Discovery, Genomic Sequence Characterization and Phylogenetic Analysis of Novel RNA Viruses in the Turfgrass Pathogenic Colletotrichum spp. in Japan
Abstract
1. Introduction
2. Material and Methods
2.1. Turfgrass Pathogenic Colletotrichum Strains and Culture Conditions
2.2. Extraction and Purification of dsRNA
2.3. dsRNA Sequencing to Identify Mycovirus Genome
2.4. Analysis of FLDS-Generated Sequence Reads
2.5. Validation of Genome Segments of Mycoviruses by RT-PCR
2.6. Analysis of Genomes of Identified Viruses
2.7. Phylogenetic Analysis of Identified Viruses
3. Results
3.1. Detection of Mycoviruses in Turfgrass Pathogenic Colletotrichum Strains
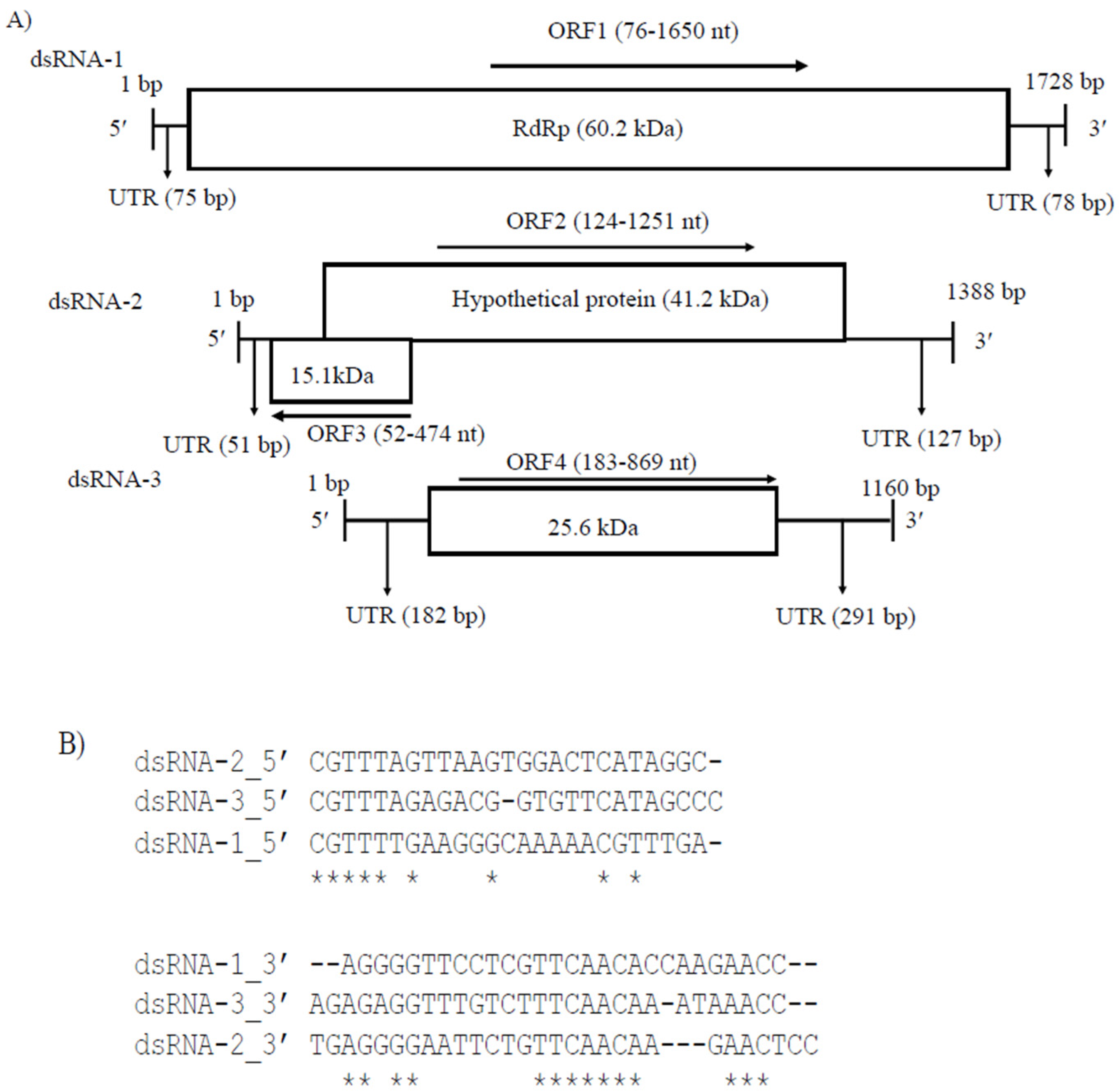
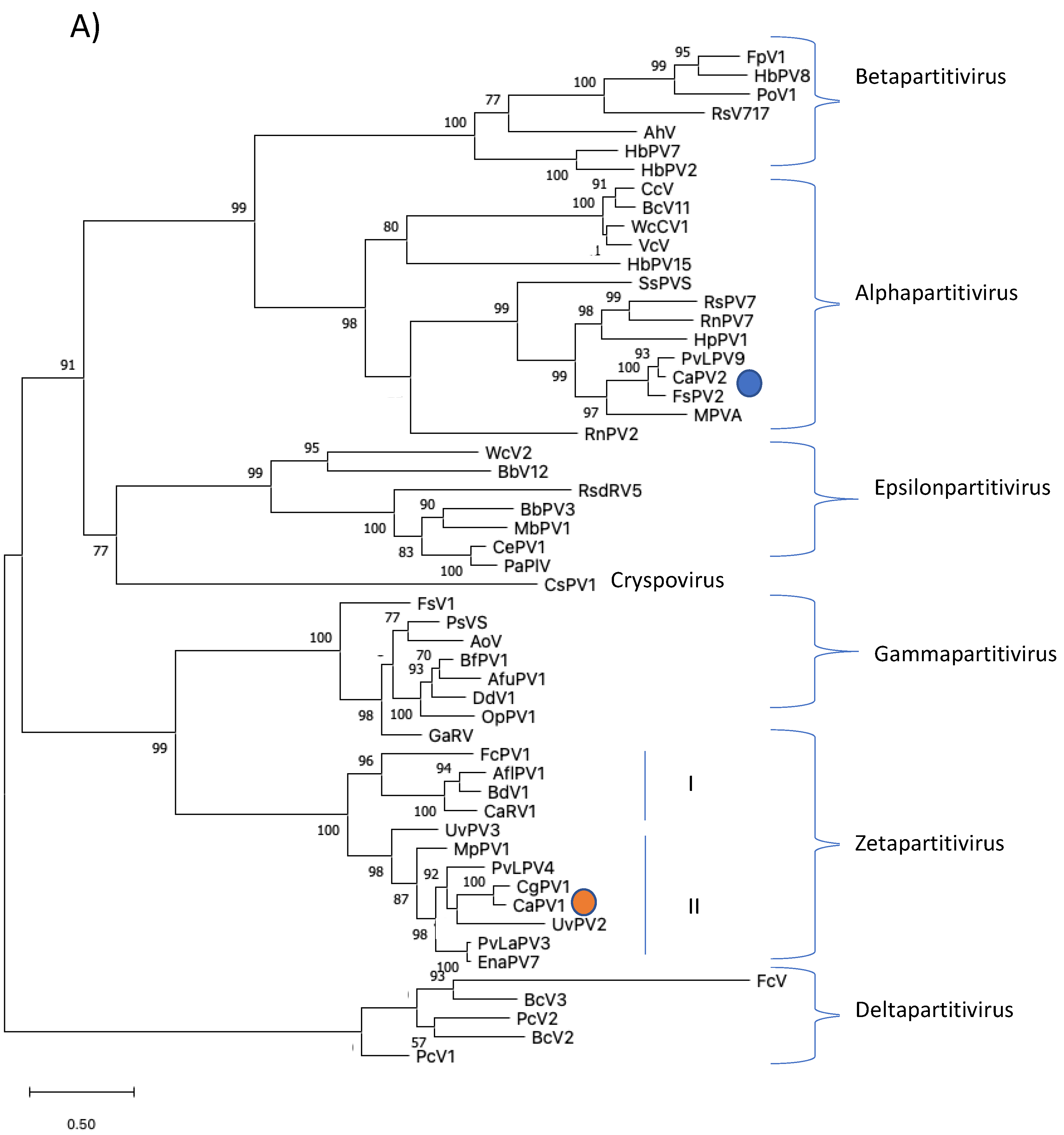
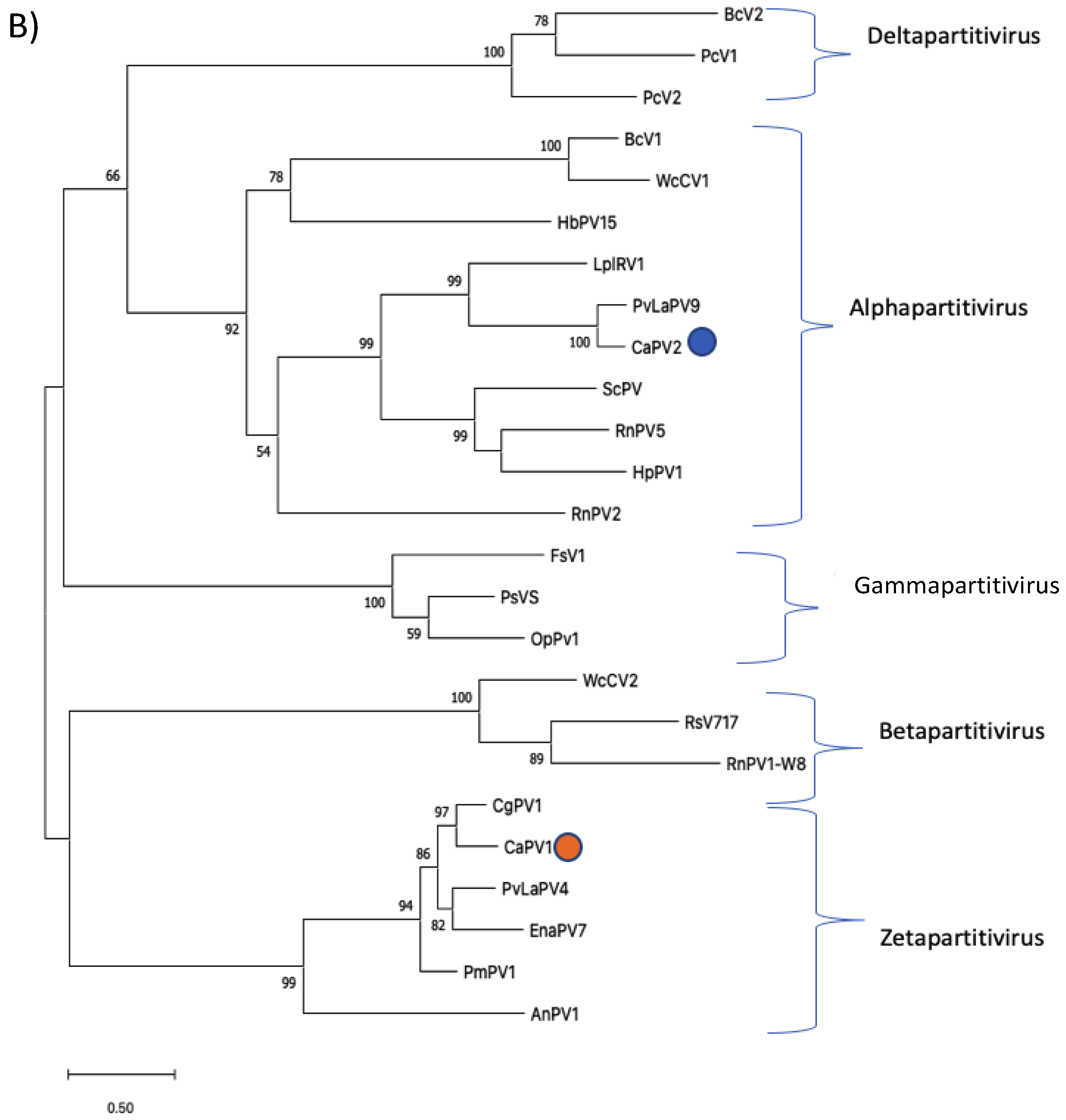
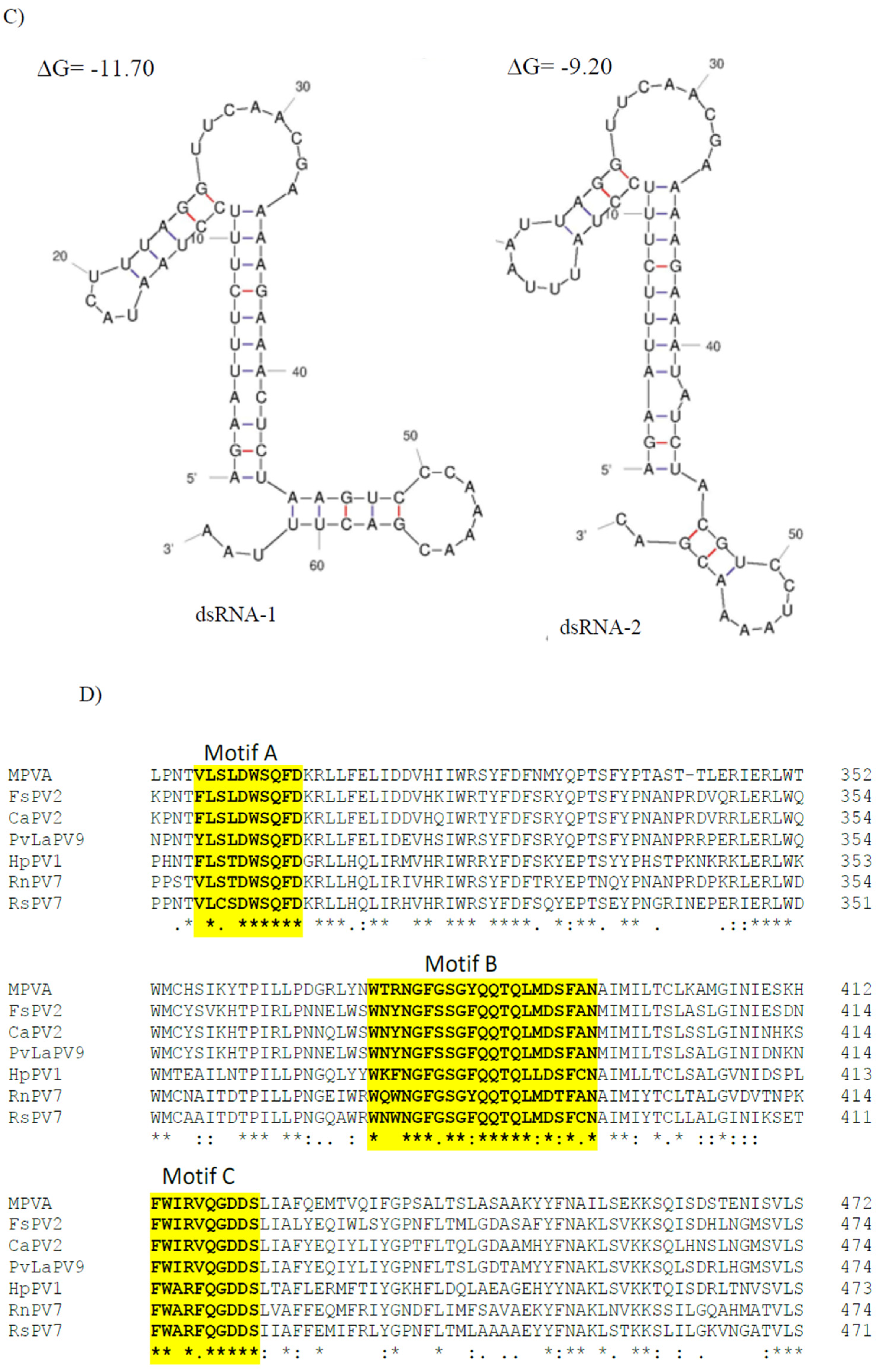
3.2. Genome Characterization and Phylogenetic Analysis of Colletotrichum Associated Partiti-Virus 1 (CaPV1)
3.3. Genome Characterization and Phylogenetic Analysis of Colletotrichum Associated Partiti-Virus 2 (CaPV2)
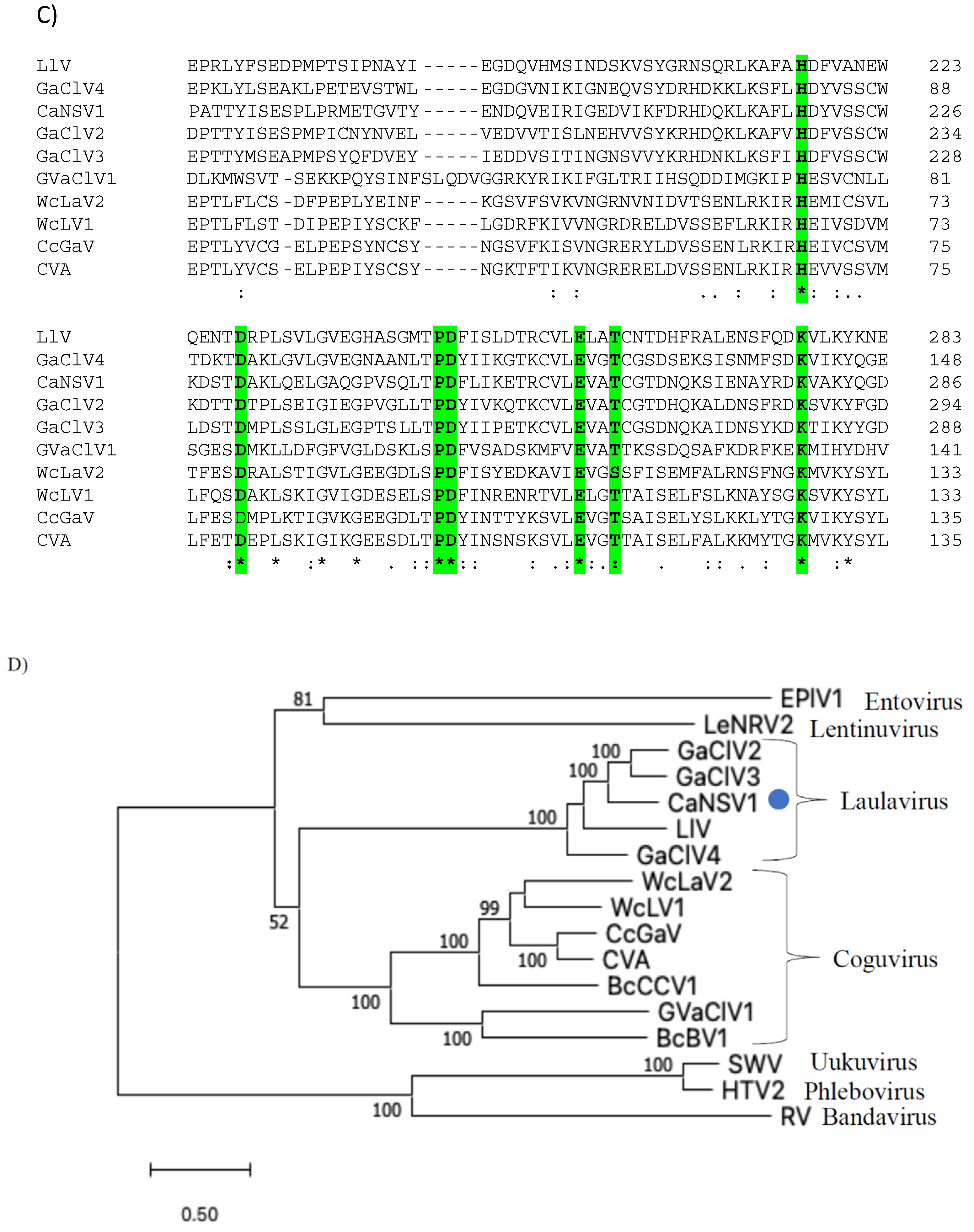
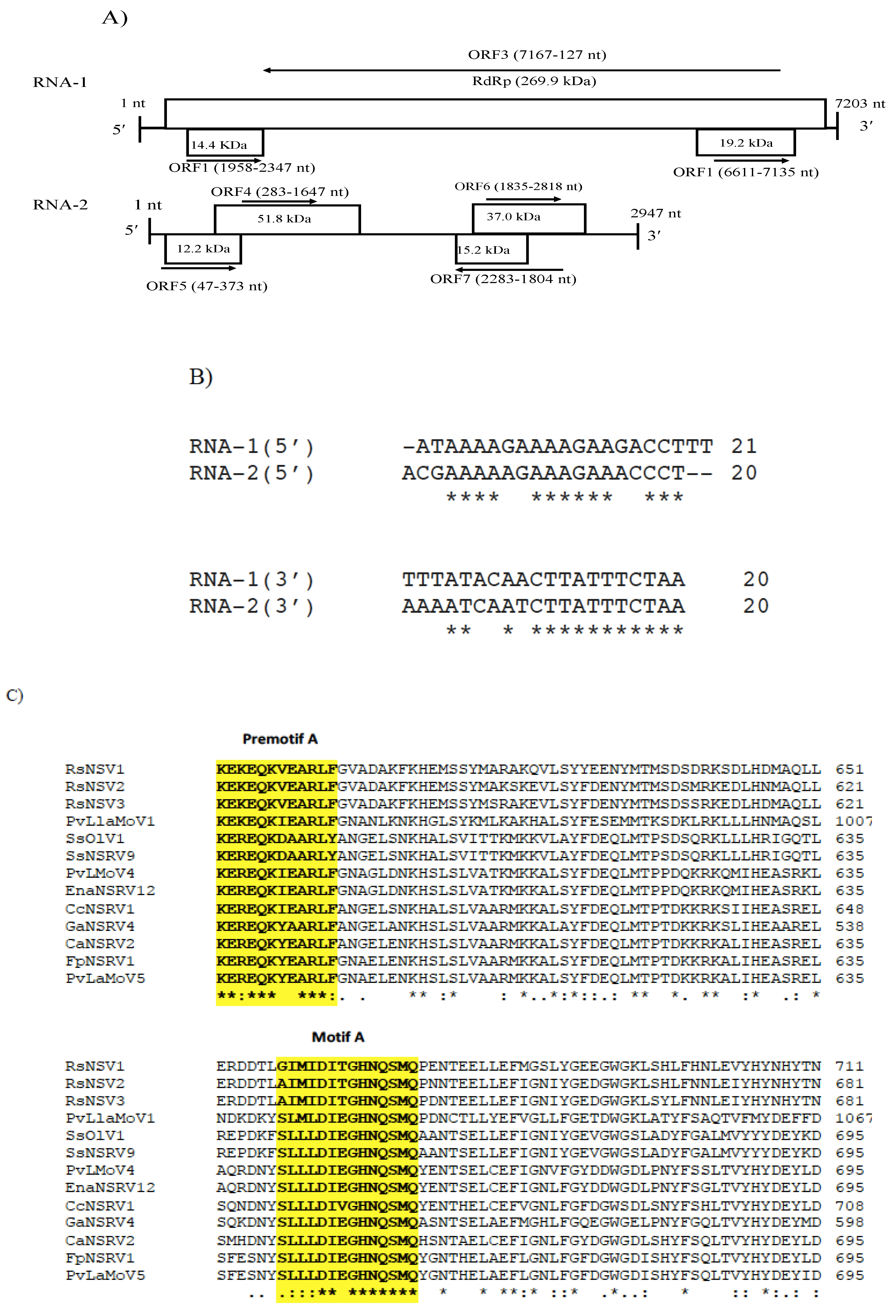
3.4. Genome Characterization of Colletotrichum Associated Negative-Stranded RNA Virus 1 (CaNSRV1) and Phylogenetic Relationship with Related Viruses
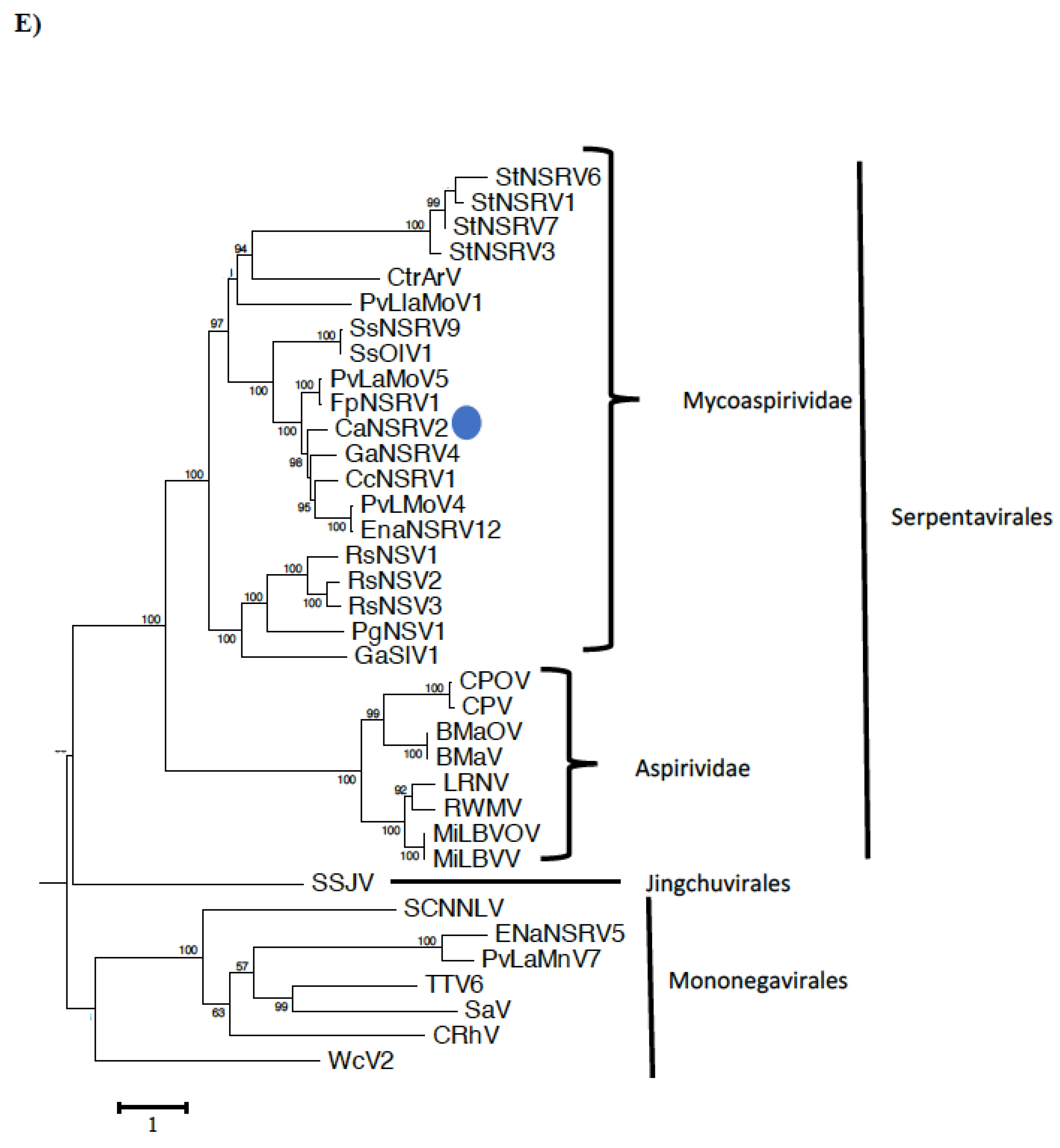
3.5. Genomic Characterization of Colletotrichum Associated Negative-Stranded RNA Virus 2 (CaNSRV2) and Phylogenetic Relationship with Related Viruses
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Casas, L.L.; Azevedo, J.L.; Almeida, L.N.; Costa-Neto, P.Q.; Bianco, R.A.; Pereira, J.O. Mycoviruses infecting Colletotrichum spp.: A comprehensive review. Braz. J. Biol. 2021, 83. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.; Wong, F.; Tredway, L.; Crouch, J.A.; Inguagiato, J.; Clarke, B.; Hsiang, T.; Rossi, F. Best management practices for anthracnose on annual bluegrass turf. Golf Course Manag. 2008, 24, 93–104. [Google Scholar]
- Zhai, L.; Zhang, M.; Hong, N.; Xiao, F.; Fu, M.; Xiang, J.; Wang, G. Identification and characterization of a novel hepta-segmented dsRNA virus from the phytopathogenic fungus Colletotrichum fructicola. Front. Microbiol. 2018, 9, 754. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Zhu, J.Z.; Zhou, X.Y.; Zhong, J.; Li, C.H.; Zhang, Z.G.; Zhu, H.J. A novel ourmia-like mycovirus isolated from the plant pathogenic fungus Colletotrichum gloeosporioides. Arch. Virol. 2019, 164, 2631–2635. [Google Scholar] [CrossRef]
