Effect of Oral Infection of Mayaro Virus on Fitness Correlates and Expression of Immune Related Genes in Aedes aegypti
Abstract
1. Introduction
2. Materials and Methods
2.1. Mosquito Populations and Rearing
2.2. Mayaro Virus Isolate and Propagation
2.3. Infection Study
2.4. Viral RNA Isolation and qRT-PCR
2.5. Statistical Analyses
2.6. Gene Expression
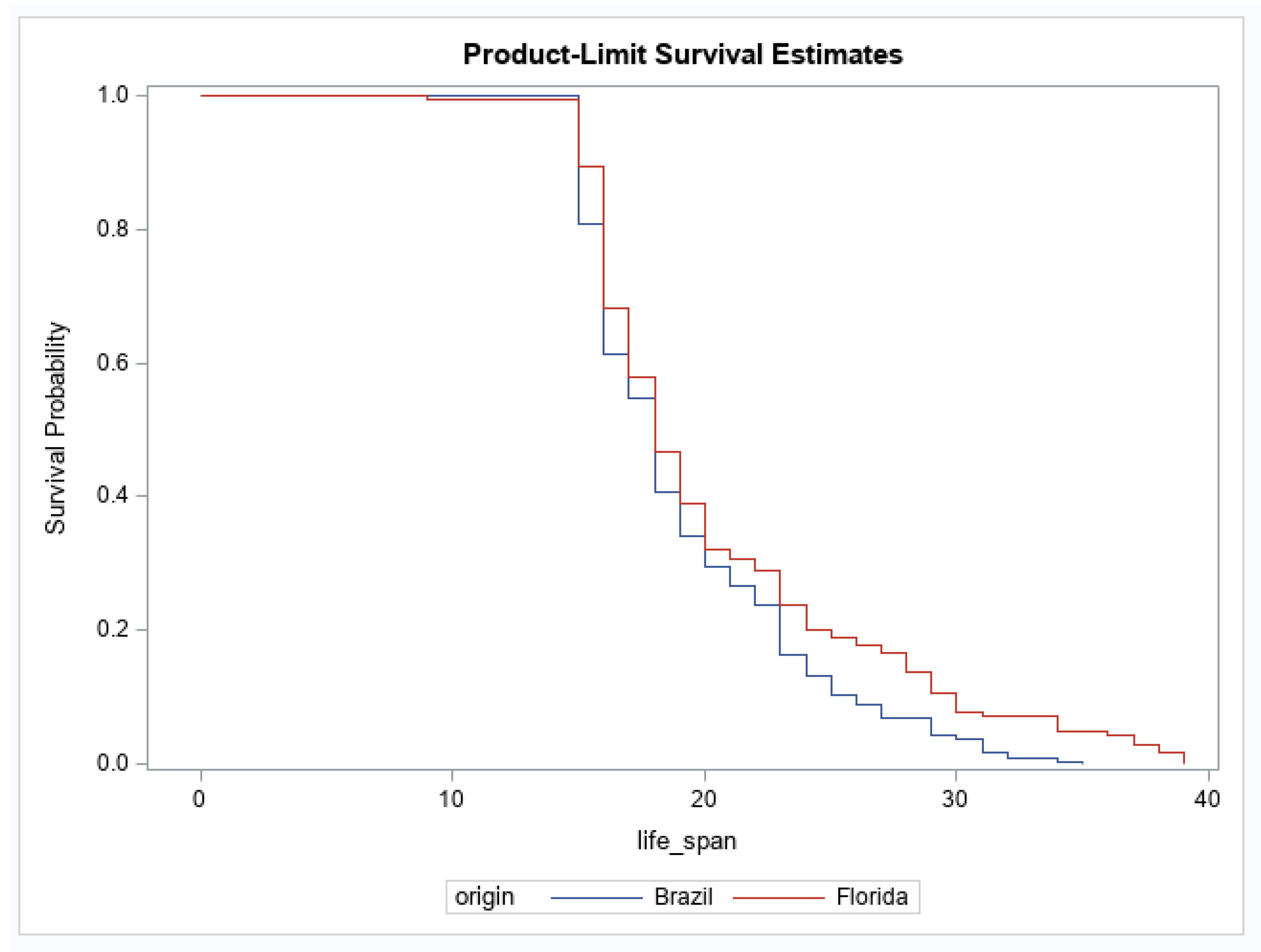
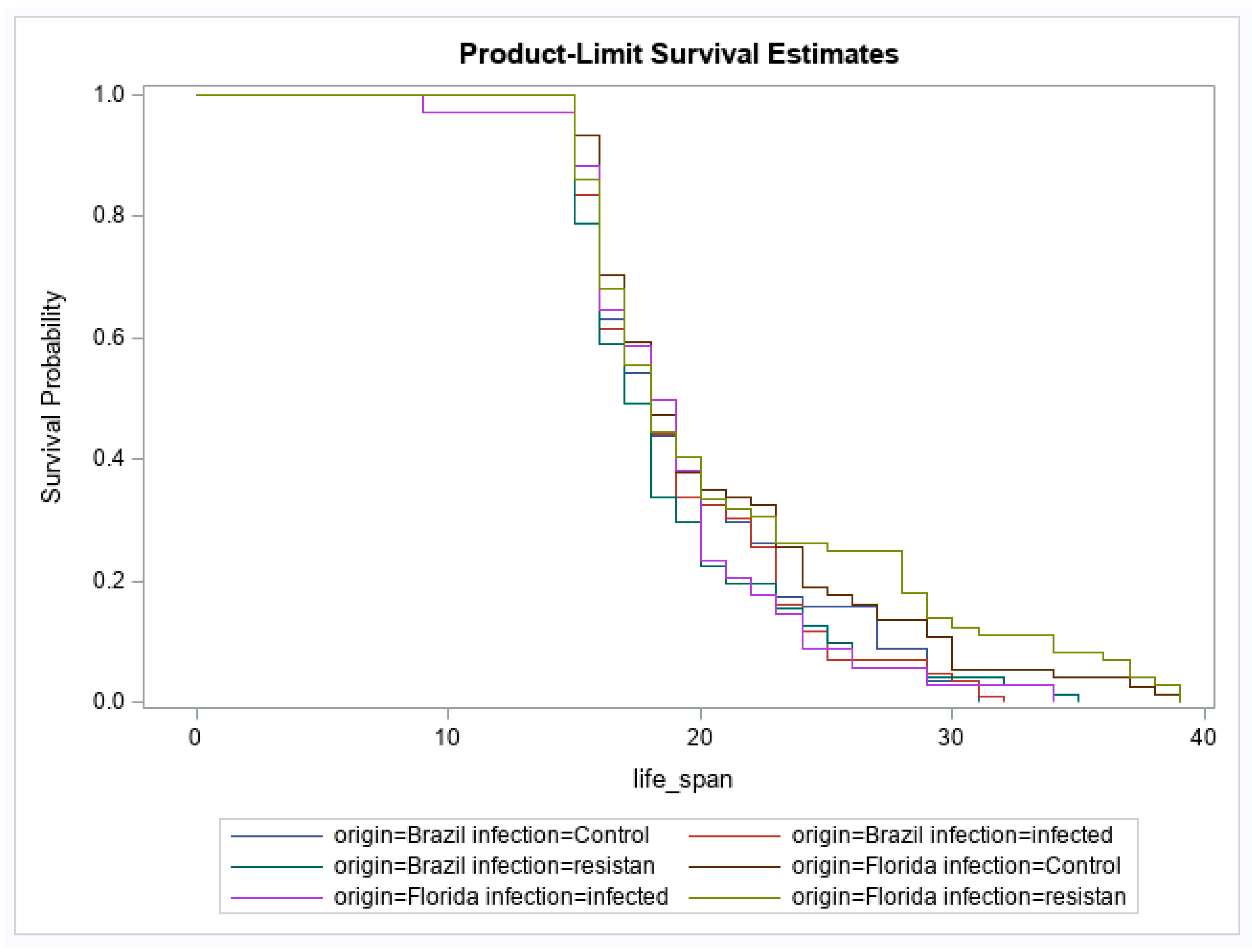
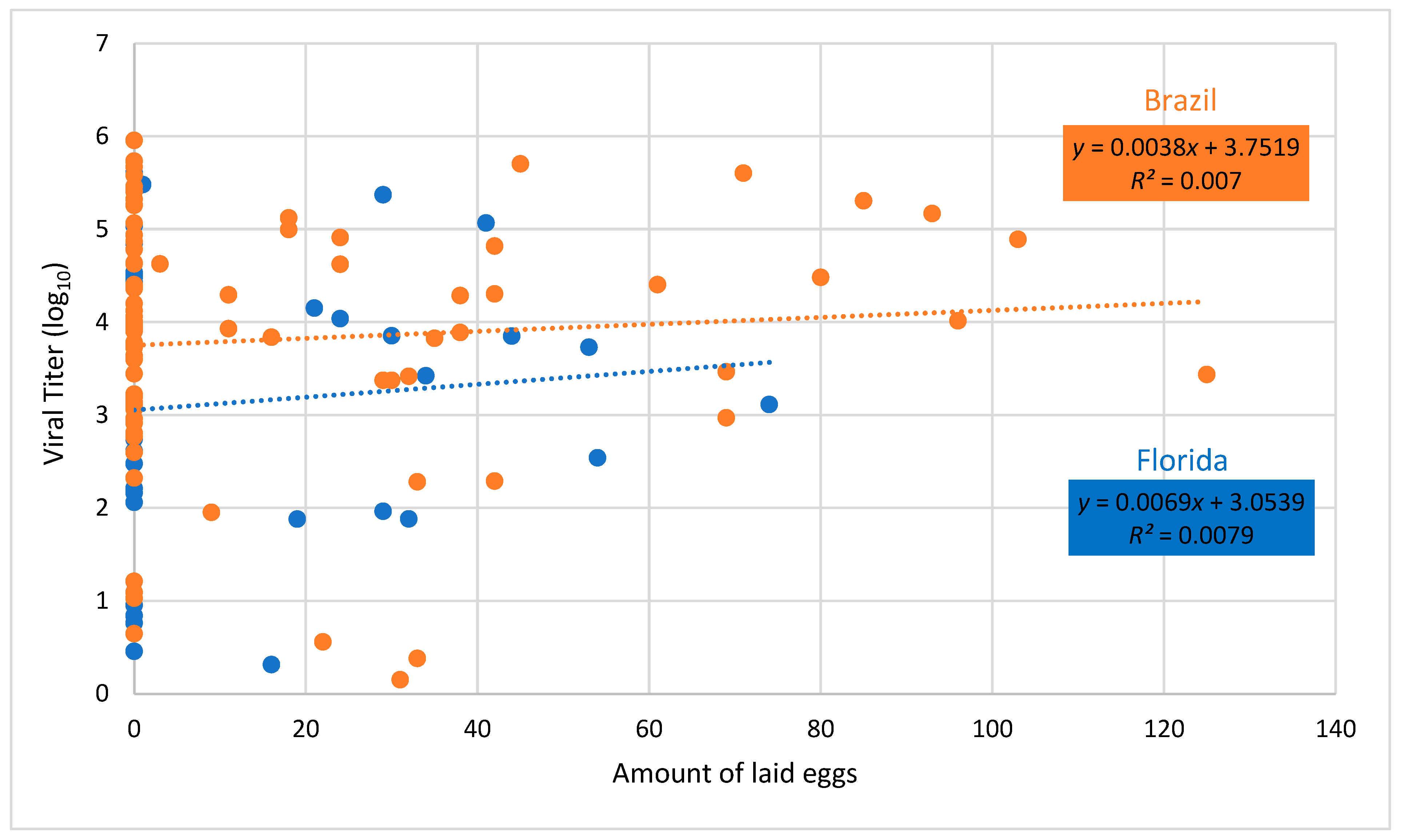
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Anderson, C.R.; Downs, W.G.; Wattley, G.H.; Ahin, N.W.; Reese, A.A. Mayaro virus: A new human disease agent. II. Isolation from blood of patients in Trinidad. B.W.I. Am. J. Trop. Med. Hyg. 1957, 6, 1012–1016. [Google Scholar] [CrossRef]
- Tesh, R.B.; Watts, D.M.; Russell, K.L.; Damodaran, C.; Calampa, C.; Cabezas, C.; Ramirez, G.; Vasquez, B.; Hayes, C.G.; Rossi, C.A.; et al. Mayaro virus disease: An emerging mosquito-borne zoonosis in tropical South America. Clin. Infect. Dis. 1999, 28, 67–73. [Google Scholar] [CrossRef]
- Lednicky, J.; Beau de Rochars, V.M.; Elbadry, M.; Loeb, J.; Telisma, T.; Chavannes, S.; Anilis, G.; Cella, E.; Ciccozzi, M.; Okech, B.; et al. Mayaro virus in child with acute febrile illness, Haiti, 2015. Emerg. Infect. Dis. 2016, 22, 2000–2002. [Google Scholar] [CrossRef]
- Auguste, A.J.; Liria, J.; Forrester, N.L.; Giambalvo, D.; Moncada, M.; Long, K.C.; Morón, D.; de Manzione, N.; Tesh, R.B.; Halsey, E.S.; et al. Evolutionary and ecological characterization of Mayaro virus strains isolated during an outbreak, Venezuela, 2010. Emerg. Infect. Dis. 2015, 21, 1742–1750. [Google Scholar] [CrossRef]
- Coimbra, T.L.M.; Santos, C.L.S.; Suzuki, A.; Petrella, S.M.C.; Bisordi, I.; Nagamori, A.H.; Marti, A.T.; Santos, R.N.; Fialho, D.M.; Lavigne, S.; et al. Mayaro virus: Imported cases of human infection in São Paulo State, Brazil. Rev. Inst. Med. Trop. S. Paulo 2007, 49, 221–224. [Google Scholar] [CrossRef]
- Azevedo, R.S.; Silva, E.V.; Carvalho, V.L.; Rodrigues, S.G.; Neto, J.P.N.; Monteiro, H.A.O.; Peixoto, V.S.; Chiang, J.O.; Nunes, M.R.T.; Vasconcelos, P.F.C. Mayaro fever virus, Brazilian Amazon. Emerg. Infect. Dis. 2009, 15, 1830–1832. [Google Scholar] [CrossRef]
- Zuchi, N.; da Silva Heinen, L.B.; dos Santos, M.A.M.; Pereira, F.C.; Slhessarenko, R.D. Molecular detection of Mayaro virus during a dengue outbreak in the state of Mato Grosso, Central-West Brazil. Mem. Inst. Oswaldo Cruz 2014, 109, 820–823. [Google Scholar] [CrossRef] [PubMed]
- Brunini, S.; França, D.D.S.; Silva, J.B.; Silva, L.N.; Silva, F.P.A.; Spadoni, M.; Rezza, G. High frequency of Mayaro virus IgM among febrile patients, Central Brazil. Emerg. Infect. Dis. 2017, 23, 1025–1026. [Google Scholar] [CrossRef] [PubMed]
- Hoch, A.L.; Peterson, N.E.; LeDuc, J.W.; Pinheiro, F.P. An outbreak of Mayaro virus disease in Belterra, Brazil. III. Entomological and ecological studies. Am. J. Trop. Med. Hyg. 1981, 30, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Hassing, R.-J.; Leparc-Goffart, I.; Blank, S.N.; Thevarayan, S.; Tolou, H.; van Doornum, G.; van Genderen, P.J. Imported Mayaro virus infection in the Netherlands. J. Infect. 2010, 61, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, F.P.; LeDuc, J.W. Mayaro virus disease. In The Arboviruses: Epidemiology and Ecology; Monath, T.P., Ed.; CRC Press: Boca Raton, FL, USA, 1986; pp. 137–150. [Google Scholar]
