Phage Therapy for Multi-Drug Resistant Respiratory Tract Infections
Abstract
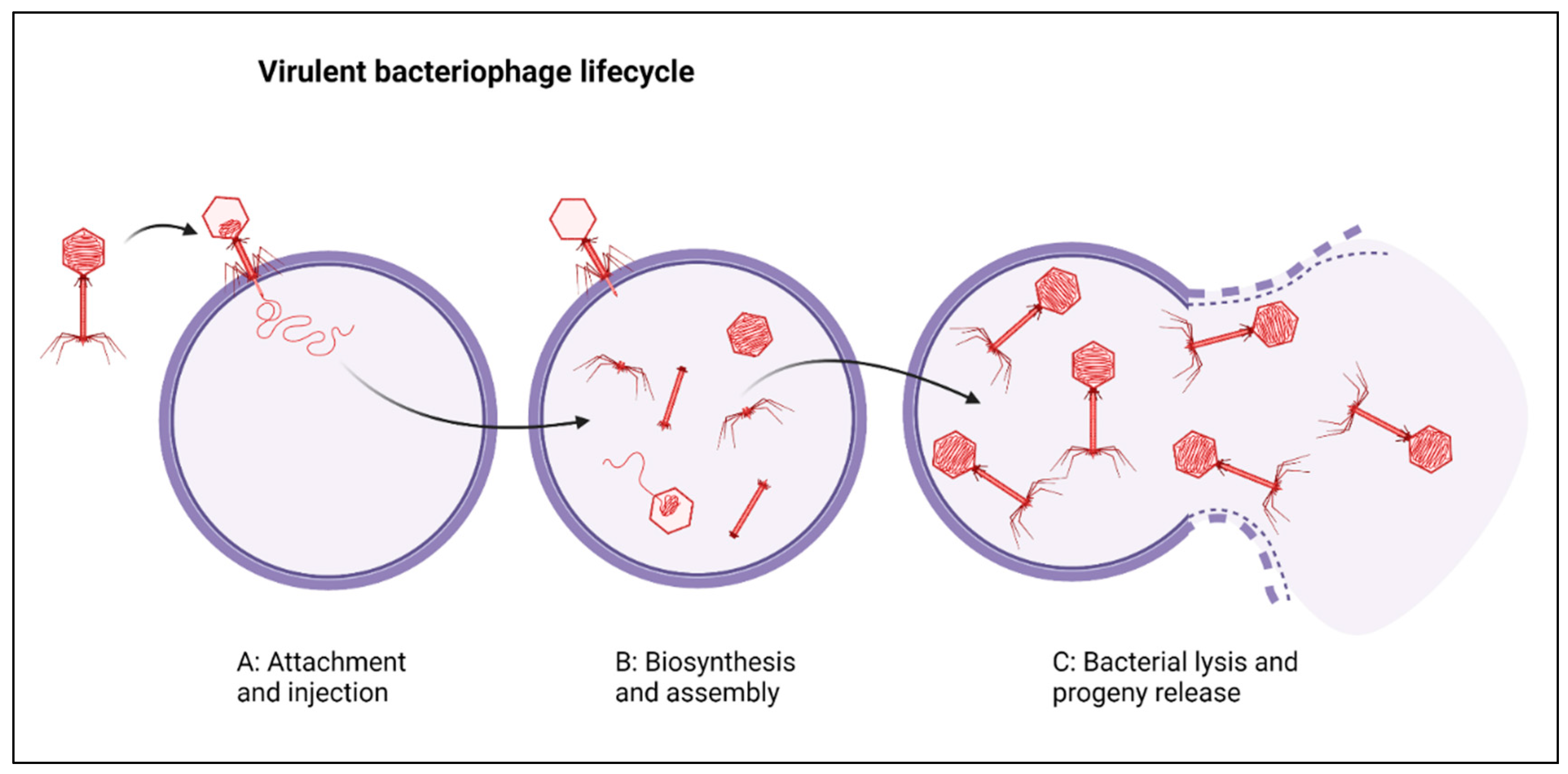
:1. Introduction
2. Pulmonary Phage Therapy
Pulmonary Delivery
3. Preclinical Data Generation
3.1. In Vitro Models
3.2. In Vivo Models
4. Clinical Trials Involving Phage for Respiratory Infections
5. Resistance
6. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azoulay, E.; the Nine-i Investigators; Russell, L.; Van De Louw, A.; Metaxa, V.; Bauer, P.; Povoa, P.; Montero, J.G.; Loeches, I.M.; Mehta, S.; et al. Diagnosis of severe respiratory infections in immunocompromised patients. Intensiv. Care Med. 2020, 46, 298–314. [Google Scholar] [CrossRef] [Green Version]
- Pragman, A.A.; Berger, J.P.; Williams, B.J. Understanding Persistent Bacterial Lung Infections. Clin. Pulm. Med. 2016, 23, 57–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Orgeur, M.; Brosch, R. Evolution of virulence in the Mycobacterium tuberculosis complex. Curr. Opin. Microbiol. 2018, 41, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Troeger, C.; Forouzanfar, M.; Rao, P.C.; Khalil, I.; Brown, A.; Swartz, S.; Fullman, N.; Mosser, J.; Thompson, R.L.; Reiner, R.C.; et al. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of lower respiratory tract infections in 195 countries: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Infect. Dis. 2017, 17, 1133–1161. [Google Scholar] [CrossRef] [Green Version]
- Chang, R.Y.K.; Wallin, M.; Lin, Y.; Leung, S.; Wang, H.; Morales, S.; Chan, H.-K. Phage therapy for respiratory infections. Adv. Drug Deliv. Rev. 2018, 133, 76–86. [Google Scholar] [CrossRef]
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; E Kretzschmar, M.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2018, 19, 56–66. [Google Scholar] [CrossRef] [Green Version]
- Principi, N.; Silvestri, E.; Esposito, S. Advantages and Limitations of Bacteriophages for the Treatment of Bacterial Infections. Front. Pharmacol. 2019, 10, 513. [Google Scholar] [CrossRef] [Green Version]
- Melo, L.D.R.; Oliveira, H.; Pires, D.P.; Dabrowska, K.; Azeredo, J. Phage therapy efficacy: A review of the last 10 years of preclinical studies. Crit. Rev. Microbiol. 2020, 46, 78–99. [Google Scholar] [CrossRef]
- Hoe, S.; Semler, D.D.; Goudie, A.D.; Lynch, K.H.; Matinkhoo, S.; Finlay, W.H.; Dennis, J.; Vehring, R. Respirable Bacteriophages for the Treatment of Bacterial Lung Infections. J. Aerosol Med. Pulm. Drug Deliv. 2013, 26, 317–335. [Google Scholar] [CrossRef]
- Wittebole, X.; De Roock, S.; Opal, S.M. A historical overview of bacteriophage therapy as an alternative to antibiotics for the treatment of bacterial pathogens. Virulence 2013, 5, 226–235. [Google Scholar] [CrossRef]
- Sulakvelidze, A.; Alavidze, Z.; Morris, J.G. Bacteriophage Therapy. Antimicrob. Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef] [Green Version]
- Wienhold, S.-M.; Lienau, J.; Witzenrath, M. Towards Inhaled Phage Therapy in Western Europe. Viruses 2019, 11, 295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bodner, K.; Melkonian, A.L.; Covert, M.W. The Enemy of My Enemy: New Insights Regarding Bacteriophage–Mammalian Cell Interactions. Trends Microbiol. 2020, 29, 528–541. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Międzybrodzki, R.; Żaczek, M.; Borysowski, J. Phages in the fight against COVID-19? Futur. Microbiol. 2020, 15, 1095–1100. [Google Scholar] [CrossRef] [PubMed]
