Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin
Abstract
1. Introduction
2. Materials and Methods
2.1. Database
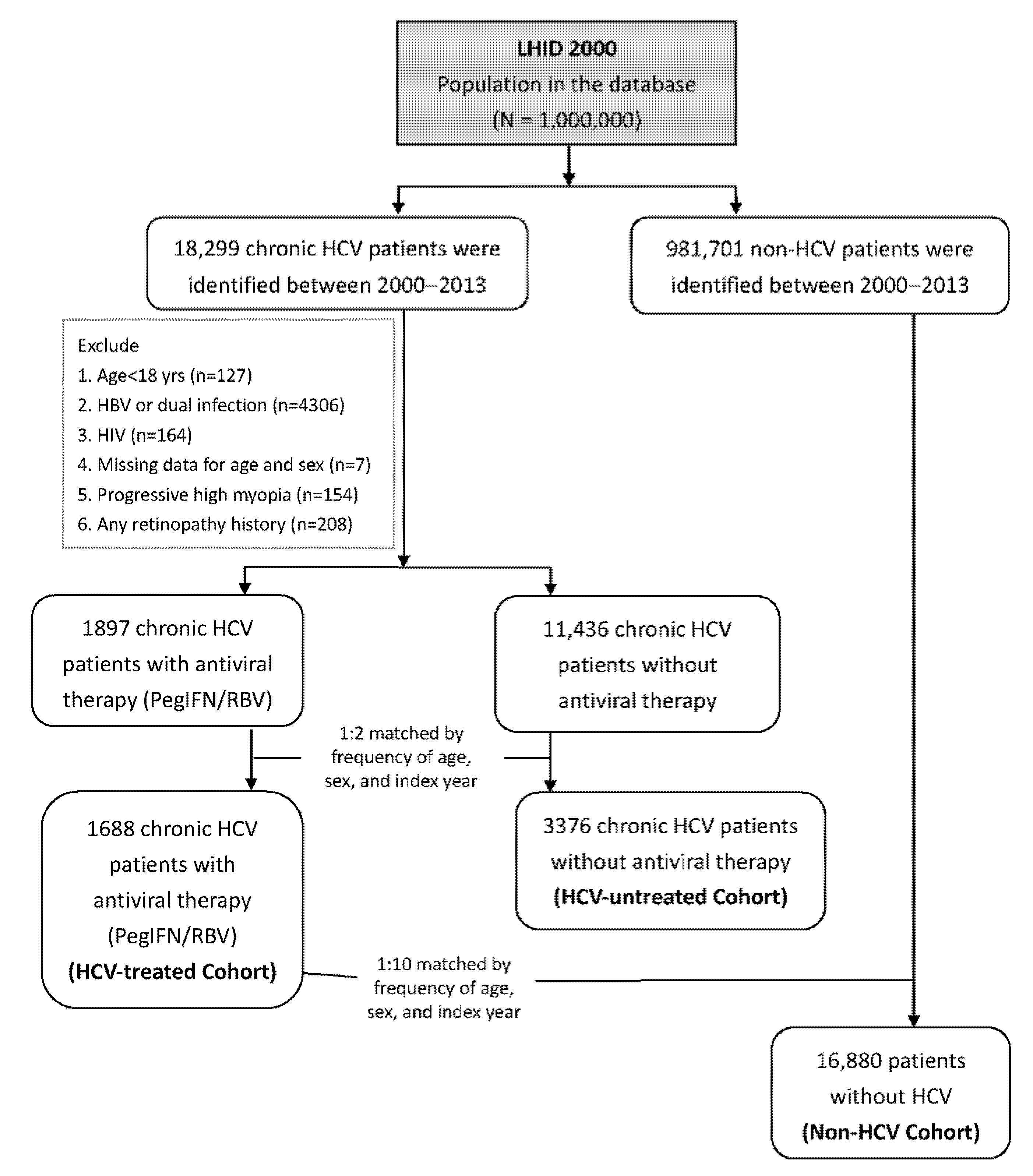
2.2. Study Sample
2.3. Statistical Analysis
3. Results
3.1. General Characteristics of Patients
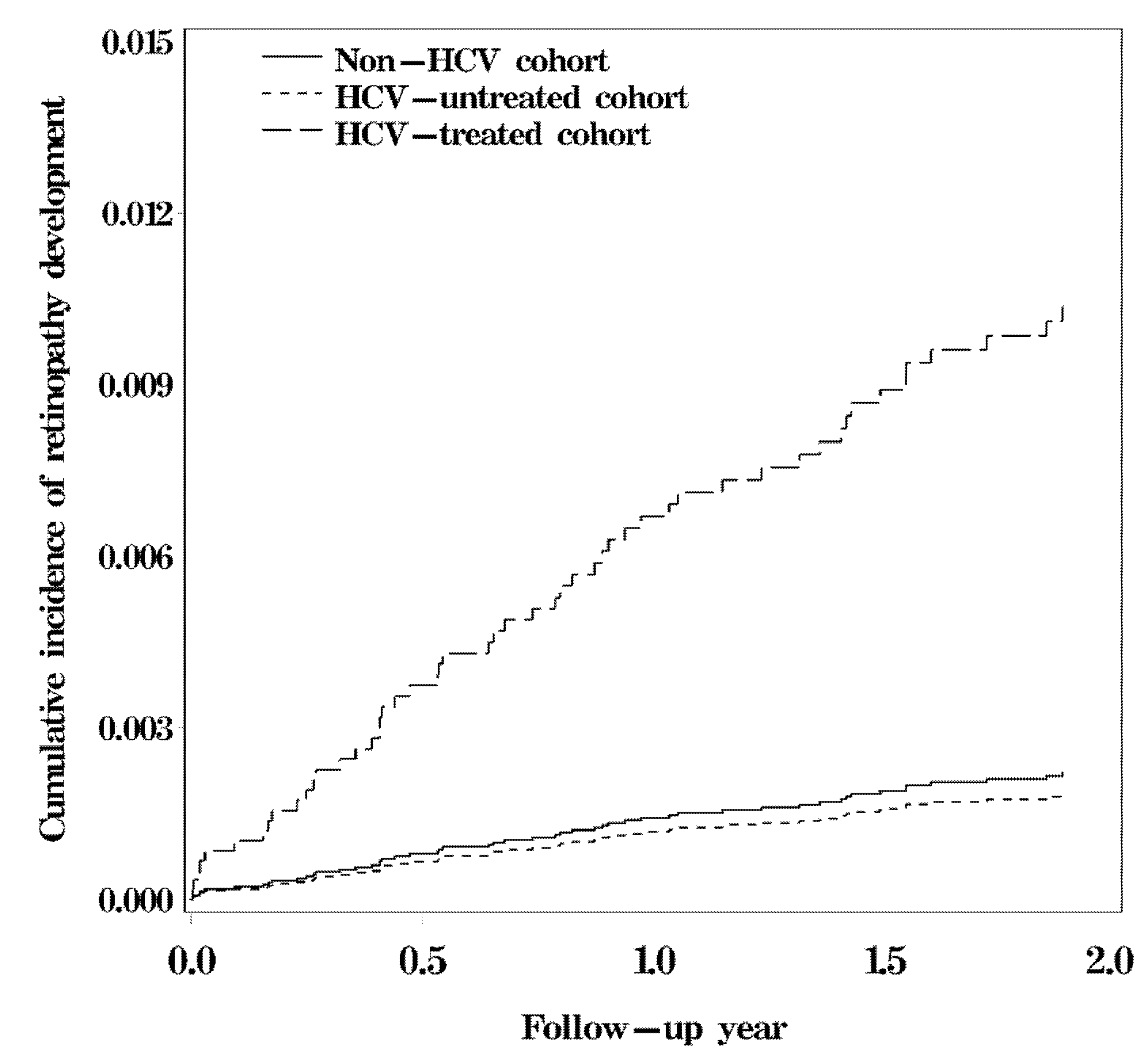
3.2. Survival Analysis for Interferon-Associated Retinopathy among Study Cohorts
3.3. Risk of Interferon-Associated Retinopathy among Study Cohorts
3.4. Risk of Side Effects Induced by PegIFN/RBV Treatment among Study Cohorts
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Petruzziello, A.; Marigliano, S.; Loquercio, G.; Cozzolino, A.; Cacciapuoti, C. Global epidemiology of hepatitis C virus infection: An up-date of the distribution and circulation of hepatitis C virus genotypes. World J. Gastroenterol. 2016, 22, 7824–7840. [Google Scholar] [CrossRef]
- Younossi, Z.; Park, H.; Henry, L.; Adeyemi, A.; Stepanova, M. Extrahepatic Manifestations of Hepatitis C: A Meta-analysis of Prevalence, Quality of Life, and Economic Burden. Gastroenterology 2016, 150, 1599–1608. [Google Scholar] [CrossRef]
- Tsoumani, A.; Theopistos, V.; Katsanos, K.; Asproudis, I.; Tsianos, E.V. Treatment and non-treatment related ocular manifestations in patients with chronic hepatitis B or C. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 1123–1131. [Google Scholar] [PubMed]
- Shiffman, M.L. Pegylated interferons: What role will they play in the treatment of chronic hepatitis C? Curr. Gastroenterol. Rep. 2001, 3, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.L.; Dai, C.Y.; Huang, J.F.; Hou, N.J.; Lee, L.P.; Hsieh, M.Y.; Chiu, C.F.; Lin, Z.Y.; Chen, S.C.; Hsieh, M.Y.; et al. A randomised study of peginterferon and ribavirin for 16 versus 24 weeks in patients with genotype 2 chronic hepatitis C. Gut 2007, 56, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.L.; Dai, C.Y.; Huang, J.F.; Chiu, C.F.; Yang, Y.H.; Hou, N.J.; Lee, L.P.; Hsieh, M.Y.; Lin, Z.Y.; Chen, S.C.; et al. Rapid virological response and treatment duration for chronic hepatitis C genotype 1 patients: A randomized trial. Hepatology 2008, 47, 1884–1893. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.L.; Chuang, W.L. Treatment of chronic hepatitis C in Asia: When East meets West. J. Gastroenterol. Hepatol. 2009, 24, 336–345. [Google Scholar] [CrossRef]
