Genetic Associations and Differential mRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient and Control Samples
2.1.1. Samples Used for the Association Analysis
2.1.2. Samples Used for RNA Expression Analysis
2.2. Selection of Candidate Genes
2.3. Genotyping
2.4. RNA-seq
2.5. Statistical Analysis
2.6. In Silico Analysis
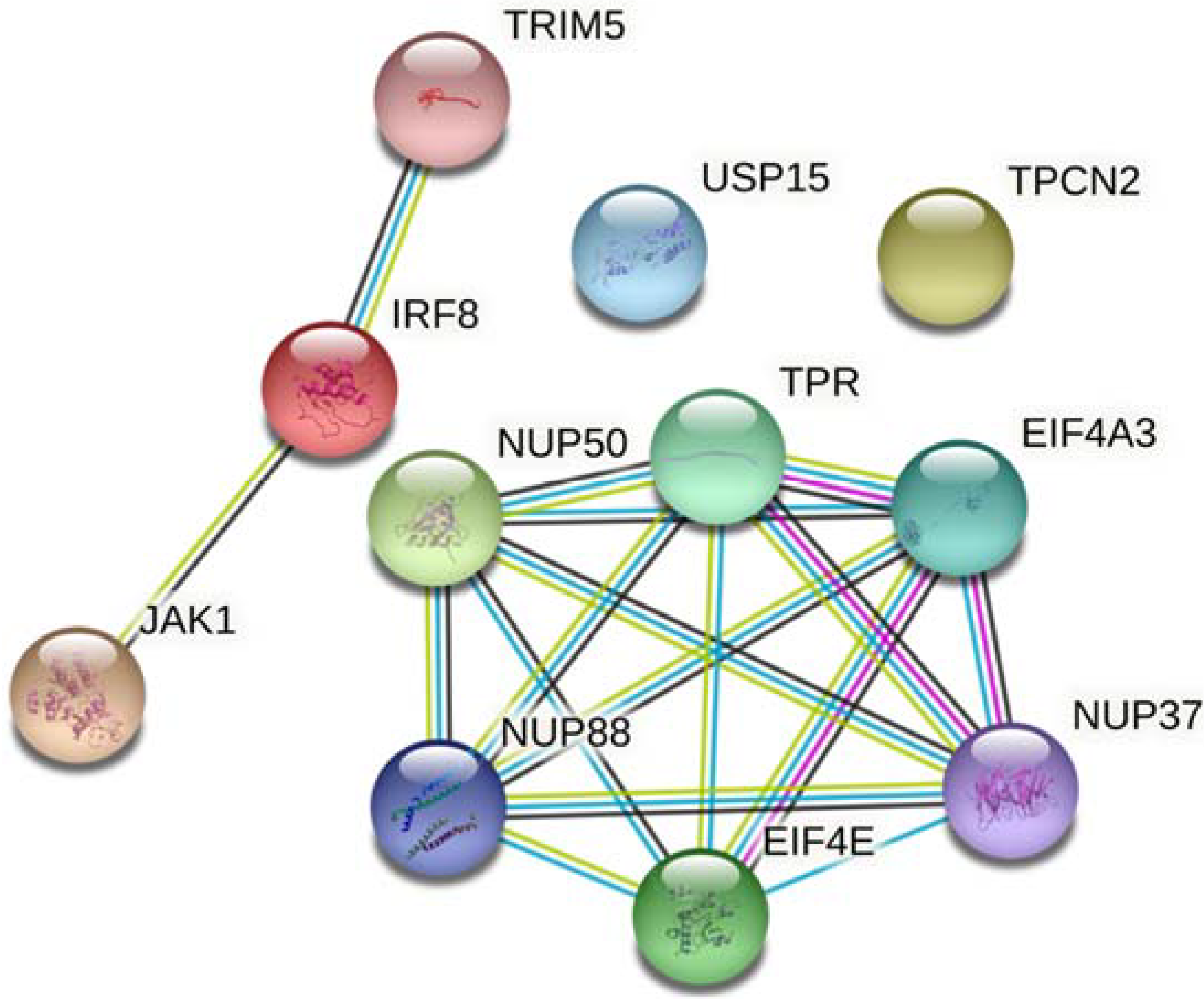
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schmidt, E.; Kasperkiewicz, M.; Joly, P. Pemphigus. Lancet 2019, 394, 882–894. [Google Scholar] [CrossRef]
- Alpsoy, E.; Akman-Karakas, A.; Uzun, S. Geographic Variations in Epidemiology of Two Autoimmune Bullous Diseases: Pemphigus and Bullous Pemphigoid. Arch. Dermatol. Res. 2015, 307, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Kridin, K.; Schmidt, E. Epidemiology of Pemphigus. JID Innov. 2021, 1, 100004. [Google Scholar] [CrossRef] [PubMed]
- Aoki, V.; Millikan, R.C.; Rivitti, E.A.; Hans-Filho, G.; Eaton, D.P.; Warren, S.J.P.; Li, N.; Hilario-Vargas, J.; Hoffmann, R.G.; Diaz, L.A.; et al. Environmental Risk Factors in Endemic Pemphigus Foliaceus (Fogo Selvagem). J. Investig. Dermatol. Symp. Proc. 2004, 9, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Petzl-Erler, M.L. Beyond the HLA Polymorphism: A Complex Pattern of Genetic Susceptibility to Pemphigus. Genet. Mol. Biol. 2020, 43, e20190369. [Google Scholar] [CrossRef]
- Brenner, S.; Bialy-Golan, A.; Ruocco, V. Drug-Induced Pemphigus. Clin. Dermatol. 1998, 16, 393–397. [Google Scholar] [CrossRef]
- Pile, H.D.; Yarrarapu, S.N.S.; Crane, J.S. Drug Induced Pemphigus. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Ruocco, V.; Ruocco, E.; Lo Schiavo, A.; Brunetti, G.; Guerrera, L.P.; Wolf, R. Pemphigus: Etiology, Pathogenesis, and Inducing or Triggering Factors: Facts and Controversies. Clin. Dermatol. 2013, 31, 374–381. [Google Scholar] [CrossRef]
- Tavakolpour, S. Pemphigus Trigger Factors: Special Focus on Pemphigus Vulgaris and Pemphigus Foliaceus. Arch. Dermatol. Res. 2018, 310, 95–106. [Google Scholar] [CrossRef]
- Lombardi, C.; Borges, P.C.; Chaul, A.; Sampaio, S.A.; Rivitti, E.A.; Friedman, H.; Martins, C.R.; Sanches Júnior, J.A.; Cunha, P.R.; Hoffmann, R.G. Environmental Risk Factors in Endemic Pemphigus Foliaceus (Fogo Selvagem). The Cooperative Group on Fogo Selvagem Research. J. Investig. Dermatol. 1992, 98, 847–850. [Google Scholar] [CrossRef]
- Vernal, S.; Pepinelli, M.; Casanova, C.; Goulart, T.M.; Kim, O.; De Paula, N.A.; Pinto, M.C.; Sá-Nunes, A.; Roselino, A.M. Insights into the Epidemiological Link between Biting Flies and Pemphigus Foliaceus in Southeastern Brazil. Acta Trop. 2017, 176, 455–462. [Google Scholar] [CrossRef]
- Qian, Y.; Culton, D.A.; Jeong, J.S.; Trupiano, N.; Valenzuela, J.G.; Diaz, L.A. Non-Infectious Environmental Antigens as a Trigger for the Initiation of an Autoimmune Skin Disease. Autoimmun. Rev. 2016, 15, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Tufano, M.A.; Baroni, A.; Buommino, E.; Ruocco, E.; Lombardi, M.L.; Ruocco, V. Detection of Herpesvirus DNA in Peripheral Blood Mononuclear Cells and Skin Lesions of Patients with Pemphigus by Polymerase Chain Reaction. Br. J. Dermatol. 1999, 141, 1033–1039. [Google Scholar] [CrossRef] [PubMed]
- Machado, A.R.D.S.R.; La Serra, L.; Turatti, A.; Machado, A.M.; Roselino, A.M. Herpes Simplex Virus 1 and Cytomegalovirus Are Associated with Pemphigus Vulgaris but Not with Pemphigus Foliaceus Disease. Exp. Dermatol. 2017, 26, 966–968. [Google Scholar] [CrossRef] [PubMed]
- Dominari, A.; Iii, H.D.; Pandav, K.; Matos, W.; Biswas, S.; Reddy, G.; Thevuthasan, S.; Khan, M.A.; Mathew, A.; Makkar, S.S.; et al. Thymosin Alpha 1: A Comprehensive Review of the Literature. World J. Virol. 2020, 9, 67–78. [Google Scholar] [CrossRef]
- Roscoe, J.T.; Naylor, P.H.; Diaz, L.A.; Labib, R.S.; Patel, H.P.; Goldstein, A.L.; Sampaio, S.A.; Anhalt, G.J. Elevated Thymosin Alpha I Levels in Brazilian Pemphigus Foliaceus. Br. J. Dermatol. 1986, 115, 147–150. [Google Scholar] [CrossRef]
- Getts, D.R.; Chastain, E.M.L.; Terry, R.L.; Miller, S.D. Virus Infection, Antiviral Immunity, and Autoimmunity. Immunol. Rev. 2013, 255, 197–209. [Google Scholar] [CrossRef]
- Houen, G.; Trier, N.H. Epstein-Barr Virus and Systemic Autoimmune Diseases. Front. Immunol. 2020, 11, 587380. [Google Scholar] [CrossRef]
- Afrasiabi, A.; Keane, J.T.; Ong, L.T.C.; Alinejad-Rokny, H.; Fewings, N.L.; Booth, D.R.; Parnell, G.P.; Swaminathan, S. Genetic and Transcriptomic Analyses Support a Switch to Lytic Phase in Epstein Barr Virus Infection as an Important Driver in Developing Systemic Lupus Erythematosus. J. Autoimmun. 2022, 127, 102781. [Google Scholar] [CrossRef]
- Kraberger, S.; Schmidlin, K.; Fontenele, R.S.; Walters, M.; Varsani, A. Unravelling the Single-Stranded DNA Virome of the New Zealand Blackfly. Viruses 2019, 11, E532. [Google Scholar] [CrossRef]
- Lu, G.; Ye, Z.-X.; He, Y.-J.; Zhang, Y.; Wang, X.; Huang, H.-J.; Zhuo, J.-C.; Sun, Z.-T.; Yan, F.; Chen, J.-P.; et al. Discovery of Two Novel Negeviruses in a Dungfly Collected from the Arctic. Viruses 2020, 12, E692. [Google Scholar] [CrossRef]
- Kobayashi, D.; Watanabe, M.; Faizah, A.N.; Amoa-Bosompem, M.; Higa, Y.; Tsuda, Y.; Sawabe, K.; Isawa, H. Discovery of a Novel Flavivirus (Flaviviridae) From the Horse Fly, Tabanus Rufidens (Diptera: Tabanidae): The Possible Coevolutionary Relationships Between the Classical Insect-Specific Flaviviruses and Host Dipteran Insects. J. Med. Entomol. 2021, 58, 880–890. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Pang, R.; Cheng, T.; Xue, L.; Zeng, H.; Lei, T.; Chen, M.; Wu, S.; Ding, Y.; Zhang, J.; et al. Abundant and Diverse RNA Viruses in Insects Revealed by RNA-Seq Analysis: Ecological and Evolutionary Implications. mSystems 2020, 5, e00039-20. [Google Scholar] [CrossRef] [PubMed]
