Viruses with U-DNA: New Avenues for Biotechnology
Abstract
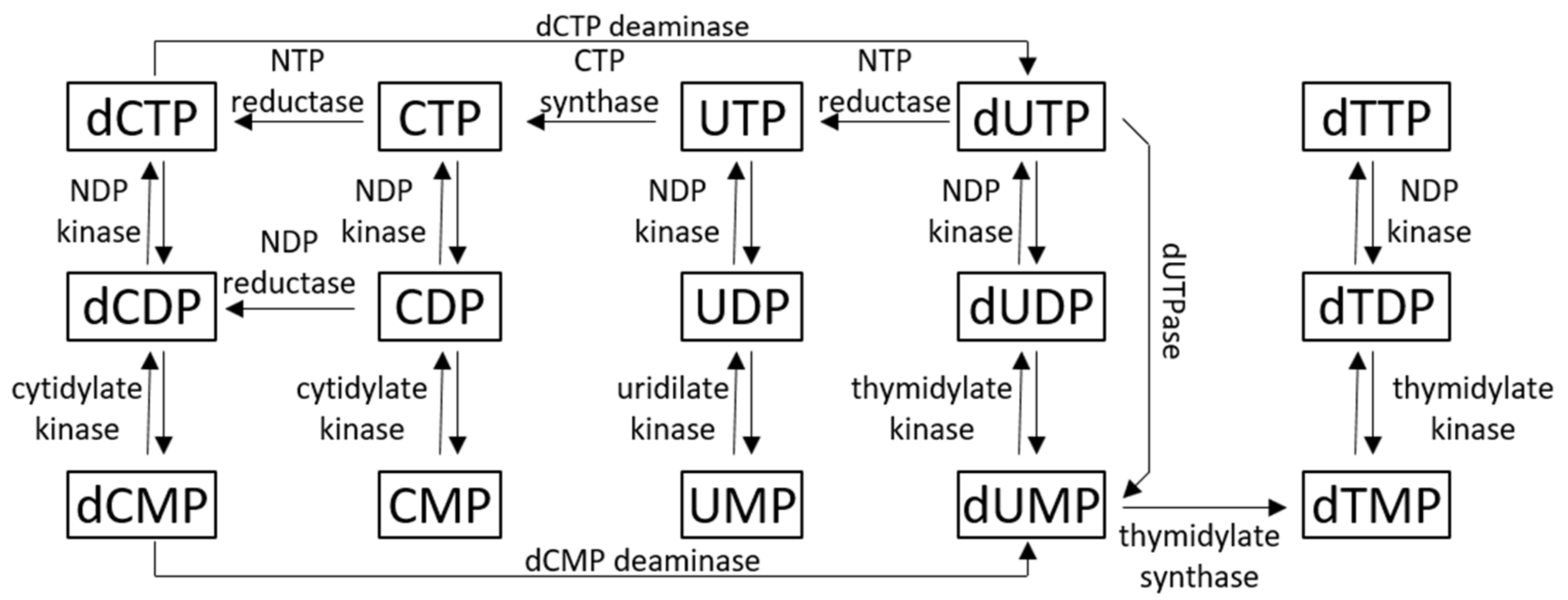
1. Uracil-DNA Metabolism
1.1. Families of Uracil-DNA Glycosylases
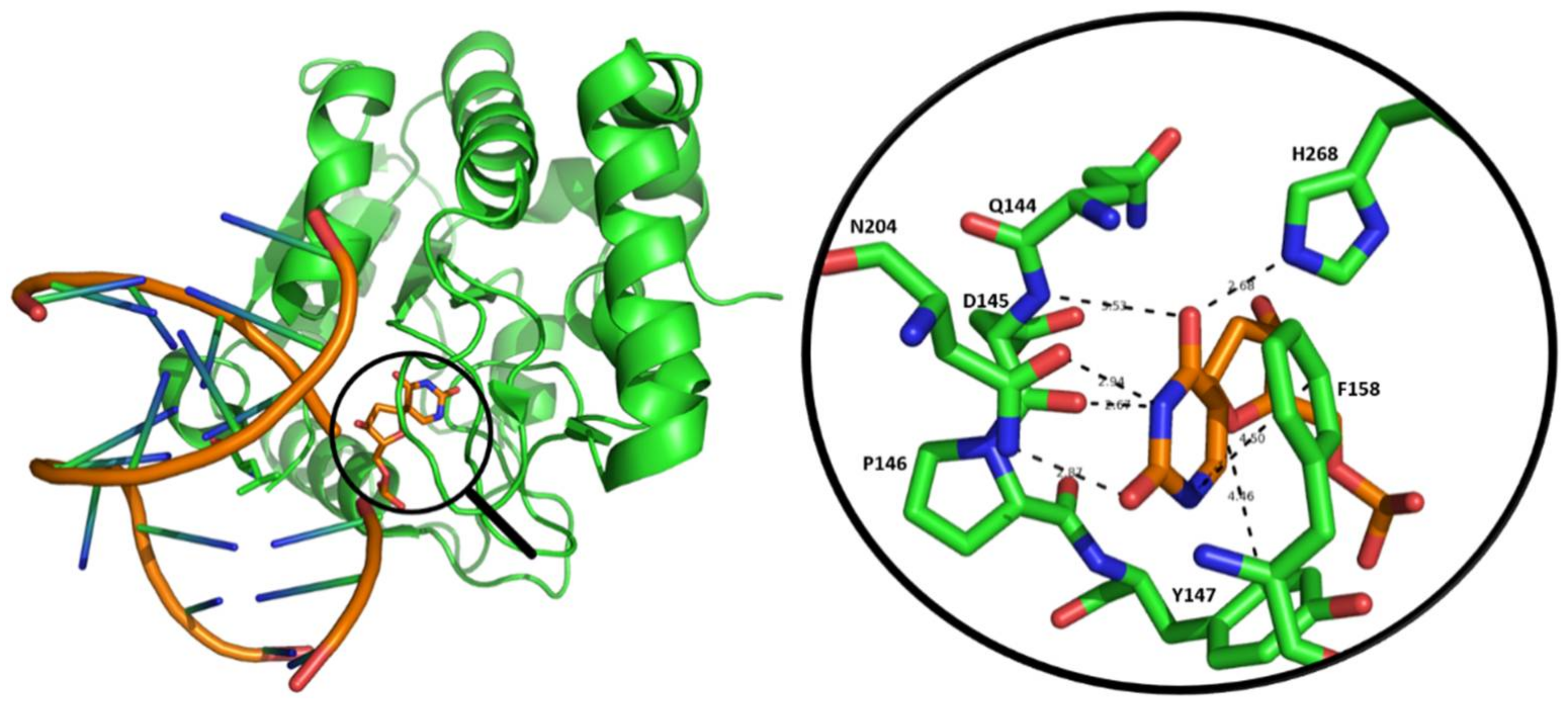
1.2. Diverse Roles for Uracil in DNA
2. Intriguing Bacteriophages with Uracil in DNA
3. UNG Inhibitors and Their Potential Biotechnological Applications
3.1. Small Inhibitory Molecules
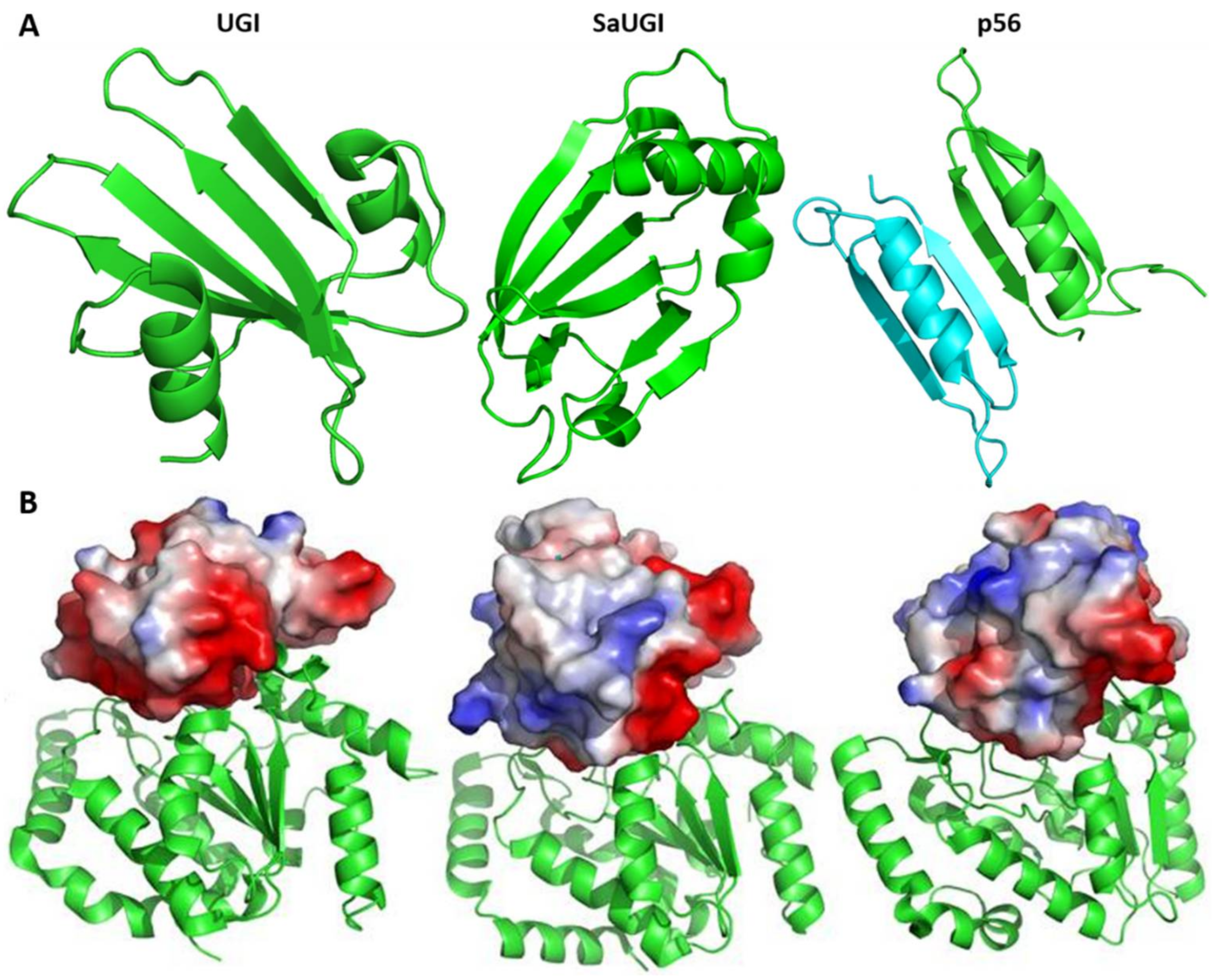
3.2. Protein Inhibitors of Phage Origin
- (1)
- Recently, CRISPR-driven genome engineering has become the gold standard for not only gene destruction, but also for introducing site specific mutations into highly complex genomes. Such CRISPR-based gene editing application required two major modification in the Cas9 nuclease. On the one hand, the nuclease