Progress towards Eradication of Peste des Petits Ruminants through Vaccination
Abstract
1. Introduction
2. PPR Vaccine Development
2.1. Progress in PPR TT Vaccines
2.2. Progress in PPR DIVA Vaccines
3. PPR Outbreaks under the Vaccination Campaign
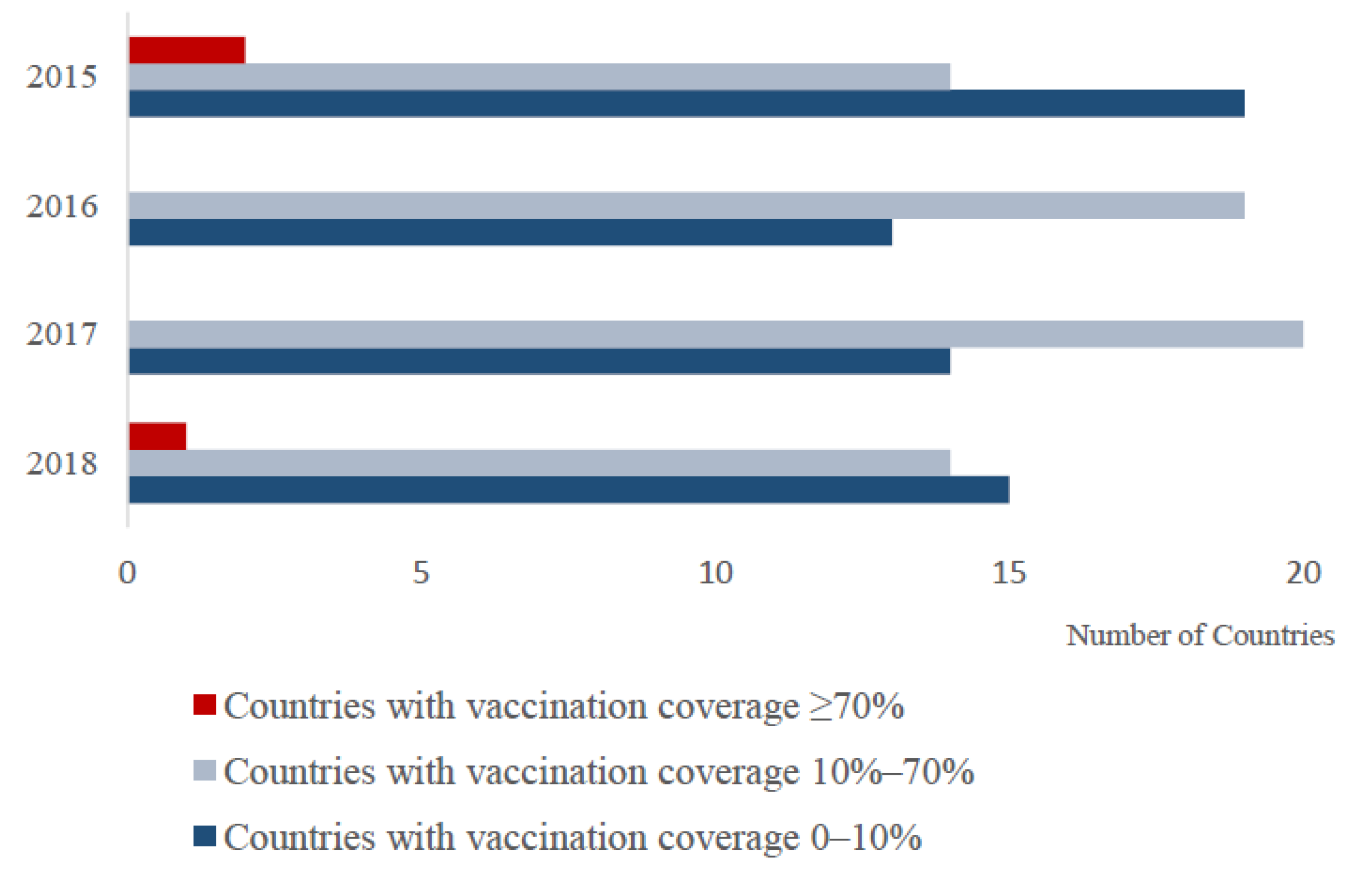
3.1. Global PPR Vaccine Use during 2015–2018
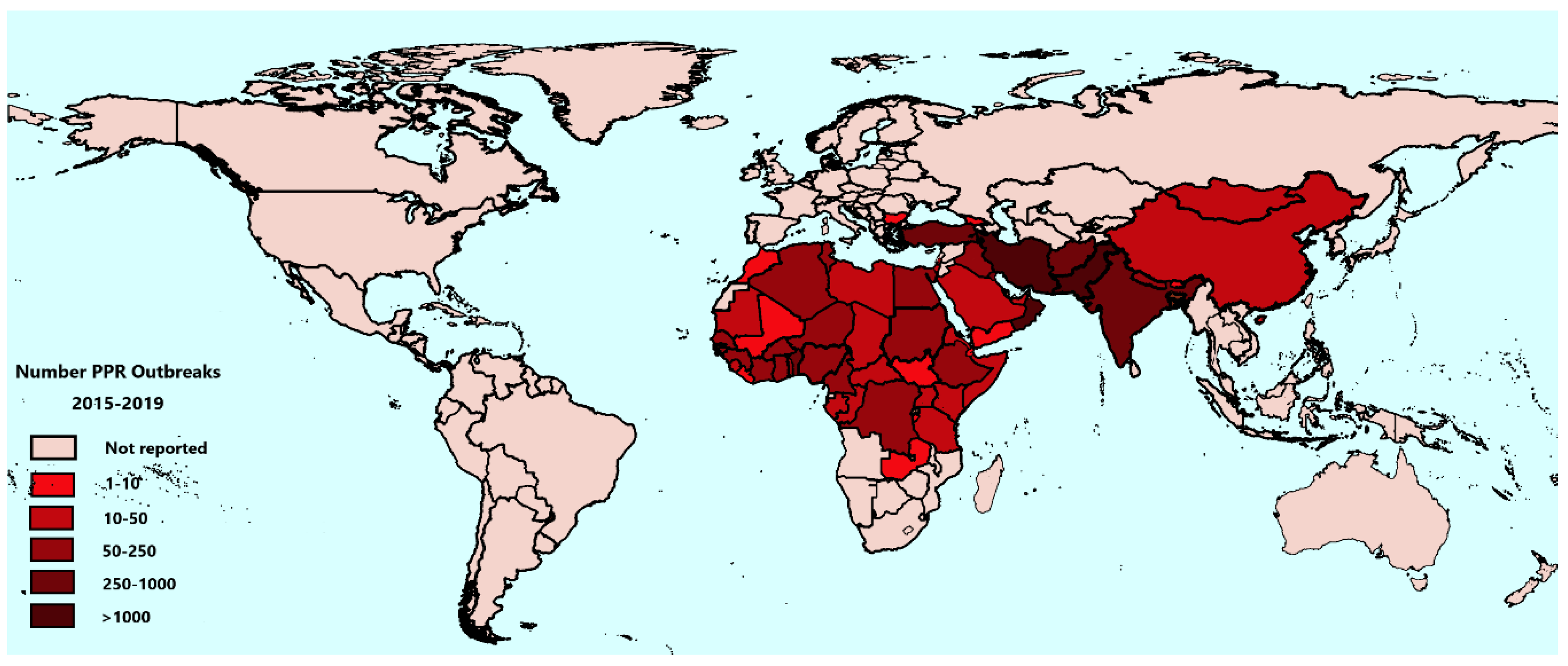
3.2. PPR Outbreaks from 2015 to 2019
4. Vaccination Strategies
4.1. PPR Vaccination Strategies in Countries from 2015 to 2018
4.1.1. Targeted Cost-Effective Vaccination Strategies Can Be Used to Maintain Uninfected Status or Control Sporadic Cases
4.1.2. Vaccination Coverage May Not Be Sufficient to Achieve PPR Elimination
4.1.3. Some Countries with Many PPR Outbreaks due to Lack of Sufficient or Timely Vaccine Coverage
4.2. Research Advances to Inform Evidence-Based PPR Control and Vaccination Strategies
5. Management of PPR Vaccination
5.1. Implementation of Vaccination Campaigns
5.2. Challenges for the Distribution and Use of PPR Vaccines
5.3. Funding Gaps for Operationalising Effective Vaccination Campaigns Still Remain