- Law, N.; Logan, C.; Yung, G.; Furr, C.-L.L.; Lehman, S.M.; Morales, S.; Rosas, F.; Gaidamaka, A.; Bilinsky, I.; Grint, P.; et al. Successful adjunctive use of bacteriophage therapy for treatment of multidrug-resistant Pseudomonas aeruginosa infection in a cystic fibrosis patient. Infection 2019, 47, 665–668. [Google Scholar] [CrossRef]
- Gainey, A.B.; Burch, A.; Brownstein, M.J.; Brown, D.E.; Ms, J.F.; Bs, B.H.; Biswas, B.; Bivens, B.N.; Malagon, F.; Daniels, R. Combining bacteriophages with cefiderocol and meropenem/vaborbactam to treat a pan-drug resistant Achromobacter species infection in a pediatric cystic fibrosis patient. Pediatr. Pulmonol. 2020, 55. [Google Scholar] [CrossRef] [PubMed]
- Trend, S.; Fonceca, A.M.; Ditcham, W.G.; Kicic, A.; Cf, A. The potential of phage therapy in cystic fibrosis: Essential human-bacterial-phage interactions and delivery considerations for use in Pseudomonas aeruginosa-infected airways. J. Cyst. Fibros. 2017, 16, 663–670. [Google Scholar] [CrossRef] [Green Version]
- Rhoads, D.; Wolcott, R.; Kuskowski, M.; Wolcott, B.; Ward, L.; Sulakvelidze, A. Bacteriophage therapy of venous leg ulcers in humans: Results of a phase I safety trial. J. Wound Care 2009, 18, 237–243. [Google Scholar] [CrossRef]
- Wright, A.; Hawkins, C.; Änggård, E.; Harper, D. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistantPseudomonas aeruginosa; a preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef]
- Kutateladze, M.; Adamia, R. Phage therapy experience at the Eliava Institute. Médecine Mal. Infect. 2008, 38, 426–430. [Google Scholar] [CrossRef]
- Emattila, S.; Eruotsalainen, P.; Ejalasvuori, M. On-Demand Isolation of Bacteriophages Against Drug-Resistant Bacteria for Personalized Phage Therapy. Front. Microbiol. 2015, 6, 1271. [Google Scholar] [CrossRef] [Green Version]
- Jurczak-Kurek, A.; Gąsior, T.; Nejman-Faleńczyk, B.; Bloch, S.; Dydecka, A.; Topka, G.; Necel, A.; Jakubowska-Deredas, M.; Narajczyk, M.; Richert, M.; et al. Biodiversity of bacteriophages: Morphological and biological properties of a large group of phages isolated from urban sewage. Sci. Rep. 2016, 6, 34338. [Google Scholar] [CrossRef] [PubMed]
- González-Menéndez, E.; Fernández, L.; Gutiérrez, D.; Rodríguez, A.; Martínez, B.; García, P. Comparative analysis of different preservation techniques for the storage of Staphylococcus phages aimed for the industrial development of phage-based antimicrobial products. PLoS ONE 2018, 13, e0205728. [Google Scholar] [CrossRef]
- Furfaro, L.L.; Payne, M.S.; Chang, B.J. Bacteriophage Therapy: Clinical Trials and Regulatory Hurdles. Front. Cell. Infect. Microbiol. 2018, 8, 376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCallin, S.; Sacher, J.; Zheng, J.; Chan, B.K. Current State of Compassionate Phage Therapy. Viruses 2019, 11, 343. [Google Scholar] [CrossRef] [Green Version]
- Fabijan, A.P.; Lin, R.C.Y.; Ho, J.; Maddocks, S.; Ben Zakour, N.L.; Iredell, J.R.; Khalid, A.; Venturini, C.; Chard, R.; Morales, S.; et al. Safety of bacteriophage therapy in severe Staphylococcus aureus infection. Nat. Microbiol. 2020, 5, 465–472. [Google Scholar] [CrossRef]
- Boucher, R.C. Muco-Obstructive Lung Diseases. New Engl. J. Med. 2019, 380, 1941–1953. [Google Scholar] [CrossRef] [PubMed]
- Łubowska, N.; Grygorcewicz, B.; Kosznik-Kwaśnicka, K.; Zauszkiewicz-Pawlak, A.; Węgrzyn, A.; Dołęgowska, B.; Piechowicz, L. Characterization of the Three New Kayviruses and Their Lytic Activity Against Multidrug-Resistant Staphylococcus aureus. Microorganisms 2019, 7, 471. [Google Scholar] [CrossRef] [Green Version]