- Ghabrial, S.A.; Castón, J.R.; Jiang, D.; Nibert, M.L.; Suzuki, N. 50-plus years of fungal viruses. Virology 2015, 479, 356–368. [Google Scholar] [CrossRef]
- Nuss, D.L. Hypovirulence: Mycoviruses at the fungal–plant interface. Nat. Rev. Microbiol. 2005, 3, 632–642. [Google Scholar] [CrossRef]
- Xie, J.; Jiang, D. New insights into mycoviruses and exploration for the biological control of crop fungal diseases. Annu. Rev. Phytopathol. 2014, 52, 45–68. [Google Scholar] [CrossRef]
- Lau, S.K.; Lo, G.C.; Chow, F.W.; Fan, R.Y.; Cai, J.J.; Yuen, K.Y.; Woo, P.C. Novel partitivirus enhances virulence of and causes aberrant gene expression in Talaromyces marneffei. MBio 2018, 9, e00947-18. [Google Scholar] [CrossRef]
- Okada, R.; Ichinose, S.; Takeshita, K.; Urayama, S.I.; Fukuhara, T.; Komatsu, K.; Arie, T.; Ishihara, A.; Egusa, M.; Kodama, M.; et al. Molecular characterization of a novel mycovirus in Alternaria alternata manifesting two-sided effects: Down-regulation of host growth and up-regulation of host plant pathogenicity. Virology 2018, 519, 23–32. [Google Scholar] [CrossRef]
- Morris, T.J.; Dodds, J.A. Isolation and analysis of double-stranded RNA from virus-infected plant and fungal tissue. Phytopathology 1979, 69, 854–858. [Google Scholar] [CrossRef]
- Okada, R.; Kiyota, E.; Moriyama, H.; Fukuhara, T.; Natsuaki, T. A simple and rapid method to purify viral dsRNA from plant and fungal tissue. J. Gen. Plant Pathol. 2015, 81, 103–107. [Google Scholar] [CrossRef]
- Khankhum, S.; Escalante, C.; Valverde, R.A. Extraction and electrophoretic analysis of large dsRNAs from desiccated plant tissues infected with plant viruses and biotrophic fungi. Eur. J. Plant Pathol. 2017, 147, 431–441. [Google Scholar] [CrossRef]
- Arjona-Lopez, J.M.; Telengech, P.; Jamal, A.; Hisano, S.; Kondo, H.; Yelin, M.D.; Arjona-Girona, I.; Kanematsu, S.; Lopez-Herrera, C.J.; Suzuki, N. Novel, diverse RNA viruses from Mediterranean isolates of the phytopathogenic fungus, Rosellinia necatrix: Insights into evolutionary biology of fungal viruses. Environ. Microbiol. 2018, 20, 1464–1483. [Google Scholar] [CrossRef] [PubMed]
- Sommers, P.; Chatterjee, A.; Varsani, A.; Trubl, G. Integrating viral metagenomics into an ecological framework. Annu. Rev. Virol. 2021, 8, 133–158. [Google Scholar] [CrossRef] [PubMed]
- Urayama, S.I.; Takaki, Y.; Chiba, Y.; Zhao, Y.; Kuroki, M.; Hagiwara, D.; Nunoura, T. Eukaryotic Microbial RNA Viruses—Acute or Persistent? Insights into Their Function in the Aquatic Ecosystem. Microbes Environ. 2022, 37, ME22034. [Google Scholar] [CrossRef] [PubMed]
