Prevalence of Neutralizing Antibodies to Canine Distemper Virus and Response to Vaccination in Client-Owned Adult Healthy Dogs
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Protocol
2.3. Detection of CDV Antibodies by VN
2.4. Reverse Transcription (RT)-PCR and Sequencing of the Full-Length Canine Distemper Hemagglutinin (H) Gene
2.5. Statistical Analysis
3. Results
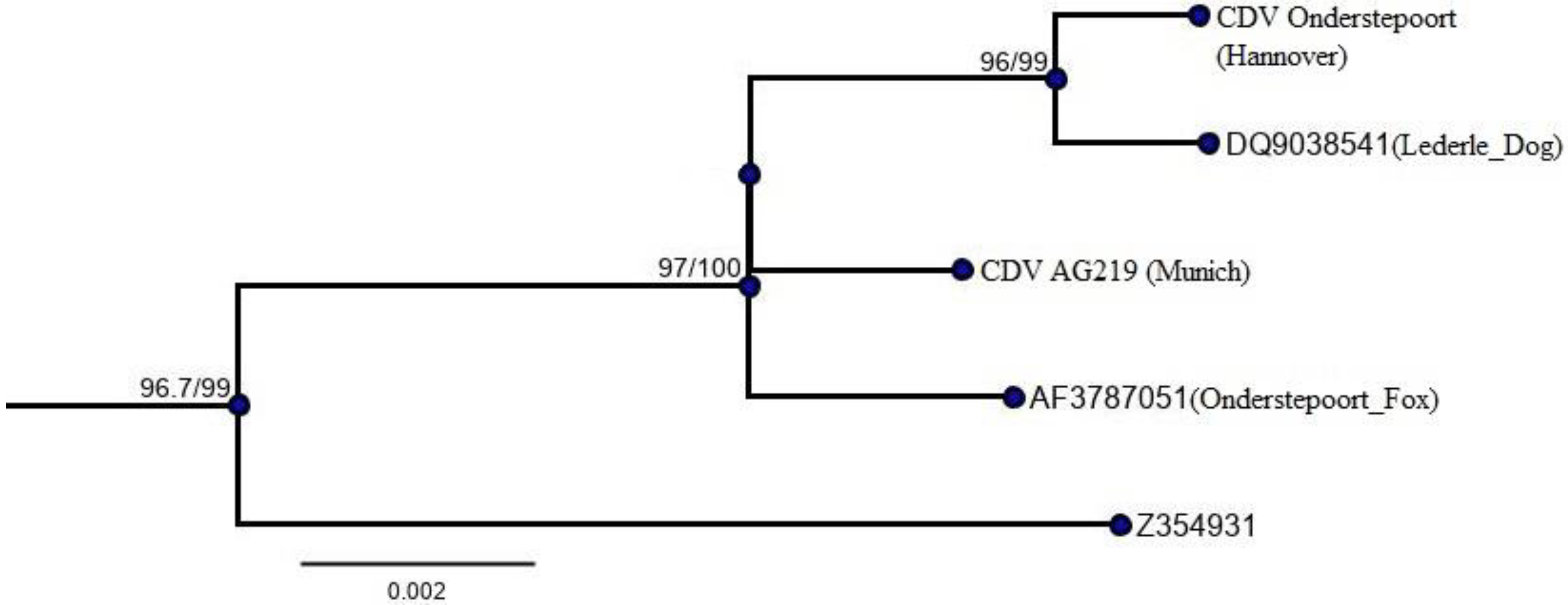
3.1. Sequences of Isolate CDV AG219 and CDV Onderstepoort
3.2. Comparison of CDV Antibodies in VN Using Isolate Ag219 and Onderstepoort
3.3. Pre-Vaccination Antibodies against Isolate CDV AG219
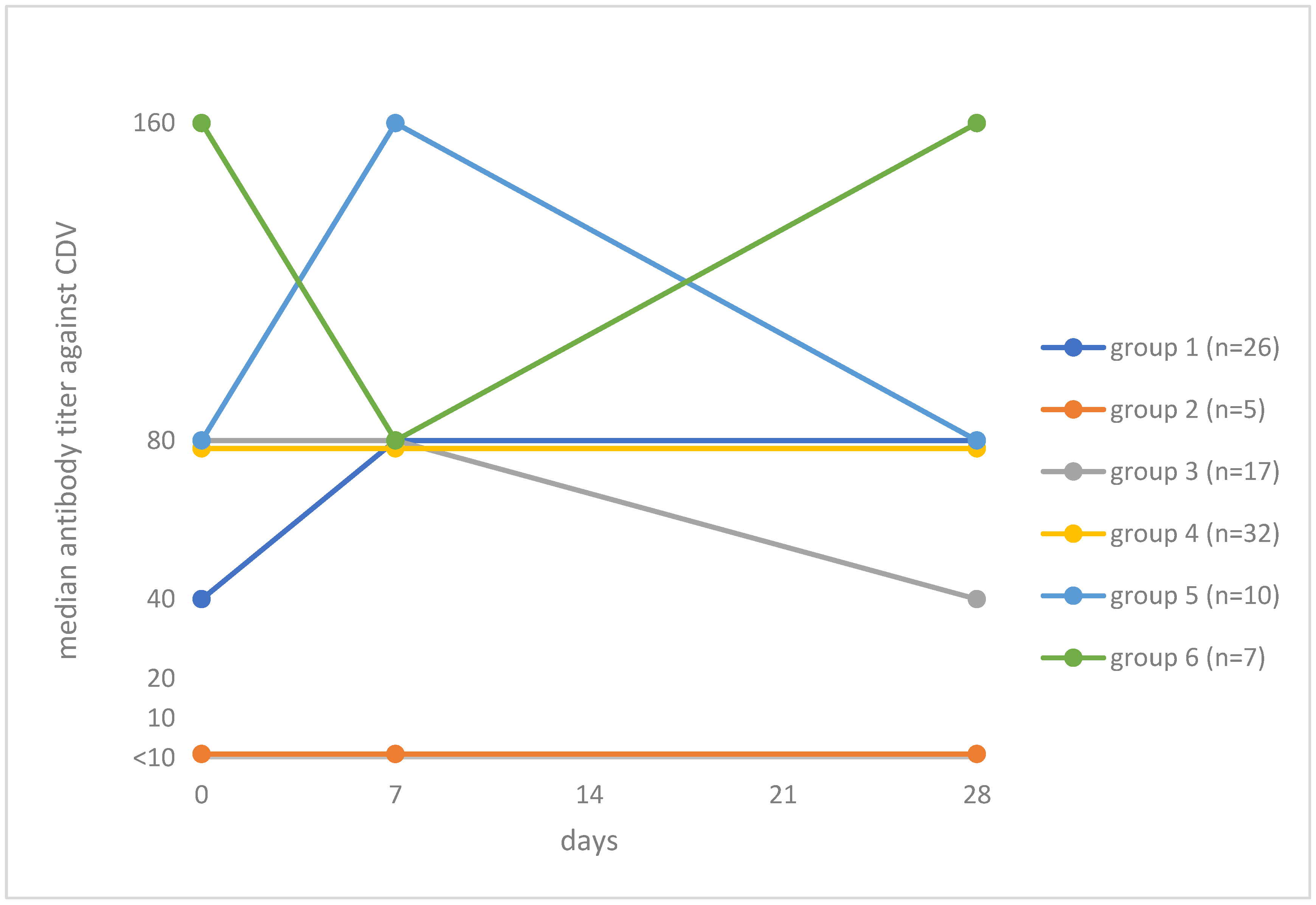
3.4. Titer Increase after Vaccination against Isolate CDV AG219
- Group 1: Titer ≥ 10 on day 0 and subsequent increase in titer by day 28 (n = 26; 26.8%). Median titers: day 0, 40; day 7, 80; day 28, 80;
- Group 2: Titer < 10 pre- and titer < 10 post-vaccination (n = 5; 5.2%). Median titers on each day <10;
- Group 3: Titer ≥ 10 on day 0 and subsequent decrease in titer by day 28 (n = 17; 17.5%). Median titers: day 0, 80; day 7, 80; day 28, 40;
- Group 4: Titer ≥ 10 on day 0 and no change in titer after vaccination (n = 32; 33.0%). Median titers: day 0, 80; day 7, 80; day 28, 80;
- Group 5: Titer ≥ 10 on day 0, an increase in titer by day 7 and a decrease in titer by day 28 (n = 10; 10.3%). Median titers: day 0, 80; day 7, 160; day 28, 80;
- Group 6: Titer ≥ 10 on day 0, a decrease in titer by day 7 and an increase in titer by day 28 (n = 7; 7.2%). Median titers: day 0, 160; day 7, 80; day 28, 160.
