Time-of-Day Variation in SARS-CoV-2 RNA Levels during the Second Wave of COVID-19
Abstract
:1. Introduction
2. Methods
2.1. Sample Collection
2.2. SARS-CoV-2 RNA Quantification
2.3. Statistical Analysis
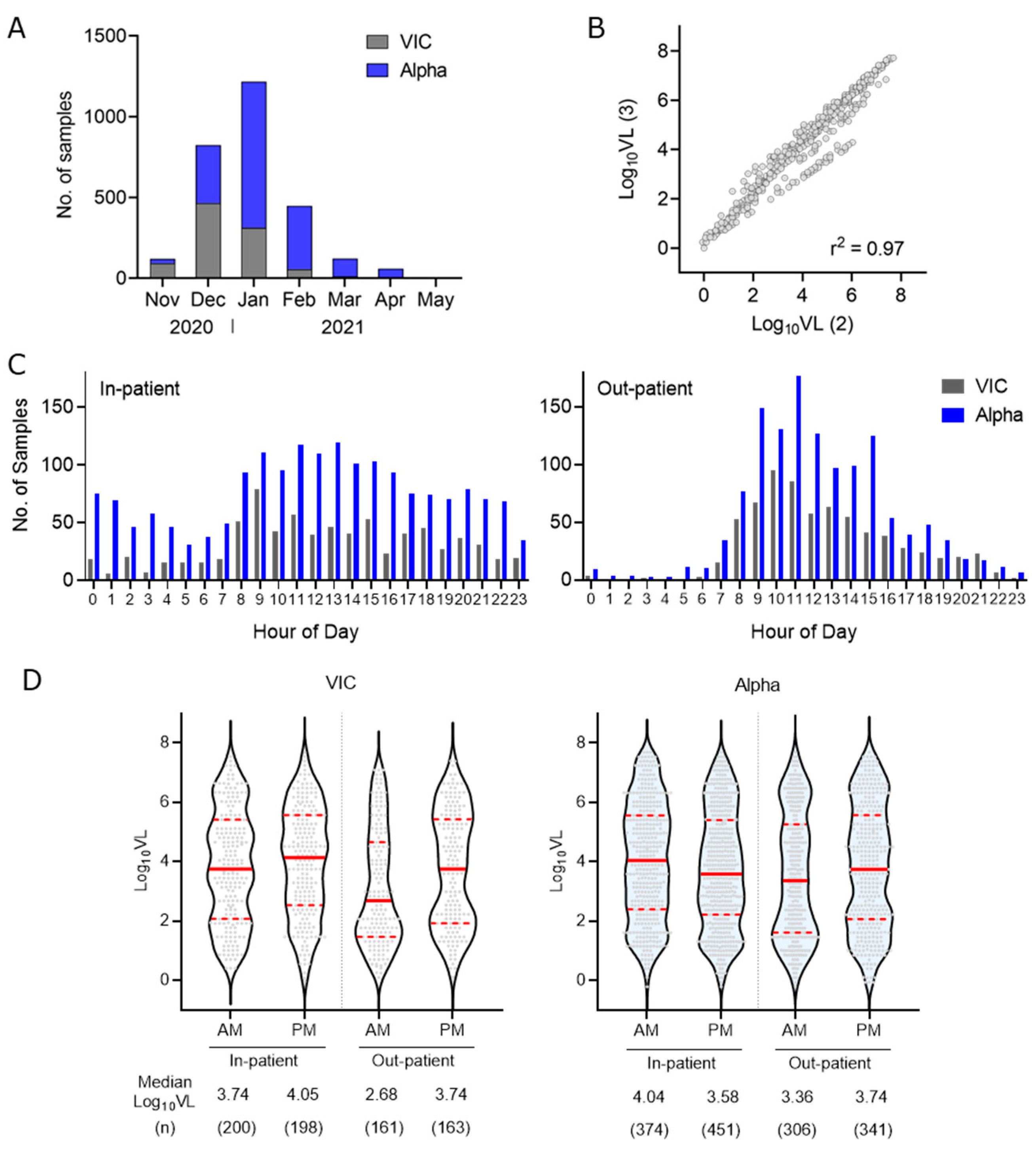
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Edwards, A.M.; Baric, R.S.; Saphire, E.O.; Ulmer, J.B. Stopping pandemics before they start: Lessons learned from SARS-CoV-2. Science 2022, 375, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Lutes, L.K.; Barnoud, C.; Scheiermann, C. The circadian immune system. Sci. Immunol. 2022, 7, eabm2465. [Google Scholar] [CrossRef] [PubMed]
- Wyse, C.; O’Malley, G.; Coogan, A.N.; McConkey, S.; Smith, D.J. Seasonal and daytime variation in multiple immune parameters in humans: Evidence from 329,261 participants of the UK Biobank cohort. iScience 2021, 24, 102255. [Google Scholar] [CrossRef]
- Sengupta, S.; Tang, S.Y.; Devine, J.C.; Anderson, S.T.; Nayak, S.; Zhang, S.L.; Valenzuela, A.; Fisher, D.G.; Grant, G.R.; Lopez, C.B.; et al. Circadian control of lung inflammation in influenza infection. Nat. Commun. 2019, 10, 4107. [Google Scholar] [CrossRef] [Green Version]
- Scheiermann, C.; Kunisaki, Y.; Lucas, D.; Chow, A.; Jang, J.E.; Zhang, D.; Hashimoto, D.; Merad, M.; Frenette, P.S. Adrenergic nerves govern circadian leukocyte recruitment to tissues. Immunity 2012, 37, 290–301. [Google Scholar] [CrossRef] [Green Version]
- Sutton, C.E.; Finlay, C.M.; Raverdeau, M.; Early, J.O.; DeCourcey, J.; Zaslona, Z.; O’Neill, L.A.J.; Mills, K.H.G.; Curtis, A.M. Loss of the molecular clock in myeloid cells exacerbates T cell-mediated CNS autoimmune disease. Nat. Commun. 2017, 8, 1923. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hunter, L.; Wu, G.; Maidstone, R.; Mizoro, Y.; Vonslow, R.; Fife, M.; Hopwood, T.; Begley, N.; Saer, B.; et al. Genome-wide effect of pulmonary airway epithelial cell-specific Bmal1 deletion. FASEB J. 2019, 33, 6226–6238. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, J.; Ince, L.; Matthews, L.; Mei, J.; Bell, T.; Yang, N.; Saer, B.; Begley, N.; Poolman, T.; Pariollaud, M.; et al. An epithelial circadian clock controls pulmonary inflammation and glucocorticoid action. Nat. Med. 2014, 20, 919–926. [Google Scholar] [CrossRef] [Green Version]
