Current Trends in Diagnostics of Viral Infections of Unknown Etiology
Abstract
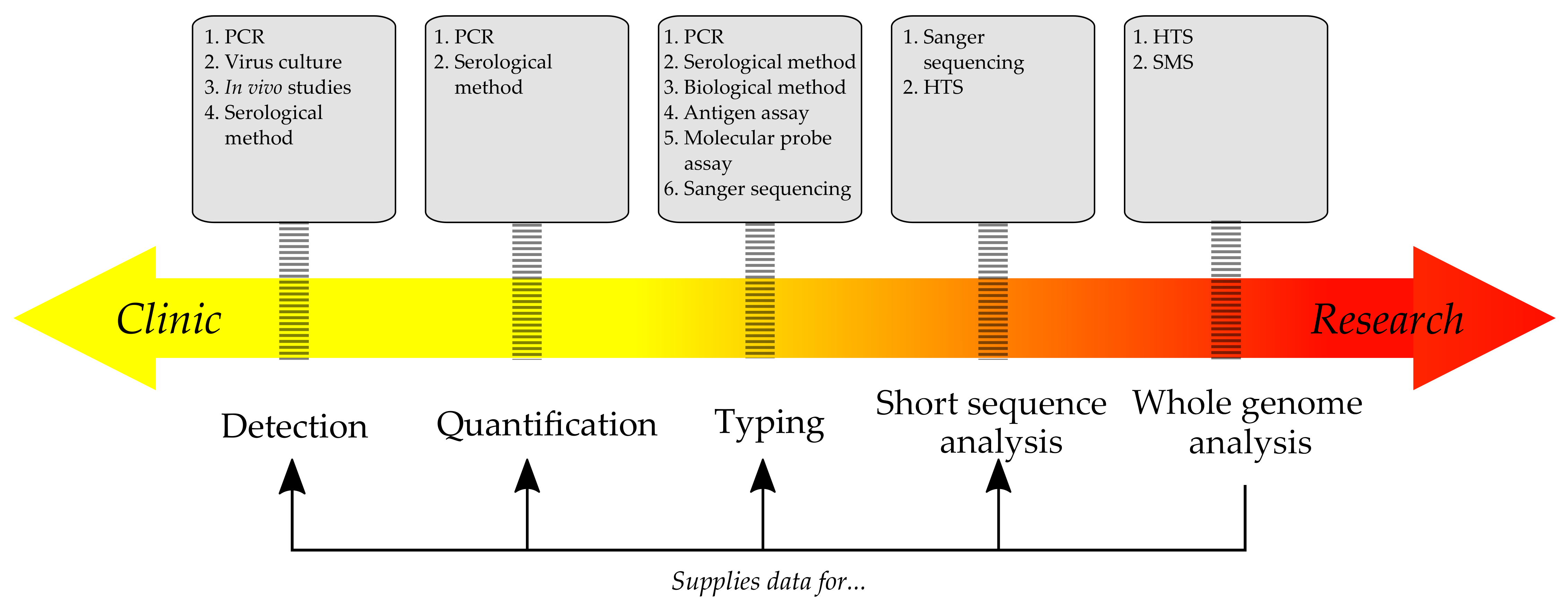
1. Introduction
2. Traditional Methods of Diagnosing Infections
3. Studying Viral Pathogens with High Throughput Sequencing (HTS)
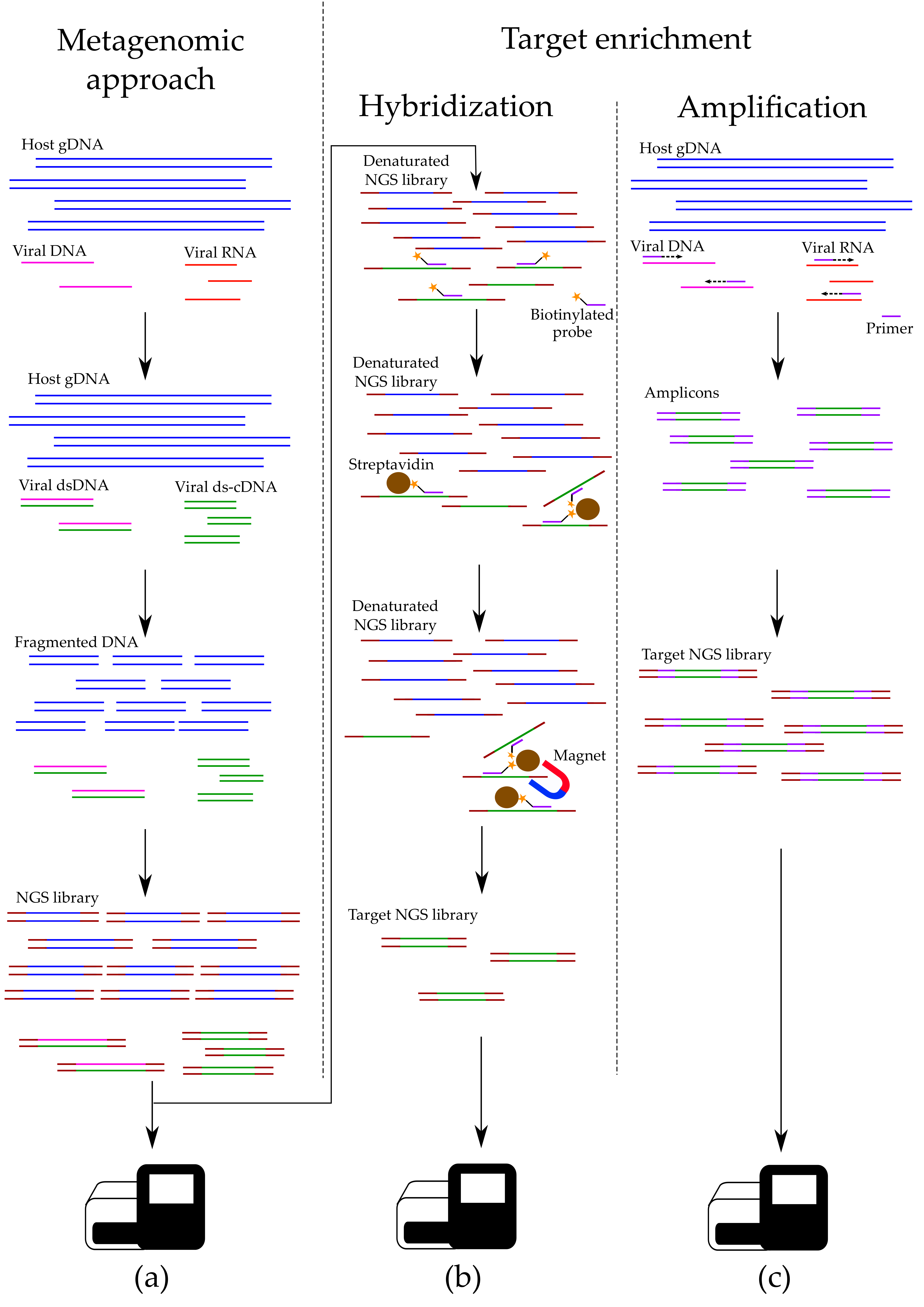
3.1. Metagenomic Approach
3.2. Problems of Metagenomic Approach
3.3. Methods for Improving Sequencing Output
3.3.1. Nucleic Acids Depletion
3.3.2. Hybridization-Based Enrichment
3.3.3. Target Amplification
3.4. Whole Viral Genome Sequencing
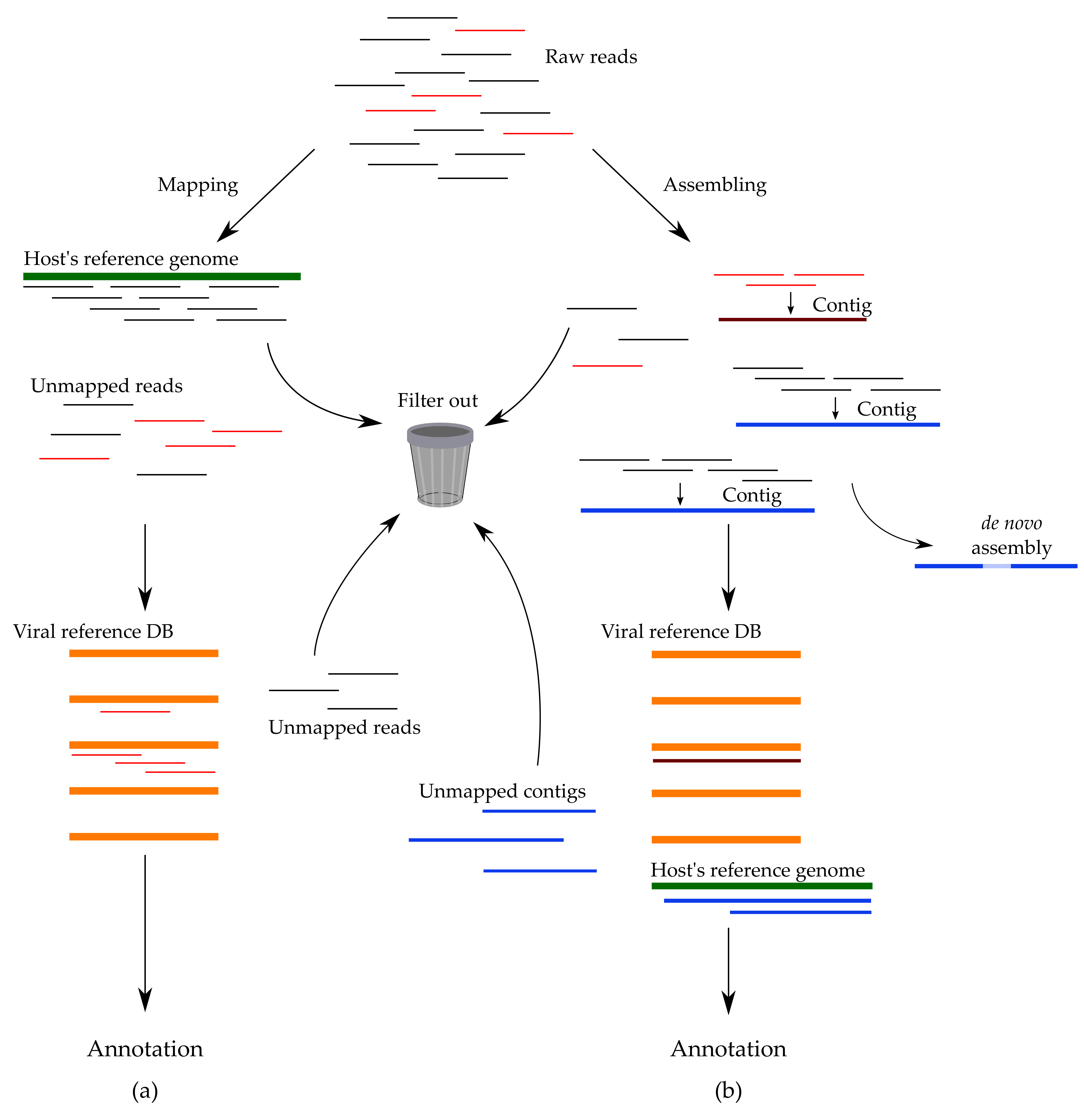
3.5. Methods of Sequencing Data Analysis
4. Long Read Sequencing
5. Obstacles to Overcome in the Nearest Future
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anthony, S.J.; Epstein, J.H.; Murray, K.A.; Navarrete-Macias, I.; Zambrana-Torrelio, C.M.; Solovyov, A.; Ojeda-Flores, R.; Arrigo, N.C.; Islam, A.; Ali Khan, S.; et al. A Strategy to Estimate Unknown Viral Diversity in Mammals. mBio 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Woolhouse, M.; Scott, F.; Hudson, Z.; Howey, R.; Chase-Topping, M. Human viruses: Discovery and emergence. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 2864–2871. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Vijayanand, P.; Wilkins, E.; Woodhead, M. Severe acute respiratory syndrome (SARS): A review. Clin. Med. 2004, 4, 152–160. [Google Scholar] [CrossRef]
- Rather, I.A.; Lone, J.B.; Bajpai, V.K.; Paek, W.K.; Lim, J. Zika Virus: An Emerging Worldwide Threat. Front. Microbiol. 2017, 8, 1417. [Google Scholar] [CrossRef]
- Hui, D.S.; I Azhar, E.; Madani, T.A.; Ntoumi, F.; Kock, R.; Dar, O.; Ippolito, G.; Mchugh, T.D.; Memish, Z.A.; Drosten, C.; et al. The continuing 2019-nCoV epidemic threat of novel coronaviruses to global health—The latest 2019 novel coronavirus outbreak in Wuhan, China. Int. J. Infect. Dis. 2020, 91, 264–266. [Google Scholar] [CrossRef]
- Beal, S.G.; Tremblay, E.E.; Toffel, S.; Velez, L.; Rand, K.H. A Gastrointestinal PCR Panel Improves Clinical Management and Lowers Health Care Costs. J. Clin. Microbiol. 2017, 56, e01457-17. [Google Scholar] [CrossRef]
- Beck, E.T.; Henrickson, K.J. Molecular diagnosis of respiratory viruses. Future Microbiol. 2010, 5, 901–916. [Google Scholar] [CrossRef]
- Schlaberg, R.; Chiu, C.Y.; Miller, S.; Procop, G.W.; Weinstock, G.; the Professional Practice Committee and Committee on Laboratory Practices of the American Society for Microbiology; the Microbiology Resource Committee of the College of American Pathologists. Validation of Metagenomic Next-Generation Sequencing Tests for Universal Pathogen Detection. Arch. Pathol. Lab. Med. 2017, 141, 776–786. [Google Scholar] [CrossRef]
- Arnold, J.C.; Singh, K.K.; Spector, S.A.; Sawyer, M.H. Undiagnosed Respiratory Viruses in Children. Pediatrics 2008, 121, e631–e637. [Google Scholar] [CrossRef]
- Vu, D.-L.; Sabrià, A.; Aregall, N.; Michl, K.; Rodriguez Garrido, V.; Goterris, L.; Bosch, A.; Pintó, R.M.; Guix, S. Novel Human Astroviruses: Prevalence and Association with Common Enteric Viruses in Undiagnosed Gastroenteritis Cases in Spain. Viruses 2019, 11, 585. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, P.; Quan, P.-L.; Lipkin, W. Viral Encephalitis of Unknown Cause: Current Perspective and Recent Advances. Viruses 2017, 9, 138. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Hamid, M.; El-Daly, M.; El-Kafrawy, S.; Mikhail, N.; Strickland, G.T.; Fix, A.D. Comparison of Second- and Third-Generation Enzyme Immunoassays for Detecting Antibodies to Hepatitis C Virus. J. Clin. Microbiol. 2002, 40, 1656–1659. [Google Scholar] [CrossRef] [PubMed]
- Döller, G.; Schuy, W.; Tjhen, K.Y.; Stekeler, B.; Gerth, H.J. Direct detection of influenza virus antigen in nasopharyngeal specimens by direct enzyme immunoassay in comparison with quantitating virus shedding. J. Clin. Microbiol. 1992, 30, 866–869. [Google Scholar] [CrossRef] [PubMed]
- Hammond, G.W.; Smith, S.J.; Noble, G.R. Sensitivity and Specificity of Enzyme Immunoassay for Serodiagnosis of Influenza A Virus Infections. J. Infect. Dis. 1980, 141, 644–651. [Google Scholar] [CrossRef] [PubMed]
- Kasprowicz, V.; Ward, S.M.; Turner, A.; Grammatikos, A.; Nolan, B.E.; Lewis-Ximenez, L.; Sharp, C.; Woodfruff, J.; Fleming, V.M.; Sims, S.; et al. Defining the directionality and quality of influenza virus–specific CD8+ T cell cross-reactivity in individuals infected with hepatitis C virus. J. Clin. Investig. 2008, JCI33082. [Google Scholar] [CrossRef]
- Hoofnagle, A.N.; Wener, M.H. The fundamental flaws of immunoassays and potential solutions using tandem mass spectrometry. J. Immunol. Methods 2009, 347, 3–11. [Google Scholar] [CrossRef]
- Cogswell, F.B.; Bantar, C.E.; Hughes, T.G.; Gu, Y.; Philipp, M.T. Host DNA can interfere with detection of Borrelia burgdorferi in skin biopsy specimens by PCR. J. Clin. Microbiol. 1996, 34, 980–982. [Google Scholar] [CrossRef]
- Reyes, G.R.; Kim, J.P. Sequence-independent, single-primer amplification (SISPA) of complex DNA populations. Mol. Cell. Probes 1991, 5, 473–481. [Google Scholar] [CrossRef]
- Castrignano, S.B.; Nagasse-Sugahara, T.K.; Kisielius, J.J.; Ueda-Ito, M.; Brandão, P.E.; Curti, S.P. Two novel circo-like viruses detected in human feces: Complete genome sequencing and electron microscopy analysis. Virus Res. 2013, 178, 364–373. [Google Scholar] [CrossRef]
- DePew, J.; Zhou, B.; McCorrison, J.M.; Wentworth, D.E.; Purushe, J.; Koroleva, G.; Fouts, D.E. Sequencing viral genomes from a single isolated plaque. Virol. J. 2013, 10, 181. [Google Scholar] [CrossRef] [PubMed]
- Djikeng, A.; Halpin, R.; Kuzmickas, R.; DePasse, J.; Feldblyum, J.; Sengamalay, N.; Afonso, C.; Zhang, X.; Anderson, N.G.; Ghedin, E.; et al. Viral genome sequencing by random priming methods. BMC Genom. 2008, 9, 5. [Google Scholar] [CrossRef] [PubMed]
- Ambrose, H.E.; Clewley, J.P. Virus discovery by sequence-independent genome amplification. Rev. Med. Virol. 2006, 16, 365–383. [Google Scholar] [CrossRef] [PubMed]
- de Vries, M.; Deijs, M.; Canuti, M.; van Schaik, B.D.C.; Faria, N.R.; van de Garde, M.D.B.; Jachimowski, L.C.M.; Jebbink, M.F.; Jakobs, M.; Luyf, A.C.M.; et al. A Sensitive Assay for Virus Discovery in Respiratory Clinical Samples. PLoS ONE 2011, 6, e16118. [Google Scholar] [CrossRef] [PubMed]
- VanDevanter, D.R.; Warrener, P.; Bennett, L.; Schultz, E.R.; Coulter, S.; Garber, R.L.; Rose, T.M. Detection and analysis of diverse herpesviral species by consensus primer PCR. J. Clin. Microbiol. 1996, 34, 1666–1671. [Google Scholar] [CrossRef]
- Weiss, S.; Witkowski, P.T.; Auste, B.; Nowak, K.; Weber, N.; Fahr, J.; Mombouli, J.-V.; Wolfe, N.D.; Drexler, J.F.; Drosten, C.; et al. Hantavirus in Bat, Sierra Leone. Emerg. Infect. Dis. 2012, 18, 159–161. [Google Scholar] [CrossRef]
- Drosten, C.; Günther, S.; Preiser, W.; van der Werf, S.; Brodt, H.-R.; Becker, S.; Rabenau, H.; Panning, M.; Kolesnikova, L.; Fouchier, R.A.M.; et al. Identification of a Novel Coronavirus in Patients with Severe Acute Respiratory Syndrome. N. Engl. J. Med. 2003, 348, 1967–1976. [Google Scholar] [CrossRef]
