Functional Analyses of Bovine Foamy Virus-Encoded miRNAs Reveal the Importance of a Defined miRNA for Virus Replication and Host–Virus Interaction
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture and DNA and miRNA Transfection Techniques
2.2. Molecular Cloning
2.3. Dual Luciferase Reporter Assays
2.4. Target Gene Quantification
2.5. In Silico Target Gene Prediction and Statistical Analyses
2.6. Virological Methods
2.7. Protein Analysis by Immunoblotting
3. Results
3.1. In Silico Target Prediction and Ranking
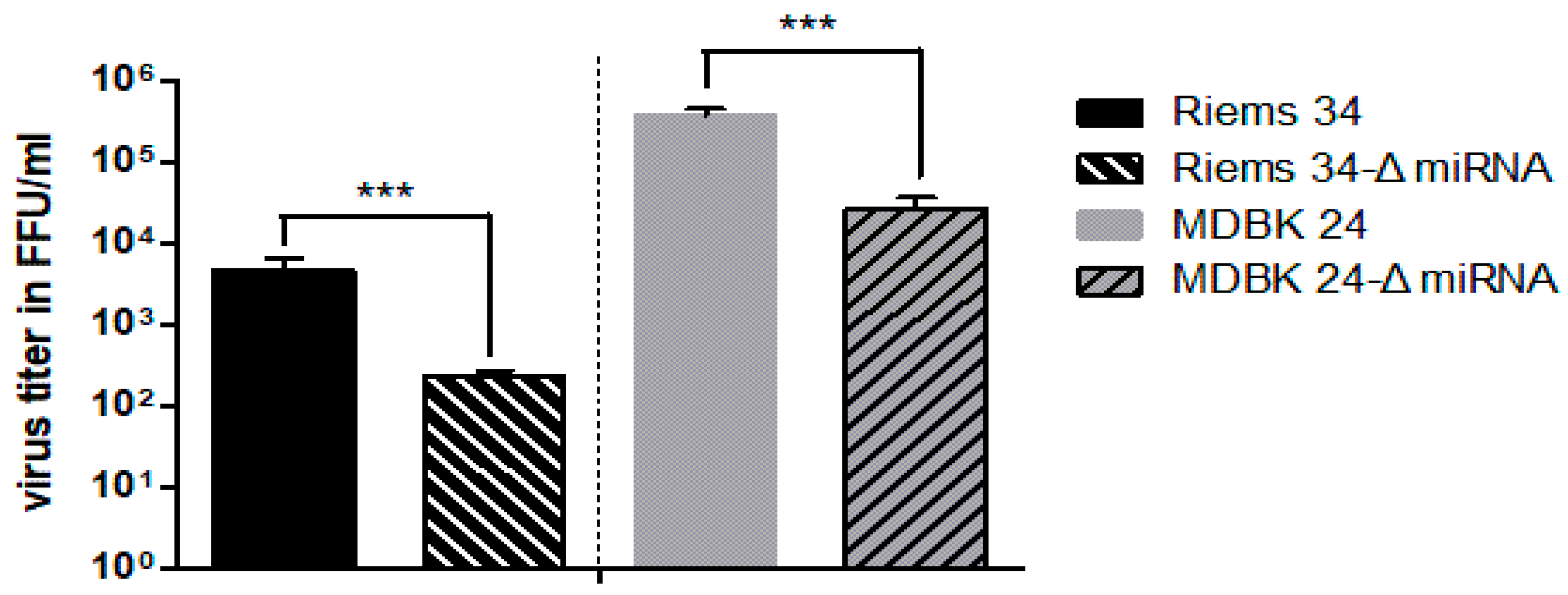
3.2. Importance of BFV miRNA Expression for Overall BFV Replication
3.3. Deletion of the miRNA Cassette Does Not Inhibit Particle Release and Gag and Pol Expression, Processing and Packaging
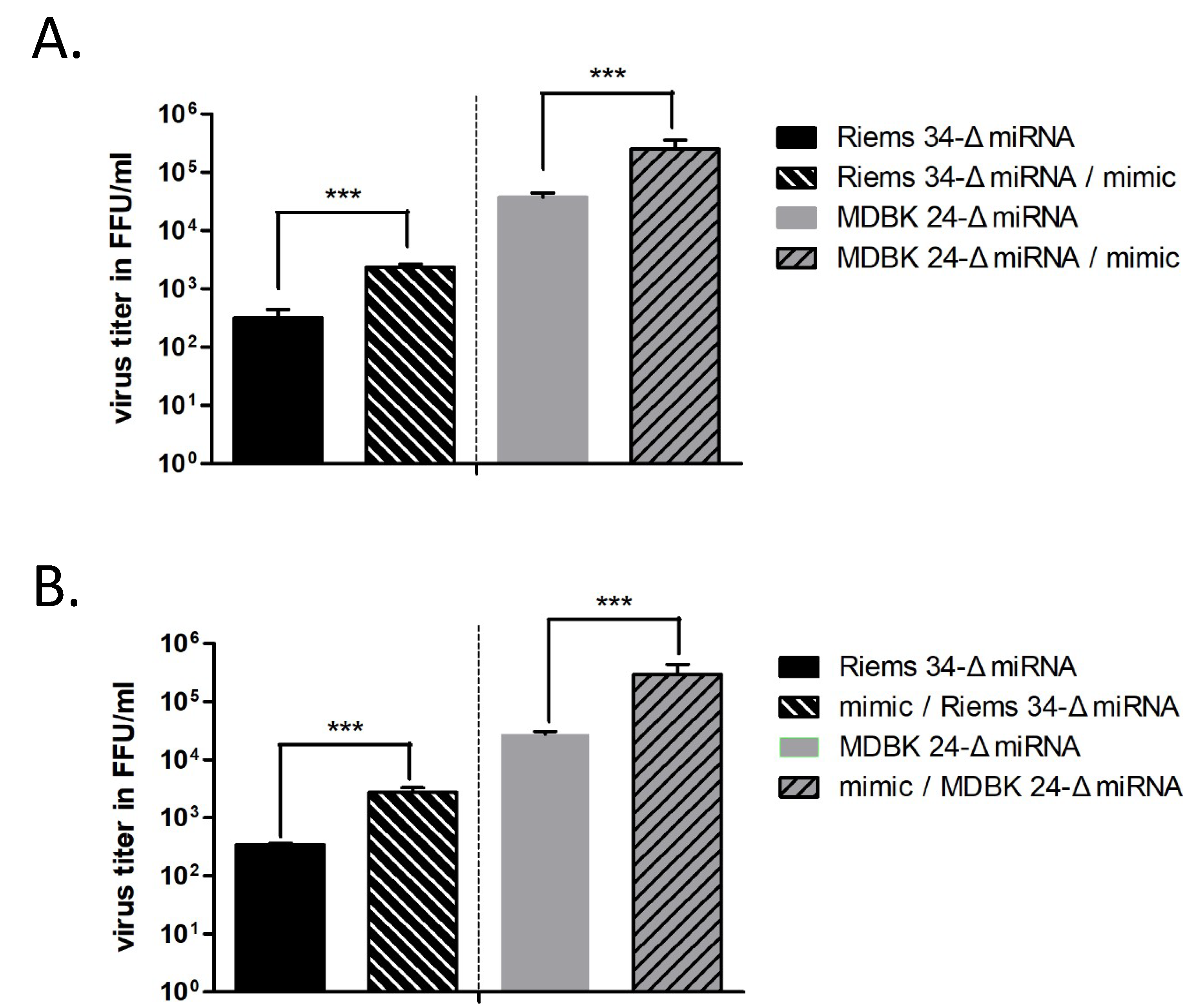
3.4. Importance of the BF2-5p miRNA for BFV Replication
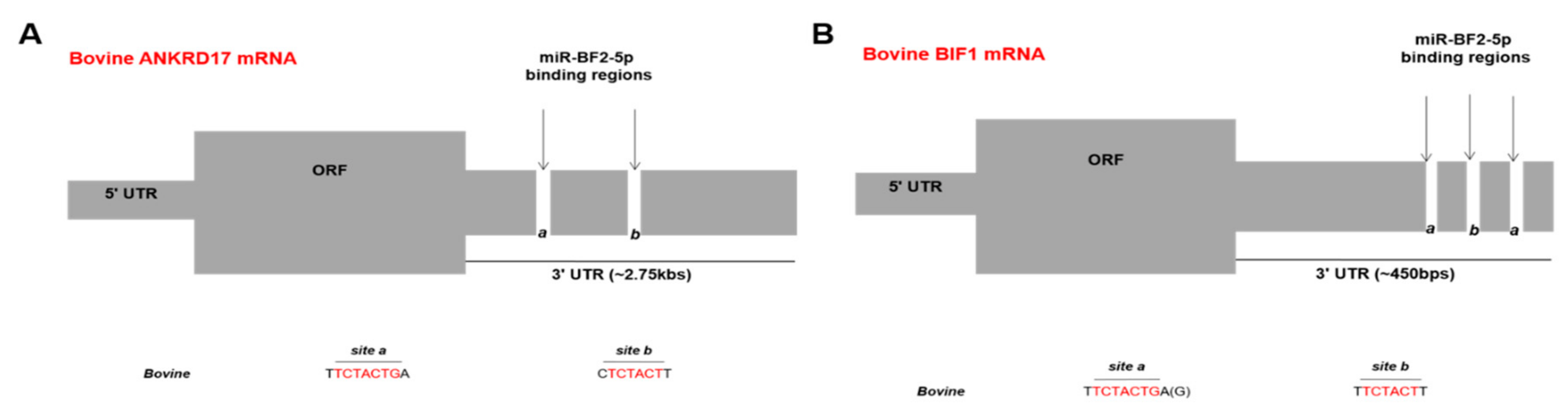
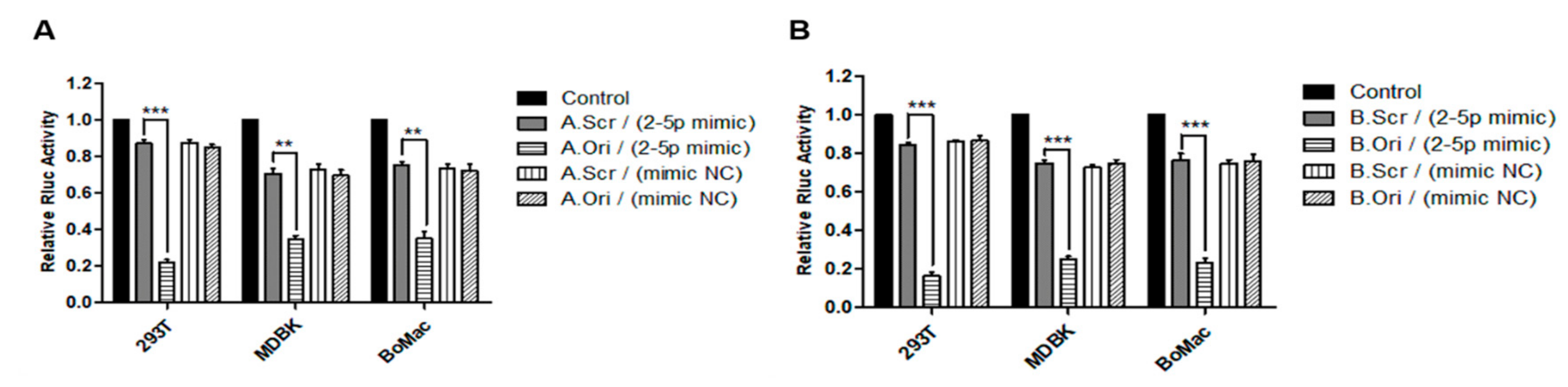
3.5. In Vitro Validation of ANKRD17 and Bif1 as Direct Targets of miR-BF2-5p
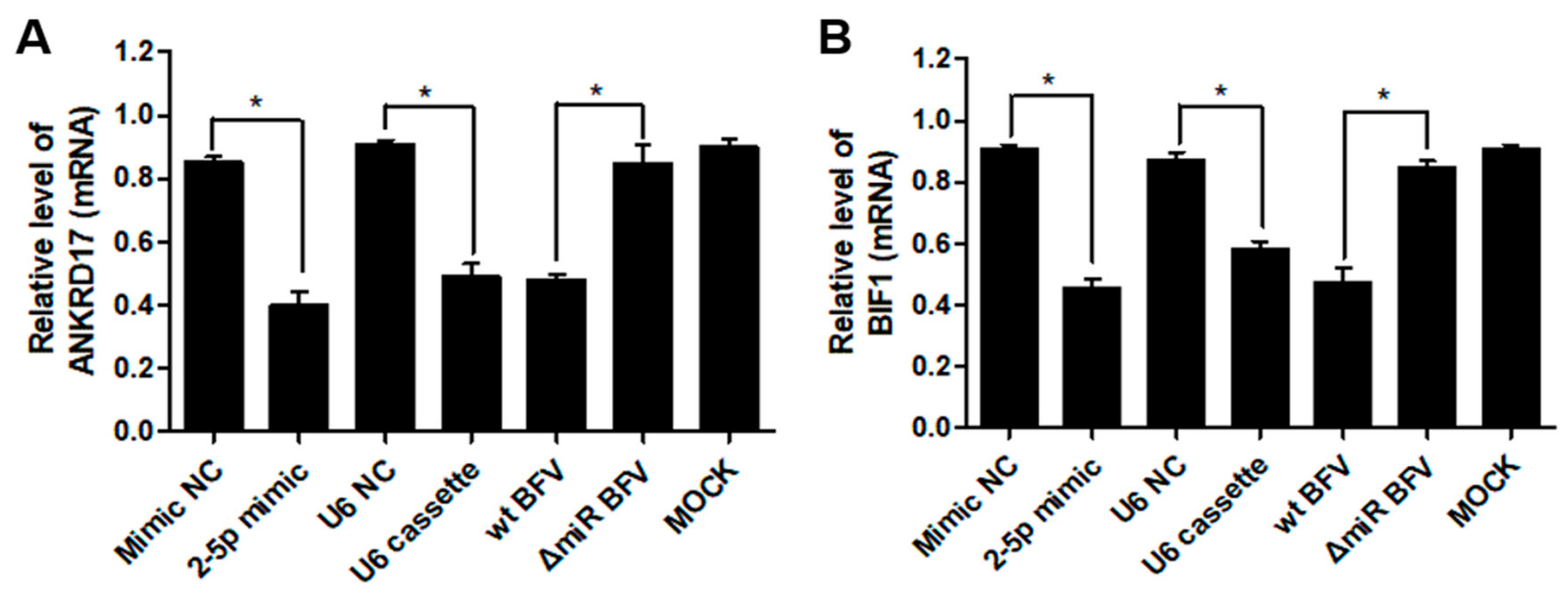
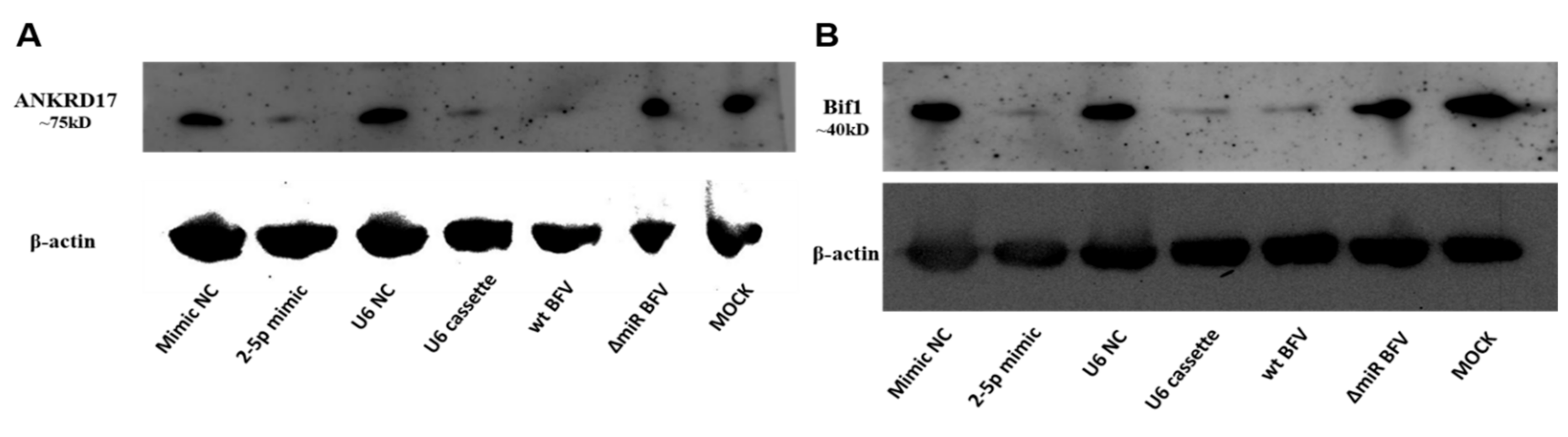
3.6. MiR-BF2-5p Suppresses Expression of Innate Immunity Genes as Predicted Downstream Targets of ANKRD17
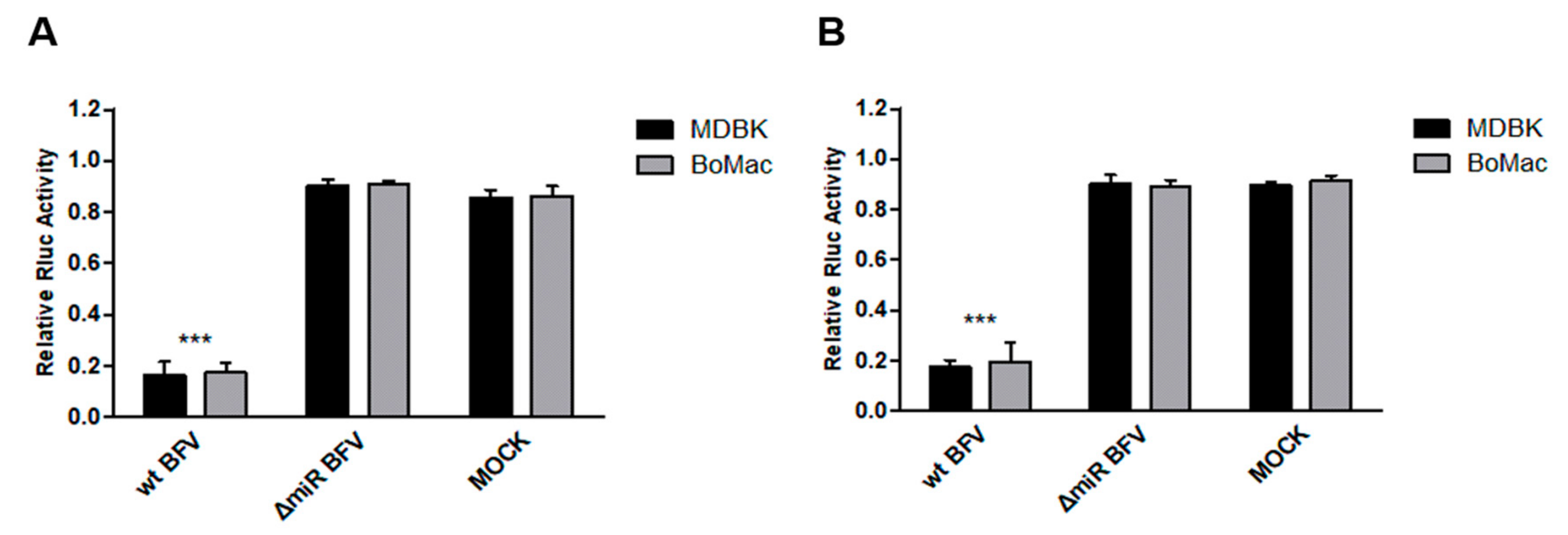
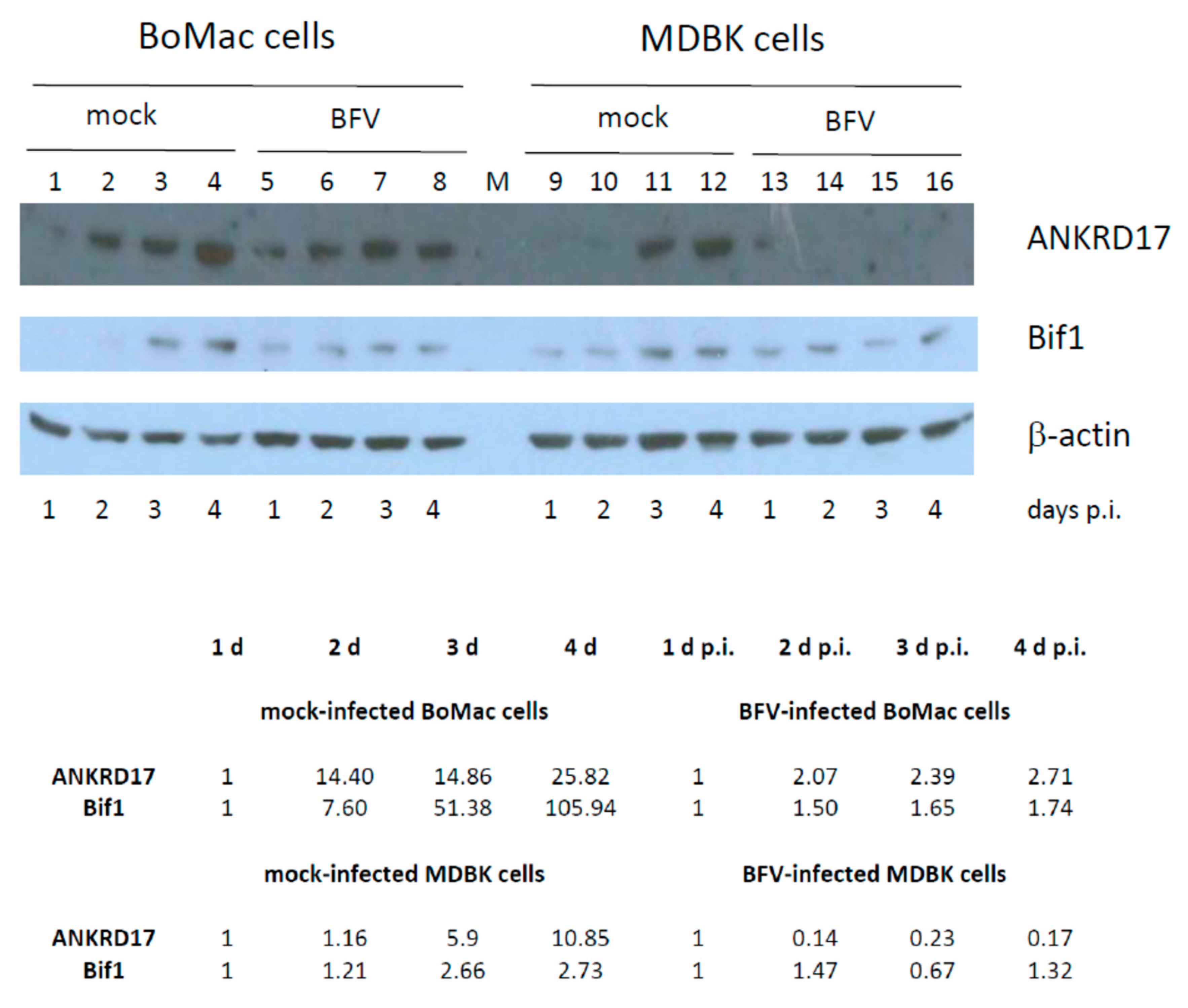
3.7. Kinetics of ANKRD17 and Bif1 Suppression during Productive BFV Infection of MDBK and BoMac Cells
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cullen, B.R. MicroRNA expression by an oncogenic retrovirus. Proc. Natl. Acad. Sci. USA 2012, 109, 2695–2696. [Google Scholar] [CrossRef]
- Cullen, B.R. MicroRNAs as mediators of viral evasion of the immune system. Nat. Immunol. 2013, 14, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef] [PubMed]
- Skalsky, R.L.; Cullen, B.R. Viruses, microRNAs, and host interactions. Annu. Rev. Microbiol. 2010, 64, 123–141. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Smith, L.P.; Nair, V.; Watson, M. An avian retrovirus uses canonical expression and processing mechanisms to generate viral microRNA. J. Virol. 2014, 88, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Balasubramaniam, M.; Pandhare, J.; Dash, C. Are microRNAs Important Players in HIV-1 Infection? An Update. Viruses 2018, 10, 110. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R.P.; Burke, J.M.; Sullivan, C.S. RNA virus microRNA that mimics a B-cell oncomiR. Proc. Natl. Acad. Sci. USA 2012, 109, 3077–3082. [Google Scholar] [CrossRef]
