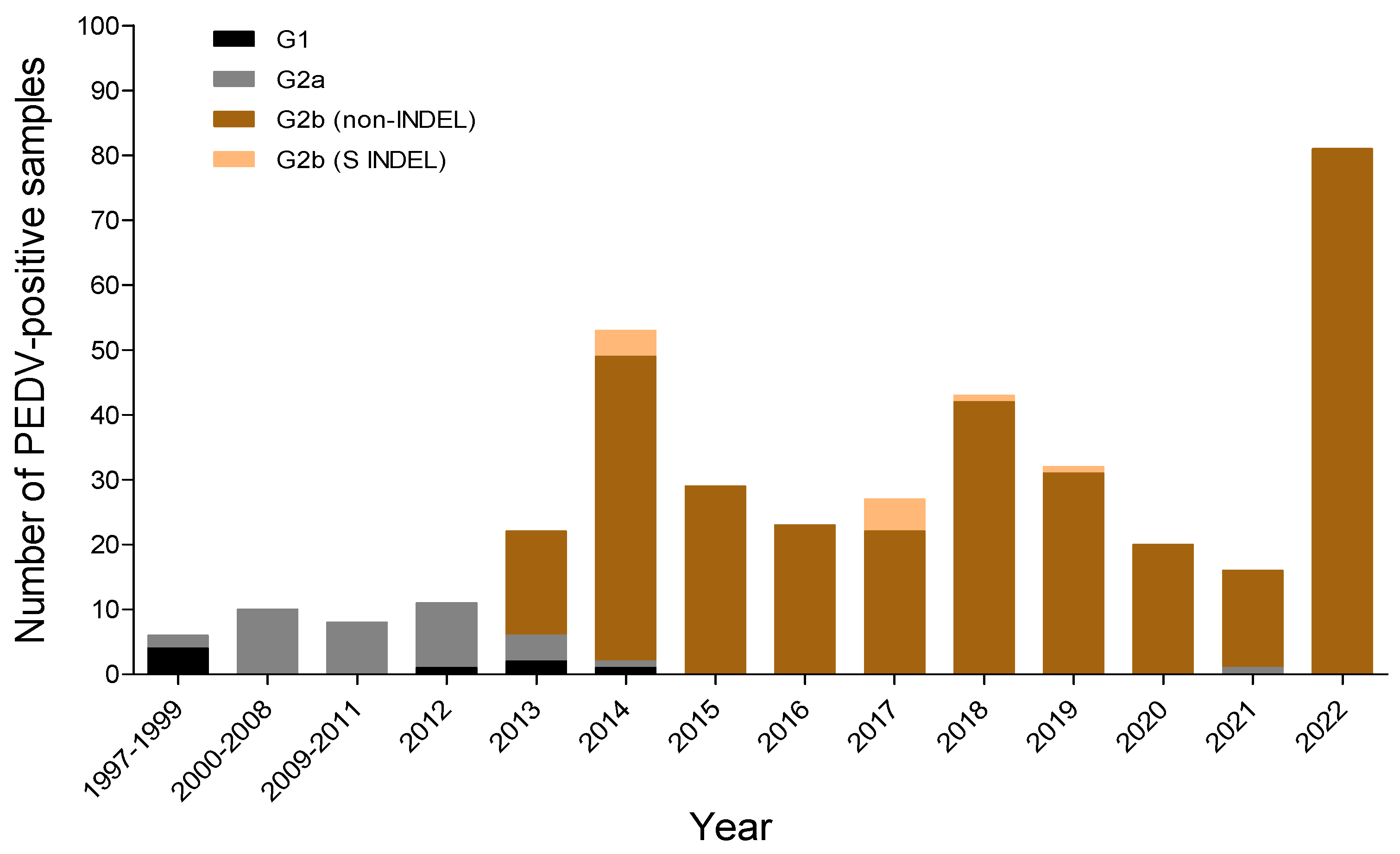
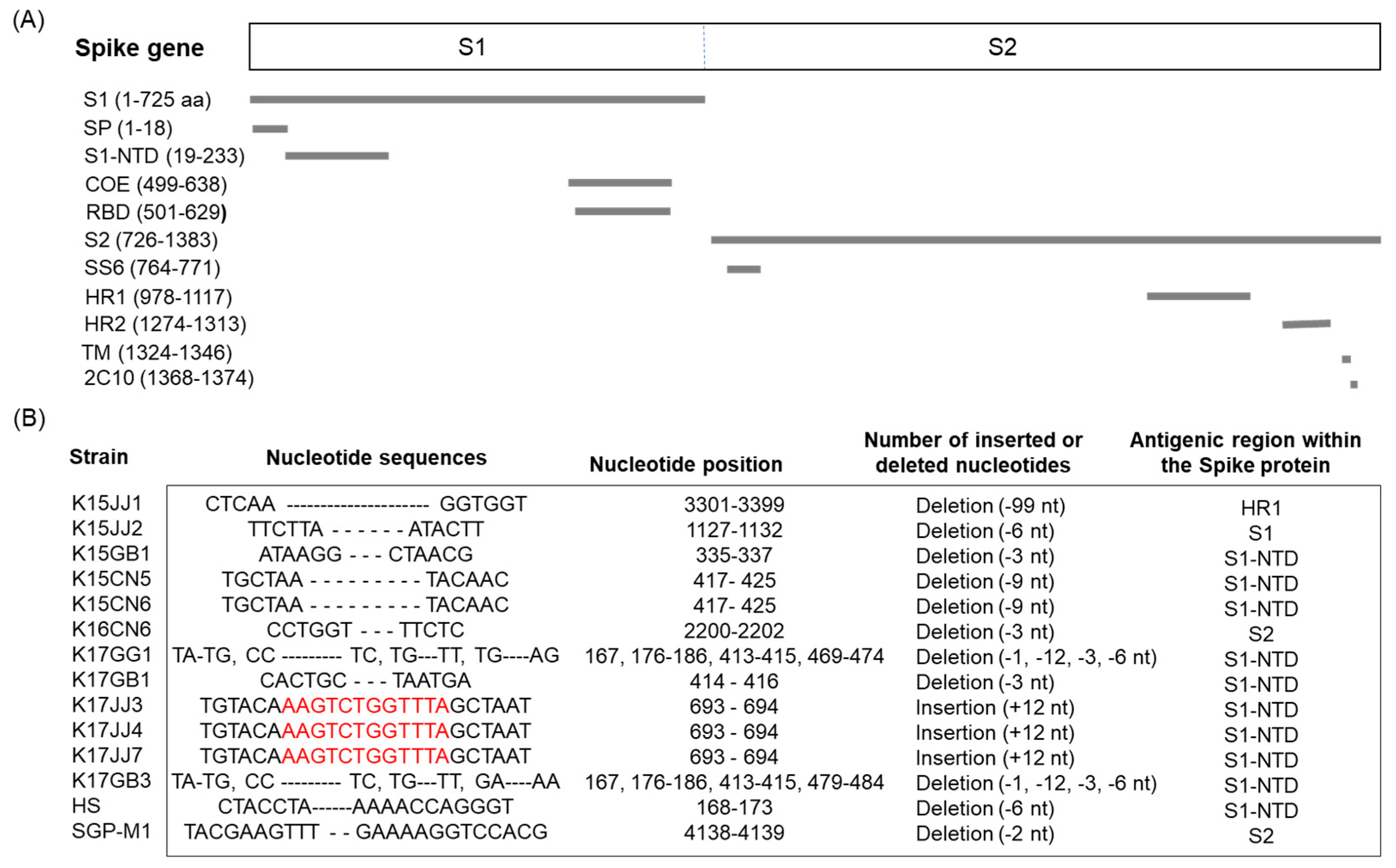
| Year | No. PED Outbreaks on Pig Farms * (No. of PEDV Samples Collected in This Study) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| GG ** | GW | CN | CB | GN | GB | JN | JB | JJ | Total | |
| 2013 | 2 (2) | 0 | 5 (2) | 0 | 3 (1) | 2 | 0 | 0 | 0 | 12 (5) |
| 2014 | 22 | 3 | 47 (11) | 0 | 12 | 13 | 8 | 21 (6) | 43 (9) | 169 (26) |
| 2015 | 11 (4) | 2 (1) | 15 (6) | 0 | 3 | 6 (2) | 4 | 6 (3) | 48 (12) | 95 (28) |
| 2016 | 12 | 0 | 11 (6) | 0 | 5 | 5 | 1 | 7 (2) | 41 (14) | 82 (22) |
| 2017 | 11 (1) | 1 | 13 (2) | 0 | 11 | 10 (3) | 6 (1) | 2 | 47 (10) | 101 (17) |
| 2018 | 29 (4) | 10 | 62 | 4 | 22 (2) | 17 | 3 | 41 (4) | 34 (1) | 222 (11) |
| 2019 | 1 | 4 | 42 | 2 | 9 (3) | 13 | 9 | 44 (2) | 10 (2) | 134 (7) |
| 2020 | 0 | 0 | 5 | 1 | 1 | 7 | 10 | 10 | 11 (2) | 45 (2) |
| 2021 | 6 | 0 | 0 | 1 | 5 | 11 (3) | 2 | 4 | 11 | 40 (3) |
| 2022 | 16 (1) | 2 | 36 | 3 | 16 | 14 (4) | 24 | 47 (9) | 73 (5) | 235 (19) |
