A Novel Frameshifting Inhibitor Having Antiviral Activity against Zoonotic Coronaviruses
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture and Virus Infection
2.2. Plasmids
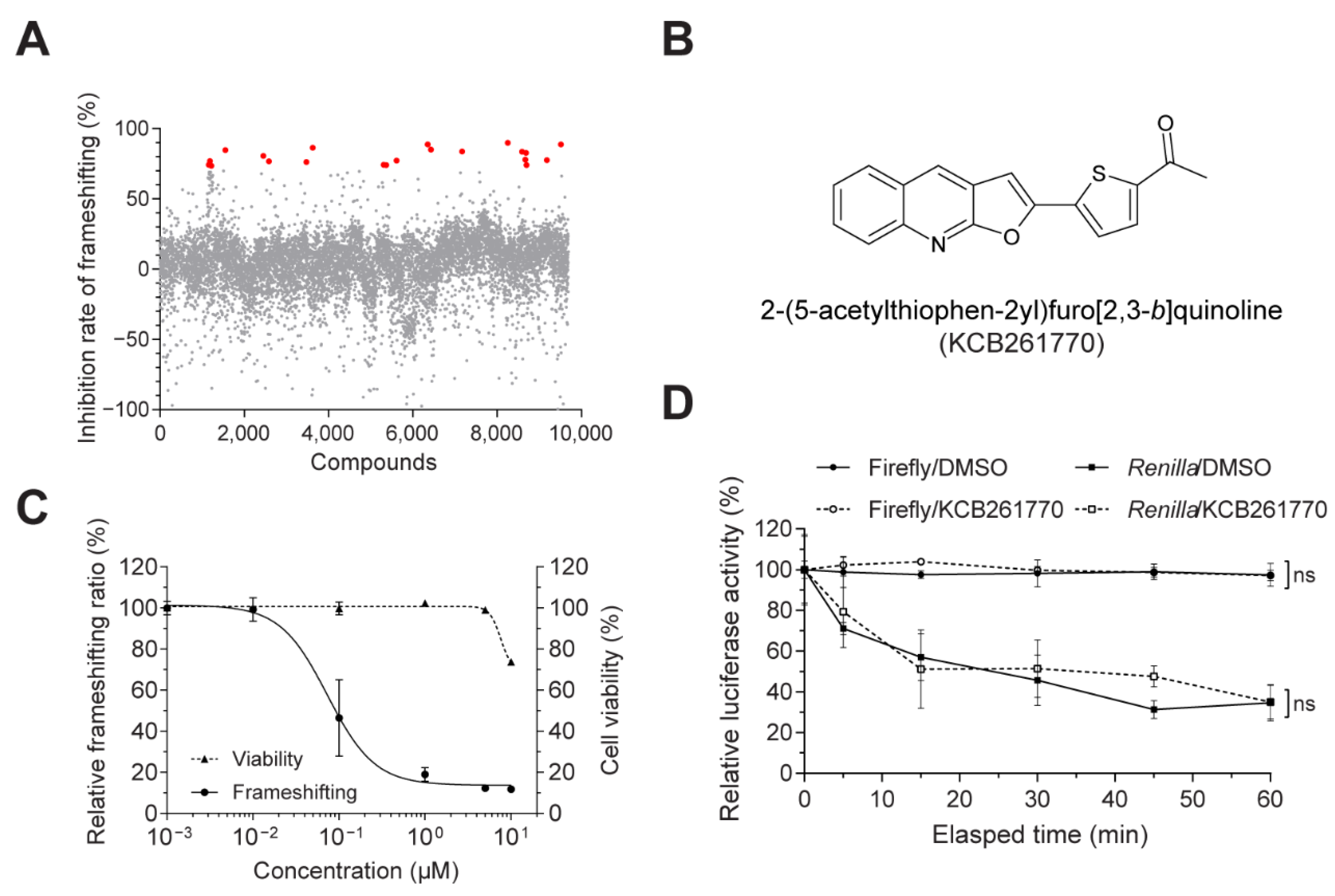
2.3. Chemical Library Screening and Dual-Luciferase Assays
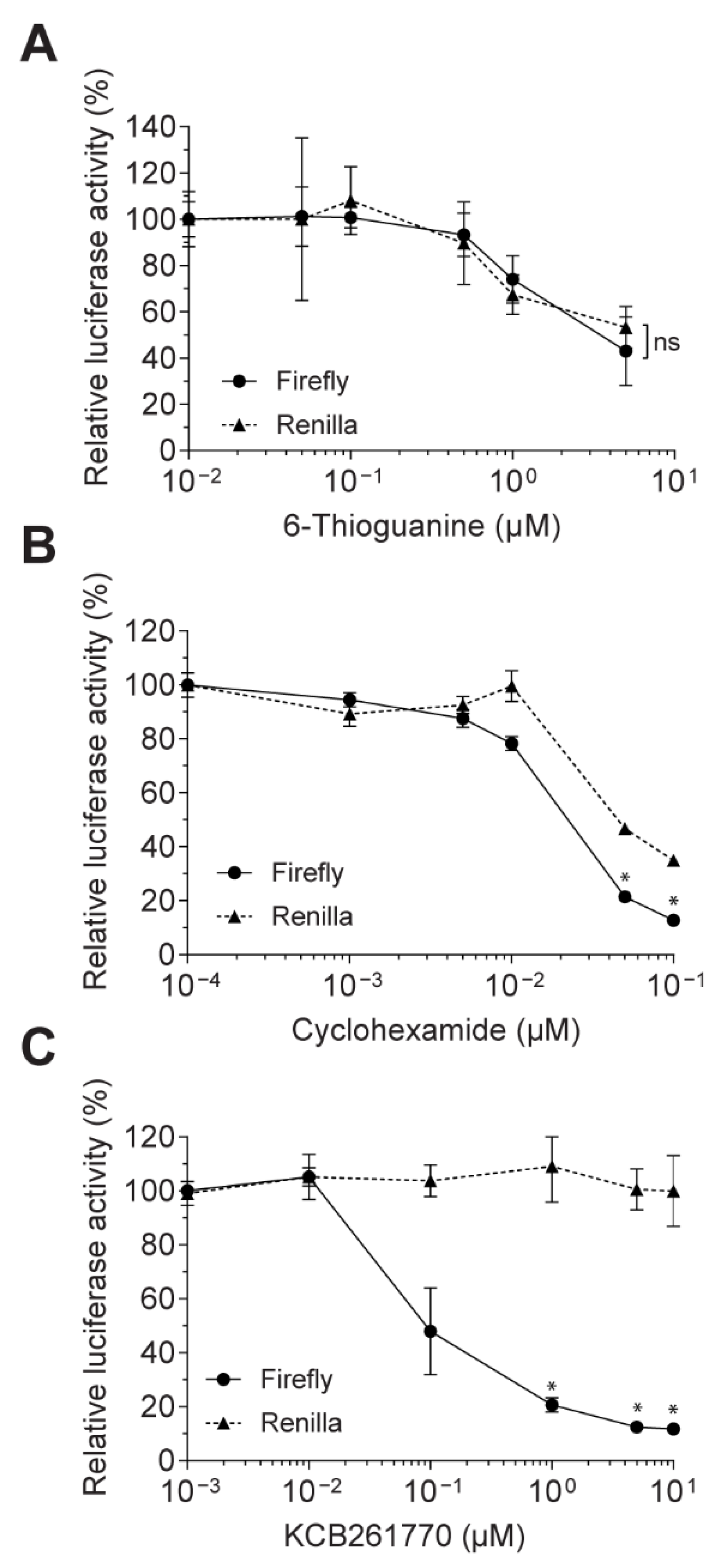
2.4. Cell Viability
2.5. Quantitative RT-PCR
2.6. Statistical Analysis
3. Results
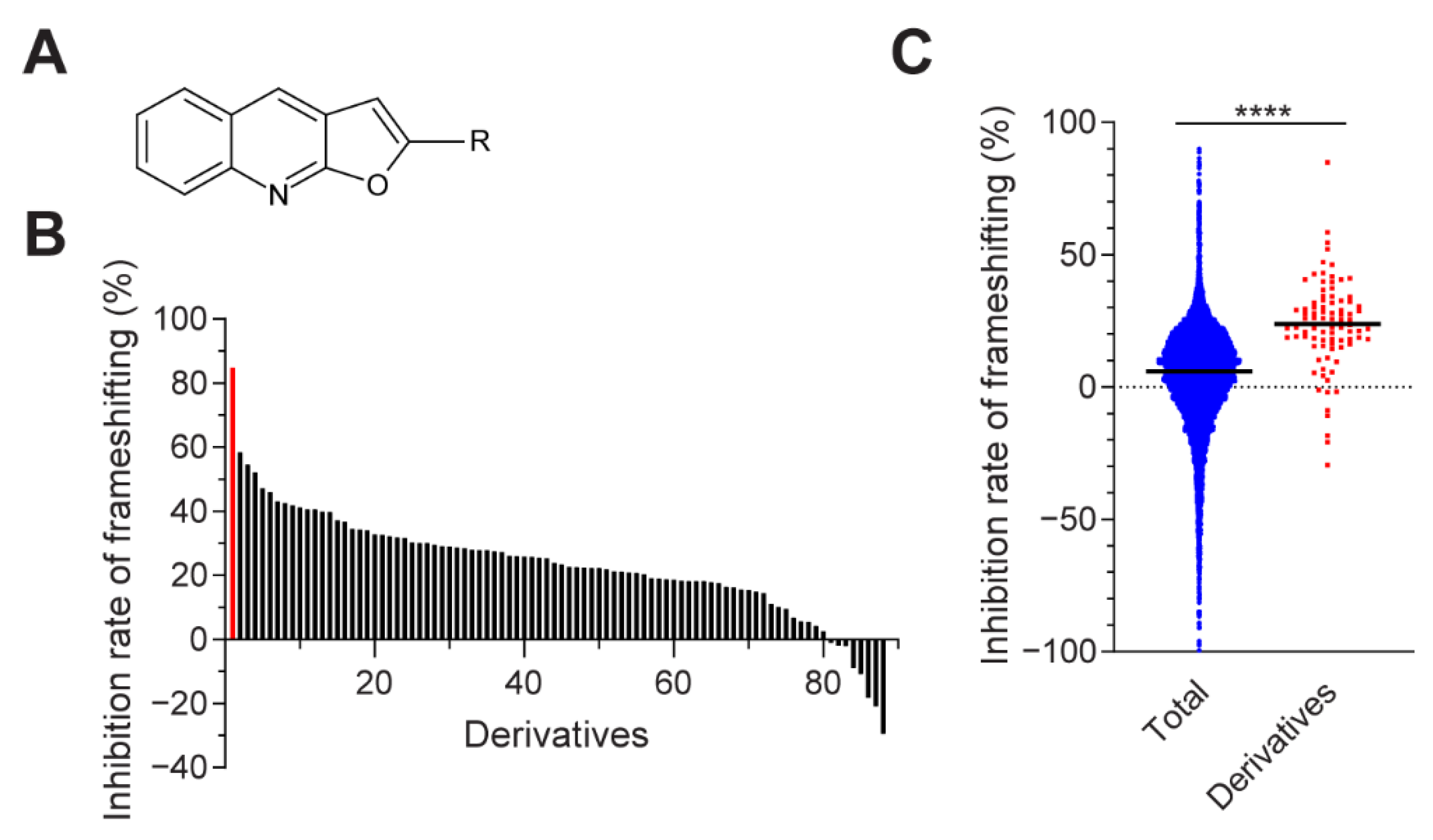
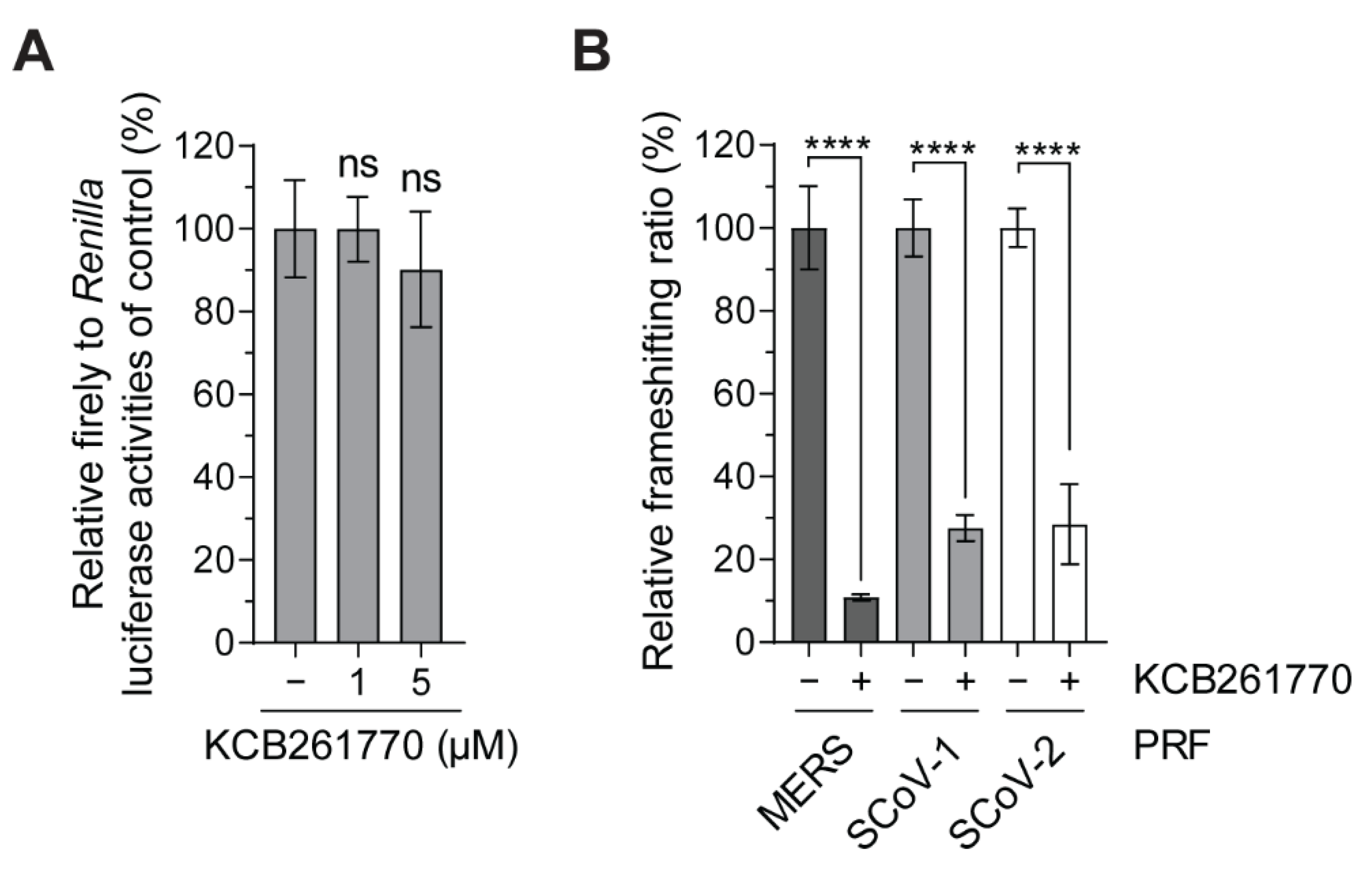
3.1. Novel Compound Inhibiting MERS-CoV Frameshifting
3.2. Inhibition of the Frameshifting of Zoonotic Coronaviruses by Compound KCB261770
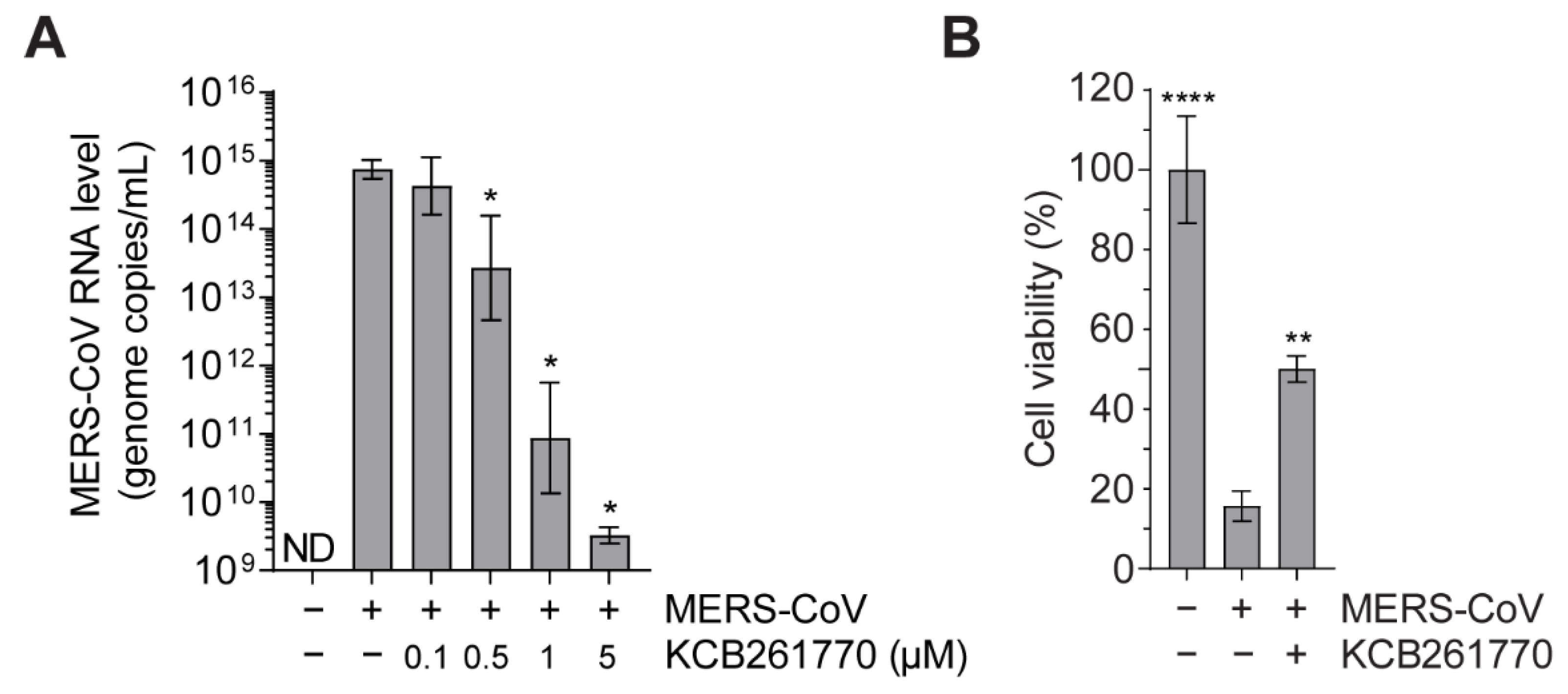
3.3. Compound KCB261770 Efficiently Suppresses MERS-CoV Propagation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Coronavirus disease (COVID-19) Weekly Epidemiological Update and Weekly Operational Update; World Health Organization: Geneva, Switzerland, 2021.
- Zaki, A.M.; van Boheemen, S.; Bestebroer, T.M.; Osterhaus, A.D.; Fouchier, R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012, 367, 1814–1820. [Google Scholar] [CrossRef]
- WHO. MERS Situation Update, March 2021; World Health Organization: Geneva, Switzerland, 2021. [Google Scholar]
- de Groot, R.J.; Baker, S.C.; Baric, R.S.; Brown, C.S.; Drosten, C.; Enjuanes, L.; Fouchier, R.A.; Galiano, M.; Gorbalenya, A.E.; Memish, Z.A.; et al. Middle East respiratory syndrome coronavirus (MERS-CoV): Announcement of the Coronavirus Study Group. J. Virol. 2013, 87, 7790–7792. [Google Scholar] [CrossRef] [Green Version]