3.5. Vaccine-Associated Adverse Events
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dabbagh, A.; Patel, M.K.; Gacic-Dobo, M.; Mulders, M.N.; Okwo-Bele, J.M.; Kretsinger, K.; Papania, M.J.; Rota, P.A.; Goodson, J.L. Progress toward regional measles elimination—Worldwide, 2000–2016. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1148–1153. [Google Scholar] [CrossRef]
- World Health Organization. Measles. Available online: https://who.int/news-room/fact-sheets/detail/measles (accessed on 5 December 2019).
- Koch, A.; Krönert, C.; Lotti, T.; Vojvodic, A.; Wollina, U. Adult measles—Case reports of a highly contagious disease. Open Access Maced. J. Med. Sci. 2019, 7, 3009–3012. [Google Scholar] [CrossRef] [PubMed]
- Willi, B.; Spiri, A.M.; Meli, M.L.; Grimm, F.; Beatrice, L.; Riond, B.; Bley, T.; Jordi, R.; Dennler, M.; Hofmann-Lehmann, R. Clinical and molecular investigation of a canine distemper outbreak and vector-borne infections in a group of rescue dogs imported from Hungary to Switzerland. BMC Vet. Res. 2015, 11, 154. [Google Scholar] [CrossRef]
- Day, M.J.; Karkare, U.; Schultz, R.D.; Squires, R.; Tsujimoto, H. Recommendations on vaccination for Asian small animal practitioners: A report of the WSAVA Vaccination Guidelines Group. J. Small Anim. Pract. 2015, 56, 77–95. [Google Scholar] [CrossRef] [PubMed]
- Jensen, W.A.; Totten, J.S.; Lappin, M.R.; Schultz, R.D. Use of serologic tests to predict resistance to canine distemper virus-induced disease in vaccinated dogs. J. Vet. Diagn. Investig. 2015, 27, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Schultz, R.D.; Thiel, B.; Mukhtar, E.; Sharp, P.; Larson, L.J. Age and long-term protective immunity in dogs and cats. J. Comp. Pathol. 2010, 142, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Plotkin, S.A. Correlates of protection induced by vaccination. Clin. Vaccine Immunol. 2010, 17, 1055–1065. [Google Scholar] [CrossRef]
- Schultz, R.D. Duration of immunity for canine and feline vaccines: A review. Vet. Microbiol. 2006, 117, 75–79. [Google Scholar] [CrossRef] [PubMed]
- Olson, P.; Finnsdottir, H.; Klingeborn, B.; Hedhammar, A. Duration of antibodies elicited by canine distemper virus vaccinations in dogs. Vet. Rec. 1997, 141, 654–655. [Google Scholar]
- Böhm, M.; Thompson, H.; Weir, A.; Hasted, A.M.; Maxwell, N.S.; Herrtage, M.E. Serum antibody titres to canine parvovirus, adenovirus and distemper virus in dogs in the UK which had not been vaccinated for at least three years. Vet. Rec. 2004, 154, 457–463. [Google Scholar] [CrossRef]
- McCaw, D.L.; Thompson, M.; Tate, D.; Bonderer, A.; Chen, Y.J. Serum distemper virus and parvovirus antibody titers among dogs brought to a veterinary hospital for revaccination. J. Am. Vet. Med. Assoc. 1998, 213, 72–75. [Google Scholar]