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Rahman, A.U.; Munir, M.; Shabbir, M.Z. A comparative phylogenomic analysis of peste des petits ruminants virus isolated from wild and unusual hosts. Mol. Biol. Rep. 2019, 46, 5587–5593. [Google Scholar] [CrossRef] [PubMed]
- Fine, A.E.; Pruvot, M.; Benfield, C.T.O.; Caron, A.; Cattoli, G.; Chardonnet, P.; Dioli, M.; Dulu, T.; Gilbert, M.; Kock, R.; et al. Eradication of Peste des Petits Ruminants Virus and the Wildlife-Livestock Interface. Front. Vet. Sci. 2020, 7, 50. [Google Scholar] [CrossRef]
- Rahman, A.U.; Dhama, K.; Ali, Q.; Hussain, I.; Oneeb, M.; Chaudhary, U.; Wensman, J.J.; Shabbir, M.Z. Peste des petits ruminants in large ruminants, camels and unusual hosts. Vet. Q. 2020, 40, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, E.P.; Taylor, W.P.; Lawman, M.J.; Bryant, J. Classification of peste des petits ruminants virus as the fourth member of the genus Morbillivirus. Intervirology 1979, 11, 268–274. [Google Scholar] [CrossRef] [PubMed]
- Implementation of Taxon-wide Non-Latinized Binomial Species Names in the Family Paramyxoviridae. Available online: https://talk.ictvonline.org/ictv/proposals/2016.011aM.A.v2.Paramyxoviridae_spren.pdf (accessed on 15 October 2020).