- Fehr, A.R.; Channappanavar, R.; Perlman, S. Middle East Respiratory Syndrome: Emergence of a Pathogenic Human Coronavirus. Annu. Rev. Med. 2017, 68, 387–399. [Google Scholar] [CrossRef] [Green Version]
- Fung, T.S.; Liu, D.X. Human Coronavirus: Host-Pathogen Interaction. Annu. Rev. Microbiol. 2019, 73, 529–557. [Google Scholar] [CrossRef] [Green Version]
- Plant, E.P.; Dinman, J.D. The role of programmed-1 ribosomal frameshifting in coronavirus propagation. Front. Biosci. 2008, 13, 4873–4881. [Google Scholar] [CrossRef] [Green Version]
- Brierley, I.; Digard, P.; Inglis, S.C. Characterization of an efficient coronavirus ribosomal frameshifting signal: Requirement for an RNA pseudoknot. Cell 1989, 57, 537–547. [Google Scholar] [CrossRef]
- Decroly, E.; Imbert, I.; Coutard, B.; Bouvet, M.; Selisko, B.; Alvarez, K.; Gorbalenya, A.E.; Snijder, E.J.; Canard, B. Coronavirus nonstructural protein 16 is a cap-0 binding enzyme possessing (nucleoside-2′O)-methyltransferase activity. J. Virol. 2008, 82, 8071–8084. [Google Scholar] [CrossRef] [Green Version]
- Minskaia, E.; Hertzig, T.; Gorbalenya, A.E.; Campanacci, V.; Cambillau, C.; Canard, B.; Ziebuhr, J. Discovery of an RNA virus 3′–5′ exoribonuclease that is critically involved in coronavirus RNA synthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 5108–5113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivanov, K.A.; Thiel, V.; Dobbe, J.C.; van der Meer, Y.; Snijder, E.J.; Ziebuhr, J. Multiple enzymatic activities associated with severe acute respiratory syndrome coronavirus helicase. J. Virol. 