Optimization and Clinical Evaluation of a Multi-Target Loop-Mediated Isothermal Amplification Assay for the Detection of SARS-CoV-2 in Nasopharyngeal Samples
Abstract
1. Introduction
2. Materials and Methods
2.1. Viral Controls and Gene Screening
2.2. Viral Target Evaluation
2.2.1. RT-LAMP Reaction Setup
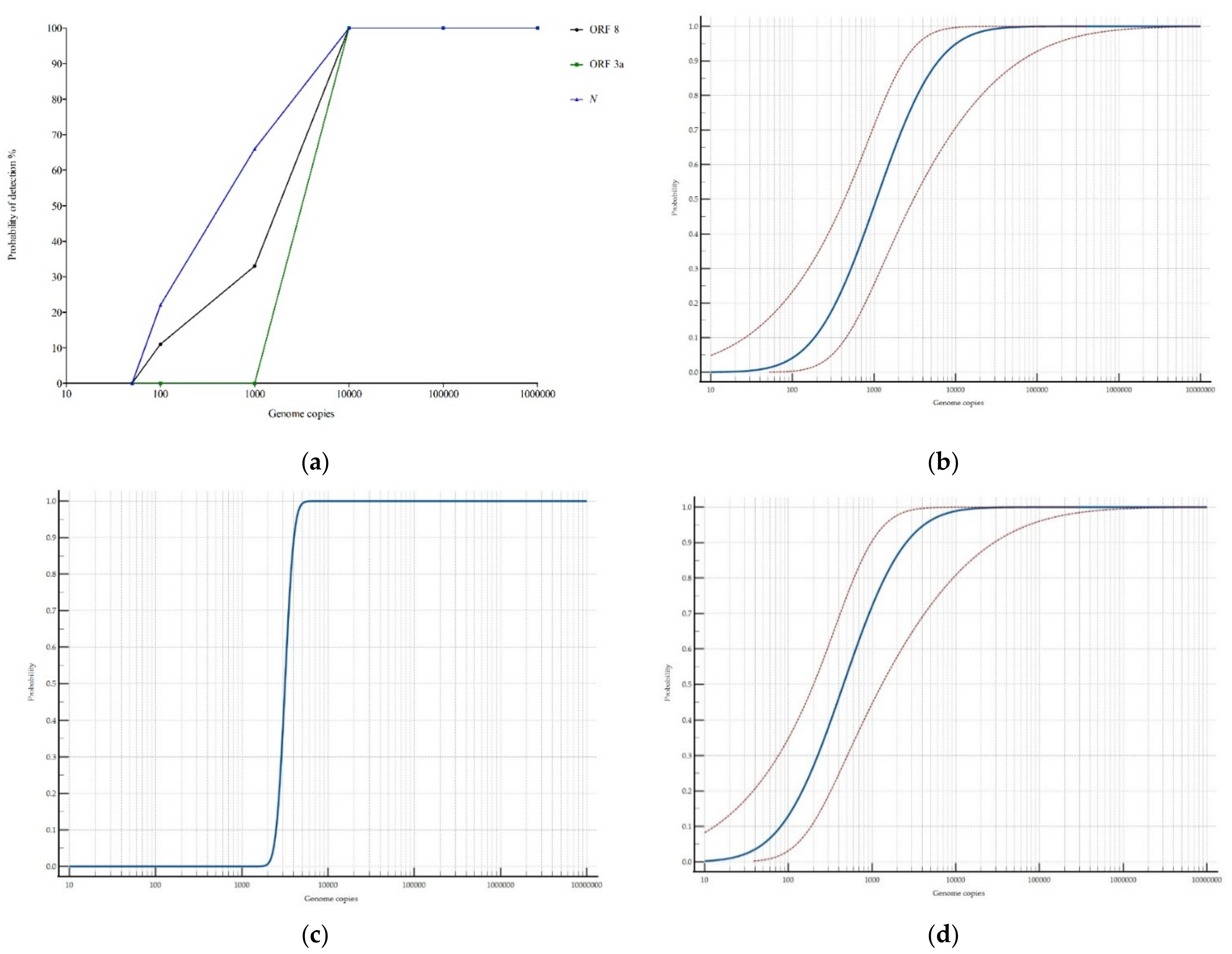
2.2.2. Determination of the Dynamic Range and Limit of Detection
2.3. Clinical Samples Processing and Reference Method
2.4. Evaluation of the Methodology in a Clinical Setting
3. Results and Discussion
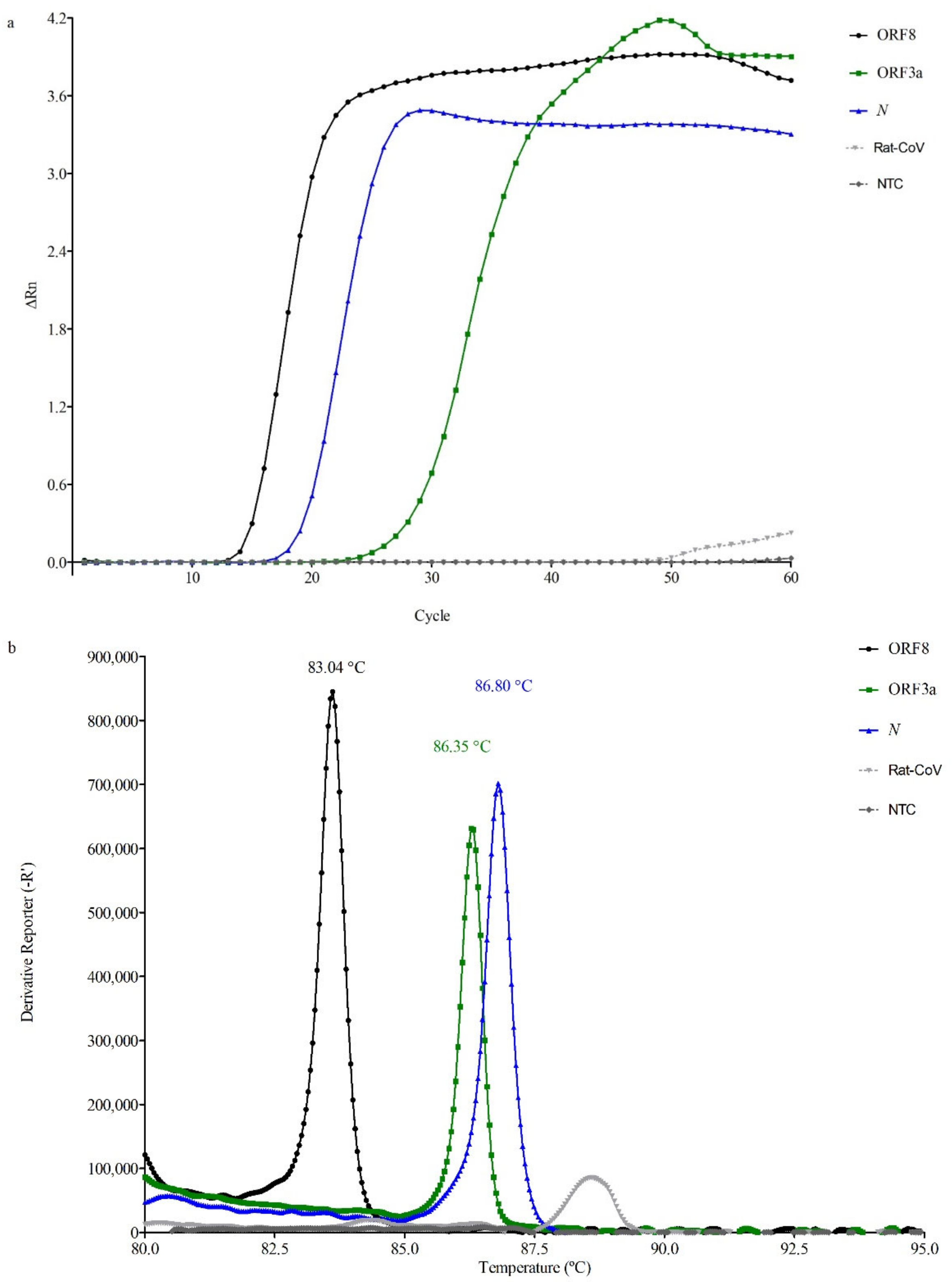
3.1. Genetic Targets Performance
3.1.1. RT-LAMP Reaction Setup
3.1.2. Dynamic Range
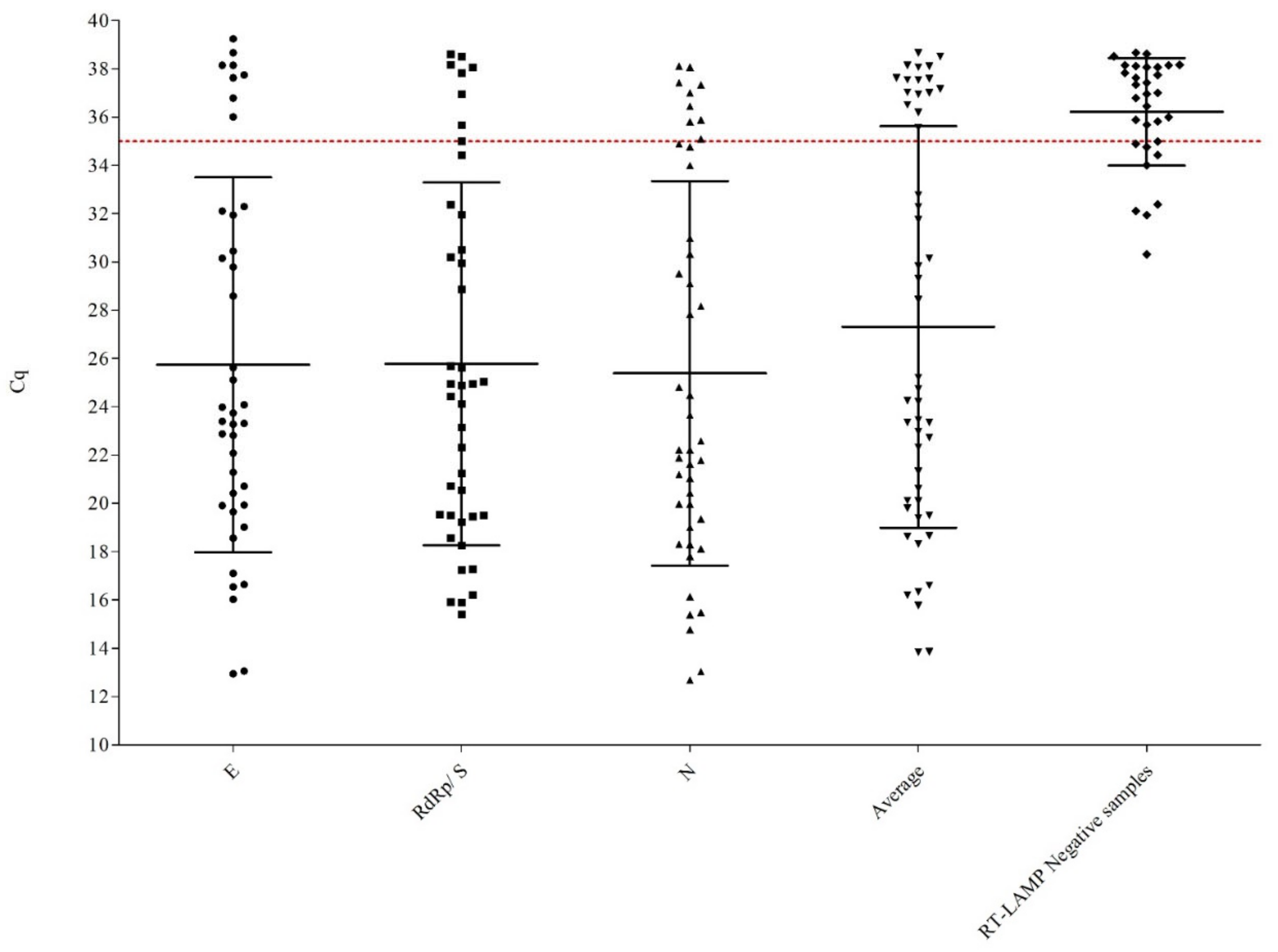