- Pariollaud, M.; Gibbs, J.E.; Hopwood, T.W.; Brown, S.; Begley, N.; Vonslow, R.; Poolman, T.; Guo, B.; Saer, B.; Jones, D.H.; et al. Circadian clock component REV-ERBalpha controls homeostatic regulation of pulmonary inflammation. J. Clin. Investig. 2018, 128, 2281–2296. [Google Scholar] [CrossRef] [Green Version]
- Early, J.O.; Menon, D.; Wyse, C.A.; Cervantes-Silva, M.P.; Zaslona, Z.; Carroll, R.G.; Palsson-McDermott, E.M.; Angiari, S.; Ryan, D.G.; Corcoran, S.E.; et al. Circadian clock protein BMAL1 regulates IL-1beta in macrophages via NRF2. Proc. Natl. Acad. Sci. USA 2018, 115, E8460–E8468. [Google Scholar] [CrossRef] [Green Version]
- Ehlers, A.; Xie, W.; Agapov, E.; Brown, S.; Steinberg, D.; Tidwell, R.; Sajol, G.; Schutz, R.; Weaver, R.; Yu, H.; et al. BMAL1 links the circadian clock to viral airway pathology and asthma phenotypes. Mucosal. Immunol. 2018, 11, 97–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scheiermann, C.; Gibbs, J.; Ince, L.; Loudon, A. Clocking in to immunity. Nat. Rev. Immunol. 2018, 18, 423–437. [Google Scholar] [CrossRef] [PubMed]
- Ince, L.M.; Zhang, Z.; Beesley, S.; Vonslow, R.M.; Saer, B.R.; Matthews, L.C.; Begley, N.; Gibbs, J.E.; Ray, D.W.; Loudon, A.S.I. Circadian variation in pulmonary inflammatory responses is independent of rhythmic glucocorticoid signaling in airway epithelial cells. FASEB J. 2019, 33, 126–139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edgar, R.S.; Stangherlin, A.; Nagy, A.D.; Nicoll, M.P.; Efstathiou, S.; O’Neill, J.S.; Reddy, A.B. Cell autonomous regulation of herpes and influenza virus infection by the circadian clock. Proc. Natl. Acad. Sci. USA 2016, 113, 10085–10090. [Google Scholar] [CrossRef] [Green Version]
- Borrmann, H.; McKeating, J.A.; Zhuang, X. The Circadian Clock and Viral Infections. J. Biol. Rhythm. 2021, 36, 9–22. [Google Scholar] [CrossRef]
- Zhuang, X.; Tsukuda, S.; Wrensch, F.; Wing, P.A.C.; Schilling, M.; Harris, J.M.; Borrmann, H.; Morgan, S.B.; Cane, J.L.; Mailly, L.; et al. The circadian clock component BMAL1 regulates SARS-CoV-2 entry and replication in lung epithelial cells. iScience 2021, 24, 103144. [Google Scholar] [CrossRef]
- Dean, C.B.; Nielsen, J.D. Generalized linear mixed models: A review and some extensions. Lifetime Data Anal. 2007, 13, 497–512. [Google Scholar] [CrossRef]
- McNaughton, C.D.; Adams, N.M.; Hirschie Johnson, C.; Ward, M.J.; Schmitz, J.E.; Lasko, T.A. Diurnal Variation in SARS-CoV-2 PCR Test Results: Test Accuracy May Vary by Time of Day. J. Biol. Rhythm. 2021, 36, 595–601. [Google Scholar] [CrossRef]