- Olmo, R.P.; Martins, N.E.; Aguiar, E.R.G.R.; Marques, J.T.; Imler, J.-L. The Insect Reservoir of Biodiversity for Viruses and for Antiviral Mechanisms. An. Acad. Bras. Cienc. 2019, 91 (Suppl. 3), e20190122. [Google Scholar] [CrossRef] [PubMed]
- Green, M.R.; Sambrook, J.; Sambrook, J. Molecular Cloning: A Laboratory Manual, 4th ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 2012. [Google Scholar]
- Calonga-Solís, V.; Amorim, L.M.; Farias, T.D.J.; Petzl-Erler, M.L.; Malheiros, D.; Augusto, D.G. Variation in Genes Implicated in B-Cell Development and Antibody Production Affects Susceptibility to Pemphigus. Immunology 2021, 162, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Joly, P.; Horvath, B.; Patsatsi, A.; Uzun, S.; Bech, R.; Beissert, S.; Bergman, R.; Bernard, P.; Borradori, L.; Caproni, M.; et al. Updated S2K Guidelines on the Management of Pemphigus Vulgaris and Foliaceus Initiated by the European Academy of Dermatology and Venereology (EADV). J. Eur. Acad. Dermatol. Venereol. 2020, 34, 1900–1913. [Google Scholar] [CrossRef] [PubMed]
- Salviano-Silva, A.; Becker, M.; Augusto, D.G.; Busch, H.; Adelman Cipolla, G.; Farias, T.D.-J.; Bumiller-Bini, V.; Calonga-Solís, V.; Munz, M.; Franke, A.; et al. Genetic Association and Differential Expression of HLA Complex Group LncRNAs in Pemphigus. J. Autoimmun. 2021, 123, 102705. [Google Scholar] [CrossRef]
- Wu, G.; Haw, R. Functional Interaction Network Construction and Analysis for Disease Discovery. Methods Mol. Biol. Clifton NJ 2017, 1558, 235–253. [Google Scholar] [CrossRef]
- Griss, J.; Viteri, G.; Sidiropoulos, K.; Nguyen, V.; Fabregat, A.; Hermjakob, H. ReactomeGSA-Efficient Multi-Omics Comparative Pathway Analysis. Mol. Cell. Proteom. MCP 2020, 19, 2115–2125. [Google Scholar] [CrossRef]
- Augusto, D.G.; de Almeida, R.C.; Farias, T.D.J.; Magalhães, W.C.S.; Malheiros, D.; Lima-Costa, M.F.; Barreto, M.L.; Horta, B.L.; Kumar, V.; Wittig, M.; et al. Unsuspected Associations of Variants within the Genes NOTCH4 and STEAP2-AS1 Uncovered by a GWAS in Endemic Pemphigus Foliaceus. J. Investig. Dermatol. 2021, 141, 2741–2744. [Google Scholar] [CrossRef]
- Bumiller-Bini, V.; Cipolla, G.A.; de Almeida, R.C.; Petzl-Erler, M.L.; Augusto, D.G.; Boldt, A.B.W. Sparking Fire Under the Skin? Answers From the Association of Complement Genes With Pemphigus Foliaceus. Front. Immunol. 2018, 9, 695. [Google Scholar] [CrossRef]
- Bumiller-Bini, V.; Cipolla, G.A.; Spadoni, M.B.; Augusto, D.G.; Petzl-Erler, M.L.; Beltrame, M.H.; Boldt, A.B.W. Condemned or Not to Die? Gene Polymorphisms Associated with Cell Death in Pemphigus Foliaceus. Front. Immunol. 2019, 10, 2416. [Google Scholar] [CrossRef] [PubMed]
- Purcell, S.; Neale, B.; Todd-Brown, K.; Thomas, L.; Ferreira, M.A.R.; Bender, D.; Maller, J.; Sklar, P.; de Bakker, P.I.W.; Daly, M.J.; et al. PLINK: A Tool Set for Whole-Genome Association and Population-Based Linkage Analyses. Am. J. Hum. Genet. 2007, 81, 559–575. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, D.J.; Berger, J.O.; Johannesson, M.; Nosek, B.A.; Wagenmakers, E.-J.; Berk, R.; Bollen, K.A.; Brembs, B.; Brown, L.; Camerer, C.; et al. Redefine Statistical Significance. Nat. Hum. Behav. 2018, 2, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Ioannidis, J.P.A. The Proposal to Lower P Value Thresholds to 0.005. JAMA 2018, 319, 1429–1430. [Google Scholar] [CrossRef]
- Di Leo, G.; Sardanelli, F. Statistical Significance: P Value, 0.05 Threshold, and Applications to Radiomics-Reasons for a Conservative Approach. Eur. Radiol. Exp. 2020, 4, 18. [Google Scholar] [CrossRef]
- Xie, Z.; Bailey, A.; Kuleshov, M.V.; Clarke, D.J.B.; Evangelista, J.E.; Jenkins, S.L.; Lachmann, A.; Wojciechowicz, M.L.; Kropiwnicki, E.; Jagodnik, K.M.; et al. Gene Set Knowledge Discovery with Enrichr. Curr. Protoc. 2021, 1, e90. [Google Scholar] [CrossRef]
- Lee, B.-K.; Bhinge, A.A.; Iyer, V.R. Wide-Ranging Functions of E2F4 in Transcriptional Activation and Repression Revealed by Genome-Wide Analysis. Nucleic Acids Res. 2011, 39, 3558–3573. [Google Scholar] [CrossRef]
- Malheiros, D.; Panepucci, R.A.; Roselino, A.M.; Araújo, A.G.; Zago, M.A.; Petzl-Erler, M.L. Genome-Wide Gene Expression Profiling Reveals Unsuspected Molecular Alterations in Pemphigus Foliaceus. Immunology 2014, 143, 381–395. [Google Scholar] [CrossRef]
- Zhou, A.; Dong, X.; Liu, M.; Tang, B. Comprehensive Transcriptomic Analysis Identifies Novel Antiviral Factors Against Influenza A Virus Infection. Front. Immunol. 2021, 12, 632798. [Google Scholar] [CrossRef]
- Mogensen, T.H. IRF and STAT Transcription Factors-From Basic Biology to Roles in Infection, Protective Immunity, and Primary Immunodeficiencies. Front. Immunol. 2018, 9, 3047. [Google Scholar] [CrossRef]
- Froggatt, H.M.; Harding, A.T.; Chaparian, R.R.; Heaton, N.S. ETV7 Limits Antiviral Gene Expression and Control of Influenza Viruses. Sci. Signal. 2021, 14, eabe1194. [Google Scholar] [CrossRef] [PubMed]
- Vieira, J.P. Pemphigus foliaceus (fogo salvagem): An endemic disease of the state of são paulo (Brazil). Arch. Dermatol. Syphilol. 1940, 41, 858. [Google Scholar] [CrossRef]
