The Impact of Urbanization and Human Mobility on Seasonal Influenza in Northern China
Abstract
1. Introduction
2. Methods
2.1. Data
2.2. SEIRS Model
2.3. GLM Model
2.4. Estimation of Transmissibility
2.5. Regression Analysis with Transmissibility and Rt of Each Influenza Season
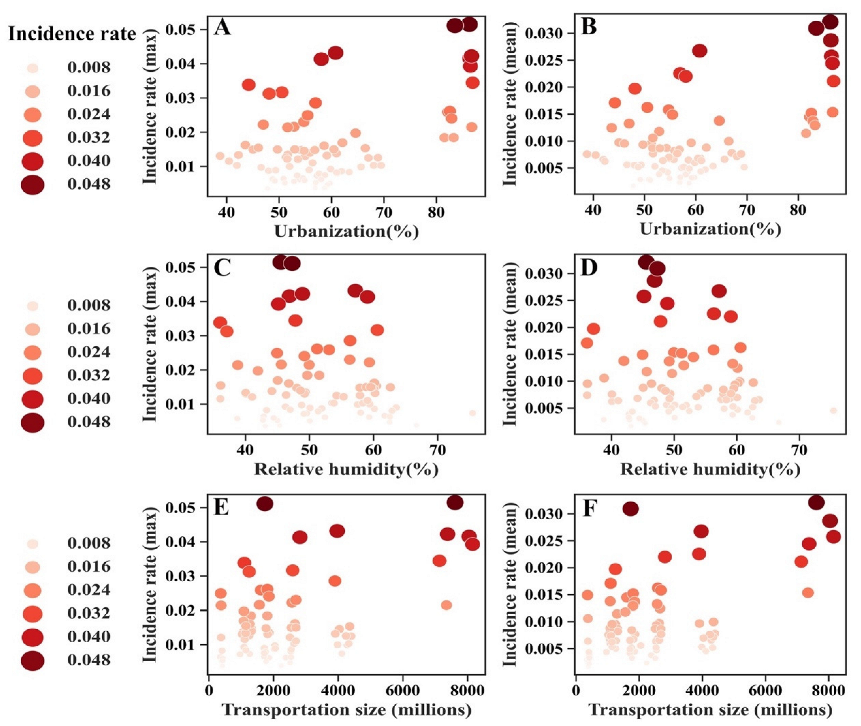
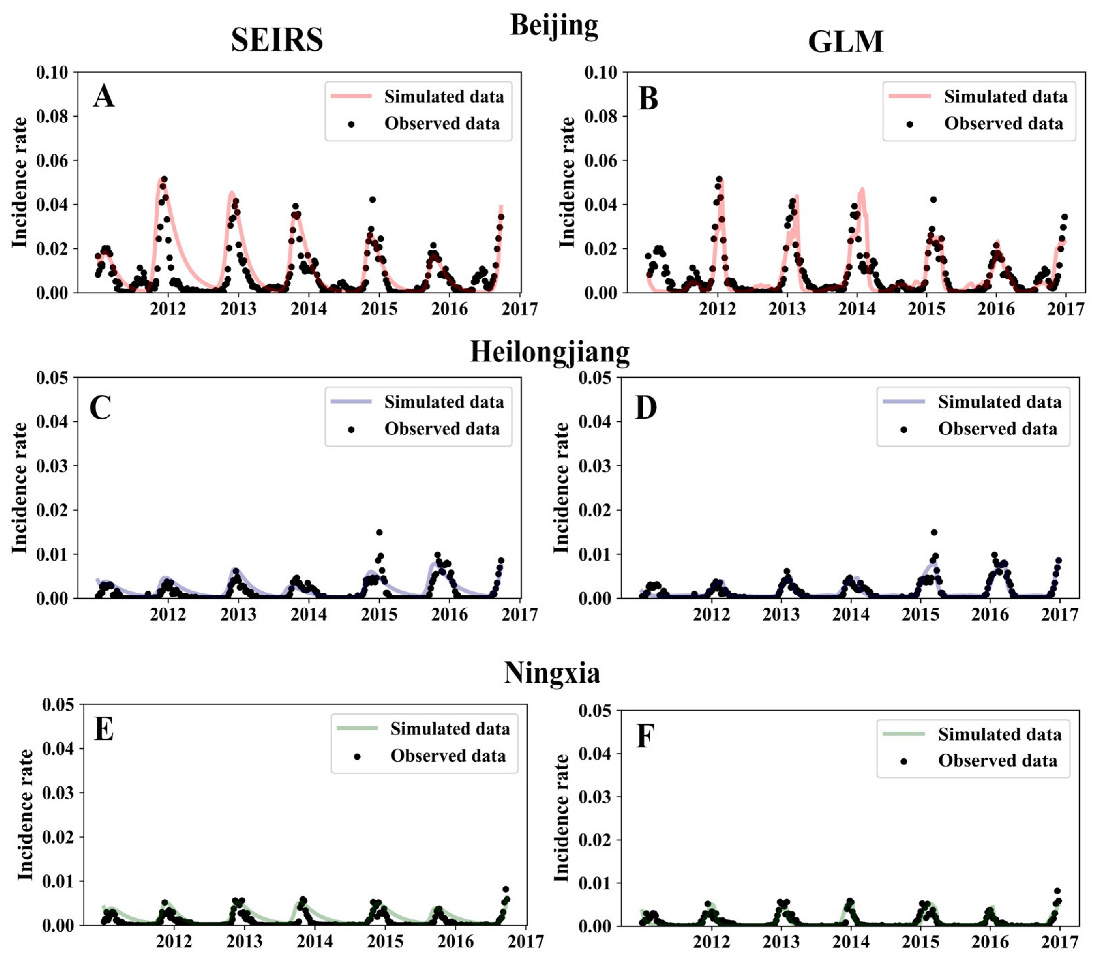
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tamerius, J.; Nelson, M.I.; Zhou, S.Z.; Viboud, C.; Miller, M.A.; Alonso, W.J. Global influenza seasonality: Reconciling patterns across temperate and tropical regions. Environ. Health Perspect. 2011, 119, 439–445. [Google Scholar] [CrossRef]
- Lei, H.; Jiang, H.; Zhang, N.; Duan, X.; Chen, T.; Yang, L.; Wang, D.; Shu, Y. Increased urbanization reduced the effectiveness of school closures on seasonal influenza epidemics in China. Infect. Dis. Poverty 2021, 10, 127. [Google Scholar] [CrossRef]
- Ali, S.T.; Cowling, B.J.; Wong, J.Y.; Chen, D.; Shan, S.; Lau, E.H.Y.; He, D.; Tian, L.; Li, Z.; Wu, P. Influenza seasonality and its environmental driving factors in mainland China and Hong Kong. Sci. Total Environ. 2022, 818, 151724. [Google Scholar] [CrossRef]
- Harper, G.J. Airborne micro-organisms: Survival tests with four viruses. J. Hyg. 1961, 59, 479–486. [Google Scholar] [CrossRef]
- Lowen, A.C.; Mubareka, S.; Steel, J.; Palese, P. Influenza virus transmission is dependent on relative humidity and temperature. PLoS Pathog. 2007, 3, 1470–1476. [Google Scholar] [CrossRef]
- Peci, A.; Winter, A.L.; Li, Y.; Gnaneshan, S.; Liu, J.; Mubareka, S.; Gubbay, J.B. Effects of Absolute Humidity, Relative Humidity, Temperature, and Wind Speed on Influenza Activity in Toronto, Ontario, Canada. Appl. Environ. Microbiol. 2019, 85, e02426-18. [Google Scholar] [CrossRef]
- Zachreson, C.; Fair, K.M.; Cliff, O.M.; Harding, N.; Piraveenan, M.; Prokopenko, M. Urbanization affects peak timing, prevalence, and bimodality of influenza pandemics in Australia: Results of a census-calibrated model. Sci. Adv. 2018, 4, eaau5294. [Google Scholar] [CrossRef]
- Brownstein, J.S.; Wolfe, C.J.; Mandl, K.D. Empirical evidence for the effect of airline travel on inter-regional influenza spread in the United States. PLoS Med. 2006, 3, e401. [Google Scholar] [CrossRef]
