Identification of a Neutralizing Monoclonal Antibody That Recognizes a Unique Epitope on Domain III of the Envelope Protein of Tembusu Virus
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Cells, Virus and Antiserum
2.3. Preparation of rEDIII Protein
2.4. MAb Preparation
2.5. MAb Isotyping
2.6. Indirect ELISA
2.7. Detection of Reactivity of MAb to EDIII
2.8. Indirect Immunofluorescence (IIF) Assay
2.9. Neutralization Assay
2.10. PRNT
2.11. Identification of the Epitope Recognized by 12F11
2.12. Pre- and Post-Adsorption Inhibition Assays
2.13. Statistical Analysis
3. Results
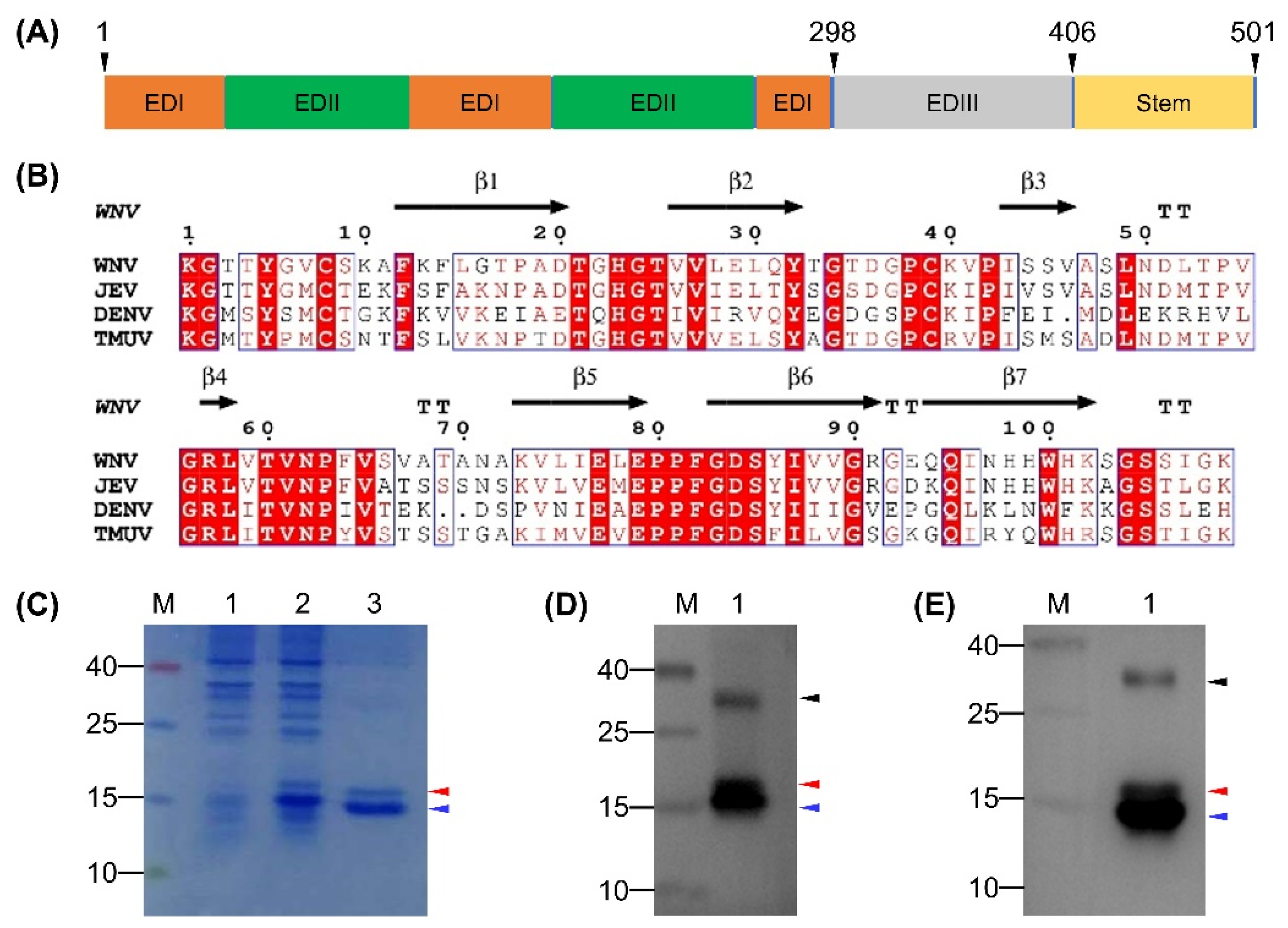
3.1. Expression and Characterization of the rEDIII Protein
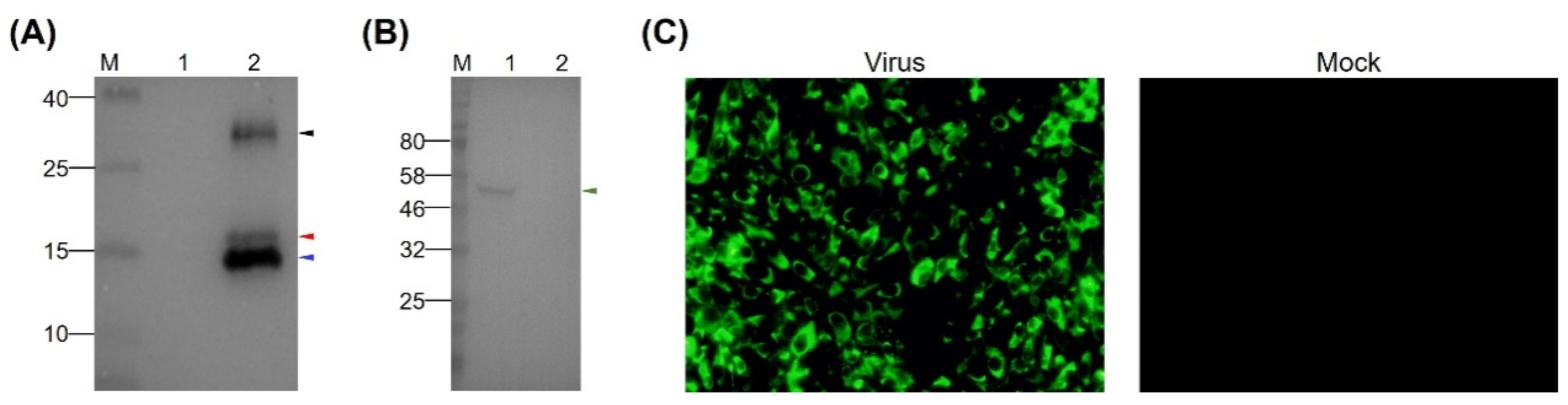
3.2. Preparation of MAb Directed Against TMUV EDIII
3.3. Analysis of Neutralizing Activity of MAb 12F11
3.4. Identification of Epitope Recognized by MAb 12F11
3.5. Inhibitory Effect of MAb 12F11 on Virus Adsorption and a Step after Virus Adsorption
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kono, Y.; Tsukamoto, K.; Hamid, M.A.; Darus, A.; Lian, T.C.; Sam, L.S.; Yok, C.N.; Di, K.B.; Lim, K.T.; Yamaguchi, S.; et al. Encephalitis and retarded growth of chicks caused by Sitiawan virus, a new isolate belonging to the genus Flavivirus. Am. J. Trop. Med. Hyg. 2000, 63, 94–101. [Google Scholar] [CrossRef]
- Cao, Z.; Zhang, C.; Liu, Y.; Liu, Y.; Ye, W.; Han, J.; Ma, G.; Zhang, D.; Xu, F.; Gao, X.; et al. Tembusu virus in ducks, China. Emerg. Infect. Dis. 2011, 17, 1873–1875. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Li, S.; Hu, X.; Yu, X.; Wang, Y.; Liu, P.; Lu, X.; Zhang, G.; Hu, X.; Liu, D.; et al. Duck egg-drop syndrome caused by BYD virus, a new Tembusu-related flavivirus. PLoS ONE 2011, 6, e18106. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Zhao, Y.; Zhang, X.; Xu, D.; Dai, X.; Teng, Q.; Yan, L.; Zhou, J.; Ji, X.; Zhang, S.; et al. An infectious disease of ducks caused by a newly emerged Tembusu virus strain in mainland China. Virology 2011, 417, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Liu, J.; Jiang, Y.; Zhao, Y.; Li, Q.; Wu, L.; He, X.; Chen, H. The vaccine efficacy of recombinant duck enteritis virus expressing secreted E with or without PrM proteins of duck Tembusu virus. Vaccine 2014, 32, 5271–5277. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Gao, X.; Xiao, Y.; Liu, S.; Peng, S.; Li, X.; Shi, Y.; Zhang, Y.; Yu, L.; Wu, X.; et al. Development of a live attenuated vaccine candidate against duck Tembusu viral disease. Virology 2014, 450–451, 233–242. [Google Scholar] [CrossRef]
- Ma, T.; Liu, Y.; Cheng, J.; Liu, Y.; Fan, W.; Cheng, Z.; Zhu, Q. Liposomes containing recombinant E protein vaccine against duck Tembusu virus in ducks. Vaccine 2016, 34, 2157–2163. [Google Scholar] [CrossRef]
- Sun, L.; Li, Y.; Zhang, Y.; Han, Z.; Xu, Y.; Kong, X.; Liu, S. Adaptation and attenuation of duck Tembusu virus strain Du/CH/LSD/110128 following serial passage in chicken embryos. Clin. Vaccine Immunol. 2014, 21, 1046–1053. [Google Scholar] [CrossRef][Green Version]
- Zou, Z.; Liu, Z.; Jin, M. Efficient strategy to generate a vectored duck enteritis virus delivering envelope of duck Tembusu virus. Viruses 2014, 6, 2428–2443. [Google Scholar] [CrossRef]
- Zhao, D.; Huang, X.; Liu, Y.; Han, K.; Zhang, J.; Yang, J.; Xie, X.; Li, Y. Domain I and II from newly emerging goose tembusu virus envelope protein functions as a dominant-negative inhibitor of virus infectivity. Res. Vet. Sci. 2015, 98, 121–126. [Google Scholar] [CrossRef]
- Zhang, L.; Li, Z.; Zhang, Q.; Sun, M.; Li, S.; Su, W.; Hu, X.; He, W.; Su, J. Efficacy assessment of an inactivated Tembusu virus vaccine candidate in ducks. Res. Vet. Sci. 2017, 110, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ji, Y.; Huang, X.; Fu, Y.; Wei, J.; Cai, X.; Zhu, Q. An adapted duck Tembusu virus induces systemic infection and mediates antibody-dependent disease severity in mice. Virus Res. 2013, 176, 216–222. [Google Scholar] [CrossRef]
- Bai, X.; Lv, R.; Liu, C.; Qiu, N.; He, Y.; Yin, X.; Li, X.; Liu, M.; Zhang, Y. Molecular characterization of a duck Tembusu virus from China. Virus Genes 2013, 47, 478–482. [Google Scholar] [CrossRef]