2004, 78, 5619–5632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, D.G.; Choi, J.K.; Taylor, D.R.; Oh, J.W. Biochemical characterization of a recombinant SARS coronavirus nsp12 RNA-dependent RNA polymerase capable of copying viral RNA templates. Arch. Virol. 2012, 157, 2095–2104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, S.; Chen, B.; Tay, F.P.; Ng, B.S.; Liu, D.X. An arginine-to-proline mutation in a domain with undefined functions within the helicase protein (Nsp13) is lethal to the coronavirus infectious bronchitis virus in cultured cells. Virology 2007, 358, 136–147. [Google Scholar] [CrossRef] [Green Version]
- Almazan, F.; Dediego, M.L.; Galan, C.; Escors, D.; Alvarez, E.; Ortego, J.; Sola, I.; Zuniga, S.; Alonso, S.; Moreno, J.L.; et al. Construction of a severe acute respiratory syndrome coronavirus infectious cDNA clone and a replicon to study coronavirus RNA synthesis. J. Virol. 2006, 80, 10900–10906. [Google Scholar] [CrossRef] [Green Version]
- Kaplan, A.H.; Zack, J.A.; Knigge, M.; Paul, D.A.; Kempf, D.J.; Norbeck, D.W.; Swanstrom, R. Partial inhibition of the human immunodeficiency virus type 1 protease results in aberrant virus assembly and the formation of noninfectious particles. J. Virol. 1993, 67, 4050–4055. [Google Scholar] [CrossRef] [Green Version]
- Biswas, P.; Jiang, X.; Pacchia, A.L.; Dougherty, J.P.; Peltz, S.W. The human immunodeficiency virus type 1 ribosomal frameshifting site is an invariant sequence determinant and an important target for antiviral therapy. J. Virol. 2004, 78, 2082–2087. [Google Scholar] [CrossRef] [Green Version]
- Jacks, T.; Power, M.D.; Masiarz, F.R.; Luciw, P.A.; Barr, P.J.; Varmus, H.E. Characterization of ribosomal frameshifting in HIV-1 gag-pol expression. Nature 1988, 331, 280–283. [Google Scholar] [CrossRef]
