1. Introduction
The virus family
Caliciviridae contains five established genera (
Norovirus,
Sapovirus,
Lagovirus,
Vesivirus, and
Nebovirus) and at least six proposed genera (
Recovirus,
Valovirus,
Bavovirus,
Nacovirus,
Minovirus, and
Salovirus) that infect many different animal species including humans. Most of these agents are enteric pathogens whose replication and chief clinical manifestations are gastroenteritis and potentially life-threatening diarrhea. Examples of these viruses include human norovirus (HuNoV), porcine norovirus, bovine norovirus, human sapovirus, porcine sapovirus, and the recently discovered Tulane virus. HuNoV is the major food- and water-borne virus that accounts for more than 95% of nonbacterial acute gastroenteritis worldwide, but this percentage may be underestimated due to the large number of asymptomatic HuNoV infections and lack of proper detection methods [
1,
2,
3,
4]. In addition, HuNoV is responsible for over 50% of the outbreaks of foodborne illnesses in the USA [
5]. The symptoms often involve projectile vomiting, diarrhea, nausea, and low-grade fever [
1,
2,
6]. HuNoV is transmitted primarily through the fecal–oral route, either by direct person-to-person contact or by fecally contaminated food or water. Although HuNoV infection is usually self-limited disease, it is highly contagious, and only a few particles are thought to be sufficient to cause an infection [
1,
2,
7]. Currently, the National Institute of Allergy and Infectious Diseases (NIAID) classify HuNoV and other caliciviruses as category B priority biodefense pathogens.
Unfortunately, researches on HuNoVs have been hampered due to the fact that it cannot efficiently be grown in cell culture system and lack a robust small animal model for infectivity and pathogenesis study [
1,
2,
4]. Recently, two separate groups in the US reported the replication of HuNoV in cell culture. Jones et al. showed that HuNoV can be grown in human B cells, and that commensal bacteria (such as
Enterobacter cloacae) facilitate such replication [
8]. Ettayebi et al. reported that multiple HuNoV strains can replicate in stem cell-derived human enteroids [
9]. Although these studies are highly promising, the robustness of these culture systems needs to be further validated. Despite major efforts, there are still no FDA-approved vaccines or antiviral drugs are available for HuNoV. Recent epidemiological studies found that severe clinical outcomes including death are often associated with high-risk populations such as the elderly, children, and immunocompromised individuals [
5,
10,
11,
12]. From 1999 to 2007 HuNoV caused, on average, 797 deaths per year in the USA. Mortality of HuNoV associated infection increases during the epidemic seasons and the burden of HuNoV is much greater in the developing world. The CDC estimates that HuNoV causes the death of 200,000 children under the age of five every year in developing countries. Therefore, there is an urgent need to develop an efficacious vaccine and therapeutic agent for control and prevent HuNoV.
Antibody-based passive immunization has been shown to be an effective strategy to prevent and treat infectious diseases [
13]. Also, rapid and immediate protection can be achieved after passive immunization, for example, against agents of bioterrorism [
14]. Using mammalian serum to produce antibodies for oral administration has been described previously [
15]. However, its application is limited by the high cost of large-scale antibody production and time-consuming [
16,
17]. In addition, passive immunization with polyclonal antibodies has also been shown to have higher levels of protection compared to monoclonal antibodies [
18]. Immunoglobulin Y (IgY)—the egg yolk antibodies generated as a passive immunity to embryos and baby chicks [
19]—can be a good alternative for large-scale production of polyclonal antibodies at a lower cost. Chicken IgYs are transferred from blood to the egg yolk during embryo development [
16]. IgY can be easily produced and purified with high yields from egg yolks of immunized hens. In addition, production of antibodies via laying hens only requires the collection of eggs, and the animal number can be reduced due to the high and long-lasting titers produced in chickens [
18,
19]. Therefore, the IgY technology is a safe, convenient, and inexpensive strategy to prevent and control infectious diseases, especially for gastrointestinal infections [
20,
21].
Previous reports have shown that HuNoV-specific IgY could be produced by immunization of hens with HuNoV virus-like particles (VLPs) or P particles, which were purified from insect cells using a baculovirus expression system [
18,
22]. In addition, these IgYs can block the binding of HuNoV VLPs to its receptors, the histo-blood group antigens (HBGA), suggesting that IgY may potentially be used as passive immunization and therapeutic agent for HuNoV. Although these results are promising, purification of VLPs from insect cells is time-consuming and expensive which may limit the large-scale production of IgY. Previously, our laboratory developed a more efficient, convenient, and economical strategy to produce HuNoV VLPs [
23,
24]. We constructed a recombinant vesicular stomatitis virus expressing VP1 gene of a HuNoV GII.4 strain (rVSV-VP1). The VLP production by rVSV-VP1 was significantly higher than that by insect cells-baculovirus expression system. Vaccination of mice with rVSV-VP1 triggered significantly higher HuNoV-specific humoral, cellular, and mucosal immunities than traditional VLP vaccination [
23,
24].