- Van Norman, G.A. Drugs, Devices, and the FDA: Part 1. JACC: Basic Transl. Sci. 2016, 1, 170–179. [Google Scholar] [CrossRef] [Green Version]
- O’Neill, J. Tackling a Crisis for the Health and Wealth of Nations; Review on Antimicrobial Resistance: London, UK, 2014. [Google Scholar]
- Kim, H.; Chang, R.; Morales, S.; Chan, H.-K. Bacteriophage-Delivering Hydrogels: Current Progress in Combating Antibiotic Resistant Bacterial Infection. Antibiotics 2021, 10, 130. [Google Scholar] [CrossRef]
- Chang, R.Y.K.; Wallin, M.; Kutter, E.; Morales, S.; Britton, W.; Li, J.; Chan, H.-K. Storage stability of inhalable phage powders containing lactose at ambient conditions. Int. J. Pharm. 2019, 560, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Astudillo, A.; Leung, S.S.Y.; Kutter, E.; Morales, S.; Chan, H.-K. Nebulization effects on structural stability of bacteriophage PEV 44. Eur. J. Pharm. Biopharm. 2018, 125, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Leung, S.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Carter, E.; Britton, W.J.; Kutter, E.; Chan, H.-K. Jet nebulization of bacteriophages with different tail morphologies–Structural effects. Int. J. Pharm. 2018, 554, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Semler, D.D.; Goudie, A.D.; Finlay, W.H.; Dennis, J.J. Aerosol Phage Therapy Efficacy in Burkholderia cepacia Complex Respiratory Infections. Antimicrob. Agents Chemother. 2014, 58, 4005–4013. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, R.Y.K.; Wong, J.; Mathai, A.; Morales, S.; Kutter, E.; Britton, W.; Li, J.; Chan, H.-K. Production of highly stable spray dried phage formulations for treatment of Pseudomonas aeruginosa lung infection. Eur. J. Pharm. Biopharm. 2017, 121, 1–13. [Google Scholar] [CrossRef]
- Leung, S.; Parumasivan, T.; Gao, F.G.; Carter, E.; Carrigy, N.B.; Vehring, R.; Finlay, W.H.; Morales, S.; Britton, W.J.; Kutter, E.; et al. Effects of storage conditions on the stability of spray dried, inhalable bacteriophage powders. Int. J. Pharm. 2017, 521, 141–149. [Google Scholar] [CrossRef] [Green Version]
- Chang, R.Y.K.; Chen, K.; Wang, J.; Wallin, M.; Britton, W.; Morales, S.; Kutter, E.; Li, J.; Chan, H.-K. Proof-of-Principle Study in a Murine Lung Infection Model of Antipseudomonal Activity of Phage PEV20 in a Dry-Powder Formulation. Antimicrob. Agents Chemother. 2018, 62, e01714-17. [Google Scholar] [CrossRef] [Green Version]
- Abatángelo, V.; Bacci, N.P.; Boncompain, C.A.; Amadio, A.F.; Carrasco, S.; Suárez, C.A.; Morbidoni, H.R. Broad-range lytic bacteriophages that kill Staphylococcus aureus local field strains. PLoS ONE 2017, 12, e0181671. [Google Scholar] [CrossRef]
- Merabishvili, M.; Pirnay, J.-P.; Verbeken, G.; Chanishvili, N.; Tediashvili, M.; Lashkhi, N.; Glonti, T.; Krylov, V.; Mast, J.; Van Parys, L.; et al. Quality-Controlled Small-Scale Production of a Well-Defined Bacteriophage Cocktail for Use in Human Clinical Trials. PLoS ONE 2009, 4, e4944. [Google Scholar] [CrossRef]
- Lehman, S.M.; Mearns, G.; Rankin, D.; Cole, R.A.; Smrekar, F.; Branston, S.D.; Morales, S. Design and Preclinical Development of a Phage Product for the Treatment of Antibiotic-Resistant Staphylococcus aureus Infections. Viruses 2019, 11, 88. [Google Scholar] [CrossRef] [Green Version]