- Whisnant, A.W.; Kehl, T.; Bao, Q.; Materniak, M.; Kuzmak, J.; Löchelt, M.; Cullen, B.R. Identification of novel, highly expressed retroviral microRNAs in cells infected by bovine foamy virus. J. Virol. 2014, 88, 4679–4686. [Google Scholar] [CrossRef] [PubMed]
- Kincaid, R.P.; Chen, Y.; Cox, J.E.; Rethwilm, A.; Sullivan, C.S. Noncanonical microRNA (miRNA) biogenesis gives rise to retroviral mimics of lymphoproliferative and immunosuppressive host miRNAs. MBio 2014, 5, e00074. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto-Gotoh, A.; Kitao, K.; Miyazawa, T. Persistent Infection of Simian Foamy Virus Derived from the Japanese Macaque Leads to the High-Level Expression of microRNA that Resembles the miR-1 microRNA Precursor Family. Microbes Environ. 2020, 35. [Google Scholar] [CrossRef]
- Khan, A.S.; Bodem, J.; Buseyne, F.; Gessain, A.; Johnson, W.; Kuhn, J.H.; Kuzmak, J.; Lindemann, D.; Linial, M.L.; Löchelt, M.; et al. Spumaretroviruses: Updated taxonomy and nomenclature. Virology 2018, 516, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Kehl, T.; Tan, J.; Materniak, M. Non-simian foamy viruses: Molecular virology, tropism and prevalence and zoonotic/interspecies transmission. Viruses 2013, 5, 2169. [Google Scholar] [CrossRef]
- Materniak, M.; Tan, J.; Heit-Mondrzyk, A.; Hotz-Wagenblatt, A.; Löchelt, M. Bovine Foamy Virus: Shared and unique molecular features in vitro and in vivo. Viruses 2019, 11, 1084. [Google Scholar] [CrossRef]
- Lindemann, D.; Rethwilm, A. Foamy virus biology and its application for vector development. Viruses 2011, 3, 561–585. [Google Scholar] [CrossRef] [PubMed]
- Rethwilm, A. Molecular biology of foamy viruses. Med. Microbiol. Immunol. 2010, 199, 197–207. [Google Scholar] [CrossRef]
- Rethwilm, A. Foamy virus vectors: An awaited alternative to gammaretro- and lentiviral vectors. Curr. Gene Ther. 2007, 7, 261–271. [Google Scholar] [CrossRef]
- Pinto-Santini, D.M.; Stenbak, C.R.; Linial, M.L. Foamy virus zoonotic infections. Retrovirology 2017, 14, 55. [Google Scholar] [CrossRef]