- Hung, D.L.; Li, X.; Chiu, K.H.; Yip, C.C.; To, K.K.; Chan, J.F.; Sridhar, S.; Chung, T.W.; Lung, K.C.; Liu, R.W.; et al. Early-Morning vs Spot Posterior Oropharyngeal Saliva for Diagnosis of SARS-CoV-2 Infection: Implication of Timing of Specimen Collection for Community-Wide Screening. Open Forum Infect. Dis. 2020, 7, ofaa210. [Google Scholar] [CrossRef]
- Katayama, Y.; Murai, R.; Moriai, M.; Nirasawa, S.; Saeki, M.; Yakuwa, Y.; Sato, Y.; Asanuma, K.; Fujiya, Y.; Kuronuma, K.; et al. Does the timing of saliva collection affect the diagnosis of SARS-CoV-2 infection? J. Infect. Chemother. 2022, 28, 1012–1014. [Google Scholar] [CrossRef]
- Yavropoulou, M.P.; Filippa, M.G.; Mantzou, A.; Ntziora, F.; Mylona, M.; Tektonidou, M.G.; Vlachogiannis, N.I.; Paraskevis, D.; Kaltsas, G.A.; Chrousos, G.P.; et al. Alterations in cortisol and interleukin-6 secretion in patients with COVID-19 suggestive of neuroendocrine-immune adaptations. Endocrine 2022, 75, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Haspel, J.; Kim, M.; Zee, P.; Schwarzmeier, T.; Montagnese, S.; Panda, S.; Albani, A.; Merrow, M. A Timely Call to Arms: COVID-19, the Circadian Clock, and Critical Care. J. Biol. Rhythm. 2021, 36, 55–70. [Google Scholar] [CrossRef] [PubMed]
- Telias, I.; Wilcox, M.E. Sleep and Circadian Rhythm in Critical Illness. Crit. Care 2019, 23, 82. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meira, E.C.M.; Miyazawa, M.; Gozal, D. Putative contributions of circadian clock and sleep in the context of SARS-CoV-2 infection. Eur Respir J. 2020, 55, 2001023. [Google Scholar] [CrossRef]
- Lee, J.Y.; Wing, P.A.; Gala, D.S.; Noerenberg, M.; Jarvelin, A.I.; Titlow, J.; Zhuang, X.; Palmalux, N.; Iselin, L.; Thompson, M.K.; et al. Absolute quantitation of individual SARS-CoV-2 RNA molecules provides a new paradigm for infection dynamics and variant differences. Elife 2022, 11, e74153. [Google Scholar] [CrossRef]
- Hawkes, M.T.; Lee, B.E.; Kanji, J.N.; Zelyas, N.; Wong, K.; Barton, M.; Mukhi, S.; Robinson, J.L. Seasonality of Respiratory Viruses at Northern Latitudes. JAMA Netw. Open 2021, 4, e2124650. [Google Scholar] [CrossRef]
- Audi, A.; AlIbrahim, M.; Kaddoura, M.; Hijazi, G.; Yassine, H.M.; Zaraket, H. Seasonality of Respiratory Viral Infections: Will COVID-19 Follow Suit? Front. Public Health 2020, 8, 567184. [Google Scholar] [CrossRef]
- Wirz-Justice, A.; Wever, R.A.; Aschoff, J. Seasonality in freerunning circadian rhythms in man. Naturwissenschaften 1984, 71, 316–319. [Google Scholar] [CrossRef]