- Jamal, M.; Hussain, T.; Das, C.R.; Andleeb, S. Characterization of Siphoviridae phage Z and studying its efficacy against multidrug-resistant Klebsiella pneumoniae planktonic cells and biofilm. J. Med Microbiol. 2015, 64, 454–462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, J.E.; Kim, J.H.; Hwang, S.Y.; Choresca, C.H.; Shin, S.P.; Jun, J.W.; Chai, J.Y.; Park, Y.H.; Park, S.C. Isolation and characterization of a Myoviridae bacteriophage against Staphylococcus aureus isolated from dairy cows with mastitis. Res. Veter- Sci. 2013, 95, 758–763. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; McAuliffe, O.; Ross, R.; Coffey, A. Prevention of Staphylococcus aureus biofilm formation and reduction in established biofilm density using a combination of phage K and modified derivatives. Lett. Appl. Microbiol. 2012, 54, 286–291. [Google Scholar] [CrossRef] [PubMed]
- Ferriol-González, C.; Domingo-Calap, P. Phages for Biofilm Removal. Antibiotics 2020, 9, 268. [Google Scholar] [CrossRef]
- Trend, S.; Chang, B.J.; O’Dea, M.; Stick, S.; Kicic, A.; WAERP; AusREC; AREST CF. Use of a Primary Epithelial Cell Screening Tool to Investigate Phage Therapy in Cystic Fibrosis. Front. Pharmacol. 2018, 9, 1330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, H.; Birch, N.P.; Suresh, V. An Optimised Human Cell Culture Model for Alveolar Epithelial Transport. PLoS ONE 2016, 11, e0165225. [Google Scholar] [CrossRef] [PubMed]
- Garratt, L.W.; Sutanto, E.N.; Foo, C.J.; Ling, K.M.; Looi, K.; Kicic-Starcevich, E.; Iosifidis, T.; Martinovich, K.M.; Lannigan, F.J.; Stick, S.M.; et al. Determinants of culture success in an airway epithelium sampling program of young children with cystic fibrosis. Exp. Lung Res. 2014, 40, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Martinovich, K.; Iosifidis, T.; Buckley, A.G.; Looi, K.; Ling, K.-M.; Sutanto, E.N.; Kicic-Starcevich, E.; Garratt, L.W.; Shaw, N.C.; Montgomery, S.; et al. Conditionally reprogrammed primary airway epithelial cells maintain morphology, lineage and disease specific functional characteristics. Sci. Rep. 2017, 7, 1–13. [Google Scholar] [CrossRef]
- Looi, K.; Troy, N.M.; Garratt, L.W.; Iosifidis, T.; Bosco, A.; Buckley, A.G.; Ling, K.-M.; Martinovich, K.; Kicic-Starcevich, E.; Shaw, N.C.; et al. Effect of human rhinovirus infection on airway epithelium tight junction protein disassembly and transepithelial permeability. Exp. Lung Res. 2016, 42, 380–395. [Google Scholar] [CrossRef] [Green Version]
- Ng, R.N.; Tai, A.S.; Chang, B.J.; Stick, S.M.; Kicic, A. Overcoming Challenges to Make Bacteriophage Therapy Standard Clinical Treatment Practice for Cystic Fibrosis. Front. Microbiol. 2021, 11, 593988. [Google Scholar] [CrossRef]
- Elborn, J.S. Cystic fibrosis. Lancet 2016, 388, 2519–2531. [Google Scholar] [CrossRef]
- Międzybrodzki, R.; Fortuna, W.; Weber-Dąbrowska, B.; Gorski, A. A retrospective analysis of changes in inflammatory markers in patients treated with bacterial viruses. Clin. Exp. Med. 2009, 9, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Hietala, V.; Horsma-Heikkinen, J.; Carron, A.; Skurnik, M.; Kiljunen, S. The Removal of Endo- and Enterotoxins From Bacteriophage Preparations. Front. Microbiol. 2019, 10, 1674. [Google Scholar] [CrossRef] [Green Version]
- Liu, D.; Van Belleghem, J.; de Vries, C.; Burgener, E.; Chen, Q.; Manasherob, R.; Aronson, J.; Amanatullah, D.; Tamma, P.; Suh, G. The Safety and Toxicity of Phage Therapy: A Review of Animal and Clinical Studies. Viruses 2021, 13, 1268. [Google Scholar] [CrossRef]