- Mitchell, S.A.; Zwijnenberg, R.J.; Huang, J.; Hodge, A.; Day, M.J. Duration of serological response to canine parvovirus-type 2, canine distemper virus, canine adenovirus type 1 and canine parainfluenza virus in client-owned dogs in Australia. Aust. Vet. J. 2012, 90, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Twark, L.; Dodds, W.J. Clinical use of serum parvovirus and distemper virus antibody titers for determining revaccination strategies in healthy dogs. J. Am. Vet. Med. Assoc. 2000, 217, 1021–1024. [Google Scholar] [CrossRef] [PubMed]
- Moore, G.E.; Guptill, L.F.; Ward, M.P.; Glickman, N.W.; Faunt, K.K.; Lewis, H.B.; Glickman, L.T. Adverse events diagnosed within three days of vaccine administration in dogs. J. Am. Vet. Med. Assoc. 2005, 227, 1102–1108. [Google Scholar] [CrossRef]
- Cußler, K.; Schwedinger, E. Pharmakovigilanz für Qualität, Wirksamkeit und Unbedenklichkeit von Tierarzneimitteln, Nebenwirkungen nach Impfung von Deutschen Pinschern. DTBL 2012, 12, 1715–1716. [Google Scholar]
- Ohmori, K.; Sakaguchi, M.; Kaburagi, Y.; Maeda, S.; Masuda, K.; Ohno, K.; Tsujimoto, H. Suspected allergic reactions after vaccination in 85 dogs in Japan. Vet. Rec. 2005, 156, 87–88. [Google Scholar] [CrossRef] [PubMed]
- Bergmann, M.; Schwertler, S.; Reese, S.; Speck, S.; Truyen, U.; Hartmann, K. Antibody response to feline panleukopenia virus vaccination in healthy adult cats. J. Feline Med. Surg. 2018, 20, 1087–1093. [Google Scholar] [CrossRef]
- Riedl, M.; Truyen, U.; Reese, S.; Hartmann, K. Prevalence of antibodies to canine parvovirus and reaction to vaccination in client-owned, healthy dogs. Vet. Rec. 2015, 177, 597. [Google Scholar] [CrossRef] [PubMed]
- Welborn, L.V.; DeVries, J.G.; Ford, R.; Franklin, R.T.; Hurley, K.F.; McClure, K.D.; Paul, M.A.; Schultz, R.D. 2011 AAHA canine vaccination guidelines. J. Am. Anim. Hosp. Assoc. 2011, 47, 1–42. [Google Scholar] [PubMed]
- Seki, F.; Ono, N.; Yamaguchi, R.; Yanagi, Y. Efficient isolation of wild strains of canine distemper virus in vero cells expressing canine SLAM (CD150) and their adaptability to marmoset B95a cells. J. Virol. 2003, 77, 9943–9950. [Google Scholar] [CrossRef]
- Liao, P.; Guo, L.; Wen, Y.; Yang, Y.; Cheng, S. Phylogenetic features of hemagglutinin gene in canine distemper virus strains from different genetic lineages. Int. J. Clin. Exp. Med. 2015, 8, 6607–6612. [Google Scholar] [PubMed]
- Ke, G.M.; Ho, C.H.; Chiang, M.J.; Sanno-Duanda, B.; Chung, C.S.; Lin, M.Y.; Shi, Y.Y.; Yang, M.H.; Tyan, Y.C.; Liao, P.C.; et al. Phylodynamic analysis of the canine distemper virus hemagglutinin gene. BMC Vet. Res. 2015, 11, 164. [Google Scholar] [CrossRef] [PubMed]
- Pan American Health Organization. Measles Eradication: Field Guide; Pan American Health Organization: Washington, DC, USA, 1999; Volume 41, pp. 49–52. [Google Scholar]
- Rikula, U.; Nuotio, L.; Sihvonen, L. Vaccine coverage, herd immunity and occurrence of canine distemper from 1990–1996 in Finland. Vaccine 2007, 25, 7994–7998. [Google Scholar] [CrossRef] [PubMed]
- Litster, A.; Nichols, J.; Volpe, A. Prevalence of positive antibody test results for canine parvovirus (CPV) and canine distemper virus (CDV) and response to modified live vaccination against CPV and CDV in dogs entering animal shelters. Vet. Microbiol. 2012, 157, 86–90. [Google Scholar] [CrossRef]