- Peng, B.; Temple, B.R.; Yang, J.; Geng, S.; Culton, D.A.; Qian, Y. Identification of a Primary Antigenic Target of Epitope Spreading in Endemic Pemphigus Foliaceus. J. Autoimmun. 2021, 116, 102561. [Google Scholar] [CrossRef] [PubMed]
- Vernal, S.; Oliveira, F.; Oliveira, W.H.C.; Goulart, T.M.; Oristian, J.; Calvo, E.; Pinto, M.C.; Roselino, A.M.; Ribeiro, J.M.C. RNA-Sequencing of the Nyssomyia Neivai Sialome: A Sand Fly-Vector from a Brazilian Endemic Area for Tegumentary Leishmaniasis and Pemphigus Foliaceus. Sci. Rep. 2020, 10, 17664. [Google Scholar] [CrossRef] [PubMed]
- Mason, A.L. Is PBC a Viral Infectious Disease? Best Pract. Res. Clin. Gastroenterol. 2018, 34–35, 27–39. [Google Scholar] [CrossRef]
- Jog, N.R.; James, J.A. Epstein Barr Virus and Autoimmune Responses in Systemic Lupus Erythematosus. Front. Immunol. 2020, 11, 623944. [Google Scholar] [CrossRef]
- Volkmann, B.; Wittmann, S.; Lagisquet, J.; Deutschmann, J.; Eissmann, K.; Ross, J.J.; Biesinger, B.; Gramberg, T. Human TRIM5α Senses and Restricts LINE-1 Elements. Proc. Natl. Acad. Sci. USA 2020, 117, 17965–17976. [Google Scholar] [CrossRef]
- Ganser-Pornillos, B.K.; Pornillos, O. Restriction of HIV-1 and Other Retroviruses by TRIM5. Nat. Rev. Microbiol. 2019, 17, 546–556. [Google Scholar] [CrossRef]
- Chiramel, A.I.; Meyerson, N.R.; McNally, K.L.; Broeckel, R.M.; Montoya, V.R.; Méndez-Solís, O.; Robertson, S.J.; Sturdevant, G.L.; Lubick, K.J.; Nair, V.; et al. TRIM5α Restricts Flavivirus Replication by Targeting the Viral Protease for Proteasomal Degradation. Cell Rep. 2019, 27, 3269–3283.e6. [Google Scholar] [CrossRef]
- Rose, K.M.; Spada, S.J.; Broeckel, R.; McNally, K.L.; Hirsch, V.M.; Best, S.M.; Bouamr, F. From Capsids to Complexes: Expanding the Role of TRIM5α in the Restriction of Divergent RNA Viruses and Elements. Viruses 2021, 13, 446. [Google Scholar] [CrossRef]
- Morger, D.; Zosel, F.; Bühlmann, M.; Züger, S.; Mittelviefhaus, M.; Schuler, B.; Luban, J.; Grütter, M.G. The Three-Fold Axis of the HIV-1 Capsid Lattice Is the Species-Specific Binding Interface for TRIM5α. J. Virol. 2018, 92, e01541-17. [Google Scholar] [CrossRef] [PubMed]
- Medrano, L.M.; Rallón, N.; Berenguer, J.; Jiménez-Sousa, M.A.; Soriano, V.; Aldámiz-Echevarria, T.; Fernández-Rodríguez, A.; García, M.; Tejerina, F.; Martínez, I.; et al. Relationship of TRIM5 and TRIM22 Polymorphisms with Liver Disease and HCV Clearance after Antiviral Therapy in HIV/HCV Coinfected Patients. J. Transl. Med. 2016, 14, 257. [Google Scholar] [CrossRef] [PubMed]
- Mobasheri, S.; Irani, N.; Sepahi, A.A.; Sakhaee, F.; Jamnani, F.R.; Vaziri, F.; Siadat, S.D.; Fateh, A. Evaluation of TRIM5 and TRIM22 Polymorphisms on Treatment Responses in Iranian Patients with Chronic Hepatitis C Virus Infection. Gene 2018, 676, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Christensen, T. Association of Human Endogenous Retroviruses with Multiple Sclerosis and Possible Interactions with Herpes Viruses. Rev. Med. Virol. 2005, 15, 179–211. [Google Scholar] [CrossRef]
- Nexø, B.A.; Christensen, T.; Frederiksen, J.; Møller-Larsen, A.; Oturai, A.B.; Villesen, P.; Hansen, B.; Nissen, K.K.; Laska, M.J.; Petersen, T.S.; et al. The Etiology of Multiple Sclerosis: Genetic Evidence for the Involvement of the Human Endogenous Retrovirus HERV-Fc1. PLoS ONE 2011, 6, e16652. [Google Scholar] [CrossRef]
- Römer, C. Viruses and Endogenous Retroviruses as Roots for Neuroinflammation and Neurodegenerative Diseases. Front. Neurosci. 2021, 15, 648629. [Google Scholar] [CrossRef]
- Nexø, B.A.; Hansen, B.; Nissen, K.K.; Gundestrup, L.; Terkelsen, T.; Villesen, P.; Bahrami, S.; Petersen, T.; Pedersen, F.S.; Laska, M.J. Restriction Genes for Retroviruses Influence the Risk of Multiple Sclerosis. PLoS ONE 2013, 8, e74063. [Google Scholar] [CrossRef]
- Xu, W.K.; Byun, H.; Dudley, J.P. The Role of APOBECs in Viral Replication. Microorganisms 2020, 8, 1899. [Google Scholar] [CrossRef]
- Vieira, V.C.; Soares, M.A. The Role of Cytidine Deaminases on Innate Immune Responses against Human Viral Infections. BioMed. Res. Int. 2013, 2013, 1–18. [Google Scholar] [CrossRef]
- Sheehy, A.M.; Gaddis, N.C.; Choi, J.D.; Malim, M.H. Isolation of a Human Gene That Inhibits HIV-1 Infection and Is Suppressed by the Viral Vif Protein. Nature 2002, 418, 646–650. [Google Scholar] [CrossRef]
- Yu, Q.; Chen, D.; König, R.; Mariani, R.; Unutmaz, D.; Landau, N.R. APOBEC3B and APOBEC3C Are Potent Inhibitors of Simian Immunodeficiency Virus Replication. J. Biol. Chem. 2004, 279, 53379–53386. [Google Scholar] [CrossRef] [PubMed]
- Harris, R.S.; Dudley, J.P. APOBECs and Virus Restriction. Virology 2015, 479–480, 131–145. [Google Scholar] [CrossRef] [PubMed]
- Esnault, C.; Heidmann, O.; Delebecque, F.; Dewannieux, M.; Ribet, D.; Hance, A.J.; Heidmann, T.; Schwartz, O. APOBEC3G Cytidine Deaminase Inhibits Retrotransposition of Endogenous Retroviruses. Nature 2005, 433, 430–433. [Google Scholar] [CrossRef] [PubMed]
- Perez-Bercoff, D.; Laude, H.; Lemaire, M.; Hunewald, O.; Thiers, V.; Vignuzzi, M.; Blanc, H.; Poli, A.; Amoura, Z.; Caval, V.; et al. Sustained High Expression of Multiple APOBEC3 Cytidine Deaminases in Systemic Lupus Erythematosus. Sci. Rep. 2021, 11, 7893. [Google Scholar] [CrossRef]
- Mavragani, C.P.; Kirou, K.A.; Nezos, A.; Seshan, S.; Wild, T.; Wahl, S.M.; Moutsopoulos, H.M.; Crow, M.K. Expression of APOBEC Family Members as Regulators of Endogenous Retroelements and Malignant Transformation in Systemic Autoimmunity. Clin. Immunol. 2021, 223, 108649. [Google Scholar] [CrossRef]