| Total | 110 (12) | 22 (1) | 236 (27) | 11 | 87 (6) | 98 (12) | 67 (1) | 182 (26) | 318 (55) | 1131 (140) |
| Year | 2016 | 2017 | 2018 | 2019 | 2020 | 2021 | 2022 | Total |
|---|---|---|---|---|---|---|---|---|
| No. of pig farms | 566 | 405 | 443 | 341 | 502 | 415 | 312 | 2984 |
| No. of sows | 1713 | 1215 | 1329 | 1023 | 1506 | 1245 | 938 | 8969 |
| SN antibodies a | 1.39 ± 0.40 | 4.82 ± 1.54 | 3.94 ± 1.15 | 4.96 ± 1.47 | 4.89 ± 1.44 | 2.66 ± 0.78 | 3.77 ± 1.09 | 3.78 ± 1.12 |
| PEDV Genotype | Region | Substitution Rate (Substitutions/Site/Year) | ||
|---|---|---|---|---|
| Mean | 95% HPD Lower | 95% HPD Upper | ||
| G2b | SK * | 1.2556 × 10−3 | 8.6827 × 10−4 | 1.5997 × 10−3 |
| CN | 1.5952 × 10−3 | 1.2681 × 10−3 | 1.9121 × 10−3 | |
| JB | 1.8065 × 10−3 | 1.4644 × 10−3 | 2.1807 × 10−3 | |
| JJ | 1.5113 × 10−3 | 1.1315 × 10−3 | 1.9121 × 10−3 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, G.-N.; Song, S.; Choe, S.; Shin, J.; An, B.-H.; Kim, S.-Y.; Hyun, B.