Structural and Functional Aspects of Foamy Virus Protease-Reverse Transcriptase
Abstract
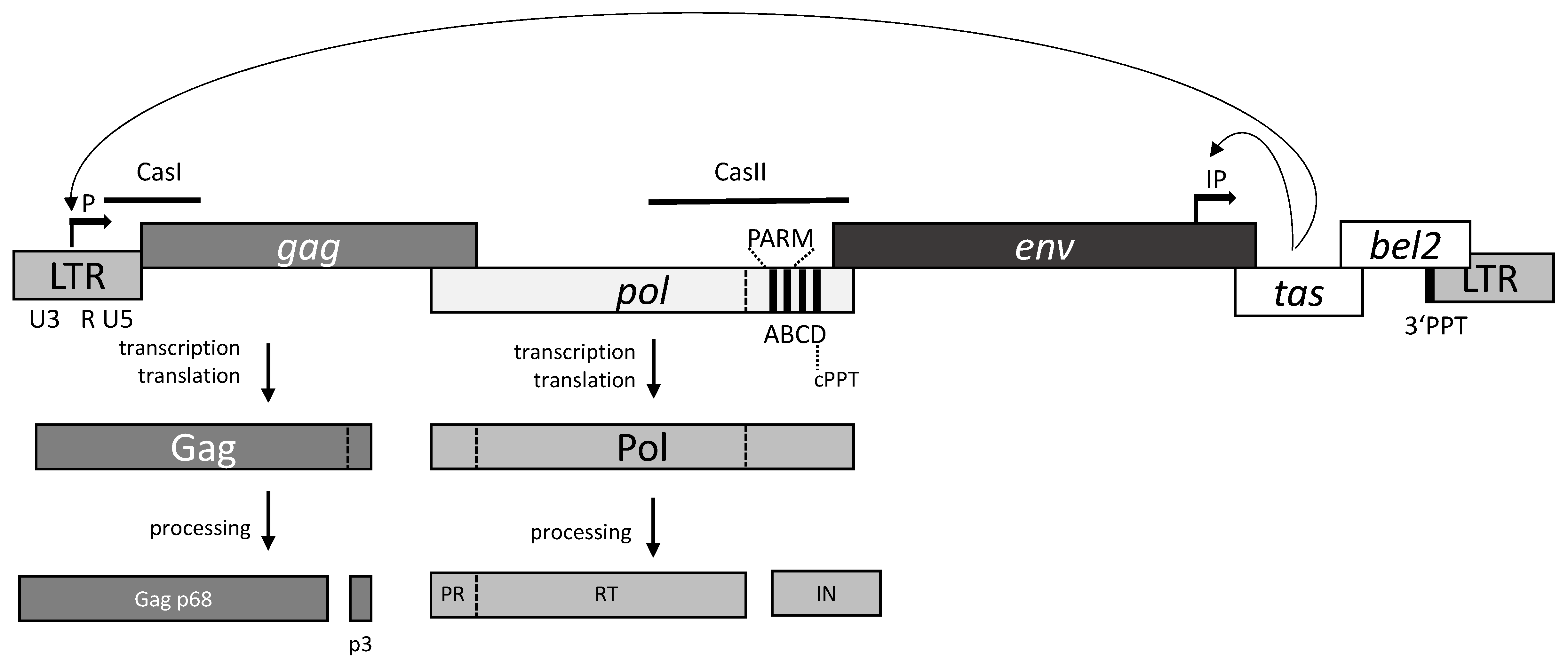
1. General Features of Foamy Virus Replication
2. The Pol Protein
3. Reverse Transcription
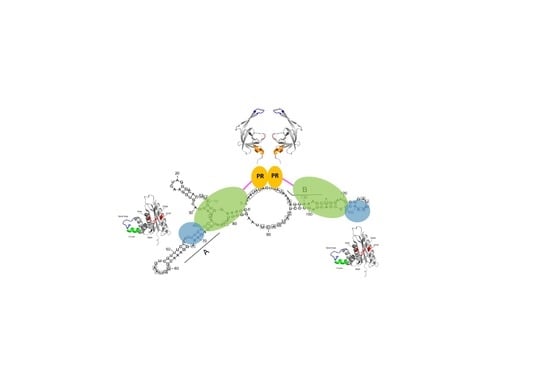
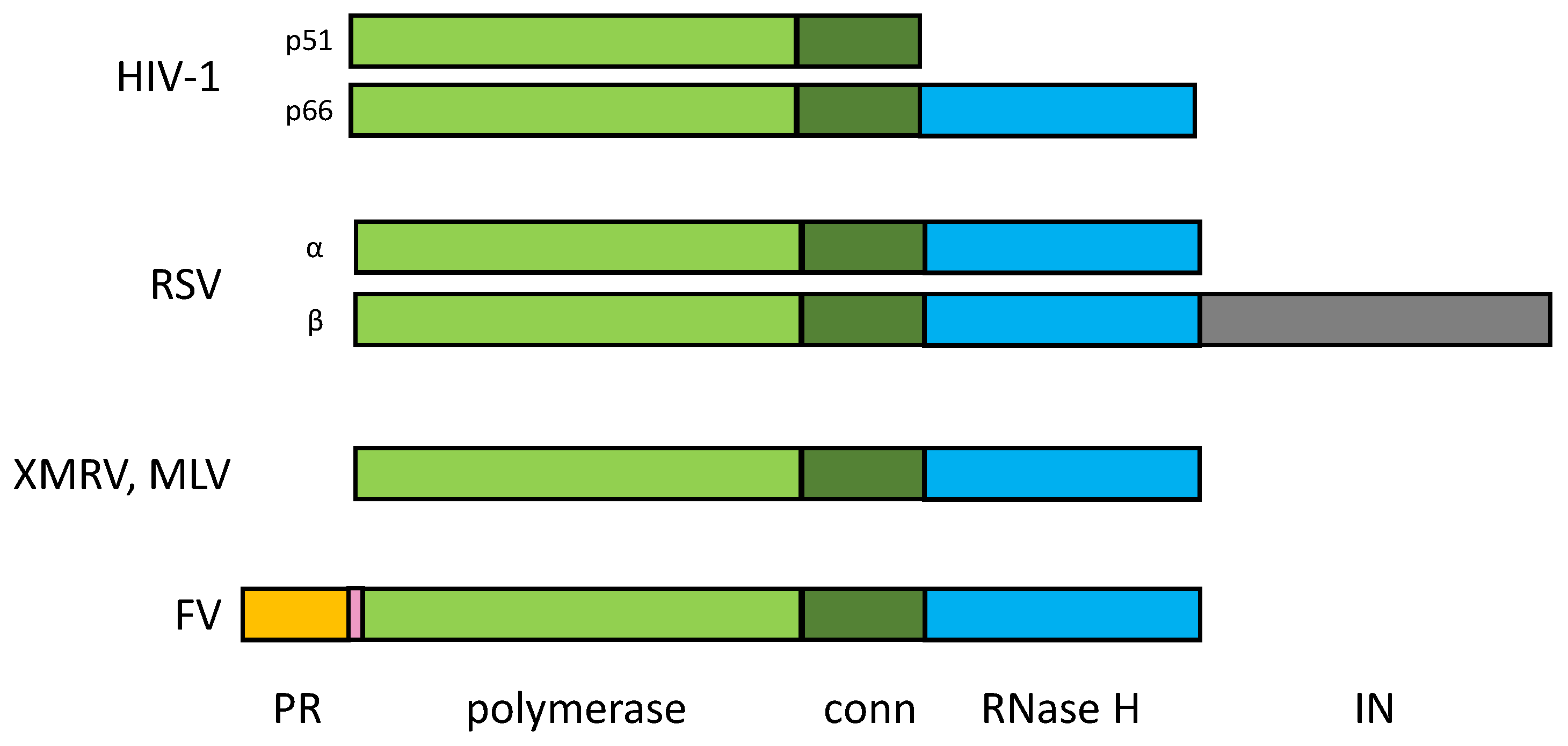
4. Foamy Virus PR-RT
4.1. Domain organization.
4.2. Polymerization Activities.
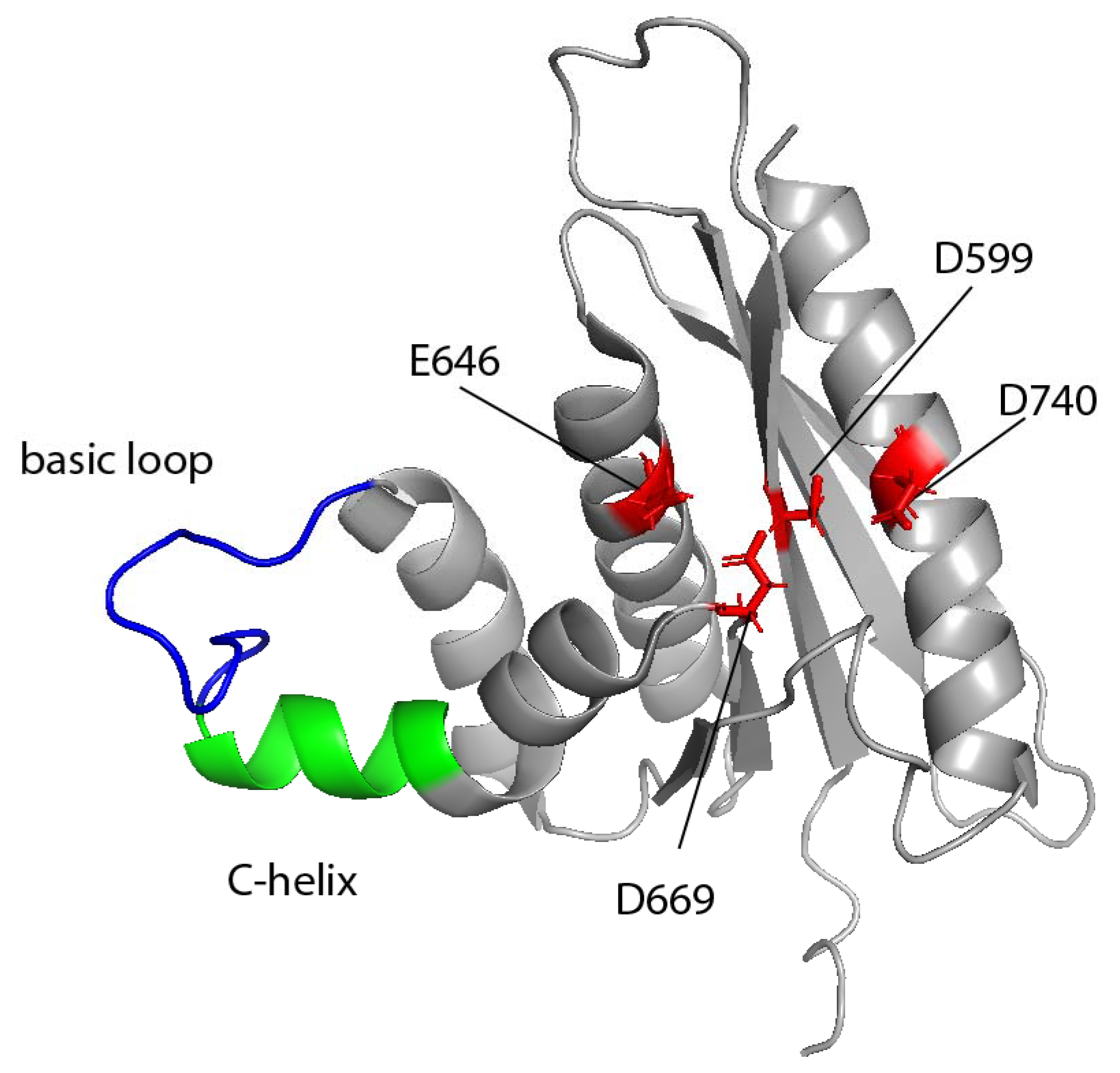
5. RNase H Activity and Structure
6. Protease Activity and Structure
7. Resistance of FV PR-RT against RT Inhibitors
8. Outlook and Persepectives
Funding
Conflicts of Interest
References
- Rethwilm, A.; Lindemann, D. Foamy Viruses. In Fields Virology; Knipe, D., Howley, P., Eds.; Lippincott, Williams & Wilkins: Philadelphia, PA, USA, 2013. [Google Scholar]
- Vogt, P.K. Historical introduction to the general properties of retroviruses. In Retroviruses; Coffin, J.M., Hughes, S.H., Varmus, H.E., Eds.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, USA, 1997. [Google Scholar]
- Achong, B.G.; Mansell, P.W.; Epstein, M.A.; Clifford, P. An unusual virus in cultures from a human nasopharyngeal carcinoma. J. Natl. Cancer Inst. 1971, 46, 299–307. [Google Scholar] [PubMed]
- Herchenröder, O.; Renne, R.; Loncar, D.; Cobb, E.K.; Murthy, K.K.; Schneider, J.; Mergia, A.; Luciw, P.A. Isolation, cloning, and sequencing of simian foamy viruses from chimpanzees (SFVcpz): High homology to human foamy virus (HFV). Virology 1994, 201, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.F.; Baldwin, D.N.; Gwynn, S.R.; Yendapalli, S.; Linial, M.L. Human foamy virus replication: A pathway distinct from that of retroviruses and hepadnaviruses. Science 1996, 271, 1579–1582. [Google Scholar] [CrossRef] [PubMed]
- Enssle, J.; Jordan, I.; Mauer, B.; Rethwilm, A. Foamy virus reverse transcriptase is expressed independently from the Gag protein. Proc. Natl. Acad. Sci. USA 1996, 93, 4137–4141. [Google Scholar] [CrossRef] [PubMed]
- Jordan, I.; Enssle, J.; Güttler, E.; Mauer, B.; Rethwilm, A. Expression of human foamy virus reverse transcriptase involves a spliced pol mRNA. Virology 1996, 224, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Löchelt, M.; Yu, S.F.; Linial, M.L.; Flügel, R.M. The human foamy virus internal promoter is required for efficient gene expression and infectivity. Virology 1995, 206, 601–610. [Google Scholar] [CrossRef]
- Campbell, M.; Renshaw-Gegg, L.; Renne, R.; Luciw, P.A. Characterization of the internal promoter of simian foamy viruses. J. Virol. 1994, 68, 4811–4820. [Google Scholar] [PubMed]
- Löchelt, M.; Muranyi, W.; Flügel, R.M. Human foamy virus genome possesses an internal, Bel-1-dependent and functional promoter. Proc. Natl. Acad. Sci. USA 1993, 90, 7317–7321. [Google Scholar] [CrossRef] [PubMed]
- Mergia, A. Simian foamy virus type 1 contains a second promoter located at the 3′ end of the env gene. Virology 1994, 199, 219–222. [Google Scholar] [CrossRef]
- Meiering, C.D.; Rubio, C.; May, C.; Linial, M.L. Cell-type-specific regulation of the two foamy virus promoters. J. Virol. 2001, 75, 6547–6557. [Google Scholar] [CrossRef]
- Alke, A.; Schwantes, A.; Kido, K.; Flötenmeyer, M.; Flügel, R.M.; Löchelt, M. The bet gene of feline foamy virus is required for virus replication. Virology 2001, 287, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Löchelt, M.; Romen, F.; Bastone, P.; Muckenfuss, H.; Kirchner, N.; Kim, Y.-B.; Truyen, U.; Rösler, U.; Battenberg, M.; Saib, A.; et al. The antiretroviral activity of APOBEC3 is inhibited by the foamy virus accessory Bet protein. Proc. Natl. Acad. Sci. USA 2005, 102, 7982–7987. [Google Scholar] [CrossRef] [PubMed]
- Chareza, S.; Slavkovic Lukic, D.; Liu, Y.; Räthe, A.-M.; Münk, C.; Zabogli, E.; Pistello, M.; Löchelt, M. Molecular and functional interactions of cat APOBEC3 and feline foamy and immunodeficiency virus proteins: Different ways to counteract host-encoded restriction. Virology 2012, 424, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Perkovic, M.; Schmidt, S.; Marino, D.; Russell, R.A.; Stauch, B.; Hofmann, H.; Kopietz, F.; Kloke, B.-P.; Zielonka, J.; Ströver, H.; et al. Species-specific inhibition of APOBEC3C by the prototype foamy virus protein bet. J. Biol. Chem. 2009, 284, 5819–5826. [Google Scholar] [CrossRef] [PubMed]
