Comparative HIV-1 Proviral Dynamics in Two Individuals That Maintained Viral Replication Control with or without Antiretroviral Therapy following Superinfection
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Subjects
2.1.1. CD4+ T- Cell Counts and Plasma HIV-1 RNA Quantification
2.1.2. Genomic DNA Isolation and Single Genome Amplification (SGA) and Sequencing
2.2. Sequence Analysis
2.3. Statistical Analysis
2.4. Availability of Data
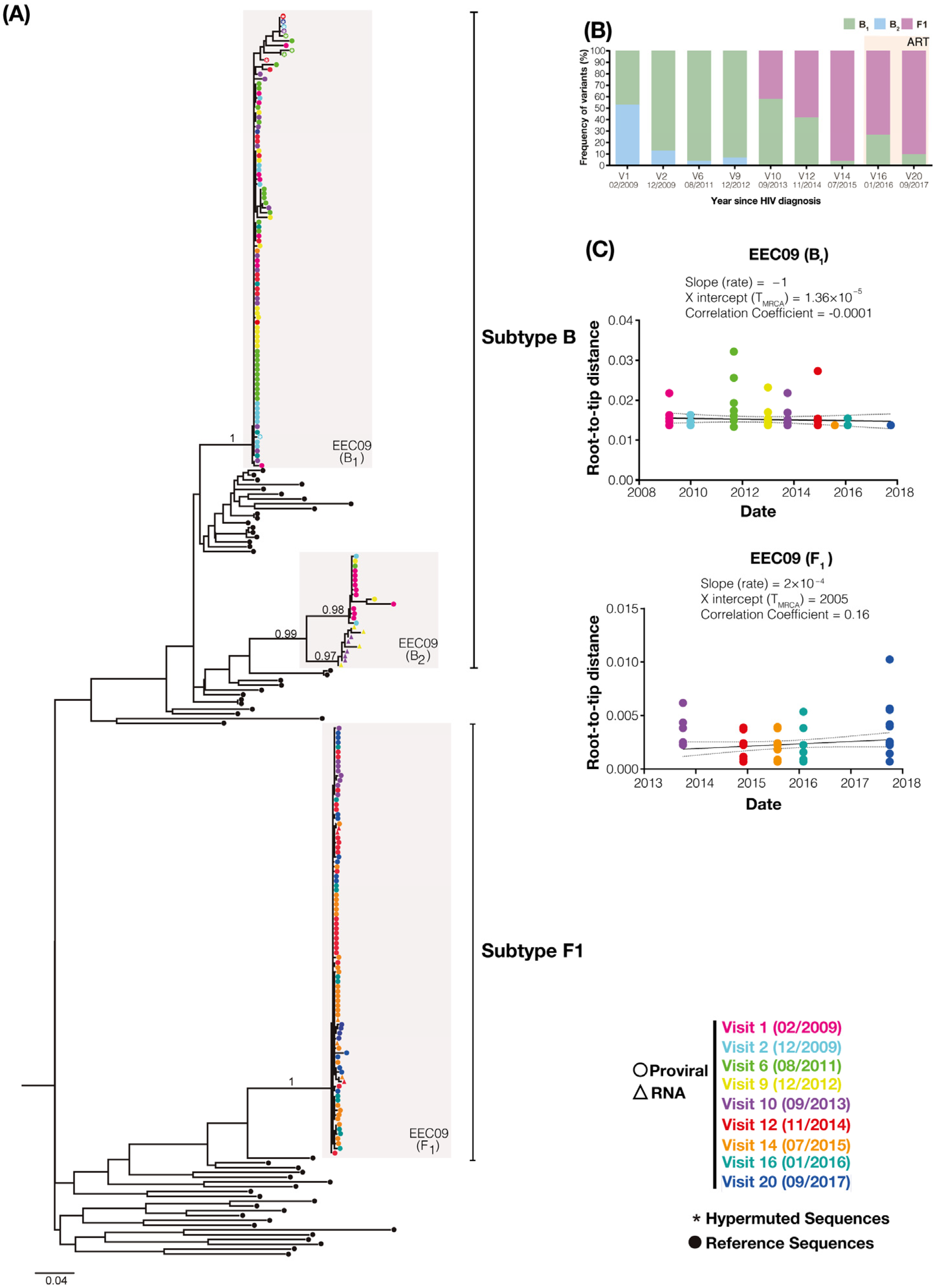
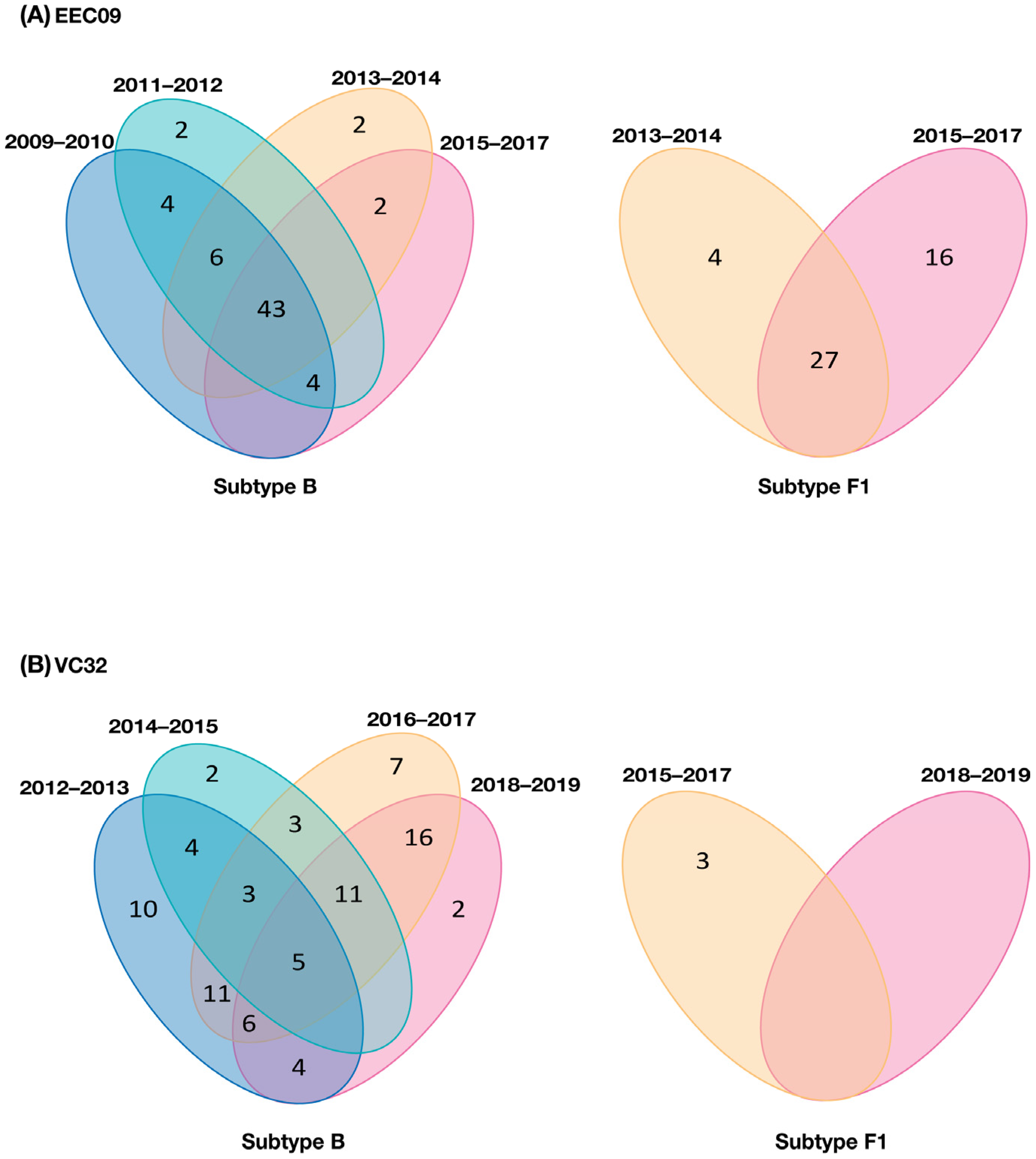
3. Results
3.1. Clinical Aspects of Patients
3.2. Diversity and Evolution of DNA Viral Quasispecies Associated with PBMC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chun, T.-W.; Carruth, L.; Finzi, D.; Shen, X.; DiGiuseppe, J.A.; Taylor, H.; Hermankova, M.; Chadwick, K.; Margolick, J.; Quinn, T.C.; et al. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature 1997, 387, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Chun, T.W.; Finzi, D.; Margolick, J.; Chadwick, K.; Schwartz, D.; Siliciano, R.F. In vivo fate of HIV-1-infected T cells: Quantitative analysis of the transition to stable latency. Nat. Med. 1995, 1, 1284–1290. [Google Scholar] [CrossRef] [PubMed]
- Deeks, S.G.; Walker, B.D. Human Immunodeficiency Virus Controllers: Mechanisms of Durable Virus Control in the Absence of Antiretroviral Therapy. Immunity 2007, 27, 406–416. [Google Scholar] [CrossRef] [PubMed]
- Deeks, S.G.; Archin, N.; Cannon, P.; Collins, S.; Jones, R.B.; de Jong, M.A.W.P.; Lambotte, O.; Lamplough, R.; Ndung’u, T.; Sugarman, J.; et al. Research priorities for an HIV cure: International AIDS Society Global Scientific Strategy 2021. Nat. Med. 2021, 27, 2085–2098. [Google Scholar] [CrossRef]
- de Azevedo, S.S.D.; Delatorre, E.; Côrtes, F.H.; Hoagland, B.; Grinsztejn, B.; Veloso, V.G.; Souza, T.M.L.; Morgado, M.G.; Bello, G. HIV controllers suppress viral replication and evolution and prevent disease progression following intersubtype HIV-1 superinfection. AIDS 2019, 33, 399–410. [Google Scholar] [CrossRef] [PubMed]