- Groot, H.; Morales, A.; Vidales, H. Virus isolations from forest mosquitoes in San Vicente de Chucuri, Colombia. Am. J. Trop. Med. Hyg. 397–402. [CrossRef]
- Karabatsos, N. International Catalogue of Arboviruses Including Certain Other Viruses of Vertebrates; American Society of Tropical Medicine and Hygiene: San Antonio, TX, USA, 1985. [Google Scholar]
- Alencar, J.; Lorosa, E.S.; Dégallier, N.; Serra-Freire, N.M.; Pancheco, J.B.; Guimarães, A.E. Feeding patterns of Haemagogus janthinomys (Diptera: Culicidae) in different regions of Brazil. J. Med. Entomol. 2005, 42, 981–985. [Google Scholar] [CrossRef]
- Weaver, S.C.; Reisen, W.K. Present and future arboviral threats. Antivir. Res. 2010, 85, 328–345. [Google Scholar] [CrossRef] [PubMed]
- Long, K.C.; Ziegler, S.A.; Thangamani, S.; Hausser, N.L.; Kochel, T.J.; Higgs, S.; Tesh, R.B. Experimental transmission of Mayaro virus by Aedes aegypti. Am. J. Trop. Med. Hyg. 2011, 85, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Wiggins, K.; Eastmond, B.; Alto, B.W. Transmission potential of Mayaro virus in Florida Aedes aegypti and Ae. albopictus. Med. Vet. Entomol. 2018, 32, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.C.; Francy, D.B. Laboratory studies of a Brazilian strain of Aedes albopictus as a potential vector of Mayaro and Oropouche viruses. J. Am. Mosq. Control Assoc. 1991, 7, 89–93. [Google Scholar] [PubMed]
- Weaver, S.C.; Scott, T.W.; Lorenz, L.H.; Lerdthusnee, K.; Romoser, W.S. Togavirus-associated pathologic changes in the midgut of a natural mosquito vector. J. Virol. 1988, 62, 2083–2090. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, R.; Scott, T.W. Apoptosis in mosquito midgut epithelia associated with West Nile virus infection. Apoptosis 2006, 11, 1643–1651. [Google Scholar] [CrossRef]
- Mims, C.A.; Day, M.F.; Marshall, I.D. Cytopathic effect of Semliki Forest virus in the mosquitoes Aedes aegypti. Am. J. Trop. Med. Hyg. 1966, 15, 775–784. [Google Scholar] [CrossRef]
- Girard, Y.A.; Popov, V.; Wen, J.; Han, V.; Higgs, S. Ultrastructural study of West Nile virus pathogenesis in Culex pipiens quinquefasciatus (Diptera: Culicidae). J. Med. Entomol. 2005, 42, 429–444. [Google Scholar] [CrossRef]
- Ahmed, A.M.; Baggott, S.L.; Hurd, H. The costs of mounting an immune response are reflected in the reproductive fitness of the mosquito Anopheles gambiae. Oikos 2002, 97, 371–377. [Google Scholar] [CrossRef]
- Schwartz, A.; Koella, J.C. The cost of immunity in the yellow fever mosquito, Aedes aegypti depends on immune activation. J. Evol. Biol. 2004, 17, 834–840. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.L.; Dimopoulos, G. The Toll immune signaling pathway control conserved anti-dengue defenses across diverse Ae. aegypti strains and against multiple dengue virus serotypes. Dev. Comp. Immunol. 2010, 34, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Ciota, A.T.; Styer, L.M.; Meola, M.A.; Kramer, L.D. The costs of infection and resistance as determinants of West Nile virus susceptibility in Culex mosquitoes. BMC Ecol. 2011, 11, 23. [Google Scholar] [CrossRef]
