SARS CoV-2 Infection among Health Care Workers from Different Health Care Facilities in Western Norway: A Prospective, Cross-Sectional Study
Abstract
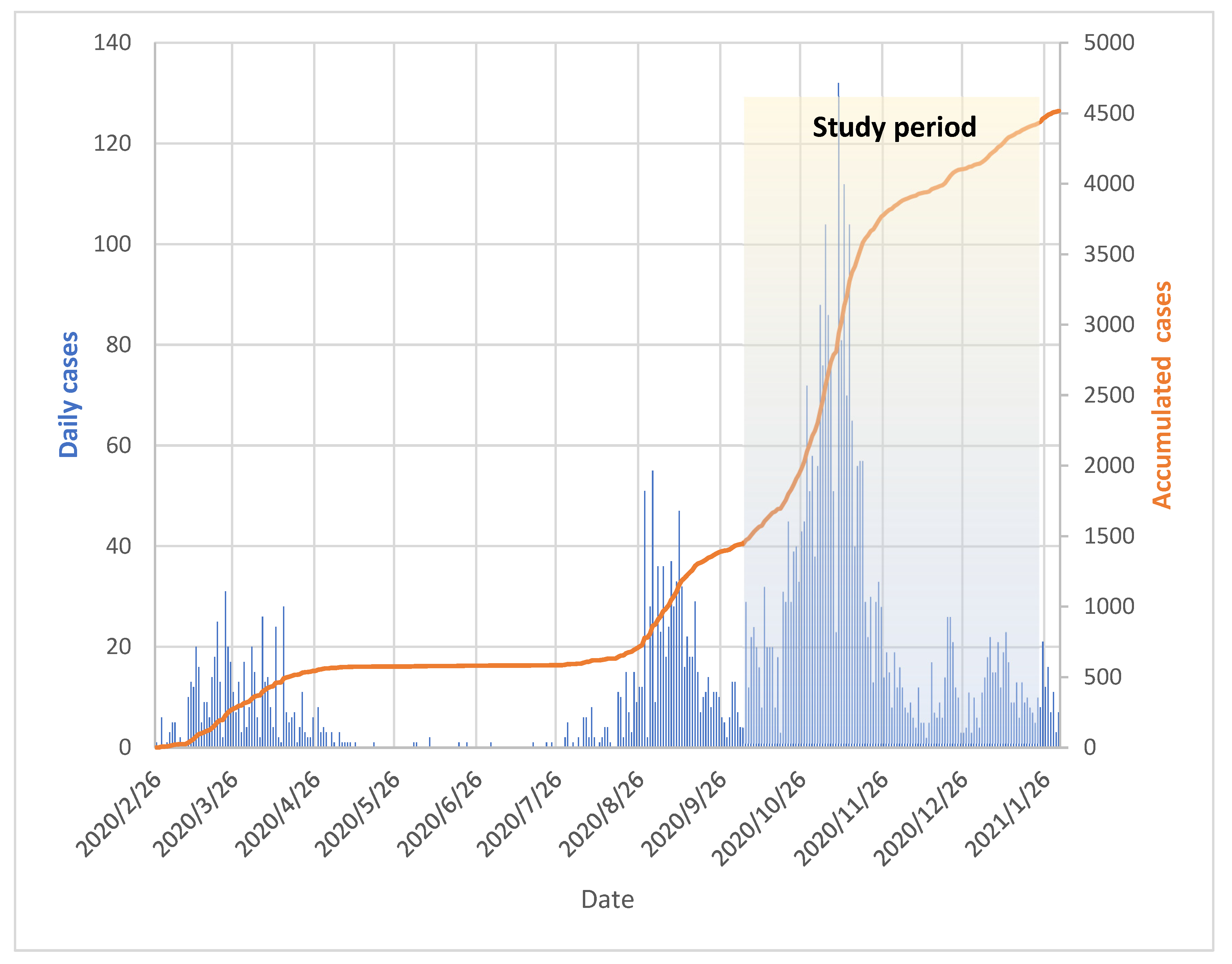
:1. Introduction
2. Materials and Methods
2.1. Study Population and Clinical Data
2.2. Laboratory Methods
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grant, J.J.; Wilmore, S.M.S.; McCann, N.S.; Donnelly, O.; Lai, R.W.L.; Kinsella, M.J.; Rochford, H.L.; Patel, T.; Kelsey, M.C.; Andrews, J.A. Seroprevalence of SARS-CoV-2 antibodies in healthcare workers at a London NHS Trust. Infect. Control Hosp. Epidemiol. 2021, 42, 212–214. [Google Scholar] [CrossRef] [PubMed]
- Rudberg, A.S.; Havervall, S.; Månberg, A.; Jernbom Falk, A.; Aguilera, K.; Ng, H.; Gabrielsson, L.; Salomonsson, A.C.; Hanke, L.; Murrell, B.; et al. SARS-CoV-2 exposure, symptoms and seroprevalence in healthcare workers in Sweden. Nat. Commun. 2020, 11, 5064. [Google Scholar] [PubMed]
- McGarry, B.E.; Grabowski, D.C.; Barnett, M.L. Severe Staffing and Personal Protective Equipment Shortages Faced by Nursing Homes during the COVID-19 Pandemic. Health Aff. 2020, 39, 1812–1821. [Google Scholar] [CrossRef] [PubMed]
- White, E.M.; Wetle, T.F.; Reddy, A.; Baier, R.R. Front-line Nursing Home Staff Experiences during the COVID-19 Pandemic. J. Am. Med. Dir. Assoc. 2021, 22, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.S.; Charland, K.; Quach, C.; Nguyen, Q.D.; Zinszer, K. Institutional, therapeutic, and individual factors associated with 30-day mortality after COVID-19 diagnosis in Canadian long-term care facilities. J. Am. Geriatr. Soc. 2022, 70, 3210–3220. [Google Scholar] [CrossRef] [PubMed]
- Blain, H.; Rolland, Y.; Benetos, A.; Giacosa, N.; Albrand, M.; Miot, S.; Bousquet, J. Atypical clinical presentation of COVID-19 infection in residents of a long-term care facility. Eur. Geriatr. Med. 2020, 11, 1085–1088. [Google Scholar] [CrossRef] [PubMed]
- Moscola, J.; Sembajwe, G.; Jarrett, M.; Farber, B.; Chang, T.; McGinn, T.; Davidson, K.W. Prevalence of SARS-CoV-2 Antibodies in Health Care Personnel in the New York City Area. JAMA 2020, 324, 893–895. [Google Scholar] [CrossRef] [PubMed]
- Plebani, M.; Padoan, A.; Fedeli, U.; Schievano, E.; Vecchiato, E.; Lippi, G.; Lo Cascio, G.; Porru, S.; Palù, G. SARS-CoV-2 serosurvey in health care workers of the Veneto Region. Clin. Chem. Lab. Med. 2020, 58, 2107–2111. [Google Scholar] [CrossRef]