- Merler, S.; Ajelli, M. The role of population heterogeneity and human mobility in the spread of pandemic influenza. Proc. Biol. Sci. 2010, 277, 557–565. [Google Scholar] [CrossRef]
- Dalziel, B.D.; Kissler, S.; Gog, J.R.; Viboud, C.; Bjornstad, O.N.; Metcalf, C.J.E.; Grenfell, B.T. Urbanization and humidity shape the intensity of influenza epidemics in U.S. cities. Science 2018, 362, 75–79. [Google Scholar] [CrossRef]
- Sun, S.; Fu, C.; Cong, J.; Li, Y.; Xie, S.; Wang, P. Epidemiological features and trends of influenza incidence in mainland China: A population-based surveillance study from 2005 to 2015. Int. J. Infect. Dis. 2019, 89, 12–20. [Google Scholar] [CrossRef] [PubMed]
- National Bureau of Statistics of China. Available online: http://www.stats.gov.cn/tjsj/ndsj/ (accessed on 16 September 2022).
- Lei, H.; Xu, M.; Wang, X.; Xie, Y.; Du, X.; Chen, T.; Yang, L.; Wang, D.; Shu, Y. Nonpharmaceutical Interventions Used to Control COVID-19 Reduced Seasonal Influenza Transmission in China. J. Infect. Dis. 2020, 222, 1780–1783. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, E.; Cobey, S.; Takahashi, S.; Miller, J.C.; Lipsitch, M. Predicting the epidemic sizes of influenza A/H1N1, A/H3N2, and B: A statistical method. PLoS Med. 2011, 8, e1001051. [Google Scholar] [CrossRef] [PubMed]
- Wong, J.Y.; Wu, P.; Nishiura, H.; Goldstein, E.; Lau, E.H.; Yang, L.; Chuang, S.K.; Tsang, T.; Peiris, J.S.; Wu, J.T.; et al. Infection fatality risk of the pandemic A(H1N1)2009 virus in Hong Kong. Am. J. Epidemiol. 2013, 177, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.T.; Cowling, B.J.; Lau, E.H.Y.; Fang, V.J.; Leung, G.M. Mitigation of Influenza B Epidemic with School Closures, Hong Kong, 2018. Emerg. Infect Dis. 2018, 24, 2071–2073. [Google Scholar] [CrossRef]
- Young, B.; Sadarangani, S.; Jiang, L.; Wilder-Smith, A.; Chen, M.I. Duration of Influenza Vaccine Effectiveness: A Systematic Review, Meta-analysis, and Meta-regression of Test-Negative Design Case-Control Studies. J. Infect. Dis. 2018, 217, 731–741. [Google Scholar] [CrossRef]
- Patel, M.M.; York, I.A.; Monto, A.S.; Thompson, M.G.; Fry, A.M. Immune-mediated attenuation of influenza illness after infection: Opportunities and challenges. Lancet Microbe 2021, 2, e715–e725. [Google Scholar] [CrossRef]