- Calcraft, P.J.; Ruas, M.; Pan, Z.; Cheng, X.; Arredouani, A.; Hao, X.; Tang, J.; Rietdorf, K.; Teboul, L.; Chuang, K.-T.; et al. NAADP Mobilizes Calcium from Acidic Organelles through Two-Pore Channels. Nature 2009, 459, 596–600. [Google Scholar] [CrossRef]
- Grimm, C.; Chen, C.-C.; Wahl-Schott, C.; Biel, M. Two-Pore Channels: Catalyzers of Endolysosomal Transport and Function. Front. Pharmacol. 2017, 8, 45. [Google Scholar] [CrossRef]
- Sakurai, Y.; Kolokoltsov, A.A.; Chen, C.-C.; Tidwell, M.W.; Bauta, W.E.; Klugbauer, N.; Grimm, C.; Wahl-Schott, C.; Biel, M.; Davey, R.A. Ebola Virus. Two-Pore Channels Control Ebola Virus Host Cell Entry and Are Drug Targets for Disease Treatment. Science 2015, 347, 995–998. [Google Scholar] [CrossRef]
- Gunaratne, G.S.; Yang, Y.; Li, F.; Walseth, T.F.; Marchant, J.S. NAADP-Dependent Ca2+ Signaling Regulates Middle East Respiratory Syndrome-Coronavirus Pseudovirus Translocation through the Endolysosomal System. Cell Calcium 2018, 75, 30–41. [Google Scholar] [CrossRef]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of Spike Glycoprotein of SARS-CoV-2 on Virus Entry and Its Immune Cross-Reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620. [Google Scholar] [CrossRef]
- Dey-Rao, R.; Seiffert-Sinha, K.; Sinha, A.A. Genome-Wide Expression Analysis Suggests Unique Disease-Promoting and Disease-Preventing Signatures in Pemphigus Vulgaris. Genes Immun. 2013, 14, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Feng, C.; Fang, Y.; Zhou, X.; Xu, L.; Wang, W.; Kong, X.; Peppelenbosch, M.P.; Pan, Q.; Yin, Y. The Eukaryotic Translation Initiation Factor 4F Complex Restricts Rotavirus Infection via Regulating the Expression of IRF1 and IRF7. Int. J. Mol. Sci. 2019, 20, 1580. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.D.; Stintzing, S.; Heinemann, V.; Yang, D.; Cao, S.; Sunakawa, Y.; Ning, Y.; Matsusaka, S.; Okazaki, S.; Miyamoto, Y.; et al. Impact of Genetic Variations in the MAPK Signaling Pathway on Outcome in Metastatic Colorectal Cancer Patients Treated with First-Line FOLFIRI and Bevacizumab: Data from FIRE-3 and TRIBE Trials. Ann. Oncol. 2017, 28, 2780–2785. [Google Scholar] [CrossRef] [PubMed]
- Ho, J.J.D.; Lee, S. A Cap for Every Occasion: Alternative EIF4F Complexes. Trends Biochem. Sci. 2016, 41, 821–823. [Google Scholar] [CrossRef] [PubMed]
- Robert, F.; Cencic, R.; Cai, R.; Schmeing, T.M.; Pelletier, J. RNA-Tethering Assay and EIF4G:EIF4A Obligate Dimer Design Uncovers Multiple EIF4F Functional Complexes. Nucleic Acids Res. 2020, 48, 8562–8575. [Google Scholar] [CrossRef] [PubMed]
- Cencic, R.; Desforges, M.; Hall, D.R.; Kozakov, D.; Du, Y.; Min, J.; Dingledine, R.; Fu, H.; Vajda, S.; Talbot, P.J.; et al. Blocking EIF4E-EIF4G Interaction as a Strategy To Impair Coronavirus Replication. J. Virol. 2011, 85, 6381–6389. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.; Mohr, I. Phosphorylation of EIF4E by Mnk-1 Enhances HSV-1 Translation and Replication in Quiescent Cells. Genes Dev. 2004, 18, 660–672. [Google Scholar] [CrossRef]
- Walsh, D. Manipulation of the Host Translation Initiation Complex EIF4F by DNA Viruses. Biochem. Soc. Trans. 2010, 38, 1511–1516. [Google Scholar] [CrossRef]
- Yángüez, E.; Rodriguez, P.; Goodfellow, I.; Nieto, A. Influenza Virus Polymerase Confers Independence of the Cellular Cap-Binding Factor EIF4E for Viral MRNA Translation. Virology 2012, 422, 297–307. [Google Scholar] [CrossRef]
- Redondo, N.; García-Moreno, M.; Sanz, M.A.; Carrasco, L. Translation of Viral MRNAs That Do Not Require EIF4E Is Blocked by the Inhibitor 4EGI-1. Virology 2013, 444, 171–180. [Google Scholar] [CrossRef]
- Connor, J.H.; Lyles, D.S. Vesicular Stomatitis Virus Infection Alters the EIF4F Translation Initiation Complex and Causes Dephosphorylation of the EIF4E Binding Protein 4E-BP1. J. Virol. 2002, 76, 10177–10187. [Google Scholar] [CrossRef] [PubMed]
- Herdy, B.; Jaramillo, M.; Svitkin, Y.V.; Rosenfeld, A.B.; Kobayashi, M.; Walsh, D.; Alain, T.; Sean, P.; Robichaud, N.; Topisirovic, I.; et al. Translational Control of the Activation of Transcription Factor NF-ΚB and Production of Type I Interferon by Phosphorylation of the Translation Factor EIF4E. Nat. Immunol. 2012, 13, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Villas-Bôas, C.S.A.; Conceição, T.M.; Ramírez, J.; Santoro, A.B.M.; Da Poian, A.T.; Montero-Lomelí, M. Dengue Virus-Induced Regulation of the Host Cell Translational Machinery. Braz. J. Med. Biol. Res. 2009, 42, 1020–1026. [Google Scholar] [CrossRef] [PubMed]
- Ou, Y.; Enarson, P.; Rattner, J.B.; Barr, S.G.; Fritzler, M.J. The Nuclear Pore Complex Protein Tpr Is a Common Autoantigen in Sera That Demonstrate Nuclear Envelope Staining by Indirect Immunofluorescence. Clin. Exp. Immunol. 2004, 136, 379–387. [Google Scholar] [CrossRef]
- Enarson, P.; Rattner, J.B.; Ou, Y.; Miyachi, K.; Horigome, T.; Fritzler, M.J. Autoantigens of the Nuclear Pore Complex. J. Mol. Med. 2004, 82, 423–433. [Google Scholar] [CrossRef]
- Duarte-Rey, C.; Bogdanos, D.; Yang, C.-Y.; Roberts, K.; Leung, P.S.C.; Anaya, J.-M.; Worman, H.J.; Gershwin, M.E. Primary Biliary Cirrhosis and the Nuclear Pore Complex. Autoimmun. Rev. 2012, 11, 898–902. [Google Scholar] [CrossRef]
- The RACI Consortium; The GARNET Consortium; Okada, Y.; Wu, D.; Trynka, G.; Raj, T.; Terao, C.; Ikari, K.; Kochi, Y.; Ohmura, K.; et al. Genetics of Rheumatoid Arthritis Contributes to Biology and Drug Discovery. Nature 2014, 506, 376–381. [Google Scholar] [CrossRef]
