The Second Wave of SARS-CoV-2 Circulation—Antibody Detection in the Domestic Cat Population in Germany
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Collection Date | RBD ELISA | iIFT SARS-CoV-2 | VNT SARS-CoV-2 |
|---|---|---|---|
| Absorbance/Result | ND100 | ||
| 15 September 2020 | 0.799/positive | >1:1024 | 1:1024 |
| 23 September 2020 | 0.685/positive | >1:1024 | neg. 1 |
| 6 October 2020 | 0.364/positive | >1:1024 | 1:101.6 |
| 28 October 2020 | 0.429/positive | 1:512 | neg. 1 |
| 10 November 2020 | 0.416/positive | >1:1024 | neg. 1 |
| 11 November 2020 | 0.798/positive | 1:64 | neg. 1 |
| 7 December2020 | 0.588/positive | >1:1024 | neg. 1 |
| 9 December 2020 | 0.822/positive | 1:64 | neg. 1 |
| 9 December 2020 | 0.364/positive | >1:1024 | neg. 1 |
| 4 January 2021 | 0.354/positive | 1:128 | neg. 1 |
| 5 January 2021 | 0.837/positive | >1:1024 | 1:40.32 |
| 8 January 2021 | 0.709/positive | >1:1024 | 1:101.6 |
| 11 January 2021 | 0.425/positive | >1:1024 | neg. 1 |
| 14 January 2021 | 0.827/positive | >1:1024 | 1:161.3 |
| 9 February 2021 | 1.108/positive | 1:256 | neg. 1 |
| 19 February 2021 | 0.539/positive | 1:256 | neg. 1 |
| 2 October 2020 | 0.264/borderline | neg. 1 | neg. 1 |
| 21 October 2020 | 0.234/borderline | neg. 1 | neg. 1 |
| 28 October 2020 | 0.208/borderline | neg. 1 | neg. 1 |
| 2 November 2020 | 0.210/borderline | neg. 1 | neg. 1 |
| 11 November 2020 | 0.265/borderline | 1/128 | neg. 1 |
| 29 December 2020 | 0.259/borderline | neg. 1 | neg. 1 |
| 11 January 2021 | 0.215/borderline | neg. 1 | neg. 1 |
| 22 January 2021 | 0.223/borderline | 1:16 | neg. 1 |
| 10 February 2021 | 0.212/borderline | neg. 1 | neg. 1 |
References
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733. [Google Scholar] [CrossRef]
- Dong, E.; Du, H.; Gardner, L. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect. Dis. 2020, 20, 533–534. [Google Scholar] [CrossRef]
- Salzberger, B.; Buder, F.; Lampl, B.; Ehrenstein, B.; Hitzenbichler, F.; Holzmann, T.; Schmidt, B.; Hanses, F. Epidemiology of SARS-CoV-2. Infection 2020. [Google Scholar] [CrossRef]
- Robert Koch-Institut. Coronavirus Disease 2019 (COVID-19)—Situation Report of the Robert Koch Institute. Available online: https://www.rki.de/EN/Content/infections/epidemiology/outbreaks/COVID-19/Situationsberichte_Tab.html (accessed on 12 March 2021).
- Graichen, H. What is the difference between the first and the second/third wave of Covid-19?—German perspective. J. Orthop. 2021. [Google Scholar] [CrossRef]
- Fontanet, A.; Autran, B.; Lina, B.; Kieny, M.P.; Karim, S.S.A.; Sridhar, D. SARS-CoV-2 variants and ending the COVID-19 pandemic. Lancet 2021, 397, 952–954. [Google Scholar] [CrossRef]
- European Centre for Disease Prevention and Control. SARS-CoV-2—Increased Circulation of Variants of Concern and Vaccine Rollout in the EU/EEA, 14th Update—15 February 2021. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/RRA-covid-19-14th-update-15-feb-2021.pdf (accessed on 25 March 2021).