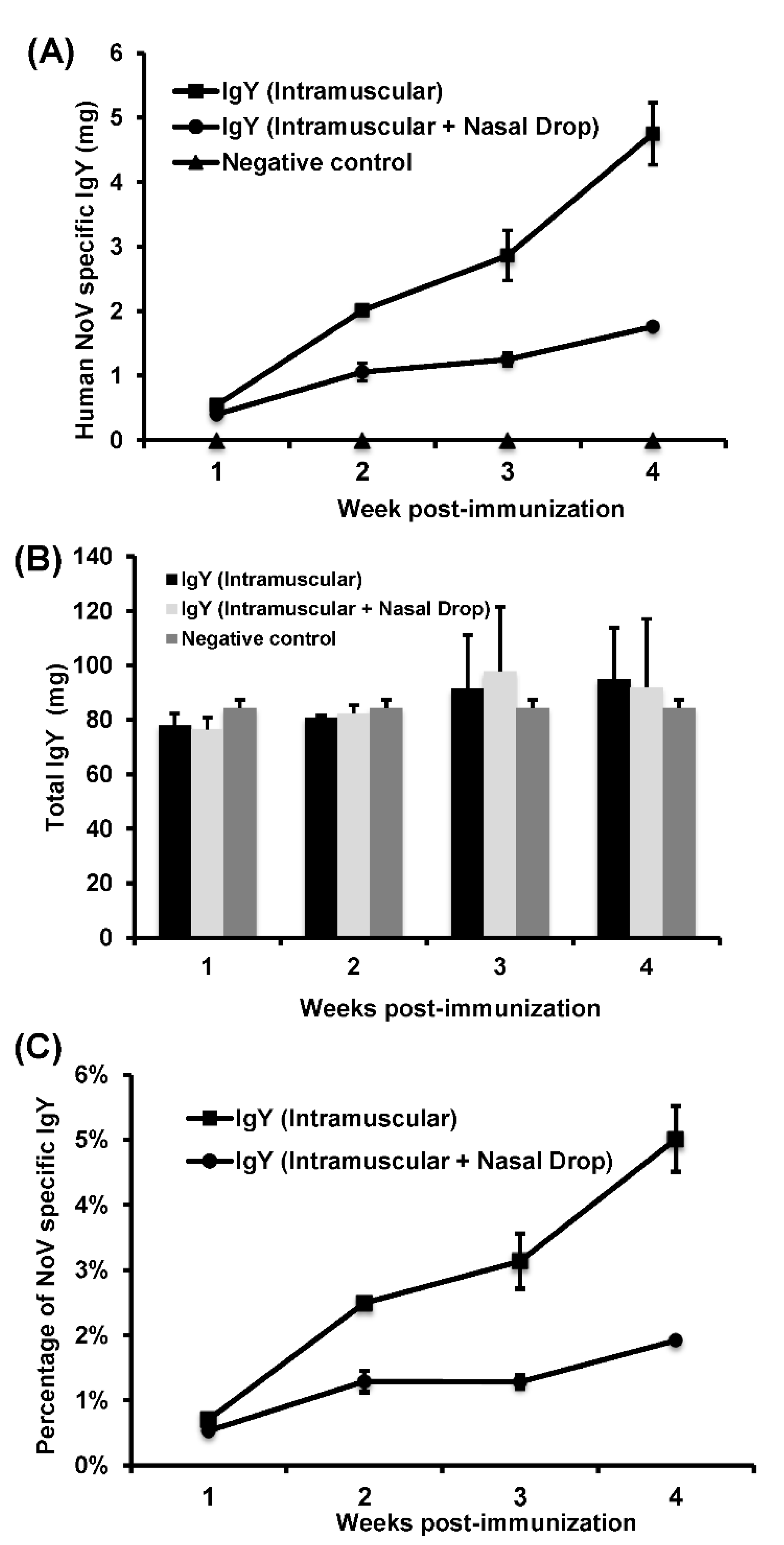
In this study, we developed a highly efficient bioreactor for large-scale production of chicken egg yolk IgY antibodies using rVSV-VP1 as an antigen. We found that HuNoV VP1 was highly expressed in chicken cells infected by rVSV-VP1 and that hens vaccinated rVSV-VP1 induced a high level of HuNoV-specific IgY in egg yolks. Interestingly, intramuscular vaccination of rVSV-VP1 produced approximately three times more HuNoV-specific IgY than combination of intramuscular and nasal drop vaccination route. Importantly, HuNoV-specific IgY produced by rVSV-VP1 vaccination was capable of blocking the binding of HuNoV VLPs to type A, B, and O HBGA receptors. In addition, egg yolk IgY antibodies remained stable at temperature below 70 °C and at pH ranging from 4 to 9. These data demonstrated that recombinant rVSV-VP1 is a highly effective antigen for large-scale production of HuNoV-specific IgY for passive immunization and therapeutic agent.
2. Materials and Methods
2.1. Virus and Cell Culture
Recombinant vesicular stomatitis virus (rVSV) expressing human NoV capsid protein (rVSV-VP1) was previously constructed in our laboratory [
23]. Working stocks of rVSV-VP1 were propagated in confluent BSRT7 cells. Briefly, BSRT7 cells in T150 flask were infected by rVSV-VP1 at a multiplicity of infection (MOI) of 10. After 1 h of absorption, the inoculum was removed, and the cells were washed twice with Dulbecco’s modified Eagle’s medium (DMEM). After addition of 15 mL fresh DMEM (supplemented with 2% fetal bovine serum), the infected cells were incubated at 37 °C in CO
2 incubator. When extensive cytopathic effects (CPE) were observed, cell culture fluid was harvested, and virus titer was determined by plaque assay in Vero cells.
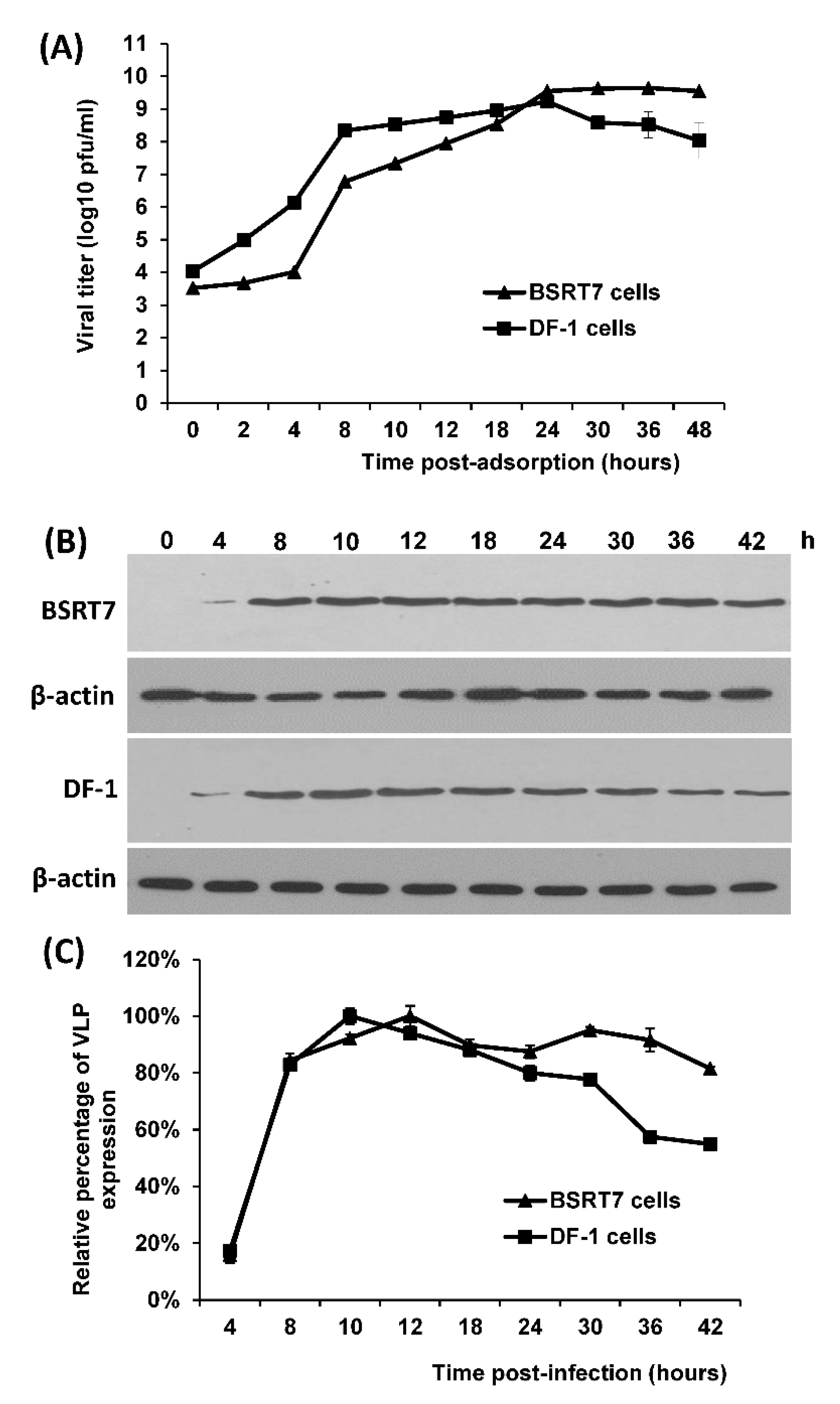
2.2. Multistep Growth Curves in BSRT7 and Chicken DF-1 Cells
Thirty-five-millimeter dishes were seeded with BSRT-7 or DF-1cells (kindly provided by Dr. Qingzhong Yu at USDA ARS, Athens, GA) and were infected with rVSV-VP1 at a multiplicity of infection (MOI) of 10. After 1 h of absorption, the inoculum was removed, the cells were washed twice with DMEM, 2 mL of fresh DMEM (supplemented with 2% fetal bovine serum) was added, and the infected cells were incubated at 37 °C. At the indicated intervals, 500-µL aliquots of the cell culture fluid were removed and the same amount of fresh DMEM was added back to the virus-infected cells. Virus titers were determined by plaque assay in Vero cells.