- Laufer, V.A.; Tiwari, H.K.; Reynolds, R.J.; Danila, M.I.; Wang, J.; Edberg, J.C.; Kimberly, R.P.; Kottyan, L.C.; Harley, J.B.; Mikuls, T.R.; et al. Genetic Influences on Susceptibility to Rheumatoid Arthritis in African-Americans. Hum. Mol. Genet. 2019, 28, 858–874. [Google Scholar] [CrossRef]
- Le Sage, V.; Mouland, A. Viral Subversion of the Nuclear Pore Complex. Viruses 2013, 5, 2019–2042. [Google Scholar] [CrossRef]
- Wubben, J.M.; Atkinson, S.C.; Borg, N.A. The Role of Protein Disorder in Nuclear Transport and in Its Subversion by Viruses. Cells 2020, 9, 2654. [Google Scholar] [CrossRef]
- De Jesús-González, L.A.; Palacios-Rápalo, S.; Reyes-Ruiz, J.M.; Osuna-Ramos, J.F.; Cordero-Rivera, C.D.; Farfan-Morales, C.N.; Gutiérrez-Escolano, A.L.; del Ángel, R.M. The Nuclear Pore Complex Is a Key Target of Viral Proteases to Promote Viral Replication. Viruses 2021, 13, 706. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.W.; Mamede, J.I.; Hope, T.J. Impact of Nucleoporin-Mediated Chromatin Localization and Nuclear Architecture on HIV Integration Site Selection. J. Virol. 2015, 89, 9702–9705. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zheng, Y.; Yang, Z.; Jin, C.; Chen, C.; Wu, N. Hsa-MiR-191-5p Inhibits Replication of Human Immunodeficiency Virus Type 1 by Downregulating the Expression of NUP50. Arch. Virol. 2021, 166, 755–766. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Tang, Y.; He, F.; Zhang, Y.; Cheng, A.; Gan, R.; Wu, Y. Screening and Functional Analysis of Differentially Expressed Genes in EBV-Transformed Lymphoblasts. Virol. J. 2012, 9, 77. [Google Scholar] [CrossRef]
- Das, T.; Song, E.J.; Kim, E.E. The Multifaceted Roles of USP15 in Signal Transduction. Int. J. Mol. Sci. 2021, 22, 4728. [Google Scholar] [CrossRef]
- Pauli, E.-K.; Chan, Y.K.; Davis, M.E.; Gableske, S.; Wang, M.K.; Feister, K.F.; Gack, M.U. The Ubiquitin-Specific Protease USP15 Promotes RIG-I-Mediated Antiviral Signaling by Deubiquitylating TRIM25. Sci. Signal. 2014, 7, ra3. [Google Scholar] [CrossRef]
- Torre, S.; Polyak, M.J.; Langlais, D.; Fodil, N.; Kennedy, J.M.; Radovanovic, I.; Berghout, J.; Leiva-Torres, G.A.; Krawczyk, C.M.; Ilangumaran, S.; et al. USP15 Regulates Type I Interferon Response and Is Required for Pathogenesis of Neuroinflammation. Nat. Immunol. 2017, 18, 54–63. [Google Scholar] [CrossRef]
- Chiang, C.; Pauli, E.-K.; Biryukov, J.; Feister, K.F.; Meng, M.; White, E.A.; Münger, K.; Howley, P.M.; Meyers, C.; Gack, M.U. The Human Papillomavirus E6 Oncoprotein Targets USP15 and TRIM25 To Suppress RIG-I-Mediated Innate Immune Signaling. J. Virol. 2018, 92, e01737-17. [Google Scholar] [CrossRef]
- Pyeon, D.; Timani, K.A.; Gulraiz, F.; He, J.J.; Park, I.-W. Function of Ubiquitin (Ub) Specific Protease 15 (USP15) in HIV-1 Replication and Viral Protein Degradation. Virus Res. 2016, 223, 161–169. [Google Scholar] [CrossRef]
- Kusakabe, S.; Suzuki, T.; Sugiyama, Y.; Haga, S.; Horike, K.; Tokunaga, M.; Hirano, J.; Zhang, H.; Chen, D.V.; Ishiga, H.; et al. USP15 Participates in Hepatitis C Virus Propagation through Regulation of Viral RNA Translation and Lipid Droplet Formation. J. Virol. 2019, 93, e01708-18. [Google Scholar] [CrossRef]
- He, Z.; Wang, F.; Ma, J.; Sen, S.; Zhang, J.; Gwack, Y.; Zhou, Y.; Sun, Z. Ubiquitination of RORγt at Lysine 446 Limits Th17 Differentiation by Controlling Coactivator Recruitment. J. Immunol. 2016, 197, 1148–1158. [Google Scholar] [CrossRef] [PubMed]
- Paiva, I.A.; Badolato-Corrêa, J.; Familiar-Macedo, D.; de-Oliveira-Pinto, L.M. Th17 Cells in Viral Infections-Friend or Foe? Cells 2021, 10, 1159. [Google Scholar] [CrossRef] [PubMed]
- Wacleche, V.S.; Landay, A.; Routy, J.-P.; Ancuta, P. The Th17 Lineage: From Barrier Surfaces Homeostasis to Autoimmunity, Cancer, and HIV-1 Pathogenesis. Viruses 2017, 9, E303. [Google Scholar] [CrossRef] [PubMed]
- Fraternale, A.; Zara, C.; De Angelis, M.; Nencioni, L.; Palamara, A.T.; Retini, M.; Di Mambro, T.; Magnani, M.; Crinelli, R. Intracellular Redox-Modulated Pathways as Targets for Effective Approaches in the Treatment of Viral Infection. Int. J. Mol. Sci. 2021, 22, 3603. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.-H.; Liu, Q.; Xu, Z.-Y.; Li, J.-H.; Hu, Z.-X.; Li, M.-J.; Zheng, W.-L.; Li, Z.-J.; Pan, H.-W. Quantitative Proteomic Analysis of Human Corneal Epithelial Cells Infected with HSV-1. Exp. Eye Res. 2019, 185, 107664. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-J.; Luo, Y.-H.; Wan, S.-W.; Lin, C.-F.; Wang, S.-T.; Hung, N.T.; Liu, C.-C.; Ho, T.-S.; Liu, H.-S.; Yeh, T.-M.; et al. Correlation between Serum Levels of Anti-Endothelial Cell Autoantigen and Anti-Dengue Virus Nonstructural Protein 1 Antibodies in Dengue Patients. Am. J. Trop. Med. Hyg. 2015, 92, 989–995. [Google Scholar] [CrossRef]
- Fu, W.; Liu, Y.; Liu, F.; Liu, C.; Li, J.; Niu, J.; Han, P.; Xu, D.; Hou, J.; Ma, Y.; et al. A Novel Autoantibody Induced by Bacterial Biofilm Conserved Components Aggravates Lupus Nephritis. Front. Immunol. 2021, 12, 656090. [Google Scholar] [CrossRef]
- Bartosik-Psujek, H.; Stelmasiak, Z. The Levels of Chemokines CXCL8, CCL2 and CCL5 in Multiple Sclerosis Patients Are Linked to the Activity of the Disease. Eur. J. Neurol. 2005, 12, 49–54. [Google Scholar] [CrossRef]
- Liu, C.; Papewalis, C.; Domberg, J.; Scherbaum, W.; Schott, M. Chemokines and Autoimmune Thyroid Diseases. Horm. Metab. Res. 2008, 40, 361–368. [Google Scholar] [CrossRef]