- Moomau, C.; Musalgaonkar, S.; Khan, Y.A.; Jones, J.E.; Dinman, J.D. Structural and Functional Characterization of Programmed Ribosomal Frameshift Signals in West Nile Virus Strains Reveals High Structural Plasticity Among cis-Acting RNA Elements. J. Biol. Chem. 2016, 291, 15788–15795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melian, E.B.; Hinzman, E.; Nagasaki, T.; Firth, A.E.; Wills, N.M.; Nouwens, A.S.; Blitvich, B.J.; Leung, J.; Funk, A.; Atkins, J.F.; et al. NS1’ of flaviviruses in the Japanese encephalitis virus serogroup is a product of ribosomal frameshifting and plays a role in viral neuroinvasiveness. J. Virol. 2010, 84, 1641–1647. [Google Scholar] [CrossRef] [Green Version]
- Firth, A.E.; Chung, B.Y.; Fleeton, M.N.; Atkins, J.F. Discovery of frameshifting in Alphavirus 6K resolves a 20-year enigma. Virol. J. 2008, 5, 108. [Google Scholar] [CrossRef] [Green Version]
- Snyder, J.E.; Kulcsar, K.A.; Schultz, K.L.; Riley, C.P.; Neary, J.T.; Marr, S.; Jose, J.; Griffin, D.E.; Kuhn, R.J. Functional characterization of the alphavirus TF protein. J. Virol. 2013, 87, 8511–8523. [Google Scholar] [CrossRef] [Green Version]
- Kendra, J.A.; de la Fuente, C.; Brahms, A.; Woodson, C.; Bell, T.M.; Chen, B.; Khan, Y.A.; Jacobs, J.L.; Kehn-Hall, K.; Dinman, J.D. Ablation of Programmed -1 Ribosomal Frameshifting in Venezuelan Equine Encephalitis Virus Results in Attenuated Neuropathogenicity. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [Green Version]