2.3. Determination of the Kinetics of VP1 Expression in Cell Culture
Six-well plates were seeded by BSRT7 or DF-1cells (2 × 105 cells per well). After 24 h incubation at 37 °C in CO2 incubator the cells were infected with rVSV-VP1 at an MOI of 10. At the indicated time points, cells were lysed in lysis buffer containing 5% β-mercaptoethanol, 0.01% NP-40, and 2% sodium dodecyl sulfate (SDS). Proteins were separated by 12% SDS-PAGE and transferred to a Hybond ECL nitrocellulose membrane (Amersham, Piscataway, NJ, USA) in a Mini Trans-Blot electrophoretic transfer cell (Bio-Rad, Hercules, CA, USA). The blot was probed with guinea pig anti-human NoV VP1 antiserum (a generous gift from Dr. Xi Jiang) at a dilution of 1:6000, followed by horseradish peroxidase-conjugated goat anti-guinea pig IgG secondary antibody (Santa Cruz Biotechnology, Inc., Dallas, TX, USA) at a dilution of 1:20,000. The blot was developed with SuperSignal West Pico Chemiluminescent Substrate (Thermo Scientific, Waltham, MA, USA) and exposed to Kodak BioMax MR film (Kodak, Rochester, NY, USA). The harvested cell lysates were also subjected to Western blot using anti β-actin antibody (Proteintech, Rosemont, IL, USA), which recognizes β-actin from multiple species, including human, mouse, hamster, and chicken. The protein bands were scanned and quantified using a Typhoon PhosphorImager and ImageQuant TL software (GE Healthcare, Piscataway, NJ, USA). The VP1 protein band of each time point was normalized by the respective β-actin band. The time point (12 h postinfection in BSRT7 cells) with highest VP1 expression was set as 100%. The percentage of VP1 expression at other time points was normalized by the VP1 expression at 12 h postinfection in BSRT7 cells.
2.4. Chickens and Immunization
The animal study was conducted in strict accordance with USDA regulations and the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health, and was approved by The Ohio State University Institutional Animal Care and Use Committee (animal protocol no. 2013A00000011). Chickens were housed in cages inside high-security isolation rooms provided with HEPA-filtered intake and exhaust air at The Ohio Agriculture Research and Development Center, The Ohio State University. The animal care facilities at The Ohio State University are AAALAC accredited. Before animal study, blood samples were collected from each chicken to confirm that they were negative for HuNoV antibody.
Six, 21-week-old, healthy White Leghorn chickens were provided by Dr. Lilburn, Department of Animal Sciences, The Ohio State University and were randomly divided into two groups (three chickens per group). Prior to the study, these chickens were negative for VSV and HuNoV antibody. Chickens in Group I were immunized intramuscularly by injecting 500 µL of DMEM containing 5 × 107 PFU of rVSV-VP1 into three different locations of the pectoral muscle. Chickens in Group II were immunized by combination of intramuscular and intranasal routes. Specifically, 300 µL of rVSV-VP1 (3 × 107 PFU) was injected into three different locations of the pectoral muscle, and the remaining 200 µL of rVSV-VP1 (2 × 107 PFU) was used for nasal drop vaccination. At week 2 postimmunization, chickens in groups I and II were boosted with 5 × 107 PFU of rVSV-VP1 via intramuscular and combination of intramuscular and intranasal routes, respectively. After immunization, eggs were collected daily until week 4 post-booster vaccination. In addition, hens were observed daily for any abnormal reactions. Eggs that were collected one week before immunization were used as negative control. Eggs were stored at 4 °C before IgY extraction.
2.5. Production and Purification of HuNoV VLPs by a Baculovirus Expression System
Purification of VLPs from insect cells was described previously with some minor modifications [
23].
Spodoptera frugiperda (Sf9) cells were infected with baculovirus expressing HuNoV VP1 at an MOI of 10, and the infected Sf9 cells and cell culture supernatants were harvested at 6 days postinoculation. The VLPs were purified from cell culture supernatants and cell lysates by ultracentrifugation through a 40% (
w/
v) sucrose cushion, followed by CsCl isopycnic gradient (0.39 g/cm
3) ultracentrifugation. Purified VLPs were analyzed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) followed by Coomassie blue staining. The protein concentrations of the VLPs were measured by using the Bradford reagent (Sigma Chemical Co., St. Louis, MO, USA).