- Maciel-de-Freitas, R.; Koella, J.C.; Lourenço-de-Oliveira, R. Lower survival rate, longevity and fecundity of Aedes aegypti (Diptera: Culicidae) females orally challenged with dengue virus serotype 2. Trans. Roy. Soc. Trop. Med. Hyg. 2011, 105, 452–458. [Google Scholar] [CrossRef]
- Faran, M.E.; Turell, M.J.; Romoser, W.S.; Routier, R.G.; Gibbs, P.H.; Cannon, T.L.; Bailey, C.L. Reduced survival of adult Culex pipiens infected with Rift Valley fever virus. Am. J. Trop. Med. Hyg. 1987, 37, 403–409. [Google Scholar] [CrossRef]
- Scott, T.W.; Lorenz, L.H. Reduction of Culiseta melanura fitness by eastern equine encephalomyelitis virus. Am. J. Trop. Med. Hyg. 1998, 59, 341–346. [Google Scholar] [CrossRef]
- Moncayo, A.C.; Edman, J.D.; Turell, M.J. Effect of eastern equine encephalomyelitis virus on the survival of Aedes albopictus, Anopheles quadrimaculatus, and Coquillettidia perturbans (Diptera: Culicidae). J. Med. Entomol. 2000, 37, 701–706. [Google Scholar] [CrossRef]
- Styer, L.M.; Meola, M.A.; Kramer, L.D. West Nile virus infection decreases fecundity of Culex tarsalis females. J. Med. Entomol. 2007, 44, 1074–1085. [Google Scholar] [CrossRef]
- Lambrechts, L.; Scott, T.W. Mode of transmission and the evolution of arbovirus virulence in mosquito vectors. Proc. R. Soc. B 2009, 276, 1369–1378. [Google Scholar] [CrossRef]
- Gabitzsch, E.S.; Blair, C.D.; Beaty, B.J. Effect of La Crosse virus on insemination rates in female Aedes triseriatus. J. Med. Entomol. 2006, 43, 850–852. [Google Scholar] [CrossRef] [PubMed]
- Reese, S.M.; Beaty, M.K.; Gabitzsch, E.; Blair, C.D.; Beaty, B.J. Aedes triseriatus females transovarially infected with La Crosse virus mate more efficiently than uninfected mosquitoes. J. Med. Entomol. 2009, 46, 1152–1158. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Binn, L.N.; Harrison, V.R.; Randall, R. Patterns of viremia and antibody observed in Rhesus monkeys inoculated with chikungunya and other serologically related Group A arboviruses. Am. J. Trop. Med. Hyg. 1967, 16, 782–785. [Google Scholar] [CrossRef]
- Pinheiro, F.P.; Freitas, R.B.; Travassos da Rosa, J.F.; Gabbay, Y.B.; Mello, W.A.; LeDuc, J.W. An outbreak of Mayaro virus disease in Belterra, Brazil. I. Clinical and Virological Findings. Am. J. Trop. Med. Hyg. 1981, 30, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Buckner, E.A.; Alto, B.W.; Lounibos, L.P. Vertical transmission of Key West dengue-1 virus by Aedes aegypti and Aedes albopictus (Diptera: Culicidae) mosquitoes from Florida. J. Med. Entomol. 2013, 50, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Buckner, E.A.; Alto, B.W.; Lounibos, L.P. Larval temperature-food effects on adult mosquito infection and vertical transmission of dengue-1 virus. J. Med. Entomol. 2016, 53, 91–98. