Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases
Abstract
1. Introduction
2. The Necessity of E. faecalis Phage Therapy
3. Strategies for Obtaining E. faecalis Phages for Phage Therapy
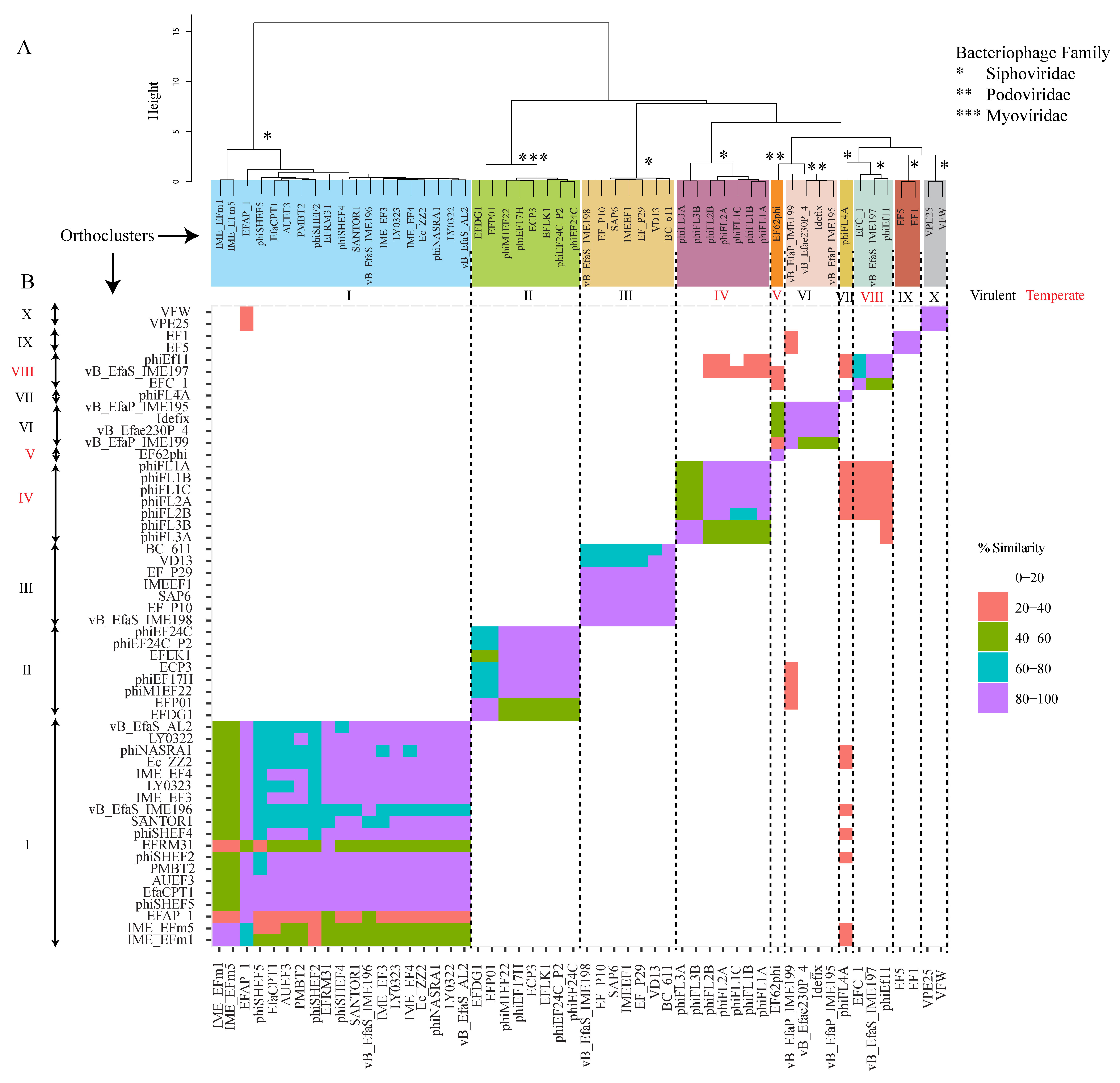
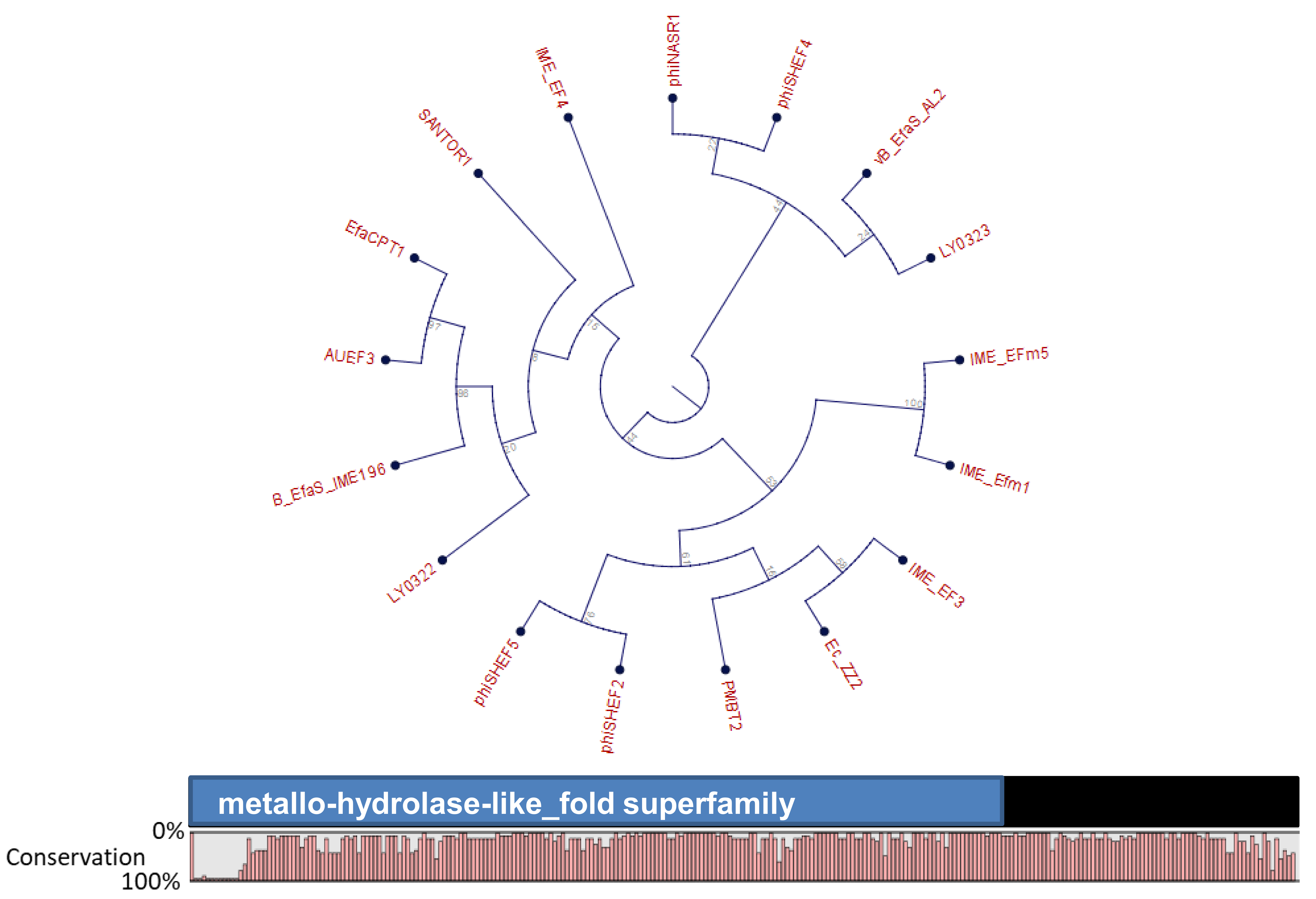
4. Orthocluster Analysis of E. faecalis Phages
5. E. faecalis Phage Therapy in In Vitro Models
5.1. Biofilm Eradication
5.2. Human Root Canal Model (In Vitro/Ex Vivo)
5.3. Fibrin Clot Model
5.4. E. faecalis Phages as Biocontrol Agents
6. E. faecalis Phage Therapy in In Vivo Models
6.1. Vertebrate Models
6.2. Invertebrate Models
7. E. faecalis Phage Endolysins as Viable Alternatives for Phage Therapy
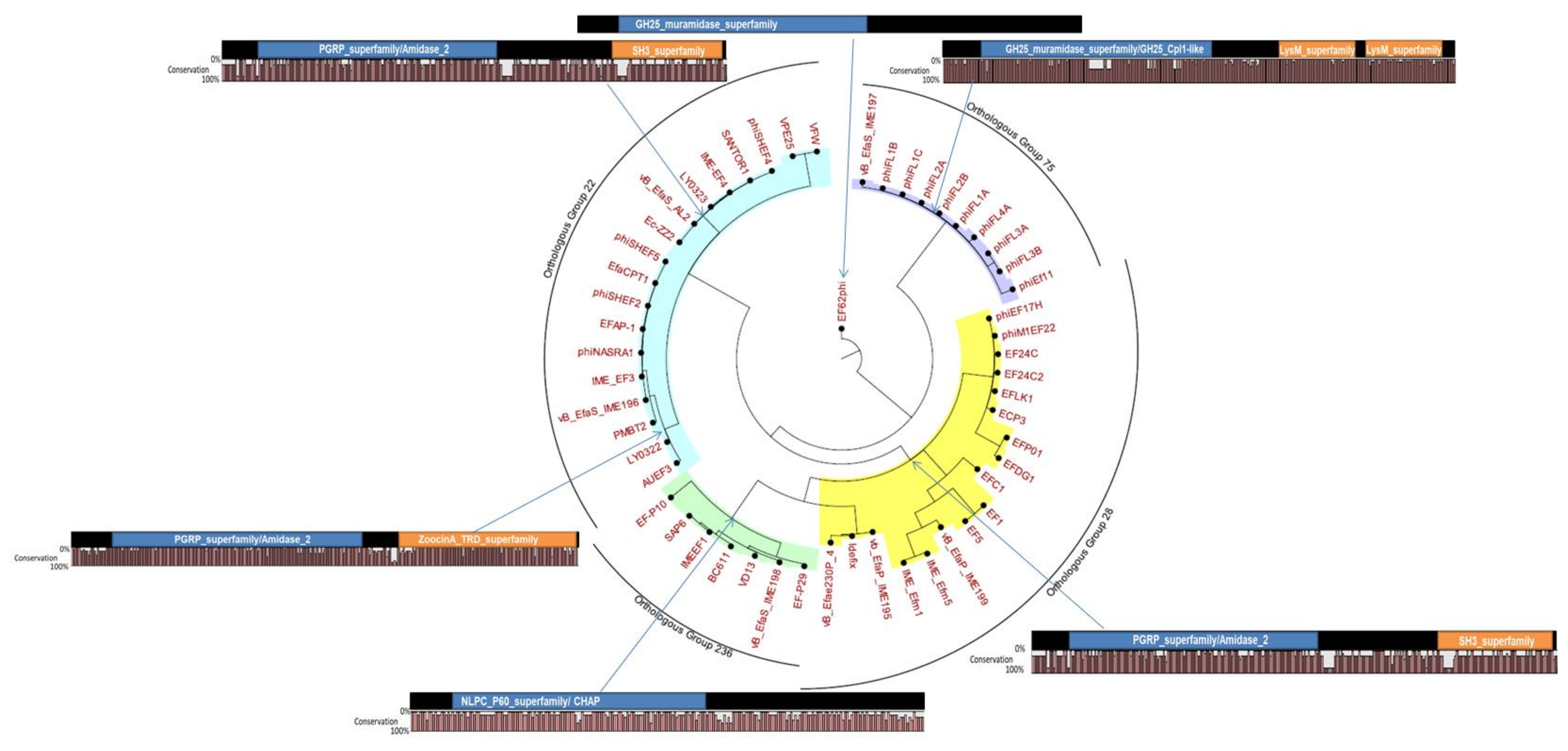
7.1. Domain Architecture of E. faecalis Phage Endolysins
7.2. Applications of E. faecalis Phage Endolysins
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Arias, C.A.; Contreras, G.A.; Murray, B.E. Management of multidrug-resistant enterococcal infections. Clin. Microbiol. Infect. 2010, 16, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Sava, I.G.; Heikens, E.; Huebner, J. Pathogenesis and immunity in enterococcal infections. Clin. Microbiol. Infect. 2010, 16, 533–540. [Google Scholar] [CrossRef]