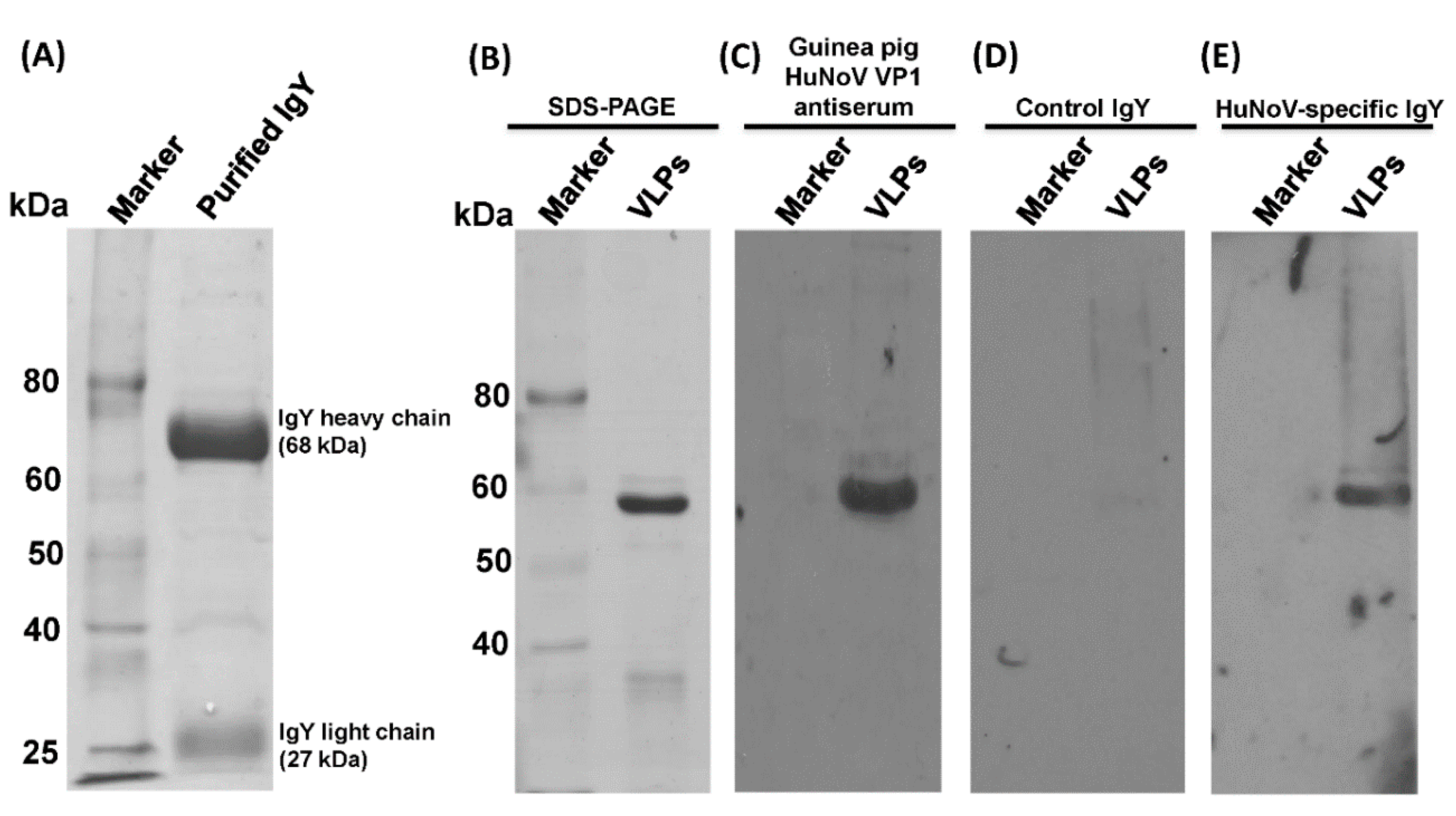
2.6. Extraction and Purification of IgY from Chicken Egg Yolks
IgY was extracted and purified from egg yolks using polyethylene glycol 8000 (PEG 8000, Sigma, St. Louis, MO, USA) precipitation method [
25] with some modifications. Briefly, egg yolks were diluted in three volumes of PBS (pH 7.4) and mixed, and PEG 8000 was added to a final concentration of 3.5%. After vortexing, the mixture continued to roll on a rolling mixer for 20 min. The mixtures were centrifuged at 13,000×
g for 20 min at 4 °C, and the precipitated debris were removed. Subsequently, PEG 8000 was added to the supernatant to a final concentration of 8.5%, and the samples were mixed on a rolling mixer for 20 min. The mixtures were centrifuged again at 13,000×
g for 20 min at 4 °C. The precipitated pellets containing IgY were dissolved in 10 mL of PBS and then precipitated again with 12% of PEG 8000 using the same procedures described above. The final pellets was dissolved in 2.0 mL of PBS, filtered through a 0.45 µm filter, and stored at −20 °C. The purity of the IgY was determined by SDS-PAGE followed by Coomassie blue staining.
2.7. Determination of HuNoV-Specific IgY and Total IgY Titers in Egg Yolk
Standard ELISA measured HuNoV-specific IgY antibody titers. Briefly, 96-well microtiter plates were coated with 100 μL of purified HuNoV VLP antigen (200 ng/well) and incubated overnight at 4 °C. After blocking with 5% nonfat milk, 10 times serially diluted chicken IgYs were added to the antigen-coated wells and incubated at 37 °C for 1 h. After washing with PBST (PBS containing 0.05% Tween), goat anti-chicken IgY-HRP (1:5000) (Santa Cruz Biotechnology) was added for 1 h. Plates were washed and developed with 75 µL of 3,3’,5,5’-tetramethylbenzidine (TMB), and the optical density (OD) at 450 nm was determined using an enzyme-linked immunosorbent assay (ELISA) plate reader. The IgYs from pre-immunized chicken yolks were used as controls. To calculate the amount of total IgY and HuNoV-specific IgY, a standard curve was set-up: wells were coated with 100 μL of serially diluted pure chicken IgY (Promega, Madison, WI, USA) at a concentration from 0.0075 μg/mL to 1 μg/mL. After washing with PBST, 100 μL of goat anti-chicken IgY-HRP (Santa Cruz Biotechnology) at a dilution of 1:1000) were added and incubated at 37 °C for 1 h. The bound HRP was colorized by substrate reagent (Kirkegaard and Perry Laboratories, Inc., Gaithersburg, Maryland, USA), followed by a reading of the signal intensity at 450 nm (Epoch Micro-Volume Spectrophotometer System, BioTek, Winooski, VT, USA). The resulting standard curve of absorbance was used to quantify the relative concentration of total IgY and HuNoV-specific IgY from the egg yolks by coating plates with either HuNoV VLP or rabbit anti-chicken IgY antibodies (10 μg/mL, Sigma) to capture the total IgY or HuNoV-specific IgY.
2.8. Analysis of Chicken IgY by SDS-PAGE
The purified IgYs from egg yolks were analyzed by SDS-PAGE. Samples were boiled for 5 min in loading buffer containing 1% SDS, 2.5% β-mercaptoethanol, 6.25 mM Tris-HCl (pH 6.8), and 5% glycerol and loaded onto a 12% polyacrylamide gel. Proteins were visualized by Coomassie blue staining.
2.9. Characterization of IgY by Western Blot Analysis
Specific reactions of the chicken IgY with HuNoV capsid protein were examined by Western blot analysis. The purified HuNoV virus-like particles (VLPs) were separated by conventional 12% SDS-PAGE and transferred to a Hybond ECL nitrocellulose membrane (Amersham) in a Mini Trans-Blot electrophoretic transfer cell (Bio-Rad). After blocking with 5% nonfat milk, the membrane was incubated with anti-HuNoV-specific IgY or nonspecific IgY (1:1000) in 1% nonfat milk-PBS at 4 °C overnight, followed by horseradish peroxidase (HRP)-conjugated goat anti- chicken IgY secondary antibody (Santa Cruz Biotechnology) at a dilution of 1:5000. The blot was developed with SuperSignal West Pico chemiluminescent substrate (Thermo Scientific) and exposed to Kodak BioMax MR film (Kodak).