- Jaguva Vasudevan, A.A.; Perkovic, M.; Bulliard, Y.; Cichutek, K.; Trono, D.; Häussinger, D.; Münk, C. Prototype foamy virus Bet impairs the dimerization and cytosolic solubility of human APOBEC3G. J. Virol. 2013, 87, 9030–9040. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.A.; Wiegand, H.L.; Moore, M.D.; Schäfer, A.; McClure, M.O.; Cullen, B.R. Foamy virus Bet proteins function as novel inhibitors of the APOBEC3 family of innate antiretroviral defense factors. J. Virol. 2005, 79, 8724–8731. [Google Scholar] [CrossRef]
- Baldwin, D.N.; Linial, M.L. Proteolytic activity, the carboxy terminus of Gag, and the primer binding site are not required for Pol incorporation into foamy virus particles. J. Virol. 1999, 73, 6387–6393. [Google Scholar]
- Enssle, J.; Fischer, N.; Moebes, A.; Mauer, B.; Smola, U.; Rethwilm, A. Carboxy-terminal cleavage of the human foamy virus Gag precursor molecule is an essential step in the viral life cycle. J. Virol. 1997, 71, 7312–7317. [Google Scholar]
- Lehmann-Che, J.; Giron, M.-L.; Delelis, O.; Löchelt, M.; Bittoun, P.; Tobaly-Tapiero, J.; de Thé, H.; Saïb, A. Protease-dependent uncoating of a complex retrovirus. J. Virol. 2005, 79, 9244–9253. [Google Scholar] [CrossRef]
- Cartellieri, M.; Rudolph, W.; Herchenröder, O.; Lindemann, D.; Rethwilm, A. Determination of the relative amounts of Gag and Pol proteins in foamy virus particles. Retrovirology 2005, 2, 44. [Google Scholar] [CrossRef]
- Hütter, S.; Müllers, E.; Stanke, N.; Reh, J.; Lindemann, D. Prototype foamy virus protease activity is essential for intraparticle reverse transcription initiation but not absolutely required for uncoating upon host cell entry. J. Virol. 2013, 87, 3163–3176. [Google Scholar] [CrossRef] [PubMed]
- Stenbak, C.R.; Linial, M.L. Role of the C terminus of foamy virus Gag in RNA packaging and Pol expression. J. Virol. 2004, 78, 9423–9430. [Google Scholar] [CrossRef] [PubMed]
- Zemba, M.; Wilk, T.; Rutten, T.; Wagner, A.; Flügel, R.M.; Löchelt, M. The carboxy-terminal p3Gag domain of the human foamy virus Gag precursor is required for efficient virus infectivity. Virology 1998, 247, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Spannaus, R.; Schneider, A.; Hartl, M.J.; Wöhrl, B.M.; Bodem, J. Foamy virus Gag p71–p68 cleavage is required for template switch of the reverse transcriptase. J. Virol. 2013, 87, 7774–7776. [Google Scholar] [CrossRef] [PubMed]
- Löchelt, M.; Flügel, R.M. The human foamy virus pol gene is expressed as a Pro-Pol polyprotein and not as a Gag-Pol fusion protein. J. Virol. 1996, 70, 1033–1040. [Google Scholar] [PubMed]
- Bodem, J.; Löchelt, M.; Winkler, I.; Flower, R.P.; Delius, H.; Flügel, R.M. Characterization of the spliced pol transcript of feline foamy virus: The splice acceptor site of the pol transcript is located in gag of foamy viruses. J. Virol. 1996, 70, 9024–9027. [Google Scholar] [PubMed]
- Konvalinka, J.; Löchelt, M.; Zentgraf, H.; Flügel, R.M.; Kräusslich, H.G. Active foamy virus proteinase is essential for virus infectivity but not for formation of a Pol polyprotein. J. Virol. 1995, 69, 7264–7268. [Google Scholar] [PubMed]
- Kögel, D.; Aboud, M.; Flügel, R.M. Molecular biological characterization of the human foamy virus reverse transcriptase and ribonuclease H domains. Virology 1995, 213, 97–108. [Google Scholar] [CrossRef][Green Version]
- Netzer, K.O.; Schliephake, A.; Maurer, B.; Watanabe, R.; Aguzzi, A.; Rethwilm, A. Identification of pol-related gene products of human foamy virus. Virology 1993, 192, 336–338. [Google Scholar] [CrossRef]
- Goff, S.P. Retroviridae: The retroviruses and their replication. In Fields Virology; Knipe, D., Howley, P., Eds.; Lippincott, Williams & Wilkins: Philadelphia, PA, USA, 2007; pp. 1999–2069. [Google Scholar]