3.2. Clinical Samples Analyzed
3.3. Clinical Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Andersen, K.G.; Rambaut, A.; Lipkin, W.I.; Holmes, E.C.; Garry, R.F. The proximal origin of SARS-CoV-2. Nat. Med. 2020, 26, 450–452. [Google Scholar] [CrossRef]
- Enjuanes, L.; Sola, I.; Zúñiga, S. Position Paper-Enjuanes, Sola & Zúñiga: Novel Human Pathogenic Coronavirus SARS-CoV-2; Digital.CSIC: Madrid, Spain, 2020; pp. 1–5. [Google Scholar]
- Lai, C.C.; Shih, T.P.; Ko, W.C.; Tang, H.J.; Hsueh, P.R. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and coronavirus disease-2019 (COVID-19): The epidemic and the challenges. Int. J. Antimicrob. Agents 2020, 55, 105924. [Google Scholar] [CrossRef] [PubMed]
- Chu, H.; Chan, J.F.-W.; Yuen, T.T.-T.; Shuai, H.; Yuan, S.; Wang, Y.; Hu, B.; Yip, C.C.-Y.; Tsang, J.O.-L.; Huang, X.; et al. Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: An observational study. Lancet Microbe 2020, 1, e14–e23. [Google Scholar] [CrossRef]
- Pasteur, I. Protocol: Real-Time RT-PCR Assays for the Detection of SARS-CoV-2; WHO: Geneva, Switzerland, 2020; pp. 1–3. [Google Scholar]
- Corman, V.; Bleicker, T.; Brünink, S.; Drosten, C.; Landt, O.; Koopmans, M.; Zambon Public Health England, M. Diagnostic Detection of 2019-nCoV by Real-Time RT-RCR incl. Euro Surveill. 2020, 25, 2000045. [Google Scholar] [CrossRef] [PubMed]
- CDC Real-Time RT-PCR Diagnostic Panel for Emergency Use Only; CDC: Atlanta, GA, USA, 2020; Volume 3.