- Romen, F.; Backes, P.; Materniak, M.; Sting, R.; Vahlenkamp, T.W.; Riebe, R.; Pawlita, M.; Kuzmak, J.; Löchelt, M. Serological detection systems for identification of cows shedding bovine foamy virus via milk. Virology 2007, 364, 123–131. [Google Scholar] [CrossRef]
- Cao, W.; Heit, A.; Hotz-Wagenblatt, A.; Löchelt, M. Functional characterization of the bovine foamy virus miRNA expression cassette and its dumbbell-shaped pri-miRNA. Virus Genes 2018, 54, 550–560. [Google Scholar] [CrossRef]
- Menning, M.; Kufer, T.A. A role for the Ankyrin repeat containing protein Ankrd17 in Nod1- and Nod2-mediated inflammatory responses. FEBS Lett. 2013, 587, 2137–2142. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, Y.; Wu, J.; Roizman, B.; Zhou, G.G. Innate responses to gene knockouts impact overlapping gene networks and vary with respect to resistance to viral infection. Proc. Natl. Acad. Sci. USA 2018, 115, E3230–E3237. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tong, X.; Li, G.; Li, J.; Deng, M.; Ye, X. Ankrd17 positively regulates RIG-I-like receptor (RLR)-mediated immune signaling. Eur. J. Immunol. 2012, 42, 1304–1315. [Google Scholar] [CrossRef] [PubMed]
- Piersanti, R.L.; Horlock, A.D.; Block, J.; Santos, J.E.P.; Sheldon, I.M.; Bromfield, J.J. Persistent effects on bovine granulosa cell transcriptome after resolution of uterine disease. Reproduction 2019, 158, 35–46. [Google Scholar] [CrossRef]
- Takahashi, Y.; Meyerkord, C.L.; Hori, T.; Runkle, K.; Fox, T.E.; Kester, M.; Loughran, T.P.; Wang, H.G. Bif-1 regulates Atg9 trafficking by mediating the fission of Golgi membranes during autophagy. Autophagy 2011, 7, 61–73. [Google Scholar] [CrossRef]
- Xu, L.; Wang, Z.; He, S.Y.; Zhang, S.F.; Luo, H.J.; Zhou, K.; Li, X.F.; Qiu, S.P.; Cao, K.Y. Bax-interacting factor-1 inhibits cell proliferation and promotes apoptosis in prostate cancer cells. Oncol. Rep. 2016, 36, 3513–3521. [Google Scholar] [CrossRef]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Stabel, J.R.; Stabel, T.J. Immortalization and characterization of bovine peritoneal macrophages transfected with SV40 plasmid DNA. Vet. Immunol. Immunopathol. 1995, 45, 211–220. [Google Scholar] [CrossRef]
