Novel Picobirnaviruses in Respiratory and Alimentary Tracts of Cattle and Monkeys with Large Intra- and Inter-Host Diversity
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Mammal and Poultry Surveillance and Sample Collection
2.3. RNA Extraction
2.4. RT-PCR for PBVs and DNA Sequencing
2.5. Complete/Near-Complete Segment 2 Sequencing of Genogroups I and II PBVs
2.6. Genome Segment 2 Analysis
2.7. Estimation of Synonymous and Non-Synonymous Substitution Rates
2.8. Nucleotide Sequence Accession Numbers
3. Results
3.1. Detection of PBVs in Respiratory Specimens of Animals
3.2. Complete/Near-Complete Segment 2 Sequence Analysis of Cattle and Monkey PBVs
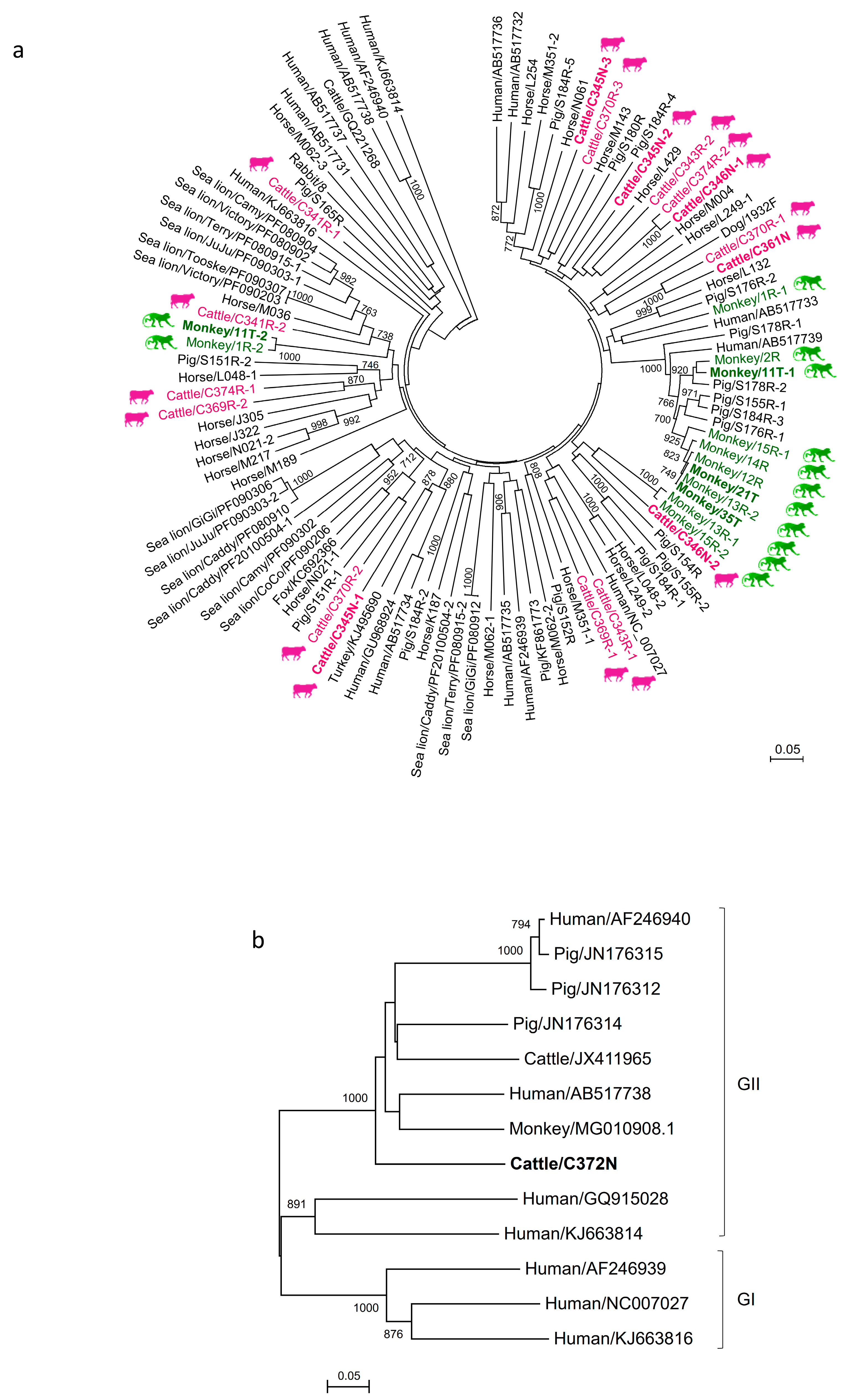
3.3. Phylogenetic Analysis of Cattle and Monkey PBVs’ Complete/Near-Complete Segment 2 Sequences
3.4. Estimation of Substitution Rates
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pereira, H.G.; Fialho, A.M.; Flewett, T.H.; Teixeira, J.M.; Andrade, Z.P. Novel viruses in human faeces. Lancet 1988, 2, 103–104. [Google Scholar] [CrossRef]
- Pereira, H.G.; Flewett, T.H.; Candeias, J.A.; Barth, O.M. A virus with a bisegmented double-stranded RNA genome in rat (oryzomys nigripes) intestines. J. Gen. Virol. 1988, 69 Pt 11, 2749–2754. [Google Scholar] [CrossRef]
- Bodewes, R.; van der Giessen, J.; Haagmans, B.L.; Osterhaus, A.D.; Smits, S.L. Identification of multiple novel viruses, including a parvovirus and a hepevirus, in feces of red foxes. J. Virol. 2013, 87, 7758–7764. [Google Scholar] [CrossRef] [PubMed]
- Fregolente, M.C.; de Castro-Dias, E.; Martins, S.S.; Spilki, F.R.; Allegretti, S.M.; Gatti, M.S. Molecular characterization of picobirnaviruses from new hosts. Virus Res. 2009, 143, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, B.; Nagashima, S.; Ghosh, S.; Nataraju, S.M.; Rajendran, K.; Manna, B.; Ramamurthy, T.; Niyogi, S.K.; Kanungo, S.; Sur, D.; et al. Detection and molecular characterization of multiple strains of picobirnavirus causing mixed infection in a diarrhoeic child: Emergence of prototype genogroup II-like strain in kolkata, india. Int. J. Mol. Epidemiol. Genet. 2011, 2, 61–72. [Google Scholar] [PubMed]
- Ghosh, S.; Kobayashi, N.; Nagashima, S.; Naik, T.N. Molecular characterization of full-length genomic segment 2 of a bovine picobirnavirus (PBV) strain: Evidence for high genetic diversity with genogroup I PBVs. J. Gen. Virol. 2009, 90, 2519–2524. [Google Scholar] [CrossRef] [PubMed]