- Vujosevic, S.; Tempesta, D.; Noventa, F.; Midena, E.; Sebastiani, G. Pegylated interferon-associated retinopathy is frequent in hepatitis C virus patients with hypertension and justifies ophthalmologic screening. Hepatology 2012, 56, 455–463. [Google Scholar] [CrossRef]
- Hayasaka, S.; Nagaki, Y.; Matsumoto, M.; Sato, S. Interferon associated retinopathy. Br. J. Ophthalmol. 1998, 82, 323–325. [Google Scholar] [CrossRef] [PubMed]
- Raza, A.; Mittal, S.; Sood, G.K. Interferon-associated retinopathy during the treatment of chronic hepatitis C: A systematic review. J. Viral. Hepat. 2013, 20, 593–599. [Google Scholar] [CrossRef]
- Abd El-Badie Mohamed, M.; Abd-El Azeem Eed, K. Retinopathy associated with interferon therapy in patients with hepatitis C virus. Clin. Ophthalmol. 2012, 6, 1341–1345. [Google Scholar] [CrossRef]
- Fraunfelder, F.W.; Fraunfelder, F.T. Interferon alfa-associated anterior ischemic optic neuropathy. Ophthalmology 2011, 118, 408–411. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.T.; Kim, L.H.; Lee, J.I.; Chin, H.S. Retinopathy in hepatitis C patients due to combination therapy with pegylated interferon and ribavirin. Jpn. J. Ophthalmol. 2009, 53, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Saito, H.; Ebinuma, H.; Nagata, H.; Inagaki, Y.; Saito, Y.; Wakabayashi, K.; Takagi, T.; Nakamura, M.; Katsura, H.; Oguchi, Y.; et al. Interferon-associated retinopathy in a uniform regimen of natural interferon-alpha therapy for chronic hepatitis C. Liver 2001, 21, 192–197. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.H.; Wang, I.H.; Woung, L.C.; Jou, J.R. Anterior ischemic optic neuropathy associated with pegylated interferon therapy for chronic hepatitis C. Ocul. Immunol. Inflamm. 2009, 17, 191–194. [Google Scholar] [CrossRef]