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bustin, S.A. Absolute quantification of mRNA using real-time reverse transcription polymerase chain reaction assays. J. Mol. Endocrinol. 2000, 25, 169–193. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.; Civana, A.; Acevedo, C.; Smartt, C.T. Transcriptomics of differential vector competence: West Nile virus infection in two populations of Culex pipiens quinquefasciatus linked to ovary development. BMC Genomics 2014, 15, 513. [Google Scholar] [CrossRef]
- Carissimo, G.; Pondeville, E.; McFarlane, M.; Dietrich, I.; Mitri, C.; Bischoff, E.; Antoniewski, C.; Bourgouin, C.; Failloux, A.-B.; Kohl, A.; et al. Antiviral immunity of Anopheles gambiae is highly compartmentalized, with distinct roles for RNA interference and gut microbiota. Proc. Natl. Acad. Sci. USA 2015, 112, E176–E185. [Google Scholar] [CrossRef]
- Sanders, H.R.; Foy, B.D.; Evans, A.M.; Ross, L.S.; Beaty, B.J.; Olson, K.E.; Gill, S.S. Sindbis virus induces transport processes and alters expression of innate immunity pathway genes in the midgut of the disease vector, Aedes aegypti. Insect Biochem. Mol. Biol. 2005, 35, 1293–1307. [Google Scholar] [CrossRef]
- Sim, S.; Ramirez, J.L.; Dimopoulos, G. Dengue virus infection of the Aedes aegypti salivary gland and chemosensory apparatus induces genes that modulate infection and blood-feeding behavior. PLoS Pathog. 2012, 8, e1002631. [Google Scholar] [CrossRef] [PubMed]
- Xi, Z.; Ramirez, J.L.; Dimopoulos, G. The Aedes aegypti toll pathway controls dengue virus infection. PLoS Pathog. 2008, 4, e1000098. [Google Scholar] [CrossRef] [PubMed]
- Smartt, C.T.; Shin, D.; Alto, B.W. Dengue serotype-specific immune response in Aedes aegypti and Aedes albopictus. Mem. Inst. Oswaldo Cruz. 2017, 112, 829–837. [Google Scholar] [CrossRef]
- Vega-Rúa, A.; Zouache, K.; Girod, R.; Failloux, A.-B.; Lourenço-de-Oliveira, R. High level of vector competence of Aedes aegypti and Aedes albopictus from ten American countries as a crucial factor in the spread of Chikungunya virus. J. Virol. 2014, 88, 6294–6306. [Google Scholar]
- Honório, N.A.; Wiggins, K.; Câmara, D.C.P.; Eastmond, B.; Alto, B.W. Chikunguny virus competency of Brazilian and Florida mosquito vectors. PloS Negl. Trop. Dis. 2018, 12, e0006521. [Google Scholar] [CrossRef]
- Alto, B.W.; Wiggins, K.; Eastmond, B.; Velez, D.; Lounibos, L.P.; Lord, C.C. Transmission risk of two chikungunya lineages by invasive mosquito vectors from Florida and the Dominican Republic. PLoS Negl. Trop. Dis. 2017, 11, e0005724. [Google Scholar] [CrossRef]
- Tabachnick, W.J.; Wallis, G.P.; Aitken, T.H.G.; Miller, B.R.; Amato, G.D.; Lorenz, L.; Powell, J.R.; Beaty, B.J. Oral infection of Aedes aegypti with yellow fever virus: Geographic variation and genetic considerations. Am. J. Trop. Med. Hyg. 1985, 34, 1219–1224. [Google Scholar] [CrossRef]
- Gubler, D.J.; Nalim, S.; Tan, R.; Saipan, H.; Saroso, J.S. Variation in susceptibility to oral infection with dengue viruses among geographic strains of Aedes aegypti. Am. J. Trop. Med. Hyg. 1979, 28, 1045–1052. [Google Scholar] [CrossRef]