- Hosie, M.J.; Hofmann-Lehmann, R.; Hartmann, K.; Egberink, H.; Truyen, U.; Addie, D.D.; Belák, S.; Boucraut-Baralon, C.; Frymus, T.; Lloret, A.; et al. Anthropogenic infection of cats during the 2020 COVID-19 pandemic. Viruses 2021, 13, 185. [Google Scholar] [CrossRef]
- Shi, J.; Wen, Z.; Zhong, G.; Yang, H.; Wang, C.; Huang, B.; Liu, R.; He, X.; Shuai, L.; Sun, Z.; et al. Susceptibility of ferrets, cats, dogs, and other domesticated animals to SARS-coronavirus 2. Science 2020, 368, 1016–1020. [Google Scholar] [CrossRef] [PubMed]
- Bosco-Lauth, A.M.; Hartwig, A.E.; Porter, S.M.; Gordy, P.W.; Nehring, M.; Byas, A.D.; VandeWoude, S.; Ragan, I.K.; Maison, R.M.; Bowen, R.A. Experimental infection of domestic dogs and cats with SARS-CoV-2: Pathogenesis, transmission, and response to reexposure in cats. Proc. Natl. Acad. Sci. USA 2020, 117, 26382–26388. [Google Scholar] [CrossRef] [PubMed]
- Gaudreault, N.N.; Trujillo, J.D.; Carossino, M.; Meekins, D.A.; Morozov, I.; Madden, D.W.; Indran, S.V.; Bold, D.; Balaraman, V.; Kwon, T.; et al. SARS-CoV-2 infection, disease and transmission in domestic cats. Emerg. Microbes Infect. 2020, 9, 2322–2332. [Google Scholar] [CrossRef] [PubMed]
- Halfmann, P.J.; Hatta, M.; Chiba, S.; Maemura, T.; Fan, S.; Takeda, M.; Kinoshita, N.; Hattori, S.I.; Sakai-Tagawa, Y.; Iwatsuki-Horimoto, K.; et al. Transmission of SARS-CoV-2 in domestic cats. N. Engl. J. Med. 2020, 383, 592–594. [Google Scholar] [CrossRef]
- Barrs, V.; Peiris, M.; Tam, K.W.S.; Law, P.Y.T.; Brackman, C.; To, E.M.W.; Yu, V.Y.T.; Chu, D.K.W.; Perera, R.A.P.M.; Sit, T.H.C. SARS-CoV-2 in quarantined domestic cats from COVID-19 households or close contacts, Hong Kong, China. Emerg. Infect. Dis. 2020, 26, 3071–3074. [Google Scholar] [CrossRef] [PubMed]
- Neira Ramirez, V.; Brito, B.; Aguero, B.; Berrios, F.; Valdes, V.; Gutierrez, A.; Ariyama, N.; Espinoza, P.; Retamal, P.; Holmes, E.C.; et al. A household case evidences shorter shedding of SARS-CoV-2 in naturally infected cats compared to their human owners. Emerg. Microbes Infect. 2020, 10, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.; Smith, D.; Ghai, R.R.; Wallace, R.M.; Torchetti, M.K.; Loiacono, C.; Murrell, L.S.; Carpenter, A.; Moroff, S.; Rooney, J.A.; et al. First reported cases of SARS-CoV-2 infection in companion animals—New York, March-April 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 710–713. [Google Scholar] [CrossRef]
- Sailleau, C.; Dumarest, M.; Vanhomwegen, J.; Delaplace, M.; Caro, V.; Kwasiborski, A.; Hourdel, V.; Chevaillier, P.; Barbarino, A.; Comtet, L.; et al. First detection and genome sequencing of SARS-CoV-2 in an infected cat in France. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Segalés, J.; Puig, M.; Rodon, J.; Avila-Nieto, C.; Carrillo, J.; Cantero, G.; Terrón, M.T.; Cruz, S.; Parera, M.; Noguera-Julián, M.; et al. Detection of SARS-CoV-2 in a cat owned by a COVID-19-affected patient in Spain. Proc. Natl. Acad. Sci. USA 2020, 117, 24790–24793. [Google Scholar] [CrossRef]
- Garigliany, M.; Van Laere, A.S.; Clercx, C.; Giet, D.; Escriou, N.; Huon, C.; van der Werf, S.; Eloit, M.; Desmecht, D. SARS-CoV-2 natural transmission from human to cat, Belgium, March 2020. Emerg. Infect. Dis. 2020, 26, 3069. [Google Scholar] [CrossRef] [PubMed]
- OIE. Events in Animals. Available online: https://www.oie.int/en/scientific-expertise/specific-information-and-recommendations/questions-and-answers-on-2019novel-coronavirus/events-in-animals/ (accessed on 25 January 2021).
