Two Years of Genomic Surveillance in Belgium during the SARS-CoV-2 Pandemic to Attain Country-Wide Coverage and Monitor the Introduction and Spread of Emerging Variants
Abstract
1. Introduction
2. Prior to the National Genomic Surveillance Initiative
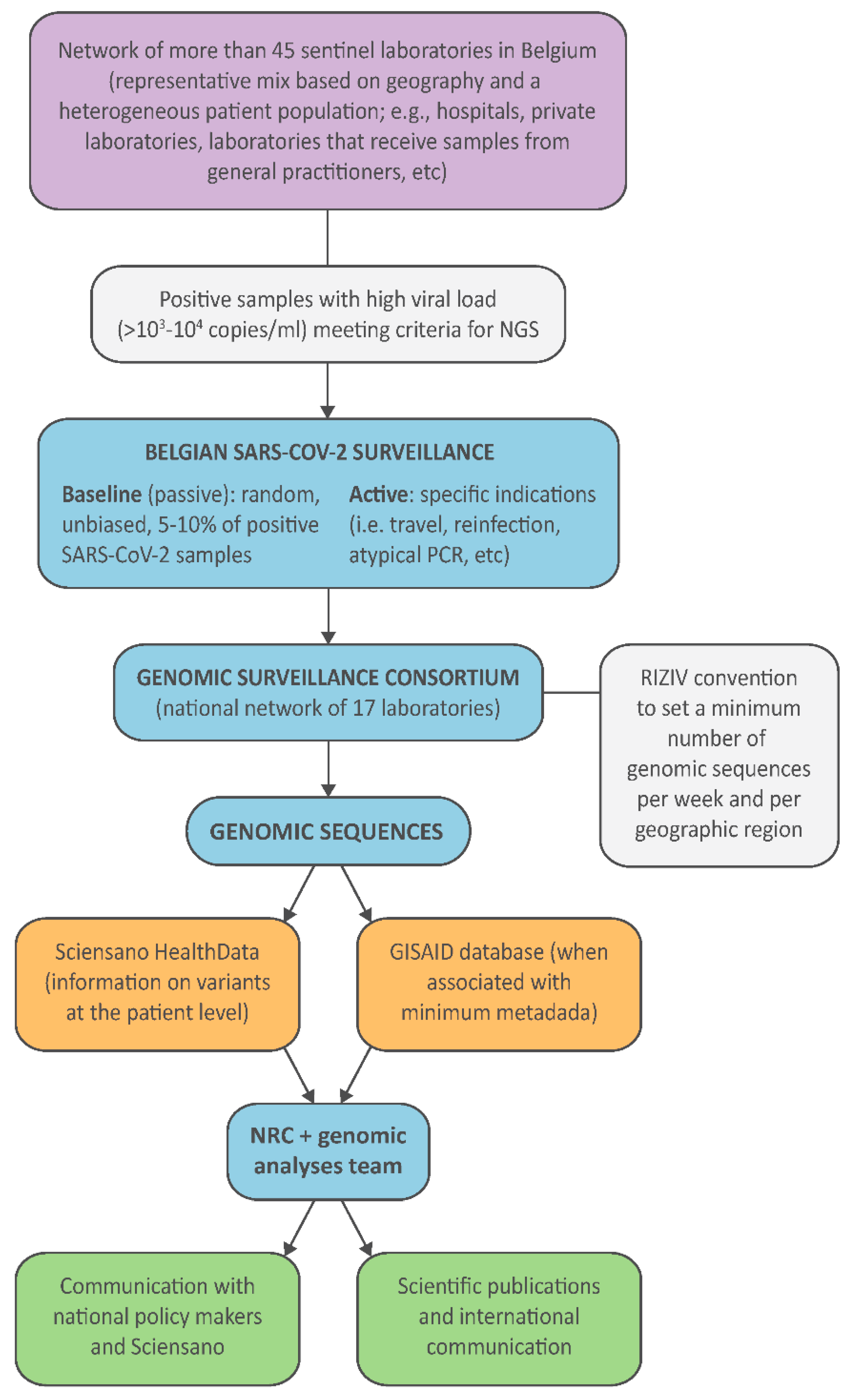
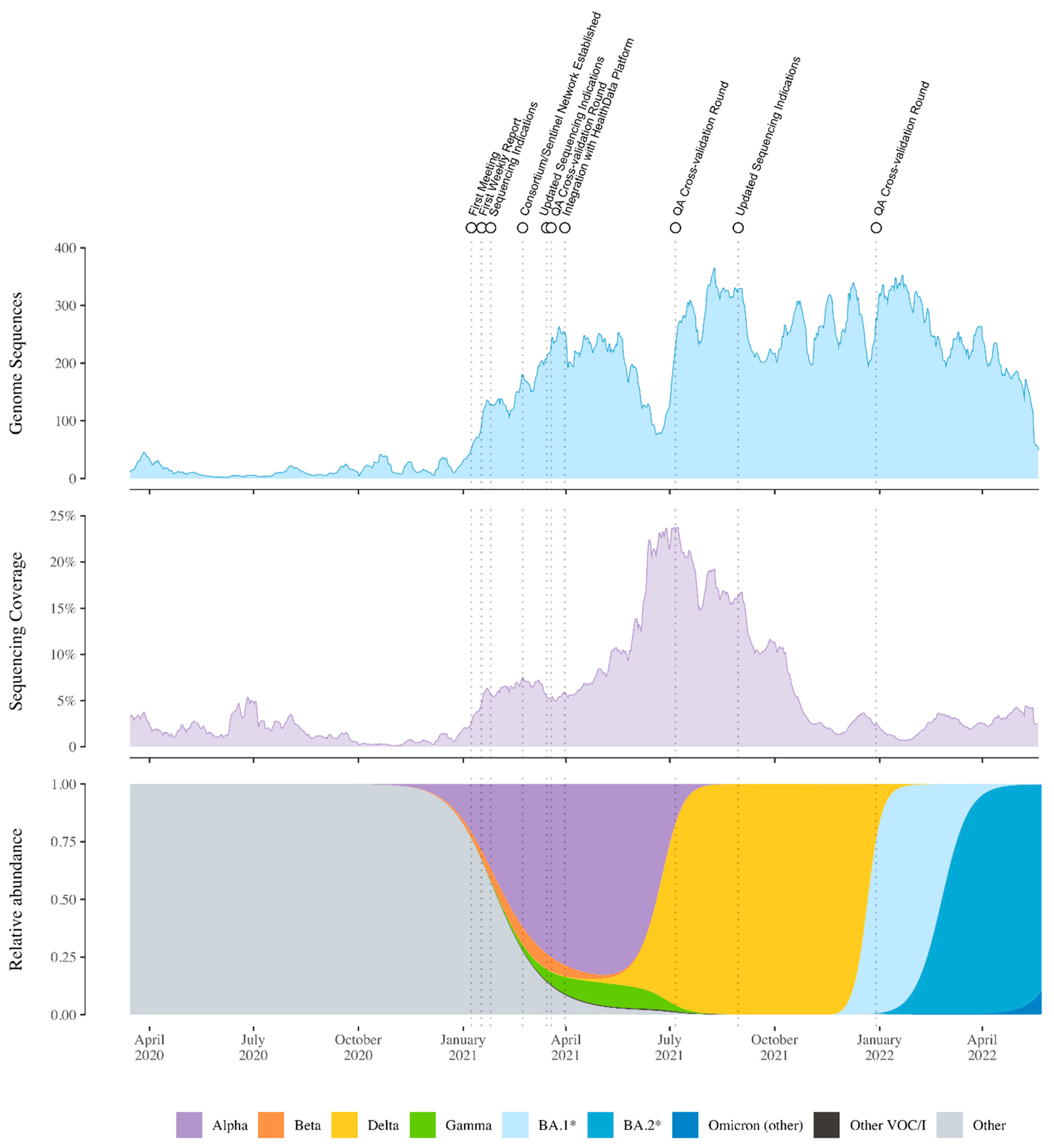
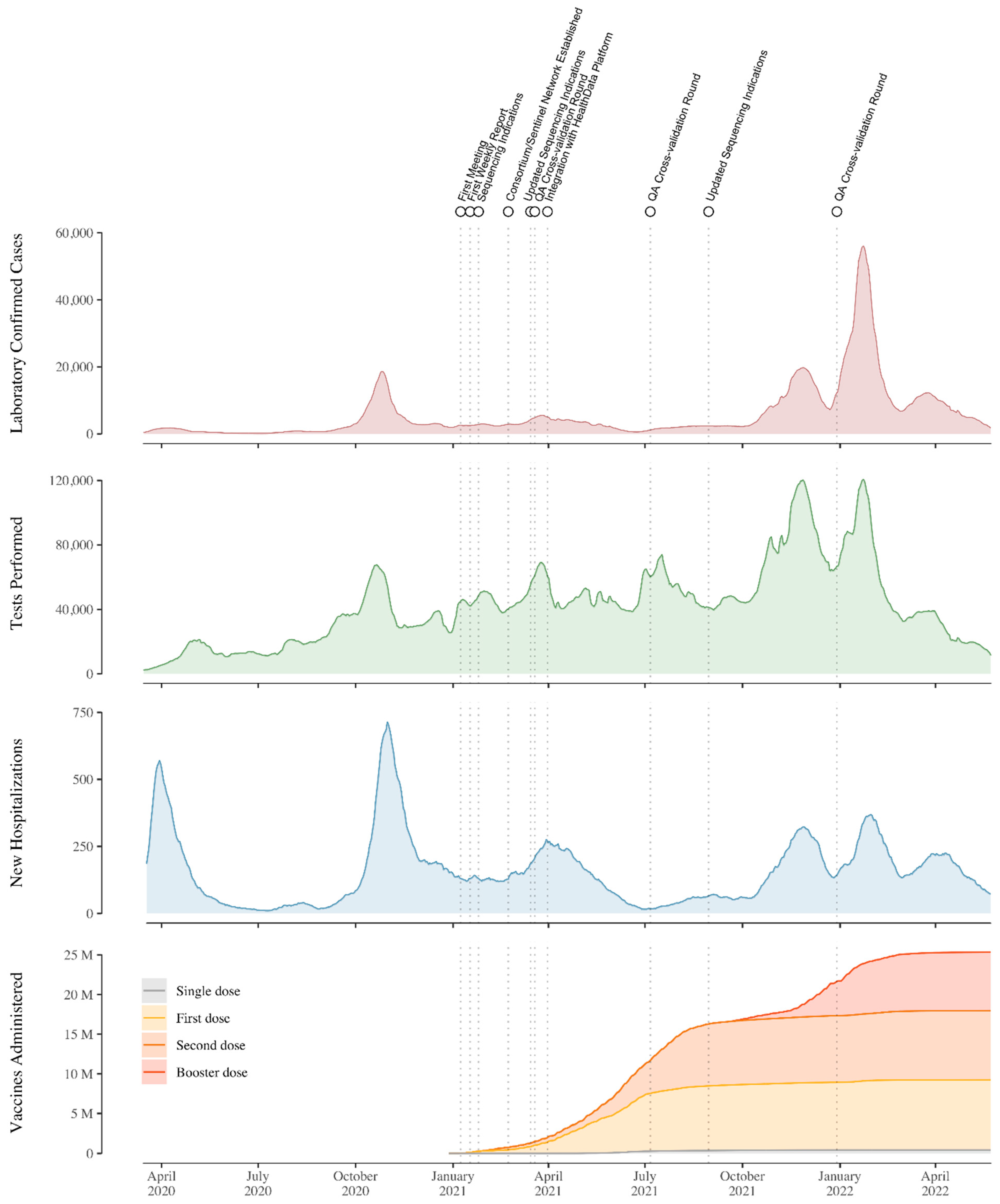
3. Setting Up the National Genomic Surveillance Platform for SARS-CoV-2 in Belgium
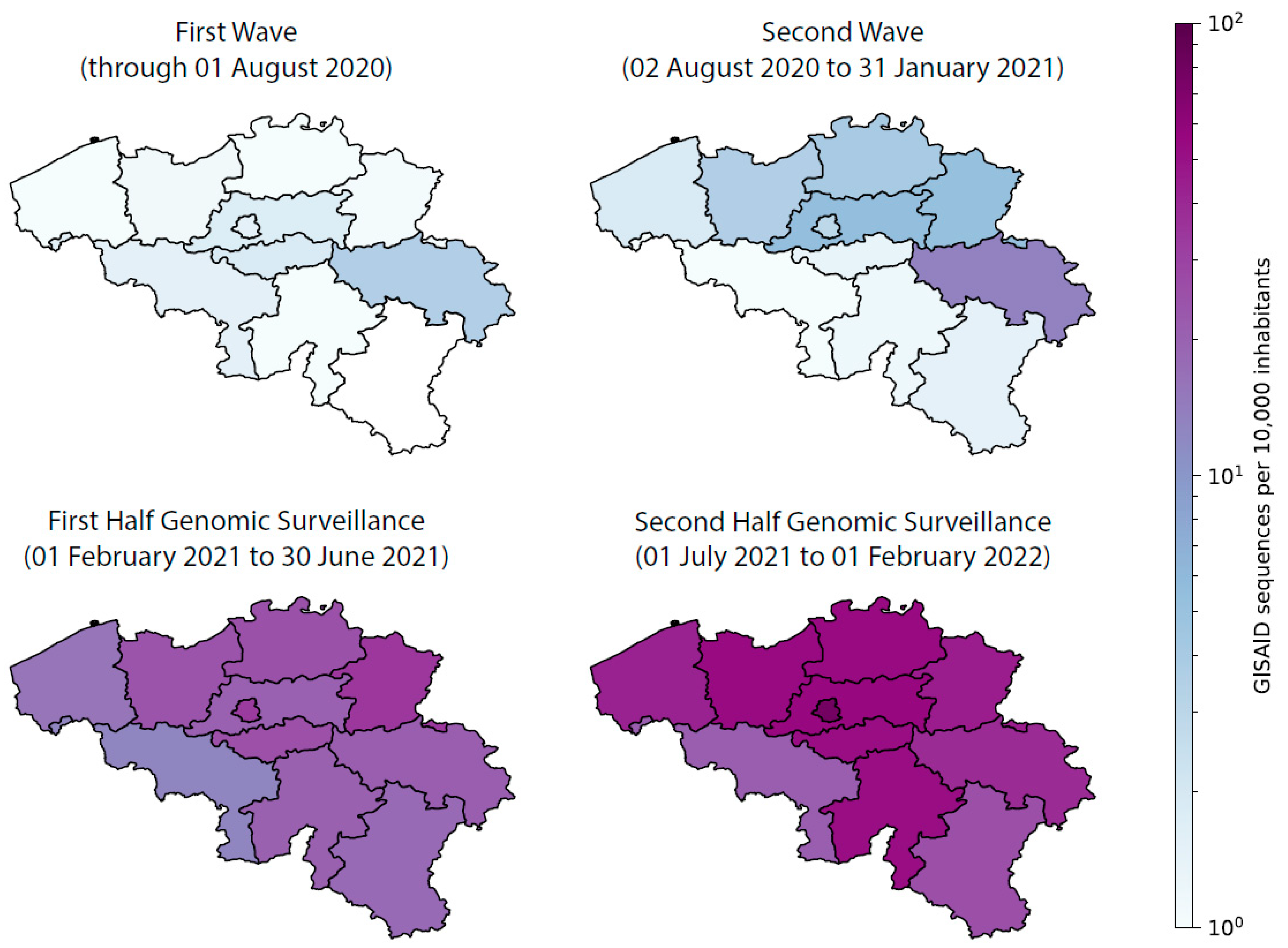
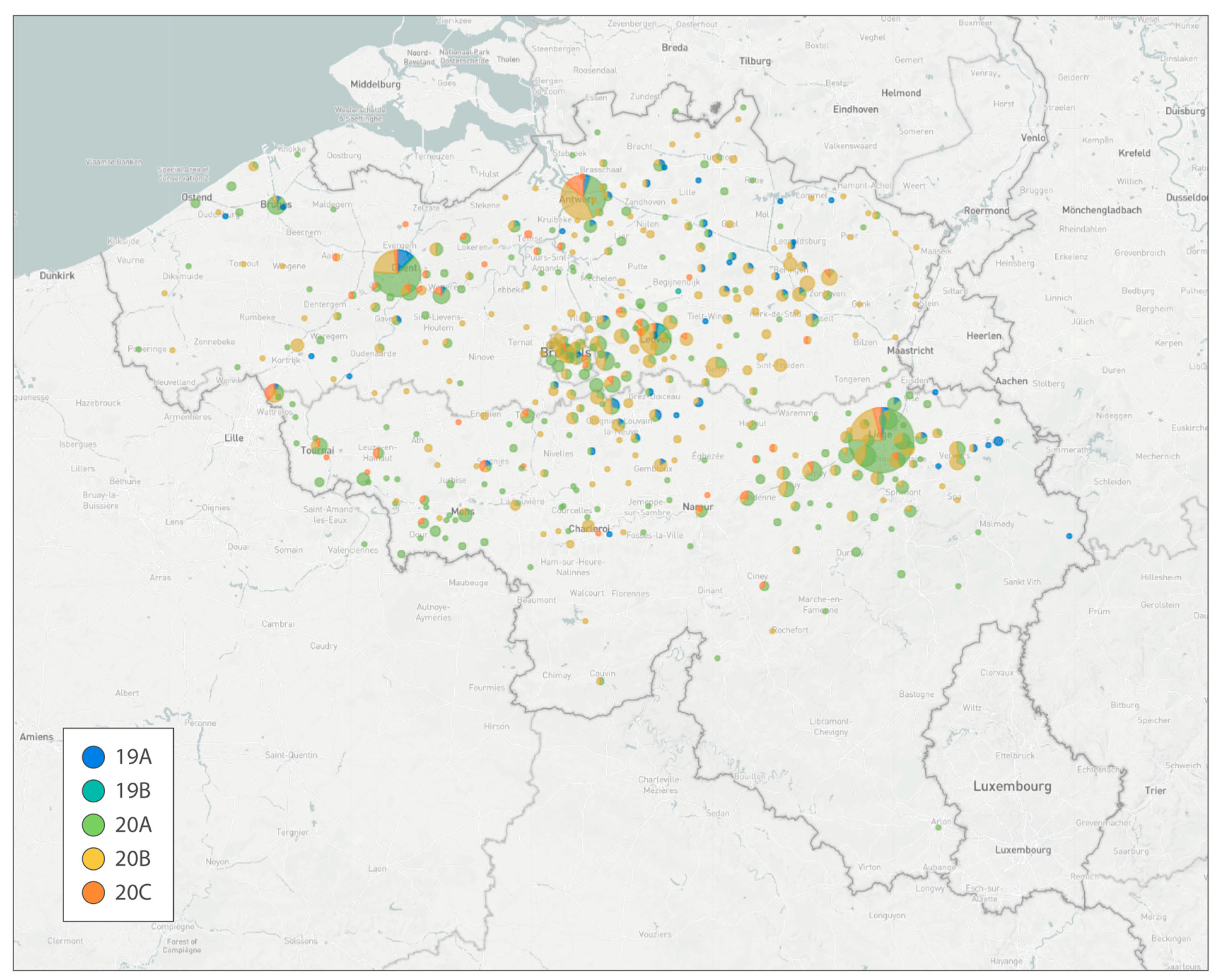
4. Studies Resulting from the Genomic Surveillance Initiative
5. Conclusions and Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zivin, J.G.; Sanders, N. The Spread of COVID-19 Shows the Importance of Policy Coordination. Proc. Natl. Acad. Sci. USA 2020, 117, 32842–32844. [Google Scholar] [CrossRef] [PubMed]