- Boyer, P.L.; Stenbak, C.R.; Clark, P.K.; Linial, M.L.; Hughes, S.H. Characterization of the polymerase and RNase H activities of human foamy virus reverse transcriptase. J. Virol. 2004, 78, 6112–6121. [Google Scholar] [CrossRef]
- Rinke, C.S.; Boyer, P.L.; Sullivan, M.D.; Hughes, S.H.; Linial, M.L. Mutation of the catalytic domain of the foamy virus reverse transcriptase leads to loss of processivity and infectivity. J. Virol. 2002, 76, 7560–7570. [Google Scholar] [CrossRef] [PubMed]
- Erlwein, O.; Bieniasz, P.D.; McClure, M.O. Sequences in pol are required for transfer of human foamy virus-based vectors. J. Virol. 1998, 72, 5510–5516. [Google Scholar] [PubMed]
- Heinkelein, M.; Dressler, M.; Jármy, G.; Rammling, M.; Imrich, H.; Thurow, J.; Lindemann, D.; Rethwilm, A. Improved primate foamy virus vectors and packaging constructs. J. Virol. 2002, 76, 3774–3783. [Google Scholar] [CrossRef] [PubMed]
- Heinkelein, M.; Leurs, C.; Rammling, M.; Peters, K.; Hanenberg, H.; Rethwilm, A. Pregenomic RNA is required for efficient incorporation of pol polyprotein into foamy virus capsids. J. Virol. 2002, 76, 10069–10073. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Chari, S.; Yanchis, T.; Mergia, A. Cis-Acting sequences required for simian foamy virus type 1 vectors. J. Virol. 1998, 72, 3451–3454. [Google Scholar] [PubMed]
- Peters, K.; Wiktorowicz, T.; Heinkelein, M.; Rethwilm, A. RNA and protein requirements for incorporation of the Pol protein into foamy virus particles. J. Virol. 2005, 79, 7005–7013. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.-G.; Linial, M.L. The C terminus of foamy retrovirus Gag contains determinants for encapsidation of Pol protein into virions. J. Virol. 2008, 82, 10803–10810. [Google Scholar] [CrossRef] [PubMed]
- Temin, H.M.; Mizutani, S. RNA-dependent DNA polymerase in virions of Rous sarcoma virus. Nature 1970, 226, 1211–1213. [Google Scholar] [CrossRef] [PubMed]
- Baltimore, D. RNA-dependent DNA polymerase in virions of RNA tumour viruses. Nature 1970, 226, 1209–1211. [Google Scholar] [CrossRef]
- Cain, D.; Erlwein, O.; Grigg, A.; Russell, R.A.; McClure, M.O. Palindromic sequence plays a critical role in human foamy virus dimerization. J. Virol. 2001, 75, 3731–3739. [Google Scholar] [CrossRef]
- Erlwein, O.; Cain, D.; Fischer, N.; Rethwilm, A.; McClure, M.O. Identification of sites that act together to direct dimerization of human foamy virus RNA in vitro. Virology 1997, 229, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Moebes, A.; Enssle, J.; Bieniasz, P.D.; Heinkelein, M.; Lindemann, D.; Bock, M.; McClure, M.O.; Rethwilm, A. Human foamy virus reverse transcription that occurs late in the viral replication cycle. J. Virol. 1997, 71, 7305–7311. [Google Scholar] [PubMed]
- Yu, S.F.; Sullivan, M.D.; Linial, M.L. Evidence that the human foamy virus genome is DNA. J. Virol. 1999, 73, 1565–1572. [Google Scholar] [PubMed]
- Delelis, O.; Saïb, A.; Sonigo, P. Biphasic DNA synthesis in spumaviruses. J. Virol. 2003, 77, 8141–8146. [Google Scholar] [CrossRef] [PubMed]
- Zamborlini, A.; Renault, N.; Saïb, A.; Delelis, O. Early Reverse Transcription Is Essential for Productive Foamy Virus Infection. PLoS ONE 2010, 5, e11023. [Google Scholar] [CrossRef]
- Trono, D. Partial reverse transcripts in virions from human immunodeficiency and murine leukemia viruses. J. Virol. 1992, 66, 4893–4900. [Google Scholar] [PubMed]
- Nassal, M. The arginine-rich domain of the hepatitis B virus core protein is required for pregenome encapsidation and productive viral positive-strand DNA synthesis but not for virus assembly. J. Virol. 1992, 66, 4107–4116. [Google Scholar]
- Hatton, T.; Zhou, S.; Standring, D.N. RNA- and DNA-binding activities in hepatitis B virus capsid protein: A model for their roles in viral replication. J. Virol. 1992, 66, 5232–5241. [Google Scholar]
- Nassal, M.; Schaller, H. Hepatitis B virus replication. Trends Microbiol. 1993, 1, 221–228. [Google Scholar] [CrossRef]
- Beck, J.; Nassal, M. Hepatitis B virus replication. World J. Gastroenterol. WJG 2007, 13, 48–64. [Google Scholar] [CrossRef]