- de Azevedo, S.S.D.; Caetano, D.G.; Côrtes, F.H.; Teixeira, S.L.M.; Dos Santos Silva, K.; Hoagland, B.; Grinsztejn, B.; Veloso, V.G.; Morgado, M.G.; Bello, G. Highly divergent patterns of genetic diversity and evolution in proviral quasispecies from HIV controllers. Retrovirology 2017, 14, 29. [Google Scholar] [CrossRef]
- Rose, P.P.; Korber, B.T. Detecting hypermutations in viral sequences with an emphasis on G —> A hypermutation. Bioinformatics 2000, 16, 400–401. [Google Scholar] [CrossRef]
- Guindon, S.; Lethiec, F.; Duroux, P.; Gascuel, O. PHYML Online—A web server for fast maximum likelihood-based phylogenetic inference. Nucleic Acids Res. 2005, 33, W557–W559. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: More models, new heuristics and parallel computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef]
- Anisimova, M.; Gascuel, O. Approximate likelihood-ratio test for branches: A fast, accurate, and powerful alternative. Syst. Biol. 2006, 55, 539–552. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A.; Lam, T.T.; Max Carvalho, L.; Pybus, O.G. Exploring the temporal structure of heterochronous sequences using TempEst (formerly Path-O-Gen). Virus Evol. 2016, 2, vew007. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Gaschen, B.; Blay, W.; Foley, B.; Haigwood, N.; Kuiken, C.; Korber, B. Tracking global patterns of N-linked glycosylation site variation in highly variable viral glycoproteins: HIV, SIV, and HCV envelopes and influenza hemagglutinin. Glycobiology 2004, 14, 1229–1246. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Decker, J.M.; Wang, S.; Hui, H.; Kappes, J.C.; Wu, X.; Salazar-Gonzalez, J.F.; Salazar, M.G.; Kilby, J.M.; Saag, M.S.; et al. Antibody neutralization and escape by HIV-1. Nature 2003, 422, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tan, S.; Zhang, C.; Chai, Y.; He, M.; Zhang, C.W.H.; Wang, Q.; Tong, Z.; Liu, K.; Lei, Y.; et al. Limited Cross-Linking of 4-1BB by 4-1BB Ligand and the Agonist Monoclonal Antibody Utomilumab. Cell Rep. 2018, 25, 909–920.e4. [Google Scholar] [CrossRef] [PubMed]