- Gillman, L.; Sanchez, A.M.; Arbiza, J. Picobirnavirus in captive animals from uruguay: Identification of new hosts. Intervirology 2013, 56, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Kluge, M.; Campos, F.S.; Tavares, M.; de Amorim, D.B.; Valdez, F.P.; Giongo, A.; Roehe, P.M.; Franco, A.C. Metagenomic survey of viral diversity obtained from feces of subantarctic and south american fur seals. PLoS ONE 2016, 11, e0151921. [Google Scholar] [CrossRef]
- Luo, X.L.; Lu, S.; Jin, D.; Yang, J.; Wu, S.S.; Xu, J. Marmota himalayana in the qinghai-tibetan plateau as a special host for bi-segmented and unsegmented picobirnaviruses. Emerg. Microbes Infect. 2018, 7, 20. [Google Scholar] [CrossRef]
- Malik, Y.S.; Chandrashekar, K.M.; Sharma, K.; Haq, A.A.; Vaid, N.; Chakravarti, S.; Batra, M.; Singh, R.; Pandey, A.B. Picobirnavirus detection in bovine and buffalo calves from foothills of himalaya and central india. Trop. Anim. Health Prod. 2011, 43, 1475–1478. [Google Scholar] [CrossRef]
- Martinez, L.C.; Masachessi, G.; Carruyo, G.; Ferreyra, L.J.; Barril, P.A.; Isa, M.B.; Giordano, M.O.; Ludert, J.E.; Nates, S.V. Picobirnavirus causes persistent infection in pigs. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2010, 10, 984–988. [Google Scholar] [CrossRef] [PubMed]
- Ng, T.F.; Mesquita, J.R.; Nascimento, M.S.; Kondov, N.O.; Wong, W.; Reuter, G.; Knowles, N.J.; Vega, E.; Esona, M.D.; Deng, X.; et al. Feline fecal virome reveals novel and prevalent enteric viruses. Vet. Microbiol. 2014, 171, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Pankovics, P.; Boros, A.; Nemes, C.; Kapusinszky, B.; Delwart, E.; Reuter, G. Molecular characterization of a novel picobirnavirus in a chicken. Arch. Virol. 2018, 163, 3455–3458. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro Silva, R.; Bezerra, D.A.; Kaiano, J.H.; Oliveira Dde, S.; Silvestre, R.V.; Gabbay, Y.B.; Ganesh, B.; Mascarenhas, J.D. Genogroup I avian picobirnavirus detected in brazilian broiler chickens: A molecular epidemiology study. J. Gen. Virol. 2014, 95, 117–122. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shi, M.; Lin, X.D.; Tian, J.H.; Chen, L.J.; Chen, X.; Li, C.X.; Qin, X.C.; Li, J.; Cao, J.P.; Eden, J.S.; et al. Redefining the invertebrate rna virosphere. Nature 2016, 540, 539–543. [Google Scholar] [CrossRef] [PubMed]
- Symonds, E.M.; Griffin, D.W.; Breitbart, M. Eukaryotic viruses in wastewater samples from the United States. Appl. Environ. Microbiol. 2009, 75, 1402–1409. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Banyai, K.; Tu, X.; Jiang, B. Simian genogroup I picobirnaviruses: Prevalence, genetic diversity, and zoonotic potential. J. Clin. Microbiol. 2012, 50, 2779–2782. [Google Scholar] [CrossRef]
