Phage Diving: An Exploration of the Carcharhinid Shark Epidermal Virome
Abstract
:1. Introduction
2. Materials and Methods
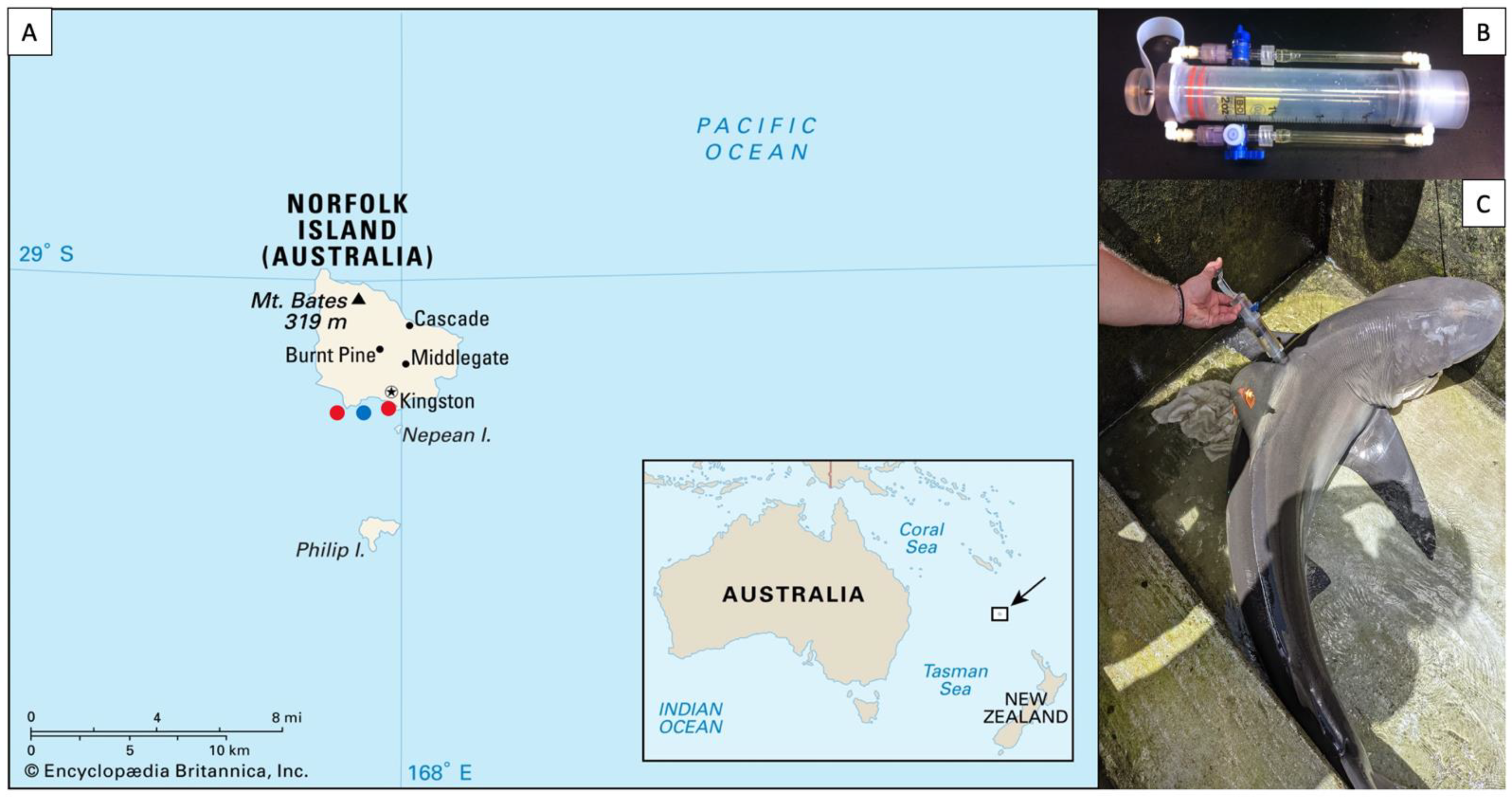
2.1. Sample Collection
2.2. Virome Processing and Sequencing
2.3. Bioinformatics and Statistical Analysis
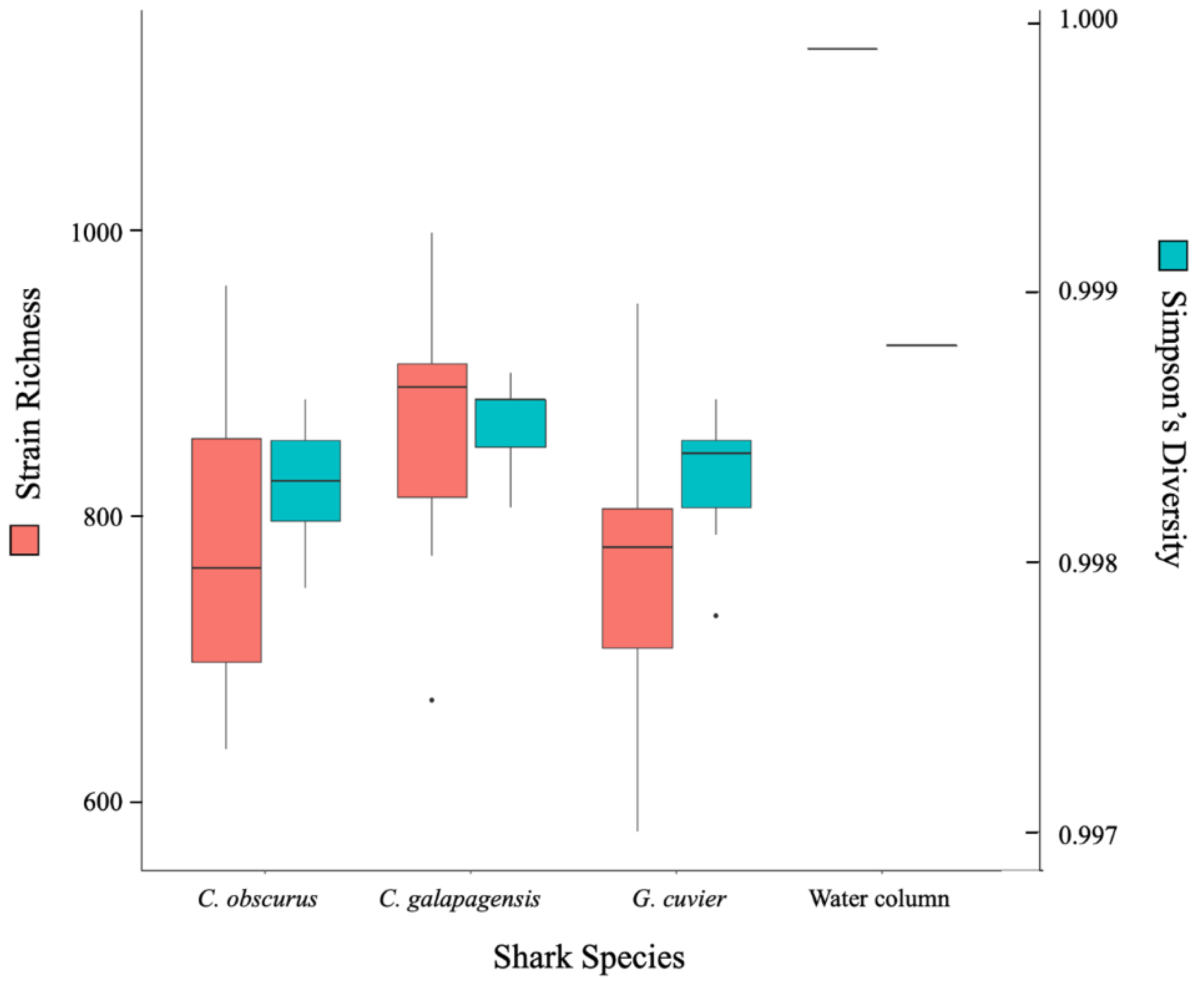
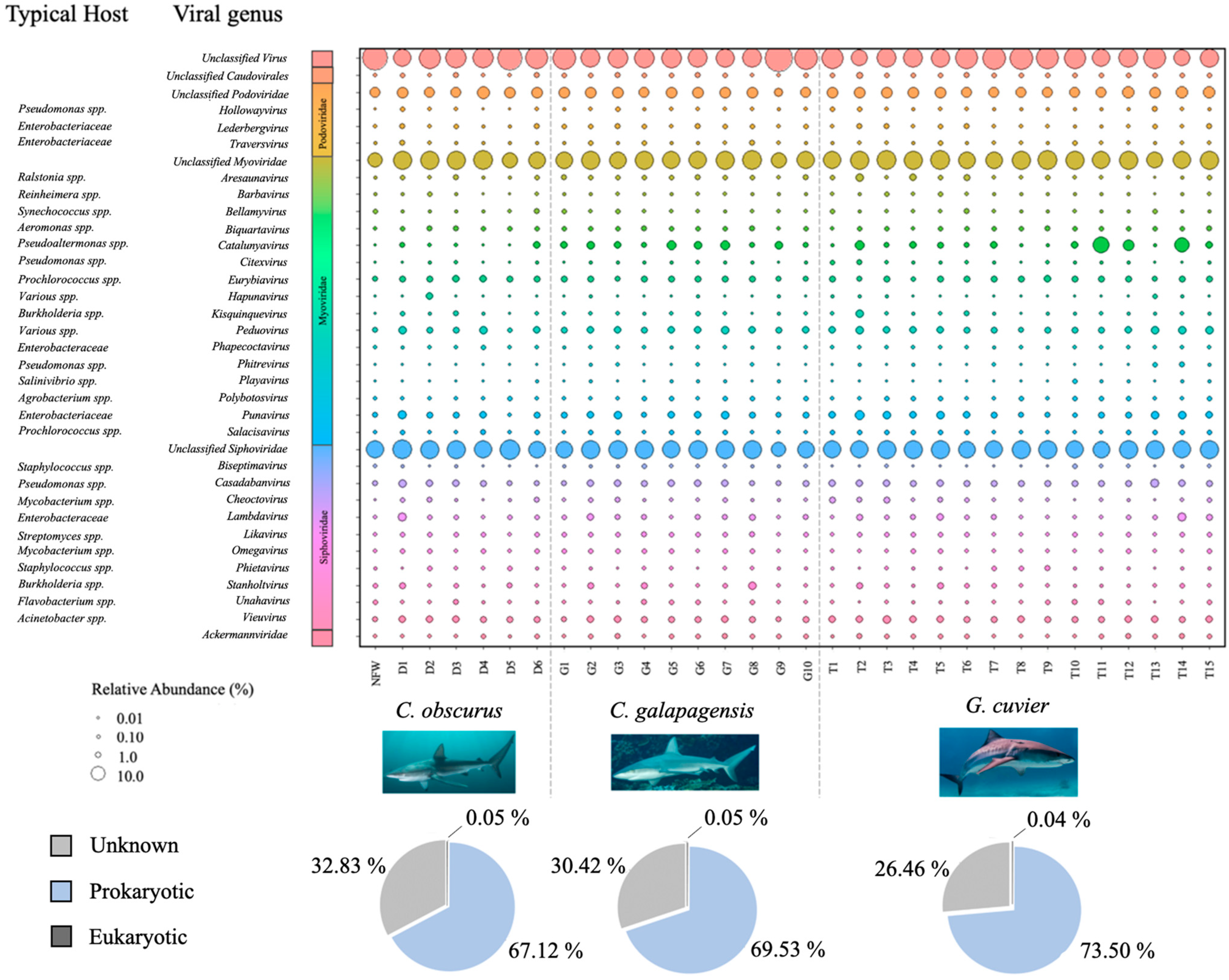
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shnit-Orland, M.; Kushmaro, A. Coral mucus-associated bacteria: A possible first line of defense. FEMS Microbiol. Ecol. 2009, 67, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Wu, E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes 2012, 3, 4–14. [Google Scholar] [CrossRef]
- Egerton, S.; Culloty, S.; Whooley, J.; Stanton, C.; Ross, R.P. The Gut Microbiota of Marine Fish. Front. Microbiol. 2018, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- Silveira, C.B.; Rohwer, F. Piggyback-the-Winner in host-associated microbial communities. NPJ Biofilms Microbiomes 2016, 2, 16010. [Google Scholar] [CrossRef]
- Hansen, M.F.; Svenningsen, S.L.; Røder, H.L.; Middelboe, M.; Burmølle, M. Big Impact of the Tiny: Bacteriophage-Bacteria Interactions in the Biofilm. Trends Microbiol. 2019, 27, 739–753. [Google Scholar] [CrossRef]
- Wahl, M.; Goecke, F.; Labes, A.; Dobretsov, S.; Weinberger, F. The second skin: Ecological role of epibiotic biofilms on marine organisms. Front. Microbiol. 2012, 3, 292. [Google Scholar] [CrossRef]