- Lackermair, K.; William, F.; Grzanna, N.; Lehmann, E.; Fichtner, S.; Kucher, H.B.; Wilhelm, K.; Estner, H. Infection with SARS-CoV-2 in primary care health care workers assessed by antibody testing. Fam. Pract. 2021, 38, 76–79. [Google Scholar] [PubMed]
- Hussein, R.; Lami, F. Seroprevalence of COVID-19 Among Health Care Workers in Primary Health Care Centers in Al-Sader City District, Baghdad, Iraq. JMIR Publ. 2022, 8, e36473. [Google Scholar] [CrossRef]
- Baker, J.M.; Nelson, K.N.; Overton, E.; Lopman, B.A.; Lash, T.L.; Photakis, M.; Jacob, J.T.; Roback, J.D.; Fridkin, S.K.; Steinberg, J.P. Quantification of Occupational and Community Risk Factors for SARS-CoV-2 Seropositivity among Health Care Workers in a Large U.S. Health Care System. Ann. Intern. Med. 2021, 174, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Zangoue, M.; Safari, H.; Royce, S.G.; Zangooie, A.; Rezapour, H.; Zangouei, A.; Fereidouni, M. The high level of adherence to personal protective equipment in health care workers efficiently protects them from COVID-19 infection. Work 2021, 69, 1191–1196. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hayakawa, K.; Ainai, A.; Iwata-Yoshikawa, N.; Sano, K.; Nagata, N.; Suzuki, T.; Wakimoto, Y.; Akiyama, Y.; Miyazato, Y.; et al. Effectiveness of personal protective equipment in preventing severe acute respiratory syndrome coronavirus 2 infection among healthcare workers. J. Infect. Chemother. 2021, 27, 120–122. [Google Scholar] [CrossRef]
- Trieu, M.C.; Bansal, A.; Madsen, A.; Zhou, F.; Sævik, M.; Vahokoski, J.; Brokstad, K.A.; Krammer, F.; Tøndel, C.; Mohn, K.G.I.; et al. SARS-CoV-2-Specific Neutralizing Antibody Responses in Norwegian Health Care Workers after the First Wave of COVID-19 Pandemic: A Prospective Cohort Study. J. Infect Dis. 2021, 223, 589–599. [Google Scholar] [CrossRef]
- Brainard, J.; Rushton, S.; Winters, T.; Hunter, P.R. Introduction to and spread of COVID-19-like illness in care homes in Norfolk, UK. J. Public Health 2021, 43, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Galanis, P.; Vraka, I.; Fragkou, D.; Bilali, A.; Kaitelidou, D. Seroprevalence of SARS-CoV-2 antibodies and associated factors in healthcare workers: A systematic review and meta-analysis. J. Hosp. Infect. 2021, 108, 120–134. [Google Scholar] [CrossRef]
- Arons, M.M.; Hatfield, K.M.; Reddy, S.C.; Kimball, A.; James, A.; Jacobs, J.R.; Taylor, J.; Spicer, K.; Bardossy, A.C.; Oakley, L.P.; et al. Presymptomatic SARS-CoV-2 Infections and Transmission in a Skilled Nursing Facility. N. Engl. J. Med. 2020, 382, 2081–2090. [Google Scholar] [CrossRef] [PubMed]