- Huh, H.; Wong, S.; Jean, J.S.; Slavcev, R. Bacteriophage interactions with mammalian tissue: Therapeutic applications. Adv. Drug Deliv. Rev. 2019, 145, 4–17. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Dąbrowska, K.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Łusiak-Szelachowska, M.; Jończyk-Matysiak, E.; Borysowski, J. Phages and immunomodulation. Futur. Microbiol. 2017, 12, 905–914. [Google Scholar] [CrossRef] [Green Version]
- Cafora, M.; Brix, A.; Forti, F.; Loberto, N.; Aureli, M.; Briani, F.; Pistocchi, A. Phages as immunomodulators and their promising use as anti-inflammatory agents in a cftr loss-of-function zebrafish model. J. Cyst. Fibros. 2020. [Google Scholar] [CrossRef]
- Forrest, O.A.; Ingersoll, S.A.; Preininger, M.; Laval, J.; Limoli, D.; Brown, M.R.; Lee, F.E.; Bedi, B.; Sadikot, R.T.; Goldberg, J.B.; et al. Frontline Science: Pathological conditioning of human neutrophils recruited to the airway milieu in cystic fibrosis. J. Leukoc. Biol. 2018, 104, 665–675. [Google Scholar] [CrossRef]
- Pires, D.P.; Melo, L.; Boas, D.V.; Sillankorva, S.; Azeredo, J. Phage therapy as an alternative or complementary strategy to prevent and control biofilm-related infections. Curr. Opin. Microbiol. 2017, 39, 48–56. [Google Scholar] [CrossRef] [Green Version]
- Lepper, P.; Held, T.; Schneider, E.; Bölke, E.; Gerlach, H.; Trautmann, M. Clinical implications of antibiotic-induced endotoxin release in septic shock. Intensiv. Care Med. 2002, 28, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Górski, A.; Jończyk-Matysiak, E.; Łusiak-Szelachowska, M.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Borysowski, J. The Potential of Phage Therapy in Sepsis. Front. Immunol. 2017, 8, 1783. [Google Scholar] [CrossRef] [Green Version]
- Kiedrowski, M.R.; Gaston, J.R.; Kocak, B.R.; Coburn, S.L.; Lee, S.; Pilewski, J.M.; Myerburg, M.M.; Bomberger, J.M. Staphylococcus aureus Biofilm Growth on Cystic Fibrosis Airway Epithelial Cells Is Enhanced during Respiratory Syncytial Virus Coinfection. mSphere 2018, 3, e00341-18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hosseinidoust, Z.; Tufenkji, N.; Van De Ven, T.G. Formation of biofilms under phage predation: Considerations concerning a biofilm increase. Biofouling 2013, 29, 457–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, R.Y.K.; Das, T.; Manos, J.; Kutter, E.; Morales, S.; Chan, H.-K. Bacteriophage PEV20 and Ciprofloxacin Combination Treatment Enhances Removal of Pseudomonas aeruginosa Biofilm Isolated from Cystic Fibrosis and Wound Patients. AAPS J. 2019, 21, 49. [Google Scholar] [CrossRef]
- Dufour, N.; Delattre, R.; Ricard, J.-D.; Debarbieux, L. The Lysis of Pathogenic Escherichia coli by Bacteriophages Releases Less Endotoxin Than by β-Lactams. Clin. Infect. Dis. 2017, 64, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Dufour, N.; Delattre, R.; Chevallereau, A.; Ricard, J.D.; Debarbieux, L. Phage therapy of pneumonia is not associated with an overstimulation of the inflammatory response compared to antibiotic treatment in mice. Antimicrob. Agents Chemother. 2019, 63, e00379-19. [Google Scholar] [CrossRef] [Green Version]