- Huang, W.E.; Lim, B.; Hsu, C.C.; Xiong, D.; Wu, W.; Yu, Y.; Jia, H.; Wang, Y.; Zeng, Y.; Ji, M.; et al. RT-LAMP for rapid diagnosis of coronavirus SARS-CoV-2. Microb. Biotechnol. 2020, 13, 950–961. [Google Scholar] [CrossRef] [PubMed]
- Lamb, L.E.; Bartolone, S.N.; Ward, E.; Chancellor, M.B. Rapid detection of novel coronavirus/Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) by reverse transcription-loop-mediated isothermal amplification. PLoS ONE 2020, 15, 1–15. [Google Scholar] [CrossRef]
- Park, G.-S.; Ku, K.; Beak, S.-H.; Kim, S.-J.; Kim, S.I.; Kim, B.-T.; Maeng, J.-S. Development of Reverse Transcription Loop-mediated Isothermal Amplification (RT-LAMP) Assays Targeting SARS-CoV-2. J. Mol. Diagn. 2020, 1–8. [Google Scholar] [CrossRef]
- Zhang, Y.; Ren, G.; Buss, J.; Barry, A.J.; Patton, G.C.; Tanner, N.A. Enhancing colorimetric loop-mediated isothermal amplification speed and sensitivity with guanidine chloride. Biotechniques 2020, 69, 178–185. [Google Scholar] [CrossRef]
- Lee, J.Y.H.; Best, N.; Mcauley, J.; Porter, J.L.; Seemann, T.; Mark, B.; Sait, M.; Orlando, N.; Mercoulia, K.; Ballard, S.A.; et al. Validation of a single-step, single-tube reverse transcription-loop-mediated isothermal amplification assay for rapid detection of SARS-CoV-2 RNA. J. Med. Microbiol. 2020, 69, 1169. [Google Scholar] [CrossRef]
- Sherrill-Mix, S.; Hwang, Y.; Roche, A.M.; Weiss, S.R.; Li, Y.; Graham-Wooten, J.; Taylor, L.J.; Collman, R.G.; Van Duyne, G.D.; Bushman, F.D. LAMP-BEAC: Detection of SARS-CoV-2 RNA Using RT-LAMP and Molecular Beacons. medRxiv 2020. [Google Scholar] [CrossRef]
- Hu, X.; Deng, Q.; Li, J.; Chen, J.; Wang, Z.; Zhang, X.; Fang, Z.; Li, H.; Zhao, Y.; Yu, P.; et al. Development and Clinical Application of a Rapid and Sensitive Loop-Mediated Isothermal Amplification Test for SARS-CoV-2 Infection. mSphere 2020, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Odiwuor, N.; Xiong, J.; Sun, L.; Nyaruaba, R.O.; Wei, H.; Tanner, N.A. Rapid Molecular Detection of SARS-CoV-2 (COVID-19) Virus RNA Using Colorimetric LAMP. medRxiv 2020, 2. [Google Scholar] [CrossRef]
- Park, G.S.; Ku, K.; Baek, S.H.; Kim, S.J.; Kim, S.I.; Kim, B.T.; Maeng, J.S. Development of Reverse Transcription Loop-Mediated Isothermal Amplification Assays Targeting Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). J. Mol. Diagn. 2020, 22, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Broughton, J.P.; Deng, X.; Yu, G.; Fasching, C.L.; Singh, J.; Streithorst, J.; Granados, A.; Sotomayor-Gonzalez, A.; Zorn, K.; Gopez, A.; et al. Rapid Detection of 2019 Novel Coronavirus SARS-CoV-2 Using a CRISPR-based DETECTR Lateral Flow Assay. medRxiv 2020. [Google Scholar] [CrossRef]