- Jozwik, A.; Frymus, T.; Mizak, B.; Rzezutka, A. Antibody titres against canine distemper virus in vaccinated and unvaccinated dogs. J. Vet. Med. B Infect. Dis. Vet. Public Health 2004, 51, 99–103. [Google Scholar] [CrossRef]
- Appel, M.J.G.; Gillespie, J.H. Canine distemper virus. In Virology Monographs 11: Canine Distemper Virus Marburg Virus; Gard, S., Hallauer, C., Meyer, K.F., Eds.; Springer: New York, NY, USA, 1972; pp. 1–96. [Google Scholar]
- Martella, V.; Elia, G.; Buonavoglia, C. Canine distemper virus. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 787–797. [Google Scholar] [CrossRef]
- Newbury, S.; Larson, L.J.; Schultz, R. Canine distemper virus. In Infectious Disease Management in Animal Shelters, 1st ed.; Miller, L., Hurley, K., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2009; pp. 161–172. [Google Scholar]
- Schulz, B.S.; Kurz, S.; Weber, K.; Balzer, H.J.; Hartmann, K. Detection of respiratory viruses and Bordetella bronchiseptica in dogs with acute respiratory tract infections. Vet. J. 2014, 201, 365–369. [Google Scholar] [CrossRef]
- Schulz, B.S.; Strauch, C.; Mueller, R.S.; Eichhorn, W.; Hartmann, K. Comparison of the prevalence of enteric viruses in healthy dogs and those with acute haemorrhagic diarrhoea by electron microscopy. J. Small Anim. Pract. 2008, 49, 84–88. [Google Scholar] [CrossRef]
- Haralambieva, I.C.H.; Kennedy, R.B.; Ovsyannikova, I.G.; Whitaker, J.A.; Poland, G.A. Variability in humoral immunity to measles vaccine: New Developments. Trends Mol. Med. 2015, 21, 789–801. [Google Scholar] [CrossRef]
- Larson, L.J.; Schultz, R.D. Three-year duration of immunity in dogs vaccinated with a canarypox-vectored recombinant canine distemper virus vaccine. Vet. Ther. 2007, 8, 101–106. [Google Scholar]
- Filippelli, M.; Lionetti, E.; Gennaro, A.; Lanzafame, A.; Arrigo, T.; Salpietro, C.; La Rosa, M.; Leonardi, S. Hepatitis B vaccine by intradermal route in non responder patients: An update. World J. Gastroenterol. 2014, 20, 10383–10394. [Google Scholar] [CrossRef]
- Lambkin, R.; Novelli, P.; Oxford, J.; Gelder, C. Human genetics and responses to influenza vaccination: Clinical implications. Am. J. Pharm. 2004, 4, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Day, M.J. Immune system development in the dog and cat. J. Comp. Pathol. 2007, 137, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Day, M.J.; Schultz, R.D. Vaccination. In Veterinary Immunology—Principles and Practice, 2nd ed.; Day, M.J., Schultz, R.D., Eds.; CRC Press: Boca Raton, FL, USA; Taylor & Francis Group: Abingdon, UK, 2014; pp. 233–247. [Google Scholar]
- Tizard, I. Dendritic cells and antigen processing. In Veterinary Immunology, 10th ed.; Tizard, I., Ed.; Elsevier Saunders: St. Louis, MI, USA, 2018; pp. 89–99. [Google Scholar]
- Tizard, I. The major histocompatibility complex. In Veterinary Immunology, 10th ed.; Tizard, I., Ed.; Elsevier Saunders: St. Louis, MI, USA, 2018; pp. 100–107. [Google Scholar]