- Berg, R.D. The indigenous gastrointestinal microflora. Trends Microbiol. 1996, 4, 430–435. [Google Scholar] [CrossRef]
- Facklam, R.R.; da Carvalho, M.G.S.; Teixeira, L.M. History, Taxonomy, Biochemical Characteristics, and Antibiotic Susceptibility Testing of Enterococci. In The Enterococci; American Society of Microbiology: Washington, DC, USA, 2002; pp. 1–54. [Google Scholar]
- Simonsen, G.S.; Småbrekke, L.; Monnet, D.L.; Sørensen, T.L.; Møller, J.K.; Kristinsson, K.G.; Lagerqvist-Widh, A.; Torell, E.; Digranes, A.; Harthug, S.; et al. Prevalence of resistance to ampicillin, gentamicin and vancomycin in Enterococcus faecalis and Enterococcus faecium isolates from clinical specimens and use of antimicrobials in five Nordic hospitals. J. Antimicrob. Chemother. 2003, 51, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Hanchi, H.; Mottawea, W.; Sebei, K.; Hammami, R. The Genus Enterococcus: Between Probiotic Potential and Safety Concerns—An Update. Front. Microbiol. 2018, 9, 1791. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J. AMR Review Paper-Tackling a Crisis for the Health and Wealth of Nations. Available online: http://www.jpiamr.eu/wp-content/uploads/2014/12/AMR-Review-Paper-Tackling-a-crisis-for-the-health-and-wealth-of-nations_1-2.pdf (accessed on 19 April 2019).