- Zheng, L.; Tang, J.; Clover, G.R.G.; Spackman, M.E.; Freeman, A.J.; Rodoni, B.C. Novel genus-specific broad range primers for the detection of furoviruses, hordeiviruses and rymoviruses and their application in field surveys in South-east Australia. J. Virol. Methods 2015, 214, 1–9. [Google Scholar] [CrossRef]
- Choi, S.K.; Choi, J.K.; Park, W.M.; Ryu, K.H. RT–PCR detection and identification of three species of cucumoviruses with a genus-specific single pair of primers. J. Virol. Methods 1999, 83, 67–73. [Google Scholar] [CrossRef]
- Proebster, B.; Kinney, R.M.; Kaaden, O.-R.; Pfeffer, M. Genus-Specific Detection of Alphaviruses by a Semi-Nested Reverse Transcription-Polymerase Chain Reaction. Am. J. Trop. Med. Hyg. 1997, 57, 709–718. [Google Scholar] [CrossRef]
- Radford, A.D.; Chapman, D.; Dixon, L.; Chantrey, J.; Darby, A.C.; Hall, N. Application of next-generation sequencing technologies in virology. J. Gen. Virol. 2012, 93, 1853–1868. [Google Scholar] [CrossRef]
- Jerome, H.; Taylor, C.; Sreenu, V.B.; Klymenko, T.; Filipe, A.D.S.; Jackson, C.; Davis, C.; Ashraf, S.; Wilson-Davies, E.; Jesudason, N.; et al. Metagenomic next-generation sequencing aids the diagnosis of viral infections in febrile returning travellers. J. Infect. 2019, 79, 383–388. [Google Scholar] [CrossRef] [PubMed]
- Prachayangprecha, S.; Schapendonk, C.M.E.; Koopmans, M.P.; Osterhaus, A.D.M.E.; Schurch, A.C.; Pas, S.D.; van der Eijk, A.A.; Poovorawan, Y.; Haagmans, B.L.; Smits, S.L. Exploring the Potential of Next-Generation Sequencing in Detection of Respiratory Viruses. J. Clin. Microbiol. 2014, 52, 3722–3730. [Google Scholar] [CrossRef]
- Dhiman, N.; Smith, D.I.; Poland, G.A. Next-generation sequencing: A transformative tool for vaccinology. Expert Rev. Vaccines 2009, 8, 963–967. [Google Scholar] [CrossRef] [PubMed]
- Luciani, F.; Bull, R.A.; Lloyd, A.R. Next generation deep sequencing and vaccine design: Today and tomorrow. Trends Biotechnol. 2012, 30, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Kruglyak, K.M.; Lin, E.; Ong, F.S. Next-Generation Sequencing and Applications to the Diagnosis and Treatment of Lung Cancer. In Lung Cancer and Personalized Medicine: Novel Therapies and Clinical Management; Ahmad, A., Gadgeel, S.M., Eds.; Springer International Publishing: Cham, Switzerland, 2016; Volume 890, pp. 123–136. ISBN 978-3-319-24931-5. [Google Scholar]
- Derache, A.; Iwuji, C.C.; Baisley, K.; Danaviah, S.; Marcelin, A.-G.; Calvez, V.; de Oliveira, T.; Dabis, F.; Porter, K.; Pillay, D. Impact of Next-generation Sequencing Defined Human Immunodeficiency Virus Pretreatment Drug Resistance on Virological Outcomes in the ANRS 12249 Treatment-as-Prevention Trial. Clin. Infect. Dis. 2019, 69, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Riley, L.W.; Blanton, R.E. Advances in Molecular Epidemiology of Infectious Diseases: Definitions, Approaches, and Scope of the Field *. Microbiol. Spectr. 2018, 6. [Google Scholar] [CrossRef]
- Wilson, M.R.; Sample, H.A.; Zorn, K.C.; Arevalo, S.; Yu, G.; Neuhaus, J.; Federman, S.; Stryke, D.; Briggs, B.; Langelier, C.; et al. Clinical Metagenomic Sequencing for Diagnosis of Meningitis and Encephalitis. N. Engl. J. Med. 2019, 380, 2327–2340. [Google Scholar] [CrossRef]
- Sukhum, K.V.; Diorio-Toth, L.; Dantas, G. Genomic and Metagenomic Approaches for Predictive Surveillance of Emerging Pathogens and Antibiotic Resistance. Clin. Pharmacol. Ther. 2019, 106, 512–524. [Google Scholar] [CrossRef]
- Chen, K.; Pachter, L. Bioinformatics for Whole-Genome Shotgun Sequencing of Microbial Communities. PLoS Comput. Biol. 2005, 1, e24. [Google Scholar] [CrossRef]
- Worthey, E.A.; Mayer, A.N.; Syverson, G.D.; Helbling, D.; Bonacci, B.B.; Decker, B.; Serpe, J.M.; Dasu, T.; Tschannen, M.R.; Veith, R.L.; et al. Making a definitive diagnosis: Successful clinical application of whole exome sequencing in a child with intractable inflammatory bowel disease. Genet. Med. 2011, 13, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-X.; Shi, M.; Tian, J.-H.; Lin, X.-D.; Kang, Y.-J.; Chen, L.-J.; Qin, X.-C.; Xu, J.; Holmes, E.C.; Zhang, Y.-Z. Unprecedented genomic diversity of RNA viruses in arthropods reveals the ancestry of negative-sense RNA viruses. eLife 2015, 4, e05378. [Google Scholar] [CrossRef] [PubMed]
- Shi, M.; Lin, X.-D.; Tian, J.-H.; Chen, L.-J.; Chen, X.; Li, C.-X.; Qin, X.-C.; Li, J.; Cao, J.-P.; Eden, J.-S.; et al. Redefining the invertebrate RNA virosphere. Nature 2016, 540, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Venter, J.C. Environmental Genome Shotgun Sequencing of the Sargasso Sea. Science 2004, 304, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Mulcahy-O’Grady, H.; Workentine, M.L. The Challenge and Potential of Metagenomics in the Clinic. Front. Immunol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Graf, E.H.; Simmon, K.E.; Tardif, K.D.; Hymas, W.; Flygare, S.; Eilbeck, K.; Yandell, M.; Schlaberg, R. Unbiased Detection of Respiratory Viruses by Use of RNA Sequencing-Based Metagenomics: A Systematic Comparison to a Commercial PCR Panel. J. Clin. Microbiol. 2016, 54, 1000–1007. [Google Scholar] [CrossRef]
- Fischer, N.; Indenbirken, D.; Meyer, T.; Lütgehetmann, M.; Lellek, H.; Spohn, M.; Aepfelbacher, M.; Alawi, M.; Grundhoff, A. Evaluation of Unbiased Next-Generation Sequencing of RNA (RNA-seq) as a Diagnostic Method in Influenza Virus-Positive Respiratory Samples. J. Clin. Microbiol. 2015, 53, 2238–2250. [Google Scholar] [CrossRef]
- Alquezar-Planas, D.E.; Mourier, T.; Bruhn, C.A.W.; Hansen, A.J.; Vitcetz, S.N.; Mørk, S.; Gorodkin, J.; Nielsen, H.A.; Guo, Y.; Sethuraman, A.; et al. Discovery of a divergent HPIV4 from respiratory secretions using second and third generation metagenomic sequencing. Sci. Rep. 2013, 3, 2468. [Google Scholar] [CrossRef]
- Gu, W.; Miller, S.; Chiu, C.Y. Clinical Metagenomic Next-Generation Sequencing for Pathogen Detection. Annu. Rev. Pathol. Mech. Dis. 2019, 14, 319–338. [Google Scholar] [CrossRef]
- McLaren, M.R.; Willis, A.D.; Callahan, B.J. Consistent and correctable bias in metagenomic sequencing experiments. eLife 2019, 8, e46923. [Google Scholar] [CrossRef]
- Boers, S.A.; Jansen, R.; Hays, J.P. Understanding and overcoming the pitfalls and biases of next-generation sequencing (NGS) methods for use in the routine clinical microbiological diagnostic laboratory. Eur. J. Clin. Microbiol. Infect. Dis. 2019, 38, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Liu, T.; Yu, C.-H.; Chiang, T.-Y.; Hwang, C.-C. Effects of GC Bias in Next-Generation-Sequencing Data on De Novo Genome Assembly. PLoS ONE 2013, 8, e62856. [Google Scholar] [CrossRef] [PubMed]
- Thomson, E.; Ip, C.L.C.; Badhan, A.; Christiansen, M.T.; Adamson, W.; Ansari, M.A.; Bibby, D.; Breuer, J.; Brown, A.; Bowden, R.; et al. Comparison of Next-Generation Sequencing Technologies for Comprehensive Assessment of Full-Length Hepatitis C Viral Genomes. J. Clin. Microbiol. 2016, 54, 2470–2484. [Google Scholar] [CrossRef] [PubMed]
- Mlakar, J.; Korva, M.; Tul, N.; Popović, M.; Poljšak-Prijatelj, M.; Mraz, J.; Kolenc, M.; Resman Rus, K.; Vesnaver Vipotnik, T.; Fabjan Vodušek, V.; et al. Zika Virus Associated with Microcephaly. N. Engl. J. Med. 2016, 374, 951–958. [Google Scholar] [CrossRef]