- Lee, E.-G.; Sinicrope, A.; Jackson, D.L.; Yu, S.F.; Linial, M.L. Foamy virus Pol protein expressed as a Gag-Pol fusion retains enzymatic activities, allowing for infectious virus production. J. Virol. 2012, 86, 5992–6001. [Google Scholar] [CrossRef] [PubMed]
- Spannaus, R.; Hartl, M.J.; Wöhrl, B.M.; Rethwilm, A.; Bodem, J. The prototype foamy virus protease is active independently of the integrase domain. Retrovirology 2012, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Swiersy, A.; Wiek, C.; Reh, J.; Zentgraf, H.; Lindemann, D. Orthoretroviral-like prototype foamy virus Gag-Pol expression is compatible with viral replication. Retrovirology 2011, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Jackson, D.L.; Lee, E.-G.; Linial, M.L. Expression of prototype foamy virus pol as a Gag-Pol fusion protein does not change the timing of reverse transcription. J. Virol. 2013, 87, 1252–1254. [Google Scholar] [CrossRef] [PubMed]
- Kupiec, J.J.; Tobaly-Tapiero, J.; Canivet, M.; Santillana-Hayat, M.; Flügel, R.M.; Périès, J.; Emanoil-Ravier, R. Evidence for a gapped linear duplex DNA intermediate in the replicative cycle of human and simian spumaviruses. Nucleic Acids Res. 1988, 16, 9557–9565. [Google Scholar] [CrossRef] [PubMed]
- Peters, K.; Barg, N.; Gärtner, K.; Rethwilm, A. Complex effects of foamy virus central purine-rich regions on viral replication. Virology 2008, 373, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Tobaly-Tapiero, J.; Kupiec, J.J.; Santillana-Hayat, M.; Canivet, M.; Peries, J.; Emanoil-Ravier, R. Further characterization of the gapped DNA intermediates of human spumavirus: Evidence for a dual initiation of plus-strand DNA synthesis. J. Gen. Virol. 1991, 72, 605–608. [Google Scholar] [CrossRef]
- Moschall, R.; Denk, S.; Erkelenz, S.; Schenk, C.; Schaal, H.; Bodem, J. A purine-rich element in foamy virus pol regulates env splicing and gag/pol expression. Retrovirology 2017, 14, 10. [Google Scholar] [CrossRef]
- Rausch, J.W.; Le Grice, S.F.J. “Binding, bending and bonding”: Polypurine tract-primed initiation of plus-strand DNA synthesis in human immunodeficiency virus. Int. J. Biochem. Cell Biol. 2004, 36, 1752–1766. [Google Scholar] [CrossRef]
- Bieniasz, P.D.; Weiss, R.A.; McClure, M.O. Cell cycle dependence of foamy retrovirus infection. J. Virol. 1995, 69, 7295–7299. [Google Scholar]
- Chen, D.; Song, J.; Sun, Y.; Li, Z.; Wen, D.; Liu, Q.; Liu, W.; He, X. The fourth central polypurine tract guides the synthesis of prototype foamy virus plus-strand DNA. Virus Genes 2017, 53, 259–265. [Google Scholar] [CrossRef] [PubMed]
- Le Grice, S.F.; Grüninger-Leitch, F. Rapid purification of homodimer and heterodimer HIV-1 reverse transcriptase by metal chelate affinity chromatography. Eur. J. Biochem. 1990, 187, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Rodgers, D.W.; Gamblin, S.J.; Harris, B.A.; Ray, S.; Culp, J.S.; Hellmig, B.; Woolf, D.J.; Debouck, C.; Harrison, S.C. The structure of unliganded reverse transcriptase from the human immunodeficiency virus type 1. Proc. Natl. Acad. Sci. USA 1995, 92, 1222–1226. [Google Scholar] [CrossRef] [PubMed]
- Rho, H.M.; Grandgenett, D.P.; Green, M. Sequence relatedness between the subunits of avian myeloblastosis virus reverse transcriptase. J. Biol. Chem. 1975, 250, 5278–5280. [Google Scholar] [PubMed]
- Hizi, A.; Joklik, W.K. RNA-dependent DNA polymerase of avian sarcoma virus B77. I. Isolation and partial characterization of the alpha, beta2, and alphabeta forms of the enzyme. J. Biol. Chem. 1977, 252, 2281–2289. [Google Scholar] [PubMed]
- Werner, S.; Wöhrl, B.M. Asymmetric subunit organization of heterodimeric Rous sarcoma virus reverse transcriptase alphabeta: Localization of the polymerase and RNase H active sites in the alpha subunit. J. Virol. 2000, 74, 3245–3252. [Google Scholar] [CrossRef]