- Balamurugan, V.; Sen, A.; Venkatesan, G.; Yadav, V.; Bhanot, V.; Riyesh, T.; Bhanuprakash, V.; Singh, R.K. Sequence and phylogenetic analyses of the structural genes of virulent isolates and vaccine strains of peste des petits ruminants virus from India. Transbound. Emerg. Dis. 2010, 57, 352–364. [Google Scholar] [CrossRef] [PubMed]
- Muniraju, M.; Munir, M.; Banyard, A.C.; Ayebazibwe, C.; Wensman, J.; Zohari, S.; Berg, M.; Parthiban, A.R.; Mahapatra, M.; Libeau, G.; et al. Complete Genome Sequences of Lineage III Peste des Petits Ruminants Viruses from the Middle East and East Africa. Genome Announc. 2014, 2. [Google Scholar] [CrossRef]
- Muniraju, M.; Munir, M.; Parthiban, A.R.; Banyard, A.C.; Bao, J.; Wang, Z.; Ayebazibwe, C.; Ayelet, G.; El Harrak, M.; Mahapatra, M.; et al. Molecular evolution of peste des petits ruminants virus. Emerg. Infect. Dis. 2014, 20, 2023–2033. [Google Scholar] [CrossRef]
- Kwiatek, O.; Ali, Y.H.; Saeed, I.K.; Khalafalla, A.I.; Mohamed, O.I.; Obeida, A.A.; Abdelrahman, M.B.; Osman, H.M.; Taha, K.M.; Abbas, Z.; et al. Asian lineage of peste des petits ruminants virus, Africa. Emerg. Infect. Dis. 2011, 17, 1223–1231. [Google Scholar] [CrossRef]
- Dundon, W.G.; Diallo, A.; Cattoli, G. Peste des petits ruminants in Africa: A review of currently available molecular epidemiological data, 2020. Arch. Virol. 2020, 165, 2147–2163. [Google Scholar] [CrossRef]
- Shaila, M.S.; Shamaki, D.; Forsyth, M.A.; Diallo, A.; Goatley, L.; Kitching, R.P.; Barrett, T. Geographic distribution and epidemiology of peste des petits ruminants virus. Virus Res. 1996, 43, 149–153. [Google Scholar] [CrossRef]
- Dhar, P.; Sreenivasa, B.P.; Barrett, T.; Corteyn, M.; Singh, R.P.; Bandyopadhyay, S.K. Recent epidemiology of peste des petits ruminants virus (PPRV). Vet. Microbiol. 2002, 88, 153–159. [Google Scholar] [CrossRef]
- Abraham, G.; Sintayehu, A.; Libeau, G.; Albina, E.; Roger, F.; Laekemariam, Y.; Abayneh, D.; Awoke, K.M. Antibody seroprevalences against peste des petits ruminants (PPR) virus in camels, cattle, goats and sheep in Ethiopia. Prev. Vet. Med. 2005, 70, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Maherchandani, S.; Kashyap, S.K.; Singh, S.V.; Sharma, S.; Chaubey, K.K.; Ly, H. Peste des petits ruminants virus infection of small ruminants: A comprehensive review. Viruses 2014, 6, 2287–2327. [Google Scholar] [CrossRef] [PubMed]
- Khan, H.A.; Siddique, M.; Sajjad ur, R.; Abubakar, M.; Ashraf, M. The detection of antibody against peste des petits ruminants virus in sheep, goats, cattle and buffaloes. Trop. Anim. Health Prod. 2008, 40, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Abubakar, M.; Mahapatra, M.; Muniraju, M.; Arshed, M.J.; Khan, E.H.; Banyard, A.C.; Ali, Q.; Parida, S. Serological Detection of Antibodies to Peste des Petits Ruminants Virus in Large Ruminants. Transbound. Emerg. Dis. 2017, 64, 513–519. [Google Scholar] [CrossRef]
- Mahapatra, M.; Sayalel, K.; Muniraju, M.; Eblate, E.; Fyumagwa, R.; Shilinde, L.; Mdaki, M.; Keyyu, J.; Parida, S.; Kock, R. Spillover of Peste des Petits Ruminants Virus from Domestic to Wild Ruminants in the Serengeti Ecosystem, Tanzania. Emerg. Infect. Dis. 2015, 21, 2230–2234. [Google Scholar] [CrossRef]