2.10. HBGA Binding Assay
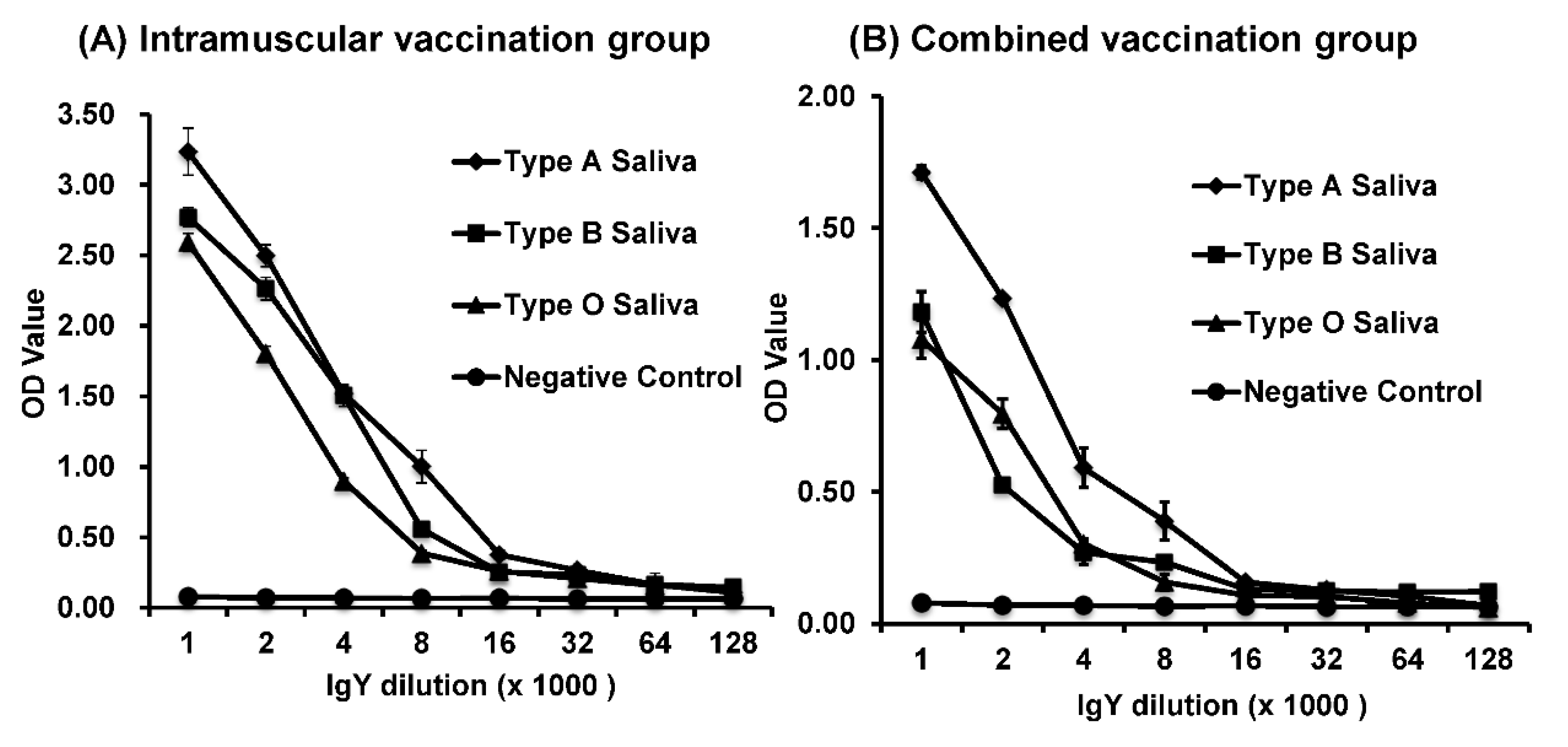
The saliva-based HBGA binding and blocking assays were performed as described previously [
18,
26,
27]. To avoid potential HuNoV-specific antibodies in the saliva that may interfere with the receptor-binding assay, saliva samples were boiled before being used in the assays. The boiled human saliva samples with known HBGA phenotypes (A, B, or O) were diluted 1000-fold and coated on 96-well microtiter plates at 4 °C overnight. After blocking with 5% nonfat milk in PBS, HuNoV VLPs were added to a final concentration of 4 μg/mL. The bound VLPs were detected by using serially diluted IgYs (from 1:1000 to 1:128,000), followed by the addition of HRP-conjugated goat anti-chicken IgY at a dilution of 1:5000. The color was then developed by adding tetramethylbenzidine peroxidase liquid substrate (Kirkegaard and Perry Laboratory) and stopped after 10 min of incubation at 22 °C by adding 1 mol/L sulfuric acid. Optical density (OD) was measured at 450 nm with the use of an Epoch Micro-Volume Spectrophotometer System (BioTek, Winooski, VT, USA).
2.11. HBGA Blocking Assay
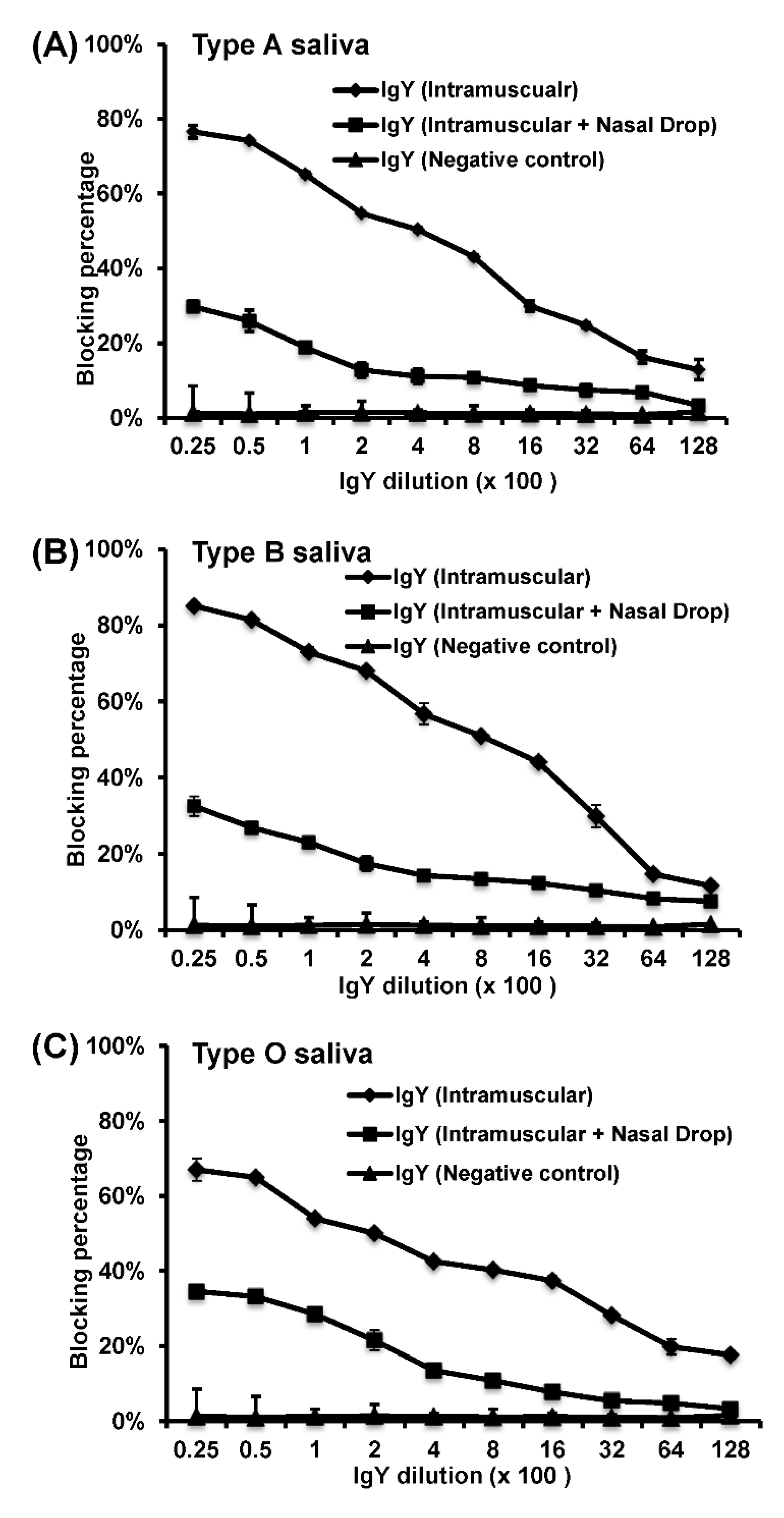
HBGA blocking assay was performed to determine the inhibitory activity of IgY against the binding of HuNoV VLPs to the HBGA antigens [