- Nerva, L.; Varese, G.C.; Falk, B.W.; Turina, M.J.S.R. Mycoviruses of an endophytic fungus can replicate in plant cells: Evolutionary implications. Sci. Rep. 2017, 7, 1–11. [Google Scholar]
- Urayama, S.I.; Takaki, Y.; Nunoura, T. FLDS: A comprehensive dsRNA sequencing method for intracellular RNA virus surveillance. Microbes Environ. 2016, 31, 33–40. [Google Scholar] [CrossRef]
- Urayama, S.I.; Takaki, Y.; Nishi, S.; Yoshida-Takashima, Y.; Deguchi, S.; Takai, K.; Nunoura, T. Unveiling the RNA virosphere associated with marine microorganisms. Mol. Ecol. Resour. 2018, 18, 1444–1455. [Google Scholar] [CrossRef]
- Tanaka, A.; Matsuda, K. Anthracnose of zoysiagrass and bermudagrass caused by Colletotrichum sp. J. Jpn. Soc. Turfgrass Sci. 2021, 50, 47. (In Japanese) [Google Scholar]
- Zhang, W.; Damm, U.; Crous, P.W.; Groenewald, J.Z.; Niu, X.; Lin, J.; Li, Y. Anthracnose Disease of Carpetgrass (Axonopus compressus) Caused by Colletotrichum hainanense sp. nov. Plant Dis. 2020, 104, 1744–1750. [Google Scholar] [CrossRef]
- Hirai, J.; Urayama, S.I.; Takaki, Y.; Hirai, M.; Nagasaki, K.; Nunoura, T. RNA Virosphere in a Marine Zooplankton Community in the Subtropical Western North Pacific. Microbes Environ. 2022, 37, ME21066. [Google Scholar] [CrossRef] [PubMed]
- Milne, I.; Bayer, M.; Cardle, L.; Shaw, P.; Stephen, G.; Wright, F.; Marshall, D. Tablet—next generation sequence assemblyvisualization. Bioinformatics 2010, 26, 401–402. [Google Scholar] [CrossRef] [PubMed]
- Chiba, Y.; Oiki, S.; Yaguchi, T.; Urayama, S.I.; Hagiwara, D. Discovery of divided RdRp sequences and a hitherto unknown genomic complexity in fungal viruses. Virus Evol. 2021, 7, veaa101. [Google Scholar] [CrossRef] [PubMed]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 1–9. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [PubMed]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef]
- Zuker, M. Mfold web server for nucleic acid folding and hybridization prediction. Nucleic Acids Res. 2003, 31, 3406–3415. [Google Scholar] [CrossRef]
- Sievers, F.; Wilm, A.; Dineen, D.; Gibson, T.J.; Karplus, K.; Li, W.; Lopez, R.; McWilliam, H.; Remmert, M.; Söding, J.; et al. Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol. Syst. Biol. 2011, 7, 539. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular evolutionary genetics analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res. 2016, 44, W232–W235. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.T.; Chernomor, O.; Von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Fu, Y.; Jiang, D.; Mu, F.; Cheng, J.; Lin, Y.; Li, B.; Marzano, S.Y.L.; Xie, J. Interannual dynamics, diversity and evolution of the virome in Sclerotinia sclerotiorum from a single crop field. Virus Evol. 2021, 7, veab032. [Google Scholar] [CrossRef] [PubMed]