- Bunnik, E.M.; Pisas, L.; van Nuenen, A.C.; Schuitemaker, H. Autologous Neutralizing Humoral Immunity and Evolution of the Viral Envelope in the Course of Subtype B Human Immunodeficiency Virus Type 1 Infection. J. Virol. 2008, 82, 7932–7941. [Google Scholar] [CrossRef]
- Wagh, K.; Kreider, E.F.; Li, Y.; Barbian, H.J.; Learn, G.H.; Giorgi, E.; Hraber, P.T.; Decker, T.G.; Smith, A.G.; Gondim, M.V.; et al. Completeness of HIV-1 Envelope Glycan Shield at Transmission Determines Neutralization Breadth. Cell Rep. 2018, 25, 893–908.e7. [Google Scholar] [CrossRef]
- Jones, B.R.; Kinloch, N.N.; Horacsek, J.; Ganase, B.; Harris, M.; Harrigan, P.R.; Jones, R.B.; Brockman, M.A.; Joy, J.B.; Poon, A.F.Y.; et al. Phylogenetic approach to recover integration dates of latent HIV sequences within-host. Proc. Natl. Acad. Sci. USA 2018, 115, E8958–E8967. [Google Scholar] [CrossRef]
- Brooks, K.; Jones, B.R.; Dilernia, D.A.; Wilkins, D.J.; Claiborne, D.T.; McInally, S.; Gilmour, J.; Kilembe, W.; Joy, J.B.; Allen, S.A.; et al. HIV-1 variants are archived throughout infection and persist in the reservoir. PLoS Pathog. 2020, 16, e1008378. [Google Scholar] [CrossRef]
- Brodin, J.; Zanini, F.; Thebo, L.; Lanz, C.; Bratt, G.; Neher, R.A.; Albert, J. Establishment and stability of the latent HIV-1 DNA reservoir. eLife 2016, 5, e18889. [Google Scholar] [CrossRef]
- Abrahams, M.-R.; Joseph, S.B.; Garrett, N.; Tyers, L.; Moeser, M.; Archin, N.; Council, O.D.; Matten, D.; Zhou, S.; Doolabh, D.; et al. The replication-competent HIV-1 latent reservoir is primarily established near the time of therapy initiation. Sci. Transl. Med. 2019, 11, eaaw5589. [Google Scholar] [CrossRef] [PubMed]
- Pankau, M.D.; Reeves, D.B.; Harkins, E.; Ronen, K.; Jaoko, W.; Mandaliya, K.; Graham, S.M.; McClelland, R.S.; Matsen, F.A.; Schiffer, J.T.; et al. Dynamics of HIV DNA reservoir seeding in a cohort of superinfected Kenyan women. PLoS Pathog. 2020, 16, e1008286. [Google Scholar] [CrossRef] [PubMed]