- Cocchi, F.; DeVico, A.L.; Garzino-Demo, A.; Arya, S.K.; Gallo, R.C.; Lusso, P. Identification of RANTES, MIP-1α, and MIP-1β as the Major HIV-Suppressive Factors Produced by CD8+ T Cells. Science 1995, 270, 1811–1815. [Google Scholar] [CrossRef]
- Hudspeth, K.; Fogli, M.; Correia, D.V.; Mikulak, J.; Roberto, A.; Della Bella, S.; Silva-Santos, B.; Mavilio, D. Engagement of NKp30 on Vδ1 T Cells Induces the Production of CCL3, CCL4, and CCL5 and Suppresses HIV-1 Replication. Blood 2012, 119, 4013–4016. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.; Temerozo, J.R.; do Vale, G.; Ferreira, A.C.; Soares, V.C.; Dias, S.S.G.; Sardella, G.; Bou-Habib, D.C.; Siqueira, M.; Souza, T.M.L.; et al. The Chemokine CCL5 Inhibits the Replication of Influenza A Virus Through SAMHD1 Modulation. Front. Cell. Infect. Microbiol. 2021, 11, 549020. [Google Scholar] [CrossRef] [PubMed]
- Katsounas, A.; Schlaak, J.F.; Lempicki, R.A. CCL5: A Double-Edged Sword in Host Defense Against the Hepatitis C Virus. Int. Rev. Immunol. 2011, 30, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Li, M.-Z.; Pang, L.-L.; Bai, A.-Y.; Yu, S.-C.; Gong, X.; Liu, N.; Cai, K.; Xie, G.-C.; Gao, W.-J.; Jin, Y.; et al. Association of Chemotactic Chemokine Ligand 5 Polymorphisms with the Risk of Developing Severe Enterovirus 71 Infection. Am. J. Trop. Med. Hyg. 2015, 93, 709–713. [Google Scholar] [CrossRef]
- Sheng, Y.-F.; Qi, Q. Association of Chemotactic Chemokine Ligand 5 Rs2107538 Polymorphism with Tuberculosis Susceptibility: A Meta-Analysis. Innate Immun. 2020, 26, 358–363. [Google Scholar] [CrossRef]
- De Jager, P.L.; Jia, X.; Wang, J.; de Bakker, P.I.W.; Ottoboni, L.; Aggarwal, N.T.; Piccio, L.; Raychaudhuri, S.; Tran, D.; Aubin, C.; et al. Meta-Analysis of Genome Scans and Replication Identify CD6, IRF8 and TNFRSF1A as New Multiple Sclerosis Susceptibility Loci. Nat. Genet. 2009, 41, 776–782. [Google Scholar] [CrossRef]
- International Multiple Sclerosis Genetics Consortium. The Genetic Association of Variants in CD6, TNFRSF1A and IRF8 to Multiple Sclerosis: A Multicenter Case-Control Study. PLoS ONE 2011, 6, e18813. [Google Scholar] [CrossRef]
- Gorlova, O.; Martin, J.-E.; Rueda, B.; Koeleman, B.P.C.; Ying, J.; Teruel, M.; Diaz-Gallo, L.-M.; Broen, J.C.; Vonk, M.C.; Simeon, C.P.; et al. Identification of Novel Genetic Markers Associated with Clinical Phenotypes of Systemic Sclerosis through a Genome-Wide Association Strategy. PLoS Genet. 2011, 7, e1002178. [Google Scholar] [CrossRef]
- Arismendi, M.; Giraud, M.; Ruzehaji, N.; Dieudé, P.; Koumakis, E.; Ruiz, B.; Airo, P.; Cusi, D.; Matucci-Cerinic, M.; Salvi, E.; et al. Identification of NF-ΚB and PLCL2 as New Susceptibility Genes and Highlights on a Potential Role of IRF8 through Interferon Signature Modulation in Systemic Sclerosis. Arthritis Res. Ther. 2015, 17, 71. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, H.; Yu, H.; Li, L.; Xu, D.; Hou, S.; Kijlstra, A.; Yang, P. Two Genetic Variations in the IRF8 Region Are Associated with Behçet’s Disease in Han Chinese. Sci. Rep. 2016, 6, 19651. [Google Scholar] [CrossRef]
- Sezin, T.; Vorobyev, A.; Sadik, C.D.; Zillikens, D.; Gupta, Y.; Ludwig, R.J. Gene Expression Analysis Reveals Novel Shared Gene Signatures and Candidate Molecular Mechanisms between Pemphigus and Systemic Lupus Erythematosus in CD4+ T Cells. Front. Immunol. 2017, 8, 1992. [Google Scholar] [CrossRef] [PubMed]
- Stark, G.R.; Darnell, J.E. The JAK-STAT Pathway at Twenty. Immunity 2012, 36, 503–514. [Google Scholar] [CrossRef] [PubMed]
- Johnson, H.M.; Noon-Song, E.; Ahmed, C.M. Noncanonical IFN Signaling, Steroids, and STATs: A Probable Role of V-ATPase. Mediat. Inflamm. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Saevarsdottir, S.; Olafsdottir, T.A.; Ivarsdottir, E.V.; Halldorsson, G.H.; Gunnarsdottir, K.; Sigurdsson, A.; Johannesson, A.; Sigurdsson, J.K.; Juliusdottir, T.; Lund, S.H.; et al. FLT3 Stop Mutation Increases FLT3 Ligand Level and Risk of Autoimmune Thyroid Disease. Nature 2020, 584, 619–623. [Google Scholar] [CrossRef] [PubMed]
- International Multiple Sclerosis Genetics Consortium; Patsopoulos, N.A.; Baranzini, S.E.; Santaniello, A.; Shoostari, P.; Cotsapas, C.; Wong, G.; Beecham, A.H.; James, T.; Replogle, J.; et al. Multiple Sclerosis Genomic Map Implicates Peripheral Immune Cells and Microglia in Susceptibility. Science 2019, 365, eaav7188. [Google Scholar] [CrossRef]
- Sayed, K.S.; EL-Komy, M.H.M.; Shehata, H.; ElShazly, S.H.; El Desouky, E.D.; Amr, K.S.; ElAraby, N.M.; AlOrbani, A.M. JAK1 Rs310241 and JAK3 Rs3008 Genotypes May Increase Susceptibility to Psoriasis: A Case Control Study. Skin Pharmacol. Physiol. 2020, 33, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Hur, M.S.; Choi, B.G.; Kim, M.J.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. A Preliminary Study of New Single Polymorphisms in the T Helper Type 17 Pathway for Psoriasis in the Korean Population. Clin. Exp. Immunol. 2017, 187, 251–258. [Google Scholar] [CrossRef]
- Hou, S.; Qi, J.; Zhang, Q.; Liao, D.; Li, Q.; Hu, K.; Zhou, Y.; Kijlstra, A.; Yang, P. Genetic Variants in the JAK1 Gene Confer Higher Risk of Behcet’s Disease with Ocular Involvement in Han Chinese. Hum. Genet. 2013, 132, 1049–1058. [Google Scholar] [CrossRef]
- Hu, K.; Hou, S.; Li, F.; Xiang, Q.; Kijlstra, A.; Yang, P. JAK1, but Not JAK2 and STAT3, Confers Susceptibility to Vogt–Koyanagi–Harada (VKH) Syndrome in a Han Chinese Population. Investig. Opthalmology Vis. Sci. 2013, 54, 3360. [Google Scholar] [CrossRef][Green Version]