- Bruenn, J.A. A structural and primary sequence comparison of the viral RNA-dependent RNA polymerases. Nucleic Acids Res. 2003, 31, 1821–1829. [Google Scholar] [CrossRef]
- Jiang, Y.; Wang, J.; Yang, B.; Wang, Q.; Zhou, J.; Yu, W. Molecular characterization of a debilitation-associated partitivirus infecting the pathogenic fungus Aspergillus flavus. Front. Microbiol. 2019, 10, 626. [Google Scholar] [CrossRef]
- Ahmed, I.; Li, P.; Zhang, L.; Jiang, X.; Bhattacharjee, P.; Guo, L.; Wang, S. First report of a novel partitivirus from the phytopathogenic fungus Fusarium cerealis in China. Arch. Virol. 2020, 165, 2979–2983. [Google Scholar] [CrossRef] [PubMed]
- Vainio, E.J.; Chiba, S.; Ghabrial, S.A.; Maiss, E.; Roossinck, M.; Sabanad-zovic, S.; Suzuki, N.; Xie, J.T.; Nibert, M.; Consortium, I.R. ICTV virus taxonomy profile: Partitiviridae. J. Gener Virol. 2018, 99, 17–18. [Google Scholar] [CrossRef]
- Gilbert, K.B.; Holcomb, E.E.; Allscheid, R.L.; Carrington, J.C. Hiding in plain sight: New virus genomes discovered via a sys- tematic analysis of fungal public transcriptomes. PLoS ONE 2019, 14, e0219207. [Google Scholar] [CrossRef]
- Huerta-Cepas, J.; Serra, F.; Bork, P. ETE 3: Reconstruction, analysis, and visualization of phylogenomic data. Mol. Biol. Evol. 2016, 33, 1635–1638. [Google Scholar] [CrossRef]
- Katoh, K.; Kuma, K.I.; Toh, H.; Miyata, T. MAFFT version 5: Improvement in accuracy of multiple sequence alignment. Nucleic Acids Res. 2005, 33, 511–518. [Google Scholar] [CrossRef]
- Nguyen, L.T.; Schmidt, H.A.; Von Haeseler, A.; Minh, B.Q. IQ-TREE: A fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 2015, 32, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Nibert, M.L.; Ghabrial, S.A.; Maiss, E.; Lesker, T.; Vainio, E.J.; Jiang, D.; Suzuki, N. Taxonomic reorganization of family Partitiviridae and other recent progress in partitivirus research. Virus Res. 2014, 188, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Sahin, E.; Keskin, E.; Akata, I. Novel and diverse mycoviruses co-inhabiting the hypogeous ectomycorrhizal fungus Picoa juniperi. Virology 2021, 552, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Chiapello, M.; Rodríguez-Romero, J.; Ayllón, M.A.; Turina, M. Analysis of the virome associated to grapevine downy mildew lesions reveals new mycovirus lineages. Virus Evol. 2020, 6, veaa058. [Google Scholar] [CrossRef]