- Korber, B.; Fischer, W.M.; Gnanakaran, S.; Yoon, H.; Theiler, J.; Abfalterer, W.; Hengartner, N.; Giorgi, E.E.; Bhattacharya, T.; Foley, B.; et al. Tracking Changes in SARS-CoV-2 Spike: Evidence That D614G Increases Infectivity of the COVID-19 Virus. Cell 2020, 182, 812–827.e19. [Google Scholar] [CrossRef]
- Plante, J.A.; Liu, Y.; Liu, J.; Xia, H.; Johnson, B.A.; Lokugamage, K.G.; Zhang, X.; Muruato, A.E.; Zou, J.; Fontes-Garfias, C.R.; et al. Spike Mutation D614G Alters SARS-CoV-2 Fitness. Nature 2021, 592, 116–121. [Google Scholar] [CrossRef]
- COG-UK Update on SARS-CoV-2 Spike Mutations of Special Interest Report 1. Available online: https://www.attogene.com/wp-content/uploads/2020/12/Report-1_COG-UK_19-December-2020_SARS-CoV-2-Mutations.pdf (accessed on 1 June 2022).
- Volz, E.; Hill, V.; McCrone, J.T.; Price, A.; Jorgensen, D.; O’Toole, Á.; Southgate, J.; Johnson, R.; Jackson, B.; Nascimento, F.F.; et al. Evaluating the Effects of SARS-CoV-2 Spike Mutation D614G on Transmissibility and Pathogenicity. Cell 2021, 184, 64–75.e11. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control Rapid Increase of a SARS-CoV-2 Variant with Multiple Spike Protein Mutations Observed in the United Kingdom. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/SARS-CoV-2-variant-multiple-spike-protein-mutations-United-Kingdom.pdf (accessed on 1 June 2022).
- Rambaut, A.; Holmes, E.C.; O’Toole, Á.; Hill, V.; McCrone, J.T.; Ruis, C.; du Plessis, L.; Pybus, O.G. A Dynamic Nomenclature Proposal for SARS-CoV-2 Lineages to Assist Genomic Epidemiology. Nat. Microbiol. 2020, 5, 1403–1407. [Google Scholar] [CrossRef] [PubMed]