- Woo, P.C.; Teng, J.L.; Bai, R.; Wong, A.Y.; Martelli, P.; Hui, S.W.; Tsang, A.K.; Lau, C.C.; Ahmed, S.S.; Yip, C.C.; et al. High diversity of genogroup i picobirnaviruses in mammals. Front. Microbiol. 2016, 7, 1886. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, L.; Deng, X.; Kapusinszky, B.; Pesavento, P.A.; Delwart, E. Faecal virome of cats in an animal shelter. J. Gen. Virol. 2014, 95, 2553–2564. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Bai, R.; Teng, J.L.; Lee, P.; Martelli, P.; Hui, S.W.; Yuen, K.Y. Complete genome sequence of a novel picobirnavirus, otarine picobirnavirus, discovered in california sea lions. J. Virol. 2012, 86, 6377–6378. [Google Scholar] [CrossRef]
- Woo, P.C.; Lau, S.K.; Teng, J.L.; Tsang, A.K.; Joseph, M.; Wong, E.Y.; Tang, Y.; Sivakumar, S.; Bai, R.; Wernery, R.; et al. Metagenomic analysis of viromes of dromedary camel fecal samples reveals large number and high diversity of circoviruses and picobirnaviruses. Virology 2014, 471–473, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Wakuda, M.; Pongsuwanna, Y.; Taniguchi, K. Complete nucleotide sequences of two RNA segments of human picobirnavirus. J. Virol. Methods 2005, 126, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Rosen, B.I.; Fang, Z.Y.; Glass, R.I.; Monroe, S.S. Cloning of human picobirnavirus genomic segments and development of an rt-pcr detection assay. Virology 2000, 277, 316–329. [Google Scholar] [CrossRef] [PubMed]
- Delmas, B.; Attoui, H.; Ghosh, S.; Malik, Y.S.; Mundt, E.; Vakharia, V.N.; ICTV Report Consortium. ICTV virus taxonomy profile: Picobirnaviridae. J. Gen. Virol. 2019, 100, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Kirkwood, C.D. Genetic and antigenic diversity of human rotaviruses: Potential impact on vaccination programs. J. Infect. Dis. 2010, 202 (Suppl. 1), S43–S48. [Google Scholar] [CrossRef]
- Komoto, S.; Apondi, E.W.; Shah, M.; Odoyo, E.; Nyangao, J.; Tomita, M.; Wakuda, M.; Maeno, Y.; Shirato, H.; Tsuji, T.; et al. Whole genomic analysis of human G12P[6] and G12P[8] rotavirus strains that have emerged in kenya: Identification of porcine-like NSP4 genes. Infect. Genet. Evol. 2014, 27, 277–293. [Google Scholar] [CrossRef] [PubMed]
- Smits, S.L.; Poon, L.L.; van Leeuwen, M.; Lau, P.N.; Perera, H.K.; Peiris, J.S.; Simon, J.H.; Osterhaus, A.D. Genogroup I and II picobirnaviruses in respiratory tracts of pigs. Emerg. Infect. Dis. 2011, 17, 2328–2330. [Google Scholar] [CrossRef] [PubMed]
- Smits, S.L.; van Leeuwen, M.; Schapendonk, C.M.; Schurch, A.C.; Bodewes, R.; Haagmans, B.L.; Osterhaus, A.D. Picobirnaviruses in the human respiratory tract. Emerg. Infect. Dis. 2012, 18, 1539–1540. [Google Scholar] [CrossRef] [PubMed]