- Tagliabue, A.; Rappuoli, R. Changing Priorities in Vaccinology: Antibiotic Resistance Moving to the Top. Front. Immunol. 2018, 9, 1068. [Google Scholar] [CrossRef] [PubMed]
- Bolocan, A.S.; Callanan, J.; Forde, A.; Ross, P.; Hill, C. Phage therapy targeting Escherichia coli-a story with no end? FEMS Microbiol. Lett. 2016, 363. [Google Scholar] [CrossRef] [PubMed]
- Kortright, K.E.; Chan, B.K.; Koff, J.L.; Turner, P.E. Phage Therapy: A Renewed Approach to Combat Antibiotic-Resistant Bacteria. Cell Host Microbe 2019, 25, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Brüssow, H. Phage therapy: The Escherichia coli experience. Microbiology 2005, 151, 2133–2140. [Google Scholar] [CrossRef]
- Pires, D.P.; Cleto, S.; Sillankorva, S.; Azeredo, J.; Lu, T.K. Genetically Engineered Phages: A Review of Advances over the Last Decade. Microbiol. Mol. Biol. Rev. 2016, 80, 523–543. [Google Scholar]
- Moelling, K.; Broecker, F.; Willy, C. A Wake-Up Call: We Need Phage Therapy Now. Viruses 2018, 10, 688. [Google Scholar] [CrossRef] [PubMed]
- Patey, O.; McCallin, S.; Mazure, H.; Liddle, M.; Smithyman, A.; Dublanchet, A. Clinical Indications and Compassionate Use of Phage Therapy: Personal Experience and Literature Review with a Focus on Osteoarticular Infections. Viruses 2018, 11, 18. [Google Scholar] [CrossRef] [PubMed]
- Gong, P.; Cheng, M.; Li, X.; Jiang, H.; Yu, C.; Kahaer, N.; Li, J.; Zhang, L.; Xia, F.; Hu, L.; et al. Characterization of Enterococcus faecium bacteriophage IME-EFm5 and its endolysin LysEFm5. Virology 2016, 492, 11–20. [Google Scholar] [CrossRef]
- Khalifa, L.; Gelman, D.; Shlezinger, M.; Dessal, A.L.; Coppenhagen-Glazer, S.; Beyth, N.; Hazan, R. Defeating Antibiotic- and Phage-Resistant Enterococcus faecalis Using a Phage Cocktail in Vitro and in a Clot Model. Front. Microbiol. 2018, 9, 326. [Google Scholar] [CrossRef]
- Lossouarn, J.; Briet, A.; Moncaut, E.; Furlan, S.; Bouteau, A.; Son, O.; Leroy, M.; DuBow, M.; Lecointe, F.; Serror, P.; et al. Enterococcus faecalis Countermeasures Defeat a Virulent Picovirinae Bacteriophage. Viruses 2019, 11, 48. [Google Scholar] [CrossRef] [PubMed]
- Khalifa, L.; Brosh, Y.; Gelman, D.; Coppenhagen-Glazer, S.; Beyth, S.; Poradosu-Cohen, R.; Que, Y.A.; Beyth, N.; Hazan, R. Targeting Enterococcus faecalis biofilms with phage therapy. Appl. Environ. Microbiol. 2015, 81, 2696–2705. [Google Scholar] [CrossRef] [PubMed]
- Fanaro, S.; Chierici, R.; Guerrini, P.; Vigi, V. Intestinal microflora in early infancy: Composition and development. Acta Paediatr. 2007, 92, 48–55. [Google Scholar] [CrossRef]
- Huycke, M.M.; Abrams, V.; Moore, D.R. Enterococcus faecalis produces extracellular superoxide and hydrogen peroxide that damages colonic epithelial cell DNA. Carcinogenesis 2002, 23, 529–536. [Google Scholar] [CrossRef]
- Bhatty, M.; Cruz, M.R.; Frank, K.L.; Laverde Gomez, J.A.; Andrade, F.; Garsin, D.A.; Dunny, G.M.; Kaplan, H.B.; Christie, P.J. Enterococcus faecalis pCF10-encoded surface proteins PrgA, PrgB (aggregation substance) and PrgC contribute to plasmid transfer, biofilm formation and virulence. Mol. Microbiol. 2015, 95, 660–677. [Google Scholar] [CrossRef] [PubMed]
- Kayaoglu, G.; Ørstavik, D. Virulence factors of Enterococcus faecalis: Relationship to endodontic disease. Crit. Rev. Oral Biol. Med. 2004, 15, 308–320. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, P.; Ravikumar, K.; Umapathy, B. Review of virulence factors of enterococcus: An emerging nosocomial pathogen. Indian J. Med. Microbiol. 2009, 27, 301. [Google Scholar] [CrossRef]
- Singh, K.V.; Nallapareddy, S.R.; Murray, B.E. Importance of the ebp (Endocarditis- and Biofilm-Associated Pilus) Locus in the Pathogenesis of Enterococcus faecalis Ascending Urinary Tract Infection. J. Infect. Dis. 2007, 195, 1671–1677. [Google Scholar] [CrossRef]
- Singh, K.V.; Nallapareddy, S.R.; Sillanpää, J.; Murray, B.E. Importance of the Collagen Adhesin Ace in Pathogenesis and Protection against Enterococcus faecalis Experimental Endocarditis. PLoS Pathol. 2010, 6, e1000716. [Google Scholar] [CrossRef]
- Toledo-Arana, A.; Valle, J.; Solano, C.; Arrizubieta, M.J.; Cucarella, C.; Lamata, M.; Amorena, B.; Leiva, J.; Penadés, J.R.; Lasa, I. The enterococcal surface protein, Esp, is involved in Enterococcus faecalis biofilm formation. Appl. Environ. Microbiol. 2001, 67, 4538–4545. [Google Scholar] [CrossRef] [PubMed]
- Nallapareddy, S.R.; Singh, K.V.; Duh, R.W.; Weinstock, G.M.; Murray, B.E. Diversity of ace, a gene encoding a microbial surface component recognizing adhesive matrix molecules, from different strains of Enterococcus faecalis and evidence for production of ace during human infections. Infect. Immun. 2000, 68, 5210–5217. [Google Scholar] [CrossRef] [PubMed]
- Montealegre, M.C.; La Rosa, S.L.; Roh, J.H.; Harvey, B.R.; Murray, B.E. The Enterococcus faecalis EbpA Pilus Protein: Attenuation of Expression, Biofilm Formation, and Adherence to Fibrinogen Start with the Rare Initiation Codon ATT. MBio 2015, 6, e00467-15. [Google Scholar] [CrossRef] [PubMed]