- Wang, Q.; Yue, N.; Zheng, M.; Wang, D.; Duan, C.; Yu, X.; Zhang, X.; Bao, C.; Jin, H. Influenza vaccination coverage of population and the factors influencing influenza vaccination in mainland China: A meta-analysis. Vaccine 2018, 36, 7262–7269. [Google Scholar] [CrossRef] [PubMed]
- Te Beest, D.E.; Van Boven, M.; Hooiveld, M.; Van Den Dool, C.; Wallinga, J. Driving factors of influenza transmission in the Netherlands. Am. J. Epidemiol. 2013, 178, 1469–1477. [Google Scholar] [CrossRef]
- Cori, A.; Ferguson, N.M.; Fraser, C.; Cauchemez, S. A new framework and software to estimate time-varying reproduction numbers during epidemics. Am. J. Epidemiol. 2013, 178, 1505–1512. [Google Scholar] [CrossRef]
- Fraser, C. Estimating individual and household reproduction numbers in an emerging epidemic. PLoS ONE 2007, 2, e758. [Google Scholar] [CrossRef] [PubMed]
- Hemmes, J.H.; Winkler, K.C.; Kool, S.M. Virus survival as a seasonal factor in influenza and polimyelitis. Nature 1960, 188, 430–431. [Google Scholar] [CrossRef]
- Cooley, P.; Brown, S.; Cajka, J.; Chasteen, B.; Ganapathi, L.; Grefenstette, J.; Hollingsworth, C.R.; Lee, B.Y.; Levine, B.; Wheaton, W.D.; et al. The role of subway travel in an influenza epidemic: A New York City simulation. J. Urban Health. 2011, 88, 982–995. [Google Scholar] [CrossRef]
- Russell, C.A.; Jones, T.C.; Barr, I.G.; Cox, N.J.; Garten, R.J.; Gregory, V.; Gust, I.D.; Hampson, A.W.; Hay, A.J.; Hurt, A.C.; et al. The global circulation of seasonal influenza A (H3N2) viruses. Science 2008, 320, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Brockmann, D.; Helbing, D. The hidden geometry of complex, network-driven contagion phenomena. Science 2013, 342, 1337–1342. [Google Scholar] [CrossRef] [PubMed]
- Grais, R.F.; Ellis, J.H.; Glass, G.E. Assessing the impact of airline travel on the geographic spread of pandemic influenza. Eur. J. Epidemiol. 2003, 18, 1065–1072. [Google Scholar] [CrossRef]
- Tizzoni, M.; Bajardi, P.; Poletto, C.; Ramasco, J.J.; Balcan, D.; Gonçalves, B.; Perra, N.; Colizza, V.; Vespignani, A. Real-time numerical forecast of global epidemic spreading: Case study of 2009 A/H1N1pdm. BMC Med. 2012, 10, 165. [Google Scholar] [CrossRef]