- Fernandez Aguilar, X.; Mahapatra, M.; Begovoeva, M.; Kalema-Zikusoka, G.; Driciru, M.; Ayebazibwe, C.; Adwok, D.S.; Kock, M.; Lukusa, J.K.; Muro, J.; et al. Peste des Petits Ruminants at the Wildlife-Livestock Interface in the Northern Albertine Rift and Nile Basin, East Africa. Viruses 2020, 12, 293. [Google Scholar] [CrossRef]
- Pruvot, M.; Fine, A.E.; Hollinger, C.; Strindberg, S.; Damdinjav, B.; Buuveibaatar, B.; Chimeddorj, B.; Bayandonoi, G.; Khishgee, B.; Sandag, B.; et al. Outbreak of Peste des Petits Ruminants among Critically Endangered Mongolian Saiga and Other Wild Ungulates, Mongolia, 2016–2017. Emerg. Infect. Dis. 2020, 26, 51–62. [Google Scholar] [CrossRef]
- Aguilar, X.F.; Fine, A.E.; Pruvot, M.; Njeumi, F.; Walzer, C.; Kock, R.; Shiilegdamba, E. PPR virus threatens wildlife conservation. Science 2018, 362, 165–166. [Google Scholar] [CrossRef]
- Asil, R.M.; Ludlow, M.; Ballal, A.; Alsarraj, S.; Ali, W.H.; Mohamed, B.A.; Mutwakil, S.M.; Osman, N.A. First detection and genetic characterization of peste des petits ruminants virus from dorcas gazelles “Gazella dorcas” in the Sudan, 2016–2017. Arch. Virol. 2019, 164, 2537–2543. [Google Scholar] [CrossRef]
- Furley, C.W.; Taylor, W.P.; Obi, T.U. An outbreak of peste des petits ruminants in a zoological collection. Vet. Rec. 1987, 121, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Berkowitz, A.; Avni-Magen, N.; Bouznach, A.; Waner, T.; Litvak, A.; Friedgut, O.; Bombarov, V.; Guini-Rubinstein, M.; Stram, Y.; Eldar, A.; et al. Pathological and molecular characterisation of peste des petits ruminants in Nubian ibex (Capra nubiana) in Israel. Arch. Virol. 2019, 164, 1997–2003. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, V.; Sen, A.; Venkatesan, G.; Bhanot, V.; Yadav, V.; Bhanuprakash, V.; Singh, R.K. Peste des petits ruminants virus detected in tissues from an Asiatic lion (Panthera leo persica) belongs to Asian lineage IV. J. Vet. Sci. 2012, 13, 203–206. [Google Scholar] [CrossRef] [PubMed]
- Sevik, M.; Oz, M.E. Detection of Peste des petits ruminants virus RNA in Culicoides imicola (Diptera: Ceratopogonidae) in Turkey. Vet. Ital. 2019, 55, 173–179. [Google Scholar] [CrossRef]
- Nawathe, D.R.; Taylor, W.P. Experimental infection of domestic pigs with the virus of peste des petits ruminants. Trop. Anim. Health Prod. 1979, 11, 120–122. [Google Scholar] [CrossRef]
- Ratta, B.; Pokhriyal, M.; Singh, S.K.; Kumar, A.; Saxena, M.; Sharma, B. Detection of Peste Des Petits Ruminants Virus (PPRV) Genome from Nasal Swabs of Dogs. Curr. Microbiol. 2016, 73, 99–103. [Google Scholar] [CrossRef]
- Tounkara, K.; Traore, A.; Traore, A.P.; Sidibe, S.; Samake, K.; Diallo, B.O.; Diallo, A. [Epidemiology of rinderpest and cattle plague in Mali: Serological surveys]. Rev. D’elevage Et De Med. Vet. Des. Pays Trop 1996, 49, 273–277. [Google Scholar]
- Ngangnou, A.; Zoyem, N.; Hamet, M.; Abdoulkadiri, S. [Evaluation of vaccinal protection against rinderpest in Cameroon. III. General evaluation]. Rev. D’elevage Et De Med. Vet. Des. Pays Trop. 1996, 49, 18–22. [Google Scholar]
- Schulz, C.; Fast, C.; Schlottau, K.; Hoffmann, B.; Beer, M. Neglected Hosts of Small Ruminant Morbillivirus. Emerg Infect. Dis. 2018, 24, 2334–2337. [Google Scholar] [CrossRef]