- Schermer, B.; Fabretti, F.; Damagnez, M.; Di Cristanziano, V.; Heger, E.; Arjune, S.; Tanner, N.A.; Imhof, T.; Koch, M.; Ladha, A.; et al. Rapid SARS-CoV-2 testing in primary material based on a novel multiplex LAMP assay. medRxiv 2020. [Google Scholar] [CrossRef]
- Joung, J.; Ladha, A.; Saito, M.; Segel, M.; Bruneau, R.; Huang, M.-L.W.; Kim, N.-G.; Yu, X.; Li, J.; Walker, B.D.; et al. Point-of-care testing for COVID-19 using SHERLOCK diagnostics. medRxiv Prepr. Serv. Health Sci. 2020. [Google Scholar] [CrossRef]
- Patchsung, M.; Jantarug, K.; Pattama, A.; Aphicho, K.; Suraritdechachai, S.; Meesawat, P.; Sappakhaw, K.; Leelahakorn, N.; Ruenkam, T.; Wongsatit, T.; et al. Clinical validation of a Cas13-based assay for the detection of SARS-CoV-2 RNA. Nat. Biomed. Eng. 2020. [Google Scholar] [CrossRef]
- Myhrvold, C.; Freije, C.A.; Gootenberg, J.S.; Abudayyeh, O.O.; Metsky, H.C.; Durbin, A.F.; Kellner, M.J.; Tan, A.L.; Paul, L.M.; Parham, L.A.; et al. Field-deployable viral diagnostics using CRISPR-Cas13. Science 2018, 360, 444–448. [Google Scholar] [CrossRef]
- Ali, Z.; Aman, R.; Mahas, A.; Rao, G.S.; Tehseen, M.; Marsic, T.; Salunke, R.; Subudhi, A.K.; Hala, S.M.; Hamdan, S.M.; et al. iSCAN: An RT-LAMP-coupled CRISPR-Cas12 module for rapid, sensitive detection of SARS-CoV-2. Virus Res. 2020, 288, 198129. [Google Scholar] [CrossRef]
- Mautner, L.; Baillie, C.K.; Herold, H.M.; Volkwein, W.; Guertler, P.; Eberle, U.; Ackermann, N.; Sing, A.; Pavlovic, M.; Goerlich, O.; et al. Rapid point-of-care detection of SARS-CoV-2 using reverse transcription loop-mediated isothermal amplification (RT-LAMP). Virol. J. 2020, 17, 1–14. [Google Scholar] [CrossRef]
- Curtis, K.A.; Morrison, D.; Rudolph, D.L.; Shankar, A.; Bloomfield, L.S.P.; Switzer, W.M.; Owen, S.M. A multiplexed RT-LAMP assay for detection of group M HIV-1 in plasma or whole blood. J. Virol. Methods 2018, 255, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, K.; Mage, P.L.; Csordas, A.T.; Eisenstein, M.; Soh, H.T. Simultaneous elimination of carryover contamination and detection of DNA with uracil-DNA-glycosylase-supplemented loop-mediated isothermal amplification (UDG-LAMP). Chem. Commun. 2014, 50, 3747–3749. [Google Scholar] [CrossRef]
- Bao, Y.; Jiang, Y.; Xiong, E.; Tian, T.; Zhang, Z.; Lv, J.; Li, Y.; Zhou, X. CUT-LAMP: Contamination-Free Loop-Mediated Isothermal Amplification Based on the CRISPR/Cas9 Cleavage. ACS Sensors 2020, 5, 1082–1091. [Google Scholar] [CrossRef]
- Karthik, K.; Rathore, R.; Thomas, P.; Arun, T.R.; Viswas, K.N.; Dhama, K.; Agarwal, R.K. New closed tube loop mediated isothermal amplification assay for prevention of product cross-contamination. MethodsX 2014, 1, e137–e143. [Google Scholar] [CrossRef] [PubMed]
- Tomas, D.; Rodrigo, A.; Hernandez, M.; Ferrus, M.A. Validation of Real-Time PCR and Enzyme-Linked Fluorescent Assay-Based Methods for Detection of Salmonella spp. in Chicken Feces Samples. Food Anal. Methods 2009, 2, 180–189. [Google Scholar] [CrossRef]