- Hubble, T.S.; Hatton, J.F.; Nallapareddy, S.R.; Murray, B.E.; Gillespie, M.J. Influence of Enterococcus faecalis proteases and the collagen-binding protein, Ace, on adhesion to dentin. Oral Microbiol. Immunol. 2003, 18, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.V.; La Rosa, S.L.; Somarajan, S.R.; Roh, J.H.; Murray, B.E. The fibronectin-binding protein EfbA contributes to pathogenesis and protects against infective endocarditis caused by Enterococcus faecalis. Infect. Immun. 2015, 83, 4487–4494. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nallapareddy, S.R.; Qin, X.; Weinstock, G.M.; Höök, M.; Murray, B.E. Enterococcus faecalis adhesin, ace, mediates attachment to extracellular matrix proteins collagen type IV and laminin as well as collagen type I. Infect. Immun. 2000, 68, 5218–5224. [Google Scholar] [CrossRef]
- Kristich, C.J.; Rice, L.B.; Arias, C.A. Enterococcal Infection—Treatment and Antibiotic Resistance; Massachusetts Eye and Ear Infirmary: Boston, MA, USA, 2014. [Google Scholar]
- Beganovic, M.; Luther, M.K.; Rice, L.B.; Arias, C.A.; Rybak, M.J.; Laplante, K.L. A Review of Combination Antimicrobial Therapy for Enterococcus Faecalis Bloodstream Infections and Infective Endocarditis Citation/Publisher Attribution. Clin. Infect. Dis. 2018, 2, 303–309. [Google Scholar] [CrossRef]
- Matar, M.J.; Safdar, A.; Rolston, K.V.I. Relationship of colonization with vancomycin-resistant enterococci and risk of systemic infection in patients with cancer. Clin. Infect. Dis. 2006, 42, 1506–1507. [Google Scholar] [CrossRef] [PubMed]
- Gedik, H.; Şimşek, F.; Kantürk, A.; Yıldırmak, T.; Arıca, D.; Aydın, D.; Yokuş, O.; Demirel, N. Vancomycin-resistant enterococci colonization in patients with hematological malignancies: Screening and its cost-effectiveness. Afr. Health Sci. 2014, 14, 899. [Google Scholar] [PubMed]
- Hicks, K.L.; Breto, L.; Halbur, L. Vancomycin Resistant Enterococcus as a Leading Cause of Early Infection-Related Mortality in Older (≥60 Years) AML Patients Admitted to a Community Hospital for Standard Induction Chemotherapy; American Society of Hematology: Washington, DC, USA, 2006. [Google Scholar]
- Messina, J.A.; Sung, A.D.; Chao, N.J.; Alexander, B.D. The Timing and Epidemiology of Enterococcus Faecium and E. Faecalis Bloodstream Infections (BSI) in Patients with Acute Leukemia Receiving Chemotherapy; American Society of Hematology: Washington, DC, USA, 2017. [Google Scholar]
- De Almeida, C.V.; Taddei, A.; Amedei, A. The controversial role of Enterococcus faecalis in colorectal cancer. Ther. Adv. Gastroenterol. 2018, 11, 175628481878360. [Google Scholar] [CrossRef] [PubMed]
- Koehler, P.; Jung, N.; Cornely, O.A.; Rybniker, J.; Fätkenheuer, G. Combination Antimicrobial Therapy for Enterococcus faecalis Infective Endocarditis. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef] [PubMed]
- Abdelkader, K.; Gerstmans, H.; Saafan, A.; Dishisha, T.; Briers, Y. The Preclinical and Clinical Progress of Bacteriophages and Their Lytic Enzymes: The Parts are Easier than the Whole. Viruses 2019, 11, 96. [Google Scholar] [CrossRef]
- Elbreki, M.; Ross, R.P.; Hill, C.; O’Mahony, J.; McAuliffe, O.; Coffey, A. Bacteriophages and Their Derivatives as Biotherapeutic Agents in Disease Prevention and Treatment. J. Viruses 2014, 2014, 1–20. [Google Scholar] [CrossRef]
- Purnell, S.E.; Ebdon, J.E.; Taylor, H.D. Bacteriophage lysis of enterococcus host strains: A tool for microbial source tracking? Environ. Sci. Technol. 2011, 45, 10699–10705. [Google Scholar] [CrossRef] [PubMed]
- Yasmin, A.; Kenny, J.G.; Shankar, J.; Darby, A.C.; Hall, N.; Edwards, C.; Horsburgh, M.J. Comparative genomics and transduction potential of Enterococcus faecalis temperate bacteriophages. J. Bacteriol. 2010, 192, 1122–1130. [Google Scholar] [CrossRef]
- Uchiyama, J.; Rashel, M.; Maeda, Y.; Takemura, I.; Sugihara, S.; Akechi, K.; Muraoka, A.; Wakiguchi, H.; Matsuzaki, S. Isolation and characterization of a novel Enterococcus faecalis bacteriophage φEF24C as a therapeutic candidate. FEMS Microbiol. Lett. 2008, 278, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Santiago-Rodriguez, T.M.; De vila, C.; Gonzalez, J.; Bonilla, N.; Marcos, P.; Urdaneta, M.; Cadete, M.; Monteiro, S.; Santos, R.; Domingo, J.S.; et al. Characterization of Enterococcus faecalis-infecting phages (enterophages) as markers of human fecal pollution in recreational waters. Water Res. 2010, 44, 4716–4725. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, N.; Santiago, T.; Marcos, P.; Urdaneta, M.; Santo Domingo, J.; Toranzos, G.A. Enterophages, a group of phages infecting Enterococcus faecalis, and their potential as alternate indicators of human faecal contamination. Water Sci. Technol. 2010, 61, 293–300. [Google Scholar] [CrossRef] [PubMed]
- Mullan, M. Factors Affecting Plaque Formation by Bacteriophages. Dairy Sci. 2002, 5, 1–12. [Google Scholar]