- Kennedy, L.J.; Barnes, A.; Happ, G.M.; Quinnell, R.J.; Courtenay, O.; Carter, S.D.; Ollier, W.E.R.; Thomson, W. Evidence for extensive DLA polymorphism in different dog populations. Tissue Antigens 2002, 60, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Faldyna, M.; Leva, L.; Knotigova, P.; Toman, M. Lymphocyte subsets in peripheral blood of dogs-a flow cytometric study. Vet. Immunol. Immunopathol. 2001, 82, 23–37. [Google Scholar] [CrossRef]
- Kennedy, L.J.; Lunt, M.; Barnes, A.; McElhinney, L.; Fooks, A.R.; Baxter, D.N.; Ollier, W.E.R. Factors influencing the antibody response of dogs vaccinated against rabies. Vaccine 2007, 25, 8500–8507. [Google Scholar] [CrossRef]
- Debenham, S.L.; Millington, A.; Kijast, J.; Andersson, L.; Binns, M. Canine leucocyte adhesion deficiency in Irish Red and White Setters. J. Small Anim. Pract. 2002, 43, 74–75. [Google Scholar] [CrossRef] [PubMed]
- Foureman, P.; Whiteley, M.; Giger, U. Canine leukocyte adhesion deficiency: Presence of the Cys36Ser beta-2 integrin mutation in an affected US Irish Setter cross-breed dog and in US Irish Red and White Setters. J. Vet. Intern. Med. 2002, 16, 518–523. [Google Scholar] [CrossRef]
- Kijas, J.M.; Bauer, T.R., Jr.; Gafvert, S.; Marklund, S.; Trowald-Wigh, G.; Johannisson, A.; Hedhammar, A.; Binns, M.; Juneja, R.K.; Hickstein, D.D.; et al. A missense mutation in the beta-2 integrin gene (ITGB2) causes canine leukocyte adhesion deficiency. Genomics 1999, 61, 101–107. [Google Scholar] [CrossRef]
- Pfeiffer, I.; Brenig, B. Frequency of the canine leucocyte adhesion deficiency (CLAD) mutation among Irish Red Setters in Germany. J. Anim. Breed. Genet. 2005, 122, 140–142. [Google Scholar] [CrossRef]
- Gerber, J.D.; Marron, A.E. Cell-mediated immunity and age at vaccination associated with measles inoculation and protection of dogs against canine distemper. Am. J. Vet. Res. 1976, 37, 133–138. [Google Scholar] [PubMed]
- Rikula, U.; Nuotio, L.; Sihvonen, L. Canine distemper virus neutralising antibodies in vaccinated dogs. Vet. Rec. 2000, 147, 598–603. [Google Scholar] [CrossRef] [PubMed]
- Schoder, D.; Benetka, V.; Sommerfeld-Stur, I.; Kopf, N.; Weissenbacher, E.; Pallan, C.; Walk, K.; Möstl, K. Untersuchungen zum Antikörper-Status gegen Hundestaupe-Virus und canines Parvovirus-2 bei Hunden in Niederösterreich und Wien nach unterschiedlichen Impfintervallen. Vet. Med. Austria/Wien. Tierärztl. Mschr. 2006, 93, 176–182. [Google Scholar]
- Olson, P.; Klingeborn, B.; Hedhammar, A. Serum antibody response to canine parvovirus, canine adenovirus-1, and canine distemper virus in dogs with known status of immunization: Study of dogs in Sweden. Am. J. Vet. Res. 1988, 49, 1460–1466. [Google Scholar]
- Lechner, E.S.; Crawford, P.C.; Levy, J.K.; Edinboro, C.H.; Dubovi, E.J.; Caligiuri, R. Prevalence of protective antibody titers for canine distemper virus and canine parvovirus in dogs entering a Florida animal shelter. J. Am. Vet. Med. Assoc. 2010, 236, 1317–1321. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, M.; Namikawa, K.; Maruo, T.; Orito, K.; Lynch, J.; Sahara, H. Antibody titers for canine parvovirus type-2, canine distemper virus, and canine adenovirus type-1 in adult household dogs. Can. Vet. J. 2011, 52, 983–986. [Google Scholar] [PubMed]