activity was destroyed creating a Cas9 variant that is still capable of site-specific DNA binding but does not cleave the DMS strand. On the other hand, a cytidine deaminase domain (originally present in, e.g., AID) was fused to the catalytically inactive Cas9 protein (Cas9D10A). In this fused Cas9D10A construct, the deaminase activity will get constrained to the cytosine base(s) present within the sequence recognition window on the DNA strand. Site specific cytosine deamination leads to a point mutation (C to U, followed by U to T in the next replication cycle). However, this clever system necessarily requires abrogation of uracil directed repair, which is constituted by a further modification of the Cas9D10A-deaminase molecular tool. Namely, the uracil DNA glycosylase inhibitor (UGI) is linked to the C-terminus of the Cas9D10A-deaminase complex [59].
- (2)
- The appearance of uracil in the DNA, as reviewed above, may report on activation of cytosine-deaminases or spontaneous events of deamination, as well as on dNTP pool perturbations. In all these circumstances the primary effect, i.e., appearance of uracil, is only temporary. Through the highly efficient uracil directed repair, the transient uracil mark is quickly erased, impeding analysis of uracil patterns. To visualize nascent uracils is an important task in order to gain mechanistic insights into the different cellular processes involved in uracil-DNA metabolism. The application of the UNG inhibitor UGI, or other inhibitors with this function is indispensable to decipher uracil patterns [3,60].