- Garrido, A.; Chapela, M.J.; Román, B.; Fajardo, P.; Lago, J.; Vieites, J.M.; Cabado, A.G. A new multiplex real-time PCR developed method for Salmonella spp. and Listeria monocytogenes detection in food and environmental samples. Food Control 2013, 30, 76–85. [Google Scholar] [CrossRef]
- Davies, N.G.; Abbott, S.; Barnard, R.C.; Jarvis, C.I.; Kucharski, A.J.; Munday, J.D.; Pearson, C.A.B.; Russell, T.W.; Tully, D.C.; Washburne, A.D.; et al. Estimated transmissibility and impact of SARS-CoV-2 lineage B.1.1.7 in England. Science 2021, 3055, 1–16. [Google Scholar] [CrossRef]
- WHO. Statement of the WHO Working Group on COVID-19 Animal Models (WHO-COM) about the UK and South African SARS-CoV-2 New Variants; WHO: Geneva, Switzerland, 2020; Volume 21. [Google Scholar]
- Jang, W.S.; Lim, D.H.; Yoon, J.; Kim, A.; Lim, M.; Nam, J.; Yanagihara, R.; Ryu, S.W.; Jung, B.K.; Ryoo, N.H.; et al. Development of a multiplex Loop-Mediated Isothermal Amplification (LAMP) assay for onsite diagnosis of SARS CoV-2. PLoS ONE 2021, 16, 1–14. [Google Scholar] [CrossRef]
- Danilla, S.; Andrades, P.; Gomez, M.E.; Chamorro, M.; Leniz, P.; Pineros, J.L.; Llanos, S.; Roco, H.; Correa, G.; Pasten, J.A.; et al. Concordance between qualitative and quantitative cultures in burned patients: Analysis of 2886 cultures. Burns 2005, 31, 967–971. [Google Scholar] [CrossRef] [PubMed]
- Rödel, J.; Egerer, R.; Suleyman, A.; Sommer-Schmid, B.; Baier, M.; Henke, A.; Edel, B.; Löffler, B. Use of the variplexTM SARS-CoV-2 RT-LAMP as a rapid molecular assay to complement RT-PCR for COVID-19 diagnosis. J. Clin. Virol. 2020, 132. [Google Scholar] [CrossRef]
- L’Helgouach, N.; Champigneux, P.; Santos-Schneider, F.; Molina, L.; Espeut, J.; Alali, M.; Baptiste, J.; Cardeur, L.; Dubuc, B.; Foulongne, V.; et al. EasyCOV: LAMP based rapid detection of SARS-CoV-2 in saliva. medRxiv 2020. [Google Scholar] [CrossRef]
- Tom, M.R.; Mina, M.J. To Interpret the SARS-CoV-2 Test, Consider the Cycle Threshold Value. Clin. Infect. Dis. 2020, 02115, 1–3. [Google Scholar] [CrossRef]
- Basu, A.; Zinger, T.; Inglima, K.; Woo, K.M.; Atie, O.; Yurasits, L.; See, B.; Aguero-Rosenfeld, M.E. Performance of abbott id now covid-19 rapid nucleic acid amplification test using nasopharyngeal swabs transported in viral transport media and dry nasal swabs in a New York city academic institution. J. Clin. Microbiol. 2020, 58, 1–19. [Google Scholar] [CrossRef]
- Mina, M.; Miller, S.; Quigley, M.; Prentiss, T.; McKinnon, J.E.; Comer, S.; McKinnon, E.J.; Comer, S. Analyzing Conflicting Results in Rapid Point-of-Care COVID-19 Testing; Office for Scholarly Communication: Cambridge, MA, USA, 2020; p. 16. [Google Scholar]