- Volz, E.; Mishra, S.; Chand, M.; Barrett, J.C.; Johnson, R.; Geidelberg, L.; Hinsley, W.R.; Laydon, D.J.; Dabrera, G.; O’Toole, Á.; et al. Assessing Transmissibility of SARS-CoV-2 Lineage B.1.1.7 in England. Nature 2021, 593, 266–269. [Google Scholar] [CrossRef]
- Tegally, H.; Wilkinson, E.; Giovanetti, M.; Iranzadeh, A.; Fonseca, V.; Giandhari, J.; Doolabh, D.; Pillay, S.; San, E.J.; Msomi, N.; et al. Detection of a SARS-CoV-2 Variant of Concern in South Africa. Nature 2021, 592, 438–443. [Google Scholar] [CrossRef]
- Faria, N.R.; Mellan, T.A.; Whittaker, C.; Claro, I.M.; Candido, D.d.S.; Mishra, S.; Crispim, M.A.E.; Sales, F.C.S.; Hawryluk, I.; McCrone, J.T.; et al. Genomics and Epidemiology of the P.1 SARS-CoV-2 Lineage in Manaus, Brazil. Science 2021, 372, 815–821. [Google Scholar] [CrossRef]
- Mlcochova, P.; Kemp, S.A.; Dhar, M.S.; Papa, G.; Meng, B.; Ferreira, I.A.T.M.; Datir, R.; Collier, D.A.; Albecka, A.; Singh, S.; et al. SARS-CoV-2 B.1.617.2 Delta Variant Replication and Immune Evasion. Nature 2021, 599, 114–119. [Google Scholar] [CrossRef]
- Martin, D.P.; Lytras, S.; Lucaci, A.G.; Maier, W.; Grüning, B.; Shank, S.D.; Weaver, S.; MacLean, O.A.; Orton, R.J.; Lemey, P.; et al. Selection Analysis Identifies Clusters of Unusual Mutational Changes in Omicron Lineage BA.1 That Likely Impact Spike Function. Mol. Biol. Evol. 2022, 39, msac061. [Google Scholar] [CrossRef]
- Viana, R.; Moyo, S.; Amoako, D.G.; Tegally, H.; Scheepers, C.; Althaus, C.L.; Anyaneji, U.J.; Bester, P.A.; Boni, M.F.; Chand, M.; et al. Rapid Epidemic Expansion of the SARS-CoV-2 Omicron Variant in Southern Africa. Nature 2022, 603, 679–686. [Google Scholar] [CrossRef] [PubMed]
- European Centre for Disease Prevention and Control Sequencing of SARS-CoV-2. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/sequencing-of-SARS-CoV-2.pdf (accessed on 3 July 2022).