- Osaki, H.; Sasaki, A.; Nomiyama, K.; Sekiguchi, H.; Tomioka, K.; Takehara, T. Isolation and characterization of two mitoviruses and a putative alphapartitivirus from Fusarium spp. Virus Genes 2015, 50, 466–473. [Google Scholar] [CrossRef]
- Te Velthuis, A.J. Common and unique features of viral RNA-dependent polymerases. Cell. Mol. Life Sci. 2014, 71, 4403–4420. [Google Scholar] [CrossRef]
- van Poelwijk, F.; Prins, M.; Goldbach, R. Completion of the impatiens necrotic spot virus genome sequence and genetic comparison of the L proteins within the family Bunyaviridae. J. Gen. Virol. 1997, 78, 543–546. [Google Scholar] [CrossRef]
- Kormelink, R.; Garcia, M.L.; Goodin, M.; Sasaya, T.; Haenni, A.L. Negative-strand RNA viruses: The plant-infecting counterparts. Virus Res. 2011, 162, 184–202. [Google Scholar] [CrossRef]
- Elbeaino, T.; Digiaro, M.; Alabdullah, A.; De Stradis, A.; Minafra, A.; Mielke, N.; Castellano, M.A.; Martelli, G.P. A multipartite single-stranded negative-sense RNA virus is the putative agent of fig mosaic disease. J. Gen. Virol. 2009, 90, 1281–1288. [Google Scholar] [CrossRef]
- Olschewski, S.; Cusack, S.; Rosenthal, M. The cap-snatching mechanism of Bunyaviruses. Trends Microbiol. 2020, 28, 293–303. [Google Scholar] [CrossRef]
- Osaki, H.; Sasaki, A.; Nomiyama, K.; Tomioka, K. Multiple virus infection in a single strain of Fusarium poae shown by deep sequencing. Virus Genes 2016, 52, 835–847. [Google Scholar] [CrossRef] [PubMed]
- Nigg, E.A. Nucleocytoplasmic transport: Signals, mechanisms and regulation. Nature 1997, 386, 779–787. [Google Scholar] [CrossRef] [PubMed]
- Chun, J.; Yang, H.E.; Kim, D.H. Identification of a novel partitivirus of Trichoderma harzianum NFCF319 and evidence for the related antifungal activity. Front. Plant Sci. 2018, 9, 1699. [Google Scholar] [CrossRef] [PubMed]
- Magae, Y.; Sunagawa, M. Characterization of a mycovirus associated with the brown discoloration of edible mushroom, Flammulina velutipes. Virol. J. 2010, 7, 1–8. [Google Scholar] [CrossRef]
- Bhatti, M.F.; Jamal, A.; Petrou, M.A.; Cairns, T.C.; Bignell, E.M.; Coutts, R.H. The effects of dsRNA mycoviruses on growth and murine virulence of Aspergillus umigatus. Fungal Genet. Biol. 2011, 48, 1071–1075. [Google Scholar] [CrossRef]