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Fadda, E.; Pomès, R. On the molecular basis of uracil recognition in DNA: Comparative study of T-A versus U-A structure, dynamics and open base pair kinetics. Nucleic Acids Res. 2011, 39, 767–780. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Gizzi, A.; Stivers, J.T. Deoxyuridine in DNA has an inhibitory and promutagenic effect on RNA transcription by diverse RNA polymerases. Nucleic Acids Res. 2019, 47, 4153–4168. [Google Scholar] [CrossRef] [PubMed]
- Róna, G.; Marfori, M.; Borsos, M.; Scheer, I.; Takács, E.; Tóth, J.; Babos, F.; Magyar, A.; Erdei, A.; Bozóky, Z.; et al. Phosphorylation adjacent to the nuclear localization signal of human dUTPase abolishes nuclear import: Structural and mechanistic insights. Acta Crystallogr. Sect. D Biol. Crystallogr. 2013, 69, 2495–2505. [Google Scholar] [CrossRef] [PubMed]
- Róna, G.; Scheer, I.; Nagy, K.; Pálinkás, H.L.; Tihanyi, G.; Borsos, M.; Békési, A.; Vértessy, B.G. Detection of uracil within DNA using a sensitive labeling method for in vitro and cellular applications. Nucleic Acids Res. 2016, 44, e28. [Google Scholar] [CrossRef]
- Schormann, N.; Ricciardi, R.; Chattopadhyay, D. Uracil-DNA glycosylases-structural and functional perspectives on an essential family of DNA repair enzymes. Protein Sci. 2014, 23, 1667–1685. [Google Scholar] [CrossRef]
- Otterlei, M.; Warbrick, E.; Nagelhus, T.A.; Haug, T.; Slupphaug, G.; Akbari, M.; Aas, P.A.; Steinsbekk, K.; Bakke, O.; Krokan, H.E. Post-replicative base excision repair in replication foci. EMBO J. 1999, 18, 3834–3844. [Google Scholar] [CrossRef]
- Krokan, H.E.; Bjørås, M. Base excision repair. Cold Spring Harb. Perspect. Biol. 2013, 5, a012583. [Google Scholar] [CrossRef]
- Meyers, G.; Ng, Y.-S.; Bannock, J.M.; Lavoie, A.; Walter, J.E.; Notarangelo, L.D.; Kilic, S.S.; Aksu, G.; Debré, M.; Rieux-Laucat, F.; et al. Activation-induced cytidine deaminase (AID) is required for B-cell tolerance in humans. Proc. Natl. Acad. Sci. USA 2011, 108, 11554–11559. [Google Scholar] [CrossRef]
- Muramatsu, M.; Kinoshita, K.; Fagarasan, S.; Yamada, S.; Shinkai, Y.; Honjo, T. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 2000, 102, 553–563. [Google Scholar] [CrossRef]
- Sire, J.; Quérat, G.; Esnault, C.; Priet, S. Uracil within DNA: An actor of antiviral immunity. Retrovirology 2008, 5, 45. [Google Scholar] [CrossRef]
- Priet, S.; Sire, J.; Quérat, G. Uracils as a cellular weapon against viruses and mechanisms of viral escape. Curr. HIV Res. 2006, 4, 31–42. [Google Scholar] [CrossRef]
- Imahashi, M.; Nakashima, M.; Iwatani, Y. Antiviral Mechanism and Biochemical Basis of the Human APOBEC3 Family. Front. Microbiol. 2012, 3, 250. [Google Scholar] [CrossRef]
- Revy, P.; Muto, T.; Levy, Y.; Geissmann, F.; Plebani, A.; Sanal, O.; Catalan, N.; Forveille, M.; Dufourcq-Lagelouse, R.; Gennery, A.; et al. Activation-Induced Cytidine Deaminase (AID) Deficiency Causes the Autosomal Recessive Form of the Hyper-IgM Syndrome (HIGM2). Cell 2000, 102, 565–575. [Google Scholar] [CrossRef]
- Takahashi, I.; Marmur, J. Replacement of thymidylic acid by deoxyuridylic acid in the deoxyribonucleic acid of a transducing phage for Bacillus subtilis. Nature 1963, 197, 794–795. [Google Scholar] [CrossRef]
- Skurnik, M.; Venho, R.; Toivanen, P.; Al-Hendy, A. A novel locus of Yersinia enterocolitica serotype O:3 involved in lipopolysaccharide outer core biosynthesis. Mol. Microbiol. 1995, 17, 575–594. [Google Scholar] [CrossRef]