- Shu, Y.; McCauley, J. GISAID: Global Initiative on Sharing All Influenza Data—From Vision to Reality. Euro. Surveill. 2017, 22, 30494. [Google Scholar] [CrossRef]
- Greaney, A.J.; Loes, A.N.; Crawford, K.H.D.; Starr, T.N.; Malone, K.D.; Chu, H.Y.; Bloom, J.D. Comprehensive Mapping of Mutations in the SARS-CoV-2 Receptor-Binding Domain That Affect Recognition by Polyclonal Human Plasma Antibodies. Cell Host Microbe 2021, 29, 463–476.e6. [Google Scholar] [CrossRef] [PubMed]
- Martin, M.A.; VanInsberghe, D.; Koelle, K. Insights from SARS-CoV-2 Sequences. Science 2021, 371, 466–467. [Google Scholar] [CrossRef] [PubMed]
- Wawina-Bokalanga, T.; Martí-Carreras, J.; Vanmechelen, B.; Bloemen, M.; Wollants, E.; Laenen, L.; Cuypers, L.; Beuselinck, K.; Lagrou, K.; André, E.; et al. Genetic Diversity and Evolution of SARS-CoV-2 in Belgium during the First Wave Outbreak. bioRxiv 2021, bioRxiv:2021.06.29.450330. [Google Scholar]
- Hadfield, J.; Megill, C.; Bell, S.M.; Huddleston, J.; Potter, B.; Callender, C.; Sagulenko, P.; Bedford, T.; Neher, R.A. Nextstrain: Real-Time Tracking of Pathogen Evolution. Bioinformatics 2018, 34, 4121–4123. [Google Scholar] [CrossRef]
- Alm, E.; Broberg, E.K.; Connor, T.; Hodcroft, E.B.; Komissarov, A.B.; Maurer-Stroh, S.; Melidou, A.; Neher, R.A.; O’Toole, Á.; Pereyaslov, D.; et al. Geographical and Temporal Distribution of SARS-CoV-2 Clades in the WHO European Region, January to June 2020. Eur. Surveill. 2020, 25, 2001410. [Google Scholar] [CrossRef]
- Dellicour, S.; Durkin, K.; Hong, S.L.; Vanmechelen, B.; Martí-Carreras, J.; Gill, M.S.; Meex, C.; Bontems, S.; André, E.; Gilbert, M.; et al. A Phylodynamic Workflow to Rapidly Gain Insights into the Dispersal History and Dynamics of SARS-CoV-2 Lineages. Mol. Biol. Evol. 2021, 38, 1608–1613. [Google Scholar] [CrossRef]
- Bollen, N.; Artesi, M.; Durkin, K.; Hong, S.L.; Potter, B.; Boujemla, B.; Vanmechelen, B.; Martí-Carreras, J.; Wawina-Bokalanga, T.; Meex, C.; et al. Exploiting Genomic Surveillance to Map the Spatio-Temporal Dispersal of SARS-CoV-2 Spike Mutations in Belgium across 2020. Sci. Rep. 2021, 11, 18580. [Google Scholar] [CrossRef]
- Pirnay, J.-P.; Selhorst, P.; Cochez, C.; Petrillo, M.; Claes, V.; Van der Beken, Y.; Verbeken, G.; Degueldre, J.; T’Sas, F.; Van den Eede, G.; et al. Study of a SARS-CoV-2 Outbreak in a Belgian Military Education and Training Center in Maradi, Niger. Viruses 2020, 12, 949. [Google Scholar] [CrossRef] [PubMed]
- Van Cleemput, J.; van Snippenberg, W.; Lambrechts, L.; Dendooven, A.; D’Onofrio, V.; Couck, L.; Trypsteen, W.; Vanrusselt, J.; Theuns, S.; Vereecke, N.; et al. Organ-Specific Genome Diversity of Replication-Competent SARS-CoV-2. Nat. Commun. 2021, 12, 6612. [Google Scholar] [CrossRef]
- Vuylsteke, B.; Cuypers, L.; Baele, G.; Stranger, M.; Paralovo, S.L.; André, E.; Dirks, J.; Maes, P.; Laga, M. The Role of Airborne Transmission in a Large Single Source Outbreak of SARS-CoV-2 in a Belgian Nursing Home in 2020. Epidemics 2022, 40, 100589. [Google Scholar] [CrossRef]
- Lemey, P.; Hong, S.L.; Hill, V.; Baele, G.; Poletto, C.; Colizza, V.; O’Toole, Á.; McCrone, J.T.; Andersen, K.G.; Worobey, M.; et al. Accommodating Individual Travel History and Unsampled Diversity in Bayesian Phylogeographic Inference of SARS-CoV-2. Nat. Commun. 2020, 11, 5110. [Google Scholar] [CrossRef] [PubMed]
- Lemey, P.; Ruktanonchai, N.; Hong, S.L.; Colizza, V.; Poletto, C.; Van den Broeck, F.; Gill, M.S.; Ji, X.; Levasseur, A.; Oude Munnink, B.B.; et al. Untangling Introductions and Persistence in COVID-19 Resurgence in Europe. Nature 2021, 595, 713–717. [Google Scholar] [CrossRef]