- Lai, C.H.; Yang, Y.H.; Chen, P.C.; King, Y.C.; Liu, C.Y. Retinal vascular complications associated with interferon-ribavirin therapy for chronic hepatitis C: A population-based study. Pharmacoepidemiol. Drug Saf. 2018, 27, 191–198. [Google Scholar] [CrossRef]
- Panetta, J.D.; Gilani, N. Interferon-induced retinopathy and its risk in patients with diabetes and hypertension undergoing treatment for chronic hepatitis C virus infection. Aliment Pharmacol. Ther. 2009, 30, 597–602. [Google Scholar] [CrossRef]
- Bureau of National Health Insurance (Taiwan). National Health Insurance in Taiwan 2009; Bureau of National Health Insurance (Taiwan): Taipei City, Taiwan, 2009.
- Lunn, M.; McNeil, D. Applying Cox regression to competing risks. Biometrics 1995, 51, 524–532. [Google Scholar] [CrossRef]
- Cuthbertson, F.M.; Davies, M.; McKibbin, M. Is screening for interferon retinopathy in hepatitis C justified? Br. J. Ophthalmol. 2004, 88, 1518–1520. [Google Scholar] [CrossRef]
- Nagaoka, T.; Sato, E.; Takahashi, A.; Yokohama, S.; Yoshida, A. Retinal circulatory changes associated with interferon-induced retinopathy in patients with hepatitis C. Investig. Ophthalmol. Vis. Sci. 2007, 48, 368–375. [Google Scholar] [CrossRef]
- El Raziky, M.; Zayed, N.A.; Abdel Baki, A.; Mansour, S.A.; Shahin, R.M.H. ITPA gene polymorphism (94C>A) effects on ribavirin-induced anemia during therapy in Egyptian patients with chronic hepatitis C. J. Med. Virol. 2017, 89, 1823–1829. [Google Scholar] [CrossRef] [PubMed]
- Fried, M.W.; Shiffman, M.L.; Reddy, K.R.; Smith, C.; Marinos, G.; Goncales, F.L., Jr.; Haussinger, D.; Diago, M.; Carosi, G.; Dhumeaux, D.; et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N. Engl. J. Med. 2002, 347, 975–982. [Google Scholar] [CrossRef] [PubMed]
- Hadziyannis, S.J.; Sette, H., Jr.; Morgan, T.R.; Balan, V.; Diago, M.; Marcellin, P.; Ramadori, G.; Bodenheimer, H., Jr.; Bernstein, D.; Rizzetto, M.; et al. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: A randomized study of treatment duration and ribavirin dose. Ann. Intern. Med. 2004, 140, 346–355. [Google Scholar] [CrossRef]
