Epidemic Spread of SARS-CoV-2 Lineage B.1.1.7 in Brazil
1. Introduction
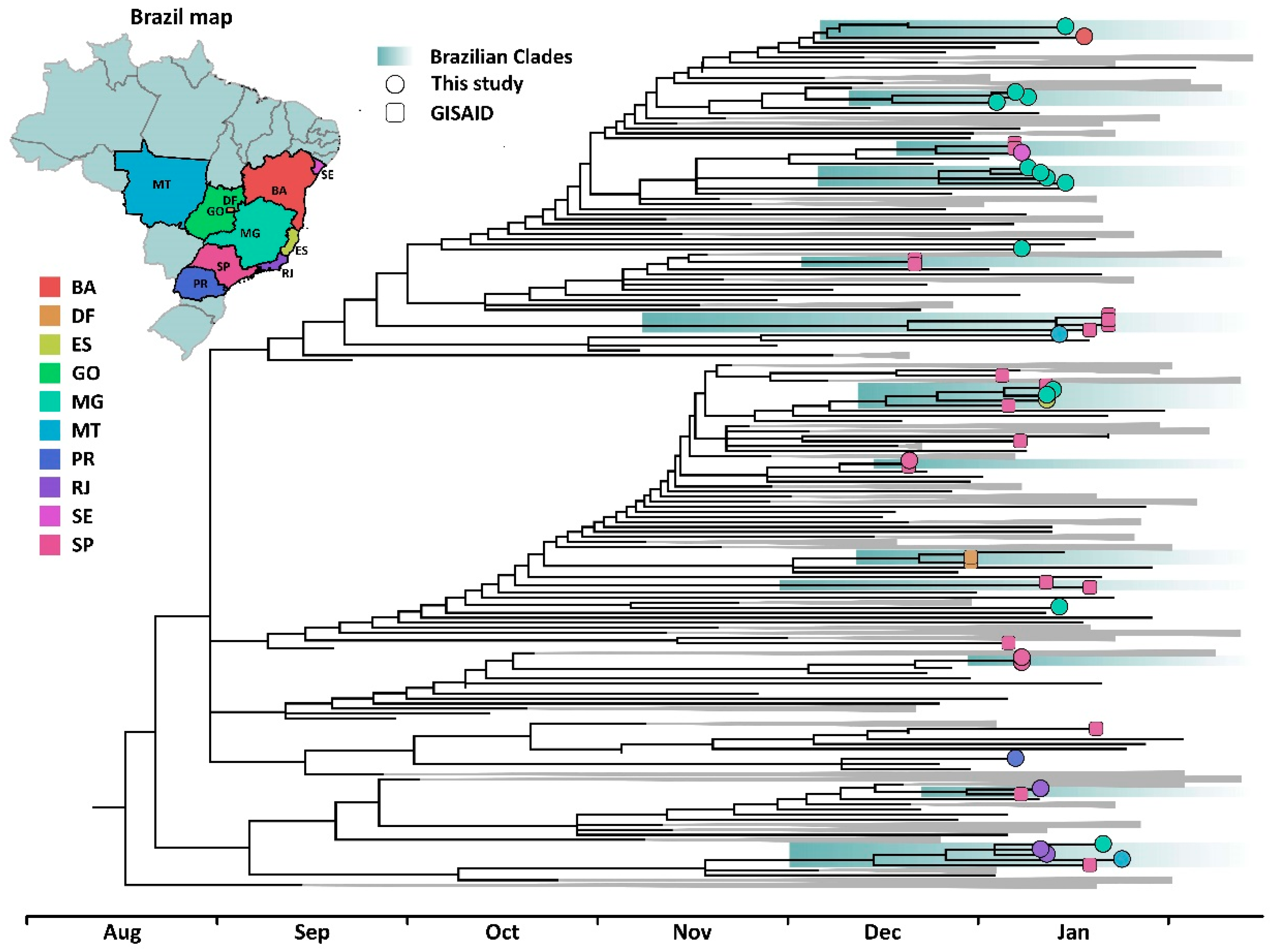
2. Materials and Methods
3. Results and Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Volz, E.; Mishra, S.; Chand, M.; Barrett, J.; Johnson, R.; Geidelberg, L.; Hinsley, W.; Laydon, D.; Dabrera, G.; O’Toole, Á.; et al. Assessing transmissibility of SARS-CoV-2 lineage B.1.1.7 in England. Nature 2021, 593, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Tegally, H.; Wilkinson, E.; Giovanetti, M.; Iranzadeh, A.; Fonseca, V.; Giandhari, J.; Doolabh, D.; Pillay, S.; San, E.; Msomi, N.; et al. Detection of a SARS-CoV-2 variant of concern in South Africa. Nature 2021, 592, 438–443. [Google Scholar] [CrossRef]
- Faria, N.; Mellan, T.; Whittaker, C.; Claro, I.; Candido, D.; Mishra, S.; Crispim, M.; Sales, F.; Hawryluk, I.; McCrone, J.; et al. Genomics and epidemiology of the P.1 SARS-CoV-2 lineage in Manaus, Brazil. Science 2021, 372, 815–821. [Google Scholar] [CrossRef]
- Voloch, C.; Francisco, R., Jr.; de Almeida, L.; Cardoso, C.; Brustolini, O.; Gerber, A.; Guimarães, A.; Mariani, D.; da Costa, R.; Ferreira, O., Jr.; et al. Genomic characterization of a novel SARS-CoV-2 lineage from Rio de Janeiro, Brazil. J. Virol. 2021, 95. [Google Scholar] [CrossRef]
- Resende, P.; Gräf, T.; Paixão, A.; Appolinario, L.; Lopes, R.; Mendonça, A.; da Rocha, A.; Motta, F.; Neto, L.; Khouri, R.; et al. A Potential SARS-CoV-2 Variant of Interest (VOI) Harboring Mutation E484K in the Spike Protein Was Identified within Lineage B.1.1.33 Circulating in Brazil. Viruses 2021, 13, 724. [Google Scholar] [CrossRef]
- Resende, P.; Gräf, T.; Lima Neto, L.; Appolinario, L.; Lopes, R.; Mendonça, A.; Rocha, A.; Motta, F.; Delatorre, E.; Wallau, G.; et al. Identification of a New B.1.1.33 SARS-CoV-2 Variant of Interest (VOI) Circulating in Brazil with Mutation E484K and Multiple Deletions in the Amino (N)-Terminal Domain of the Spike Protein. Available online: https://virological.org/t/identification-of-a-new-b-1-1-33-sars-cov-2-variant-of-interest-voi-circulating-in-brazil-with-mutation-e484k-and-multiple-deletions-in-the-amino-n-terminal-domain-of-the-spike-protein/675 (accessed on 11 May 2021).