- Kichaev, G.; Bhatia, G.; Loh, P.-R.; Gazal, S.; Burch, K.; Freund, M.K.; Schoech, A.; Pasaniuc, B.; Price, A.L. Leveraging Polygenic Functional Enrichment to Improve GWAS Power. Am. J. Hum. Genet. 2019, 104, 65–75. [Google Scholar] [CrossRef]
- Yan, B.; Freiwald, T.; Chauss, D.; Wang, L.; West, E.; Mirabelli, C.; Zhang, C.J.; Nichols, E.-M.; Malik, N.; Gregory, R.; et al. SARS-CoV-2 Drives JAK1/2-Dependent Local Complement Hyperactivation. Sci. Immunol. 2021, 6, eabg0833. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Yang, F.; Wang, Q.; Xu, N.; Xie, Y.; Chen, S.; Qin, T.; Peng, D. Influenza a Virus Antagonizes Type I and Type II Interferon Responses via SOCS1-Dependent Ubiquitination and Degradation of JAK1. Virol. J. 2020, 17, 74. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Li, F.; Ji, J.; Liu, Y.; Liu, F.; Zhao, Y.; Li, J.; Han, S.; Wang, Q.; Ding, G. Inhibition of MiR-93 Promotes Interferon Effector Signaling to Suppress Influenza A Infection by Upregulating JAK1. Int. Immunopharmacol. 2020, 86, 106754. [Google Scholar] [CrossRef] [PubMed]
- Marshall, E.E.; Geballe, A.P. Multifaceted Evasion of the Interferon Response by Cytomegalovirus. J. Interferon Cytokine Res. 2009, 29, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; You, J.; You, H.; Zheng, C. Herpes Simplex Virus 1 UL36USP Antagonizes Type I Interferon-Mediated Antiviral Innate Immunity. J. Virol. 2018, 92, e01161-18. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, Y.; Yamaguchi, M.; Kohno, M.; Sakai, M.; Itoh, M.; Gotoh, B. Respirovirus C Protein Inhibits Activation of Type I Interferon Receptor-associated Kinases to Block JAK-STAT Signaling. FEBS Lett. 2020, 594, 864–877. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, S.; Martin, M.P.; Carrington, M. The Yin and Yang of HLA and KIR in Human Disease. Semin. Immunol. 2008, 20, 343–352. [Google Scholar] [CrossRef]
- Björkström, N.K.; Strunz, B.; Ljunggren, H.-G. Natural Killer Cells in Antiviral Immunity. Nat. Rev. Immunol. 2021, 22, 112–123. [Google Scholar] [CrossRef]
- Augusto, D.G.; Lobo-Alves, S.C.; Melo, M.F.; Pereira, N.F.; Petzl-Erler, M.L. Activating KIR and HLA Bw4 Ligands Are Associated to Decreased Susceptibility to Pemphigus Foliaceus, an Autoimmune Blistering Skin Disease. PLoS ONE 2012, 7, e39991. [Google Scholar] [CrossRef]
- Augusto, D.G.; O’Connor, G.M.; Lobo-Alves, S.C.; Bass, S.; Martin, M.P.; Carrington, M.; McVicar, D.W.; Petzl-Erler, M.L. Pemphigus Is Associated with KIR3DL2 Expression Levels and Provides Evidence That KIR3DL2 May Bind HLA-A3 and A11 in Vivo: Innate Immunity. Eur. J. Immunol. 2015, 45, 2052–2060. [Google Scholar] [CrossRef]
- Feng, H.; Zhang, Y.-B.; Gui, J.-F.; Lemon, S.M.; Yamane, D. Interferon Regulatory Factor 1 (IRF1) and Anti-Pathogen Innate Immune Responses. PLoS Pathog. 2021, 17, e1009220. [Google Scholar] [CrossRef] [PubMed]
- Schoggins, J.W.; Wilson, S.J.; Panis, M.; Murphy, M.Y.; Jones, C.T.; Bieniasz, P.; Rice, C.M. A Diverse Range of Gene Products Are Effectors of the Type I Interferon Antiviral Response. Nature 2011, 472, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Campos, R.K.; Wong, B.; Xie, X.; Lu, Y.-F.; Shi, P.-Y.; Pompon, J.; Garcia-Blanco, M.A.; Bradrick, S.S. RPLP1 and RPLP2 Are Essential Flavivirus Host Factors That Promote Early Viral Protein Accumulation. J. Virol. 2017, 91, e01706-16. [Google Scholar] [CrossRef] [PubMed]
- Lanza, A.; Cirillo, N.; Rossiello, R.; Rienzo, M.; Cutillo, L.; Casamassimi, A.; de Nigris, F.; Schiano, C.; Rossiello, L.; Femiano, F.; et al. Evidence of Key Role of Cdk2 Overexpression in Pemphigus Vulgaris. J. Biol. Chem. 2008, 283, 8736–8745. [Google Scholar] [CrossRef]
- Sarig, O.; Bercovici, S.; Zoller, L.; Goldberg, I.; Indelman, M.; Nahum, S.; Israeli, S.; Sagiv, N.; Martinez de Morentin, H.; Katz, O.; et al. Population-Specific Association between a Polymorphic Variant in ST18, Encoding a Pro-Apoptotic Molecule, and Pemphigus Vulgaris. J. Investig. Dermatol. 2012, 132, 1798–1805. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Arredondo, J.; Chernyavsky, A.I.; Kitajima, Y.; Pittelkow, M.; Grando, S.A. Pemphigus Vulgaris IgG and Methylprednisolone Exhibit Reciprocal Effects on Keratinocytes. J. Biol. Chem. 2004, 279, 2135–2146. [Google Scholar] [CrossRef]
| Endemic Pemphigus Foliaceus | Sporadic Pemphigus Foliaceus | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GENE | SNP | MAF (%) | MODEL | CTR | PAT | OR | 95% CI | p | MAF (%) | MODEL | CTR | PAT | OR | 95% CI | p | ||
| CTR | PAT | CTR | PAT | ||||||||||||||
| TRIM5 | rs4992800 | 7.25 | 14.64 | add | 28/360 | 65/379 | 2.21 | [1.37–3.56] | 0.0012 | 2.40 | 3.33 | add | 7/285 | 5/145 | 1.34 | [0.45–4.0] | 0.5941 |
| 11p15.4 | a > C | rec | 1/193 | 5/217 | 4.53 | [0.52–39.17] | 0.17 | rec | 1/145 | 0/75 | NA | NA | NA | ||||
| Intron 1 | dom | 27/167 | 60/162 | 2.30 | [1.38–3.86] | 0.0014 | dom | 6/140 | 5/70 | 1.67 | [0.49–5.65] | 0.4122 | |||||
| USP15 | rs10160955 | 22.16 | 13.66 | add | 86/302 | 62/392 | 0.56 | [0.39–0.81] | 0.0017 | 15.69 | 25.33 | add | 43/231 | 38/112 | 1.74 | [1.07–2.79] | 0.0234 |
| 12q14.1 | T > c | rec | 12/182 | 4/223 | 0.27 | [0.08–0.84] | 0.0246 | rec | 8/129 | 3/72 | 0.67 | [0.17–2.61] | 0.566 | ||||
| Intron 20 | dom | 74/120 | 58/169 | 0.17 | [0.00–17.43] | 0.4496 | dom | 35/102 | 35/40 | 2.55 | [1.40–4.62] | 0.0020 | |||||
| NUP37 | rs11111162 | 19.37 | 26.89 | add | 74/308 | 121/329 | 1.44 | [1.03–2.00] | 0.0292 | 23.08 | 19.33 | add | 66/220 | 29/121 | 0.80 | [0.49–1.30] | 0.3671 |