- Kiljunen, S.; Hakala, K.; Pinta, E.; Huttunen, S.; Pluta, P.; Gador, A.; Lönnberg, H.; Skurnik, M. Yersiniophage phiR1-37 is a tailed bacteriophage having a 270 kb DNA genome with thymidine replaced by deoxyuridine. Microbiology 2005, 151, 4093–4102. [Google Scholar] [CrossRef]
- Skurnik, M.; Hyytiäinen, H.J.; Happonen, L.J.; Kiljunen, S.; Datta, N.; Mattinen, L.; Williamson, K.; Kristo, P.; Szeliga, M.; Kalin-Mänttäri, L.; et al. Characterization of the genome, proteome, and structure of yersiniophage ϕR1-37. J. Virol. 2012, 86, 12625–12642. [Google Scholar] [CrossRef][Green Version]
- Uchiyama, J.; Takemura-Uchiyama, I.; Sakaguchi, Y.; Gamoh, K.; Kato, S.; Daibata, M.; Ujihara, T.; Misawa, N.; Matsuzaki, S. Intragenus generalized transduction in Staphylococcus spp. by a novel giant phage. ISME J. 2014, 8, 1949–1952. [Google Scholar] [CrossRef]
- Belyaeva, N.N.; Azizbekyan, R.R. Fine structure of new Bacillus subtilis phage AR9 with complex morphology. Virology 1968, 34, 176–179. [Google Scholar] [CrossRef]
- Lavysh, D.; Sokolova, M.; Minakhin, L.; Yakunina, M.; Artamonova, T.; Kozyavkin, S.; Makarova, K.S.; Koonin, E.V.; Severinov, K. The genome of AR9, a giant transducing Bacillus phage encoding two multisubunit RNA polymerases. Virology 2016, 495, 185–196. [Google Scholar] [CrossRef]
- Ackermann, H.-W. Phage Classification and Characterization. In Bacteriopages; Methods in Molecular Biology; Clokie, M.R.J., Kropinski, A.M., Eds.; Humana Press: Totowa, NJ, USA, 2009; Volume 501, pp. 127–140. [Google Scholar]
- Ackermann, H.-W. Frequency of morphological phage descriptions in the year 2000. Arch. Virol. 2001, 146, 843–857. [Google Scholar] [CrossRef]
- Nagy, G.N.; Leveles, I.; Vértessy, B.G. Preventive DNA repair by sanitizing the cellular (deoxy)nucleoside triphosphate pool. FEBS J. 2014, 281, 4207–4223. [Google Scholar] [CrossRef]
- Vodenkova, S.; Buchler, T.; Cervena, K.; Veskrnova, V.; Vodicka, P.; Vymetalkova, V. 5-fluorouracil and other fluoropyrimidines in colorectal cancer: Past, present and future. Pharmacol. Ther. 2020, 206, 107447. [Google Scholar] [CrossRef]
- Baratelli, C.; Zichi, C.; Di Maio, M.; Brizzi, M.P.; Sonetto, C.; Scagliotti, G.V.; Tampellini, M. A systematic review of the safety profile of the different combinations of fluoropyrimidines and oxaliplatin in the treatment of colorectal cancer patients. Crit. Rev. Oncol. Hematol. 2018, 122, 21–29. [Google Scholar] [CrossRef]
- Wilson, P.M.; Danenberg, P.V.; Johnston, P.G.; Lenz, H.-J.; Ladner, R.D. Standing the test of time: Targeting thymidylate biosynthesis in cancer therapy. Nat. Rev. Clin. Oncol. 2014, 11, 282–298. [Google Scholar] [CrossRef]
- Surányi, É.V.; Hírmondó, R.; Nyíri, K.; Tarjányi, S.; Kőhegyi, B.; Tóth, J.; Vértessy, B.G. Exploiting a Phage-Bacterium Interaction System as a Molecular Switch to Decipher Macromolecular Interactions in the Living Cell. Viruses 2018, 10, 168. [Google Scholar] [CrossRef]
- Yokogawa, T.; Yano, W.; Tsukioka, S.; Osada, A.; Wakasa, T.; Ueno, H.; Hoshino, T.; Yamamura, K.; Fujioka, A.; Fukuoka, M.; et al. dUTPase inhibition confers susceptibility to a thymidylate synthase inhibitor in DNA-repair-defective human cancer cells. Cancer Sci. 2020, 112, 422–432. [Google Scholar] [CrossRef]