- Huang, X.; Han, K.; Zhao, D.; Liu, Y.; Zhang, J.; Niu, H.; Zhang, K.; Zhu, J.; Wu, D.; Gao, L.; et al. Identification and molecular characterization of a novel flavivirus isolated from geese in China. Res. Vet. Sci. 2013, 94, 774–780. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.; Lu, H.; Li, S.; Moureau, G.; Deng, Y.Q.; Wang, Y.; Zhang, L.; Jiang, T.; de Lamballerie, X.; Qin, C.F.; et al. Genomic and antigenic characterization of the newly emerging Chinese duck egg-drop syndrome flavivirus: Genomic comparison with Tembusu and Sitiawan viruses. J. Gen. Virol. 2012, 93, 2158–2170. [Google Scholar] [CrossRef] [PubMed]
- Yun, T.; Zhang, D.; Ma, X.; Cao, Z.; Chen, L.; Ni, Z.; Ye, W.; Yu, B.; Hua, J.; Zhang, Y.; et al. Complete genome sequence of a novel flavivirus, duck Tembusu virus, isolated from ducks and geese in china. J. Virol. 2012, 86, 3406–3407. [Google Scholar] [CrossRef]
- Diamond, M.S.; Pierson, T.C.; Fremont, D.H. The structural immunology of antibody protection against West Nile virus. Immunol. Rev. 2008, 225, 212–225. [Google Scholar] [CrossRef]
- Heinz, F.X.; Stiasny, K. Flaviviruses and their antigenic structure. J. Clin. Virol. 2012, 55, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.C.; Fremont, D.H.; Kuhn, R.J.; Diamond, M.S. Structural insights into the mechanisms of antibody-mediated neutralization of flavivirus infection: Implications for vaccine development. Cell Host Microbe 2008, 4, 229–238. [Google Scholar] [CrossRef]
- Modis, Y.; Ogata, S.; Clements, D.; Harrison, S.C. A ligand-binding pocket in the dengue virus envelope glycoprotein. Proc. Natl. Acad. Sci. USA 2003, 100, 6986–6991. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, W.; Ogata, S.; Clements, D.; Strauss, J.H.; Baker, T.S.; Kuhn, R.J.; Rossmann, M.G. Conformational changes of the flavivirus E glycoprotein. Structure 2004, 12, 1607–1618. [Google Scholar] [CrossRef] [PubMed]