- Örmälä, A.-M.; Jalasvuori, M. Phage therapy. Bacteriophage 2013, 3, e24219. [Google Scholar] [CrossRef]
- Rostøl, J.T.; Marraffini, L. (Ph)ighting Phages: How Bacteria Resist Their Parasites. Cell Host Microbe 2019, 25, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Duerkop, B.A.; Palmer, K.L.; Horsburgh, M.J. Enterococcal Bacteriophages and Genome Defense. In Enterococci: From Commensals to Leading Causes of Drug Resistant Infection; Eye and Ear Infirmary: Boston, MA, USA, 2014. [Google Scholar]
- Swift, S.M.; Rowley, D.T.; Young, C.; Franks, A.; Hyman, P.; Donovan, D.M. The endolysin from the Enterococcus faecalis bacteriophage VD13 and conditions stimulating its lytic activity. FEMS Microbiol. Lett. 2016, 363, fnw216. [Google Scholar] [CrossRef]
- Zhang, W.; Mi, Z.; Yin, X.; Fan, H.; An, X.; Zhang, Z.; Chen, J.; Tong, Y. Characterization of Enterococcus faecalis phage IME-EF1 and its endolysin. PLoS ONE 2013, 8, e80435. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, W.; Lv, Y.; Zheng, W.; Mi, Z.; Pei, G.; An, X.; Xu, X.; Han, C.; Liu, J.; et al. Characterization and complete genome sequence analysis of novel bacteriophage IME-EFm1 infecting Enterococcus faecium. J. Gen. Virol. 2014, 95, 2565–2575. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Xing, S.; Zhao, F.; Li, P.; Mi, Z.; Shi, T.; Liu, H.; Tong, Y. Characterization and genome analysis of novel phage vB_EfaP_IME195 infecting Enterococcus faecalis. Virus Genes 2018, 54, 804–811. [Google Scholar] [CrossRef]
- Hyatt, D.; Chen, G.-L.; LoCascio, P.F.; Land, M.L.; Larimer, F.W.; Hauser, L.J. Prodigal: Prokaryotic gene recognition and translation initiation site identification. BMC Bioinform. 2010, 11, 119. [Google Scholar] [CrossRef]
- Li, L.; Stoeckert, C.J.; Roos, D.S.; Roos, D.S. OrthoMCL: Identification of ortholog groups for eukaryotic genomes. Genome Res. 2003, 13, 2178–2189. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ding, P.; Han, C.; Fan, H.; Wang, Y.; Mi, Z.; Feng, F.; Tong, Y. Genome analysis of Enterococcus faecalis bacteriophage IME-EF3 harboring a putative metallo-beta-lactamase gene. Virus Genes 2014, 49, 145–151. [Google Scholar] [CrossRef]
- Son, J.S.; Jun, S.Y.; Kim, E.B.; Park, J.E.; Paik, H.R.; Yoon, S.J.; Kang, S.H.; Choi, Y.-J. Complete genome sequence of a newly isolated lytic bacteriophage, EFAP-1 of Enterococcus faecalis, and antibacterial activity of its endolysin EFAL-1. J. Appl. Microbiol. 2010, 108, 1769–1779. [Google Scholar] [CrossRef] [PubMed]
- Mazaheri Nezhad Fard, R.; Barton, M.D.; Heuzenroeder, M.W. Bacteriophage-mediated transduction of antibiotic resistance in enterococci. Lett. Appl. Microbiol. 2011, 52, 559–564. [Google Scholar] [CrossRef]
- Gelman, D.; Beyth, S.; Lerer, V.; Adler, K.; Poradosu-Cohen, R.; Coppenhagen-Glazer, S.; Hazan, R. Combined bacteriophages and antibiotics as an efficient therapy against VRE Enterococcus faecalis in a mouse model. Res. Microbiol. 2018, 169, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-D.; Park, J.-H. Complete genome sequence of enterococcal bacteriophage SAP6. J. Virol. 2012, 86, 5402–5403. [Google Scholar] [CrossRef]
- São-José, C.; Proença, D.; Leandro, C.; Fernandes, S.; Pimentel, M.; Mato, R.; Lopes, F.; Santos, S.; Cavaco-Silva, P.; Silva, F.A. Phage Endolysins with Broad Antimicrobial Activity Against Enterococcus faecalis Clinical Strains. Microb. Drug Resist. 2012, 18, 322–332. [Google Scholar]
- Brede, D.A.; Snipen, L.G.; Ussery, D.W.; Nederbragt, A.J.; Nes, I.F. Complete genome sequence of the commensal Enterococcus faecalis 62, isolated from a healthy Norwegian infant. J. Bacteriol. 2011, 193, 2377–2378. [Google Scholar] [CrossRef] [PubMed]
- Xing, S.; Zhang, X.; Sun, Q.; Wang, J.; Mi, Z.; Pei, G.; Huang, Y.; An, X.; Fu, K.; Zhou, L.; et al. Complete genome sequence of a novel, virulent Ahjdlikevirus bacteriophage that infects Enterococcus faecium. Arch. Virol. 2017, 162, 3843–3847. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.H.; Porras, O.D.; Delisle, A.L. Bacteriophages induced from lysogenic root canal isolates of Enterococcus faecalis. Oral Microbiol. Immunol. 2009, 24, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Yoon, B.H.; Chang, H.-I. Genomic annotation for the temperate phage EFC-1, isolated from Enterococcus faecalis KBL101. Arch. Virol. 2015, 160, 601–604. [Google Scholar] [CrossRef]
- Zhang, H.; Fouts, D.E.; DePew, J.; Stevens, R.H. Genetic modifications to temperate Enterococcus faecalis phage Ef11 that abolish the establishment of lysogeny and sensitivity to repressor, and increase host range and productivity of lytic infection. Microbiology 2013, 159, 1023–1035. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.H.; Zhang, H.; Hsiao, C.; Kachlany, S.; Tinoco, E.M.B.; Depew, J.; Fouts, D.E.; Roy, F.; Stevens, H. Structural proteins of Enterococcus faecalis bacteriophage φEf11. Bacteriophage 2016, 6, e1251381. [Google Scholar] [CrossRef] [PubMed]