- Lau, S.K.; Woo, P.C.; Li, K.S.; Huang, Y.; Tsoi, H.W.; Wong, B.H.; Wong, S.S.; Leung, S.Y.; Chan, K.H.; Yuen, K.Y. Severe acute respiratory syndrome coronavirus-like virus in chinese horseshoe bats. Proc. Natl. Acad. Sci. USA 2005, 102, 14040–14045. [Google Scholar] [CrossRef]
- Kumar, S.; Nei, M.; Dudley, J.; Tamura, K. Mega: A biologist-centric software for evolutionary analysis of DNA and protein sequences. Brief. Bioinform. 2008, 9, 299–306. [Google Scholar] [CrossRef]
- Attoui, H.; Billoir, F.; Cantaloube, J.F.; Biagini, P.; de Micco, P.; de Lamballerie, X. Strategies for the sequence determination of viral dsrna genomes. J. Virol. Methods 2000, 89, 147–158. [Google Scholar] [CrossRef]
- Lambden, P.R.; Cooke, S.J.; Caul, E.O.; Clarke, I.N. Cloning of noncultivatable human rotavirus by single primer amplification. J. Virol. 1992, 66, 1817–1822. [Google Scholar] [PubMed]
- Hofacker, I.L. Vienna RNA secondary structure server. Nucleic Acids Res. 2003, 31, 3429–3431. [Google Scholar] [CrossRef] [PubMed]
- Nates, S.V.; Gatti, M.S.V.; Ludert, J.E. The picobirnavirus: An integrated view on its biology, epidemiology and pathogenic potential. Future Virol. 2011, 6, 223–235. [Google Scholar] [CrossRef]
- Ng, T.F.; Kondov, N.O.; Deng, X.; Van Eenennaam, A.; Neibergs, H.L.; Delwart, E. A metagenomics and case-control study to identify viruses associated with bovine respiratory disease. J. Virol. 2015, 89, 5340–5349. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Sun, H.; Lan, D.; Hua, X.; Cui, L.; Yuan, C.; Yang, Z. Molecular detection of genogroup I and II picobirnaviruses in pigs in china. Virus Genes 2014, 48, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Ganesh, B.; Nataraju, S.M.; Rajendran, K.; Ramamurthy, T.; Kanungo, S.; Manna, B.; Nagashima, S.; Sur, D.; Kobayashi, N.; Krishnan, T. Detection of closely related picobirnaviruses among diarrhoeic children in kolkata: Evidence of zoonoses? Infect. Genet. Evol. 2010, 10, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, M.; Williams, M.M.; Koraka, P.; Simon, J.H.; Smits, S.L.; Osterhaus, A.D. Human picobirnaviruses identified by molecular screening of diarrhea samples. J. Clin. Microbiol. 2010, 48, 1787–1794. [Google Scholar] [CrossRef]
- Banyai, K.; Martella, V.; Bogdan, A.; Forgach, P.; Jakab, F.; Meleg, E.; Biro, H.; Melegh, B.; Szucs, G. Genogroup I picobirnaviruses in pigs: Evidence for genetic diversity and relatedness to human strains. J. Gen. Virol. 2008, 89, 534–539. [Google Scholar] [CrossRef]