- Kittang, B.R.; Hofacker, S.V.; Solheim, S.P.; Krüger, K.; Løland, K.K.; Jansen, K. Outbreak of COVID-19 at three nursing homes in Bergen. Tidsskr. Den Nor. Legeforening 2020, 140. [Google Scholar] [CrossRef]
- LeRose, J.J.; Merlo, C.; Duong, P.; Harden, K.; Rush, R.; Artzberger, A.; Sidhu, N.; Sandhu, A.; Chopra, T. The role of the social vulnerability index in personal protective equipment shortages, number of cases, and associated mortality during the coronavirus disease 2019 (COVID-19) pandemic in Michigan skilled nursing facilities. Infect. Control Hosp. Epidemiol. 2021, 42, 877–880. [Google Scholar] [CrossRef]
- McMichael, T.M.; Currie, D.W.; Clark, S.; Pogosjans, S.; Kay, M.; Schwartz, N.G.; Lewis, J.; Baer, A.; Kawakami, V.; Lukoff, M.D.; et al. Epidemiology of Covid-19 in a Long-Term Care Facility in King County, Washington. N. Engl. J. Med. 2020, 382, 2005–2011. [Google Scholar] [CrossRef]
- Hossain, A.; Nasrullah, S.M.; Tasnim, Z.; Hasan, M.K.; Hasan, M.M. Seroprevalence of SARS-CoV-2 IgG antibodies among health care workers prior to vaccine administration in Europe, the USA and East Asia: A systematic review and meta-analysis. EClinicalMedicine 2021, 33, 100770. [Google Scholar] [CrossRef] [PubMed]
- Cevik, M.; Kuppalli, K.; Kindrachuk, J.; Peiris, M. Virology, transmission, and pathogenesis of SARS-CoV-2. BMJ 2020, 371, m3862. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.P.; Service, B.C.; Gupta, S.; Mubarak, N.; Zeini, I.M.; Osbahr, D.C.; Romeo, A.A. N95 respirator and surgical mask effectiveness against respiratory viral illnesses in the healthcare setting: A systematic review and meta-analysis. J. Am. Coll. Emerg. Physicians Open 2021, 2, e12582. [Google Scholar] [CrossRef] [PubMed]
- Blomberg, B.; Mohn, K.G.; Brokstad, K.A.; Zhou, F.; Linchausen, D.W.; Hansen, B.A.; Lartey, S.; Onyango, T.B.; Kuwelker, K.; Sævik, M.; et al. Long COVID in a prospective cohort of home-isolated patients. Nat. Med. 2021, 27, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Kuwelker, K.; Zhou, F.; Blomberg, B.; Lartey, S.; Brokstad, K.A.; Trieu, M.C.; Bansal, A.; Madsen, A.; Krammer, F.; Mohn, K.G.; et al. Attack rates amongst household members of outpatients with confirmed COVID-19 in Bergen, Norway: A case-ascertained study. Lancet Reg. Health Eur. 2021, 3, 100014. [Google Scholar] [CrossRef] [PubMed]

| Nursing Home (n = 313) | Hospital and ER (n = 434) | OR (95% CI) | p-Value | |
|---|---|---|---|---|