- Nyíri, K.; Mertens, H.D.T.; Tihanyi, B.; Nagy, G.N.; Kőhegyi, B.; Matejka, J.; Harris, M.J.; Szabó, J.E.; Papp-Kádár, V.; Németh-Pongrácz, V.; et al. Structural model of human dUTPase in complex with a novel proteinaceous inhibitor. Sci. Rep. 2018, 8, 4326. [Google Scholar] [CrossRef]
- Szabó, J.E.; Németh, V.; Papp-Kádár, V.; Nyíri, K.; Leveles, I.; Bendes, A.Á.; Zagyva, I.; Róna, G.; Pálinkás, H.L.; Besztercei, B.; et al. Highly potent dUTPase inhibition by a bacterial repressor protein reveals a novel mechanism for gene expression control. Nucleic Acids Res. 2014, 42, 11912–11920. [Google Scholar] [CrossRef]
- Doi, T.; Yoh, K.; Shitara, K.; Takahashi, H.; Ueno, M.; Kobayashi, S.; Morimoto, M.; Okusaka, T.; Ueno, H.; Morizane, C.; et al. First-in-human phase 1 study of novel dUTPase inhibitor TAS-114 in combination with S-1 in Japanese patients with advanced solid tumors. Investig. New Drugs 2019, 37, 507–518. [Google Scholar] [CrossRef]
- Hirmondó, R.; Szabó, J.E.; Nyíri, K.; Tarjányi, S.; Dobrotka, P.; Tóth, J.; Vértessy, B.G. Cross-species inhibition of dUTPase via the Staphylococcal Stl protein perturbs dNTP pool and colony formation in Mycobacterium. DNA Repair 2015, 30, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Kawazoe, A.; Takahari, D.; Keisho, C.; Nakamura, Y.; Ikeno, T.; Wakabayashi, M.; Nomura, S.; Tamura, H.; Fukutani, M.; Hirano, N.; et al. A multicenter phase II study of TAS-114 in combination with S-1 in patients with pretreated advanced gastric cancer (EPOC1604). Gastric Cancer 2021, 24, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Nyíri, K.; Harris, M.J.; Matejka, J.; Ozohanics, O.; Vékey, K.; Borysik, A.J.; Vértessy, B.G. HDX and Native Mass Spectrometry Reveals the Different Structural Basis for Interaction of the Staphylococcal Pathogenicity Island Repressor Stl with Dimeric and Trimeric Phage dUTPases. Biomolecules 2019, 9, 488. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Han, X.; Qing, Y.; Condie, A.G.; Gorityala, S.; Yang, S.; Xu, Y.; Zhang, Y.; Gerson, S.L. Inhibition of uracil DNA glycosylase sensitizes cancer cells to 5-fluorodeoxyuridine through replication fork collapse-induced DNA damage. Oncotarget 2016, 7, 59299–59313. [Google Scholar] [CrossRef]
- Liao, Y.-T.; Lin, S.-J.; Ko, T.-P.; Liu, C.-Y.; Hsu, K.-C.; Wang, H.-C. Structural insight into the differential interactions between the DNA mimic protein SAUGI and two gamma herpesvirus uracil-DNA glycosylases. Int. J. Biol. Macromol. 2020, 160, 903–914. [Google Scholar] [CrossRef]
- Mehta, A.; Raj, P.; Sundriyal, S.; Gopal, B.; Varshney, U. Use of a molecular beacon based fluorescent method for assaying uracil DNA glycosylase (Ung) activity and inhibitor screening. Biochem. Biophys. Rep. 2021, 26, 100954. [Google Scholar] [CrossRef]
- Mechetin, G.V.; Endutkin, A.V.; Diatlova, E.A.; Zharkov, D.O. Inhibitors of DNA Glycosylases as Prospective Drugs. Int. J. Mol. Sci. 2020, 21, 3118. [Google Scholar] [CrossRef]
- Jiang, Y.L.; Krosky, D.J.; Seiple, L.; Stivers, J.T. Uracil-Directed Ligand Tethering: An Efficient Strategy for Uracil DNA Glycosylase (UNG) Inhibitor Development. J. Am. Chem. Soc. 2005, 127, 17412–17420. [Google Scholar] [CrossRef] [PubMed]
- Focher, F.; Verri, A.; Spadari, S.; Manservigi, R.; Gambino, J.; Wright, G.E. Herpes simplex virus type 1 uracil-DNA glycosylase: Isolation and selective inhibition by novel uracil derivatives. Biochem. J. 1993, 292, 883–889. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suksangpleng, T.; Leartsakulpanich, U.; Moonsom, S.; Siribal, S.; Boonyuen, U.; Wright, G.E.; Chavalitshewinkoon-Petmitr, P. Molecular characterization of Plasmodium falciparum uracil-DNA glycosylase and its potential as a new anti-malarial drug target. Malar. J. 2014, 13, 149. [Google Scholar] [CrossRef] [PubMed]