- Plant, E.P.; Sims, A.C.; Baric, R.S.; Dinman, J.D.; Taylor, D.R. Altering SARS coronavirus frameshift efficiency affects genomic and subgenomic RNA production. Viruses 2013, 5, 279–294. [Google Scholar] [CrossRef] [Green Version]
- Plant, E.P.; Rakauskaite, R.; Taylor, D.R.; Dinman, J.D. Achieving a golden mean: Mechanisms by which coronaviruses ensure synthesis of the correct stoichiometric ratios of viral proteins. J. Virol. 2010, 84, 4330–4340. [Google Scholar] [CrossRef] [Green Version]
- Plant, E.P.; Perez-Alvarado, G.C.; Jacobs, J.L.; Mukhopadhyay, B.; Hennig, M.; Dinman, J.D. A three-stemmed mRNA pseudoknot in the SARS coronavirus frameshift signal. PLoS Biol. 2005, 3, e172. [Google Scholar] [CrossRef] [Green Version]
- Ahn, D.G.; Lee, W.; Choi, J.K.; Kim, S.J.; Plant, E.P.; Almazan, F.; Taylor, D.R.; Enjuanes, L.; Oh, J.W. Interference of ribosomal frameshifting by antisense peptide nucleic acids suppresses SARS coronavirus replication. Antivir. Res. 2011, 91, 1–10. [Google Scholar] [CrossRef]
- Kim, Y.J.; Cho, Y.J.; Kim, D.W.; Yang, J.S.; Kim, H.; Park, S.; Han, Y.W.; Yun, M.R.; Lee, H.S.; Kim, A.R.; et al. Complete Genome Sequence of Middle East Respiratory Syndrome Coronavirus KOR/KNIH/002_05_2015, Isolated in South Korea. Genome Announc. 2015, 3. [Google Scholar] [CrossRef] [Green Version]
- Loughran, G.; Howard, M.T.; Firth, A.E.; Atkins, J.F. Avoidance of reporter assay distortions from fused dual reporters. RNA 2017, 23, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Small Molecule Libraries Obtained from the Korea Chemical Bank; Korea Research Institute of Chemical Technology: Daejeon, Korea. Available online: http://www.chembank.org (accessed on 12 July 2021).