- Bennett, K.E.; Olson, K.W.; Munoz, M.L.; Bernandez-Salas, I.; Farfan-ale, J.A.; Higgs, S.; Black, W.C., 4th; Beaty, B.J. Variation in vector competence for dengue 2 virus among 24 collections of Aedes aegypti from Mexico and the United States. Am. J. Trop. Med. Hyg. 2002, 67, 85–92. [Google Scholar] [CrossRef]
- Failloux, A.-B.; Vazeille, M.; Rodhain, F. Geographic genetic variation in populations of the dengue virus vector Aedes aegypti. J. Mol. Evol. 2002, 55, 653–663. [Google Scholar] [CrossRef]
- Gonçlaves, C.M.; Melo, F.F.; Bezerra, J.M.T.; Chaves, B.A.; Silva, B.M.; Silva, L.D.; Pessanha, J.E.M.; Arias, J.R.; Secundino, N.F.C.; Norris, D.E.; et al. Distinct variation in vector competence among nine field populations of Aedes aegypti form a Brazilian dengue-endemic risk city. Parasit. Vectors 2014, 7, 320. [Google Scholar] [CrossRef] [PubMed]
- Boromisa, R.D.; Rai, K.S.; Grimstad, P.R. Variation in the vector competence of geographic strains of Aedes albopictus for dengue 1 virus. J. Am. Mosq. Control Assoc. 1987, 3, 378–386. [Google Scholar]
- Terradas, G.; McGraw, E.A. Using genetic variation in Aedes aegypti to identify candidate anti-dengue virus genes. BMC Infect. Dis. 2019, 19, 580. [Google Scholar] [CrossRef]
- Mahmood, F.; Reisen, W.K.; Chiles, R.E.; Fang, Y. Western equine encephalomyelitis virus infection affects the life table characteristics of Culex tarsalis (Diptera: Culicidae). J. Med. Entomol. 2004, 41, 982–986. [Google Scholar] [CrossRef] [PubMed]
- Padilha, K.P.; Resck, M.E.B.; da Cunha, A.T.; Teles-de-Freitas, R.; Campos, S.S.; Sorgine, M.H.F.; Lourenço-de-Oliveira, R.; Farneisi, L.C.; Bruno, R.V. Zika infection decreases Aedes aegypti locomotor activity but does not influence egg production or viability. Mem. Inst. Oswaldo Cruz 2018, 113, e180290. [Google Scholar] [CrossRef] [PubMed]
- da Silveira, I.D.; Petersen, M.T.; Sylvestre, G.; Garcia, G.A.; David, M.R.; Pavan, M.G.; Maciel-de-Freitas, R. Zika virus infection produces a reduction on Aedes aegypti lifespan but no effects on mosquito fecundity and oviposition success. Front. Microbiol. 2018, 9, 3011. [Google Scholar] [CrossRef] [PubMed]
- Petersen, M.T.; da Silveira, I.D.; Tátila-Ferreira, A.; David, M.R.; Chouin-Carneiro, T.; Van den Wouwer, L.; Maes, L.; Maciel-de-Freitas, R. The impact of the age of first blood meal and Zika virus infection on Aedes aegypti egg production and longevity. PLoS ONE 2018, 13, e0200766. [Google Scholar] [CrossRef]
- Carrington, L.B.; Nguyen, H.L.; Nguyen, N.M.; Duong, T.H.K.; Tuan, T.V.; Giang, N.T.; Tuyet, N.V.; Thi, D.L.; Thi, L.V.; Tran, C.N.; et al. Naturally-acquired dengue virus infections do not reduce short-term survival of infected Aedes aegypti from Ho Chi Minh City, Vietnam. Am. J. Trop. Med. Hyg. 2015, 92, 492–496. [Google Scholar] [CrossRef][Green Version]
- Alto, B.W.; Richards, S.L.; Anderson, S.L.; Lord, C.C. Survival of West Nile virus-challenged Southern house mosquitoes, Culex pipiens quinquefasciatus, in relation to environmental temperatures. J. Vector Ecol. 2014, 39, 123–133. [Google Scholar] [CrossRef]