- Pierson, T.C.; Diamond, M.S. A game of numbers: The stoichiometry of antibody-mediated neutralization of flavivirus infection. Prog. Mol. Biol. Transl. Sci. 2015, 129, 141–166. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Shaozhou, W.; Zhang, Q.; Li, C.; Qiu, N.; Meng, R.; Liu, M.; Zhang, Y. Characterization of monoclonal antibodies against duck Tembusu virus E protein: An antigen-capture ELISA for the detection of Tembusu virus infection. Arch. Virol. 2015, 160, 757–764. [Google Scholar] [CrossRef]
- Han, K.; Zhao, D.; Liu, Y.; Liu, Q.; Huang, X.; Yang, J.; Bi, K.; Xu, T.; Li, Y. Generation and characterization of a monoclonal antibody against duck Tembusu virus envelope protein. Pol. J. Vet. Sci. 2016, 19, 877–883. [Google Scholar] [CrossRef]
- Li, C.; Bai, X.; Meng, R.; Shaozhou, W.; Zhang, Q.; Hua, R.; Liu, J.H.; Liu, M.; Zhang, Y. Identification of a new broadly cross-reactive epitope within domain III of the duck Tembusu virus E protein. Sci. Rep. 2016, 6, 36288. [Google Scholar] [CrossRef]
- Li, C.; Liu, J.; Shaozhou, W.; Bai, X.; Zhang, Q.; Hua, R.; Liu, J.H.; Liu, M.; Zhang, Y. Epitope identification and application for diagnosis of duck Tembusu virus infections in ducks. Viruses 2016, 8, 306. [Google Scholar] [CrossRef]
- Chen, X.; Li, C.; Lin, W.; Li, T.; Li, X.; Bai, X.; Wulin, S.; Zhang, Q.; Li, S.; Liu, M.; et al. A novel neutralizing antibody targeting a unique cross-reactive epitope on the hi-loop of domain II of the envelope protein protects mice against duck Tembusu virus. J. Immunol. 2020, 204, 1836–1848. [Google Scholar] [CrossRef]
- Marchler-Bauer, A.; Lu, S.; Anderson, J.B.; Chitsaz, F.; Derbyshire, M.K.; DeWeese-Scott, C.; Fong, J.H.; Geer, L.Y.; Geer, R.C.; Gonzales, N.R.; et al. CDD: A Conserved Domain Database for the functional annotation of proteins. Nucleic Acids Res. 2011, 39, D225–D229. [Google Scholar] [CrossRef]
- Köhler, G.; Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 1975, 256, 495–497. [Google Scholar] [CrossRef]
- Wang, X.; Wang, Y.; Xie, X.; Zhang, B.; Zhang, D. Expression of the C-terminal ORF2 protein of duck astrovirus for application in a serological test. J. Virol. Methods 2011, 171, 8–12. [Google Scholar] [CrossRef]