- Li, T.; Mbala-Kingebeni, P.; Naccache, S.N.; Thézé, J.; Bouquet, J.; Federman, S.; Somasekar, S.; Yu, G.; Sanchez-San Martin, C.; Achari, A.; et al. Metagenomic Next-Generation Sequencing of the 2014 Ebola Virus Disease Outbreak in the Democratic Republic of the Congo. J. Clin. Microbiol. 2019, 57, e00827-19. [Google Scholar] [CrossRef]
- Palacios, G.; Druce, J.; Du, L.; Tran, T.; Birch, C.; Briese, T.; Conlan, S.; Quan, P.-L.; Hui, J.; Marshall, J.; et al. A New Arenavirus in a Cluster of Fatal Transplant-Associated Diseases. N. Engl. J. Med. 2008, 358, 991–998. [Google Scholar] [CrossRef]
- Kustin, T.; Ling, G.; Sharabi, S.; Ram, D.; Friedman, N.; Zuckerman, N.; Bucris, E.D.; Glatman-Freedman, A.; Stern, A.; Mandelboim, M. A method to identify respiratory virus infections in clinical samples using next-generation sequencing. Sci. Rep. 2019, 9, 2606. [Google Scholar] [CrossRef]
- Choi, S.-H.; Hong, S.-B.; Ko, G.-B.; Lee, Y.; Park, H.J.; Park, S.-Y.; Moon, S.M.; Cho, O.-H.; Park, K.-H.; Chong, Y.P.; et al. Viral Infection in Patients with Severe Pneumonia Requiring Intensive Care Unit Admission. Am. J. Respir. Crit. Care Med. 2012, 186, 325–332. [Google Scholar] [CrossRef]
- Karhu, J.; Ala-Kokko, T.I.; Vuorinen, T.; Ohtonen, P.; Syrjala, H. Lower Respiratory Tract Virus Findings in Mechanically Ventilated Patients With Severe Community-Acquired Pneumonia. Clin. Infect. Dis. 2014, 59, 62–70. [Google Scholar] [CrossRef]
- Honkinen, M.; Lahti, E.; Österback, R.; Ruuskanen, O.; Waris, M. Viruses and bacteria in sputum samples of children with community-acquired pneumonia. Clin. Microbiol. Infect. 2012, 18, 300–307. [Google Scholar] [CrossRef]
- Jain, S.; Williams, D.J.; Arnold, S.R.; Ampofo, K.; Bramley, A.M.; Reed, C.; Stockmann, C.; Anderson, E.J.; Grijalva, C.G.; Self, W.H.; et al. Community-Acquired Pneumonia Requiring Hospitalization among U.S. Children. N. Engl. J. Med. 2015, 372, 835–845. [Google Scholar] [CrossRef] [PubMed]
- Thorburn, F.; Bennett, S.; Modha, S.; Murdoch, D.; Gunson, R.; Murcia, P.R. The use of next generation sequencing in the diagnosis and typing of respiratory infections. J. Clin. Virol. 2015, 69, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Heinonen, S.; Jartti, T.; Garcia, C.; Oliva, S.; Smitherman, C.; Anguiano, E.; de Steenhuijsen Piters, W.A.A.; Vuorinen, T.; Ruuskanen, O.; Dimo, B.; et al. Rhinovirus Detection in Symptomatic and Asymptomatic Children: Value of Host Transcriptome Analysis. Am. J. Respir. Crit. Care Med. 2016, 193, 772–782. [Google Scholar] [CrossRef] [PubMed]
- Pavia, A.T. Viral Infections of the Lower Respiratory Tract: Old Viruses, New Viruses, and the Role of Diagnosis. Clin. Infect. Dis. 2011, 52, S284–S289. [Google Scholar] [CrossRef] [PubMed]
- Ruuskanen, O.; Jarvinen, A. Editorial Commentary: What is the Real Role of Respiratory Viruses in Severe Community-Acquired Pneumonia? Clin. Infect. Dis. 2014, 59, 71–73. [Google Scholar] [CrossRef] [PubMed]
- Ruuskanen, O.; Lahti, E.; Jennings, L.C.; Murdoch, D.R. Viral pneumonia. The Lancet 2011, 377, 1264–1275. [Google Scholar] [CrossRef]
- Self, W.H.; Williams, D.J.; Zhu, Y.; Ampofo, K.; Pavia, A.T.; Chappell, J.D.; Hymas, W.C.; Stockmann, C.; Bramley, A.M.; Schneider, E.; et al. Respiratory Viral Detection in Children and Adults: Comparing Asymptomatic Controls and Patients With Community-Acquired Pneumonia. J. Infect. Dis. 2016, 213, 584–591. [Google Scholar] [CrossRef]
- Pérez-Sautu, U.; Wiley, M.R.; Iglesias-Caballero, M.; Pozo, F.; Prieto, K.; Chitty, J.A.; García-García, M.L.; Calvo, C.; Casas, I.; Palacios, G. Target-independent high-throughput sequencing methods provide evidence that already known human viral pathogens play a main role in respiratory infections with unexplained etiology. Emerg. Microbes Infect. 2019, 8, 1054–1065. [Google Scholar] [CrossRef]
- Otto, M. Next-generation sequencing to monitor the spread of antimicrobial resistance. Genome Med. 2017, 9, 68. [Google Scholar] [CrossRef]
- Trotter, A.J.; Aydin, A.; Strinden, M.J.; O’Grady, J. Recent and emerging technologies for the rapid diagnosis of infection and antimicrobial resistance. Curr. Opin. Microbiol. 2019, 51, 39–45. [Google Scholar] [CrossRef]
- Datta, S. Next-generation sequencing in clinical virology: Discovery of new viruses. World J. Virol. 2015, 4, 265. [Google Scholar] [CrossRef]
- DNA Sequencing Costs: Data. Available online: https://www.genome.gov/about-genomics/fact-sheets/DNA-Sequencing-Costs-Data (accessed on 26 September 2019).
- Hillmann, B.; Al-Ghalith, G.A.; Shields-Cutler, R.R.; Zhu, Q.; Gohl, D.M.; Beckman, K.B.; Knight, R.; Knights, D. Evaluating the Information Content of Shallow Shotgun Metagenomics. mSystems 2018, 3, e00069-18. [Google Scholar] [CrossRef] [PubMed]
- Wood, D.E.; Salzberg, S.L. Kraken: Ultrafast metagenomic sequence classification using exact alignments. Genome Biol. 2014, 15, R46. [Google Scholar] [CrossRef] [PubMed]
- Menzel, P.; Ng, K.L.; Krogh, A. Fast and sensitive taxonomic classification for metagenomics with Kaiju. Nat. Commun. 2016, 7, 11257. [Google Scholar] [CrossRef]
- Wang, Q.; Jia, P.; Zhao, Z. VirusFinder: Software for Efficient and Accurate Detection of Viruses and Their Integration Sites in Host Genomes through Next Generation Sequencing Data. PLoS ONE 2013, 8, e64465. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.H.; Siddle, K.J.; Park, D.J.; Sabeti, P.C. Benchmarking Metagenomics Tools for Taxonomic Classification. Cell 2019, 178, 779–794. [Google Scholar] [CrossRef] [PubMed]
- Breitwieser, F.P.; Lu, J.; Salzberg, S.L. A review of methods and databases for metagenomic classification and assembly. Brief. Bioinform. 2019, 20, 1125–1136. [Google Scholar] [CrossRef]
- Nooij, S.; Schmitz, D.; Vennema, H.; Kroneman, A.; Koopmans, M.P.G. Overview of Virus Metagenomic Classification Methods and Their Biological Applications. Front. Microbiol. 2018, 9, 749. [Google Scholar] [CrossRef]
- Gargis, A.S.; Kalman, L.; Berry, M.W.; Bick, D.P.; Dimmock, D.P.; Hambuch, T.; Lu, F.; Lyon, E.; Voelkerding, K.V.; Zehnbauer, B.A.; et al. Assuring the quality of next-generation sequencing in clinical laboratory practice. Nat. Biotechnol. 2012, 30, 1033–1036. [Google Scholar] [CrossRef]
- Gargis, A.S.; Kalman, L.; Bick, D.P.; da Silva, C.; Dimmock, D.P.; Funke, B.H.; Gowrisankar, S.; Hegde, M.R.; Kulkarni, S.; Mason, C.E.; et al. Good laboratory practice for clinical next-generation sequencing informatics pipelines. Nat. Biotechnol. 2015, 33, 689–693. [Google Scholar] [CrossRef]
- Oulas, A.; Pavloudi, C.; Polymenakou, P.; Pavlopoulos, G.A.; Papanikolaou, N.; Kotoulas, G.; Arvanitidis, C.; Iliopoulos, L. Metagenomics: Tools and Insights for Analyzing Next-Generation Sequencing Data Derived from Biodiversity Studies. Bioinforma. Biol. Insights 2015, 9. [Google Scholar] [CrossRef]
- Prakash, T.; Taylor, T.D. Functional assignment of metagenomic data: Challenges and applications. Brief. Bioinform. 2012, 13, 711–727. [Google Scholar] [CrossRef] [PubMed]