- Tinoco, J.M.; Buttaro, B.; Zhang, H.; Liss, N.; Sassone, L.; Stevens, R. Effect of a genetically engineered bacteriophage on Enterococcus faecalis biofilms. Arch. Oral Biol. 2016, 71, 80–86. [Google Scholar] [CrossRef]
- Duerkop, C.A.; Huo, B.A.; Bhardwaj, W.; Palmer, P.L.; Hooper, K.L. Molecular basis for lytic bacteriophage resistance in enterococci. MBio 2016, 7, 1304–1320. [Google Scholar] [CrossRef] [PubMed]
- Dalmasso, M.; de Haas, E.; Neve, H.; Strain, R.; Cousin, F.J.; Stockdale, S.R.; Ross, R.P.; Hill, C. Isolation of a Novel Phage with Activity against Streptococcus mutans Biofilms. PLoS ONE 2015, 10, e0138651. [Google Scholar] [CrossRef]
- Nazareth, N.; Magro, F.; Machado, E.; Ribeiro, T.G.; Martinho, A.; Rodrigues, P.; Alves, R.; Macedo, G.N.; Gracio, D.; Coelho, R.; et al. Prevalence of Mycobacterium avium subsp. paratuberculosis and Escherichia coli in blood samples from patients with inflammatory bowel disease. Med. Microbiol. Immunol. 2015, 204, 681–692. [Google Scholar] [CrossRef]
- Fong, S.A.; Drilling, A.; Morales, S.; Cornet, M.E.; Woodworth, B.A.; Fokkens, W.J.; Psaltis, A.J.; Vreugde, S.; Wormald, P.-J. Activity of Bacteriophages in Removing Biofilms of Pseudomonas aeruginosa Isolates from Chronic Rhinosinusitis Patients. Front. Cell. Infect. Microbiol. 2017, 7, 418. [Google Scholar] [CrossRef]
- Abdulamir, A.S.; Jassim, S.A.A.; Hafidh, R.R.; Bakar, F.A. The potential of bacteriophage cocktail in eliminating Methicillin-resistant Staphylococcus aureus biofilms in terms of different extracellular matrices expressed by PIA, ciaA-D and FnBPA genes. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 49. [Google Scholar] [CrossRef]
- Khalifa, L.; Shlezinger, M.; Beyth, S.; Houri-Haddad, Y.; Coppenhagen-Glazer, S.; Beyth, N.; Hazan, R. Phage therapy against Enterococcus faecalis in dental root canals. J. Oral Microbiol. 2016, 8, 32157. [Google Scholar] [CrossRef] [PubMed]
- Szafrański, S.P.; Winkel, A.; Stiesch, M. The use of bacteriophages to biocontrol oral biofilms. J. Biotechnol. 2017, 250, 29–44. [Google Scholar] [CrossRef] [PubMed]
- Sutherland, I.W.; Hughes, K.A.; Skillman, L.C.; Tait, K. The interaction of phage and biofilms. FEMS Microbiol. Lett. 2004, 232, 1–6. [Google Scholar] [CrossRef]
- Stuart, C.H.; Schwartz, S.A.; Beeson, T.J.; Owatz, C.B. Enterococcus faecalis: Its role in root canal treatment failure and current concepts in retreatment. J. Endod. 2006, 32, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.Q.; Zhang, C.F.; Chu, C.H.; Zhu, X.F. Prevalence of Enterococcus faecalis in saliva and filled root canals of teeth associated with apical periodontitis. Int. J. Oral Sci. 2012, 4, 19–23. [Google Scholar] [CrossRef]
- Sundqvist, G.; Figdor, D.; Persson, S.; Sjögren, U. Microbiologic analysis of teeth with failed endodontic treatment and the outcome of conservative re-treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1998, 85, 86–93. [Google Scholar] [CrossRef]
- Paisano, A.F.; Spira, B.; Cai, S.; Bombana, A.C. In vitro antimicrobial effect of bacteriophages on human dentin infected with Enterococcus faecalis ATCC 29212. Oral Microbiol. Immunol. 2004, 19, 327–330. [Google Scholar] [CrossRef] [PubMed]
- Furie, B.; Furie, B.C. The molecular basis of blood coagulation. Cell 1988, 53, 505–518. [Google Scholar] [CrossRef]
- Hershberger, E.; Coyle, E.A.; Kaatz, G.W.; Zervos, M.J.; Rybak, M.J. Comparison of a Rabbit Model of Bacterial Endocarditis and an In Vitro Infection Model with Simulated Endocardial Vegetations. Antimicrob. Agents Chemother. 2000, 44, 1921–1924. [Google Scholar] [CrossRef]
- Kastrup, C.J.; Boedicker, J.Q.; Pomerantsev, A.P.; Moayeri, M.; Bian, Y.; Pompano, R.R.; Kline, T.R.; Sylvestre, P.; Shen, F.; Leppla, S.H.; et al. Spatial localization of bacteria controls coagulation of human blood by “quorum acting”. Nat. Chem. Biol. 2008, 4, 742–750. [Google Scholar] [CrossRef]
- McGrath, B.J.; Kang, S.L.; Kaatz, G.W.; Rybak, M.J. Bactericidal activities of teicoplanin, vancomycin, and gentamicin alone and in combination against Staphylococcus aureus in an in vitro pharmacodynamic model of endocarditis. Antimicrob. Agents Chemother. 1994, 38, 2034–2040. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Houlihan, H.H.; Stokes, D.P.; Rybak, M.J. Pharmacodynamics of vancomycin and ampicillin alone and in combination with gentamicin once daily or thrice daily against Enterococcus faecalis in an in vitro infection model. J. Antimicrob. Chemother. 2000, 46, 79–86. [Google Scholar] [CrossRef][Green Version]
- Ladero, V.; Gómez-Sordo, C.; Sánchez-Llana, E.; del Rio, B.; Redruello, B.; Fernández, M.; Martín, M.C.; Alvarez, M.A. Q69 (an E. faecalis-Infecting Bacteriophage) As a Biocontrol Agent for Reducing Tyramine in Dairy Products. Front. Microbiol. 2016, 7, 445. [Google Scholar] [CrossRef]