- Hannay, K.M.; Forger, D.B.; Booth, V. Seasonality and light phase-resetting in the mammalian circadian rhythm. Sci. Rep. 2020, 10, 19506. [Google Scholar] [CrossRef]
- Prather, A.A.; Pressman, S.D.; Miller, G.E.; Cohen, S. Temporal Links Between Self-Reported Sleep and Antibody Responses to the Influenza Vaccine. Int. J. Behav. Med. 2021, 28, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Lange, T.; Perras, B.; Fehm, H.L.; Born, J. Sleep enhances the human antibody response to hepatitis A vaccination. Psychosom. Med. 2003, 65, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Despres, H.W.; Mills, M.G.; Shirley, D.J.; Schmidt, M.M.; Huang, M.L.; Roychoudhury, P.; Jerome, K.R.; Greninger, A.L.; Bruce, E.A. Measuring infectious SARS-CoV-2 in clinical samples reveals a higher viral titer:RNA ratio for Delta and Epsilon vs. Alpha variants. Proc. Natl. Acad. Sci. USA 2022, 119, e2116518119. [Google Scholar] [CrossRef] [PubMed]
- Fajnzylber, J.; Regan, J.; Coxen, K.; Corry, H.; Wong, C.; Rosenthal, A.; Worrall, D.; Giguel, F.; Piechocka-Trocha, A.; Atyeo, C.; et al. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat. Commun. 2020, 11, 5493. [Google Scholar] [CrossRef] [PubMed]
| Female | Male | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| In-Patients | Out-Patients | In-Patients | Out-Patients | ||||||
| Age | AM | PM | AM | PM | AM | PM | AM | PM | Total |
| 18–39 | 31 | 39 | 71 | 84 | 23 | 28 | 50 | 77 | 403 |
| 40–59 | 44 | 67 | 56 | 56 | 88 | 84 | 62 | 62 | 519 |
| 60–79 | 90 | 89 | 70 | 66 | 159 | 164 | 67 | 62 | 767 |
| 80–104 | 83 | 87 | 40 | 50 | 56 | 91 | 50 | 46 | 503 |
| Total | 248 | 282 | 237 | 256 | 326 | 367 | 229 | 247 | 2192 |
| Main Effects: | Num DF | F-Value | p-Value |
|---|---|---|---|
| Time (AM/PM) | 1 | 4.09 | 0.044 |
| Age | 3 | 0.14 | 0.935 |
| Sex | 1 | 4.19 | 0.041 |
| In/Out patient | 1 | 7.28 | 0.007 |
| VIC/Alpha | 1 | 2.94 | 0.087 |
| Interaction terms: | |||
| Time × Age | 3 | 1.69 | 0.169 |
| Time × Sex | 1 | 0.48 | 0.487 |
| Time × In/Out patient | 1 | 8.45 | 0.004 |
| Time × VIC/Alpha | 1 | 4.17 | 0.042 |
| Age × Sex | 3 | 0.93 | 0.426 |
| Age × In/Out patient | 3 | 7.93 | <0.0001 |
| Age × VIC/Alpha | 3 | 2.02 | 0.11 |
| Sex × In/Out patient | 1 | 2.76 | 0.097 |
| Sex × VIC/Alpha | 1 | 0.04 | 0.847 |
| In/Out patient × VIC/Alpha | 1 | 0.18 | 0.671 |