- Duraisamy, R.; Akiana, J.; Davoust, B.; Mediannikov, O.; Michelle, C.; Robert, C.; Parra, H.J.; Raoult, D.; Biagini, P.; Desnues, C. Detection of novel rna viruses from free-living gorillas, republic of the congo: Genetic diversity of picobirnaviruses. Virus Genes 2018, 54, 256–271. [Google Scholar] [CrossRef]
- Ghosh, S.; Shiokawa, K.; Aung, M.S.; Malik, Y.S.; Kobayashi, N. High detection rates of picobirnaviruses in free roaming rats (Rattus spp.): Molecular characterization of complete gene segment-2. Infect. Genet. Evol. 2018, 65, 131–135. [Google Scholar] [CrossRef] [PubMed]
- Kuhar, U.; Jamnikar-Ciglenecki, U. High detection rate and high genetic diversity of genogroup I picobirnaviruses from roe deer. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2019, 73, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Malik, Y.S.; Kumar, N.; Sharma, K.; Sharma, A.K.; Sircar, S.; Jeena, L.M.; Singh, N.K.; Mondal, A.; Joardar, S.N.; Balasubramanian, G. Molecular characterization of a genetically diverse bubaline picobirnavirus strain, india. Thai J. Vet. Med. 2013, 43, 609–613. [Google Scholar]
- Malik, Y.S.; Sharma, A.K.; Kumar, N.; Sharma, K.; Ganesh, B.; Kobayashi, N. Identification and characterisation of a novel genogroup ii picobirnavirus in a calf in india. Vet. Rec. 2014, 174, 278. [Google Scholar] [CrossRef] [PubMed]
- Malik, Y.S.; Sircar, S.; Dhama, K.; Singh, R.; Ghosh, S.; Banyai, K.; Vlasova, A.N.; Nadia, T.; Singh, R.K. Molecular epidemiology and characterization of picobirnaviruses in small ruminant populations in india. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2018, 63, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, R.; Sahoo, G.C.; Nayak, M.K.; Rajendran, K.; Dutta, P.; Mitra, U.; Bhattacharya, M.K.; Naik, T.N.; Bhattacharya, S.K.; Krishnan, T. Detection of genogroup i and ii human picobirnaviruses showing small genomic rna profile causing acute watery diarrhoea among children in kolkata, india. Infect. Genet. Evol. 2007, 7, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Krishnamurthy, S.R.; Wang, D. Extensive conservation of prokaryotic ribosomal binding sites in known and novel picobirnaviruses. Virology 2018, 516, 108–114. [Google Scholar] [CrossRef]

| Animal Type | Sample Types | No. of Specimens Obtained | No. (%) of Specimens Positive for Genogroup I PBVs | No. (%) of Specimens Positive for Genogroup II PBVs |
|---|---|---|---|---|
| Mammals | ||||
| Cattle | Nasopharyngeal swab | 51 | 3 (5.9%) | 1 (1.9%) |
| Monkey 1 | Throat swab | 52 | 3 (5.8%) | 0 (0%) |
| Dog | Nasopharyngeal swab | 50 | 0 (0%) | 0 (0%) |
| Cat | Nasopharyngeal swab | 50 | 0 (0%) | 0 (0%) |
| Bat 2 | Mouth swab | 157 | 0 (0%) | 0 (0%) |
| Horse 3 | Nasopharyngeal swab | 116 | 0 (0%) | 0 (0%) |