- Francisco, R., Jr.; Benites, L.; Lamarca, A.; Almeida, L.; Hansen, A.; Gularte, J.; Demoliner, M.; Gerber, A.; Guimarães, A.; Antunes, A.; et al. Pervasive transmission of E484K and emergence of VUI-NP13L with evidence of SARS-CoV-2 co-infection events by two different lineages in Rio Grande do Sul, Brazil. Virus Res. 2021, 296, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Claro, I.; Cristina, F.; Ramundo, M.; Candido, D.; Silva, C.; de Jesus, J.; Manuli, E.; de Oliveira, C.; Scarpelli, L.; Campana, G.; et al. Local Transmission of SARS-CoV-2 Lineage B.1.1.7, Brazil, December 2020. Emerg Infect Dis. 2021, 3, 970–972. [Google Scholar] [CrossRef] [PubMed]
- Bal, A.; Destras, G.; Gaymard, A.; Stefic, K.; Marlet, J.; Eymieux, S.; Regue, H.; Semanas, Q.; d’Aubarede, C.; Billaud, G.; et al. Two-step strategy for the identification of SARS-CoV-2 variant of concern 202012/01 and other variants with spike deletion H69-V70, France, August to December 2020. medRxiv 2021. [Google Scholar] [CrossRef]
- Minh, B.; Schmidt, H.; Chernomor, O.; Schrempf, D.; Woodhams, M.; Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Kalyaanamoorthy, S.; Minh, B.; Wong, T.; Haeseler, A.; Jermiin, L. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Sagulenko, P.; Puller, V.; Neher, R. TreeTime: Maximum-likelihood phylodynamic analysis. Virus Evol. 2018, 4, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Candido, D.; Claro, I.; de Jesus, J.; Souza, W.; Moreira, F.; Dellicour, S.; Mellan, T.; Plessis, L.; Pereira, R.; Sales, F.; et al. Evolution and epidemic spread of SARS-CoV-2 in Brazil. Science 2020, 369, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Davies, N.; Abbott, S.; Barnard, R.; Jarvis, C.; Kucharski, A.; Munday, J.; Pearson, C.; Russell, T.; Tully, D.; Washburne, A.; et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 2021, 372. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreira, F.R.R.; Bonfim, D.M.; Zauli, D.A.G.; Silva, J.P.; Lima, A.B.; Malta, F.S.V.; Ferreira, A.C.S.; Pardini, V.C.; Magalhães, W.C.S.; Queiroz, D.C.; et al. Epidemic Spread of SARS-CoV-2 Lineage B.1.1.7 in Brazil. Viruses 2021, 13, 984. https://doi.org/10.3390/v13060984
Moreira FRR, Bonfim DM, Zauli DAG, Silva JP, Lima AB, Malta FSV, Ferreira ACS, Pardini VC, Magalhães WCS, Queiroz DC, et al. Epidemic Spread of SARS-CoV-2 Lineage B.1.1.7 in Brazil. Viruses. 2021; 13(6):984. https://doi.org/10.3390/v13060984
Chicago/Turabian StyleMoreira, Filipe R. R., Diego M. Bonfim, Danielle A. G. Zauli, Joice P. Silva, Aline B. Lima, Frederico S. V. Malta, Alessandro C. S. Ferreira, Victor C. Pardini, Wagner C. S. Magalhães, Daniel C. Queiroz, and et al. 2021. "Epidemic Spread of SARS-CoV-2 Lineage B.1.1.7 in Brazil" Viruses 13, no. 6: 984. https://doi.org/10.3390/v13060984
APA StyleMoreira, F. R. R., Bonfim, D. M., Zauli, D. A. G., Silva, J. P., Lima, A. B., Malta, F. S. V., Ferreira, A. C. S., Pardini, V. C., Magalhães, W. C. S., Queiroz, D. C., Souza, R. M., Geddes, V. E. V., Costa, W. C., Moreira, R. G., Faria, N. R., Voloch, C. M., Souza, R. P., & Aguiar, R. S. (2021). Epidemic Spread of SARS-CoV-2 Lineage B.1.1.7 in Brazil. Viruses, 13(6), 984. https://doi.org/10.3390/v13060984