- Kenah, E.; Chao, D.L.; Matrajt, L.; Halloran, M.E.; Longini, I.M., Jr. The global transmission and control of influenza. PLoS ONE 2011, 6, e19515. [Google Scholar] [CrossRef]
- Lemey, P.; Rambaut, A.; Bedford, T.; Faria, N.; Bielejec, F.; Baele, G.; Russell, C.A.; Smith, D.J.; Pybus, O.G.; Brockmann, D.; et al. Unifying viral genetics and human transportation data to predict the global transmission dynamics of human influenza H3N2. PLoS Pathog. 2014, 10, e1003932. [Google Scholar] [CrossRef]
- Cauchemez, S.; Valleron, A.J.; Boëlle, P.Y.; Flahault, A.; Ferguson, N.M. Estimating the impact of school closure on influenza transmission from Sentinel data. Nature 2008, 452, 750–754. [Google Scholar] [CrossRef]
- Charu, V.; Zeger, S.; Gog, J.; Bjornstad, O.N.; Kissler, S.; Simonsen, L.; Grenfell, B.T.; Viboud, C. Human mobility and the spatial transmission of influenza in the United States. PLoS Comput. Biol. 2017, 13, e1005382. [Google Scholar] [CrossRef] [PubMed]
- Eubank, S.; Guclu, H.; Kumar, V.S.; Marathe, M.V.; Srinivasan, A.; Toroczkai, Z.; Wang, N. Modelling disease outbreaks in realistic urban social networks. Nature 2004, 429, 180–184. [Google Scholar] [CrossRef] [PubMed]
- Yashima, K.; Sasaki, A. Epidemic process over the commute network in a metropolitan area. PLoS ONE 2014, 9, e98518. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.; Srebric, J.; Spengler, J.D.; Demokritou, P. An advanced numerical model for the assessment of airborne transmission of influenza in bus microenvironments. Build Environ. 2012, 47, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.M.; Stallone, R.; Zhang, F.; Gerolimatos, M.; Korologos, D.D.; Sweetapple, C.; De Geronimo, M.; Dlugacz, Y.; Armellino, D.M.; Ginocchio, C.C. Laboratory surge response to pandemic (H1N1) 2009 outbreak, New York City metropolitan area, USA. Emerg. Infect. Dis. 2010, 16, 8–13. [Google Scholar] [CrossRef]
- Arinaminpathy, N.; Kim, I.K.; Gargiullo, P.; Haber, M.; Foppa, I.M.; Gambhir, M.; Bresee, J. Estimating Direct and Indirect Protective Effect of Influenza Vaccination in the United States. Am. J. Epidemiol. 2017, 186, 92–100. [Google Scholar] [CrossRef]
- Feng, L.; Zhang, T.; Wang, Q.; Xie, Y.; Peng, Z.; Zheng, J.; Qin, Y.; Zhang, M.; Lai, S.; Wang, D.; et al. Impact of COVID-19 outbreaks and interventions on influenza in China and the United States. Nat. Commun. 2021, 12, 3249. [Google Scholar] [CrossRef]