-H.; An, D.-J. Spike Gene Analysis and Prevalence of Porcine Epidemic Diarrhea Virus from Pigs in South Korea: 2013–2022. Viruses 2023, 15, 2165. https://doi.org/10.3390/v15112165
Park G-N, Song S, Choe S, Shin J, An B-H, Kim S-Y, Hyun B-H, An D-J. Spike Gene Analysis and Prevalence of Porcine Epidemic Diarrhea Virus from Pigs in South Korea: 2013–2022. Viruses. 2023; 15(11):2165. https://doi.org/10.3390/v15112165
Chicago/Turabian StylePark, Gyu-Nam, Sok Song, SeEun Choe, Jihye Shin, Byung-Hyun An, Song-Yi Kim, Bang-Hun Hyun, and Dong-Jun An. 2023. "Spike Gene Analysis and Prevalence of Porcine Epidemic Diarrhea Virus from Pigs in South Korea: 2013–2022" Viruses 15, no. 11: 2165. https://doi.org/10.3390/v15112165
APA StylePark, G.-N., Song, S., Choe, S., Shin, J., An, B.-H., Kim, S.-Y., Hyun, B.-H., & An, D.-J. (2023). Spike Gene Analysis and Prevalence of Porcine Epidemic Diarrhea Virus from Pigs in South Korea: 2013–2022. Viruses, 15(11), 2165. https://doi.org/10.3390/v15112165