- Bao, Q.; Hipp, M.; Hugo, A.; Lei, J.; Liu, Y.; Kehl, T.; Hechler, T.; Löchelt, M. In Vitro Evolution of Bovine Foamy Virus Variants with Enhanced Cell-Free Virus Titers and Transmission. Viruses 2015, 7, 5855–5874. [Google Scholar] [CrossRef]
- Hechler, T.; Materniak, M.; Kehl, T.; Kuzmak, J.; Löchelt, M. Complete genome sequences of two novel European clade bovine foamy viruses from Germany and Poland. J. Virol. 2012, 86, 10905–10906. [Google Scholar] [CrossRef]
- Bao, Q.; Hotz-Wagenblatt, A.; Betts, M.J.; Hipp, M.; Hugo, A.; Pougialis, G.; Lei-Rossmann, J.; Löchelt, M. Shared and cell type-specific adaptation strategies of Gag and Env yield high titer bovine foamy virus variants. Infect. Genet. Evol. 2020, 82. [Google Scholar] [CrossRef]
- Schwantes, A.; Ortlepp, I.; Löchelt, M. Construction and functional characterization of feline foamy virus-based retroviral vectors. Virology 2002, 301, 53–63. [Google Scholar] [CrossRef]
- Lei, J.; Osen, W.; Gardyan, A.; Hotz-Wagenblatt, A.; Wei, G.; Gissmann, L.; Eichmüller, S.; Löchelt, M. Replication-Competent Foamy Virus Vaccine Vectors as Novel Epitope Scaffolds for Immunotherapy. PLoS ONE 2015, 10, e0138458. [Google Scholar] [CrossRef] [PubMed]
- Gibson, D.G.; Young, L.; Chuang, R.Y.; Venter, J.C.; Hutchison, C.A., 3rd; Smith, H.O. Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat. Methods 2009, 6, 343–345. [Google Scholar] [CrossRef] [PubMed]
- Stricker, E.; Pougialis, G.; Löchelt, M. Features of Engineered Bovine Foamy Virus Genomes; German Cancer Research Center (DKFZ): Heidelberg, Germany, 2018. [Google Scholar]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 2001, 29, e45. [Google Scholar] [CrossRef]
- Lewis, B.P.; Shih, I.H.; Jones-Rhoades, M.W.; Bartel, D.P.; Burge, C.B. Prediction of mammalian microRNA targets. Cell 2003, 115, 787–798. [Google Scholar] [CrossRef]
- John, B.; Enright, A.J.; Aravin, A.; Tuschl, T.; Sander, C.; Marks, D.S. Human MicroRNA targets. PLoS Biol. 2004, 2, e363. [Google Scholar] [CrossRef]
- Gebert, L.F.R.; MacRae, I.J. Regulation of microRNA function in animals. Nat. Rev. Mol. Cell Biol. 2019, 20, 21–37. [Google Scholar] [CrossRef]