- Das, D.; Georgiadis, M.M. The crystal structure of the monomeric reverse transcriptase from Moloney murine leukemia virus. Structure 2004, 12, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Nowak, E.; Potrzebowski, W.; Konarev, P.V.; Rausch, J.W.; Bona, M.K.; Svergun, D.I.; Bujnicki, J.M.; Le Grice, S.F.J.; Nowotny, M. Structural analysis of monomeric retroviral reverse transcriptase in complex with an RNA/DNA hybrid. Nucleic Acids Res. 2013, 41, 3874–3887. [Google Scholar] [CrossRef]
- Roy, J.; Linial, M.L. Role of the Foamy Virus Pol Cleavage Site in Viral Replication. J. Virol. 2007, 81, 4956–4962. [Google Scholar] [CrossRef]
- Pfrepper, K.-I.; Rackwitz, H.-R.; Schnölzer, M.; Heid, H.; Löchelt, M.; Flügel, R.M. Molecular Characterization of Proteolytic Processing of the Pol Proteins of Human Foamy Virus Reveals Novel Features of the Viral Protease. J. Virol. 1998, 72, 7648–7652. [Google Scholar]
- Hartl, M.J.; Mayr, F.; Rethwilm, A.; Wöhrl, B.M. Biophysical and enzymatic properties of the simian and prototype foamy virus reverse transcriptases. Retrovirology 2010, 7, 5. [Google Scholar] [CrossRef] [PubMed]
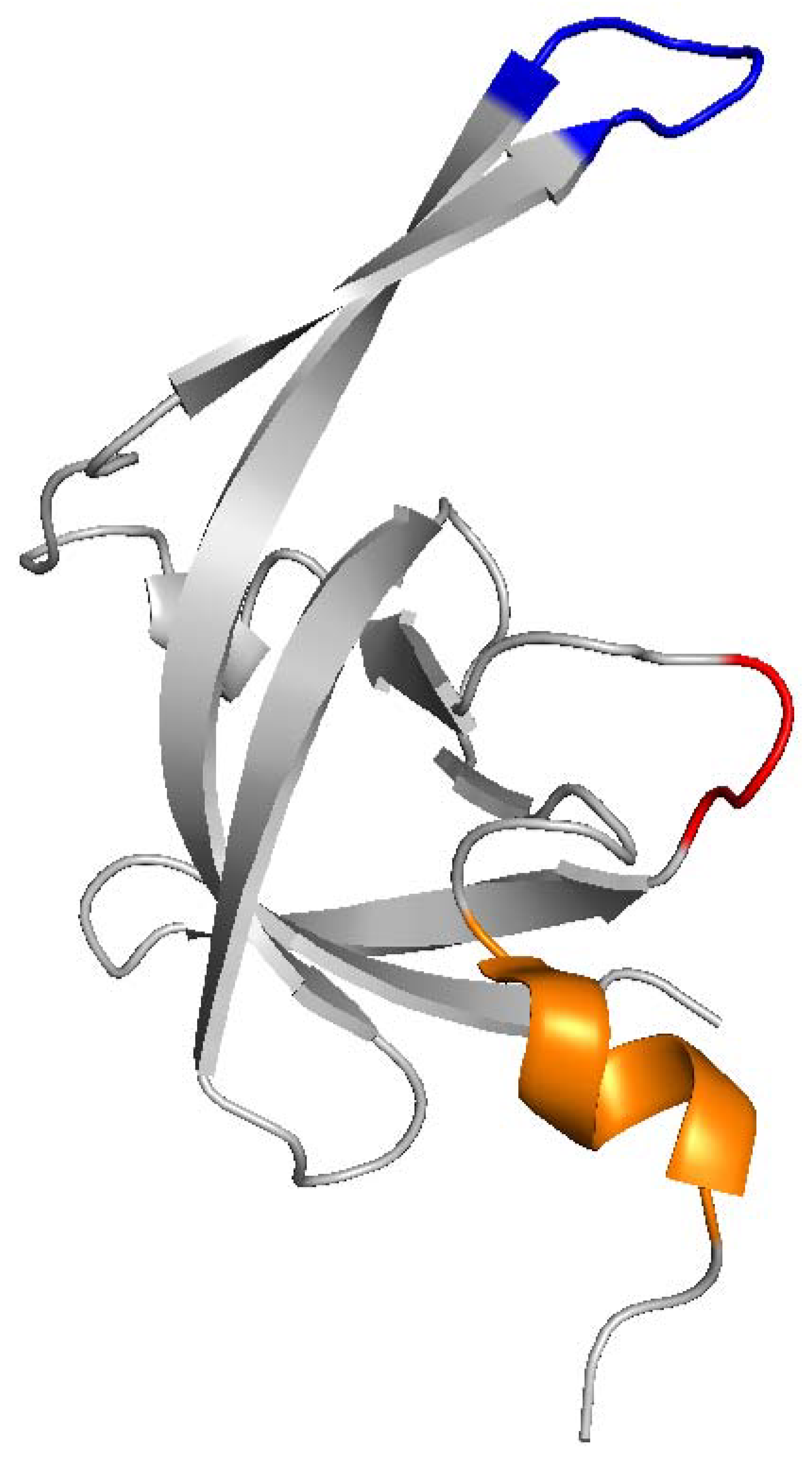
- Hartl, M.J.; Wöhrl, B.M.; Rösch, P.; Schweimer, K. The solution structure of the simian foamy virus protease reveals a monomeric protein. J. Mol. Biol. 2008, 381, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Hartl, M.J.; Kretzschmar, B.; Frohn, A.; Nowrouzi, A.; Rethwilm, A.; Wöhrl, B.M. AZT resistance of simian foamy virus reverse transcriptase is based on the excision of AZTMP in the presence of ATP. Nucleic Acids Res. 2008, 36, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Leo, B.; Hartl, M.J.; Schweimer, K.; Mayr, F.; Wöhrl, B.M. Insights into the structure and activity of prototype foamy virus RNase H. Retrovirology 2012, 9, 14. [Google Scholar] [CrossRef] [PubMed]
- Leo, B.; Schweimer, K.; Rösch, P.; Hartl, M.J.; Wöhrl, B.M. The solution structure of the prototype foamy virus RNase H domain indicates an important role of the basic loop in substrate binding. Retrovirology 2012, 9, 73. [Google Scholar] [CrossRef] [PubMed]
- Schultz, S.J.; Champoux, J.J. RNase H domain of Moloney murine leukemia virus reverse transcriptase retains activity but requires the polymerase domain for specificity. J. Virol. 1996, 70, 8630–8638. [Google Scholar]
- Hartl, M.J.; Schweimer, K.; Reger, M.H.; Schwarzinger, S.; Bodem, J.; Rösch, P.; Wöhrl, B.M. Formation of transient dimers by a retroviral protease. Biochem. J. 2010, 427, 197–203. [Google Scholar] [CrossRef]
- Hartl, M.J.; Burmann, B.M.; Prasch, S.J.; Schwarzinger, C.; Schweimer, K.; Wöhrl, B.M.; Rösch, P.; Schwarzinger, S. Fast mapping of biomolecular interfaces by Random Spin Labeling (RSL). J. Biomol. Struct. Dyn. 2012, 29, 793–798. [Google Scholar] [CrossRef]
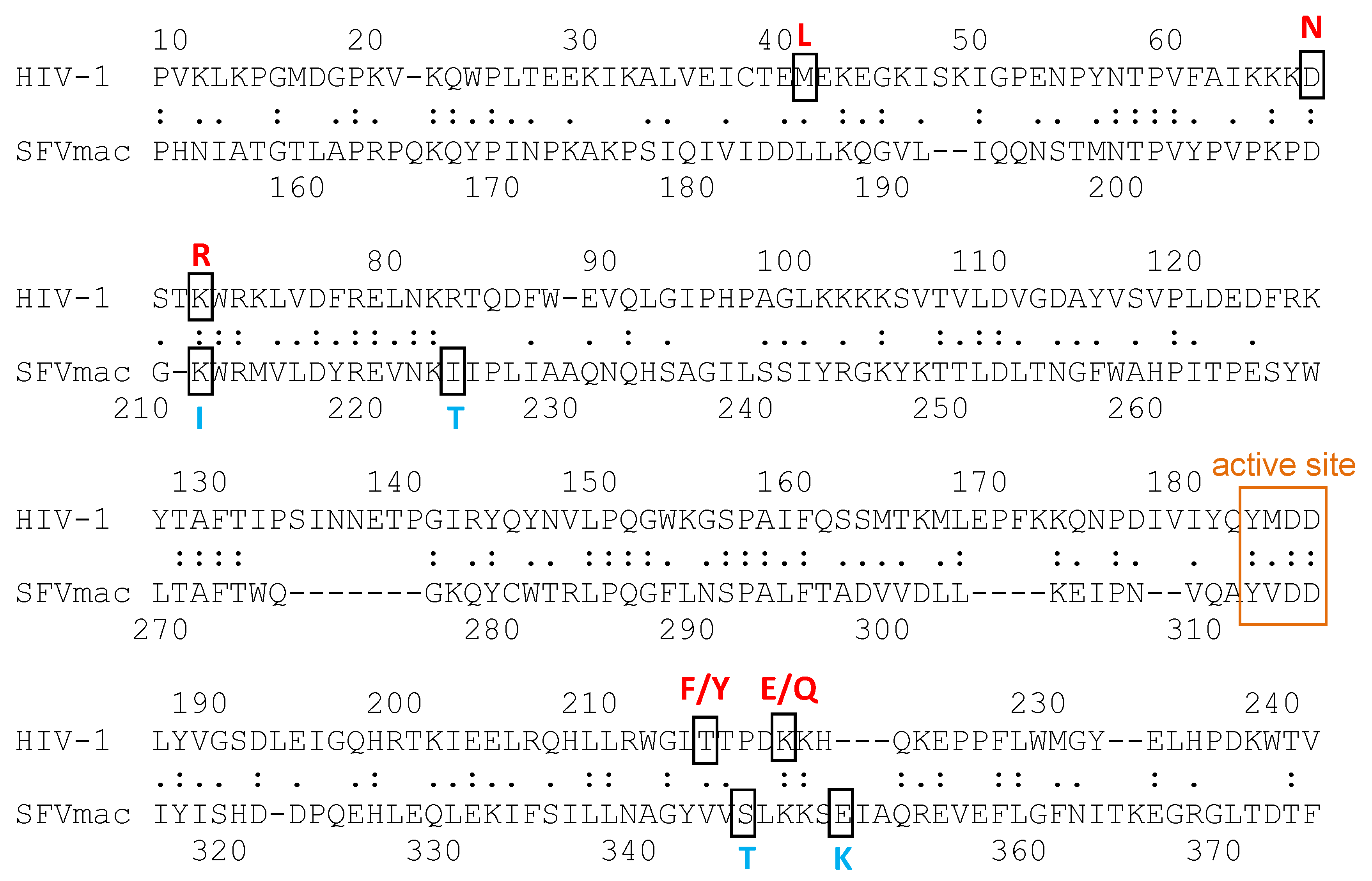
- Schneider, A.; Peter, D.; Schmitt, J.; Leo, B.; Richter, F.; Rösch, P.; Wöhrl, B.M.; Hartl, M.J. Structural requirements for enzymatic activities of foamy virus protease-reverse transcriptase. Proteins 2014, 82, 375–385. [Google Scholar] [CrossRef]
- Steitz, T.A. DNA polymerases: Structural diversity and common mechanisms. J. Biol. Chem. 1999, 274, 17395–17398. [Google Scholar] [CrossRef]
- Singh, K.; Marchand, B.; Kirby, K.A.; Michailidis, E.; Sarafianos, S.G. Structural Aspects of Drug Resistance and Inhibition of HIV-1 Reverse Transcriptase. Viruses 2010, 2, 606–638. [Google Scholar] [CrossRef] [PubMed]
- Gu, Z.; Gao, Q.; Li, X.; Parniak, M.A.; Wainberg, M.A. Novel mutation in the human immunodeficiency virus type 1 reverse transcriptase gene that encodes cross-resistance to 2′,3′-dideoxyinosine and 2′,3′-dideoxycytidine. J. Virol. 1992, 66, 7128–7135. [Google Scholar] [PubMed]