- Klaus, J.; Palizzotto, C.; Zini, E.; Meli, M.L.; Leo, C.; Egberink, H.; Zhao, S.; Hofmann-Lehmann, R. SARS-CoV-2 infection and antibody response in a symptomatic cat from Italy with intestinal B-cell lymphoma. Viruses 2021, 13, 527. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, H.; Gao, J.; Huang, K.; Yang, Y.; Hui, X.; He, X.; Li, C.; Gong, W.; Zhang, Y.; et al. A serological survey of SARS-CoV-2 in cat in Wuhan. Emerg. Microbes Infect. 2020, 9, 2013–2019. [Google Scholar] [CrossRef]
- Fritz, M.; Rosolen, B.; Krafft, E.; Becquart, P.; Elguero, E.; Vratskikh, O.; Denolly, S.; Boson, B.; Vanhomwegen, J.; Gouilh, M.A.; et al. High prevalence of SARS-CoV-2 antibodies in pets from COVID-19+ households. One Health 2020, 11, 100192. [Google Scholar] [CrossRef]
- Spada, E.; Vitale, F.; Bruno, F.; Castelli, G.; Reale, S.; Perego, R.; Baggiani, L.; Proverbio, D. A pre- and during pandemic survey of Sars-Cov-2 infection in stray colony and shelter cats from a high endemic area of Northern Italy. Viruses 2021, 13, 618. [Google Scholar] [CrossRef]
- Patterson, E.I.; Elia, G.; Grassi, A.; Giordano, A.; Desario, C.; Medardo, M.; Smith, S.L.; Anderson, E.R.; Prince, T.; Patterson, G.T.; et al. Evidence of exposure to SARS-CoV-2 in cats and dogs from households in Italy. Nat. Commun. 2020, 11, 6231. [Google Scholar] [CrossRef]
- Michelitsch, A.; Hoffmann, D.; Wernike, K.; Beer, M. Occurrence of antibodies against SARS-CoV-2 in the domestic cat population of Germany. Vaccines 2020, 8, 772. [Google Scholar] [CrossRef] [PubMed]