- Xiao, X.; Cheng, J.; Tang, J.; Fu, Y.; Jiang, D.; Baker, T.S.; Ghabrial, S.A.; Xie, J. A novel partitivirus that confers hypovirulence on plant pathogenic fungi. J. Virol. 2014, 88, 10120–10133. [Google Scholar] [CrossRef]
- Krishnan, S.; Manavathu, E.K.; Chandrasekar, P.H. Aspergillus flavus: An emerging non-fumigatus Aspergillus species of significance. Mycoses 2009, 52, 206–222. [Google Scholar] [CrossRef]
- Chiba, S.; Lin, Y.-H.; Kondo, H.; Kanematsu, S.; Suzuki, N. Effects of defective interfering RNA on symptom induction by, and replication of, a Novel Partitivirus from a Phytopathogenic fungus, Rosellinia necatrix. J. Virol. 2013, 87, 2330–2341. [Google Scholar] [CrossRef]
- Compel, P.; Papp, I.; Fekete, C.; Hornok, L. Genetic interrelationships and genome organization of double-stranded RNA elements of Fusarium poae. Virus Genes 1999, 18, 49–56. [Google Scholar] [CrossRef]
- Zheng, L.; Zhang, M.; Chen, Q.; Zhu, M.; Zhou, E. A novel mycovirus closely related to viruses in the genus Alphapartitivirus confers hypovirulence in the phytopathogenic fungus Rhizoctonia solani. Virology 2014, 456, 220–226. [Google Scholar] [CrossRef]
- Tokarz, R.; Sameroff, S.; Tagliafierro, T.; Jain, K.; Williams, S.H.; Cucura, D.M.; Rochlin, I.; Monzon, J.; Carpi, G.; Tufts, D.; et al. Identification of novel viruses in Amblyomma americanum, Dermacentor variabilis, and Ixodes scapularis ticks. Msphere 2018, 3, e00614-17. [Google Scholar] [CrossRef] [PubMed]
- Marzano, S.Y.L.; Domier, L.L. Novel mycoviruses discovered from metatranscriptomics survey of soybean phyllosphere phytobiomes. Virus Res. 2016, 213, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.H.; Fujita, M.; Chiba, S.; Hyodo, K.; Andika, I.B.; Suzuki, N.; Kondo, H. Two novel fungal negative-strand RNA viruses related to mymonaviruses and phenuiviruses in the shiitake mushroom (Lentinula edodes). Virology 2019, 533, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Nerva, L.; Vigani, G.; Di Silvestre, D.; Ciuffo, M.; Forgia, M.; Chitarra, W.; Turina, M. Biological and molecular characterization of Chenopodium quinoa mitovirus 1 reveals a distinct sRNA response compared to cytoplasmic RNA viruses. JVI 2019, 93, e01998-18. [Google Scholar] [CrossRef]
- Velasco, L.; Arjona-Girona, I.; Cretazzo, E.; Lopez-Herrera, C. Viromes in Xylariaceae fungi infecting avocado in Spain. Virology 2019, 532, 11–21. [Google Scholar] [CrossRef]