- Doane, M.P.; Haggerty, J.M.; Kacev, D.; Papudeshi, B.; Dinsdale, E.A. The skin microbiome of the common thresher shark (Alopias vulpinus) has low taxonomic and gene function β-diversity. Environ. Microbiol. Rep. 2017, 9, 357–373. [Google Scholar] [CrossRef]
- Minich, J.J.; Morris, M.M.; Brown, M.; Doane, M.P.; Edwards, M.S.; Michael, T.P.; Dinsdale, E.A. Elevated temperature drives kelp microbiome dysbiosis, while elevated carbon dioxide induces water microbiome disruption. PLoS ONE 2018, 13, e0192772. [Google Scholar] [CrossRef]
- Doane, M.P.; Morris, M.M.; Papudeshi, B.; Allen, L.; Pande, D.; Haggerty, J.M.; Johri, S.; Turnlund, A.C.; Peterson, M.; Kacev, D.; et al. The skin microbiome of elasmobranchs follows phylosymbiosis, but in teleost fishes, the microbiomes converge. Microbiome 2020, 8, 93. [Google Scholar] [CrossRef]
- Dinsdale, E.A.; Pantos, O.; Smriga, S.; Edwards, R.A.; Angly, F.; Wegley, L.; Hatay, M.; Hall, D.; Brown, E.; Haynes, M.; et al. Microbial ecology of four coral atolls in the Northern Line Islands. PLoS ONE 2008, 3, e1548. [Google Scholar] [CrossRef] [Green Version]
- McDole, T.; Nulton, J.; Barott, K.L.; Felts, B.; Hand, C.; Hatay, M.; Lee, H.; Nadon, M.O.; Nosrat, B.; Salamon, P.; et al. Assessing Coral Reefs on a Pacific-Wide Scale Using the Microbialization Score. PLoS ONE 2012, 7, e43233. [Google Scholar] [CrossRef]