- Vasilakis, N.; Deardorff, E.R.; Kenney, J.L.; Rossi, S.L.; Hanley, K.A.; Weaver, S.C. Mosquitoes put the brake on arbovirus evolution: Experimental evolution reveals slower mutation accumulation in mosquito than vertebrate cells. PLoS Path. 2009, 5, e1000467. [Google Scholar] [CrossRef]
- Coffey, L.L.; Beeharry, Y.; Bordería, A.V.; Blanc, H.; Vignuzzi, M. Arbovirus high fidelity variant loses fitness in mosquitoes and mice. Proc. Natl. Acad. Sci. USA 2011, 108, 16038–16043. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.R.; DeFoliart, G.R.; Yuill, T.M. Vertical transmission of La Crosse virus (California encephalitis group): Transovarial and filial infection rates in Aedes triseriatus (Diptera: Culicidae). J. Med. Entomol. 1977, 14, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.T.; Gonzalez, J.A.; Reagan, K.L.; Blair, C.D.; Beaty, B.J. Comparative potential of Aedes triseriatus, Ae. albopictus, and Ae. aegypti (Diptera: Culicidae) to transovarially transmit La Crosse Virus. J. Med. Entomol. 2006, 43, 757–761. [Google Scholar] [CrossRef]
- Ewald, P.W. Transmission modes and evolution of the parasitism-mutualism continuum. Ann. N. Y. Acad. Sci. 1987, 503, 295–306. [Google Scholar] [CrossRef]
- Lipsitch, M.S.; Siller, S.; Nowak, M.A. The evolution of virulence in pathogens with vertical and horizontal transmission. Evolution 1996, 50, 1729–1741. [Google Scholar] [CrossRef]
- Patrican, L.A.; DeFoliart, G.R. Lack of adverse effect of transovarially acquired La Crosse virus infection on the reproductive capacity of Aedes triseriatus (Diptera: Culicidae). J. Med. Entomol. 1985, 22, 604–611. [Google Scholar] [CrossRef]
- McGaw, M.M.; Chandler, L.J.; Waseiloski, L.P.; Blair, C.D.; Beaty, B.J. Effect of La Crosse virus infection on overwintering of Aedes triseriatus. Am. J. Trop. Med. Hyg. 1998, 58, 168–175. [Google Scholar] [CrossRef]
- Fragkoudis, R.; Chi, Y.; Siu, R.W.C.; Barry, G.; Attarzadeh-Yazdi, G.; Merits, A.; Nash, A.A.; Fazakerley, J.K.; Kohl, A. Semliki Forest virus strongly reduces mosquito host defence signaling. Insect Mol. Biol. 2008, 17, 647–656. [Google Scholar] [CrossRef]
- Sirisena, P.D.N.N.; Kumar, A.; Sunil, S. Evaluation of Aedes aegypti (Diptera: Culicidae) life table attributes upon chikungunya virus replication reveals impact on egg-laying pathways. J. Med. Entomol. 2018, 55, 1580–1587. [Google Scholar] [CrossRef]
- Hogg, J.C.; Hurd, H. Malaria-induced reduction of fecundity during the first gonotrophic cycle of Anopheles stephensi mosquitoes. Med. Vet. Entomol. 1995, 9, 176–180. [Google Scholar] [CrossRef]
- Jahan, N.; Hurd, H. The effects of infection with Plasmodium yoelii nigeriensis on the reproductive fitness of Anopheles stephensi. Ann. Trop. Med. Parasitol. 1997, 91, 365–369. [Google Scholar] [CrossRef] [PubMed]
- Behura, S.K.; Gomez-Machorro, C.; Harker, B.W.; deBruyn, B.; Lovin, D.D.; Hemme, R.R.; Mori, A.; Romero-Severson, J.; Severson, D.W. Global cross-talk of genes of the mosquito Aedes aegypti in response to dengue virus infection. PLoS Negl. Trop. Dis. 2011, 5, e1385. [Google Scholar] [CrossRef] [PubMed]