- Liu, L.; Xie, J.; Cheng, J.; Fu, Y.; Li, G.; Yi, X.; Jiang, D. Fungal negative-stranded RNA virus that is related to bornaviruses and nyaviruses. Proc. Natl. Acad. Sci. USA 2014, 111, 12205–12210. [Google Scholar] [CrossRef]
- Hao, F.; Ding, T.; Wu, M.; Zhang, J.; Yang, L.; Chen, W.; Li, G. Two novel hypovirulence-associated mycoviruses in the phytopathogenic fungus Botrytis cinerea: Molecular characterization and suppression of infection cushion formation. Viruses 2018, 10, 254. [Google Scholar] [CrossRef]
- Koonin, E.V.; Dolja, V.V.; Krupovic, M.; Varsani, A.; Wolf, Y.I.; Yutin, N.; Zerbini, F.M.; Kuhn, J.H. Global organization and proposed megataxonomy of the virus world. Microbiol. Mol. Biol. Rev. 2020, 84, e00061-19. [Google Scholar] [CrossRef]
- Ghabrial, S.A. Origin, adaptation and evolutionary pathways of fungal viruses. Virus Genes 1998, 16, 119–131. [Google Scholar] [CrossRef]
- Ran, H.; Liu, L.; Li, B.; Cheng, J.; Fu, Y.; Jiang, D.; Xie, J. Co-infection of a hypovirulent isolate of Sclerotinia sclerotiorum with a new botybirnavirus and a strain of a mitovirus. Virol. J. 2016, 13, 92. [Google Scholar] [CrossRef]
- Telengech, P.; Hisano, S.; Mugambi, C.; Hyodo, K.; Arjona-López, J.M.; López-Herrera, C.J.; Kanematsu, S.; Kondo, H.; Suzuki, N. Diverse partitiviruses from the phytopathogenic fungus, Rosellinia necatrix. Front. Microbiol. 2020, 11, 1064. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Cheng, J.; Fu, Y.; Chen, T.; Jiang, D.; Ghabrial, S.A.; Xie, J. Virus-mediated suppression of host non-self recognition facilitates horizontal transmission of heterologous viruses. PLoS Pathog. 2017, 13, e1006234. [Google Scholar] [CrossRef] [PubMed]







| Strain | Host | Collection Place | Identified Pathogen |
|---|---|---|---|
| MBCT-164 | Bermuda grass (Cynodon dactylon) | Ibaraki Prefecture | Colletotirchum sp. |
| MBCT-165 | Bermuda grass (Cynodon dactylon) | Ibaraki Prefecture | Colletotirchum sp. |
| MBCT-220 | Manila grass (Zoysia matrella) | Saitama Prefecture | Colletotirchum sp. |
| MBCT-261 | Manila grass (Zoysia matrella) | Tochigi Prefecture | Colletotirchum sp. |
| MBCT-264 | Manila grass (Zoysia matrella) | Shizuoka Prefecture | Colletotirchum sp. |
| MBCT-265 | Japanese lawngrass (Zoysia japonica) | Shizuoka Prefecture | Colletotirchum sp. |
| MBCT-266 | Japanese lawngrass (Zoysia japonica) | Shizuoka Prefecture | Colletotirchum sp. |
| MBCT-288 | Creeping bentgrass (Agrostis stolonifera) | Ibaraki Prefecture | Colletotirchum sp. |
| MBCT-314 | Creeping bentgrass (Agrostis stolonifera) | Ibaraki Prefecture | Colletotirchum sp. |