- Hutter, S.; Mullers, E.; Stanke, N.; Reh, J.; Lindemann, D. Prototype foamy virus protease activity is essential for intraparticle reverse transcription initiation but not absolutely required for uncoating upon host cell entry. J. Virol. 2013, 87, 3163–3176. [Google Scholar] [CrossRef]
- Materniak, M.; Hechler, T.; Löchelt, M.; Kuzmak, J. Similar patterns of infection with bovine foamy virus in experimentally inoculated calves and sheep. J. Virol. 2013, 87, 3516–3525. [Google Scholar] [CrossRef] [PubMed]
- Liebermann, H.; Riebe, R. Isolation of bovine syncytial virus in East Germany. Arch. Exp. Vet. 1981, 35, 917–919. [Google Scholar]
- Stricker, E. Influence of LTR Variants on Bovine Foamy Virus Replication Kinetics and Gene Expression. Master‘s Thesis, University of Heidelberg, Heidelberg, Germany, 2017. [Google Scholar]
- Wei, G.; Kehl, T.; Bao, Q.; Benner, A.; Lei, J.; Löchelt, M. The chromatin binding domain, including the QPQRYG motif, of feline foamy virus Gag is required for viral DNA integration and nuclear accumulation of Gag and the viral genome. Virology 2018, 524, 56–68. [Google Scholar] [CrossRef]
- Cao, W. Functional Characterization and Target Validation of the Bovine Foamy Virus Encoded miRNAs. Ph.D. Thesis, University of Heidelberg, Heidelberg, Germany, 2018. [Google Scholar]
- Hill, C.G.; Matyunina, L.V.; Walker, D.; Benigno, B.B.; McDonald, J.F. Transcriptional override: A regulatory network model of indirect responses to modulations in microRNA expression. BMC Syst. Biol. 2014, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Hao, P.; Jia, R.; Yang, W.; Liu, R.; Wang, J.; Xi, Z.; Geng, Y.; Qiao, W. Identification and functional characterization of BTas transactivator as a DNA-binding protein. Virology 2010, 405, 408–413. [Google Scholar] [CrossRef] [PubMed]
- Swiersy, A.; Wiek, C.; Reh, J.; Zentgraf, H.; Lindemann, D. Orthoretroviral-like prototype foamy virus Gag-Pol expression is compatible with viral replication. Retrovirology 2011, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Wilk, T.; Geiselhart, V.; Frech, M.; Fuller, S.D.; Flugel, R.M.; Löchelt, M. Specific interaction of a novel foamy virus Env leader protein with the N-terminal Gag domain. J. Virol. 2001, 75, 7995–8007. [Google Scholar] [CrossRef]