- Jeon, J.; Yong, D. Two Novel Bacteriophages Improve Survival in Galleria mellonella Infection and Mouse Acute Pneumonia Models Infected with Extensively Drug-Resistant Pseudomonas aeruginosa. Appl. Environ. Microbiol. 2019, 85, e02900-18. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Zheng, P.; Ji, W.; Fu, Q.; Wang, H.; Yan, Y.; Sun, J. SLPW: A Virulent Bacteriophage Targeting Methicillin-Resistant Staphylococcus aureus In vitro and In vivo. Front. Microbiol. 2016, 7, 934. [Google Scholar] [CrossRef] [Green Version]
- Prazak, J.; Iten, M.; Cameron, D.; Save, J.; Grandgirard, D.; Resch, G.; Goepfert, C.; Leib, S.L.; Takala, J.; Jakob, S.M.; et al. Bacteriophages Improve Outcomes in Experimental Staphylococcus aureus Ventilator-associated Pneumonia. Am. J. Respir. Crit. Care Med. 2019, 200, 1126–1133. [Google Scholar] [CrossRef] [PubMed]
- Huff, W.; Huff, G.; Rath, N.; Balog, J.; Donoghue, A. Evaluation of aerosol spray and intramuscular injection of bacteriophage to treat an Escherichia coli respiratory infection. Poult. Sci. 2003, 82, 1108–1112. [Google Scholar] [CrossRef]
- Carmody, L.A.; Gill, J.; Summer, E.J.; Sajjan, U.S.; Gonzalez, C.F.; Young, R.F.; Lipuma, J.J. Efficacy of Bacteriophage Therapy in a Model ofBurkholderia cenocepaciaPulmonary Infection. J. Infect. Dis. 2010, 201, 264–271. [Google Scholar] [CrossRef] [Green Version]
- Morello, E.; Saussereau, E.; Maura, D.; Huerre, M.; Touqui, L.; Debarbieux, L. Pulmonary Bacteriophage Therapy on Pseudomonas aeruginosa Cystic Fibrosis Strains: First Steps Towards Treatment and Prevention. PLoS ONE 2011, 6, e16963. [Google Scholar] [CrossRef] [Green Version]
- Fothergill, J.; Neill, D.; Loman, N.; Winstanley, C.; Kadioglu, A. Pseudomonas aeruginosa adaptation in the nasopharyngeal reservoir leads to migration and persistence in the lungs. Nat. Commun. 2014, 5, 4780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alemayehu, D.; Casey, P.G.; McAuliffe, O.; Guinane, C.M.; Martin, J.G.; Shanahan, F.; Coffey, A.; Ross, R.; Hill, C. Bacteriophages ϕMR299-2 and ϕNH-4 Can Eliminate Pseudomonas aeruginosa in the Murine Lung and on Cystic Fibrosis Lung Airway Cells. mBio 2012, 3, e00029-12. [Google Scholar] [CrossRef] [Green Version]
- Nir-Paz, R.; Gelman, D.; Khouri, A.; Sisson, B.M.; Fackler, J.; Alkalay-Oren, S.; Khalifa, L.; Rimon, A.; Yerushalmy, O.; Bader, R.; et al. Successful Treatment of Antibiotic-resistant, Poly-microbial Bone Infection With Bacteriophages and Antibiotics Combination. Clin. Infect. Dis. 2019, 69, 2015–2018. [Google Scholar] [CrossRef] [PubMed]
- Secor, P.R.; Sass, G.; Nazik, H.; Stevens, D.A. Effect of acute predation with bacteriophage on intermicrobial aggression by Pseudomonas aeruginosa. PLoS ONE 2017, 12, e0179659. [Google Scholar] [CrossRef] [PubMed]
- Park, K.; Cha, K.; Myung, H. Observation of inflammatory responses in mice orally fed with bacteriophage T7. J. Appl. Microbiol. 2014, 117, 627–633. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.-A.; Resch, G.; Rousseau, A.-F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2018, 19, 35–45. [Google Scholar] [CrossRef]
- Bretaudeau, L.; Tremblais, K.; Aubrit, F.; Meichenin, M.; Arnaud, I. Good Manufacturing Practice (GMP) Compliance for Phage Therapy Medicinal Products. Front. Microbiol. 2020, 11, 1161. [Google Scholar] [CrossRef]
- Pirnay, J.P.; Verbeken, G.; Ceyssens, P.J.; Huys, I.; De Vos, D.; Ameloot, C.; Fauconnier, A. The Magistral Phage. Viruses 2018, 10, 64. [Google Scholar] [CrossRef] [Green Version]
- Onsea, J.; Uyttebroek, S.; Chen, B.; Wagemans, J.; Lood, C.; Van Gerven, L.; Spriet, I.; Devolder, D.; Debaveye, Y.; Depypere, M.; et al. Bacteriophage Therapy for Difficult-to-Treat Infections: The Implementation of a Multidisciplinary Phage Task Force (The PHAGEFORCE Study Protocol). Viruses 2021, 13, 1543. [Google Scholar] [CrossRef] [PubMed]