- Murray, S.M.; Picker, L.J.; Axthelm, M.K.; Linial, M.L. Expanded tissue targets for foamy virus replication with simian immunodeficiency virus-induced immunosuppression. J. Virol. 2006, 80, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Martin, J.L.; Wilson, J.E.; Haynes, R.L.; Furman, P.A. Mechanism of resistance of human immunodeficiency virus type 1 to 2′,3′-dideoxyinosine. Proc. Natl. Acad. Sci. USA 1993, 90, 6135–6139. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Shirasaka, T.; Mitsuya, H. Enzymatic characterization of human immunodeficiency virus type 1 reverse transcriptase resistant to multiple 2′,3′-dideoxynucleoside 5′-triphosphates. J. Biol. Chem. 1995, 270, 23605–23611. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.E.; Aulabaugh, A.; Caligan, B.; McPherson, S.; Wakefield, J.K.; Jablonski, S.; Morrow, C.D.; Reardon, J.E.; Furman, P.A. Human immunodeficiency virus type-1 reverse transcriptase. Contribution of Met-184 to binding of nucleoside 5′-triphosphate. J. Biol. Chem. 1996, 271, 13656–13662. [Google Scholar] [CrossRef] [PubMed]
- Krebs, R.; Immendörfer, U.; Thrall, S.H.; Wöhrl, B.M.; Goody, R.S. Single-step kinetics of HIV-1 reverse transcriptase mutants responsible for virus resistance to nucleoside inhibitors zidovudine and 3-TC. Biochemistry 1997, 36, 10292–10300. [Google Scholar] [CrossRef]
- Wöhrl, B.M.; Krebs, R.; Thrall, S.H.; Le Grice, S.F.; Scheidig, A.J.; Goody, R.S. Kinetic analysis of four HIV-1 reverse transcriptase enzymes mutated in the primer grip region of p66 implications for DNA synthesis and dimerization. J. Biol. Chem. 1997, 272, 17581–17587. [Google Scholar] [CrossRef]
- Santos-Velazquez, J.; Kim, B. Deoxynucleoside triphosphate incorporation mechanism of foamy virus (FV) reverse transcriptase: Implications for cell tropism of FV. J. Virol. 2008, 82, 8235–8238. [Google Scholar] [CrossRef]
- Skasko, M.; Weiss, K.K.; Reynolds, H.M.; Jamburuthugoda, V.; Lee, K.; Kim, B. Mechanistic differences in RNA-dependent DNA polymerization and fidelity between murine leukemia virus and HIV-1 reverse transcriptases. J. Biol. Chem. 2005, 280, 12190–12200. [Google Scholar] [CrossRef]
- Tisdale, M.; Schulze, T.; Larder, B.A.; Moelling, K. Mutations within the RNase H domain of human immunodeficiency virus type 1 reverse transcriptase abolish virus infectivity. J. Gen. Virol. 1991, 72, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Repaske, R.; Hartley, J.W.; Kavlick, M.F.; O’Neill, R.R.; Austin, J.B. Inhibition of RNase H activity and viral replication by single mutations in the 3′ region of Moloney murine leukemia virus reverse transcriptase. J. Virol. 1989, 63, 1460–1464. [Google Scholar] [PubMed]
- Krug, M.S.; Berger, S.L. Ribonuclease H activities associated with viral reverse transcriptases are endonucleases. Proc. Natl. Acad. Sci. USA 1989, 86, 3539–3543. [Google Scholar] [CrossRef] [PubMed]
- DeStefano, J.J.; Buiser, R.G.; Mallaber, L.M.; Bambara, R.A.; Fay, P.J. Human immunodeficiency virus reverse transcriptase displays a partially processive 3′ to 5′ endonuclease activity. J. Biol. Chem. 1991, 266, 24295–24301. [Google Scholar] [PubMed]
- Wöhrl, B.M.; Volkmann, S.; Moelling, K. Mutations of a conserved residue within HIV-1 ribonuclease H affect its exo- and endonuclease activities. J. Mol. Biol. 1991, 220, 801–818. [Google Scholar] [CrossRef]
- Huber, H.E.; Richardson, C.C. Processing of the primer for plus strand DNA synthesis by human immunodeficiency virus 1 reverse transcriptase. J. Biol. Chem. 1990, 265, 10565–10573. [Google Scholar] [PubMed]
- Luo, G.X.; Sharmeen, L.; Taylor, J. Specificities involved in the initiation of retroviral plus-strand DNA. J. Virol. 1990, 64, 592–597. [Google Scholar]
- Wöhrl, B.M.; Moelling, K. Interaction of HIV-1 ribonuclease H with polypurine tract containing RNA-DNA hybrids. Biochemistry 1990, 29, 10141–10147. [Google Scholar] [CrossRef]
- Furfine, E.S.; Reardon, J.E. Human immunodeficiency virus reverse transcriptase ribonuclease H: Specificity of tRNA(Lys3)-primer excision. Biochemistry 1991, 30, 7041–7046. [Google Scholar] [CrossRef]
- Smith, J.S.; Roth, M.J. Specificity of human immunodeficiency virus-1 reverse transcriptase-associated ribonuclease H in removal of the minus-strand primer, tRNA(Lys3). J. Biol. Chem. 1992, 267, 15071–15079. [Google Scholar]
- Yang, W.; Lee, J.Y.; Nowotny, M. Making and breaking nucleic acids: Two-Mg2+-ion catalysis and substrate specificity. Mol. Cell 2006, 22, 5–13. [Google Scholar] [CrossRef]