- Wernike, K.; Aebischer, A.; Michelitsch, A.; Hoffmann, D.; Freuling, C.; Balkema-Buschmann, A.; Graaf, A.; Müller, T.; Osterrieder, N.; Rissmann, M.; et al. Multi-species ELISA for the detection of antibodies against SARS-CoV-2 in animals. Transbound. Emerg. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Schlottau, K.; Rissmann, M.; Graaf, A.; Schön, J.; Sehl, J.; Wylezich, C.; Höper, D.; Mettenleiter, T.C.; Balkema-Buschmann, A.; Harder, T.; et al. SARS-CoV-2 in fruit bats, ferrets, pigs, and chickens: An experimental transmission study. Lancet Microbe 2020, 15, e218–e225. [Google Scholar] [CrossRef]
- Schulz, C.; Wylezich, C.; Wernike, K.; Gründl, M.; Dangl, A.; Baechlein, C.; Hoffmann, D.; Röhrs, S.; Hepner, S.; Ackerermann, N.; et al. Natural SARS-CoV-2 infection of a therapy cat during a cluster outbreak in a retirement home. In Proceedings of the 30th Annual Meeting of the Society for Virology, Online Conference, 24–26 March 2021. [Google Scholar]
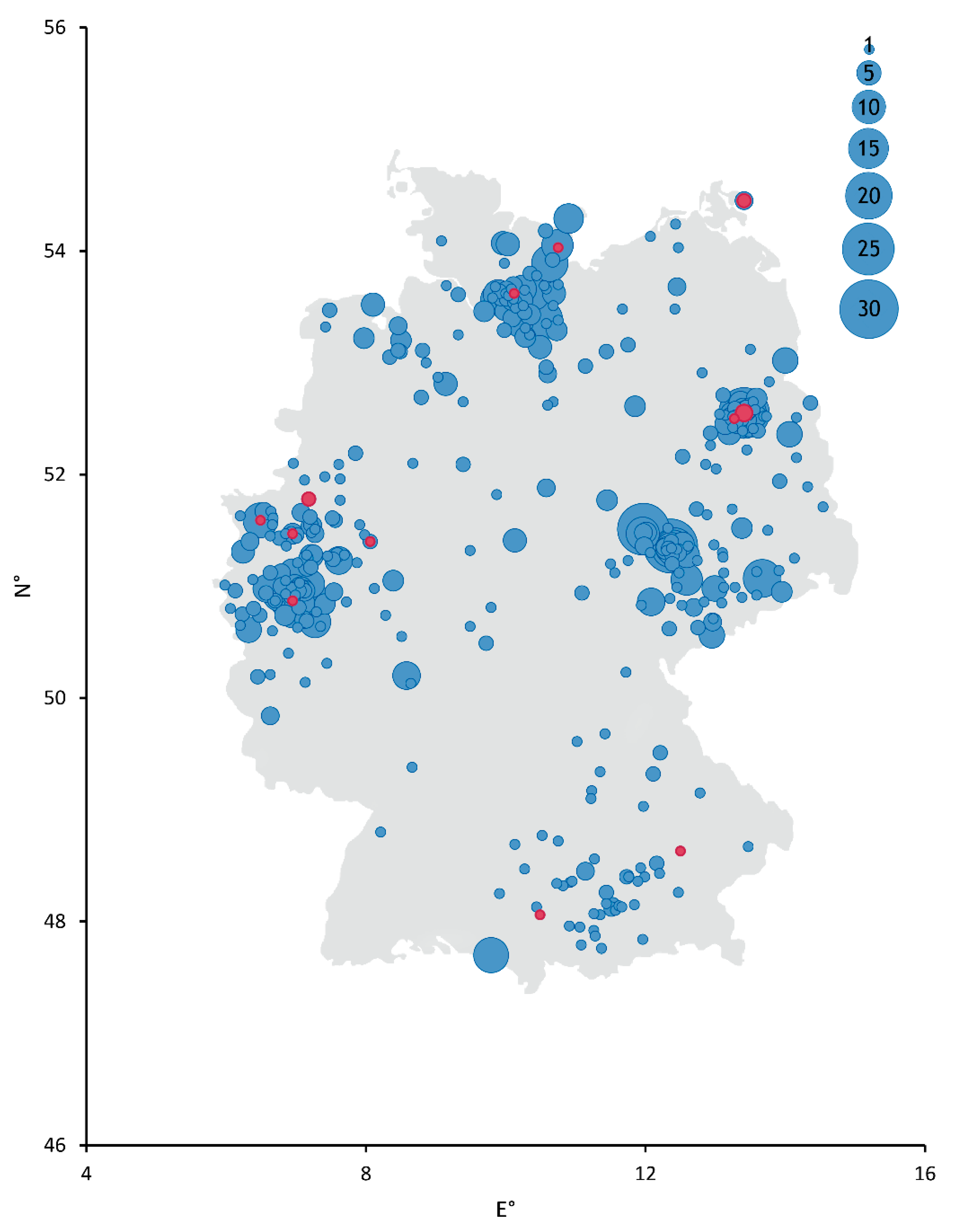
| Federal State | Month 2020/21 | ||||||
|---|---|---|---|---|---|---|---|
| September | October | November | December | January | February | Σ | |
| Baden-Württemberg | 0/0 | 0/0 | 0/2 | 0/1 | 0/0 | 0/0 | 0/3 |
| Bavaria | 0/19 | 0/1 | 2/27 | 0/7 | 0/5 | 0/0 | 2/59 |
| Berlin | 0/3 | 1/74 | 0/4 | 1/31 | 1/47 | 0/1 | 3/160 |
| Brandenburg | 0/0 | 0/17 | 0/3 | 1/13 | 0/19 | 0/15 | 1/67 |
| Bremen | 0/0 | 0/0 | 0/0 | 0/1 | 0/0 | 0/2 | 0/3 |
| Hamburg | 0/10 | 0/27 | 0/33 | 0/31 | 1/25 | 0/34 | 1/160 |
| Hesse | 0/7 | 0/3 | 0/3 | 0/1 | 0/2 | 0/0 | 0/16 |
| Lower Saxony | 0/2 | 0/12 | 0/6 | 0/12 | 0/18 | 0/7 | 0/57 |