- Oechslin, F. Resistance Development to Bacteriophages Occurring during Bacteriophage Therapy. Viruses 2018, 10, 351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Barceló, C. Phage Therapy Faces Evolutionary Challenges. Viruses 2018, 10, 323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moller, A.G.; Lindsay, J.; Read, T.D. Determinants of Phage Host Range in Staphylococcus Species. Appl. Environ. Microbiol. 2019, 85. [Google Scholar] [CrossRef] [Green Version]
- Rohde, C.; Resch, G.; Pirnay, J.-P.; Blasdel, B.G.; Debarbieux, L.; Gelman, D.; Górski, A.; Hazan, R.; Huys, I.; Kakabadze, E.; et al. Expert Opinion on Three Phage Therapy Related Topics: Bacterial Phage Resistance, Phage Training and Prophages in Bacterial Production Strains. Viruses 2018, 10, 178. [Google Scholar] [CrossRef] [Green Version]
| Characterisation | Data Obtained | References |
|---|---|---|
| Infectivity: Host-range and specificity | Host-range and specificity assays determine the ability for a single phage to infect different bacterial strains, species, and genera. However, to determine how well a phage can infect any given strain of bacteria, an efficiency of plating assay must be performed by spotting a serial dilution of phage onto lawns of bacterial culture. Many studies utilise standard microbiological plating techniques to do these, such as the spot test on double agar overlays inoculated with the target bacteria. | [21,28,39,42,43] |
| Morphological analysis | Homogenous phage particles are prepared, via staining of high titre phage lysates, for observation via transmission electron microscopy. From this, the head size and shape, as well as tail length, are determined. | [28,39,42,43] |
| Adsorption assay | Adsorption assays are used to determine how fast a phage attaches to its target bacterium and the percentage of phage within a sample that can attach to the target bacterium at a given multiplicity of infection (MOI). | [28,42] |
| One-step growth curve | Latency period is the period between adsorption of phage to the target bacteria and the first burst as indicated by the rise in phage titre within a sample. The burst size of a phage refers to how many virions are released per infected bacterial cell and both are typically calculated using the one-step growth curve. | [28,42,43] |
| Kill-curves/Lysis profiles | These are bacteriolytic activity tests which determine a phages lytic capability in vitro by infecting early exponential phase bacteria at various MOIs and comparing with a non-infected control. Bacterial cell density is typically measured via spectrophotometry at an optical density of 600 nm. | [28,42,43] |
| Stability/Sensitivity | The stability of phages at various temperatures and pH ranges is important to determine how fast phages will degrade under different storage or relevant test conditions, such as body temperature. | [21,28,39,42] |
| Genomic analysis | Genomic analysis can be carried out in various ways such as restriction enzyme analysis and whole genome sequencing of genomic DNA (gDNA) isolated from phage. Restriction enzyme analysis is performed using restriction endonucleases to cleave the gDNA, followed by gel electrophoresis to visualise restriction patterns and estimate the size of the genome. Whole genome sequencing gives the ability to analyse and annotate the full genome of a phage, perform whole genome-based phylogeny, and search for lysogeny-associated genes such as integrases. | [28,42,43] |