- Ottiger, H.P.; Neimeier-Forster, M.; Stark, K.D.; Duchow, K.; Bruckner, L. Serological responses of adult dogs to revaccination against distemper, parvovirus and rabies. Vet. Rec. 2006, 159, 7–12. [Google Scholar] [CrossRef]
- Pawelec, G.; Larbi, A. Immunity and ageing in man: Annual Review 2006/2007. Exp. Gerontol. 2008, 43, 34–38. [Google Scholar] [CrossRef]
- Mansfield, K.L.; Burr, P.D.; Snodgrass, D.R.; Sayers, R.; Fooks, A.R. Factors affecting the serological response of dogs and cats to rabies vaccination. Vet. Rec. 2004, 154, 423–426. [Google Scholar] [CrossRef]
| Variable | Category | Number of Dogs Tested | Dogs with Pre- Vaccination Antibodies Against CDV 1 | Univariate Analysis | ||
|---|---|---|---|---|---|---|
| P2 | Odds Ratio | 95% CI 3 | ||||
| Age | <2 years | 7/97 | 5/7 | 0.018 | 26.8 | 1.2–1763.4 |
| 2 ≤ 9 years | 76/97 | 75/76 | Ref.value 4 | n. a. 5 | n. a. 5 | |
| >9 years | 14/97 | 12/14 | 0.062 | 11.9 | 0.6–742.6 | |
| Sex | Female | 57/97 | 55/57 | 0.401 | - | - |
| Male | 40/97 | 37/40 | ||||
| Weight | <10 kg | 16/97 | 15/16 | 0.511 | - | - |
| 10–20 kg | 23/97 | 23/23 | ||||
| 20–30 kg | 31/97 | 28/31 | ||||
| >30 kg | 27/97 | 26/27 | ||||
| Neutering status | Intact | 47/97 | 44/47 | 0.671 | - | - |
| Neutered | 50/97 | 48/50 | ||||
| Origin | Breeder | 33/97 | 32/33 | 0.641 | - | - |
| Private | 23/97 | 21/23 | ||||
| Shelter | 13/97 | 12/13 | ||||
| Humane society | 28/97 | 27/28 | ||||
| Environment | Urban | 56/97 | 54/56 | 0.647 | - | - |
| Rural | 41/97 | 38/41 | ||||
| Lifestyle | Family | 70/97 | 66/70 | 1.000 | - | - |
| Breeding | 9/97 | 9/9 | ||||
| Farm | 4/97 | 4/4 | ||||
| Utility | 14/97 | 13/14 | ||||
| History abroad | Yes | 65/97 | 62/65 | 1.000 | - | - |
| No | 32/97 | 30/32 | ||||
| Housing conditions | Other dogs/cats | 62/97 | 57/62 | 0.156 | - | - |
| No other dogs/cats | 35/97 | 35/35 | ||||
| Daily contact with other dogs | <2 | 20/97 | 20/20 | 0.079 | - | - |
| 3–5 | 58/97 | 56/58 | ||||
| >5 | 19/97 | 16/19 | ||||
| Time since last vaccination | 1–3 years | 77/97 | 73/77 | 0.203 | - | - |
| 3–4 years | 11/97 | 11/11 | ||||
| 4–5 years | 4/97 | 4/4 | ||||
| 5–7 years | 3/97 | 3/3 | ||||
| >7 years | 2/97 | 1/2 | ||||
| Complete vaccination series | Yes | 19/97 | 18/19 | 1.000 | - | - |
| No | 78/97 | 74/78 | ||||
| Nucleotide Mismatch/Percent Identity; Amino Acid Mismatch/Percent Identity | ||
|---|---|---|
| CDV Onderstepoort (Hannover) | CDV Onderstepoort (AF378705.1) | |
| CDV AG219 (Munich) | 9/99.404; 6/98.927 | 7/99.585; 3/99.466 |
| CDV Lederle (DQ903854.1) | 4/99.702; 3/99.463 | 11/99.394; 6/99.007 |
| Serum | Dog | Sample | VN isolate Ag219 | VN isolate Onderstepoort |
|---|---|---|---|---|
| 1 | 1 | day 0 | 20 | 20 |
| 2 | 1 | day 7 | 20 | 40 |