- Hijano, D.R.; Brazelton de Cardenas, J.; Maron, G.; Garner, C.D.; Ferrolino, J.A.; Dallas, R.H.; Gu, Z.; Hayden, R.T. Clinical correlation of influenza and respiratory syncytial virus load measured by digital PCR. PLoS ONE 2019, 14, e0220908. [Google Scholar] [CrossRef] [PubMed]
- Allen, U.D.; Hu, P.; Pereira, S.L.; Robinson, J.L.; Paton, T.A.; Beyene, J.; Khodai-Booran, N.; Dipchand, A.; Hébert, D.; Ng, V.; et al. The genetic diversity of Epstein-Barr virus in the setting of transplantation relative to non-transplant settings: A feasibility study. Pediatr. Transplant. 2016, 20, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Matranga, C.B.; Andersen, K.G.; Winnicki, S.; Busby, M.; Gladden, A.D.; Tewhey, R.; Stremlau, M.; Berlin, A.; Gire, S.K.; England, E.; et al. Enhanced methods for unbiased deep sequencing of Lassa and Ebola RNA viruses from clinical and biological samples. Genome Biol. 2014, 15, 519. [Google Scholar] [CrossRef] [PubMed]
- Calvet, G.; Aguiar, R.S.; Melo, A.S.O.; Sampaio, S.A.; de Filippis, I.; Fabri, A.; Araujo, E.S.M.; de Sequeira, P.C.; de Mendonça, M.C.L.; de Oliveira, L.; et al. Detection and sequencing of Zika virus from amniotic fluid of fetuses with microcephaly in Brazil: A case study. Lancet Infect. Dis. 2016, 16, 653–660. [Google Scholar] [CrossRef]
- Khan, A.S.; Ng, S.H.S.; Vandeputte, O.; Aljanahi, A.; Deyati, A.; Cassart, J.-P.; Charlebois, R.L.; Taliaferro, L.P. A Multicenter Study To Evaluate the Performance of High-Throughput Sequencing for Virus Detection. mSphere 2017, 2, e00307-17. [Google Scholar] [CrossRef]
- Oechslin, C.P.; Lenz, N.; Liechti, N.; Ryter, S.; Agyeman, P.; Bruggmann, R.; Leib, S.L.; Beuret, C.M. Limited Correlation of Shotgun Metagenomics Following Host Depletion and Routine Diagnostics for Viruses and Bacteria in Low Concentrated Surrogate and Clinical Samples. Front. Cell. Infect. Microbiol. 2018, 8, 375. [Google Scholar] [CrossRef]
- Kunin, V.; Copeland, A.; Lapidus, A.; Mavromatis, K.; Hugenholtz, P. A Bioinformatician’s Guide to Metagenomics. Microbiol. Mol. Biol. Rev. 2008, 72, 557–578. [Google Scholar] [CrossRef]
- Shi, M.; Lin, X.-D.; Chen, X.; Tian, J.-H.; Chen, L.-J.; Li, K.; Wang, W.; Eden, J.-S.; Shen, J.-J.; Liu, L.; et al. The evolutionary history of vertebrate RNA viruses. Nature 2018, 556, 197–202. [Google Scholar] [CrossRef]
- Zhang, Y.-Z.; Shi, M.; Holmes, E.C. Using Metagenomics to Characterize an Expanding Virosphere. Cell 2018, 172, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Wilder, C.N.; Lopera, J.; Mittar, D.; Horton, C. Optimizing microbiome research applications with cutting-edge standards. Nature. Available online: https://www.nature.com/articles/d42473-019-00005-x (accessed on 26 September 2019). [CrossRef]
- Kulkarni, P.; Frommolt, P. Challenges in the Setup of Large-scale Next-Generation Sequencing Analysis Workflows. Comput. Struct. Biotechnol. J. 2017, 15, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Greninger, A.L. The challenge of diagnostic metagenomics. Expert Rev. Mol. Diagn. 2018, 18, 605–615. [Google Scholar] [CrossRef] [PubMed]
- Dewey, F.E.; Pan, S.; Wheeler, M.T.; Quake, S.R.; Ashley, E.A. DNA Sequencing: Clinical Applications of New DNA Sequencing Technologies. Circulation 2012, 125, 931–944. [Google Scholar] [CrossRef] [PubMed]
- Totomoch-Serra, A.; Marquez, M.F.; Cervantes-Barragán, D.E. Sanger sequencing as a first-line approach for molecular diagnosis of Andersen-Tawil syndrome. F1000Research 2017, 6, 1016. [Google Scholar] [CrossRef] [PubMed]
- Oude Munnink, B.B.; Jazaeri Farsani, S.M.; Deijs, M.; Jonkers, J.; Verhoeven, J.T.P.; Ieven, M.; Goossens, H.; de Jong, M.D.; Berkhout, B.; Loens, K.; et al. Autologous Antibody Capture to Enrich Immunogenic Viruses for Viral Discovery. PLoS ONE 2013, 8, e78454. [Google Scholar] [CrossRef] [PubMed]
- Rabinowicz, P.D. Constructing Gene-Enriched Plant Genomic Libraries Using Methylation Filtration Technology. In Plant Functional Genomics; Humana Press: Totowa, NJ, USA, 2003; Volume 236, pp. 21–36. ISBN 978-1-59259-413-9. [Google Scholar]
- Kohl, C.; Brinkmann, A.; Dabrowski, P.W.; Radonić, A.; Nitsche, A.; Kurth, A. Protocol for Metagenomic Virus Detection in Clinical Specimens1. Emerg. Infect. Dis. 2015, 21. [Google Scholar] [CrossRef]
- Charalampous, T.; Kay, G.L.; Richardson, H.; Aydin, A.; Baldan, R.; Jeanes, C.; Rae, D.; Grundy, S.; Turner, D.J.; Wain, J.; et al. Nanopore metagenomics enables rapid clinical diagnosis of bacterial lower respiratory infection. Nat. Biotechnol. 2019, 37, 783–792. [Google Scholar] [CrossRef]
- Marotz, C.A.; Sanders, J.G.; Zuniga, C.; Zaramela, L.S.; Knight, R.; Zengler, K. Improving saliva shotgun metagenomics by chemical host DNA depletion. Microbiome 2018, 6, 42. [Google Scholar] [CrossRef]
- Sabina, J.; Leamon, J.H. Bias in Whole Genome Amplification: Causes and Considerations. In Whole Genome Amplification; Kroneis, T., Ed.; Springer New York: New York, NY, USA, 2015; Volume 1347, pp. 15–41. ISBN 978-1-4939-2989-4. [Google Scholar]
- Jensen, R.H.; Mollerup, S.; Mourier, T.; Hansen, T.A.; Fridholm, H.; Nielsen, L.P.; Willerslev, E.; Hansen, A.J.; Vinner, L. Target-Dependent Enrichment of Virions Determines the Reduction of High-Throughput Sequencing in Virus Discovery. PLoS ONE 2015, 10, e0122636. [Google Scholar] [CrossRef]
- Denesvre, C.; Dumarest, M.; Rémy, S.; Gourichon, D.; Eloit, M. Chicken skin virome analyzed by high-throughput sequencing shows a composition highly different from human skin. Virus Genes 2015, 51, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Crawford, E.D.; O’Donovan, B.D.; Wilson, M.R.; Chow, E.D.; Retallack, H.; DeRisi, J.L. Depletion of Abundant Sequences by Hybridization (DASH): Using Cas9 to remove unwanted high-abundance species in sequencing libraries and molecular counting applications. Genome Biol. 2016, 17, 41. [Google Scholar] [CrossRef] [PubMed]
- Kraus, A.J.; Brink, B.G.; Siegel, T.N. Efficient and specific oligo-based depletion of rRNA. Sci. Rep. 2019, 9, 12281. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.T.; Pope, C.E.; Marsh, R.L.; Wolter, D.J.; Weiss, E.J.; Hager, K.R.; Vo, A.T.; Brittnacher, M.J.; Radey, M.C.; Hayden, H.S.; et al. Human and Extracellular DNA Depletion for Metagenomic Analysis of Complex Clinical Infection Samples Yields Optimized Viable Microbiome Profiles. Cell Rep. 2019, 26, 2227–2240.e5. [Google Scholar] [CrossRef] [PubMed]
- Tsalik, E.L.; Bonomo, R.A.; Fowler, V.G. New Molecular Diagnostic Approaches to Bacterial Infections and Antibacterial Resistance. Annu. Rev. Med. 2018, 69, 379–394. [Google Scholar] [CrossRef] [PubMed]
- Lindbaek, M. Broad-spectrum antibiotics gave no clinical benefit and more adverse effects than narrow-spectrum antibiotics in treating acute respiratory tract infections in US children. BMJ Evid.-Based Med. 2019, 24, 119–120. [Google Scholar] [CrossRef]
- Briese, T.; Kapoor, A.; Mishra, N.; Jain, K.; Kumar, A.; Jabado, O.J.; Lipkin, W.I. Virome Capture Sequencing Enables Sensitive Viral Diagnosis and Comprehensive Virome Analysis. mBio 2015, 6, e01491-15. [Google Scholar] [CrossRef]