- Maciel-de-Freitas, R.; Sylvestre, G.; Gandini, M.; Koella, J.C. The influence of dengue virus serotype-2 infection on Aedes aegypti (Diptera: Culicidae) motivation and avidity to blood feed. PLoS ONE 2013, 8, e65252. [Google Scholar] [CrossRef]
- Reiskind, M.H.; Westbrook, C.J.; Lounibos, L.P. Exposure to chikungunya virus and adult longevity in Aedes aegypti (L.) and Aedes albopictus (Skuse). J. Vector Ecol. 2009, 35, 61–68. [Google Scholar] [CrossRef]

| Gene ID | Description | Primer Sequences |
|---|---|---|
| AAEL007696 | Rel1A | Forward CTCACTCACTCACCGACATAAC |
| Toll pathway | Reverse CAAACTAGGCGCCGAATCATA | |
| AAEL007768 | MYD (MyD88) | Forward GGAGCTTCCTGCAAACCTAA |
| Toll pathway | Reverse TATGGCATCTTCCAGCTTGTC | |
| AAEL007624 | REL2 | Forward AGCTACCGGCATGAGTTATTC |
| IMD pathway | Reverse GCGATACAGATTCCATCGAGAG | |
| AAEL001929 | Spatzle5 | Forward ACCTCCGGTGAATCACAATC |
| Toll pathway | Reverse CAACCATTCCGCTGGACTAA | |
| AAEL027860 | Caspar | Forward TCTGAGAATCGCGAGGAGAT |
| IMD pathway | Reverse GCGGACAGTAGATCCCAATTAC |
| Treatment | Mosquito Strain | No. Tested | No. infected (%) | Body Titer † (Non-Disseminated Infection) | No. Disseminated Infection (%) | Body Titer † (Disseminated Infection) | Leg Titer † |
|---|---|---|---|---|---|---|---|
| Unexposed (control) | Brazil | 57 | 0 (0) | . | . | . | . |
| Unexposed (control) | USA (Florida) | 74 | 0 (0) | . | . | . | . |
| MAYV exposed | Brazil | 157 | 86 (54.78) a | 2.78 a | 59 (68.60) a | 4.30 a | 2.52 a |
| MAYV exposed | USA (Florida) | 106 | 34 (32.08) b | 2.58 a | 14 (41.18) b | 3.98 a | 3.02 a |
| Source | d.f. | χ2 | P |
|---|---|---|---|
| Geographic origin | 1 | 7.55 | 0.0060 |
| Infection status | 2 | 2.06 | 0.3566 |
| Origin × infection status | 5 | 11.28 | 0.0461 |
| Source | d.f. | F | P |
|---|---|---|---|
| Geographic origin | 1 | 0.16 | 0.6870 |
| Infection status | 2 | 6.39 | 0.0019 |
| Origin × infection status | 2 | 0.10 | 0.9070 |
| Error | 387 |
| Brazil (Control) | Brazil (Mayaro) | Florida (Control) | Florida (Mayaro) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Gene | Time (h) | Mean | Stdev | Mean | Stdev | Mean | Stdev | Mean | Stdev |
| caspar | 0 h | 1.007 | 0.138 | 1.001 | 0.054 | 1.019 | 0.237 | 1.003 | 0.088 |
| 4 h | 1.002 | 0.077 | 1.293 | 0.847 | 1.049 | 0.414 | 1.006 | 0.128 | |
| 8 h | 1.006 | 0.135 | 1.015 | 0.206 | 1.001 | 0.041 | 1.008 | 0.160 | |
| 12 h | 1.008 | 0.158 | 1.008 | 0.160 | 1.022 | 0.248 | 1.154 | 0.624 | |
| 24 h | 1.001 | 0.044 | 1.095 | 0.584 | 1.005 | 0.127 | 1.001 | 0.064 | |
| 72 h | 1.001 | 0.041 | 1.003 | 0.100 | 1.002 | 0.071 | 1.003 | 0.101 | |
| 168 h | 2.716 | 2.301 | 1.853 | 1.974 | 0.626 | 0.834 | 0.765 | 1.245 | |
| spz5 | 0 h | 1.012 | 0.191 | 1.137 | 0.615 | 1.005 | 0.129 | 1.007 | 0.144 |
| 4 h | 1.002 | 0.078 | 1.034 | 0.303 | 1.018 | 0.225 | 1.012 | 0.185 | |
| 8 h | 1.083 | 0.543 | 1.203 | 0.753 | 1.001 | 0.045 | 1.018 | 0.234 | |
| 12 h | 1.109 | 0.596 | 1.103 | 0.608 | 1.001 | 0.054 | 1.002 | 0.081 | |
| 24 h | 1.111 | 0.533 | 1.237 | 0.848 | 1.005 | 0.120 | 1.044 | 0.390 | |
| 72 h | 1.110 | 0.557 | 1.072 | 0.510 | 1.056 | 0.447 | 1.128 | 0.663 | |