- Cohen, B.J.; Audet, S.; Andrews, N.; Beeler, J. WHO working group on measles plaque reduction neutralization test Plaque reduction neutralization test for measles antibodies: Description of a standardised laboratory method for use in immunogenicity studies of aerosol vaccination. Vaccine 2007, 26, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Hung, S.L.; Lee, P.L.; Chen, H.W.; Chen, L.K.; Kao, C.L.; King, C.C. Analysis of the steps involved in dengue virus entry into host cells. Virology 1999, 257, 156–167. [Google Scholar] [CrossRef] [PubMed]
- Fibriansah, G.; Tan, J.L.; Smith, S.A.; de Alwis, R.; Ng, T.S.; Kostyuchenko, V.A.; Jadi, R.S.; Kukkaro, P.; de Silva, A.M.; Crowe, J.E.; et al. A highly potent human antibody neutralizes dengue virus serotype 3 by binding across three surface proteins. Nat. Commun. 2015, 6, 6341. [Google Scholar] [CrossRef] [PubMed]
- Gromowski, G.D.; Barrett, A.D. Characterization of an antigenic site that contains a dominant, type-specific neutralization determinant on the envelope protein domain III (ED3) of dengue 2 virus. Virology 2007, 366, 349–360. [Google Scholar] [CrossRef]
- Schnabel, D.; Castillo-Robles, J.; Lomeli, H. Protein purification and Western blot detection from single zebrafish embryo. Zebrafish 2019, 16, 505–507. [Google Scholar] [CrossRef] [PubMed]
- Towbin, H.; Staehelin, T.; Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc. Natl. Acad. Sci. USA 1979, 76, 4350–4354. [Google Scholar] [CrossRef]


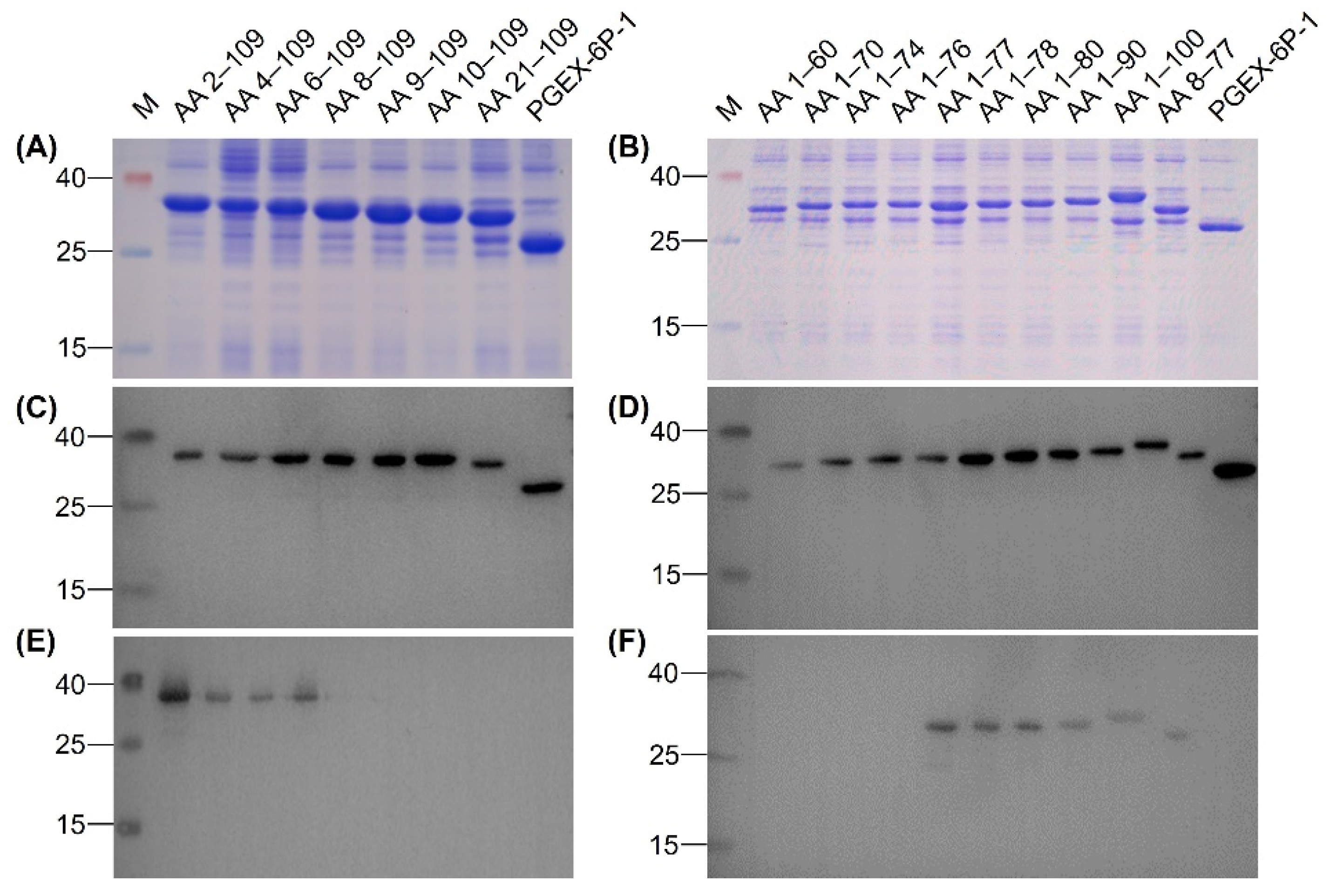

| Primer | Sequence (5′‒3′) d | Recombinant Plasmid e | Mr of Recombinant Protein (kDa) f |
|---|---|---|---|
| 1f | CGCGGATCCAAAGGAATGACCTACCCGATGTGTAGC (BamH I) | / | / |
| 109.1r a | CCGCCTCGAGTTTTTCCAATTGTGCTCCCACTTCTATG | PET-28a-EDIII | 16.7 |
| 100r b | CCGCTCGAGCCACTGGTACCTGATCTGTC (Xho I) | PGEX-6P-1(AA1‒100) | 38.7 |
| 90r b | CCGCTCGAGCCCTACTAAGATGAATGAATCCCCGAATGG | PGEX-6P-1(AA1‒90) | 37.5 |
| 80r b | CCGCTCGAGAGGTTCCACTTCCACCATTATCTTG | PGEX-6P-1(AA1‒80) | 36.4 |
| 70r b | CCGCTCGAGCGTGGAGGAGGTCGACACGTATGGGTTGAC | PGEX-6P-1(AA1‒70) | 35.4 |
| 60r b | CCGCTCGAGTTGTTATCAAGCGTCCAACTGGTGTC | PGEX-6P-1(AA1‒60) | 34.3 |
| 78r b | CCGCTCGAGCACTTCCACCATTATCTTGGCAC | PGEX-6P-1(AA1‒78) | 36.2 |