- Mukherjee, T.; Hovingh, E.S.; Foerster, E.G.; Abdel-Nour, M.; Philpott, D.J.; Girardin, S.E. NOD1 and NOD2 in inflammation, immunity and disease. Arch. Biochem. Biophys. 2019, 670, 69–81. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.M.; Chow, V.T. The VP1 structural protein of enterovirus 71 interacts with human ornithine decarboxylase and gene trap ankyrin repeat. Microb. Pathog. 2007, 42, 129–137. [Google Scholar] [CrossRef]
- Wang, H.; Liu, X.; Shu, S.; Zhang, J.; Huang, Y.; Fang, F. Murine cytomegalovirus IE3 protein interacts with Ankrd17. J. Huazhong Univ. Sci. Technol. Med. Sci. 2011, 31, 285–289. [Google Scholar] [CrossRef]
- Bergez, M.; Weber, J.; Riess, M.; Erdbeer, A.; Seifried, J.; Stanke, N.; Munz, C.; Hornung, V.; Konig, R.; Lindemann, D. Insights into Innate Sensing of Prototype Foamy Viruses in Myeloid Cells. Viruses 2019, 11, 1095. [Google Scholar] [CrossRef]
- Rua, R.; Lepelley, A.; Gessain, A.; Schwartz, O. Innate sensing of foamy viruses by human hematopoietic cells. J. Virol. 2012, 86, 909–918. [Google Scholar] [CrossRef]
- Bahr, A.; Singer, A.; Hain, A.; Vasudevan, A.A.; Schilling, M.; Reh, J.; Riess, M.; Panitz, S.; Serrano, V.; Schweizer, M.; et al. Interferon but not MxB inhibits foamy retroviruses. Virology 2016, 488, 51–60. [Google Scholar] [CrossRef]
- Rola-Luszczak, M.; Materniak, M.; Pluta, A.; Hulst, M.; Kuzmak, J. Transcriptomic microarray analysis of BoMac cells after infection with bovine foamy virus. Arch. Virol. 2014, 159, 1515–1519. [Google Scholar] [CrossRef] [PubMed]
- Serfass, J.M.; Takahashi, Y.; Zhou, Z.; Kawasawa, Y.I.; Liu, Y.; Tsotakos, N.; Young, M.M.; Tang, Z.; Yang, L.; Atkinson, J.M.; et al. Endophilin B2 facilitates endosome maturation in response to growth factor stimulation, autophagy induction, and influenza A virus infection. J. Biol. Chem. 2017, 292, 10097–10111. [Google Scholar] [CrossRef] [PubMed]
- Santamaria, E.; Mora, M.I.; Potel, C.; Fernandez-Irigoyen, J.; Carro-Roldan, E.; Hernandez-Alcoceba, R.; Prieto, J.; Epstein, A.L.; Corrales, F.J. Identification of replication-competent HSV-1 Cgal+ strain signaling targets in human hepatoma cells by functional organelle proteomics. Mol. Cell. Proteom. 2009, 8, 805–815. [Google Scholar] [CrossRef]