- Ichihara, H.; Koh, S.; Aoyama, Y.; Kumura, T.; Ohta, T.; Furukawa, Y.; Terada, Y.; Yamane, T.; Hino, M.; Mugitani, A. [Complication of pernicious anemia during interferon-beta treatment for type C chronic hepatitis]. Rinsho Ketsueki 2012, 53, 352–356. [Google Scholar] [PubMed]
- Manns, M.P.; McHutchison, J.G.; Gordon, S.C.; Rustgi, V.K.; Shiffman, M.; Reindollar, R.; Goodman, Z.D.; Koury, K.; Ling, M.; Albrecht, J.K. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: A randomised trial. Lancet 2001, 358, 958–965. [Google Scholar] [CrossRef]
- Roomer, R.; Hansen, B.E.; Janssen, H.L.; de Knegt, R.J. Thrombocytopenia and the risk of bleeding during treatment with peginterferon alfa and ribavirin for chronic hepatitis C. J. Hepatol. 2010, 53, 455–459. [Google Scholar] [CrossRef] [PubMed]
- Takaki, S.; Tsubota, A.; Hosaka, T.; Akuta, N.; Someya, T.; Kobayashi, M.; Suzuki, F.; Suzuki, Y.; Saitoh, S.; Arase, Y.; et al. Factors contributing to ribavirin dose reduction due to anemia during interferon alfa2b and ribavirin combination therapy for chronic hepatitis C. J. Gastroenterol. 2004, 39, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Carraro, M.C.; Rossetti, L.; Gerli, G.C. Prevalence of retinopathy in patients with anemia or thrombocytopenia. Eur. J. Haematol. 2001, 67, 238–244. [Google Scholar] [CrossRef] [PubMed]
- D’Amore, P.A. Mechanisms of retinal and choroidal neovascularization. Investig. Ophthalmol. Vis. Sci. 1994, 35, 3974–3979. [Google Scholar]
- Klagsbrun, M.; Soker, S. VEGF/VPF: The angiogenesis factor found? Curr. Biol. 1993, 3, 699–702. [Google Scholar] [CrossRef]
- Shweiki, D.; Itin, A.; Soffer, D.; Keshet, E. Vascular endothelial growth factor induced by hypoxia may mediate hypoxia-initiated angiogenesis. Nature 1992, 359, 843–845. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Non-HCV (n = 16,880) | HCV-Untreated (n = 3376) | HCV-Treated (n = 1688) | p Value |
|---|---|---|---|---|