| Poultry | ||||
| Chicken | Tracheal swab | 100 | 0 (0%) | 0 (0%) |
| PBV | Genome Features | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Size (bp) | G+C Content (%) | ORF Features | 5′ UTR Features | 3′ UTR Features | ||||||||||
| ORF | Location (nt) | Length (nt) | Length (aa) | Frame | Length (nt) | G+C Content (%) | Beginning Bases | Length (nt) | G+C Content (%) | Ending Bases | ||||
| Reference genogroup I PBVs segment 2 sequences | ||||||||||||||
| Human 1 | 1745 | 46.36 | RdRp | 94-1698 | 1605 | 534 | +1 | 93 | 21.51 | GTAAA | 47 | 53.19 | CTGC | |
| Human 2 | 1696 | 43.63 | RdRp | 58-1650 | 1593 | 530 | +1 | 57 | 24.56 | GTAAA | 46 | 50.00 | CTGC | |
| Reference genogroup II PBV segment 2 sequence | ||||||||||||||
| Human 3 | 1674 | 43.91 | RdRp | 88-1641 | 1554 | 517 | +1 | 87 | 16.09 | GTAAA | 33 | 57.58 | CTC | |
| Genogroup I PBVs detected in this study | ||||||||||||||
| PBVs (Clade) | GenBank accession no. | |||||||||||||
| Monkey/11T-1 (M-R1;) | KY120190 | 1678 | 42.55 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/11T-2 (M-R2) | KY120191 | 1681 | 41.05 | RdRp | 47-1636 | 1590 | 529 | +2 | 46 | 26.09 | GTAAA | 45 | 51.11 | CTGC |
| Monkey/35T (M-R1) | KY120193 | 1678 | 42.61 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/21T (M-R1) | KY120192 | 1678 | 42.49 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/1R-1 4 (M-R3) | KY120194 | 1613 | 42.72 | RdRp | 2-1564 | 1563 | 520 | NA | NA | NA | NA | 49 | 53.06 | CTGC |
| Monkey/1R-2 (M-R2) | KY120182 | 1681 | 41.52 | RdRp | 47-1636 | 1590 | 529 | +2 | 46 | 26.09 | GTAAA | 45 | 51.11 | CTGC |
| Monkey/2R (M-R1) | KY120183 | 1678 | 42.37 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 50.00 | CTGC |
| Monkey/12R (M-R1) | KY120184 | 1678 | 42.61 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/13R-1 (M-R3) | KY120185 | 1734 | 41.64 | RdRp | 75-1685 | 1611 | 536 | +3 | 74 | 25.68 | GTAAA | 49 | 53.06 | CTGC |
| Monkey/13R-2 (M-R1) | KY120186 | 1678 | 42.55 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/14R (M-R1) | KY120187 | 1678 | 42.55 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 52.17 | CTGC |
| Monkey/15R-1 (M-R1) | KY120188 | 1678 | 42.31 | RdRp | 28-1632 | 1605 | 534 | +1 | 27 | 14.81 | GTAAA | 46 | 50.00 | CTGC |
| Monkey/15R-2 (M-R3) | KY120189 | 1734 | 41.64 | RdRp | 75-1685 | 1611 | 536 | +3 | 74 | 25.68 | GTAAA | 49 | 53.06 | CTGC |
| Cattle/C345N-2 | KY120170 | 1671 | 43.93 | RdRp | 49-1629 | 1581 | 526 | +1 | 48 | 25.00 | GTAAA | 42 | 52.38 | CATC |
| Cattle/C345N-3 | KY120171 | 1694 | 42.15 | RdRp | 44-1657 | 1614 | 537 | +2 | 43 | 23.26 | GTAAA | 37 | 59.46 | CTGC |