- Cowling, B.J.; Lau, E.H.; Lam, C.L.; Cheng, C.K.; Kovar, J.; Chan, K.H.; Peiris, J.S.; Leung, G.M. Effects of school closures, 2008 winter influenza season, Hong Kong. Emerg. Infect. Dis. 2008, 14, 1660–1662. [Google Scholar] [CrossRef]
- Wu, J.T.; Cowling, B.J.; Lau, E.H.; Ip, D.K.; Ho, L.M.; Tsang, T.; Chuang, S.K.; Leung, P.Y.; Lo, S.V.; Liu, S.H.; et al. School closure and mitigation of pandemic (H1N1) 2009, Hong Kong. Emerg. Infect. Dis. 2010, 16, 538–541. [Google Scholar] [CrossRef]
- Lam, E.K.S.; Morris, D.H.; Hurt, A.C.; Barr, I.G.; Russell, C.A. The impact of climate and antigenic evolution on seasonal influenza virus epidemics in Australia. Nat. Commun. 2020, 11, 2741. [Google Scholar] [CrossRef]


| Year | Province | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beijing | Tianjin | Hebei | Shanxi | Inner Mongolia | Liaoning | Jilin | Heilongjiang | Shandong | Henan | Shaanxi | Gansu | Qinghai | Ningxia | ||
| Population size (millions) | 2012 | 20.69 | 14.13 | 72.88 | 36.11 | 24.90 | 43.89 | 27.50 | 38.34 | 96.85 | 94.06 | 37.53 | 25.78 | 5.73 | 6.47 |
| 2013 | 21.15 | 14.72 | 73.33 | 36.30 | 24.98 | 43.90 | 27.51 | 38.35 | 97.33 | 94.13 | 37.64 | 25.82 | 5.78 | 6.54 | |
| 2014 | 21.52 | 15.17 | 73.84 | 36.48 | 25.05 | 43.91 | 27.52 | 38.33 | 97.89 | 94.36 | 37.75 | 25.91 | 5.83 | 6.62 | |
| 2015 | 21.71 | 15.47 | 74.25 | 36.64 | 25.11 | 43.82 | 27.53 | 38.12 | 98.47 | 94.80 | 37.93 | 26.00 | 5.88 | 6.68 | |
| 2016 | 21.73 | 15.62 | 74.70 | 36.82 | 25.20 | 43.78 | 27.33 | 37.99 | 99.47 | 95.32 | 38.13 | 26.10 | 5.93 | 6.75 | |
| 2017 | 21.71 | 15.57 | 75.20 | 37.02 | 25.29 | 43.69 | 27.17 | 37.89 | 100.06 | 95.59 | 38.35 | 26.26 | 5.98 | 6.82 | |
| Urbanization (%) | 2012 | 86.20 | 81.55 | 46.80 | 51.26 | 57.14 | 65.65 | 53.70 | 59.60 | 52.43 | 42.43 | 50.02 | 38.75 | 47.44 | 50.67 |
| 2013 | 86.30 | 82.01 | 48.12 | 52.66 | 58.71 | 66.45 | 54.20 | 57.40 | 53.75 | 43.80 | 51.31 | 40.13 | 48.51 | 52.01 | |
| 2014 | 86.35 | 82.27 | 49.33 | 53.79 | 59.51 | 67.05 | 54.81 | 58.01 | 55.01 | 45.20 | 52.57 | 41.68 | 49.78 | 53.61 | |
| 2015 | 86.50 | 82.64 | 51.33 | 55.03 | 60.30 | 67.35 | 55.31 | 58.80 | 57.01 | 46.85 | 53.92 | 43.19 | 50.30 | 55.23 | |