| Age, years | 51.5 (12.0) | 51.6 (12.0) | 51.4 (11.9) | 0.88 |
| Male sex, n (%) | 9130 (54.1) | 1826 (54.1) | 913 (54.1) | >0.99 |
| Occupation | <0.0001 | |||
| White-collar | 9545 (56.6) | 1439 (42.6) | 709 (42.0) | |
| Blue-collar | 6060 (35.9) | 1646 (48.8) | 873 (51.7) | |
| Other | 1275 (7.55) | 291 (8.62) | 106 (6.28) | |
| Urbanisation | <0.0001 | |||
| Urban | 5379 (31.9) | 753 (22.3) | 362 (21.5) | |
| Suburban | 8074 (47.8) | 1572 (46.6) | 794 (47.0) | |
| Rural | 3427 (20.3) | 1051 (31.1) | 532 (31.5) | |
| Geographic region | <0.0001 | |||
| Northern | 8015 (47.5) | 1011 (30.0) | 466 (27.6) | |
| Central | 3286 (19.5) | 735 (21.8) | 366 (21.7) | |
| Southern | 4782 (28.3) | 1429 (42.3) | 764 (45.3) | |
| Eastern and outlying islands | 797 (4.72) | 201 (5.95) | 92 (5.45) | |
| Monthly Income (NT$) | <0.0001 | |||
| <15,840 | 5845 (34.6) | 1008 (29.9) | 437 (25.9) | |
| 15,840–24,999 | 6431 (38.1) | 1689 (50.0) | 884 (52.4) | |
| ≥25,000 | 4604 (27.3) | 679 (20.1) | 367 (21.7) | |
| Comorbidities | ||||
| Myocardial infarction | 2092 (12.4) | 780 (23.1) | 313 (18.5) | <0.0001 |
| Cerebrovascular disease | 1182 (7.00) | 454 (13.5) | 184 (10.9) | <0.0001 |
| Chronic pulmonary disease | 4025 (23.8) | 1204 (35.7) | 623 (36.9) | <0.0001 |
| Diabetes mellitus | 2328 (13.8) | 960 (28.4) | 475 (28.1) | <0.0001 |
| Renal disease | 489 (2.90) | 348 (10.3) | 96 (5.69) | <0.0001 |
| Hypertension | 4830 (28.6) | 1503 (44.5) | 745 (44.1) | <0.0001 |
| Hyperlipidaemia | 1916 (11.4) | 548 (16.2) | 240 (14.2) | 0.002 |
| Cataract | 1572 (9.31) | 642 (19.0) | 323 (19.1) | <0.0001 |
| Diabetic retinopathy | 242 (1.43) | 112 (3.32) | 39 (2.31) | <0.0001 |
| Liver cirrhosis | 2697 (16.0) | 2448 (72.5) | 1537 (91.1) | <0.0001 |
| Obesity | 362 (2.14) | 83 (2.46) | 45 (2.67) | 0.24 |
| Anemia | 1388 (8.22) | 595 (17.6) | 249 (14.8) | <0.0001 |
| Medications | ||||
| Statin | 1182 (7.00) | 212 (6.28) | 73 (4.32) | <0.0001 |
| Aspirin | 18 (0.11) | 8 (0.24) | 2 (0.12) | 0.15 |
| Nonsteroidal anti-inflammatory drugs | 51 (0.30) | 14 (0.41) | 10 (0.59) | 0.11 |