- Omondi, F.H.; Sudderuddin, H.; Shahid, A.; Kinloch, N.N.; Jones, B.R.; Miller, R.L.; Tsai, O.; MacMillan, D.; Trocha, A.; Brockman, M.A.; et al. HIV Proviral Burden, Genetic Diversity, and Dynamics in Viremic Controllers Who Subsequently Initiated Suppressive Antiretroviral Therapy. MBio 2021, 12, e0249021. [Google Scholar] [CrossRef] [PubMed]
- Maldarelli, F.; Wu, X.; Su, L.; Simonetti, F.R.; Shao, W.; Hill, S.; Spindler, J.; Ferris, A.L.; Mellors, J.W.; Kearney, M.F.; et al. HIV latency. Specific HIV integration sites are linked to clonal expansion and persistence of infected cells. Science 2014, 345, 179–183. [Google Scholar] [CrossRef] [PubMed]
- Wagner, T.A.; McLaughlin, S.; Garg, K.; Cheung, C.Y.K.; Larsen, B.B.; Styrchak, S.; Huang, H.C.; Edlefsen, P.T.; Mullins, J.I.; Frenkel, L.M. HIV latency. Proliferation of cells with HIV integrated into cancer genes contributes to persistent infection. Science 2014, 345, 570–573. [Google Scholar] [CrossRef] [PubMed]
- Cohn, L.B.; Silva, I.T.; Oliveira, T.Y.; Rosales, R.A.; Parrish, E.H.; Learn, G.H.; Hahn, B.H.; Czartoski, J.L.; McElrath, M.J.; Lehmann, C.; et al. HIV-1 integration landscape during latent and active infection. Cell 2015, 160, 420–432. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.Q.; Orlova-Fink, N.; Einkauf, K.; Chowdhury, F.Z.; Sun, X.; Harrington, S.; Kuo, H.-H.; Hua, S.; Chen, H.-R.; Ouyang, Z.; et al. Clonal expansion of genome-intact HIV-1 in functionally polarized Th1 CD4+ T cells. J. Clin. Investig. 2017, 127, 2689–2696. [Google Scholar] [CrossRef] [PubMed]
- Simonetti, F.R.; Sobolewski, M.D.; Fyne, E.; Shao, W.; Spindler, J.; Hattori, J.; Anderson, E.M.; Watters, S.A.; Hill, S.; Wu, X.; et al. Clonally expanded CD4+ T cells can produce infectious HIV-1 in vivo. Proc. Natl. Acad. Sci. USA 2016, 113, 1883–1888. [Google Scholar] [CrossRef] [PubMed]
- Bui, J.K.; Sobolewski, M.D.; Keele, B.F.; Spindler, J.; Musick, A.; Wiegand, A.; Luke, B.T.; Shao, W.; Hughes, S.H.; Coffin, J.M.; et al. Proviruses with identical sequences comprise a large fraction of the replication-competent HIV reservoir. PLoS Pathog. 2017, 13, e1006283. [Google Scholar] [CrossRef]