- Brito, A.F.; Semenova, E.; Dudas, G.; Hassler, G.W.; Kalinich, C.C.; Kraemer, M.U.G.; Ho, J.; Tegally, H.; Githinji, G.; Agoti, C.N.; et al. Global Disparities in SARS-CoV-2 Genomic Surveillance. medRxiv 2021. [Google Scholar] [CrossRef]
- Van der Moeren, N.; Selhorst, P.; Ha, M.; Heireman, L.; Van Gaal, P.-J.; Breems, D.; Meysman, P.; Laukens, K.; Verstrepen, W.; Van Gasse, N.; et al. Viral Evolution and Immunology of SARS-CoV-2 in a Persistent Infection after Treatment with Rituximab. Viruses 2022, 14, 752. [Google Scholar] [CrossRef] [PubMed]
- Van Goethem, N.; Vandromme, M.; Van Oyen, H.; Haarhuis, F.; Brondeel, R.; Catteau, L.; André, E.; Cuypers, L.; Belgian Collaborative Group on COVID-19 Hospital surveillance; COVID-19 Genomics Belgium consortium; et al. Severity of Infection with the SARS-CoV-2 B.1.1.7 Lineage among Hospitalized COVID-19 Patients in Belgium. PLoS ONE 2022, 17, e0269138. [Google Scholar] [CrossRef]
- Van Goethem, N.; Chung, P.Y.J.; Meurisse, M.; Vandromme, M.; De Mot, L.; Brondeel, R.; Stouten, V.; Klamer, S.; Cuypers, L.; Braeye, T.; et al. Clinical Severity of SARS-CoV-2 Omicron Variant Compared with Delta among Hospitalized COVID-19 Patients in Belgium during Autumn and Winter Season 2021–2022. Viruses 2022, 14, 1297. [Google Scholar] [CrossRef] [PubMed]
- Pirnay, J.-P.; Selhorst, P.; Hong, S.L.; Cochez, C.; Potter, B.; Maes, P.; Petrillo, M.; Dudas, G.; Claes, V.; der Beken, Y.V.; et al. Variant Analysis of SARS-CoV-2 Genomes from Belgian Military Personnel Engaged in Overseas Missions and Operations. Viruses 2021, 13, 1359. [Google Scholar] [CrossRef] [PubMed]
- Kaleta, T.; Kern, L.; Hong, S.L.; Hölzer, M.; Kochs, G.; Beer, J.; Schnepf, D.; Schwemmle, M.; Bollen, N.; Kolb, P.; et al. Antibody Escape and Global Spread of SARS-CoV-2 Lineage A.27. Nat. Commun. 2022, 13, 1152. [Google Scholar] [CrossRef] [PubMed]
- Van Elslande, J.; Kerckhofs, F.; Cuypers, L.; Wollants, E.; Potter, B.; Vankeerberghen, A.; Cattoir, L.; Holderbeke, A.; Behillil, S.; Gorissen, S.; et al. Two Separate Clusters of SARS-CoV-2 Delta Variant Infections in a Group of 41 Students Travelling from India: An Illustration of the Need for Rigorous Testing and Quarantine. Viruses 2022, 14, 1198. [Google Scholar] [CrossRef]
- Cuypers, L.; Keyaerts, E.; Hong, S.; Gorissen, S.; Menezes, S.M.; Starick, M.; Van Elslande, J.; Weemaes, M.; Wawina-Bokalanga, T.; Vanmechelen, B.; et al. Comprehensive Immunovirological and Environmental Screening Reveals Risk Factors for Fatal COVID-19 during Post-Vaccination Nursing Home Outbreaks. Res. Sq. 2022. [Google Scholar] [CrossRef]
- Vanmechelen, B.; Logist, A.-S.; Wawina-Bokalanga, T.; Verlinden, J.; Martí-Carreras, J.; Geenen, C.; Slechten, B.; Cuypers, L.; André, E.; Baele, G.; et al. Identification of the First SARS-CoV-2 Lineage B.1.1.529 Virus Detected in Europe. Microbiol. Resour. Announc. 2022, 11, e0116121. [Google Scholar] [CrossRef] [PubMed]
- Planas, D.; Saunders, N.; Maes, P.; Guivel-Benhassine, F.; Planchais, C.; Buchrieser, J.; Bolland, W.-H.; Porrot, F.; Staropoli, I.; Lemoine, F.; et al. Considerable Escape of SARS-CoV-2 Omicron to Antibody Neutralization. Nature 2022, 602, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Ariën, K.K.; Heyndrickx, L.; Michiels, J.; Vereecken, K.; Van Lent, K.; Coppens, S.; Willems, B.; Pannus, P.; Martens, G.A.; Van Esbroeck, M.; et al. Three Doses of BNT162b2 Vaccine Confer Neutralising Antibody Capacity against the SARS-CoV-2 Omicron Variant. NPJ Vaccines 2022, 7, 35. [Google Scholar] [CrossRef]