- Steitz, T.A.; Steitz, J.A. A general two-metal-ion mechanism for catalytic RNA. Proc. Natl. Acad. Sci. USA 1993, 90, 6498–6502. [Google Scholar] [CrossRef] [PubMed]
- Schultz, S.J.; Champoux, J.J. RNase H activity: Structure, specificity, and function in reverse transcription. Virus Res. 2008, 134, 86–103. [Google Scholar] [CrossRef] [PubMed]
- Tanese, N.; Goff, S.P. Domain structure of the Moloney murine leukemia virus reverse transcriptase: Mutational analysis and separate expression of the DNA polymerase and RNase H activities. Proc. Natl. Acad. Sci. USA 1988, 85, 1777–1781. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.; Crouch, R.J. The isolated RNase H domain of murine leukemia virus reverse transcriptase. Retention of activity with concomitant loss of specificity. J. Biol. Chem. 1997, 272, 22023–22029. [Google Scholar] [CrossRef]
- Kanaya, S.; Katsuda-Nakai, C.; Ikehara, M. Importance of the positive charge cluster in Escherichia coli ribonuclease HI for the effective binding of the substrate. J. Biol. Chem. 1991, 266, 11621–11627. [Google Scholar] [PubMed]
- Telesnitsky, A.; Blain, S.W.; Goff, S.P. Defects in Moloney murine leukemia virus replication caused by a reverse transcriptase mutation modeled on the structure of Escherichia coli RNase H. J. Virol. 1992, 66, 615–622. [Google Scholar]
- Lim, D.; Gregorio, G.G.; Bingman, C.; Martinez-Hackert, E.; Hendrickson, W.A.; Goff, S.P. Crystal structure of the moloney murine leukemia virus RNase H domain. J. Virol. 2006, 80, 8379–8389. [Google Scholar] [CrossRef]
- Zhou, D.; Chung, S.; Miller, M.; Grice, S.F.J.L.; Wlodawer, A. Crystal structures of the reverse transcriptase-associated ribonuclease H domain of xenotropic murine leukemia-virus related virus. J. Struct. Biol. 2012, 177, 638–645. [Google Scholar] [CrossRef]
- Davies, J.F.; Hostomska, Z.; Hostomsky, Z.; Jordan, S.R.; Matthews, D.A. Crystal structure of the ribonuclease H domain of HIV-1 reverse transcriptase. Science 1991, 252, 88–95. [Google Scholar] [CrossRef]
- Corona, A.; Schneider, A.; Schweimer, K.; Rösch, P.; Wöhrl, B.M.; Tramontano, E. Inhibition of foamy virus reverse transcriptase by human immunodeficiency virus type 1 RNase H inhibitors. Antimicrob. Agents Chemother. 2014, 58, 4086–4093. [Google Scholar] [CrossRef] [PubMed]
- Pearl, L.H.; Taylor, W.R. A structural model for the retroviral proteases. Nature 1987, 329, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Hartl, M.J.; Wöhrl, B.M.; Schweimer, K. Sequence-specific 1H, 13C and 15N resonance assignments and secondary structure of a truncated protease from Simian Foamy Virus. Biomol. NMR Assign. 2007, 1, 175–177. [Google Scholar] [CrossRef] [PubMed]
- Wlodawer, A.; Gustchina, A. Structural and biochemical studies of retroviral proteases. Biochim. Biophys. Acta 2000, 1477, 16–34. [Google Scholar] [CrossRef]
- Fenyöfalvi, G.; Bagossi, P.; Copeland, T.D.; Oroszlan, S.; Boross, P.; Tözsér, J. Expression and characterization of human foamy virus proteinase. FEBS Lett. 1999, 462, 397–401. [Google Scholar] [CrossRef]
- Pfrepper, K.I.; Löchelt, M.; Schnölzer, M.; Flügel, R.M. Expression and molecular characterization of an enzymatically active recombinant human spumaretrovirus protease. Biochem. Biophys. Res. Commun. 1997, 237, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Sperka, T.; Boross, P.; Eizert, H.; Tözsér, J.; Bagossi, P. Effect of mutations on the dimer stability and the pH optimum of the human foamy virus protease. Protein Eng. Des. Sel. PEDS 2006, 19, 369–375. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tang, C.; Louis, J.M.; Aniana, A.; Suh, J.-Y.; Clore, G.M. Visualizing transient events in amino-terminal autoprocessing of HIV-1 protease. Nature 2008, 455, 693–696. [Google Scholar] [CrossRef] [PubMed]
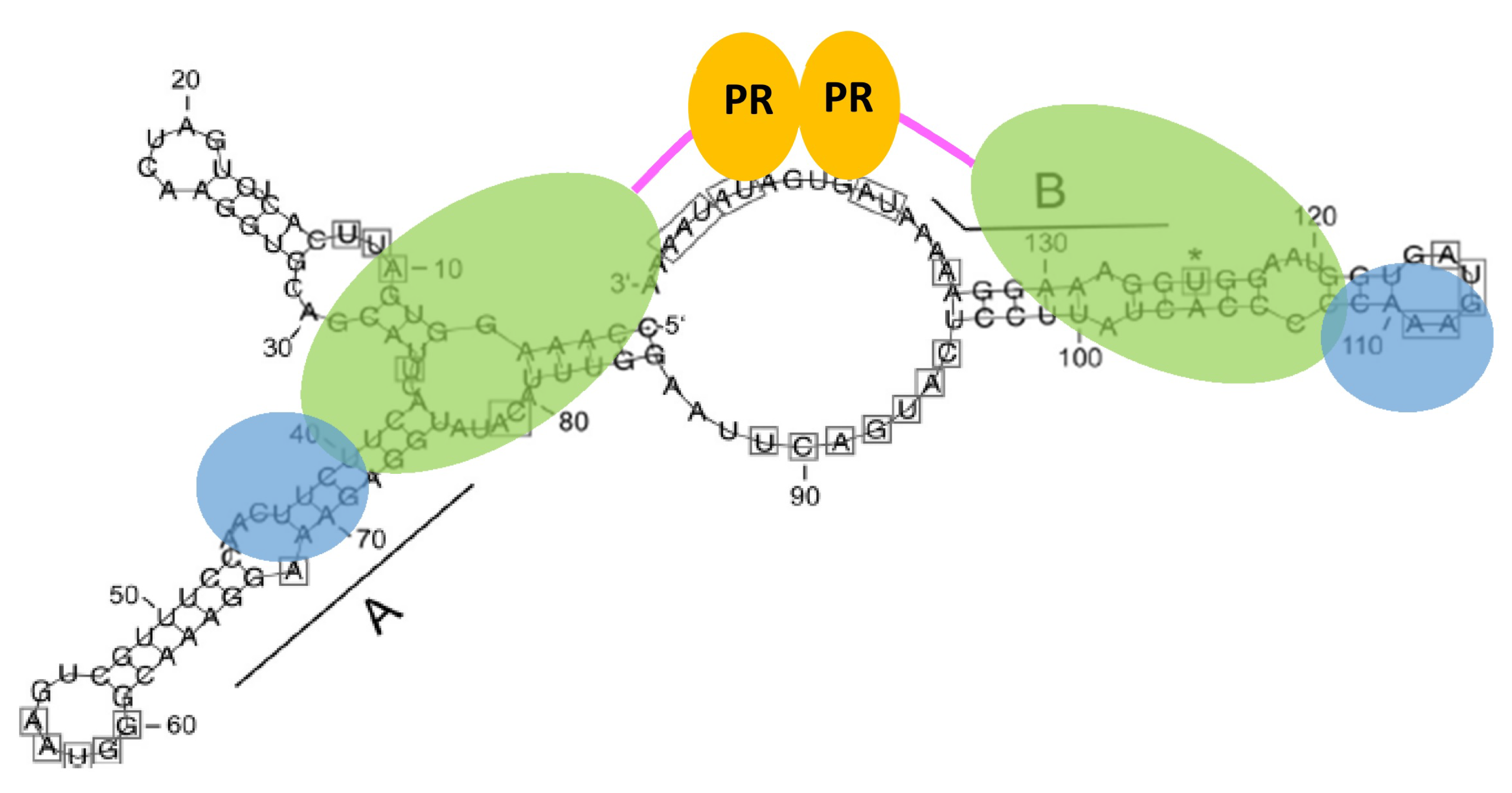
- Hartl, M.J.; Bodem, J.; Jochheim, F.; Rethwilm, A.; Rösch, P.; Wöhrl, B.M. Regulation of foamy virus protease activity by viral RNA: A novel and unique mechanism among retroviruses. J. Virol. 2011, 85, 4462–4469. [Google Scholar] [CrossRef]