- Larremore, D.B.; Wilder, B.; Lester, E.; Shehata, S.; Burke, J.M.; Hay, J.A.; Tambe, M.; Mina, M.J.; Parker, R. Test sensitivity is secondary to frequency and turnaround time for COVID-19 surveillance. medRxiv 2020. [Google Scholar] [CrossRef]
- Mina, M.J.; Parker, R.; Larremore, D.B. Rethinking Covid-19 Test Sensitivity—A Strategy for Containment. N. Engl. J. Med. 2020, 1969–1973. [Google Scholar] [CrossRef] [PubMed]
- Nagura-Ikeda, M.; Imai, K.; Tabata, S.; Miyoshi, K.; Murahara, N.; Mizuno, T. Clinical Evaluation of Self-Collected Saliva by Quantitative. J. Clin. Microbiol. 2020, 58, 1–9. [Google Scholar]
- Francois, P.; Tangomo, M.; Hibbs, J.; Bonetti, E.J.; Boehme, C.C.; Notomi, T.; Perkins, M.D.; Schrenzel, J. Robustness of a loop-mediated isothermal amplification reaction for diagnostic applications. FEMS Immunol. Med. Microbiol. 2011, 62, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Chavarria-Miró, G.; Anfruns-Estrada, E.; Guix, S.; Paraira, M.; Galofré, B.; Sáanchez, G.; Pintó, R.; Bosch, A. Sentinel surveillance of SARS-CoV-2 in wastewater anticipates the occurrence of COVID-19 cases. medRxiv 2020. [Google Scholar] [CrossRef]
- Yeo, C.; Kaushal, S.; Yeo, D. Enteric involvement of coronaviruses: Is faecal–oral transmission of SARS-CoV-2 possible? Lancet Gastroenterol. Hepatol. 2020, 5, 335–337. [Google Scholar] [CrossRef]
- Foladori, P.; Cutrupi, F.; Segata, N.; Manara, S.; Pinto, F.; Malpei, F.; Bruni, L.; La Rosa, G. SARS-CoV-2 from faeces to wastewater treatment: What do we know? A review. Sci. Total Environ. 2020, 743, 140444. [Google Scholar] [CrossRef]

| Target | Primer | Sequence (5′->3′) | Reference |
|---|---|---|---|
| ORF8 | ORF8-FIP | AGG ACA CGG GTC ATC AAC TAC AAG CTG CAT TTC ACC AAGAA | [23] |
| ORF8-BIP | AGG AGC TAG AAA ATC AGC ACC TAT GGG TGA TTT AGA ACC AGC | ||
| ORF8-F3 | ACT TGT CAC GCC TAA ACG | ||
| ORF8-B3 | CTA CCC AAT TTA GGT TCC TGG | ||
| ORF8-LF | TGG TTG ATG TTG AGT ACA TGAC | ||
| ORF8-LB | AAT TGA ATT GTG CGT GGA TGAG | ||
| ORF3a | GMP-ORF3a-FIP | GAA GCG CTCT GAA AAA CAG CAA GAA G-CCT CAC TCC CTT TCG GAT | This study |
| GMP-ORF3a-BIP | CTA GCA CTC TCC AAG GGT GTT CAC-GAG CAA AAG GTG TGA GTA AAC TG | ||
| GMP-ORF3a-F3 | CGC GCT ACT GCA ACG ATA C | ||
| GMP-ORF3a-B3 | TTC AAG GCC AGC AGC AAC | ||
| GMP-ORF3a-LF | GTG CAA CGC CAA CAA TAA GCC | ||
| N | GMP-N-FIP | AGA CGG CAT CAT ATG GGT TGC A tttt GCG GGT GCC AAT GTG ATC | This study |
| GMP-N-BIP | TCT GGC CCA GTT CCT AGG TAG T tttt GAC GAA TTC GTG GTG GTG AC | ||
| GMP-N-F3 | ATT GGC TAC TAC CGA AGA GCT | ||
| GMP-N-B3 | AGG AAG TTG TAG CAC GAT TGC | ||
| GMP-N-LF | TAC CAT CTT GGA CTG AGA TCT TTC A | ||
| GMP-N-LB | ACT GAG GGA GCC TTG AAT ACA CCA | ||
| RNaseP | FIP | GTG TGA CCC TGA AGA CTC GGT TTT AGC CAC TGA CTC GGA TC | [24] |
| BIP | CCT CCG TGA TAT GGC TCT TCG TTT TTT TCT TAC ATG GCT CTG GTC | ||
| F3 | TTG ATG AGC TGG AGC CA | ||
| B3 | CAC CCT CAA TGC AGA GTC | ||