| 3 | 1 | day 28 | 40 | 40 |
| 4 | 2 | day 0 | 40 | 20 |
| 5 | 2 | day 7 | 80 | 40 |
| 6 | 2 | day 28 | 40 | 40 |
| 7 | 3 | day 0 | 160 | 160 |
| 8 | 3 | day 7 | 160 | 160 |
| 9 | 3 | day 28 | 80 | 160 |
| 10 | 4 | day 0 | 80 | 80 |
| 11 | 4 | day 7 | 80 | 80 |
| 12 | 4 | day 28 | 80 | 40 |
| 13 | 5 | day 0 | 40 | 80 |
| 14 | 5 | day 7 | 80 | 80 |
| 15 | 5 | day 28 | 80 | 80 |
| 16 | 6 | day 0 | 40 | 80 |
| 17 | 6 | day 7 | 80 | 80 |
| 18 | 6 | day 28 | 80 | 160 |
| 19 | 7 | day 0 | 10 | 20 |
| 20 | 7 | day 7 | 10 | 20 |
| 21 | 7 | day 28 | 10 | 10 |
| 22 | 8 | day 0 | 40 | 80 |
| 23 | 8 | day 7 | 40 | 80 |
| 24 | 8 | day 28 | 40 | 80 |
| 25 | 9 | day 0 | 40 | 20 |
| 26 | 9 | day 7 | 40 | 20 |
| 27 | 9 | day 28 | 40 | 40 |
| Dog | Signalment | Weight | Origin, Lifestyle | Environment | Daily Contact to Other Dogs | Previous Vaccinations | Time since Last Vaccination | Complete Vaccination Series * | CDV 1 Antibody Titer | VAAEs 2 after Vaccination | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Day 0 | Day 7 | Day 28 | ||||||||||
| 1 | Miniature Pinscher, 2 y 3, female, intact | 4 kg 4 | Private, family dog | Urban | >5 | 7 w 5 15 w | 1 y, 7 m 6 | No | <10 | <10 | <10 | No |
| 2 | Labrador, 7 y, female, neutered | 26 kg | Private, farm dog | Rural | 3–5 | 8 m 5 y 6 y | 1 y | No | <10 | <10 | <10 | Yes Lethargy day 0–7 |
| 3 | Bavarian Mountain Hound, 2 y, male, intact | 22 kg | Breeder, utility dog | Rural | >5 | 9 w 13 w 18 w 1 y | 1 y, 2 m | Yes | <10 | <10 | <10 | Yes Lethargy day 0–7 |
| 4 | Mix, 1.5 y, male neutered | 20 kg | Humane society, family dog | Urban | >5 | 16 w 20 w | 1 y, 2 m | No | <10 | <10 | <10 | No |
| 5 | Mix, 13 y, male neutered | 43 kg | Shelter, family dog | Rural | 3–5 | 5 m 6 m | 12 y | No | <10 | <10 | <10 | Yes Local reactions day 7–28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bergmann, M.; Freisl, M.; Zablotski, Y.; Khan, M.A.A.; Speck, S.; Truyen, U.; Hartmann, K. Prevalence of Neutralizing Antibodies to Canine Distemper Virus and Response to Vaccination in Client-Owned Adult Healthy Dogs. Viruses 2021, 13, 945. https://doi.org/10.3390/v13050945
Bergmann M, Freisl M, Zablotski Y, Khan MAA, Speck S, Truyen U, Hartmann K. Prevalence of Neutralizing Antibodies to Canine Distemper Virus and Response to Vaccination in Client-Owned Adult Healthy Dogs. Viruses. 2021; 13(5):945. https://doi.org/10.3390/v13050945
Chicago/Turabian StyleBergmann, Michèle, Monika Freisl, Yury Zablotski, Md Anik Ashfaq Khan, Stephanie Speck, Uwe Truyen, and Katrin Hartmann. 2021. "Prevalence of Neutralizing Antibodies to Canine Distemper Virus and Response to Vaccination in Client-Owned Adult Healthy Dogs" Viruses 13, no. 5: 945. https://doi.org/10.3390/v13050945
APA StyleBergmann, M., Freisl, M., Zablotski, Y., Khan, M. A. A., Speck, S., Truyen, U., & Hartmann, K. (2021). Prevalence of Neutralizing Antibodies to Canine Distemper Virus and Response to Vaccination in Client-Owned Adult Healthy Dogs. Viruses, 13(5), 945. https://doi.org/10.3390/v13050945