| Biofilm susceptibility | Biofilm-forming bacteria are grown in specialised growth media to promote the formation of biofilm on abiotic surfaces. Typically, this is carried out over 24 and 48 h, as the age of biofilms has an effect on bacterial susceptibility to a number of agents including both phage and antibiotics. In addition to biofilm clearance, prevention has also been measured on abiotic surfaces. | [42,44,45] |
| Study Title | Clinical Trial ID | Primary and Secondary Outcome Measurements | Phage Treatment | Pathogen Details | Status at Time of Writing |
|---|---|---|---|---|---|
| Bacteriophage Therapy in Acute Tonsillitis. | NCT04682964 | Clinical observation of sick children. Manifestation of clinical symptoms, observation of the nasopharynx, and parental report. Experimental clinical stage: Bacteriophage therapy, efficacy will be studied based on immunological assay, cellular and humoral immunity via ELISA. | Pyobacteriophage complex liquid (PCL) administered via nebuliser inhalation. | Not available | Active, not recruiting. |
| Cystic Fibrosis bacteriophage Study at Yale (CYPHY): A Single-site, Randomized, Double-blind, Placebo-controlled Study of Bacteriophage Therapy YPT-01 for Pseudomonas aeruginosa Infections in Adults With Cystic Fibrosis. | NCT04684641 | Primary: Reduction in sputum bacterial culture. Secondary: Change in lung function based on FEV at multiple stages. Change in rate of pulmonary exacerbations. Change in the rate of hospitalisation. Change in the rate of acute antibiotic usage. Patients’ quality of life. | Yale Phage Therapy (YPT)-01 | P. aeruginosa | Recruiting. |
| A Phase 1b/2a, Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Single and Multiple Ascending Dose Study to Evaluate the Safety and Tolerability of AP-PA02 Multi-Phage Therapeutic Candidate for Inhalation in Subjects With Cystic Fibrosis and Chronic Pulmonary Pseudomonas aeruginosa (Pa) Infection. | NCT04596319 | Primary: Incidence of treatment emergent adverse events for safety and tolerability of single and multiple doses. Secondary: Explore P. aeruginosa recovery from sputum following multiple doses of AP-PA02 as measured by change in CFU/g of sputum. | AP-PA02 phage product administered via inhalation. | P. aeruginosa | Recruiting. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iszatt, J.J.; Larcombe, A.N.; Chan, H.-K.; Stick, S.M.; Garratt, L.W.; Kicic, A. Phage Therapy for Multi-Drug Resistant Respiratory Tract Infections. Viruses 2021, 13, 1809. https://doi.org/10.3390/v13091809
Iszatt JJ, Larcombe AN, Chan H-K, Stick SM, Garratt LW, Kicic A. Phage Therapy for Multi-Drug Resistant Respiratory Tract Infections. Viruses. 2021; 13(9):1809. https://doi.org/10.3390/v13091809
Chicago/Turabian StyleIszatt, Joshua J., Alexander N. Larcombe, Hak-Kim Chan, Stephen M. Stick, Luke W. Garratt, and Anthony Kicic. 2021. "Phage Therapy for Multi-Drug Resistant Respiratory Tract Infections" Viruses 13, no. 9: 1809. https://doi.org/10.3390/v13091809
APA StyleIszatt, J. J., Larcombe, A. N., Chan, H.-K., Stick, S. M., Garratt, L. W., & Kicic, A. (2021). Phage Therapy for Multi-Drug Resistant Respiratory Tract Infections. Viruses, 13(9), 1809. https://doi.org/10.3390/v13091809