| Retinopathy | Non-HCV | HCV-Untreated | p Value | HCV-Treated | p Value | |
|---|---|---|---|---|---|---|
| Any retinopathy | Cases | 32 | 5 | 15 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 1.15 | 0.99 | 5.44 | |||
| HR a (95% CI) | 1.00 (ref) | 0.71 (0.24–2.14) | 0.55 | 4.98 (2.02–12.3) | 0.0005 | |
| 1.00 (ref) | 9.02 (3.04–26.8) | <0.0001 | ||||
| Retinal venous occlusion | Cases | 11 | 3 | 3 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 0.40 | 0.60 | 1.09 | |||
| HR a (95% CI) | 1.00 (ref) | 0.70 (0.15–3.21) | 0.64 | 1.33 (0.29–6.18) | 0.72 | |
| 1.00 (ref) | 1.90 (0.40–9.00) | 0.42 | ||||
| Retinal arterial occlusion | Cases | 3 | 0 | 1 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 0.11 | 0.00 | 0.36 | |||
| HR a (95% CI) | 1.00 (ref) | NA | 0.71 | 90.6 (4.41–1865) | 0.0004 | |
| 1.00 (ref) | NA | |||||
| Retinal hemorrhage | Cases | 12 | 1 | 11 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 0.43 | 0.20 | 3.99 | |||
| HR a (95% CI) | 1.00 (ref) | 0.48 (0.07–3.40) | 0.46 | 12.7 (3.78–42.9) | <0.0001 | |
| 1.00 (ref) | 32.3 (3.94–265) | 0.001 | ||||
| Unspecified | Cases | 6 | 1 | 0 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 0.22 | 0.20 | 0.00 | |||
| HR a (95% CI) | 1.00 (ref) | 1.27 (0.03–47.3) | 0.90 | NA | ||
| 1.00 (ref) | NA |
| HCV-Untreated | HCV Patients with Antiviral Therapy (PegIFN/RBV) | |||||
|---|---|---|---|---|---|---|
| Retinopathy | <24 Weeks | p Value | ≥24 Weeks | p Value | ||
| Any retinopathy | Cases | 5 | 5 | 10 | ||
| Person-years | 5032 | 562 | 2194 | |||
| Incidence (10−3) | 0.99 | 8.90 | 4.56 | |||
| HR a (95% CI) | 1.00 (ref) | 11.3 (3.00–42.8) | 0.0003 | 8.18 (2.58–25.9) | 0.00004 | |
| Retinal venous occlusion | Cases | 3 | 2 | 1 | ||
| Person-years | 5032 | 562 | 2194 | |||
| Incidence (10−3) | 0.60 | 3.56 | 0.46 | |||
| HR a (95% CI) | 1.00 (ref) | 6.59 (0.94–46.2) | 0.06 | 0.76 (0.11–5.35) | 0.78 | |
| Retinal arterial occlusion | Cases | 0 | 1 | 0 | ||
| Person-years | 5032 | 562 | 2194 | |||