- Schulz, C.; Fast, C.; Wernery, U.; Kinne, J.; Joseph, S.; Schlottau, K.; Jenckel, M.; Hoper, D.; Patteril, N.A.G.; Syriac, G.; et al. Camelids and Cattle Are Dead-End Hosts for Peste-des-Petits-Ruminants Virus. Viruses 2019, 11, 1133. [Google Scholar] [CrossRef]
- Diallo, A.; Taylor, W.P.; Lefevre, P.C.; Provost, A. Attenuation of a virulent PPRV strain: Potential homologous live vaccine. Rev. D’elevage Med. Vet. Pays. Trop. 1989, 42, 311–319. [Google Scholar]
- Bora, M.; Yousuf, R.W.; Dhar, P.; Singh, R.P. An overview of process intensification and thermo stabilization for upscaling of Peste des petits ruminants vaccines in view of global control and eradication. Virusdisease 2018, 29, 285–296. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, J.; Sreenivasa, B.P.; Singh, R.P.; Dhar, P.; Bandyopadhyay, S.K. Comparative efficacy of various chemical stabilizers on the thermostability of a live-attenuated peste des petits ruminants (PPR) vaccine. Vaccine 2003, 21, 4728–4735. [Google Scholar] [CrossRef]
- Diallo, A.; Minet, C.; Le Goff, C.; Berhe, G.; Albina, E.; Libeau, G.; Barrett, T. The threat of peste des petits ruminants: Progress in vaccine development for disease control. Vaccine 2007, 25, 5591–5597. [Google Scholar] [CrossRef]
- Sen, A.; Saravanan, P.; Balamurugan, V.; Rajak, K.K.; Sudhakar, S.B.; Bhanuprakash, V.; Parida, S.; Singh, R.K. Vaccines against peste des petits ruminants virus. Expert Rev. Vaccines 2010, 9, 785–796. [Google Scholar] [CrossRef]
- Hodgson, S.; Moffat, K.; Hill, H.; Flannery, J.T.; Graham, S.P.; Baron, M.D.; Darpel, K.E. Comparison of the Immunogenicities and Cross-Lineage Efficacies of Live Attenuated Peste des Petits Ruminants Virus Vaccines PPRV/Nigeria/75/1 and PPRV/Sungri/96. J. Virol. 2018, 92. [Google Scholar] [CrossRef]
- Mahapatra, M.; Selvaraj, M.; Parida, S. Comparison of Immunogenicity and Protective Efficacy of PPR Live Attenuated Vaccines (Nigeria 75/1 and Sungri 96) Administered by Intranasal and Subcutaneous Routes. Vaccines 2020, 8, 168. [Google Scholar] [CrossRef]
- Cosseddu, G.M.; Polci, A.; Pinoni, C.; Capobianco Dondona, A.; Iapaolo, F.; Orsini, G.; Izzo, F.; Bortone, G.; Ronchi, F.G.; Di Ventura, M.; et al. Evaluation of Humoral Response and Protective Efficacy of an Inactivated Vaccine Against Peste des Petits Ruminants Virus in Goats. Transbound. Emerg. Dis. 2016, 63, 447–452. [Google Scholar] [CrossRef]
- Ronchi, G.F.; Monaco, F.; Harrak, M.E.; Chafiqa, L.; Capista, S.; Bortone, G.; Orsini, G.; Pinoni, C.; Iorio, M.; Iapaolo, F.; et al. Preliminary results on innocuity and immunogenicity of an inactivated vaccine against Peste des petits ruminants. Vet. Ital. 2016, 52, 101–109. [Google Scholar] [CrossRef]
- Worrall, E.E.; Litamoi, J.K.; Seck, B.M.; Ayelet, G. Xerovac: An ultra rapid method for the dehydration and preservation of live attenuated Rinderpest and Peste des Petits ruminants vaccines. Vaccine 2000, 19, 834–839. [Google Scholar] [CrossRef]
- Silva, A.C.; Carrondo, M.J.; Alves, P.M. Strategies for improved stability of Peste des Petits Ruminants Vaccine. Vaccine 2011, 29, 4983–4991. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.C.; Yami, M.; Libeau, G.; Carrondo, M.J.; Alves, P.M. Testing a new formulation for Peste des Petits Ruminants vaccine in Ethiopia. Vaccine 2014, 32, 2878–2881. [Google Scholar] [CrossRef] [PubMed]