- Vasilyev, E.V.; Trofimov, D.Y.; Tonevitsky, A.G.; Ilinsky, V.V.; Korostin, D.O.; Rebrikov, D.V. Torque Teno Virus (TTV) distribution in healthy Russian population. Virol. J. 2009, 6, 134. [Google Scholar] [CrossRef]
- Hino, S. TTV, a new human virus with single stranded circular DNA genome. Rev. Med. Virol. 2002, 12, 151–158. [Google Scholar] [CrossRef]
- Depledge, D.P.; Kundu, S.; Jensen, N.J.; Gray, E.R.; Jones, M.; Steinberg, S.; Gershon, A.; Kinchington, P.R.; Schmid, D.S.; Balloux, F.; et al. Deep Sequencing of Viral Genomes Provides Insight into the Evolution and Pathogenesis of Varicella Zoster Virus and Its Vaccine in Humans. Mol. Biol. Evol. 2014, 31, 397–409. [Google Scholar] [CrossRef]
- Tsangaras, K.; Wales, N.; Sicheritz-Pontén, T.; Rasmussen, S.; Michaux, J.; Ishida, Y.; Morand, S.; Kampmann, M.-L.; Gilbert, M.T.P.; Greenwood, A.D. Hybridization Capture Using Short PCR Products Enriches Small Genomes by Capturing Flanking Sequences (CapFlank). PLoS ONE 2014, 9, e109101. [Google Scholar] [CrossRef] [PubMed]
- Wylie, T.N.; Wylie, K.M.; Herter, B.N.; Storch, G.A. Enhanced virome sequencing using targeted sequence capture. Genome Res. 2015, 25, 1910–1920. [Google Scholar] [CrossRef] [PubMed]
- Lovett, M.; Kere, J.; Hinton, L.M. Direct selection: A method for the isolation of cDNAs encoded by large genomic regions. Proc. Natl. Acad. Sci. USA 1991, 88, 9628–9632. [Google Scholar] [CrossRef] [PubMed]
- Tweedy, J.; Spyrou, M.A.; Donaldson, C.D.; Depledge, D.; Breuer, J.; Gompels, U.A. Complete Genome Sequence of the Human Herpesvirus 6A Strain AJ from Africa Resembles Strain GS from North America. Genome Announc. 2015, 3, e01498-14. [Google Scholar] [CrossRef]
- Ebert, K.; Depledge, D.P.; Breuer, J.; Harman, L.; Elliott, G. Mode of Virus Rescue Determines the Acquisition of VHS Mutations in VP22-Negative Herpes Simplex Virus 1. J. Virol. 2013, 87, 10389–10393. [Google Scholar] [CrossRef]
- Palser, A.L.; Grayson, N.E.; White, R.E.; Corton, C.; Correia, S.; Ba abdullah, M.M.; Watson, S.J.; Cotten, M.; Arrand, J.R.; Murray, P.G.; et al. Genome Diversity of Epstein-Barr Virus from Multiple Tumor Types and Normal Infection. J. Virol. 2015, 89, 5222–5237. [Google Scholar] [CrossRef]
- Depledge, D.P.; Palser, A.L.; Watson, S.J.; Lai, I.Y.-C.; Gray, E.R.; Grant, P.; Kanda, R.K.; Leproust, E.; Kellam, P.; Breuer, J. Specific Capture and Whole-Genome Sequencing of Viruses from Clinical Samples. PLoS ONE 2011, 6, e27805. [Google Scholar] [CrossRef]
- Donaldson, C.D.; Clark, D.A.; Kidd, I.M.; Breuer, J.; Depledge, D.D. Genome Sequence of Human Herpesvirus 7 Strain UCL-1. Genome Announc. 2013, 1, e00830-13. [Google Scholar] [CrossRef]
- Mamanova, L.; Coffey, A.J.; Scott, C.E.; Kozarewa, I.; Turner, E.H.; Kumar, A.; Howard, E.; Shendure, J.; Turner, D.J. Target-enrichment strategies for next-generation sequencing. Nat. Methods 2010, 7, 111–118. [Google Scholar] [CrossRef]
- Wylie, K.M.; Wylie, T.N.; Buller, R.; Herter, B.; Cannella, M.T.; Storch, G.A. Detection of Viruses in Clinical Samples by Use of Metagenomic Sequencing and Targeted Sequence Capture. J. Clin. Microbiol. 2018, 56, e01123-18. [Google Scholar] [CrossRef]
- Viral Hemorrhagic Fever Consortium; Metsky, H.C.; Siddle, K.J.; Gladden-Young, A.; Qu, J.; Yang, D.K.; Brehio, P.; Goldfarb, A.; Piantadosi, A.; Wohl, S.; et al. Capturing sequence diversity in metagenomes with comprehensive and scalable probe design. Nat. Biotechnol. 2019, 37, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Cottam, E.M.; Wadsworth, J.; Knowles, N.J.; King, D.P. Full Sequencing of Viral Genomes: Practical Strategies Used for the Amplification and Characterization of Foot-and-Mouth Disease Virus. In Molecular Epidemiology of Microorganisms; Caugant, D.A., Ed.; Humana Press: Totowa, NJ, USA, 2009; Volume 551, pp. 217–230. ISBN 978-1-60327-998-7. [Google Scholar]
- Gardy, J.L.; Naus, M.; Amlani, A.; Chung, W.; Kim, H.; Tan, M.; Severini, A.; Krajden, M.; Puddicombe, D.; Sahni, V.; et al. Whole-Genome Sequencing of Measles Virus Genotypes H1 and D8 During Outbreaks of Infection Following the 2010 Olympic Winter Games Reveals Viral Transmission Routes. J. Infect. Dis. 2015, 212, 1574–1578. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Dhama, K.; Karthik, K.; Tiwari, R.; Khandia, R.; Munjal, A.; Iqbal, H.M.N.; Malik, Y.S.; Bueno-Marí, R. Advances in Diagnosis, Surveillance, and Monitoring of Zika Virus: An Update. Front. Microbiol. 2018, 8, 2677. [Google Scholar] [CrossRef] [PubMed]
- Cotten, M.; Petrova, V.; Phan, M.V.T.; Rabaa, M.A.; Watson, S.J.; Ong, S.H.; Kellam, P.; Baker, S. Deep Sequencing of Norovirus Genomes Defines Evolutionary Patterns in an Urban Tropical Setting. J. Virol. 2014, 88, 11056–11069. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Lockwood, J.; Depledge, D.P.; Chaudhry, Y.; Aston, A.; Rao, K.; Hartley, J.C.; Goodfellow, I.; Breuer, J. Next-Generation Whole Genome Sequencing Identifies the Direction of Norovirus Transmission in Linked Patients. Clin. Infect. Dis. 2013, 57, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Kireev, D.E.; Lopatukhin, A.E.; Murzakova, A.V.; Pimkina, E.V.; Speranskaya, A.S.; Neverov, A.D.; Fedonin, G.G.; Fantin, Y.S.; Shipulin, G.A. Evaluating the accuracy and sensitivity of detecting minority HIV-1 populations by Illumina next-generation sequencing. J. Virol. Methods 2018, 261, 40–45. [Google Scholar] [CrossRef]
- Watson, S.J.; Langat, P.; Reid, S.M.; Lam, T.T.-Y.; Cotten, M.; Kelly, M.; Van Reeth, K.; Qiu, Y.; Simon, G.; Bonin, E.; et al. Molecular Epidemiology and Evolution of Influenza Viruses Circulating within European Swine between 2009 and 2013. J. Virol. 2015, 89, 9920–9931. [Google Scholar] [CrossRef]
- Parameswaran, P.; Charlebois, P.; Tellez, Y.; Nunez, A.; Ryan, E.M.; Malboeuf, C.M.; Levin, J.Z.; Lennon, N.J.; Balmaseda, A.; Harris, E.; et al. Genome-Wide Patterns of Intrahuman Dengue Virus Diversity Reveal Associations with Viral Phylogenetic Clade and Interhost Diversity. J. Virol. 2012, 86, 8546–8558. [Google Scholar] [CrossRef]
- Newman, R.M.; Kuntzen, T.; Weiner, B.; Berical, A.; Charlebois, P.; Kuiken, C.; Murphy, D.G.; Simmonds, P.; Bennett, P.; Lennon, N.J.; et al. Whole Genome Pyrosequencing of Rare Hepatitis C Virus Genotypes Enhances Subtype Classification and Identification of Naturally Occurring Drug Resistance Variants. J. Infect. Dis. 2013, 208, 17–31. [Google Scholar] [CrossRef]
- Miia, J.-V.; Tiina, N.; Tarja, S.; Olli, V.; Liisa, S.; Anita, H. Evolutionary trends of European bat lyssavirus type 2 including genetic characterization of Finnish strains of human and bat origin 24 years apart. Arch. Virol. 2015, 160, 1489–1498. [Google Scholar] [CrossRef]