| SL | Contig | Best Match | % aa Identity | Query Cover (%) | Genome Type/Putative Gene | Family or Genus |
|---|---|---|---|---|---|---|
| FLDS of dsRNA of MBCT-264 | ||||||
| 1 | S264_5_full | Colletotrichum gloeosporioides partitivirus 1, CgPV1 | 88.95 | 90 | dsRNA/RdRp | Partitiviridae |
| 2 | S264_1_full | CgPV1 | 73.4 | 73.4 | dsRNA/hypothetical protein/CP | Partitiviridae |
| 3 | S264_2_full | No significant similarity found | ||||
| 4 | S264_3_full | Plasmopara viticola lesion associated Partitivirus 9, PvLaPV9 | 90.62 | 93 | dsRNA/RdRp | Partitiviridae |
| 5 | S264_4_full | PvLaPV9 | 73.74 | 86 | dsRNA/CP | Partitiviridae |
| 6 | S264_7_full | No significant similarity found | ||||
| 7 | S264_6_full | No significant similarity found | ||||
| 8 | S264_8_full | Grapevine associated cogu-like virus 2, GaCLV2 | 69.78 | 98 | (−) ssRNA/RdRp | Phenuiviridae |
| FLDS of dsRNA of MBCT-288 | ||||||
| 9 | S288_2_full | Fusarium poae negative-stranded virus 1, FpNSV1 | 65.69 | 97 | (−) ssRNA/RdRp | unclassified ssRNA negative-strand viruses |
| 10 | S288_1_full | Claviceps aff. purpurea | 54.58 | 30 | (−) ssRNA/hypothetical protein | unclassified ssRNA negative-strand viruses |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamim, I.; Urayama, S.-i.; Netsu, O.; Tanaka, A.; Arie, T.; Moriyama, H.; Komatsu, K. Discovery, Genomic Sequence Characterization and Phylogenetic Analysis of Novel RNA Viruses in the Turfgrass Pathogenic Colletotrichum spp. in Japan. Viruses 2022, 14, 2572. https://doi.org/10.3390/v14112572
Hamim I, Urayama S-i, Netsu O, Tanaka A, Arie T, Moriyama H, Komatsu K. Discovery, Genomic Sequence Characterization and Phylogenetic Analysis of Novel RNA Viruses in the Turfgrass Pathogenic Colletotrichum spp. in Japan. Viruses. 2022; 14(11):2572. https://doi.org/10.3390/v14112572
Chicago/Turabian StyleHamim, Islam, Syun-ichi Urayama, Osamu Netsu, Akemi Tanaka, Tsutomu Arie, Hiromitsu Moriyama, and Ken Komatsu. 2022. "Discovery, Genomic Sequence Characterization and Phylogenetic Analysis of Novel RNA Viruses in the Turfgrass Pathogenic Colletotrichum spp. in Japan" Viruses 14, no. 11: 2572. https://doi.org/10.3390/v14112572
APA StyleHamim, I., Urayama, S.-i., Netsu, O., Tanaka, A., Arie, T., Moriyama, H., & Komatsu, K. (2022). Discovery, Genomic Sequence Characterization and Phylogenetic Analysis of Novel RNA Viruses in the Turfgrass Pathogenic Colletotrichum spp. in Japan. Viruses, 14(11), 2572. https://doi.org/10.3390/v14112572