| Cattle/C346N-1 | KY120172 | 1701 | 47.68 | RdRp | 49-1656 | 1608 | 535 | +1 | 48 | 29.17 | GTAAA | 45 | 44.44 | CTGC |
| Cattle/C346N-2 | KY120173 | 1696 | 44.99 | RdRp | 52-1647 | 1596 | 531 | +1 | 51 | 27.45 | GTAAA | 49 | 46.94 | CTGC |
| Cattle/C361N | KY120174 | 1696 | 44.58 | RdRp | 49-1653 | 1605 | 534 | +1 | 48 | 35.42 | GTAAA | 43 | 44.19 | CTGC |
| Cattle/C343R-1 | KY120175 | 1692 | 44.03 | RdRp | 50-1642 | 1593 | 530 | +2 | 49 | 20.41 | GTAAA | 50 | 58.00 | CTGC |
| Cattle/C343R-2 | KY120176 | 1695 | 45.25 | RdRp | 48-1652 | 1605 | 534 | +3 | 47 | 25.53 | GTAAA | 43 | 44.19 | CTGC |
| Cattle/C369R-1 | KY120177 | 1736 | 42.45 | RdRp | 36-1721 | 1686 | 561 | +3 | 35 | 22.86 | GTAAA | 15 | 40.00 | CTTC |
| Cattle/C369R-2 5 | KY120179 | 1659 | 44.91 | RdRp | 24-1616 | 1593 | 530 | NA | 23 | 30.43 | NA | 43 | 55.81 | CTGC |
| Cattle/C374R-1 5 | KY120180 | 1651 | 44.16 | RdRp | 8-1606 | 1599 | 532 | NA | 7 | 28.57 | NA | 45 | 55.56 | CTGC |
| Cattle/C374R-2 5 | KY120181 | 1669 | 47.15 | RdRp | 24-1625 | 1602 | 533 | NA | 23 | 34.78 | NA | 44 | 47.73 | CTGC |
| Genogroup II PBVs detected in this study | ||||||||||||||
| Cattle/C372N | KY120178 | 1622 | 40.44 | RdRp | 55-1599 | 1545 | 514 | +1 | 54 | 16.67% | GTAAA | 23 | 52.17 | CTCC |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woo, P.C.Y.; Teng, J.L.L.; Bai, R.; Tang, Y.; Wong, A.Y.P.; Li, K.S.M.; Lam, C.S.F.; Fan, R.Y.Y.; Lau, S.K.P.; Yuen, K.-Y. Novel Picobirnaviruses in Respiratory and Alimentary Tracts of Cattle and Monkeys with Large Intra- and Inter-Host Diversity. Viruses 2019, 11, 574. https://doi.org/10.3390/v11060574
Woo PCY, Teng JLL, Bai R, Tang Y, Wong AYP, Li KSM, Lam CSF, Fan RYY, Lau SKP, Yuen K-Y. Novel Picobirnaviruses in Respiratory and Alimentary Tracts of Cattle and Monkeys with Large Intra- and Inter-Host Diversity. Viruses. 2019; 11(6):574. https://doi.org/10.3390/v11060574
Chicago/Turabian StyleWoo, Patrick C. Y., Jade L. L. Teng, Ru Bai, Ying Tang, Annette Y. P. Wong, Kenneth S. M. Li, Carol S. F. Lam, Rachel Y. Y. Fan, Susanna K. P. Lau, and Kwok-Yung Yuen. 2019. "Novel Picobirnaviruses in Respiratory and Alimentary Tracts of Cattle and Monkeys with Large Intra- and Inter-Host Diversity" Viruses 11, no. 6: 574. https://doi.org/10.3390/v11060574
APA StyleWoo, P. C. Y., Teng, J. L. L., Bai, R., Tang, Y., Wong, A. Y. P., Li, K. S. M., Lam, C. S. F., Fan, R. Y. Y., Lau, S. K. P., & Yuen, K.-Y. (2019). Novel Picobirnaviruses in Respiratory and Alimentary Tracts of Cattle and Monkeys with Large Intra- and Inter-Host Diversity. Viruses, 11(6), 574. https://doi.org/10.3390/v11060574