28]. The boiled human saliva samples with known HBGA phenotypes (A, B, or O) were diluted 1000-fold and coated on 96-well microtiter plates at 4 °C overnight. The HuNoV VLPs were preincubated with serially diluted IgYs for 1 h at 37 °C, and IgY-VLP solutions were added to the saliva-coated wells. Plates were washed 3 times with 0.1 mol/L sodium phosphate buffer (pH, 6.4). Then, a guinea pig anti-HuNoV VLP antiserum at a dilution of 1:1000 was added and incubated for 1 h at 4 °C. The plates were washed again, and HRP-conjugated goat anti-guinea pig IgG (at dilution of 1:5000) was added and incubated for 1 h at 4 °C. The color was then developed by adding tetramethylbenzidine peroxidase liquid substrate (Kirkegaard and Perry Laboratory) and stopped after 10 min of incubation at 22 °C by adding 1 mol/L sulfuric acid. The blocking rates were calculated by comparing the optical densities (ODs) measured with and without blocking by the chicken IgYs. The IgYs from chickens before immunization were used as controls [
26]. Blank wells were incubated with buffer instead of IgY-VLP and served as negative control whereas VLP binding to carbohydrates in the absence of IgY sample was used as a positive control.
2.12. The Effects of pH on the Stability of HuNoV-Specific IgY
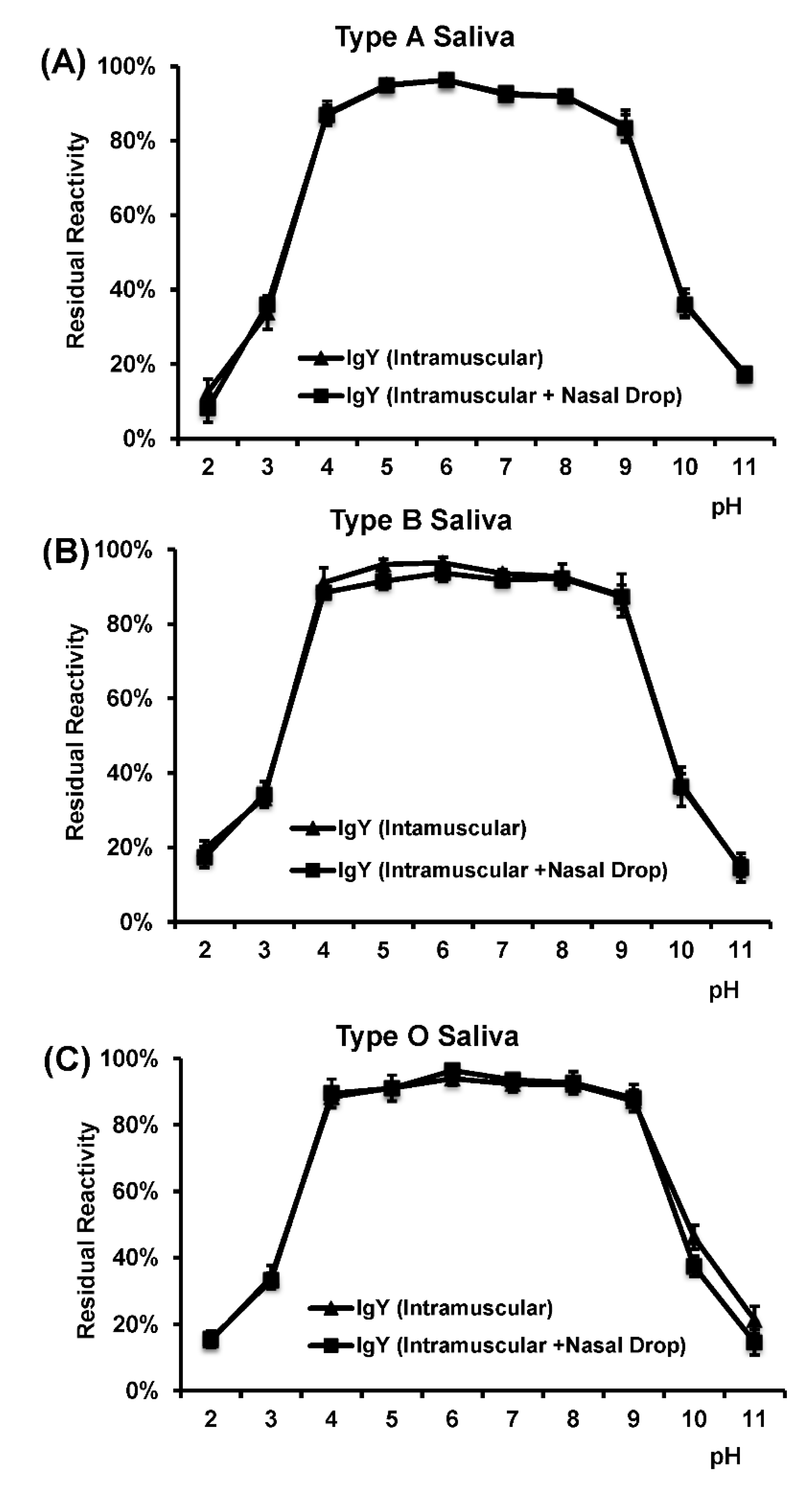
For the pH stability, the purified total IgY solution (1 mL, 1:100, pH 7.4) was diluted in 0.1 mol/L sodium phosphate buffer (pH 6.4). The pH of the solution was adjusted using either HCl or NaOH to a final pH ranging from 2 to 11. The solution was incubated at 37 °C for 3 h, followed by neutralization by adding 5 × PBS (pH 6.4) to a final pH of 7. The HBGA blocking assays were performed to measure the activity of IgY, as described above.