| Main Effects: | Num DF | F-Value | p-Value |
|---|---|---|---|
| Time (AM/Mid-day/PM) | 2 | 3.09 | 0.046 |
| Age | 3 | 0.44 | 0.725 |
| Sex | 1 | 1.92 | 0.166 |
| In/Out patient | 1 | 11.68 | 0.0007 |
| VIC/Alpha | 1 | 4.2 | 0.041 |
| Interaction terms: | |||
| Time × Age | 6 | 1.79 | 0.099 |
| Time × Sex | 2 | 1.4 | 0.248 |
| Time × In/Out patient | 2 | 3.17 | 0.043 |
| Time × VIC/Alpha | 2 | 3.8 | 0.023 |
| Age × Sex | 3 | 1.03 | 0.381 |
| Age × In/Out patient | 3 | 6.98 | 0.0001 |
| Age × VIC/Alpha | 3 | 2.08 | 0.102 |
| Sex × In/Out patient | 1 | 1.22 | 0.27 |
| Sex × VIC/Alpha | 1 | 0 | 0.97 |
| In/Out patient × VIC/Alpha | 1 | 0.48 | 0.491 |
| Main Effects: | Num DF | F-Value | p-Value |
|---|---|---|---|
| Time (AM/PM) | 1 | 6.71 | 0.0096 |
| Age (B-spline forms) | 6 | 0.96 | 0.4482 |
| Sex | 1 | 3.2 | 0.0737 |
| In/Out patient | 1 | 1.13 | 0.2882 |
| VIC/Alpha | 1 | 3.64 | 0.0567 |
| Interaction terms: | |||
| Time × Age | 6 | 2.23 | 0.0382 |
| Time × Sex | 1 | 0.22 | 0.6354 |
| Time × In/Out patient | 1 | 7.02 | 0.0081 |
| Time × VIC/Alpha | 1 | 4.11 | 0.0427 |
| Age × Sex | 6 | 1.22 | 0.2949 |
| Age × In/Out patient | 6 | 4.66 | 0.0001 |
| Age × VIC/Alpha | 6 | 1.41 | 0.2054 |
| Sex × In/Out patient | 1 | 2.81 | 0.094 |
| Sex × VIC/Alpha | 1 | 0 | 0.9711 |
| In/Out patient × VIC/Alpha | 1 | 0.45 | 0.5004 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhuang, X.; Wang, W.; Borrmann, H.; Balfe, P.; Matthews, P.C.; Eyre, D.W.; Klerman, E.B.; McKeating, J.A. Time-of-Day Variation in SARS-CoV-2 RNA Levels during the Second Wave of COVID-19. Viruses 2022, 14, 1728. https://doi.org/10.3390/v14081728
Zhuang X, Wang W, Borrmann H, Balfe P, Matthews PC, Eyre DW, Klerman EB, McKeating JA. Time-of-Day Variation in SARS-CoV-2 RNA Levels during the Second Wave of COVID-19. Viruses. 2022; 14(8):1728. https://doi.org/10.3390/v14081728
Chicago/Turabian StyleZhuang, Xiaodong, Wei Wang, Helene Borrmann, Peter Balfe, Philippa C. Matthews, David W. Eyre, Elizabeth B. Klerman, and Jane A. McKeating. 2022. "Time-of-Day Variation in SARS-CoV-2 RNA Levels during the Second Wave of COVID-19" Viruses 14, no. 8: 1728. https://doi.org/10.3390/v14081728
APA StyleZhuang, X., Wang, W., Borrmann, H., Balfe, P., Matthews, P. C., Eyre, D. W., Klerman, E. B., & McKeating, J. A. (2022). Time-of-Day Variation in SARS-CoV-2 RNA Levels during the Second Wave of COVID-19. Viruses, 14(8), 1728. https://doi.org/10.3390/v14081728