- Letkiewicz, S.; Międzybrodzki, R.; Fortuna, W.; Weber-Dąbrowska, B.; Górski, A. Eradication of Enterococcus faecalis by phage therapy in chronic bacterial prostatitis—Case report. Folia Microbiol. 2009, 54, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, J.; Rashel, M.; Takemura, I.; Wakiguchi, H.; Matsuzaki, S. In silico and in vivo evaluation of bacteriophage φEF24C, a candidate for treatment of Enterococcus faecalis infections. Appl. Environ. Microbiol. 2008, 74, 4149–4163. [Google Scholar] [CrossRef]
- Biswas, B.; Adhya, S.; Washart, P.; Paul, B.; Trostel, A.N.; Powell, B.; Carlton, R.; Merril, R.; Merril, C.R. Bacteriophage Therapy Rescues Mice Bacteremic from a Clinical Isolate of Vancomycin-Resistant Enterococcus Bacteriophage Therapy Rescues Mice Bacteremic from a Clinical Isolate of Vancomycin-Resistant Enterococcus faecium. Infect. Immun. 2002, 70, 204–210. [Google Scholar] [CrossRef]
- Cheng, M.; Liang, J.; Zhang, Y.; Hu, L.; Gong, P.; Cai, R.; Zhang, L.; Zhang, H.; Ge, J.; Ji, Y. The bacteriophage EF-P29 efficiently protects against lethal vancomycin-resistant Enterococcus faecalis and alleviates gut microbiota imbalance in a murine bacteremia model. Front. Microbiol. 2017, 8, 837. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, Y.; Li, X.; Liang, J.; Hu, L.; Gong, P.; Zhang, L.; Cai, R.; Zhang, H.; Ge, J.; et al. Endolysin LysEF-P10 shows potential as an alternative treatment strategy for multidrug-resistant Enterococcus faecalis infections. Sci. Rep. 2017, 7, 10164. [Google Scholar] [CrossRef]
- Weber-Dąbrowska, B.; Mulczyk, M.; Górski, A. Bacteriophage Therapy of Bacterial Infections: An Update of our Institute’s Experience. In Inflammation; Springer: Dordrecht, The Netherlands, 2001; pp. 201–209. [Google Scholar]
- Schooley, R.T.; Biswas, B.; Gill, J.J.; Hernandez-Morales, A.; Lancaster, J.; Lessor, L.; Barr, J.J.; Reed, S.L.; Rohwer, F.; Benler, S.; et al. Development and Use of Personalized Bacteriophage-Based Therapeutic Cocktails to Treat a Patient with a Disseminated Resistant Acinetobacter baumannii Infection. Antimicrob. Agents Chemother. 2017, 61, e00954-17. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, J.; Matsui, H.; Murakami, H.; Kato, S.; Watanabe, N.; Nasukawa, T.; Mizukami, K.; Ogata, M.; Sakaguchi, M.; Matsuzaki, S.; et al. Potential Application of Bacteriophages in Enrichment Culture for Improved Prenatal Streptococcus agalactiae Screening. Viruses 2018, 10, 552. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, K.; Fallon, J.P. Galleria mellonella larvae as models for studying fungal virulence. Fungal Biol. Rev. 2010, 24, 79–83. [Google Scholar] [CrossRef]
- Mukherjee, K.; Altincicek, B.; Hain, T.; Domann, E.; Vilcinskas, A.; Chakraborty, T. Galleria mellonella as a model system for studying Listeria pathogenesis. Appl. Environ. Microbiol. 2010, 76, 310–317. [Google Scholar] [CrossRef]
- Ramarao, N.; Nielsen-Leroux, C.; Lereclus, D. The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J. Vis. Exp. 2012, 70, e4392. [Google Scholar] [CrossRef] [PubMed]
- Kamal, F.; Dennis, J.J. Burkholderia cepacia complex phage-antibiotic synergy (PAS): Antibiotics stimulate lytic phage activity. Appl. Environ. Microbiol. 2015, 81, 1132–1138. [Google Scholar] [CrossRef] [PubMed]
- Manohar, P.; Nachimuthu, R.; Lopes, B.S. The therapeutic potential of bacteriophages targeting gram-negative bacteria using Galleria mellonella infection model. BMC Microbiol. 2018, 18, 97. [Google Scholar] [CrossRef] [PubMed]
- Goh, H.M.S.; Yong, M.H.A.; Chong, K.K.L.; Kline, K.A. Model systems for the study of Enterococcal colonization and infection. Virulence 2017, 8, 1525–1562. [Google Scholar] [CrossRef]
- Eiko Maekawa, L.; Dennis Rossoni, R.; Oliveira Barbosa, J.; Olavo Cardoso Jorge, A.; Campos Junqueira, J.; Carneiro Valera, M.; José Longo, F. Different Extracts of Zingiber Officinale Decrease Enterococcus Faecalis Infection in Galleria Mellonella. Braz. Dent. J. 2015, 26, 105–109. [Google Scholar] [CrossRef]
- Luther, M.K.; Arvanitis, M.; Mylonakis, E.; LaPlante, K.L. Activity of daptomycin or linezolid in combination with rifampin or gentamicin against biofilm-forming Enterococcus faecalis or E. faecium in an in vitro pharmacodynamic model using simulated endocardial vegetations and an in vivo survival assay using Galleria mellonella larvae. Antimicrob. Agents Chemother. 2014, 58, 4612–4620. [Google Scholar] [PubMed]
- Nale, J.Y.; Chutia, M.; Carr, P.; Hickenbotham, P.T.; Clokie, M.R.J. “Get in early”; Biofilm and wax moth (Galleria mellonella) models reveal new insights into the therapeutic potential of Clostridium difficile bacteriophages. Front. Microbiol. 2016, 7, 1383. [Google Scholar] [CrossRef]
- Seed, K.D.; Dennis, J.J. Experimental bacteriophage therapy increases survival of Galleria mellonella larvae infected with clinically relevant strains of the Burkholderia cepacia complex. Antimicrob. Agents Chemother. 2009, 53, 2205–2208. [Google Scholar] [CrossRef]