| 77r b c | CCGCTCGAGTTCCACCATTATCTTGGCACCCGTGGAG | PGEX-6P-1(AA1‒77) | 36.1 |
| 76r b | CCGCTCGAGCACCATTATCTTGGCACCCGTG | PGEX-6P-1(AA1‒76) | 36.0 |
| 74r b | CCGCTCGAGTATCTTGGCACCCGTGGAGGAGGTC | PGEX-6P-1(AA1‒74) | 35.8 |
| 2f c | CGCGGATCCGGAATGACCTACCCGATGTGTAGC | PGEX-6P-1(AA2‒109) | 39.5 |
| 4f c | CGCGGATCCACCTACCCGATGTGTAGCAACAC | PGEX-6P-1(AA4‒109) | 39.3 |
| 6f c | CGCGGATCCCCGATGTGTAGCAACACATTTTCC | PGEX-6P-1(AA6‒109) | 39.0 |
| 8f c | CGCGGATCCTGTAGCAACACATTTTCCCTAGTG | PGEX-6P-1(AA8‒109) | 38.8 |
| 9f c | CGCGGATCCAGCAACACATTTTCCCTAGTGAAG | PGEX-6P-1(AA9‒109) | 38.7 |
| 10f c | CGCGGATCCAACACATTTTCCCTAGTG | PGEX-6P-1(AA10‒109) | 38.6 |
| 21f c | CGCGGATCCACTGGGCATGGCACTGTCGTGG | PGEX-6P-1(AA21‒109) | 37.4 |
| 109.2r | CCGCCTCGAGTTTTCCAATTGTGCTCCCACTTCTATG | / | / |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, S.; Wang, X.; Yang, L.; Lv, J.; Meng, R.; Dai, W.; Li, Q.; Liu, H.; Zhang, B.; Zhang, D. Identification of a Neutralizing Monoclonal Antibody That Recognizes a Unique Epitope on Domain III of the Envelope Protein of Tembusu Virus. Viruses 2020, 12, 647. https://doi.org/10.3390/v12060647
Qu S, Wang X, Yang L, Lv J, Meng R, Dai W, Li Q, Liu H, Zhang B, Zhang D. Identification of a Neutralizing Monoclonal Antibody That Recognizes a Unique Epitope on Domain III of the Envelope Protein of Tembusu Virus. Viruses. 2020; 12(6):647. https://doi.org/10.3390/v12060647
Chicago/Turabian StyleQu, Shenghua, Xiaoyan Wang, Lixin Yang, Junfeng Lv, Runze Meng, Weiqian Dai, Qingxiangzi Li, Huicong Liu, Bing Zhang, and Dabing Zhang. 2020. "Identification of a Neutralizing Monoclonal Antibody That Recognizes a Unique Epitope on Domain III of the Envelope Protein of Tembusu Virus" Viruses 12, no. 6: 647. https://doi.org/10.3390/v12060647
APA StyleQu, S., Wang, X., Yang, L., Lv, J., Meng, R., Dai, W., Li, Q., Liu, H., Zhang, B., & Zhang, D. (2020). Identification of a Neutralizing Monoclonal Antibody That Recognizes a Unique Epitope on Domain III of the Envelope Protein of Tembusu Virus. Viruses, 12(6), 647. https://doi.org/10.3390/v12060647