| 12q23.2 | A > g | rec | 4/187 | 22/203 | 4.80 | [1.62–14.3] | 0.0047 | rec | 7/136 | 3/72 | 0.81 | [0.20–3.22] | 0.7645 | ||||
| Intron 4 | dom | 70/121 | 99/126 | 1.28 | [0.86–1.91] | 0.2257 | dom | 59/84 | 26/49 | 0.75 | [0.42–1.35] | 0.3437 | |||||
| NUP50 | rs2138156 | 30.67 | 41.85 | add | 119/269 | 190/264 | 1.56 | [1.18–2.07] | 0.0018 | 30.14 | 38.00 | add | 88/204 | 57/93 | 1.40 | [0.93–2.10] | 0.1047 |
| 22q13.31 | C > t | rec | 19/175 | 45/182 | 2.29 | [1.28–4.07] | 0.0049 | rec | 13/133 | 13/62 | 2.14 | [0.94–4.90] | 0.0700 | ||||
| Intron 3 | dom | 100/94 | 145/82 | 1.62 | [1.09–2.41] | 0.0158 | dom | 75/71 | 44/31 | 1.34 | [0.76–2.36 | 0.3035 | |||||
| NUP88 | rs1058398 | 45.36 | 38.55 | add | 176/212 | 175/279 | 0.72 | [0.54–0.96] | 0.0259 | - | - | - | - | - | - | - | - |
| 17p13.2 | A > g | rec | 33/161 | 36/191 | 0.87 | [0.52–1.48] | 0.0517 | - | - | - | - | - | - | ||||
| 3′UTR | dom | 143/51 | 139/88 | 0.54 | [0.35–0.83] | 0.0046 | - | - | - | - | - | - | |||||
| TPR | rs3753565 | 4.40 | 9.47 | add | 17/369 | 43/411 | 2.43 | [1.35–4.4] | 0.0031 | 13.1 | 15.33 | add | 38/252 | 23/127 | 1.17 | [0.70–1.98] | 0.5491 |
| 1q31.1 | G > a Ser > Asn | rec | 1/192 | 2/225 | 1.33 | [0.11–15.94] | 0.8216 | rec | 4/141 | 4/71 | 1.99 | [0.48–8.17] | 0.3419 | ||||
| Exon 22 | dom | 16/177 | 41/186 | 2.68 | [1.44–4.99] | 0.0018 | dom | 34/111 | 19/56 | 1.11 | [0.58–2.11] | 0.7567 | |||||
| EIF4E | rs6834230 | 6.96 | 3.56 | add | 27/361 | 16/434 | 0.37 | [0.18–0.73] | 0.0045 | 0 | 0 | add | NA | NA | NA | NA | NA |
| 4q23 | C > t | rec | 1/193 | 0/225 | - | - | - | rec | NA | NA | NA | NA | NA | ||||
| Intron 1 | dom | 26/168 | 16/209 | 0.37 | [0.18–0.75] | 0.0057 | dom | NA | NA | NA | NA | NA | |||||
| EIF4E3 | rs1447904 | 27.32 | 36.73 | add | 106/282 | 166/286 | 1.49 | [1.10–2.00] | 0.0094 | 26.41 | 35.33 | add | 75/209 | 53/97 | 1.51 | [0.99–2.32] | 0.0573 |
| 3p13 | T > c | rec | 17/177 | 29/197 | 1.40 | [0.73–2.66] | 0.308 | rec | 9/133 | 11/64 | 2.54 | [1.00–6.44] | 0.0495 | ||||
| Intron 2 | dom | 89/105 | 137/89 | 1.76 | [1.19–2.60] | 0.0046 | dom | 66/76 | 42/33 | 1.47 | [0.83–2.57] | 0.183 | |||||
| JAK1 | rs310199 | 41.75 | 49.12 | add | 162/266 | 231/223 | 1.39 | [1.05–1.83] | 0.0205 | 28.67 | 27.7 | add | 82/204 | 41/107 | 0.95 | [0.60–1.67] | 0.8229 |
| 1p31.3 | A > g | rec | 39/155 | 56/171 | 1.23 | [0.77–1.97] | 0.3881 | rec | 10/133 | 3/71 | 0.56 | [0.15–2.10] | 0.3929 | ||||
| Intron 2 | dom | 123/71 | 175/52 | 1.86 | [1.21–2.86] | 0.0046 | dom | 39/155 | 56/171 | 1.04 | [0.59–1.82] | 0.8887 | |||||
| rs310202 | 30.05 | 39.6 | add | 116/270 | 179/273 | 1.48 | [1.10–2.0] | 0.0102 | 20.63 | 20.67 | add | 59/227 | 31/119 | 1.00 | [0.60–1.67] | 0.9923 | |
| A > g | rec | 19/174 | 31/195 | 1.35 | [0.73–2.50] | 0.3382 | rec | 4/139 | 2/73 | 0.95 | [0.17–5.32] | 0.9554 | |||||
| Intron 2 | dom | 97/96 | 148/78 | 1.781 | [1.20–2.65] | 0.0045 | dom | 55/88 | 29/46 | 1.00 | [0.57–1.79] | 0.9764 | |||||
| TPCN2 | rs4930263 | 2.85 | 8.62 | add | 11/375 | 39/413 | 2.76 | [1.41–5.40] | 0.0031 | 0 | 0.68 | add | 0/276 | 1/145 | NA | NA | NA |
| 11q13.3 | a > C | rec | 1/192 | 4/222 | 3.01 | [0.33–27.5] | 0.328 | rec | 0/138 | 0/73 | NA | NA | NA | ||||
| Intron 16 | dom | 10/183 | 35/191 | 3.20 | [1.53–6.69] | 0.0019 | dom | 0/138 | 1/72 | NA | NA | NA | |||||
| IRF8 | rs1044873 | 42.27 | 36.5 | add | 164/224 | 165/287 | 0.76 | [0.57–1.02] | 0.0644 | 41.9 | 38.5 | add | 119/165 | 57/91 | 0.87 | [0.58–1.30] | 0.5014 |
| 16q24.1 | C > t | rec | 39/155 | 23/203 | 0.42 | [0.24–0.73] | 0.0024 | rec | 26/116 | 11/63 | 0.78 | [0.33–0.68] | 0.5243 | ||||
| 3′ UTR | dom | 125/69 | 142/84 | 0.93 | [0.62–1.39] | 0.7186 | dom | 93/49 | 46/28 | 0.87 | [0.48–1.55] | 0.6278 | |||||
| Genes | p | Fold Change |
|---|---|---|
| CCL5 | 3.885 × 10−06 | 1.4362 |
| P4HB | 3.730 × 10−05 | 0.4185 |
| APOBEC3G | 4.212 × 10−05 | 0.5244 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoch, V.B.-B.; Kohler, A.F.; Augusto, D.G.; Lobo-Alves, S.C.; Malheiros, D.; Cipolla, G.A.; Boldt, A.B.W.; Braun-Prado, K.; Wittig, M.; Franke, A.; et al. Genetic Associations and Differential mRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus. Viruses 2022, 14, 879. https://doi.org/10.3390/v14050879
Hoch VB-B, Kohler AF, Augusto DG, Lobo-Alves SC, Malheiros D, Cipolla GA, Boldt ABW, Braun-Prado K, Wittig M, Franke A, et al. Genetic Associations and Differential mRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus. Viruses. 2022; 14(5):879. https://doi.org/10.3390/v14050879
Chicago/Turabian StyleHoch, Valéria Bumiller-Bini, Ana Flávia Kohler, Danillo G. Augusto, Sara Cristina Lobo-Alves, Danielle Malheiros, Gabriel Adelman Cipolla, Angelica Beate Winter Boldt, Karin Braun-Prado, Michael Wittig, Andre Franke, and et al. 2022. "Genetic Associations and Differential mRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus" Viruses 14, no. 5: 879. https://doi.org/10.3390/v14050879
APA StyleHoch, V. B.-B., Kohler, A. F., Augusto, D. G., Lobo-Alves, S. C., Malheiros, D., Cipolla, G. A., Boldt, A. B. W., Braun-Prado, K., Wittig, M., Franke, A., Pföhler, C., Worm, M., van Beek, N., Goebeler, M., Sárdy, M., Ibrahim, S., Busch, H., Schmidt, E., Hundt, J. E., ... Petzl-Erler, M. L. (2022). Genetic Associations and Differential mRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus. Viruses, 14(5), 879. https://doi.org/10.3390/v14050879