- Beeton, M.L.; Alves, D.R.; Enright, M.C.; Jenkins, A.T.A. Assessing phage therapy against Pseudomonas aeruginosa using a Galleria mellonella infection model. Int. J. Antimicrob. Agents 2015, 46, 196–200. [Google Scholar] [CrossRef]
- Abbasifar, R.; Kropinski, A.M.; Sabour, P.M.; Chambers, J.R.; MacKinnon, J.; Malig, T.; Griffiths, M.W. Efficiency of bacteriophage therapy against Cronobacter sakazakii in Galleria mellonella (greater wax moth) larvae. Arch. Virol. 2014, 159, 2253–2261. [Google Scholar] [CrossRef] [PubMed]
- Schmelcher, M.; Donovan, D.M.; Loessner, M.J. Bacteriophage endolysins as novel antimicrobials. Future Microbiol. 2012, 7, 1147–1171. [Google Scholar] [CrossRef]
- Fernández-Ruiz, I.; Coutinho, F.H.; Rodriguez-Valera, F. Thousands of Novel Endolysins Discovered in Uncultured Phage Genomes. Front. Microbiol. 2018, 9, 1033. [Google Scholar] [CrossRef]
- Young, R.Y. Bacteriophage holins: Deadly diversity. J. Mol. Microbiol. Biotechnol. 2002, 4, 21–36. [Google Scholar]
- Hill, C.; Mills, S.; Ross, R.P. Phages & antibiotic resistance: Are the most abundant entities on earth ready for a comeback? Future Microbiol. 2018, 13, 711–726. [Google Scholar]
- Finn, R.D.; Clements, J.; Eddy, S.R. HMMER web server: Interactive sequence similarity searching. Nucleic Acids Res. 2011, 39, W29–W37. [Google Scholar] [CrossRef] [PubMed]
- Söding, J.; Biegert, A.; Lupas, A.N. The HHpred interactive server for protein homology detection and structure prediction. Nucleic Acids Res. 2005, 33, W244–W248. [Google Scholar] [CrossRef]
- Oliveira, H.; Melo, L.D.R.; Santos, S.B.; Nóbrega, F.L.; Ferreira, E.C.; Cerca, N.; Azeredo, J.; Kluskens, L.D. Molecular aspects and comparative genomics of bacteriophage endolysins. J. Virol. 2013, 87, 4558–4570. [Google Scholar] [CrossRef]
- Proença, D.; Leandro, C.; Garcia, M.; Pimentel, M.; São-José, C. EC300: A phage-based, bacteriolysin-like protein with enhanced antibacterial activity against Enterococcus faecalis. Appl. Microbiol. Biotechnol. 2015, 99, 5137–5149. [Google Scholar] [CrossRef]
- Uchiyama, J.; Takemura, I.; Hayashi, I.; Matsuzaki, S.; Satoh, M.; Ujihara, T.; Murakami, M.; Imajoh, M.; Sugai, M.; Daibata, M. Characterization of lytic enzyme open reading frame 9 (ORF9) derived from Enterococcus faecalis bacteriophage φEF24C. Appl. Environ. Microbiol. 2011, 77, 580–585. [Google Scholar] [CrossRef]
| Disease (Target Strain) | No (n) and Type of Subjects | Form and Dosage | Application Route and Clinical Outcome | Reference |
|---|---|---|---|---|
| Chronic bacterial prostatitis | n = 3 human male | Phage lysate ~107–109 PFU/mL | Rectal Pathogen eradication, Abatement of clinical symptoms Lack of early disease recurrence | [88] |
| Infection (EF14 VRE2) | n = 20; BALB/c mice female 6 to 8 week old | CsCl; 1 × 1012 PFU/mL; | Intraperitoneal; Significantly effective, Efficiently rescued mice; | [89] |
| Bacteremia (VAN) | n = 5 BALB/c mice 1 month old | CsCl 3 × 108 PFU/mL | Intraperitoneal 100% survival 45 min after bacterial challenge 50% of moribund mice rescued after delayed phage administration | [90] |
| Sepsis 002 | n = 8 7 different dosage groups BALB/c female mice 6 to 8 weeks old | PEG 3.9 × 109 PFU/mL or 0.2 mg endolysin | Intraperitoneal 60% survival at 30 min post bacterial inoculation 40% survival at 4 h post bacterial administration | [52] |
| E. faecalis challenge | n = 10 5 different dosage groups BALB/c F 6 to 8 weeks old | CsCl 4 × 103, 4 × 104, 4 × 105, 4 × 106, 4 × 107 PFU/mouse | Intraperitoneal Mice were protected from the infection | [91] |
| Septic peritonitis | n = 15 4 groups ICR(CD-1C) | Dialyzed phage lysate 2 × 108 | Intraperitoneal 100% survival No harmful effect on the microbiome | [60] |
| E. faecalis challenge (VAN) | n = 5 4 different groups BALB/c n female mice 6 to 8 weeks old | LysEF-P10 endolysin 1 µg, 5 µg, 10 µg | Intraperitoneal Reduced E. faecalis colonization Alleviated the gut microbiota imbalance caused by VRE | [92] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolocan, A.S.; Upadrasta, A.; de Almeida Bettio, P.H.; Clooney, A.G.; Draper, L.A.; Ross, R.P.; Hill, C. Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases. Viruses 2019, 11, 366. https://doi.org/10.3390/v11040366
Bolocan AS, Upadrasta A, de Almeida Bettio PH, Clooney AG, Draper LA, Ross RP, Hill C. Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases. Viruses. 2019; 11(4):366. https://doi.org/10.3390/v11040366
Chicago/Turabian StyleBolocan, Andrei S., Aditya Upadrasta, Pedro H. de Almeida Bettio, Adam G. Clooney, Lorraine A. Draper, R. Paul Ross, and Colin Hill. 2019. "Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases" Viruses 11, no. 4: 366. https://doi.org/10.3390/v11040366
APA StyleBolocan, A. S., Upadrasta, A., de Almeida Bettio, P. H., Clooney, A. G., Draper, L. A., Ross, R. P., & Hill, C. (2019). Evaluation of Phage Therapy in the Context of Enterococcus faecalis and Its Associated Diseases. Viruses, 11(4), 366. https://doi.org/10.3390/v11040366