- Chrzastek, K.; Lee, D.; Smith, D.; Sharma, P.; Suarez, D.L.; Pantin-Jackwood, M.; Kapczynski, D.R. Use of Sequence-Independent, Single-Primer-Amplification (SISPA) for rapid detection, identification, and characterization of avian RNA viruses. Virology 2017, 509, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Quick, J.; Loman, N.J.; Duraffour, S.; Simpson, J.T.; Severi, E.; Cowley, L.; Bore, J.A.; Koundouno, R.; Dudas, G.; Mikhail, A.; et al. Real-time, portable genome sequencing for Ebola surveillance. Nature 2016, 530, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Safonova, M.V.; Shchelkanov, M.Y.; Khafizov, K.; Matsvay, A.D.; Ayginin, A.A.; Dolgova, A.S.; Shchelkanov, E.M.; Pimkina, E.V.; Speranskaya, A.S.; Galkina, I.V.; et al. Sequencing and genetic characterization of two strains Paramushir virus obtained from the Tyuleniy Island in the Okhotsk Sea (2015). Ticks Tick-Borne Dis. 2019, 10, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Bialasiewicz, S.; McVernon, J.; Nolan, T.; Lambert, S.B.; Zhao, G.; Wang, D.; Nissen, M.D.; Sloots, T.P. Detection of a divergent Parainfluenza 4 virus in an adult patient with influenza like illness using next-generation sequencing. BMC Infect. Dis. 2014, 14, 275. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; Roy, S.; Ruis, C.; Yara Romero, E.; Shah, D.; Williams, R.; Breuer, J. Norovirus Whole-Genome Sequencing by SureSelect Target Enrichment: A Robust and Sensitive Method. J. Clin. Microbiol. 2016, 54, 2530–2537. [Google Scholar] [CrossRef]
- Cristescu, M.E. From barcoding single individuals to metabarcoding biological communities: Towards an integrative approach to the study of global biodiversity. Trends Ecol. Evol. 2014, 29, 566–571. [Google Scholar] [CrossRef]
- DeSalle, R.; Goldstein, P. Review and Interpretation of Trends in DNA Barcoding. Front. Ecol. Evol. 2019, 7, 302. [Google Scholar] [CrossRef]
- Deng, X.; Achari, A.; Federman, S.; Yu, G.; Somasekar, S.; Bártolo, I.; Yagi, S.; Mbala-Kingebeni, P.; Kapetshi, J.; Ahuka-Mundeke, S.; et al. Metagenomic sequencing with spiked primer enrichment for viral diagnostics and genomic surveillance. Nat. Microbiol. 2020. [Google Scholar] [CrossRef]
- Ayginin, A.A.; Pimkina, E.V.; Matsvay, A.D.; Speranskaya, A.S.; Safonova, M.V.; Blinova, E.A.; Artyushin, I.V.; Dedkov, V.G.; Shipulin, G.A.; Khafizov, K. The Study of Viral RNA Diversity in Bird Samples Using De Novo Designed Multiplex Genus-Specific Primer Panels. Adv. Virol. 2018, 2018, 1–10. [Google Scholar] [CrossRef]
- Dessilly, G.; Goeminne, L.; Vandenbroucke, A.; Dufrasne, F.E.; Martin, A.; Kabamba-Mukabi, B. First evaluation of the Next-Generation Sequencing platform for the detection of HIV-1 drug resistance mutations in Belgium. PLoS ONE 2018, 13, e0209561. [Google Scholar] [CrossRef]
- McKeegan, K.S.; Borges-Walmsley, M.I.; Walmsley, A.R. Microbial and viral drug resistance mechanisms. Trends Microbiol. 2002, 10, s8–s14. [Google Scholar] [CrossRef]
- Aanensen, D.M.; Feil, E.J.; Holden, M.T.G.; Dordel, J.; Yeats, C.A.; Fedosejev, A.; Goater, R.; Castillo-Ramírez, S.; Corander, J.; Colijn, C.; et al. Whole-Genome Sequencing for Routine Pathogen Surveillance in Public Health: A Population Snapshot of Invasive Staphylococcus aureus in Europe. mBio 2016, 7, e00444-16. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Zhao, X.; Li, J.; Niu, P.; Yang, B.; Wu, H.; Wang, W.; Song, H.; Huang, B.; Zhu, N.; et al. Genomic characterisation and epidemiology of 2019 novel coronavirus: Implications for virus origins and receptor binding. Lancet 2020. [Google Scholar] [CrossRef]
- Mbisa, J.L.; Fearnhill, E.; Dunn, D.T.; Pillay, D.; Asboe, D.; Cane, P.A. Evidence of Self-Sustaining Drug Resistant HIV-1 Lineages Among Untreated Patients in the United Kingdom. Clin. Infect. Dis. 2015, 61, 829–836. [Google Scholar] [CrossRef] [PubMed]
- Bartha, I.; Carlson, J.M.; Brumme, C.J.; McLaren, P.J.; Brumme, Z.L.; John, M.; Haas, D.W.; Martinez-Picado, J.; Dalmau, J.; López-Galíndez, C.; et al. A genome-to-genome analysis of associations between human genetic variation, HIV-1 sequence diversity, and viral control. eLife 2013, 2, e01123. [Google Scholar] [CrossRef] [PubMed]
- Power, R.A.; Davaniah, S.; Derache, A.; Wilkinson, E.; Tanser, F.; Gupta, R.K.; Pillay, D.; de Oliveira, T. Genome-Wide Association Study of HIV Whole Genome Sequences Validated using Drug Resistance. PLoS ONE 2016, 11, e0163746. [Google Scholar] [CrossRef] [PubMed]
- Artimo, P.; Jonnalagedda, M.; Arnold, K.; Baratin, D.; Csardi, G.; de Castro, E.; Duvaud, S.; Flegel, V.; Fortier, A.; Gasteiger, E.; et al. ExPASy: SIB bioinformatics resource portal. Nucleic Acids Res. 2012, 40, W597–W603. [Google Scholar] [CrossRef]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Niu, B.; Zhu, Z.; Fu, L.; Wu, S.; Li, W. FR-HIT, a very fast program to recruit metagenomic reads to homologous reference genomes. Bioinformatics 2011, 27, 1704–1705. [Google Scholar] [CrossRef]
- Langmead, B.; Salzberg, S.L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 2012, 9, 357–359. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinforma. Oxf. Engl. 2010, 26, 589–595. [Google Scholar] [CrossRef] [PubMed]
- Buchfink, B.; Xie, C.; Huson, D.H. Fast and sensitive protein alignment using DIAMOND. Nat. Methods 2015, 12, 59–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yao, H.; Thompson, E.J.; Tannir, N.M.; Weinstein, J.N.; Su, X. VirusSeq: Software to identify viruses and their integration sites using next-generation sequencing of human cancer tissue. Bioinformatics 2013, 29, 266–267. [Google Scholar] [CrossRef] [PubMed]
- UFO Sequencing Consortium within the I-BFM Study Group; Forster, M.; Szymczak, S.; Ellinghaus, D.; Hemmrich, G.; Rühlemann, M.; Kraemer, L.; Mucha, S.; Wienbrandt, L.; Stanulla, M.; et al. Vy-PER: Eliminating false positive detection of virus integration events in next generation sequencing data. Sci. Rep. 2015, 5, 11534. [Google Scholar] [CrossRef] [PubMed]
- Petty, T.J.; Cordey, S.; Padioleau, I.; Docquier, M.; Turin, L.; Preynat-Seauve, O.; Zdobnov, E.M.; Kaiser, L. Comprehensive Human Virus Screening Using High-Throughput Sequencing with a User-Friendly Representation of Bioinformatics Analysis: A Pilot Study. J. Clin. Microbiol. 2014, 52, 3351–3361. [Google Scholar] [CrossRef]
- Langmead, B.; Trapnell, C.; Pop, M.; Salzberg, S.L. Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol. 2009, 10, R25. [Google Scholar] [CrossRef]
- Kent, W.J. BLAT---The BLAST-Like Alignment Tool. Genome Res. 2002, 12, 656–664. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef]
- Zhao, G.; Krishnamurthy, S.; Cai, Z.; Popov, V.L.; Travassos da Rosa, A.P.; Guzman, H.; Cao, S.; Virgin, H.W.; Tesh, R.B.; Wang, D. Identification of Novel Viruses Using VirusHunter -- an Automated Data Analysis Pipeline. PLoS ONE 2013, 8, e78470. [Google Scholar] [CrossRef]
- Lee, W.-P.; Stromberg, M.P.; Ward, A.; Stewart, C.; Garrison, E.P.; Marth, G.T. MOSAIK: A Hash-Based Algorithm for Accurate Next-Generation Sequencing Short-Read Mapping. PLoS ONE 2014, 9, e90581. [Google Scholar] [CrossRef]
- Kostic, A.D.; Ojesina, A.I.; Pedamallu, C.S.; Jung, J.; Verhaak, R.G.W.; Getz, G.; Meyerson, M. PathSeq: Software to identify or discover microbes by deep sequencing of human tissue. Nat. Biotechnol. 2011, 29, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Naccache, S.N.; Federman, S.; Veeraraghavan, N.; Zaharia, M.; Lee, D.; Samayoa, E.; Bouquet, J.; Greninger, A.L.; Luk, K.-C.; Enge, B.; et al. A cloud-compatible bioinformatics pipeline for ultrarapid pathogen identification from next-generation sequencing of clinical samples. Genome Res. 2014, 24, 1180–1192. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, N.A.; Rung, J.; Brazma, A.; Marioni, J.C. Tools for mapping high-throughput sequencing data. Bioinformatics 2012, 28, 3169–3177. [Google Scholar] [CrossRef] [PubMed]