- Park, H.J.; Yang, O.K.; Park, Y.C.; Yum, E.K. Synthesis of 2-Substituted Furo[2,3-b]- and Furo[3,2-c]quinolines via Heterogeneous Palladium-catalyzed Heteroannulation. Bull. Korean Chem. Soc. 2016, 37, 958–961. [Google Scholar] [CrossRef]
- Sichaem, J.; Jirasirichote, A.; Sapasuntikul, K.; Khumkratok, S.; Sawasdee, P.; Do, T.M.; Tip-pyang, S. New furoquinoline alkaloids from the leaves of Evodia lepta. Fitoterapia 2014, 92, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Nouga, A.B.; Ndom, J.C.; Mpondo, E.M.; Nyobe, J.C.; Njoya, A.; Meva’a, L.M.; Cranwell, P.B.; Howell, J.A.; Harwood, L.M.; Wansi, J.D. New furoquinoline alkaloid and flavanone glycoside derivatives from the leaves of Oricia suaveolens and Oricia renieri (Rutaceae). Nat. Prod. Res. 2016, 30, 305–310. [Google Scholar] [CrossRef]
- Yu, L.Z.; Hu, X.B.; Xu, Q.; Shi, M. Thermally induced formal [3+2] cyclization of ortho-aminoaryl-tethered alkylidenecyclopropanes: Facile synthesis of furoquinoline and thienoquinoline derivatives. Chem. Commun. (Camb) 2016, 52, 2701–2704. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Wang, L.; Zhao, L.; Zhang, Q.Y.; Zeng, K.W.; Zhao, M.B.; Jiang, Y.; Tu, P.F.; Guo, X.Y. Anti-inflammatory quinoline alkaloids from the root bark of Dictamnus dasycarpus. Phytochemistry 2020, 172, 112260. [Google Scholar] [CrossRef]
- Ratheesh, M.; Sindhu, G.; Helen, A. Anti-inflammatory effect of quinoline alkaloid skimmianine isolated from Ruta graveolens L. Inflamm. Res. 2013, 62, 367–376. [Google Scholar] [CrossRef]
- Basco, L.K.; Mitaku, S.; Skaltsounis, A.L.; Ravelomanantsoa, N.; Tillequin, F.; Koch, M.; Le Bras, J. In vitro activities of furoquinoline and acridone alkaloids against Plasmodium falciparum. Antimicrob. Agents Chemother. 1994, 38, 1169–1171. [Google Scholar] [CrossRef] [Green Version]
- Kouam, A.D.K.; Bissoue, A.N.; Tcho, A.T.; Happi, E.N.; Waffo, A.F.K.; Sewald, N.; Wansi, J.D. Antimicrobial Furoquinoline Alkaloids from Vepris lecomteana (Pierre) Cheek & T. Heller (Rutaceae). Molecules 2017, 23, 13. [Google Scholar] [CrossRef] [Green Version]
- Hilimire, T.A.; Chamberlain, J.M.; Anokhina, V.; Bennett, R.P.; Swart, O.; Myers, J.R.; Ashton, J.M.; Stewart, R.A.; Featherston, A.L.; Gates, K.; et al. HIV-1 Frameshift RNA-Targeted Triazoles Inhibit Propagation of Replication-Competent and Multi-Drug-Resistant HIV in Human Cells. ACS Chem. Biol. 2017, 12, 1674–1682. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinman, J.D.; Ruiz-Echevarria, M.J.; Czaplinski, K.; Peltz, S.W. Peptidyl-transferase inhibitors have antiviral properties by altering programmed -1 ribosomal frameshifting efficiencies: Development of model systems. Proc. Natl. Acad. Sci. USA 1997, 94, 6606–6611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neupane, K.; Munshi, S.; Zhao, M.; Ritchie, D.B.; Ileperuma, S.M.; Woodside, M.T. Anti-Frameshifting Ligand Active against SARS Coronavirus-2 Is Resistant to Natural Mutations of the Frameshift-Stimulatory Pseudoknot. J. Mol. Biol. 2020, 432, 5843–5847. [Google Scholar] [CrossRef] [PubMed]
- Park, S.J.; Kim, Y.G.; Park, H.J. Identification of RNA pseudoknot-binding ligand that inhibits the -1 ribosomal frameshifting of SARS-coronavirus by structure-based virtual screening. J. Am. Chem. Soc. 2011, 133, 10094–10100. [Google Scholar] [CrossRef] [PubMed]