2.13. The Effects of Heat Treatment on the Stability of HuNoV-Specific IgY
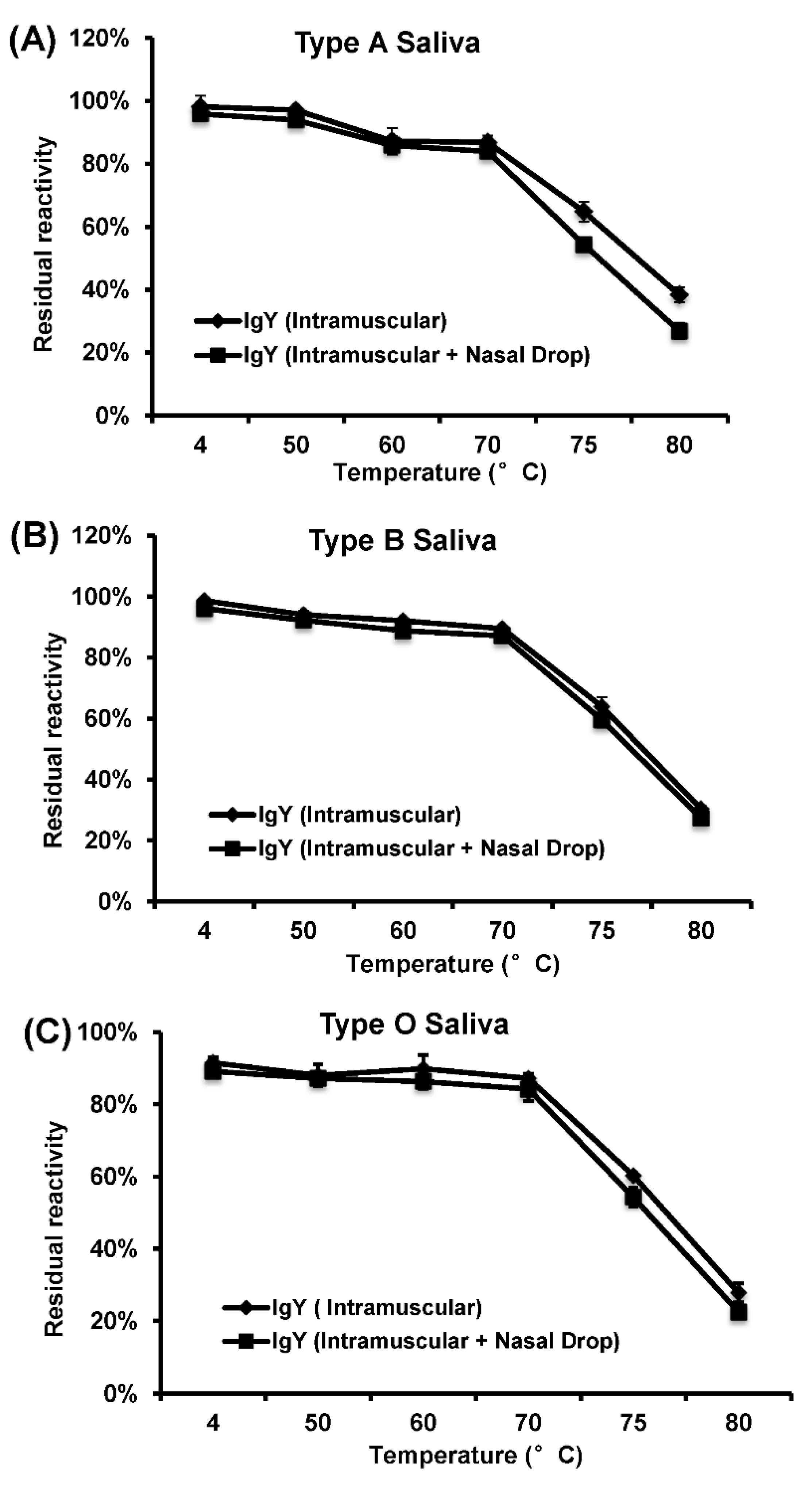
To determine heat stability of HuNoV-specific IgY, the purified total IgY solution (1 mL, 1:100, pH 7.4) was treated at temperature ranging from 4 to 80 °C for up to 30 min. After heat treatment, the samples were cooled quickly on ice. The HBGA blocking assays were performed to measure the activity of IgY as described above.
2.14. Statistical Analysis
Quantitative analysis was performed by either densitometric scanning of autoradiographs or by using a Typhoon PhosphorImager and ImageQuant TL software (GE Healthcare, Piscataway, NJ, USA). Each experiment was performed three to six times. Statistical analysis was performed by one-way multiple comparisons using SPSS software 13.0 (SPSS, Chicago, IL, USA). A p-value of <0.05 was considered statistically significant.
4. Discussion
In this study, we developed a highly efficient bioreactor to produce HuNoV-specific IgY in egg yolks by immunization of white leghorn chickens with recombinant rVSV-VP1. We found that intramuscular immunization alone was more efficient in triggering HuNoV-specific IgY compared to the combination of intramuscular and nasal drop immunization in hens. We demonstrated that IgY antibodies strongly reacted with HuNoV VLPs in ELISA, Western blot, and HBGA binding assays. The IgY antibodies were capable of blocking HuNoV-HBGA receptor interactions, and remained stable in temperature below 70 °C and at pH ranging from 4 to 9. Our results suggest that chicken egg yolk IgY antibodies (IgY) is a promising prophylactic and therapeutic agent for HuNoV.
Replication competent rVSV-VP1 is an excellent antigen for production of HuNoV-specific IgY in chickens. The natural hosts of VSV are cattle, horse, deer, and pig. However, VSV has a broad tissue tropism and can replicate to a high titer in many mammalian cell lines, insect cell, yeast, worm, and
C. elegans [
30,
31]. In this study, we found that rVSV-VP1 replicated to a high titer in DF-1 cells, a continuous cell line derived from chicken embryo fibroblasts. The virus yields in DF-1 cells were comparable to those produced in BSRT7 cell. In addition, rVSV-VP1 produced similar amounts of VP1 protein in both DF-1 and BSRT7 cells. Prior to our study, replication capability of VSV in avian species in vivo was poorly understood. The only report of VSV infection in chickens came from the study of rVSV-based influenza virus vaccine. It was found that chickens vaccinated with rVSV expressing the HA antigen of highly pathogenic avian influenza virus (H7N1) triggered a high level of serum neutralizing antibody and provided complete protection against lethal challenge of avian influenza H7N1 [
32]. In our study, we found that hens vaccinated with rVSV-VP1 triggered a high level of HuNoV-specific IgY in yolks, further supporting that chickens were susceptible to VSV infection. We also showed that immunization route affected IgY production in hens. We found that intramuscular vaccination triggered a higher HuNoV-specific IgY than combination of intramuscular and nasal drop vaccination although the total IgY levels were similar between two vaccination routes. Our rationale to include nasal drop vaccination is that it may trigger a higher mucosal immunity since egg yolk is developed in reproductive tract of chickens. In fact, our previous mice study showed that oral and intranasal vaccination of rVSV-VP1 trigged a high level of both serum and mucosal antibody [
23,
24].