- Pickett, B.E.; Sadat, E.L.; Zhang, Y.; Noronha, J.M.; Squires, R.B.; Hunt, V.; Liu, M.; Kumar, S.; Zaremba, S.; Gu, Z.; et al. ViPR: An open bioinformatics database and analysis resource for virology research. Nucleic Acids Res. 2012, 40, D593–D598. [Google Scholar] [CrossRef]
- Goodacre, N.; Aljanahi, A.; Nandakumar, S.; Mikailov, M.; Khan, A.S. A Reference Viral Database (RVDB) To Enhance Bioinformatics Analysis of High-Throughput Sequencing for Novel Virus Detection. mSphere 2018, 3, e00069-18. [Google Scholar] [CrossRef]
- Stano, M.; Beke, G.; Klucar, L. viruSITE—integrated database for viral genomics. Database 2016, 2016, baw162. [Google Scholar] [CrossRef]
- El-Gebali, S.; Mistry, J.; Bateman, A.; Eddy, S.R.; Luciani, A.; Potter, S.C.; Qureshi, M.; Richardson, L.J.; Salazar, G.A.; Smart, A.; et al. The Pfam protein families database in 2019. Nucleic Acids Res. 2019, 47, D427–D432. [Google Scholar] [CrossRef]
- The UniProt Consortium UniProt: A worldwide hub of protein knowledge. Nucleic Acids Res. 2019, 47, D506–D515. [CrossRef]
- Ari, Ş.; Arikan, M. Next-Generation Sequencing: Advantages, Disadvantages, and Future. In Plant Omics: Trends and Applications; Hakeem, K.R., Tombuloğlu, H., Tombuloğlu, G., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 109–135. ISBN 978-3-319-31701-4. [Google Scholar]
- Břinda, K.; Callendrello, A.; Ma, K.C.; MacFadden, D.R.; Charalampous, T.; Lee, R.S.; Cowley, L.; Wadsworth, C.B.; Grad, Y.H.; Kucherov, G.; et al. Rapid heuristic inference of antibiotic resistance and susceptibility by genomic neighbor typing. Bioinformatics 2018. [Google Scholar] [CrossRef]
- Ueda, Y. New sequencing technology, third-generation Nanopore sequencing, for hepatitis C virus research. Hepatol. Res. 2019. [Google Scholar] [CrossRef]
- Astbury, S.; Soares, M.M.D.C.N.; Peprah, E.; King, B.J.; Jardim, A.C.G.; Shimizu, J.F.; Jalal, P.J.; Saeed, C.H.; Sabeer, F.T.; Irving, W.L.; et al. Extraction-free direct PCR from dried serum spots permits HBV genotyping and RAS identification by Sanger and minION sequencing. Microbiology 2019. [Google Scholar] [CrossRef]
- Kafetzopoulou, L.E.; Efthymiadis, K.; Lewandowski, K.; Crook, A.; Carter, D.; Osborne, J.; Aarons, E.; Hewson, R.; Hiscox, J.A.; Carroll, M.W.; et al. Assessment of metagenomic Nanopore and Illumina sequencing for recovering whole genome sequences of chikungunya and dengue viruses directly from clinical samples. Eurosurveillance 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Faria, N.R.; Quick, J.; Claro, I.M.; Thézé, J.; de Jesus, J.G.; Giovanetti, M.; Kraemer, M.U.G.; Hill, S.C.; Black, A.; da Costa, A.C.; et al. Establishment and cryptic transmission of Zika virus in Brazil and the Americas. Nature 2017, 546, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Batovska, J.; Lynch, S.E.; Rodoni, B.C.; Sawbridge, T.I.; Cogan, N.O. Metagenomic arbovirus detection using MinION nanopore sequencing. J. Virol. Methods 2017, 249, 79–84. [Google Scholar] [CrossRef]
- Tyler, A.D.; Mataseje, L.; Urfano, C.J.; Schmidt, L.; Antonation, K.S.; Mulvey, M.R.; Corbett, C.R. Evaluation of Oxford Nanopore’s MinION Sequencing Device for Microbial Whole Genome Sequencing Applications. Sci. Rep. 2018, 8, 10931. [Google Scholar] [CrossRef]
- Wawina-Bokalanga, T.; Vanmechelen, B.; Martí-Carreras, J.; Vergote, V.; Vermeire, K.; Muyembe-Tamfum, J.-J.; Ahuka-Mundeke, S.; Maes, P. Complete Genome Sequence of a New Ebola Virus Strain Isolated during the 2017 Likati Outbreak in the Democratic Republic of the Congo. Microbiol. Resour. Announc. 2019, 8, e00360-19. [Google Scholar] [CrossRef]
- Naito, F.Y.B.; Melo, F.L.; Fonseca, M.E.N.; Santos, C.A.F.; Chanes, C.R.; Ribeiro, B.M.; Gilbertson, R.L.; Boiteux, L.S.; de Cássia Pereira-Carvalho, R. Nanopore sequencing of a novel bipartite New World begomovirus infecting cowpea. Arch. Virol. 2019, 164, 1907–1910. [Google Scholar] [CrossRef]
- Vanmechelen, B.; Bertelsen, M.F.; Rector, A.; Van den Oord, J.J.; Laenen, L.; Vergote, V.; Maes, P. Identification of a novel species of papillomavirus in giraffe lesions using nanopore sequencing. Vet. Microbiol. 2017, 201, 26–31. [Google Scholar] [CrossRef]
- Hoenen, T.; Groseth, A.; Rosenke, K.; Fischer, R.J.; Hoenen, A.; Judson, S.D.; Martellaro, C.; Falzarano, D.; Marzi, A.; Squires, R.B.; et al. Nanopore Sequencing as a Rapidly Deployable Ebola Outbreak Tool. Emerg. Infect. Dis. 2016, 22. [Google Scholar] [CrossRef]
- Kilianski, A.; Roth, P.A.; Liem, A.T.; Hill, J.M.; Willis, K.L.; Rossmaier, R.D.; Marinich, A.V.; Maughan, M.N.; Karavis, M.A.; Kuhn, J.H.; et al. Use of Unamplified RNA/cDNA–Hybrid Nanopore Sequencing for Rapid Detection and Characterization of RNA Viruses. Emerg. Infect. Dis. 2016, 22, 1448–1451. [Google Scholar] [CrossRef]
- Hill, S.C.; de Vasconcelos, J.N.; Granja, B.G.; Thézé, J.; Jandondo, D.; Neto, Z.; Mirandela, M.; dos Santos Sebastião, C.; Cândido, A.L.M.; Clemente, C.; et al. Early Genomic Detection of Cosmopolitan Genotype of Dengue Virus Serotype 2, Angola, 2018. Emerg. Infect. Dis. 2019, 25, 784–787. [Google Scholar] [CrossRef] [PubMed]
- Check Hayden, E. Pint-sized DNA sequencer impresses first users. Nature 2015, 521, 15–16. [Google Scholar] [CrossRef] [PubMed]
- Croville, G.; Le Loc’h, G.; Zanchetta, C.; Manno, M.; Camus-Bouclainville, C.; Klopp, C.; Delverdier, M.; Lucas, M.-N.; Donnadieu, C.; Delpont, M.; et al. Rapid whole-genome based typing and surveillance of avipoxviruses using nanopore sequencing. J. Virol. Methods 2018, 261, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Russell, J.A.; Campos, B.; Stone, J.; Blosser, E.M.; Burkett-Cadena, N.; Jacobs, J.L. Unbiased Strain-Typing of Arbovirus Directly from Mosquitoes Using Nanopore Sequencing: A Field-forward Biosurveillance Protocol. Sci. Rep. 2018, 8, 5417. [Google Scholar] [CrossRef] [PubMed]
- Garalde, D.R.; Snell, E.A.; Jachimowicz, D.; Sipos, B.; Lloyd, J.H.; Bruce, M.; Pantic, N.; Admassu, T.; James, P.; Warland, A.; et al. Highly parallel direct RNA sequencing on an array of nanopores. Nat. Methods 2018, 15, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Keller, M.W.; Rambo-Martin, B.L.; Wilson, M.M.; Ridenour, C.A.; Shepard, S.S.; Stark, T.J.; Neuhaus, E.B.; Dugan, V.G.; Wentworth, D.E.; Barnes, J.R. Direct RNA Sequencing of the Coding Complete Influenza A Virus Genome. Sci. Rep. 2018, 8, 14408. [Google Scholar] [CrossRef]
- Jenjaroenpun, P.; Wongsurawat, T.; Pereira, R.; Patumcharoenpol, P.; Ussery, D.W.; Nielsen, J.; Nookaew, I. Complete genomic and transcriptional landscape analysis using third-generation sequencing: A case study of Saccharomyces cerevisiae CEN.PK113-7D. Nucleic Acids Res. 2018, 46, e38. [Google Scholar] [CrossRef]