- Reeves, D.B.; Duke, E.R.; Wagner, T.A.; Palmer, S.E.; Spivak, A.M.; Schiffer, J.T. A majority of HIV persistence during antiretroviral therapy is due to infected cell proliferation. Nat. Commun. 2018, 9, 4811. [Google Scholar] [CrossRef] [PubMed]
- Patro, S.C.; Brandt, L.D.; Bale, M.J.; Halvas, E.K.; Joseph, K.W.; Shao, W.; Wu, X.; Guo, S.; Murrell, B.; Wiegand, A.; et al. Combined HIV-1 sequence and integration site analysis informs viral dynamics and allows reconstruction of replicating viral ancestors. Proc. Natl. Acad. Sci. USA 2019, 116, 25891–25899. [Google Scholar] [CrossRef] [PubMed]
- Goonetilleke, N.; Clutton, G.; Swanstrom, R.; Joseph, S.B. Blocking Formation of the Stable HIV Reservoir: A New Perspective for HIV-1 Cure. Front. Immunol. 2019, 10, 1966. [Google Scholar] [CrossRef]
- Koning, F.A.; Badhan, A.; Shaw, S.; Fisher, M.; Mbisa, J.L.; Cane, P.A. Dynamics of HIV type 1 recombination following superinfection. AIDS Res. Hum. Retrovir. 2013, 29, 963–970. [Google Scholar] [CrossRef]
- Gao, Y.; He, S.; Tian, W.; Li, D.; An, M.; Zhao, B.; Ding, H.; Xu, J.; Chu, Z.; Shang, H.; et al. First complete-genome documentation of HIV-1 intersubtype superinfection with transmissions of diverse recombinants over time to five recipients. PLoS Pathog. 2021, 17, e1009258. [Google Scholar] [CrossRef] [PubMed]
- Lian, X.; Gao, C.; Sun, X.; Jiang, C.; Einkauf, K.B.; Seiger, K.W.; Chevalier, J.M.; Yuki, Y.; Martin, M.; Hoh, R.; et al. Signatures of immune selection in intact and defective proviruses distinguish HIV-1 elite controllers. Sci. Transl. Med. 2021, 13, eabl4097. [Google Scholar] [CrossRef]
- Ho, Y.S.; Abecasis, A.B.; Theys, K.; Deforche, K.; Dwyer, D.E.; Charleston, M.; Vandamme, A.M.; Saksena, N.K. HIV-1 gp120 N-linked glycosylation differs between plasma and leukocyte compartments. Virol. J. 2008, 5, 14. [Google Scholar] [CrossRef]
- Curlin, M.E.; Zioni, R.; Hawes, S.E.; Liu, Y.; Deng, W.; Gottlieb, G.S.; Zhu, T.; Mullins, J.I. HIV-1 envelope subregion length variation during disease progression. PLoS Pathog. 2010, 6, e1001228. [Google Scholar] [CrossRef] [PubMed]