- Mariner, J.C.; Gachanja, J.; Tindih, S.H.; Toye, P. A thermostable presentation of the live, attenuated peste des petits ruminants vaccine in use in Africa and Asia. Vaccine 2017, 35, 3773–3779. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Chen, W.; Huang, K.; Baron, M.D.; Bu, Z. Rescue of recombinant peste des petits ruminants virus: Creation of a GFP-expressing virus and application in rapid virus neutralization test. Vet. Res. 2012, 43, 48. [Google Scholar] [CrossRef] [PubMed]
- Muniraju, M.; Mahapatra, M.; Buczkowski, H.; Batten, C.; Banyard, A.C.; Parida, S. Rescue of a vaccine strain of peste des petits ruminants virus: In vivo evaluation and comparison with standard vaccine. Vaccine 2015, 33, 465–471. [Google Scholar] [CrossRef]
- Liu, F.; Wu, X.; Zhao, Y.; Li, L.; Wang, Z. Budding of peste des petits ruminants virus-like particles from insect cell membrane based on intracellular co-expression of peste des petits ruminants virus M, H and N proteins by recombinant baculoviruses. J. Virol. Methods 2014, 207, 78–85. [Google Scholar] [CrossRef]
- Liu, F.; Wu, X.; Zou, Y.; Li, L.; Wang, Z. Peste des petits ruminants virus-like particles induce both complete virus-specific antibodies and virus neutralizing antibodies in mice. J. Virol. Methods 2015, 213, 45–49. [Google Scholar] [CrossRef]
- Wang, Q.; Ou, C.; Dou, Y.; Chen, L.; Meng, X.; Liu, X.; Yu, Y.; Jiang, J.; Ma, J.; Zhang, Z.; et al. M protein is sufficient for assembly and release of Peste des petits ruminants virus-like particles. Microb. Pathog. 2017, 107, 81–87. [Google Scholar] [CrossRef]
- Romero, C.H.; Barrett, T.; Kitching, R.P.; Bostock, C.; Black, D.N. Protection of goats against peste des petits ruminants with recombinant capripoxviruses expressing the fusion and haemagglutinin protein genes of rinderpest virus. Vaccine 1995, 13, 36–40. [Google Scholar] [CrossRef]
- Diallo, A.; Minet, C.; Berhe, G.; Le Goff, C.; Black, D.N.; Fleming, M.; Barrett, T.; Grillet, C.; Libeau, G. Goat immune response to capripox vaccine expressing the hemagglutinin protein of peste des petits ruminants. Ann. N. Y. Acad. Sci. 2002, 969, 88–91. [Google Scholar] [CrossRef]
- Grund, C.; Römer-Oberdörfer, A.; Mettenleiter, T.C.; Caufour, P.; Rufael, T.; Lamien, C.E.; Lancelot, R.; Kidane, M.; Awel, D.; Sertse, T.; et al. Protective efficacy of a single immunization with capripoxvirus-vectored recombinant peste des petits ruminants vaccines in presence of pre-existing immunity. Vaccines 2014, 32, 3772–3779. [Google Scholar] [CrossRef]
- Fakri, F.; Bamouh, Z.; Ghzal, F.; Baha, W.; Tadlaoui, K.; Fihri, O.F.; Chen, W.; Bu, Z.; Elharrak, M. Comparative evaluation of three capripoxvirus-vectored peste des petits ruminants vaccines. Virology 2018, 514, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Huang, H.; Ruan, Y.; Hou, X.; Yang, S.; Wang, C.; Huang, G.; Wang, T.; Feng, N.; Gao, Y.; et al. A novel recombinant Peste des petits ruminants-canine adenovirus vaccine elicits long-lasting neutralizing antibody response against PPR in goats. PLoS ONE 2012, 7, e37170. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, G.; Chen, Z.; Li, C.; Shi, L.; Li, W.; Huang, H.; Tao, C.; Cheng, C.; Xu, B.; et al. Recombinant adenovirus expressing F and H fusion proteins of peste des petits ruminants virus induces both humoral and cell-mediated immune responses in goats. Vet. Immunol. Immunopathol. 2013, 154, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Herbert, R.; Baron, J.; Batten, C.; Baron, M.; Taylor, G. Recombinant adenovirus expressing the haemagglutinin of Peste des petits ruminants virus (PPRV) protects goats against challenge with pathogenic virus; a DIVA vaccine for PPR. Vet. Res. 2014, 45, 24. [Google Scholar] [CrossRef] [PubMed]
- Rojas, J.M.; Moreno, H.; Valcárcel, F.; Peña, L.; Sevilla, N.; Martín, V. Vaccination with recombinant adenoviruses expressing the peste des petits ruminants virus F or H proteins overcomes viral immunosuppression and induces protective immunity against PPRV challenge in sheep. PLoS ONE 2014, 9, e101226. [Google Scholar] [CrossRef] [PubMed]
- Holzer, B.; Taylor, G.; Rajko-Nenow, P.; Hodgson, S.; Okoth, E.; Herbert, R.; Toye, P.; Baron, M.D. Determination of the minimum fully protective dose of adenovirus-based DIVA vaccine against peste des petits ruminants virus challenge in East African goats. Vet. Res. 2016, 47, 20. [Google Scholar] [CrossRef]
- Jones, L.; Giavedoni, L.; Saliki, J.T.; Brown, C.; Mebus, C.; Yilma, T. Protection of goats against peste des petits ruminants with a vaccinia virus double recombinant expressing the F and H genes of rinderpest virus. Vaccine 1993, 11, 961–964. [Google Scholar] [CrossRef]
- Chandran, D.; Reddy, K.B.; Vijayan, S.P.; Sugumar, P.; Rani, G.S.; Kumar, P.S.; Rajendra, L.; Srinivasan, V.A. MVA recombinants expressing the fusion and hemagglutinin genes of PPRV protects goats against virulent challenge. Indian J. Microbiol. 2010, 50, 266–274. [Google Scholar] [CrossRef]
- Murr, M.; Hoffmann, B. A Novel Recombinant Newcastle Disease Virus Vectored DIVA Vaccine against Peste des Petits Ruminants in Goats. Vaccines 2020, 8, 205. [Google Scholar] [CrossRef]
- Liu, F.; Li, L.; Liu, Y.; Sun, C.; Liu, C.; Wu, X.; Wang, Z. Development of reverse genetics system for small ruminant morbillivirus: Rescuing recombinant virus to express Echinococcus granulosus EG95 antigen. Virus Res. 2019, 261, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Yin, C.; Chen, W.; Hu, Q.; Wen, Z.; Wang, X.; Ge, J.; Yin, Q.; Zhi, H.; Xia, C.; Bu, Z. Induction of protective immune response against both PPRV and FMDV by a novel recombinant PPRV expressing FMDV VP1. Vet. Res. 2014, 45, 62. [Google Scholar] [CrossRef] [PubMed]
- Parida, S. Study on Early Events of PPR Virus Pathogenesis and Evaluation of PPR DIVA Vaccines in Goats. Available online: https://rr-asia.oie.int/wp-content/uploads/2020/06/200622_ppr-gren-2-nairobi-november-2019-final-communique.pdf (accessed on 17 November 2020).
- Xue, Q.; Xu, H.; Liu, H.; Pan, J.; Yang, J.; Sun, M.; Chen, Y.; Xu, W.; Cai, X.; Ma, H. Epitope-Containing Short Peptides Capture Distinct IgG Serodynamics That Enable Differentiating Infected from Vaccinated Animals for Live-Attenuated Vaccines. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Progress on PPR Control and Eradication Strategy. Available online: http://www.gf-tads.org/ppr/progress-on-ppr-control-and-eradication-strategy/en/ (accessed on 17 November 2020).