| Mecklenburg-Western Pomerania | 0/1 | 0/0 | 0/0 | 1/1 | 0/1 | 1/14 | 2/17 |
| North Rhine-Westphalia | 2/63 | 1/112 | 0/13 | 0/56 | 3/51 | 0/0 | 6/295 |
| Rhineland-Palatinate | 0/4 | 0/4 | 0/0 | 0/0 | 0/0 | 0/0 | 0/8 |
| Saarland | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 | 0/0 |
| Saxony | 0/35 | 0/38 | 0/40 | 0/40 | 0/44 | 0/0 | 0/197 |
| Saxony-Anhalt | 0/17 | 0/12 | 0/13 | 0/10 | 0/8 | 0/0 | 0/60 |
| Schleswig-Holstein | 0/1 | 0/13 | 0/14 | 0/16 | 0/12 | 1/7 | 1/63 |
| Thuringia | 0/2 | 0/2 | 0/1 | 0/2 | 0/1 | 0/0 | 0/8 |
| Σ | 2/164 | 2/315 | 2/159 | 3/222 | 5/233 | 2/80 | 16/1173 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michelitsch, A.; Schön, J.; Hoffmann, D.; Beer, M.; Wernike, K. The Second Wave of SARS-CoV-2 Circulation—Antibody Detection in the Domestic Cat Population in Germany. Viruses 2021, 13, 1009. https://doi.org/10.3390/v13061009
Michelitsch A, Schön J, Hoffmann D, Beer M, Wernike K. The Second Wave of SARS-CoV-2 Circulation—Antibody Detection in the Domestic Cat Population in Germany. Viruses. 2021; 13(6):1009. https://doi.org/10.3390/v13061009
Chicago/Turabian StyleMichelitsch, Anna, Jacob Schön, Donata Hoffmann, Martin Beer, and Kerstin Wernike. 2021. "The Second Wave of SARS-CoV-2 Circulation—Antibody Detection in the Domestic Cat Population in Germany" Viruses 13, no. 6: 1009. https://doi.org/10.3390/v13061009
APA StyleMichelitsch, A., Schön, J., Hoffmann, D., Beer, M., & Wernike, K. (2021). The Second Wave of SARS-CoV-2 Circulation—Antibody Detection in the Domestic Cat Population in Germany. Viruses, 13(6), 1009. https://doi.org/10.3390/v13061009