- Haas, A.F.; Fairoz, M.F.M.; Kelly, L.W.; Nelson, C.E.; Dinsdale, E.A.; Edwards, R.A.; Giles, S.; Hatay, M.; Hisakawa, N.; Knowles, B.; et al. Global microbialization of coral reefs. Nat. Microbiol. 2016, 1, 16042. [Google Scholar] [CrossRef] [PubMed]
- Silveira, C.B.; Coutinho, F.H.; Cavalcanti, G.S.; Benler, S.; Doane, M.P.; Dinsdale, E.A.; Edwards, R.A.; Francini-Filho, R.B.; Thompson, C.C.; Luque, A.; et al. Genomic and ecological attributes of marine bacteriophages encoding bacterial virulence genes. BMC Genom. 2020, 21, 126. [Google Scholar] [CrossRef] [PubMed]
- Tricas, T.C.; Le Feuvre, E.M. Mating in the reef white-tip shark Triaenodon obesus. Mar. Biol. 1985, 84, 233–237. [Google Scholar] [CrossRef]
- Pratt, H.L.; Carrier, J.C. A Review of Elasmobranch Reproductive Behavior with a Case Study on the Nurse Shark, Ginglymostoma cirratum. Environ. Biol. Fishes 2001, 60, 157–188. [Google Scholar]
- Chin, A.; Mourier, J.; Rummer, J.L. Blacktip reef sharks (Carcharhinus melanopterus) show high capacity for wound healing and recovery following injury. Conserv. Physiol. 2015, 3, cov062. [Google Scholar] [CrossRef]
- Dosay-Akbulut, M. The phylogenetic relationship within the genus Carcharhinus. Comptes Rendus Biol. 2008, 331, 500–509. [Google Scholar] [CrossRef]