- Herzog, C.M.; de Glanville, W.A.; Willett, B.J.; Cattadori, I.M.; Kapur, V.; Hudson, P.J.; Buza, J.; Swai, E.S.; Cleaveland, S.; Bjornstad, O.N. Identifying Age Cohorts Responsible for Peste Des Petits Ruminants Virus Transmission among Sheep, Goats, and Cattle in Northern Tanzania. Viruses 2020, 12, 186. [Google Scholar] [CrossRef] [PubMed]
- Wane, A.; Dione, M.; Wieland, B.; Rich, K.M.; Yena, A.S.; Fall, A. Willingness to Vaccinate (WTV) and Willingness to Pay (WTP) for Vaccination Against Peste des Petits Ruminants (PPR) in Mali. Front. Vet. Sci. 2019, 6, 488. [Google Scholar] [CrossRef]
- Tago, D.; Sall, B.; Lancelot, R.; Pradel, J. VacciCost-A tool to estimate the resource requirements for implementing livestock vaccination campaigns. Application to peste des petits ruminants (PPR) vaccination in Senegal. Prev. Vet. Med. 2017, 144, 13–19. [Google Scholar] [CrossRef]
- Govindaraj, G.N.; Roy, G.; Mohanty, B.S.; Balamurugan, V. Evaluation of effectiveness of Mass Vaccination Campaign against Peste des petits ruminants in Chhattisgarh state, India. Transbound. Emerg. Dis. 2019, 66, 1349–1359. [Google Scholar] [CrossRef]
- Njue, S.; Saeed, K.; Maloo, S.; Muchai, J.; Biaou, C.; Tetu, K. Sero-prevalence study to determine the effectiveness of Peste de Petits Ruminants vaccination in Somalia. Pastoralism 2018, 8, 17. [Google Scholar] [CrossRef]
- Domenech, J.; Bourzat, D.; Lancelot, R. Development of A Pilot Strategy for PPR Control. 2018. Available online: https://oiebulletin.com/?panorama=development-of-a-pilot-strategy-for-ppr-control (accessed on 17 November 2020).
| Storage | Standard Cold Chain | Room | Field |
|---|---|---|---|
| Temperature | 2–8 °C | 25 °C | 40 °C |
| Time Period | 2 years | 10 days | 5 days |
| Sub-Regions | No. of Outbreaks | Proportion of Total Reported Outbreaks Globally |
|---|---|---|
| Southern Africa | 171 | 1.3% |
| Central Africa | 199 | 1.6% |
| Western Africa | 1912 | 15.0% |
| Eastern Africa | 485 | 3.8% |
| Northern Africa | 394 | 3.1% |
| Middle East | 7710 | 60.4% |
| West Eurasia | 405 | 3.2% |
| South Asia | 1386 | 10.9% |
| East Asia | 58 | 0.5% |
| Countries not included in the Nine sub-regions | 37 | 0.3% |
| Country | 2010 | 2011 | 2012 | 2013 | 2014 | 2015 | 2016 | 2017 | 2018 |
|---|---|---|---|---|---|---|---|---|---|
| Eritrea | 7.7 | ||||||||
| Iraq | 73.5 | 77.6 | 91.0 | 89.8 | 78.9 | 78.5 | 67.8 | ||
| Kazakhstan | 57.0 | ||||||||
| Tajikistan | 16.0 | 8.6 | 3.2 | 5.2 | 21.9 | 10.0 | |||
| Turkey | 44.7 | 82.1 | 77.9 | 79.0 | 69.3 | ||||
| South Sudan | 1.2 | 0.8 | 0.9 | 1.5 | |||||
| Sudan | 15.5 | 12.8 | 39.7 | ||||||
| Uganda | 10.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, H.; Njeumi, F.; Parida, S.; Benfield, C.T.O. Progress towards Eradication of Peste des Petits Ruminants through Vaccination. Viruses 2021, 13, 59. https://doi.org/10.3390/v13010059
Zhao H, Njeumi F, Parida S, Benfield CTO. Progress towards Eradication of Peste des Petits Ruminants through Vaccination. Viruses. 2021; 13(1):59. https://doi.org/10.3390/v13010059
Chicago/Turabian StyleZhao, Hang, Felix Njeumi, Satya Parida, and Camilla T. O. Benfield. 2021. "Progress towards Eradication of Peste des Petits Ruminants through Vaccination" Viruses 13, no. 1: 59. https://doi.org/10.3390/v13010059
APA StyleZhao, H., Njeumi, F., Parida, S., & Benfield, C. T. O. (2021). Progress towards Eradication of Peste des Petits Ruminants through Vaccination. Viruses, 13(1), 59. https://doi.org/10.3390/v13010059