| 2016 | 86.50 | 82.93 | 53.32 | 56.21 | 61.19 | 67.37 | 55.97 | 59.20 | 59.02 | 48.50 | 55.34 | 44.69 | 51.63 | 56.29 | |
| 2017 | 86.50 | 82.93 | 55.01 | 57.34 | 62.02 | 67.49 | 56.65 | 59.40 | 60.58 | 50.16 | 56.79 | 46.39 | 53.07 | 57.98 | |
| Public transportation size (millions) | 2012 | 7615.78 | 1299.51 | 2039.54 | 1248.38 | 963.49 | 4283.67 | 1705.61 | 2239.56 | 3982.68 | 2637.18 | 2545.99 | 1028.44 | 391.35 | 391.35 |
| 2013 | 8047.75 | 1609.27 | 2027.27 | 1563.64 | 1086.85 | 4356.33 | 1713.78 | 2381.56 | 4113.11 | 2661.57 | 2507.32 | 1107.11 | 417.60 | 417.60 | |
| 2014 | 8158.48 | 1810.72 | 2053.42 | 1314.96 | 1070.99 | 4401.65 | 1768.67 | 2513.60 | 4038.54 | 2638.19 | 2692.46 | 1137.38 | 349.71 | 427.44 | |
| 2015 | 7383.84 | 1858.13 | 1872.08 | 1321.32 | 1076.74 | 4347.10 | 1754.52 | 2574.76 | 3900.26 | 2570.70 | 2692.03 | 1098.23 | 379.09 | 423.77 | |
| 2016 | 7349.53 | 1807.90 | 1860.48 | 1263.50 | 1145.88 | 4242.62 | 1713.69 | 2534.20 | 3911.36 | 2539.10 | 2694.06 | 1145.49 | 377.68 | 418.98 | |
| 2017 | 7133.96 | 1732.79 | 1818.29 | 1271.83 | 1083.90 | 4328.24 | 1772.18 | 2550.84 | 3967.77 | 2594.21 | 2817.40 | 1249.46 | 367.10 | 407.44 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, J.; Guo, X.; Zhang, T.; Wang, Q.; Zhang, X.; Yang, J.; Lai, S.; Feng, L.; Yang, W. The Impact of Urbanization and Human Mobility on Seasonal Influenza in Northern China. Viruses 2022, 14, 2563. https://doi.org/10.3390/v14112563
Yang J, Guo X, Zhang T, Wang Q, Zhang X, Yang J, Lai S, Feng L, Yang W. The Impact of Urbanization and Human Mobility on Seasonal Influenza in Northern China. Viruses. 2022; 14(11):2563. https://doi.org/10.3390/v14112563
Chicago/Turabian StyleYang, Jiao, Xudong Guo, Ting Zhang, Qing Wang, Xingxing Zhang, Jin Yang, Shengjie Lai, Luzhao Feng, and Weizhong Yang. 2022. "The Impact of Urbanization and Human Mobility on Seasonal Influenza in Northern China" Viruses 14, no. 11: 2563. https://doi.org/10.3390/v14112563
APA StyleYang, J., Guo, X., Zhang, T., Wang, Q., Zhang, X., Yang, J., Lai, S., Feng, L., & Yang, W. (2022). The Impact of Urbanization and Human Mobility on Seasonal Influenza in Northern China. Viruses, 14(11), 2563. https://doi.org/10.3390/v14112563