- Meyer, W.; Seegers, U. Basics of skin structure and function in elasmobranchs: A review. J. Fish. Biol. 2012, 80, 1940–1967. [Google Scholar] [CrossRef]
- Chien, H.-W.; Chen, X.-Y.; Tsai, W.-P.; Lee, M. Inhibition of biofilm formation by rough shark skin-patterned surfaces. Colloids Surf. B Biointerfaces 2020, 186, 110738. [Google Scholar] [CrossRef]
- Doane, M.P.; Johri, S.; Kerr, E.N.; Morris, M.M.; De Santiago, R.; Turnlund, A.C.; Goodman, A.Z.; Mora, M.F.; Lima, L.F.O.; Nosal, A.P.; et al. The Epidermal Microbiome Within an Aggregation of Leopard Sharks (Triakis semifasciata) Has Taxonomic Flexibility with Gene Functional Stability Across Three Time-Points. Microb. Ecol. 2022. [Google Scholar] [CrossRef]
- Pogoreutz, C.; Gore, M.A.; Perna, G.; Millar, C.; Nestler, R.; Ormond, R.F.; Clarke, C.R.; Voolstra, C. Similar bacterial communities on healthy and injured skin of black tip reef sharks. Anim. Microbiome 2019, 1, 9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodman, A.; Papudeshi, B.; Doane, M.P.; Mora, M.; Kerr, E.; Torres, M.; Nero, J.; Lima, L.F.O.; Nosel, A.P.; Dinsdale, E.A. Epidermal Microbiomes of Triakis semifasciata Are Consistent Across Captive and Wild Environments. Available online: https://www.researchsquare.com/article/rs-948364/v1 (accessed on 1 August 2022).