| LF | ATG TGG ATG GCT GAG TTG TT | ||
| LB | CAT GCT GAG TAC TGG ACC TC |
| RT–qPCR Limit * | PA | NA | PD | ND | SE (95% CI) | SP (95% CI) | AC (95% CI) | PPV (95% CI) | NPV (95% CI) | k |
|---|---|---|---|---|---|---|---|---|---|---|
| RT–LAMP | 31 | 102 | 1 | 18 | 63.3 (48.3–76.6) | 99.0 (94.7–100) | 87.5 (81.2–82.3) | 96.9 (83.8–99.9) | 85.0 (77.3–90.9) | 0.69 |
| RT–LAMP Cq <20 | 11 | 102 | 1 | 1 | 91.7 (61.5–99.8) | 99.0 (94.7–100) | 98.3 (95.9–100) | 91.7 (61.5–99.8) | 99.0 (94.7–100) | 0.91 |
| RT–LAMP Cq <25 | 24 | 102 | 1 | 1 | 96.0 (79.6–99.9) | 99.0 (94.7–100) | 98.4 (94.5–99.8) | 96.0 (79.6–99.9) | 99.0 (94.7–100) | 0.95 |
| RT–LAMP Cq <30 | 28 | 102 | 1 | 1 | 96.6 (82.2–99.9) | 99.0 (94.7–100) | 98.5 (94.6–99.8) | 96.6 (82.2–99.9) | 99.0 (94.7–100) | 0.96 |
| RT–LAMP Cq <35 ** | 30 | 102 | 1 | 3 | 90.9 (75.7–98.1) | 99.0 (94.7–100) | 97.1 (92.6–99.2) | 96.8 (83.3–99.9) | 97.1 (91.9–99.4) | 0.92 |
| Cq (Number of Samples) | ORF8 n (%) | ORF3a n (%) | N n (%) |
|---|---|---|---|
| <20 (n = 12) | 10 (83.3%) * | 10 (83.3%)* | 11 (91.7%)* |
| 20–25 (n = 13) | 12 (92.3%) | 11 (84.6%) | 13 (100%) |
| 25–30 (n = 4) | 3 (75%) | 0 (0%) | 4 (100%) |
| 30–35 (n = 16) | 1 (6.25%) | 0 (0%) | 2 (12.5%) |
| >35 (n = 16) | 0 (0%) | 0 (0%) | 1 (6.25%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roumani, F.; Azinheiro, S.; Sousa, H.; Sousa, A.; Timóteo, M.; Varandas, T.; Fonseca-Silva, D.; Baldaque, I.; Carvalho, J.; Prado, M.; et al. Optimization and Clinical Evaluation of a Multi-Target Loop-Mediated Isothermal Amplification Assay for the Detection of SARS-CoV-2 in Nasopharyngeal Samples. Viruses 2021, 13, 940. https://doi.org/10.3390/v13050940
Roumani F, Azinheiro S, Sousa H, Sousa A, Timóteo M, Varandas T, Fonseca-Silva D, Baldaque I, Carvalho J, Prado M, et al. Optimization and Clinical Evaluation of a Multi-Target Loop-Mediated Isothermal Amplification Assay for the Detection of SARS-CoV-2 in Nasopharyngeal Samples. Viruses. 2021; 13(5):940. https://doi.org/10.3390/v13050940
Chicago/Turabian StyleRoumani, Foteini, Sarah Azinheiro, Hugo Sousa, Ana Sousa, Mafalda Timóteo, Tatiana Varandas, Daniela Fonseca-Silva, Inês Baldaque, Joana Carvalho, Marta Prado, and et al. 2021. "Optimization and Clinical Evaluation of a Multi-Target Loop-Mediated Isothermal Amplification Assay for the Detection of SARS-CoV-2 in Nasopharyngeal Samples" Viruses 13, no. 5: 940. https://doi.org/10.3390/v13050940
APA StyleRoumani, F., Azinheiro, S., Sousa, H., Sousa, A., Timóteo, M., Varandas, T., Fonseca-Silva, D., Baldaque, I., Carvalho, J., Prado, M., & Garrido-Maestu, A. (2021). Optimization and Clinical Evaluation of a Multi-Target Loop-Mediated Isothermal Amplification Assay for the Detection of SARS-CoV-2 in Nasopharyngeal Samples. Viruses, 13(5), 940. https://doi.org/10.3390/v13050940