- Rosenblum, L.L.; Patton, G.; Grigg, A.R.; Frater, A.J.; Cain, D.; Erlwein, O.; Hill, C.L.; Clarke, J.R.; McClure, M.O. Differential susceptibility of retroviruses to nucleoside analogues. Antivir. Chem. Chemother. 2001, 12, 91–97. [Google Scholar] [CrossRef]
- Lee, C.C.I.; Ye, F.; Tarantal, A.F. Comparison of growth and differentiation of fetal and adult rhesus monkey mesenchymal stem cells. Stem Cells Dev. 2006, 15, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Kretzschmar, B.; Nowrouzi, A.; Hartl, M.J.; Gärtner, K.; Wiktorowicz, T.; Herchenröder, O.; Kanzler, S.; Rudolph, W.; Mergia, A.; Wöhrl, B.; et al. AZT-resistant foamy virus. Virology 2008, 370, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.L.; Sarafianos, S.G.; Clark, P.K.; Arnold, E.; Hughes, S.H. Why do HIV-1 and HIV-2 use different pathways to develop AZT resistance? PLoS Pathog. 2006, 2, e10. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Schweimer, K.; Rösch, P.; Wöhrl, B.M. AZT resistance alters enzymatic properties and creates an ATP-binding site in SFVmac reverse transcriptase. Retrovirology 2015, 12, 21. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tu, X.; Das, K.; Han, Q.; Bauman, J.D.; Clark, A.D.; Hou, X.; Frenkel, Y.V.; Gaffney, B.L.; Jones, R.A.; Boyer, P.L.; et al. Structural basis of HIV-1 resistance to AZT by excision. Nat. Struct. Mol. Biol. 2010, 17, 1202–1209. [Google Scholar] [CrossRef] [PubMed]
- Boyer, P.L.; Sarafianos, S.G.; Arnold, E.; Hughes, S.H. Selective excision of AZTMP by drug-resistant human immunodeficiency virus reverse transcriptase. J. Virol. 2001, 75, 4832–4842. [Google Scholar] [CrossRef]
- Ball, N.J.; Nicastro, G.; Dutta, M.; Pollard, D.J.; Goldstone, D.C.; Sanz-Ramos, M.; Ramos, A.; Müllers, E.; Stirnnagel, K.; Stanke, N.; et al. Structure of a Spumaretrovirus Gag Central Domain Reveals an Ancient Retroviral Capsid. PLoS Pathog. 2016, 12, e1005981. [Google Scholar] [CrossRef] [PubMed]
- Goldstone, D.C.; Flower, T.G.; Ball, N.J.; Sanz-Ramos, M.; Yap, M.W.; Ogrodowicz, R.W.; Stanke, N.; Reh, J.; Lindemann, D.; Stoye, J.P.; et al. A unique spumavirus Gag N-terminal domain with functional properties of orthoretroviral matrix and capsid. PLoS Pathog. 2013, 9, e1003376. [Google Scholar] [CrossRef]
- Valkov, E.; Gupta, S.S.; Hare, S.; Helander, A.; Roversi, P.; McClure, M.; Cherepanov, P. Functional and structural characterization of the integrase from the prototype foamy virus. Nucleic Acids Res. 2009, 37, 243–255. [Google Scholar] [CrossRef]
- Lesbats, P.; Serrao, E.; Maskell, D.P.; Pye, V.E.; O’Reilly, N.; Lindemann, D.; Engelman, A.N.; Cherepanov, P. Structural basis for spumavirus GAG tethering to chromatin. Proc. Natl. Acad. Sci. USA 2017, 114, 5509–5514. [Google Scholar] [CrossRef]
- Hare, S.; Gupta, S.S.; Valkov, E.; Engelman, A.; Cherepanov, P. Retroviral intasome assembly and inhibition of DNA strand transfer. Nature 2010, 464, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Nowak, E.; Miller, J.T.; Bona, M.K.; Studnicka, J.; Szczepanowski, R.H.; Jurkowski, J.; Le Grice, S.F.J.; Nowotny, M. Ty3 reverse transcriptase complexed with an RNA-DNA hybrid shows structural and functional asymmetry. Nat. Struct. Mol. Biol. 2014, 21, 389–396. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wöhrl, B.M. Structural and Functional Aspects of Foamy Virus Protease-Reverse Transcriptase. Viruses 2019, 11, 598. https://doi.org/10.3390/v11070598
Wöhrl BM. Structural and Functional Aspects of Foamy Virus Protease-Reverse Transcriptase. Viruses. 2019; 11(7):598. https://doi.org/10.3390/v11070598
Chicago/Turabian StyleWöhrl, Birgitta M. 2019. "Structural and Functional Aspects of Foamy Virus Protease-Reverse Transcriptase" Viruses 11, no. 7: 598. https://doi.org/10.3390/v11070598
APA StyleWöhrl, B. M. (2019). Structural and Functional Aspects of Foamy Virus Protease-Reverse Transcriptase. Viruses, 11(7), 598. https://doi.org/10.3390/v11070598