- Gadoin, E.; Desnues, C.; Monteil-Bouchard, S.; Bouvier, T.; Auguet, J.-C.; Roque d’Orbcastel, E.; Bettarel, Y. Fishing for the Virome of Tropical Tuna. Viruses 2021, 13, 1291. [Google Scholar] [CrossRef] [PubMed]
- Bossi, L.; Fuentes, J.A.; Mora, G.; Figueroa-Bossi, N. Prophage Contribution to Bacterial Population Dynamics. J. Bacteriol. 2003, 185, 6467–6471. [Google Scholar] [CrossRef] [PubMed]
- Suttle, C.A. Marine viruses–major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef]
- Rohwer, F.; Vega Thurber, R. Viruses manipulate the marine environment. Nature 2009, 459, 207–212. [Google Scholar] [CrossRef]
- Güemes, A.G.C.; Youle, M.; Cantú, V.A.; Felts, B.; Nulton, J.; Rohwer, F. Viruses as Winners in the Game of Life. Ann. Rev. Virol. 2016, 3, 197–214. [Google Scholar] [CrossRef]
- Argov, T.; Azulay, G.; Pasechnek, A.; Stadnyuk, O.; Ran-Sapir, S.; Borovok, I.; Sigal, N.; Herskovits, A.A. Temperate bacteriophages as regulators of host behavior. Curr. Opin. Microbiol. 2017, 38, 81–87. [Google Scholar] [CrossRef]
- Pazmiño, D.A.; van Herderden, L.; Simpfendorfer, C.A.; Junge, C.; Donnellan, S.C.; Hoyos-Padilla, E.M.; Duffy, C.A.J.; Hu-veneers, C.; Gillanders, B.M.; Butcher, P.A.; et al. Introgressive hybridisation between two widespread sharks in the east Pacific region. Mol. Phylogenet. Evol. 2019, 136, 119–127. [Google Scholar] [CrossRef]
- Lowe, C.G.; Wetherbee, B.M.; Crow, G.L.; Tester, A.L. Ontogenetic dietary shifts and feeding behavior of the tiger shark, Galeocerdo Cuvier, in Hawaiian waters. Environ. Biol Fish. 1996, 47, 203–211. [Google Scholar] [CrossRef]
- Dillon, E.M.; Norris, R.D.; O’Dea, A. Dermal Denticles as a tool to reconstruct shark communities. Mar. Ecol. Prog. Ser. 2017, 566, 117–134. [Google Scholar] [CrossRef]
- Gelsleichter, J.; Musick, J.A.; Nichols, S. Food habits of the smooth dogfish, Mustelus canis, dusky shark, Carcharhinus obscurus, Atlantic sharpnose shark, Rhizoprionodon terraenovae, and the sand tiger, Carcharias taurus, from the northwest Atlantic Ocean. Environ. Biol. Fishes 1999, 54, 205–217. [Google Scholar] [CrossRef]
- Papastamatiou, Y.P.; Meyer, C.G.; Kosaki, R.K.; Wallsgrove, N.J.; Popp, B.N. Movements and foraging of predators associated with mesophotic coral reefs and their potential for linking ecological habitats. Mar. Ecol. Prog. 2015, 521, 155–170. [Google Scholar] [CrossRef]
- Hussey, N.E.; McCarthy, I.D.; Dudley, S.F.; Mann, B.Q. Nursery grounds, movement patterns and growth rates of dusky sharks, Carcharhinus obscurus: A long-term tag and release study in South African waters. Mar. Freshw. Res. 2009, 60, 571–583. [Google Scholar] [CrossRef]