One of the major concerns of rVSV-based vaccine is the safety, particularly since VSV is neurotropic. VSV infection in ruminant animal causes vesicular lesions in the mouth, teats, and feet. In mice, wild type VSV can cause acute brain infection and fatal encephalitis [
33]. However, insertion of HuNoV VP1 into VSV vector significantly attenuated the virus in a mouse model [
23,
24]. In this study, rVSV-VP1 did not cause any abnormal reactions, feed intake, or egg production of chickens, which further proved that rVSV-VP1 was attenuated in vivo.
Although two recent reports showed that HuNoV-specific IgY can be induced in hen vaccinated with purified VLPs or P particles from insect cells using a baculovirus expression system [
18,
22], our study represents the first report of using a live attenuated recombinant virus to generate HuNoV-specific IgY. There are many potential advantages of using live attenuated rVSV-VP1 for IgY production. First, rVSV-VP1 grows to a high titer in a wide range of cell lines including chicken cells. Second, replication of rVSV-VP1 in chickens resulted in synthesis of large amount of VLPs that in turn triggered a high level of HuNoV antibody. Third, it is an economical and time-saving approach. It does not require purification of VLPs or P particles. Thus, rVSV-VP1 is a promising vaccine antigen for large-scale production of IgY in chickens.
HuNoV-specific IgY is a potential passive immunization and therapeutic agent for HuNoV. HuNoV is a leading cause of viral gastroenteritis worldwide. Despite significant social, health, and economical burden it causes, no FDA-approved vaccine or therapeutic strategy is available. Epidemiology studies showed that HuNoV could cause lethal infection in humans, particularly in high-risk populations, such as infants, young children, the elderly, and immunocompromised individuals. Thus, there is a need to develop a safe and effective therapeutic strategy.
We found that HuNoV-specific IgYs isolated from egg yolks were biologically functional in vitro. First, HuNoV-specific IgYs can react with VLPs in ELISA and Western blot. Second, similar to serum IgG, HuNoV-specific IgYs can be used as a primary antibody in HBGA binding assay. Third, HuNoV-specific IgYs, but not the control IgY, were capable of blocking the binding of HuNoV VLPs to type A, B, and O HBGAs in human saliva. Although it is unknown whether HuNoV-specific IgY can directly neutralize the infectious HuNoV, blockage of virus–receptor interaction will likely block the infectivity of HuNoV, which will prevent HuNoV infection and illness. In 2010, Reeck et al. found a direct correlation between the ability of an antibody to block VLP-HBGA binding and protection against HuNoV infection and illness in a HuNoV human challenge study [
34]. In addition, Nurminen et al., (2011) showed that children could be protected from a GII.4 HuNoV infection due to the pre-existing HBGA blocking antibodies [
35]. Thus, the IgYs is a promising passive immunization approach to prevent and treat HuNoV infection and illness. Future study will determine whether IgY can directly neutralize infectious HuNoV using the recently developed B cell culture system [
8] or human enteroids culture system [
9].
The concept of IgY passive immunization has been developed in rotavirus in vivo animal models. It was found that passive immunization of IgY could protect neonatal calves from bovine rotavirus -induced diarrhea [
36]. In a mouse model, it was found that IgY could prevent murine rotavirus infection [
37], bovine rotavirus-induced diarrhea [
38], and human rotavirus-induced gastroenteritis [
39]. Recently, human rotavirus-specific IgY administered orally as a milk supplement passively protects neonatal pigs against an enteric human rotavirus infection [
40]. Porcine epidemic diarrhea virus (PEDV) is an enteric coronavirus which causes severe diarrhea, vomiting, and mortality in young piglets. Kweon et al. (2000) found that IgY passive immunization can protect piglets against PEDV infection [
41]. Future experiments will investigate whether HuNoV-specific IgY can protect gnotobiotic piglets from HuNoV-induced gastroenteritis and viral shedding.
Chicken as a “factory” for large-scale production of antigen-specific IgY. Antibody-based passive immunization and therapy has been shown to be an effective strategy to prevent infectious diseases in many animals [
16]. However, preparation of serum antibody from mammals is expensive and time-consuming. Thus, large-scale application of serum antibody has been limited. IgY egg yolk immunoglobulins derived from hyperimmunized hens represent an economically feasible and practical strategy which has been explored for the passive treatment of enteric diseases. Chicken IgY production is a much easier, faster, and cheaper method for polyclonal antibody production than from any other sources. It is easy to raise chickens and collect eggs without involvement of any stressful procedures (such as bleeding). White leghorn chickens are highly productive in laying eggs, and they can continuously produce eggs containing antigen-specific antibodies in their yolks for a long time period after immunization [
16]. Nguyen et al. (2010) demonstrated that chicken usually lays 280 eggs/year and each egg yolk normally contains 150–200 mg of IgY, which has 2 to 10% antigen-specific antibodies [
42]. In addition, extraction of antibody from egg yolk is simple and noninvasive without affecting the immunized chickens. Therefore, a chicken is an excellent “factory” for IgY antibody production.
In order to be used as immunological supplements in infant formulas and other foods, it is important to investigate the stability of IgY during storage or following processing methods, involving thermal treatments, such as pasteurization, sterilization, or spray-drying. Based on the HBGA blocking assay, HuNoV-specific IgY remains stable at temperature below 70 °C. Thus, it is safe for pasteurization (below 72 °C) of IgY for human consumption. However, the receptor blocking activity of IgY significantly decreased when temperature reached above 75 °C, suggesting that IgY may be denatured at this temperature. For oral administration, IgY should ideally be stable in acid or alkaline environment. Our results showed that the receptor blocking activity of IgY decreased at pH below 3 or above 10. Since the stomach pH is ~2–3, it may be necessary to encapsulate the IgY in acid resistant capsules so that it can be released in intestines for neutralizing the infectious virus particles. For example, Chang et al. (1999) demonstrated that addition of sugars, glycerol, or glycine to immunoglobulin solutions was effective to protect IgYs. In addition, film coating with gum arabic was proven to be effective in reducing the degree of hydrolysis of IgY [
43].
In summary, we have developed a highly efficient bioreactor to generate a high titer of HuNoV-specific IgY by vaccination of hens with rVSV-VP1. HuNoV-specific IgY was biologically active in capturing HuNoV antigen and blocking the interaction between VLPs and HBGAs. This study will facilitate the large-scale production and purification of HuNoV-specific IgY for virus detection, diagnosis, passive immunization, and therapy.