- Yuan, P.; Dong, L.; Cheng, Q.; Wang, S.; Li, Z.; Sun, Y.; Han, S.; Yin, J.; Peng, B.; He, X.; et al. Prototype foamy virus elicits complete autophagy involving the ER stress-related UPR pathway. Retrovirology 2017, 14, 16. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhu, G.; Yan, J.; Tang, Y.; Han, S.; Yin, J.; Peng, B.; He, X.; Liu, W. The Late Domain of Prototype Foamy Virus Gag Facilitates Autophagic Clearance of Stress Granules by Promoting Amphisome Formation. J. Virol. 2020, 94. [Google Scholar] [CrossRef]
- Schmidt, M.; Herchenroder, O.; Heeney, J.; Rethwilm, A. Long terminal repeat U3 length polymorphism of human foamy virus. Virology 1997, 230, 167–178. [Google Scholar] [CrossRef]
- Cai, Q.; He, B.; Weiberg, A.; Buck, A.H.; Jin, H. Small RNAs and extracellular vesicles: New mechanisms of cross-species communication and innovative tools for disease control. PLoS Pathog. 2019, 15, e1008090. [Google Scholar] [CrossRef]
- Gurien, S.D.; Aziz, M.; Jin, H.; Wang, H.; He, M.; Al-Abed, Y.; Nicastro, J.M.; Coppa, G.F.; Wang, P. Extracellular microRNA 130b-3p inhibits eCIRP-induced inflammation. EMBO Rep. 2020, 21, e48075. [Google Scholar] [CrossRef]
- Van Looveren, K.; Van Wyngene, L.; Libert, C. An extracellular microRNA can rescue lives in sepsis. EMBO Rep. 2020, 21, e49193. [Google Scholar] [CrossRef] [PubMed]
- Safari, R.; Hamaidia, M.; De Brogniez, A.; Gillet, N.; Willems, L. Cis-drivers and trans-drivers of bovine leukemia virus oncogenesis. Curr. Opin. Virol. 2017, 26, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Gillet, N.A.; Hamaidia, M.; De Brogniez, A.; Gutierrez, G.; Renotte, N.; Reichert, M.; Trono, K.; Willems, L. Bovine Leukemia Virus Small Noncoding RNAs Are Functional Elements That Regulate Replication and Contribute to Oncogenesis In Vivo. PLoS Pathog. 2016, 12, e1005588. [Google Scholar] [CrossRef] [PubMed]


| miR-BF1-5p | miR-BF1-3p | miR-BF2-5p | ||||
|---|---|---|---|---|---|---|
| Target Rank | Target Score 1 | Gene Symbol | Target Score 1 | Gene Symbol | Target Score 1 | Gene Symbol |
| 1 | 80 | BCL7A | 62 | TMEM135 | 100 | ANKRD17 |
| 2 | 73 | DUSP4 | 58 | PHF20 | 99 | Bif1 (SH3GLB1) |
| 3 | 67 | BCKDHB | 58 | BRWD1 | 99 | AFAP |
| 4 | 64 | LYPD1 | 51 | SUN1 | 98 | AK3 |