- Dapp, M.J.; Kober, K.M.; Chen, L.; Westfall, D.H.; Wong, K.; Zhao, H.; Hall, B.M.; Deng, W.; Sibley, T.; Ghorai, S.; et al. Patterns and rates of viral evolution in HIV-1 subtype B infected females and males. PLoS ONE 2017, 12, e0182443. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Nie, J.; Prochnow, C.; Truong, C.; Jia, Z.; Wang, S.; Chen, X.S.; Wang, Y. A systematic study of the N-glycosylation sites of HIV-1 envelope protein on infectivity and antibody-mediated neutralization. Retrovirology 2013, 10, 14. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Azevedo, S.S.D.; Côrtes, F.H.; Villela, L.M.; Hoagland, B.; Grinsztejn, B.; Veloso, V.G.; Morgado, M.G.; Bello, G. Comparative HIV-1 Proviral Dynamics in Two Individuals That Maintained Viral Replication Control with or without Antiretroviral Therapy following Superinfection. Viruses 2022, 14, 2802. https://doi.org/10.3390/v14122802
de Azevedo SSD, Côrtes FH, Villela LM, Hoagland B, Grinsztejn B, Veloso VG, Morgado MG, Bello G. Comparative HIV-1 Proviral Dynamics in Two Individuals That Maintained Viral Replication Control with or without Antiretroviral Therapy following Superinfection. Viruses. 2022; 14(12):2802. https://doi.org/10.3390/v14122802
Chicago/Turabian Stylede Azevedo, Suwellen Sardinha Dias, Fernanda H. Côrtes, Larissa M. Villela, Brenda Hoagland, Beatriz Grinsztejn, Valdilea G. Veloso, Mariza G. Morgado, and Gonzalo Bello. 2022. "Comparative HIV-1 Proviral Dynamics in Two Individuals That Maintained Viral Replication Control with or without Antiretroviral Therapy following Superinfection" Viruses 14, no. 12: 2802. https://doi.org/10.3390/v14122802
APA Stylede Azevedo, S. S. D., Côrtes, F. H., Villela, L. M., Hoagland, B., Grinsztejn, B., Veloso, V. G., Morgado, M. G., & Bello, G. (2022). Comparative HIV-1 Proviral Dynamics in Two Individuals That Maintained Viral Replication Control with or without Antiretroviral Therapy following Superinfection. Viruses, 14(12), 2802. https://doi.org/10.3390/v14122802