- Meyer, C.G.; Papastamatiou, Y.P.; Holland, K.N. A multiple instrument approach to quantifying the movement patterns and habitat use of tiger (Galeocerdo cuvier) and Galapagos sharks (Carcharhinus galapagensis) at French Frigate Shoals, Hawaii. Mar. Biol. 2010, 157, 1857–1868. [Google Scholar] [CrossRef]
- Aines, A.C.; Carlson, J.K.; Boustany, A.; Mathers, A.; Kohler, N.E. Feeding habits of the tiger shark, Galeocerdo cuvier, in the northwest Atlantic Ocean and Gulf of Mexico. Environ. Biol. Fish. 2018, 101, 403–415. [Google Scholar] [CrossRef]
- Kessel, S.T.; Hussey, N.E. Tonic immobility as an anaesthetic for elasmobranchs during surgical implantation procedures. Can. J. Fish. Aquat. Sci. 2015, 72, 1287–1291. [Google Scholar] [CrossRef]
- Luong, T.; Salabarria, A.-C.; Edwards, R.A.; Roach, D.R. Standardized bacteriophage purification for personalized phage therapy. Nat. Protoc. 2020, 15, 2867–2890. [Google Scholar] [CrossRef] [PubMed]
- Schmeider, R.; Edwards, R. Quality control and preprocessing of metagenomic datasets. Bioinformatics 2011, 27, 863–864. [Google Scholar] [CrossRef]
- Roach, M.; Beecroft, S.; Mihindukulasuriya, K.A.; Wang, L.; Lima, L.F.O.; Dinsdale, E.A.; Edwards, R.A.; Handley, S.A. Hecatomb: An End-to-End Research Platform for Viral Metagenomics. bioRxiv 2022. [Google Scholar] [CrossRef]
- Clarke, K.R.; Gorley, R.N.; Somerfield, P.J.; Warwick, R.M. Changes in Marine Communities: An Approach to Statistical Analysis and Interpretation, 3rd ed.; PRIMER-E., Ltd., Phymouth Marine Laboratory: Plymouth, UK, 2014. [Google Scholar]
- Lima, L.F.O.; Weissman, M.; Reed, M.; Papudeshi, B.; Alker, A.T.; Morris, M.M.; Edwards, R.A.; de Putron, S.J.; Vaidya, N.K.; Dinsdale, E.A. Modeling of the coral microbiome: The influence of temperature and microbial network. mBio 2020, 11, 1–17. [Google Scholar] [CrossRef]
- Clarke, K.R.; Somerfield, P.J.; Gorley, R.N. Testing of null hypotheses in exploratory community analyses: Similarity profiles and biota-environment linkage. J. Exp. Mar. Biol. Ecol. 2008, 366, 56–69. [Google Scholar] [CrossRef]
- Ondov, B.D.; Treangen, T.J.; Melsted, P.; Mallonee, A.B.; Bergman, N.H.; Koren, S.; Phillippy, A.M. Mash: Fast genome and metagenome distance estimation using MinHash. Genome Biol 2016, 17, 132. [Google Scholar] [CrossRef]