| 5 | 64 | RAD23A | 51 | DUSP6 | 96 | FLRT3 |
| 6 | 62 | ANKRD26 | 50 | PRKCI | 96 | ARL6IP1 |
| 7 | 62 | GAPVD1 | 41 | FAM234B | 96 | ADAMTSL3 |
| 8 | 59 | PPP1R13B | 37 | PI4K2B | 96 | PTER |
| 9 | 59 | OOSP2 | 35 | USP38 | 96 | BTBD7 |
| 10 | 58 | ZNF701 | 33 | UBE2G1 | 96 | MON2 |
| 11 | 57 | ZMYND11 | 29 | ZFP62 | 96 | UTRN |
| 12 | 56 | MKI67 | 28 | COL4A1 | 96 | GABRA1 |
| 13 | 56 | GCC2 | 28 | ABL2 | 96 | DGKI |
| 14 | 55 | ATCAY | 27 | PPP2CA | 95 | CTDSPL2 |
| 15 | 54 | RGPD5 | 25 | STARD13 | 95 | TTC26 |
| 16 | 54 | RGPD8 | 25 | CDK19 | 95 | ITPKC |
| 17 | 54 | RGPD6 | 25 | ELOVL4 | 95 | BMP2K |
| 18 | 52 | NCCRP1 | 25 | SEL1L | 95 | SLC7A1 |
| 19 | 52 | HR | 23 | SNX18 | 95 | DPY19L4 |
| 20 | 52 | PAQR8 | 21 | ERMP1 | 95 | ZMYM2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, W.; Stricker, E.; Hotz-Wagenblatt, A.; Heit-Mondrzyk, A.; Pougialis, G.; Hugo, A.; Kuźmak, J.; Materniak-Kornas, M.; Löchelt, M. Functional Analyses of Bovine Foamy Virus-Encoded miRNAs Reveal the Importance of a Defined miRNA for Virus Replication and Host–Virus Interaction. Viruses 2020, 12, 1250. https://doi.org/10.3390/v12111250
Cao W, Stricker E, Hotz-Wagenblatt A, Heit-Mondrzyk A, Pougialis G, Hugo A, Kuźmak J, Materniak-Kornas M, Löchelt M. Functional Analyses of Bovine Foamy Virus-Encoded miRNAs Reveal the Importance of a Defined miRNA for Virus Replication and Host–Virus Interaction. Viruses. 2020; 12(11):1250. https://doi.org/10.3390/v12111250
Chicago/Turabian StyleCao, Wenhu, Erik Stricker, Agnes Hotz-Wagenblatt, Anke Heit-Mondrzyk, Georgios Pougialis, Annette Hugo, Jacek Kuźmak, Magdalena Materniak-Kornas, and Martin Löchelt. 2020. "Functional Analyses of Bovine Foamy Virus-Encoded miRNAs Reveal the Importance of a Defined miRNA for Virus Replication and Host–Virus Interaction" Viruses 12, no. 11: 1250. https://doi.org/10.3390/v12111250
APA StyleCao, W., Stricker, E., Hotz-Wagenblatt, A., Heit-Mondrzyk, A., Pougialis, G., Hugo, A., Kuźmak, J., Materniak-Kornas, M., & Löchelt, M. (2020). Functional Analyses of Bovine Foamy Virus-Encoded miRNAs Reveal the Importance of a Defined miRNA for Virus Replication and Host–Virus Interaction. Viruses, 12(11), 1250. https://doi.org/10.3390/v12111250