- Kwan, S.W.; Kwan, S.P.; Mandel, H.G. The incorporation of 6-thioguanine into RNA fractions and its effect on RNA and protein biosynthesis in mouse sarcoma 180 ascites cells. Cancer Res. 1973, 33, 950–955. [Google Scholar] [PubMed]
- Munshi, P.N.; Lubin, M.; Bertino, J.R. 6-thioguanine: A drug with unrealized potential for cancer therapy. Oncologist 2014, 19, 760–765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schneider-Poetsch, T.; Ju, J.; Eyler, D.E.; Dang, Y.; Bhat, S.; Merrick, W.C.; Green, R.; Shen, B.; Liu, J.O. Inhibition of eukaryotic translation elongation by cycloheximide and lactimidomycin. Nat. Chem. Biol. 2010, 6, 209–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, J.A.; Olson, A.N.; Neupane, K.; Munshi, S.; San Emeterio, J.; Pollack, L.; Woodside, M.T.; Dinman, J.D. Structural and functional conservation of the programmed -1 ribosomal frameshift signal of SARS coronavirus 2 (SARS-CoV-2). J. Biol. Chem. 2020, 295, 10741–10748. [Google Scholar] [CrossRef] [PubMed]
- Fourmy, D.; Yoshizawa, S. A cytosine-to-uracil change within the programmed -1 ribosomal frameshift signal of SARS-CoV-2 results in structural similarities with the MERS-CoV signal. bioRxiv 2020. [Google Scholar] [CrossRef]
- Rakauskaite, R.; Liao, P.Y.; Rhodin, M.H.; Lee, K.; Dinman, J.D. A rapid, inexpensive yeast-based dual-fluorescence assay of programmed--1 ribosomal frameshifting for high-throughput screening. Nucleic Acids Res. 2011, 39, e97. [Google Scholar] [CrossRef] [PubMed]
- Marcheschi, R.J.; Mouzakis, K.D.; Butcher, S.E. Selection and characterization of small molecules that bind the HIV-1 frameshift site RNA. ACS Chem. Biol. 2009, 4, 844–854. [Google Scholar] [CrossRef]
- Aboul-ela, F. Strategies for the design of RNA-binding small molecules. Future Med. Chem. 2010, 2, 93–119. [Google Scholar] [CrossRef] [PubMed]
- Connelly, C.M.; Moon, M.H.; Schneekloth, J.S., Jr. The Emerging Role of RNA as a Therapeutic Target for Small Molecules. Cell Chem. Biol. 2016, 23, 1077–1090. [Google Scholar] [CrossRef]
- Ng, W.C.; Soto-Acosta, R.; Bradrick, S.S.; Garcia-Blanco, M.A.; Ooi, E.E. The 5′ and 3′ Untranslated Regions of the Flaviviral Genome. Viruses 2017, 9, 137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martinez-Salas, E.; Francisco-Velilla, R.; Fernandez-Chamorro, J.; Embarek, A.M. Insights into Structural and Mechanistic Features of Viral IRES Elements. Front. Microbiol. 2017, 8, 2629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atkins, J.F.; Loughran, G.; Bhatt, P.R.; Firth, A.E.; Baranov, P.V. Ribosomal frameshifting and transcriptional slippage: From genetic steganography and cryptography to adventitious use. Nucleic Acids Res. 2016, 44, 7007–7078. [Google Scholar] [CrossRef] [Green Version]
- Wacker, A.; Weigand, J.E.; Akabayov, S.R.; Altincekic, N.; Bains, J.K.; Banijamali, E.; Binas, O.; Castillo-Martinez, J.; Cetiner, E.; Ceylan, B.; et al. Secondary structure determination of conserved SARS-CoV-2 RNA elements by NMR spectroscopy. Nucleic Acids Res. 2020, 48, 12415–12435. [Google Scholar] [CrossRef]