- Depledge, D.P.; Srinivas, K.P.; Sadaoka, T.; Bready, D.; Mori, Y.; Placantonakis, D.G.; Mohr, I.; Wilson, A.C. Direct RNA sequencing on nanopore arrays redefines the transcriptional complexity of a viral pathogen. Nat. Commun. 2019, 10, 754. [Google Scholar] [CrossRef]
- Boldogkői, Z.; Moldován, N.; Balázs, Z.; Snyder, M.; Tombácz, D. Long-Read Sequencing—A Powerful Tool in Viral Transcriptome Research. Trends Microbiol. 2019, 27, 578–592. [Google Scholar] [CrossRef]
- Xu, Y.; Lewandowski, K.; Lumley, S.; Pullan, S.; Vipond, R.; Carroll, M.; Foster, D.; Matthews, P.C.; Peto, T.; Crook, D. Detection of Viral Pathogens With Multiplex Nanopore MinION Sequencing: Be Careful With Cross-Talk. Front. Microbiol. 2018, 9, 2225. [Google Scholar] [CrossRef]
- Erlwein, O.; Robinson, M.J.; Dustan, S.; Weber, J.; Kaye, S.; McClure, M.O. DNA Extraction Columns Contaminated with Murine Sequences. PLoS ONE 2011, 6, e23484. [Google Scholar] [CrossRef] [PubMed]
- Karamitros, T.; Magiorkinis, G. A novel method for the multiplexed target enrichment of MinION next generation sequencing libraries using PCR-generated baits. Nucleic Acids Res. 2015, 43, e152. [Google Scholar] [CrossRef] [PubMed]
- Vinaya Kumar, K.; Shekhar, M.S.; Otta, S.K.; Karthic, K.; Ashok Kumar, J.; Gopikrishna, G.; Vijayan, K.K. First Report of a Complete Genome Sequence of White spot syndrome virus from India. Genome Announc. 2018, 6, e00055-18. [Google Scholar] [CrossRef] [PubMed]
- Günther, T.; Haas, L.; Alawi, M.; Wohlsein, P.; Marks, J.; Grundhoff, A.; Becher, P.; Fischer, N. Recovery of the first full-length genome sequence of a parapoxvirus directly from a clinical sample. Sci. Rep. 2017, 7, 3734. [Google Scholar] [CrossRef]
- Warwick-Dugdale, J.; Solonenko, N.; Moore, K.; Chittick, L.; Gregory, A.C.; Allen, M.J.; Sullivan, M.B.; Temperton, B. Long-read viral metagenomics captures abundant and microdiverse viral populations and their niche-defining genomic islands. PeerJ 2019, 7, e6800. [Google Scholar] [CrossRef]
- Peserico, A.; Marcacci, M.; Malatesta, D.; Di Domenico, M.; Pratelli, A.; Mangone, I.; D’Alterio, N.; Pizzurro, F.; Cirone, F.; Zaccaria, G.; et al. Diagnosis and characterization of canine distemper virus through sequencing by MinION nanopore technology. Sci. Rep. 2019, 9, 1714. [Google Scholar] [CrossRef]
- Theuns, S.; Vanmechelen, B.; Bernaert, Q.; Deboutte, W.; Vandenhole, M.; Beller, L.; Matthijnssens, J.; Maes, P.; Nauwynck, H.J. Nanopore sequencing as a revolutionary diagnostic tool for porcine viral enteric disease complexes identifies porcine kobuvirus as an important enteric virus. Sci. Rep. 2018, 8, 9830. [Google Scholar] [CrossRef]
- Bronzato Badial, A.; Sherman, D.; Stone, A.; Gopakumar, A.; Wilson, V.; Schneider, W.; King, J. Nanopore Sequencing as a Surveillance Tool for Plant Pathogens in Plant and Insect Tissues. Plant Dis. 2018, 102, 1648–1652. [Google Scholar] [CrossRef]
- Gallagher, M.D.; Matejusova, I.; Nguyen, L.; Ruane, N.M.; Falk, K.; Macqueen, D.J. Nanopore sequencing for rapid diagnostics of salmonid RNA viruses. Sci. Rep. 2018, 8, 16307. [Google Scholar] [CrossRef]
- Ameur, A.; Kloosterman, W.P.; Hestand, M.S. Single-Molecule Sequencing: Towards Clinical Applications. Trends Biotechnol. 2019, 37, 72–85. [Google Scholar] [CrossRef]
- Houldcroft, C.J.; Breuer, J. Tales from the crypt and coral reef: The successes and challenges of identifying new herpesviruses using metagenomics. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Munro, A.C.; Houldcroft, C. Human cancers and mammalian retroviruses: Should we worry about bovine leukemia virus? Future Virol. 2016, 11, 163–166. [Google Scholar] [CrossRef]
- Hue, S.; Gray, E.R.; Gall, A.; Katzourakis, A.; Tan, C.P.; Houldcroft, C.J.; McLaren, S.; Pillay, D.; Futreal, A.; Garson, J.A.; et al. Disease-associated XMRV sequences are consistent with laboratory contamination. Retrovirology 2010, 7, 111. [Google Scholar] [CrossRef] [PubMed]
- Naccache, S.N.; Greninger, A.L.; Lee, D.; Coffey, L.L.; Phan, T.; Rein-Weston, A.; Aronsohn, A.; Hackett, J.; Delwart, E.L.; Chiu, C.Y. The Perils of Pathogen Discovery: Origin of a Novel Parvovirus-Like Hybrid Genome Traced to Nucleic Acid Extraction Spin Columns. J. Virol. 2013, 87, 11966–11977. [Google Scholar] [CrossRef]
- Salter, S.J.; Cox, M.J.; Turek, E.M.; Calus, S.T.; Cookson, W.O.; Moffatt, M.F.; Turner, P.; Parkhill, J.; Loman, N.J.; Walker, A.W. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 2014, 12, 87. [Google Scholar] [CrossRef] [PubMed]
- Matsvay, A.D.; Alborova, I.E.; Pimkina, E.V.; Markelov, M.L.; Khafizov, K.; Mustafin, K.K. Experimental approaches for ancient DNA extraction and sample preparation for next generation sequencing in ultra-clean conditions. Conserv. Genet. Resour. 2019, 11, 345–353. [Google Scholar] [CrossRef]
- Morfopoulou, S.; Brown, J.R.; Davies, E.G.; Anderson, G.; Virasami, A.; Qasim, W.; Chong, W.K.; Hubank, M.; Plagnol, V.; Desforges, M.; et al. Human Coronavirus OC43 Associated with Fatal Encephalitis. N. Engl. J. Med. 2016, 375, 497–498. [Google Scholar] [CrossRef]
- Lipkin, W.I. A Vision for Investigating the Microbiology of Health and Disease. J. Infect. Dis. 2015, 212, S26–S30. [Google Scholar] [CrossRef][Green Version]
- Hall, R.J.; Draper, J.L.; Nielsen, F.G.G.; Dutilh, B.E. Beyond research: A primer for considerations on using viral metagenomics in the field and clinic. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Shafer, R.W. Rationale and Uses of a Public HIV Drug-Resistance Database. J. Infect. Dis. 2006, 194, S51–S58. [Google Scholar] [CrossRef]
- Gnaneshan, S.; Ijaz, S.; Moran, J.; Ramsay, M.; Green, J. HepSEQ: International Public Health Repository for Hepatitis B. Nucleic Acids Res. 2007, 35, D367–D370. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rhee, S.-Y.; Margeridon-Thermet, S.; Nguyen, M.H.; Liu, T.F.; Kagan, R.M.; Beggel, B.; Verheyen, J.; Kaiser, R.; Shafer, R.W. Hepatitis B virus reverse transcriptase sequence variant database for sequence analysis and mutation discovery. Antivir. Res. 2010, 88, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Kuiken, C.; Yusim, K.; Boykin, L.; Richardson, R. The Los Alamos hepatitis C sequence database. Bioinformatics 2005, 21, 379–384. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kiselev, D.; Matsvay, A.; Abramov, I.; Dedkov, V.; Shipulin, G.; Khafizov, K. Current Trends in Diagnostics of Viral Infections of Unknown Etiology. Viruses 2020, 12, 211. https://doi.org/10.3390/v12020211
Kiselev D, Matsvay A, Abramov I, Dedkov V, Shipulin G, Khafizov K. Current Trends in Diagnostics of Viral Infections of Unknown Etiology. Viruses. 2020; 12(2):211. https://doi.org/10.3390/v12020211
Chicago/Turabian StyleKiselev, Daniel, Alina Matsvay, Ivan Abramov, Vladimir Dedkov, German Shipulin, and Kamil Khafizov. 2020. "Current Trends in Diagnostics of Viral Infections of Unknown Etiology" Viruses 12, no. 2: 211. https://doi.org/10.3390/v12020211
APA StyleKiselev, D., Matsvay, A., Abramov, I., Dedkov, V., Shipulin, G., & Khafizov, K. (2020). Current Trends in Diagnostics of Viral Infections of Unknown Etiology. Viruses, 12(2), 211. https://doi.org/10.3390/v12020211