| Incidence (10−3) | 0.00 | 1.78 | 0.00 | |||
| HR a (95% CI) | 1.00 (ref) | NA | NA | |||
| Retinal hemorrhage | Cases | 1 | 2 | 9 | ||
| Person-years | 5032 | 562 | 2194 | |||
| Incidence (10−3) | 0.20 | 3.56 | 4.10 | |||
| HR a (95% CI) | 1.00 (ref) | 22.0 (1.91–254) | 0.01 | 36.2 (4.19–313) | 0.001 | |
| Unspecified | Cases | 1 | 0 | 0 | ||
| Person-years | 5032 | 562 | 2194 | |||
| Incidence (10−3) | 0.20 | 0.00 | 0.00 | |||
| HR a (95% CI) | 1.00 (ref) | NA | NA | |||
| Non-HCV | HCV-Untreated | p Value | HCV-Treated | p Value | ||
|---|---|---|---|---|---|---|
| Anemia | Cases | 179 | 77 | 152 | ||
| Person-years | 27,740 | 5007 | 2667 | |||
| Incidence (10−3) | 6.45 | 15.38 | 56.99 | |||
| HR a (95% CI) | 1.00 (ref) | 1.53 (1.10–2.13) | 0.01 | 8.10 (5.90–11.1) | <0.0001 | |
| 1.00 (ref) | 4.89 (3.66–6.55) | <0.0001 | ||||
| Thrombocytopenia | Cases | 39 | 52 | 34 | ||
| Person-years | 27,789 | 5032 | 2756 | |||
| Incidence (10−3) | 1.40 | 10.33 | 12.34 | |||
| HR a (95% CI) | 1.00 (ref) | 3.75 (2.15–6.54) | <0.0001 | 4.11 (2.22–7.61) | <0.0001 | |
| 1.00 (ref) | 1.13 (0.71–1.80) | 0.60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, C.-M.; Su, F.-H.; Muo, C.-H.; Huang, J.-C.; Wu, M.-M.; Yeh, C.-C. Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin. Viruses 2021, 13, 475. https://doi.org/10.3390/v13030475
Wu C-M, Su F-H, Muo C-H, Huang J-C, Wu M-M, Yeh C-C. Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin. Viruses. 2021; 13(3):475. https://doi.org/10.3390/v13030475
Chicago/Turabian StyleWu, Chia-Min, Fu-Hsiung Su, Chih-Hsin Muo, Jou-Chen Huang, Meei-Maan Wu, and Chih-Ching Yeh. 2021. "Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin" Viruses 13, no. 3: 475. https://doi.org/10.3390/v13030475
APA StyleWu, C.-M., Su, F.-H., Muo, C.-H., Huang, J.-C., Wu, M.-M., & Yeh, C.-C. (2021). Analysis of Different Types of Interferon-Associated Retinopathy in Patients with Chronic Hepatitis C Virus Infection Treated with Pegylated Interferon Plus Ribavirin. Viruses, 13(3), 475. https://doi.org/10.3390/v13030475