- Bruel, T.; Hadjadj, J.; Maes, P.; Planas, D.; Seve, A.; Staropoli, I.; Guivel-Benhassine, F.; Porrot, F.; Bolland, W.-H.; Nguyen, Y.; et al. Serum Neutralization of SARS-CoV-2 Omicron Sublineages BA.1 and BA.2 in Patients Receiving Monoclonal Antibodies. Nat. Med. 2022, 28, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Wawina-Bokalanga, T.; Logist, A.-S.; Sinnesael, R.; Van Holm, B.; Delforge, M.-L.; Struyven, P.; Cuypers, L.; André, E.; Baele, G.; Maes, P. Genomic Evidence of Co-Identification with Omicron and Delta SARS-CoV-2 Variants: A Report of Two Cases. Int. J. Infect. Dis. 2022, 122, 212–214. [Google Scholar] [CrossRef] [PubMed]
- Rehman, S.U.; Shafique, L.; Ihsan, A.; Liu, Q. Evolutionary Trajectory for the Emergence of Novel Coronavirus SARS-CoV-2. Pathogens 2020, 9, 240. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control Evaluation of the SARS-CoV-2 Testing Policy in Belgium from June to December 2021. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/covid-19-evaluation-SARS-CoV-2-testing-policy-Belgium-Feb-2022.pdf (accessed on 3 August 2022).
- Maher, M.C.; Bartha, I.; Weaver, S.; di Iulio, J.; Ferri, E.; Soriaga, L.; Lempp, F.A.; Hie, B.L.; Bryson, B.; Berger, B.; et al. Predicting the Mutational Drivers of Future SARS-CoV-2 Variants of Concern. Sci. Transl. Med. 2022, 14, eabk3445. [Google Scholar] [CrossRef]
- Subissi, L.; von Gottberg, A.; Thukral, L.; Worp, N.; Oude Munnink, B.B.; Rathore, S.; Abu-Raddad, L.J.; Aguilera, X.; Alm, E.; Archer, B.N.; et al. An Early Warning System for Emerging SARS-CoV-2 Variants. Nat. Med. 2022, 28, 1110–1115. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuypers, L.; Dellicour, S.; Hong, S.L.; Potter, B.I.; Verhasselt, B.; Vereecke, N.; Lambrechts, L.; Durkin, K.; Bours, V.; Klamer, S.; et al. Two Years of Genomic Surveillance in Belgium during the SARS-CoV-2 Pandemic to Attain Country-Wide Coverage and Monitor the Introduction and Spread of Emerging Variants. Viruses 2022, 14, 2301. https://doi.org/10.3390/v14102301
Cuypers L, Dellicour S, Hong SL, Potter BI, Verhasselt B, Vereecke N, Lambrechts L, Durkin K, Bours V, Klamer S, et al. Two Years of Genomic Surveillance in Belgium during the SARS-CoV-2 Pandemic to Attain Country-Wide Coverage and Monitor the Introduction and Spread of Emerging Variants. Viruses. 2022; 14(10):2301. https://doi.org/10.3390/v14102301
Chicago/Turabian StyleCuypers, Lize, Simon Dellicour, Samuel L. Hong, Barney I. Potter, Bruno Verhasselt, Nick Vereecke, Laurens Lambrechts, Keith Durkin, Vincent Bours, Sofieke Klamer, and et al. 2022. "Two Years of Genomic Surveillance in Belgium during the SARS-CoV-2 Pandemic to Attain Country-Wide Coverage and Monitor the Introduction and Spread of Emerging Variants" Viruses 14, no. 10: 2301. https://doi.org/10.3390/v14102301
APA StyleCuypers, L., Dellicour, S., Hong, S. L., Potter, B. I., Verhasselt, B., Vereecke, N., Lambrechts, L., Durkin, K., Bours, V., Klamer, S., Bayon-Vicente, G., Vael, C., Ariën, K. K., De Mendonca, R., Soetens, O., Michel, C., Bearzatto, B., Naesens, R., Gras, J., ... Baele, G. (2022). Two Years of Genomic Surveillance in Belgium during the SARS-CoV-2 Pandemic to Attain Country-Wide Coverage and Monitor the Introduction and Spread of Emerging Variants. Viruses, 14(10), 2301. https://doi.org/10.3390/v14102301