- Baranov, P.V.; Henderson, C.M.; Anderson, C.B.; Gesteland, R.F.; Atkins, J.F.; Howard, M.T. Programmed ribosomal frameshifting in decoding the SARS-CoV genome. Virology 2005, 332, 498–510. [Google Scholar] [CrossRef] [Green Version]
- Hu, H.T.; Cho, C.P.; Lin, Y.H.; Chang, K.Y. A general strategy to inhibiting viral -1 frameshifting based on upstream attenuation duplex formation. Nucleic Acids Res. 2016, 44, 256–266. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhatt, P.R.; Scaiola, A.; Loughran, G.; Leibundgut, M.; Kratzel, A.; Meurs, R.; Dreos, R.; O’Connor, K.M.; McMillan, A.; Bode, J.W.; et al. Structural basis of ribosomal frameshifting during translation of the SARS-CoV-2 RNA genome. Science 2021. [Google Scholar] [CrossRef] [PubMed]
- Napthine, S.; Ling, R.; Finch, L.K.; Jones, J.D.; Bell, S.; Brierley, I.; Firth, A.E. Protein-directed ribosomal frameshifting temporally regulates gene expression. Nat. Commun. 2017, 8, 15582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, A.; Xu, H.; Zhan, R.; Chen, W.; Liu, J.; Chi, Y.; Chen, D.; Ji, X.; Luo, C. Metabolic Profile of Skimmianine in Rats Determined by Ultra-Performance Liquid Chromatography Coupled with Quadrupole Time-of-Flight Tandem Mass Spectrometry. Molecules 2017, 22, 489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campeau, E.; Ruhl, V.E.; Rodier, F.; Smith, C.L.; Rahmberg, B.L.; Fuss, J.O.; Campisi, J.; Yaswen, P.; Cooper, P.K.; Kaufman, P.D. A versatile viral system for expression and depletion of proteins in mammalian cells. PLoS ONE 2009, 4, e6529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
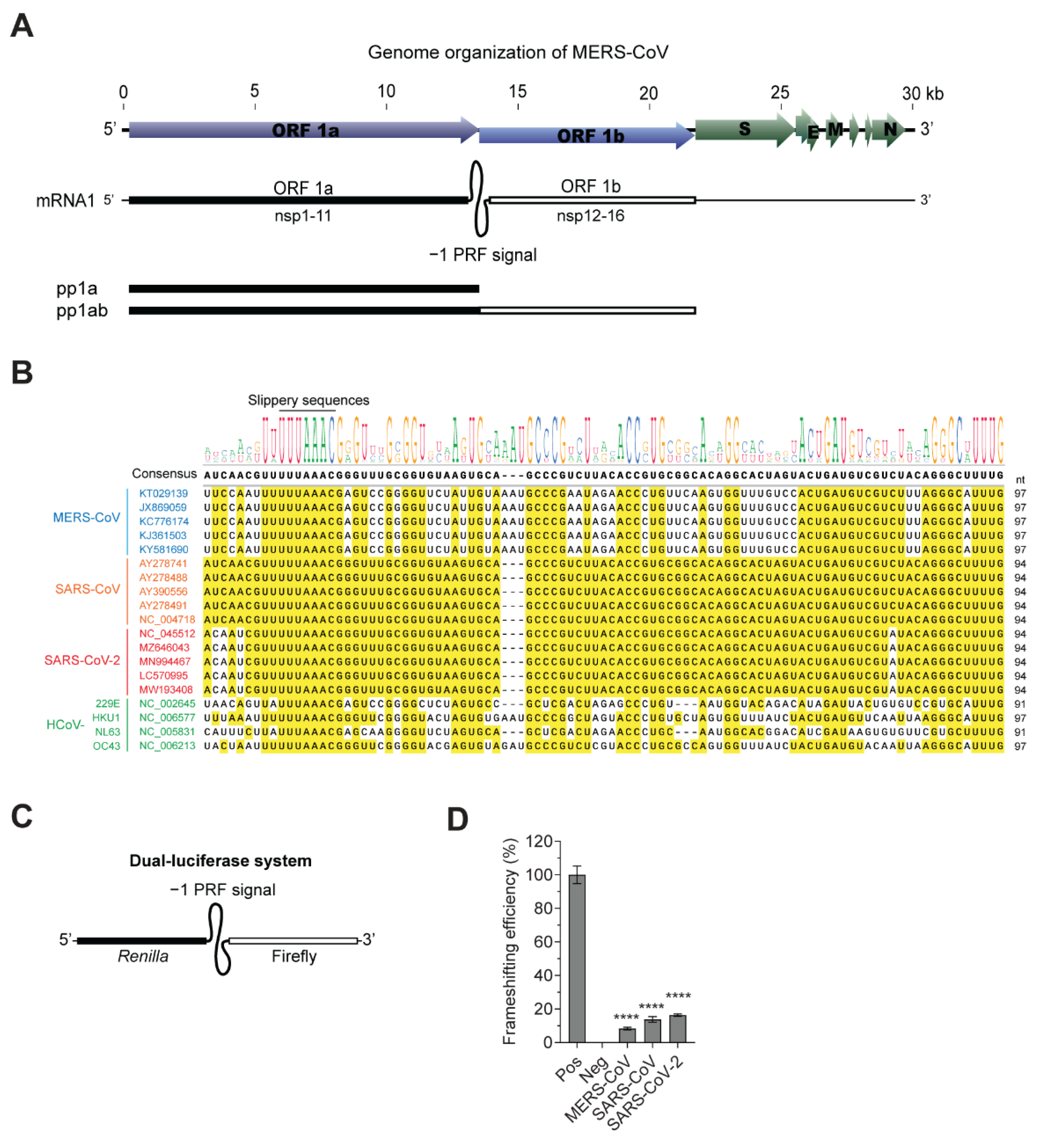
| Viruses | Inserted PRF Sequences (5′–3′) a | Accession Number and Position |
|---|---|---|
| MERS-CoV | TTCCAATTTTTTAAACGAGTCCGGGGTTCTATTGTAAATGCCCGAATAGAACCCTGTTCAAGTGGTTTGTCCACTGATGTCGTCTTTAGGGCATTTG | KT029139.1 13418–13514 nt (97 nucleotides) |
| SARS-CoV | ATCAACGTTTTTAAACGGGTTTGCGGTGTAAGTGCAGCCCGTCTTACACCGTGCGGCACAGGCACTAGTACTGATGTCGTCTACAGGGCTTTTG | AY278741 13,382–13,477 nt (94 nucleotides) |
| SARS-CoV-2 | ACAATCGTTTTTAAACGGGTTTGCGGTGTAAGTGCAGCCCGTCTTACACCGTGCGGCACAGGCACTAGTACTGATGTCGTATACAGGGCTTTTG | NC_045512.2 13,452–13,547 nt (94 nucleotides) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, D.-G.; Yoon, G.Y.; Lee, S.; Ku, K.B.; Kim, C.; Kim, K.-D.; Kwon, Y.-C.; Kim, G.-W.; Kim, B.-T.; Kim, S.-J. A Novel Frameshifting Inhibitor Having Antiviral Activity against Zoonotic Coronaviruses. Viruses 2021, 13, 1639. https://doi.org/10.3390/v13081639
Ahn D-G, Yoon GY, Lee S, Ku KB, Kim C, Kim K-D, Kwon Y-C, Kim G-W, Kim B-T, Kim S-J. A Novel Frameshifting Inhibitor Having Antiviral Activity against Zoonotic Coronaviruses. Viruses. 2021; 13(8):1639. https://doi.org/10.3390/v13081639
Chicago/Turabian StyleAhn, Dae-Gyun, Gun Young Yoon, Sunhee Lee, Keun Bon Ku, Chonsaeng Kim, Kyun-Do Kim, Young-Chan Kwon, Geon-Woo Kim, Bum-Tae Kim, and Seong-Jun Kim. 2021. "A Novel Frameshifting Inhibitor Having Antiviral Activity against Zoonotic Coronaviruses" Viruses 13, no. 8: 1639. https://doi.org/10.3390/v13081639
APA StyleAhn, D.-G., Yoon, G. Y., Lee, S., Ku, K. B., Kim, C., Kim, K.-D., Kwon, Y.-C., Kim, G.-W., Kim, B.-T., & Kim, S.-J. (2021). A Novel Frameshifting Inhibitor Having Antiviral Activity against Zoonotic Coronaviruses. Viruses, 13(8), 1639. https://doi.org/10.3390/v13081639