- Krosky, D.J.; Bianchet, M.A.; Seiple, L.; Chung, S.; Amzel, L.M.; Stivers, J.T. Mimicking damaged DNA with a small molecule inhibitor of human UNG2. Nucleic Acids Res. 2006, 34, 5872–5879. [Google Scholar] [CrossRef]
- Chang, H.-L.; Su, K.-Y.; Goodman, S.D.; Yen, R.-S.; Cheng, W.-C.; Yang, Y.-C.; Lin, L.-I.; Chang, S.-Y.; Fang, W. Measurement of uracil-DNA glycosylase activity by matrix assisted laser desorption/ionization time-of-flight mass spectrometry technique. DNA Repair 2021, 97, 103028. [Google Scholar] [CrossRef]
- Serrano-Heras, G.; Ruiz-Masó, J.A.; del Solar, G.; Espinosa, M.; Bravo, A.; Salas, M. Protein p56 from the Bacillus subtilis phage phi29 inhibits DNA-binding ability of uracil-DNA glycosylase. Nucleic Acids Res. 2007, 35, 5393–5401. [Google Scholar] [CrossRef]
- Wang, H.-C.; Hsu, K.-C.; Yang, J.-M.; Wu, M.-L.; Ko, T.-P.; Lin, S.-R.; Wang, A.H.-J. Staphylococcus aureus protein SAUGI acts as a uracil-DNA glycosylase inhibitor. Nucleic Acids Res. 2014, 42, 1354–1364. [Google Scholar] [CrossRef][Green Version]
- Pathak, D.; Kwon, E.; Kim, D.Y. Selective interactions between mimivirus uracil-DNA glycosylase and inhibitory proteins determined by a single amino acid. J. Struct. Biol. 2020, 211, 107552. [Google Scholar] [CrossRef]
- Friedberg, E.C.; Ganesan, A.K.; Minton, K. N-Glycosidase activity in extracts of Bacillus subtilis and its inhibition after infection with bacteriophage PBS2. J. Virol. 1975, 16, 315–321. [Google Scholar] [CrossRef]
- Cone, R.; Bonura, T.; Friedberg, E.C. Inhibitor of uracil-DNA glycosylase induced by bacteriophage PBS2. Purification and preliminary characterization. J. Biol. Chem. 1980, 255, 10354–10358. [Google Scholar] [CrossRef]
- Wang, Z.; Mosbaugh, D.W. Uracil-DNA glycosylase inhibitor of bacteriophage PBS2: Cloning and effects of expression of the inhibitor gene in Escherichia coli. J. Bacteriol. 1988, 170, 1082–1091. [Google Scholar] [CrossRef]
- Serrano-Heras, G.; Salas, M.; Bravo, A. A Uracil-DNA Glycosylase Inhibitor Encoded by a Non-uracil Containing Viral DNA. J. Biol. Chem. 2006, 281, 7068–7074. [Google Scholar] [CrossRef]
- Papp-Kádár, V.; Balázs, Z.; Vékey, K.; Ozohanics, O.; Vértessy, B.G. Mass spectrometry-based analysis of macromolecular complexes of Staphylococcus aureus uracil-DNA glycosylase and its inhibitor reveals specific variations due to naturally occurring mutations. FEBS Open Bio 2019, 9, 420–427. [Google Scholar] [CrossRef]
- Serrano-Heras, G.; Bravo, A.; Salas, M. Phage phi29 protein p56 prevents viral DNA replication impairment caused by uracil excision activity of uracil-DNA glycosylase. Proc. Natl. Acad. Sci. USA 2008, 105, 19044–19049. [Google Scholar] [CrossRef]
- Kerepesi, C.; Szabó, J.; Papp-Kádár, V.; Dobay, O.; Szabó, D.; Grolmusz, V.; Vértessy, B. Life without dUTPase. Front. Microbiol. 2016, 7, 1768. [Google Scholar] [CrossRef]
- Asensio, J.L.; Pérez-Lago, L.; Lázaro, J.M.; González, C.; Serrano-Heras, G.; Salas, M. Novel dimeric structure of phage ϕ29-encoded protein p56: Insights into uracil-DNA glycosylase inhibition. Nucleic Acids Res. 2011, 39, 9779–9788. [Google Scholar] [CrossRef]