- Cárdenas, A.; Ye, J.; Ziegler, M.; Payet, J.P.; McMinds, R.; Vega Thurber, R.; Voolstra, C.R. Coral-Associated Viral Assemblages from the Central Red Sea Align with Host Species and Contribute to Holobiont Genetic Diversity. Front. Microbiol. 2020, 11, 572534. [Google Scholar] [CrossRef] [PubMed]
- Vega Thurber, R.; Payet, J.P.; Thurber, A.R.; Correa, A.M.S. Virus-host interactions and their roles in coral reef health and disease. Nat. Rev. Microbiol. 2017, 15, 205–216. [Google Scholar] [CrossRef]
- Krishnamurthy, S.R.; Wang, D. Origins and challenges of viral dark matter. Vir. Res. 2017, 239, 136–142. [Google Scholar] [CrossRef]
- Garner, M.M. A Retrospective Study of Disease in Elasmobranchs. Vet. Pathol. 2013, 50, 377–389. [Google Scholar] [CrossRef]
- Koonin, E.V.; Dolja, V.V.; Krupovic, M. Origins and evolution of viruses of eukaryotes: The ultimate modularity. Virology 2015, 479–480, 2–25. [Google Scholar] [CrossRef]
- Louten, J. Virus Structure and Classification. Essent. Hum. Virol. 2016, 19–29. [Google Scholar] [CrossRef]
- Breitbart, M.; Delwart, E.; Rosario, K.; Segalés, J.; Varsani, A. ICTV Virus Taxonomy Profile: Circoviridae. J. Gen. Virol. 2017, 98, 1997–1998. [Google Scholar]
- Ritchie, K.B.; Schwarz, M.; Mueller, J.; Lapacek, V.A.; Merselis, D.; Walsh, C.J.; Luer, C.A. Survey of Antibiotic-producing Bacteria Associated with the Epidermal Mucus Layers of Rays and Skates. Front. Microbiol. 2017, 8, 1050. [Google Scholar] [CrossRef]
- Merrifield, D.L.; Rodiles, A. The fish microbiome and its interactions with mucosal tissues. Mucosal Health Aquac. 2015, 273–295. [Google Scholar] [CrossRef]
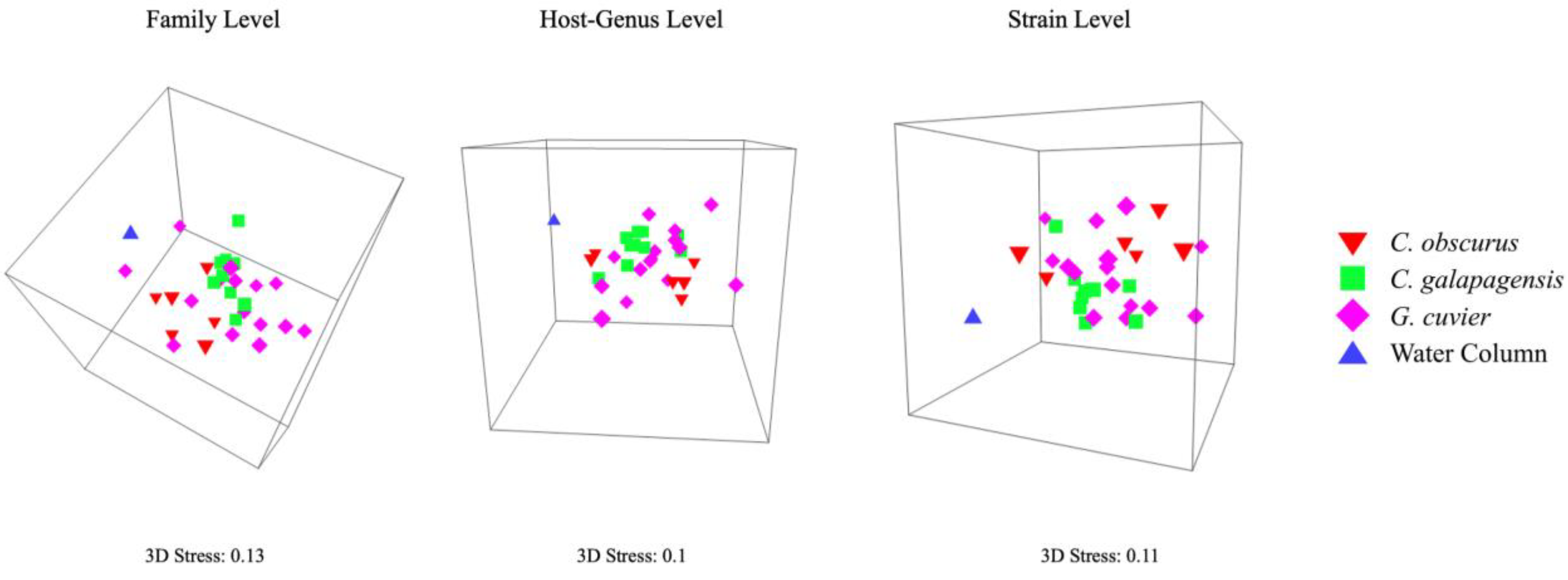

| Family Level | Water Column | C. obscurus | C. galapagensis | G. cuvier |
|---|---|---|---|---|
| C. obscurus | 84.38 | 86.18 | ||
| C. galapagensis | 84.55 | 86.99 | 90.35 | |
| G. cuvier | 82.21 | 86.09 | 88.13 | 86.63 |
| Host Genus level | Water Column | C. obscurus | C. galapagensis | G. cuvier |
| C. obscurus | 71.59 | 75.61 | ||
| C. galapagensis | 73.64 | 77.39 | 81.88 | |
| G. cuvier | 70.73 | 76.61 | 79.02 | 77.33 |
| Strain level | Water Column | C. obscurus | C. galapagensis | G. cuvier |
| C. obscurus | 60.35 | 63.32 | ||
| C. galapagensis | 63.07 | 65.57 | 71.97 | |
| G. cuvier | 58.90 | 64.20 | 67.64 | 65.17 |
| Groups | t-Statistic | Unique Permutations | Distance Between Centroids | p-Value |
|---|---|---|---|---|
| Family Level | ||||
| Water Column, C. obscurus | 1.21 | 7 | 12.88 | 0.14 |
| Water Column, C. galapagensis | 1.91 | 11 | 14.05 | 0.079 |