| 168 h | 2.326 | 2.209 | 0.250 | 0.108 | 1.406 | 1.192 | 0.209 | 0.204 | |
| myd88 | 0 h | 1.015 | 0.213 | 1.002 | 0.069 | 1.014 | 0.205 | 1.008 | 0.154 |
| 4 h | 1.030 | 0.296 | 1.002 | 0.079 | 1.012 | 0.191 | 1.007 | 0.139 | |
| 8 h | 1.012 | 0.192 | 1.073 | 0.519 | 1.014 | 0.213 | 1.109 | 0.634 | |
| 12 h | 1.013 | 0.200 | 1.002 | 0.069 | 1.012 | 0.193 | 1.026 | 0.280 | |
| 24 h | 1.020 | 0.237 | 1.167 | 0.834 | 1.002 | 0.077 | 1.002 | 0.076 | |
| 72 h | 1.061 | 0.402 | 1.010 | 0.171 | 1.003 | 0.087 | 1.064 | 0.433 | |
| 168 h | 0.322 | 0.261 | 0.383 | 0.187 | 1.533 | 1.341 | 1.319 | 1.171 | |
| rel2 | 0 h | 1.031 | 0.312 | 1.012 | 0.191 | 1.004 | 0.106 | 1.004 | 0.105 |
| 4 h | 1.002 | 0.074 | 1.003 | 0.092 | 1.062 | 0.467 | 1.007 | 0.147 | |
| 8 h | 1.022 | 0.268 | 1.016 | 0.218 | 1.003 | 0.093 | 1.022 | 0.262 | |
| 12 h | 1.019 | 0.248 | 1.000 | 0.023 | 1.007 | 0.148 | 1.007 | 0.150 | |
| 24 h | 1.102 | 0.546 | 1.260 | 1.089 | 1.010 | 0.171 | 1.022 | 0.263 | |
| 72 h | 1.026 | 0.292 | 1.006 | 0.129 | 1.009 | 0.167 | 1.025 | 0.270 | |
| 168 h | 2.521 | 3.224 | 2.269 | 1.944 | 1.693 | 1.322 | 3.964 | 3.422 | |
| rel1 | 0 h | 1.161 | 0.685 | 1.031 | 0.323 | 1.094 | 0.592 | 1.129 | 0.586 |
| 4 h | 1.064 | 0.479 | 1.042 | 0.338 | 1.140 | 0.722 | 1.101 | 0.510 | |
| 8 h | 1.060 | 0.397 | 1.289 | 1.086 | 1.098 | 0.613 | 1.245 | 0.850 | |
| 12 h | 1.010 | 0.176 | 1.054 | 0.441 | 1.061 | 0.469 | 1.332 | 1.270 | |
| 24 h | 1.280 | 0.974 | 1.104 | 0.516 | 1.500 | 1.648 | 1.878 | 2.381 | |
| 72 h | 1.083 | 0.511 | 1.403 | 1.112 | 1.016 | 0.218 | 1.115 | 0.582 | |
| 168 h | 1.354 | 1.028 | 0.433 | 0.193 | 1.960 | 2.551 | 0.388 | 0.391 | |
| Brazil | Florida | |||
|---|---|---|---|---|
| Time (h) | Mean | stdev | Mean | stdev |
| 0 h | 4.49 | 4.14 | 4.54 | 4.14 |
| 4 h | 4.41 | 4.08 | 4.51 | 4.29 |
| 8 h | 4.36 | 3.95 | 4.34 | 4.04 |
| 12 h | 4.07 | 3.65 | 4.50 | 4.09 |
| 24 h | 3.90 | 3.62 | 3.82 | 3.48 |
| 72 h | 4.98 | 4.94 | 2.90 | 2.74 |
| 168 h | 3.49 | 3.54 | 1.84 | 1.92 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alto, B.W.; Civana, A.; Wiggins, K.; Eastmond, B.; Shin, D. Effect of Oral Infection of Mayaro Virus on Fitness Correlates and Expression of Immune Related Genes in Aedes aegypti. Viruses 2020, 12, 719. https://doi.org/10.3390/v12070719
Alto BW, Civana A, Wiggins K, Eastmond B, Shin D. Effect of Oral Infection of Mayaro Virus on Fitness Correlates and Expression of Immune Related Genes in Aedes aegypti. Viruses. 2020; 12(7):719. https://doi.org/10.3390/v12070719
Chicago/Turabian StyleAlto, Barry W., Ayse Civana, Keenan Wiggins, Bradley Eastmond, and Dongyoung Shin. 2020. "Effect of Oral Infection of Mayaro Virus on Fitness Correlates and Expression of Immune Related Genes in Aedes aegypti" Viruses 12, no. 7: 719. https://doi.org/10.3390/v12070719
APA StyleAlto, B. W., Civana, A., Wiggins, K., Eastmond, B., & Shin, D. (2020). Effect of Oral Infection of Mayaro Virus on Fitness Correlates and Expression of Immune Related Genes in Aedes aegypti. Viruses, 12(7), 719. https://doi.org/10.3390/v12070719