| Sex (men) * | 42/311 (14%) | 100/433 (23%) | 0.52 (0.35–0.77) | <0.001 |
| Age (years), (median, IQR) | 44 (32, 56) | 41 (31, 53) | - | 0.028 |
| Non-occupational exposure ** | 27/298 ** (9%) | 32/401 (8%) | 1.15 (0.67–1.997) | 0.711 |
| Work in ward with COVID-19 patients *** | 189/313 (60%) | 285/434 (66%) | 0.80 (0.59–1.08) | 0.101 |
| Nursing Home, N (%) | Hospital and ER, N (%) | OR (95% CI) | p-Value | |
|---|---|---|---|---|
| Occupational exposure | ||||
| Direct exposure * total | 50/310 (16) | 197/407 (48) | 0.20 (0.14–0.29) | <0.001 |
| Direct exposure, full PPE ** | 22/50 (44) | 159/197 (81) | 0.19 (0.10–0.36) | <0.001 |
| Direct exposure, partial/no PPE *** | 28/50 (56) | 38/197 (19) | 5.33 (2.75–10.31) | <0.001 |
| SARS-CoV-2-positive during the study period **** | ||||
| Total | 11/313 (4) | 28/434 (6) | 0.53 (0.26–1.08) | 0.095 |
| Direct exposure, total | 4/50 (8) | 13/197 (7) | 1.23 (0.38–3.95) | 0.755 |
| Direct exposure, full PPE | 3/22 (14) | 10/159 (6) | 2.35 (0.59–9.31) | 0.198 |
| Direct exposure, partial/no PPE | 1/28 (4) | 3/38 (8) | 0.43 (0.04–4.39) | 0.631 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kittang, B.R.; Blomberg, B.; Sævik, M.; Olofsson, J.S.; Bergen COVID-19 Research Group; Langeland, N.; Cox, R.J. SARS CoV-2 Infection among Health Care Workers from Different Health Care Facilities in Western Norway: A Prospective, Cross-Sectional Study. Viruses 2022, 14, 2652. https://doi.org/10.3390/v14122652
Kittang BR, Blomberg B, Sævik M, Olofsson JS, Bergen COVID-19 Research Group, Langeland N, Cox RJ. SARS CoV-2 Infection among Health Care Workers from Different Health Care Facilities in Western Norway: A Prospective, Cross-Sectional Study. Viruses. 2022; 14(12):2652. https://doi.org/10.3390/v14122652
Chicago/Turabian StyleKittang, Bård Reiakvam, Bjørn Blomberg, Marianne Sævik, Jan Stefan Olofsson, Bergen COVID-19 Research Group, Nina Langeland, and Rebecca Jane Cox. 2022. "SARS CoV-2 Infection among Health Care Workers from Different Health Care Facilities in Western Norway: A Prospective, Cross-Sectional Study" Viruses 14, no. 12: 2652. https://doi.org/10.3390/v14122652
APA StyleKittang, B. R., Blomberg, B., Sævik, M., Olofsson, J. S., Bergen COVID-19 Research Group, Langeland, N., & Cox, R. J. (2022). SARS CoV-2 Infection among Health Care Workers from Different Health Care Facilities in Western Norway: A Prospective, Cross-Sectional Study. Viruses, 14(12), 2652. https://doi.org/10.3390/v14122652