- Mir-Sanchis, I.; Roman, C.A.; Misiura, A.; Pigli, Y.Z.; Boyle-Vavra, S.; Rice, P.A. Staphylococcal SCCmec elements encode an active MCM-like helicase and thus may be replicative. Nat. Struct. Mol. Biol. 2016, 23, 891–898. [Google Scholar] [CrossRef]
- Putnam, C.D.; Shroyer, M.J.N.; Lundquist, A.J.; Mol, C.D.; Arvai, A.S.; Mosbaugh, D.W.; Tainer, J.A. Protein mimicry of DNA from crystal structures of the uracil-DNA glycosylase inhibitor protein and its complex with Escherichia coli uracil-DNA glycosylase. J. Mol. Biol. 1999, 287, 331–346. [Google Scholar] [CrossRef]
- Baños-Sanz, J.I.; Mojardín, L.; Sanz-Aparicio, J.; Lázaro, J.M.; Villar, L.; Serrano-Heras, G.; González, B.; Salas, M. Crystal structure and functional insights into uracil-DNA glycosylase inhibition by phage ϕ29 DNA mimic protein p56. Nucleic Acids Res. 2013, 41, 6761–6773. [Google Scholar] [CrossRef]
- Saikrishnan, K.; Bidya Sagar, M.; Ravishankar, R.; Roy, S.; Purnapatre, K.; Handa, P.; Varshney, U.; Vijayan, M. Domain closure and action of uracil DNA glycosylase (UDG): Structures of new crystal forms containing the Escherichia coli enzyme and a comparative study of the known structures involving UDG. Acta Crystallogr. Sect. D Biol. Crystallogr. 2002, 58, 1269–1276. [Google Scholar] [CrossRef]
- Zhang, F.; Wen, Y.; Guo, X. CRISPR/Cas9 for genome editing: Progress, implications and challenges. Hum. Mol. Genet. 2014, 23, R40–R46. [Google Scholar] [CrossRef] [PubMed]
- Pálinkás, H.L.; Békési, A.; Róna, G.; Pongor, L.; Papp, G.; Tihanyi, G.; Holub, E.; Póti, Á.; Gemma, C.; Ali, S.; et al. Genome-wide alterations of uracil distribution patterns in human DNA upon chemotherapeutic treatments. eLife 2020, 9, e60498. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.B.; Azeredo, J. Bacteriophage-Based Biotechnological Applications. Viruses 2019, 11, 737. [Google Scholar] [CrossRef] [PubMed]
- Monteiro, R.; Pires, D.P.; Costa, A.R.; Azeredo, J. Phage Therapy: Going Temperate? Trends Microbiol. 2019, 27, 368–378. [Google Scholar] [CrossRef]

| U-DNA Phages | Family | Produced UNG Inhibitor | Host | Reference |
|---|---|---|---|---|
| PBS1/PBS2 | Myoviridae | UGI | Bacillus spp. | [14] |
| ΦR1-37 | ? | Yersinia spp. | [16] | |
| S6 | ? | Staphylococcus spp. | [18] | |
| AR9 | UGI | Bacillus spp. | [20] |
| Inhibitor Compound | Species | IC50 | Reference |
|---|---|---|---|
| uracil base | Mycobacterium tuberculosis | 2.05 mM | [37] |
| 6-(p-n-octylanilino)uracil (OctAU) | Herpes simplex virus type1 | 8 µM | [40] |
| 1-methoxyethyl-6-(p-n-octylanilino)uracil | Plasmodium falciparum | 16.75 μM | [41] |
| 6-(phenylhydrazino)uracil | 77.5 μM | ||
| 4-[(1E,7E)-8-(2,6-dioxo-1,2,3,6-tetrahydropyrimidin-4-YL)-3,6-dioxa-2,7-diazaocta-1,7-dien-1-YL]benzoic acid and its derivatives * | human | 9 μM | [42] |
| 0.26 μM | [39] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagy, K.K.; Skurnik, M.; Vértessy, B.G. Viruses with U-DNA: New Avenues for Biotechnology. Viruses 2021, 13, 875. https://doi.org/10.3390/v13050875
Nagy KK, Skurnik M, Vértessy BG. Viruses with U-DNA: New Avenues for Biotechnology. Viruses. 2021; 13(5):875. https://doi.org/10.3390/v13050875
Chicago/Turabian StyleNagy, Kinga K., Mikael Skurnik, and Beáta G. Vértessy. 2021. "Viruses with U-DNA: New Avenues for Biotechnology" Viruses 13, no. 5: 875. https://doi.org/10.3390/v13050875
APA StyleNagy, K. K., Skurnik, M., & Vértessy, B. G. (2021). Viruses with U-DNA: New Avenues for Biotechnology. Viruses, 13(5), 875. https://doi.org/10.3390/v13050875