| Water Column, G. cuvier | 1.52 | 16 | 15.40 | 0.06 |
| C. obscurus, C. galapagensis | 1.68 | 5709 | 7.10 | 0.014 * |
| C. obscurus, G. cuvier | 1.17 | 9058 | 5.54 | 0.47 |
| C. galapagensis, G. cuvier | 1.12 | 9910 | 4.01 | 0.085 |
| Bacterial Host Genus Level | ||||
| Water Column, C. obscurus | 1.28 | 7 | 24.05 | 0.15 |
| Water Column, C. galapagensis | 1.73 | 11 | 23.45 | 0.087 |
| Water Column, G. cuvier | 1.49 | 16 | 25.04 | 0.062 |
| C. obscurus, C. galapagensis | 1.44 | 5686 | 10.86 | 0.010 * |
| C. obscurus, G. cuvier | 0.98 | 9045 | 7.80 | 0.49 |
| C. galapagensis, G. cuvier | 1.19 | 9877 | 7.27 | 0.093 |
| Strain Level | ||||
| Water Column, C. obscurus | 1.15 | 7 | 30.19 | 0.15 |
| Water Column, C. galapagensis | 1.52 | 11 | 30.45 | 0.096 |
| Water Column, G. cuvier | 1.31 | 16 | 31.44 | 0.13 |
| C. obscurus, C. galapagensis | 1.44 | 5652 | 15.82 | 0.003 * |
| C. obscurus, G. cuvier | 1.04 | 9059 | 12.58 | 0.32 |
| C. galapagensis, G. cuvier | 1.22 | 9838 | 10.35 | 0.038 * |
| Host | Water Column Percent Shared (SE) | C. obscurus Percent Shared (SE) | C. galapagensis Percent Shared (SE) | G. cuvier Percent Shared (SE) |
|---|---|---|---|---|
| C. obscurus | 10.8 ± 1.1 | 12.3 ± 1.4 | ||
| C. galapagensis | 12.2 ± 0.8 | 15.9 ± 0.7 | 29.6 ± 0.7 | |
| G. cuvier | 10.0 ± 0.9 | 13.2 ± 0.6 | 20.3 ± 0.7 | 15.8 ± 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hesse, R.D.; Roach, M.; Kerr, E.N.; Papudeshi, B.; Lima, L.F.O.; Goodman, A.Z.; Hoopes, L.; Scott, M.; Meyer, L.; Huveneers, C.; et al. Phage Diving: An Exploration of the Carcharhinid Shark Epidermal Virome. Viruses 2022, 14, 1969. https://doi.org/10.3390/v14091969
Hesse RD, Roach M, Kerr EN, Papudeshi B, Lima LFO, Goodman AZ, Hoopes L, Scott M, Meyer L, Huveneers C, et al. Phage Diving: An Exploration of the Carcharhinid Shark Epidermal Virome. Viruses. 2022; 14(9):1969. https://doi.org/10.3390/v14091969
Chicago/Turabian StyleHesse, Ryan D., Michael Roach, Emma N. Kerr, Bhavya Papudeshi, Laís F. O. Lima, Asha Z. Goodman, Lisa Hoopes, Mark Scott, Lauren Meyer, Charlie Huveneers, and et al. 2022. "Phage Diving: An Exploration of the Carcharhinid Shark Epidermal Virome" Viruses 14, no. 9: 1969. https://doi.org/10.3390/v14091969
APA StyleHesse, R. D., Roach, M., Kerr, E. N., Papudeshi, B., Lima, L. F. O., Goodman, A. Z., Hoopes, L., Scott, M., Meyer, L., Huveneers, C., & Dinsdale, E. A. (2022). Phage Diving: An Exploration of the Carcharhinid Shark Epidermal Virome. Viruses, 14(9), 1969. https://doi.org/10.3390/v14091969





