Current State of Compassionate Phage Therapy
Abstract
1. Introduction
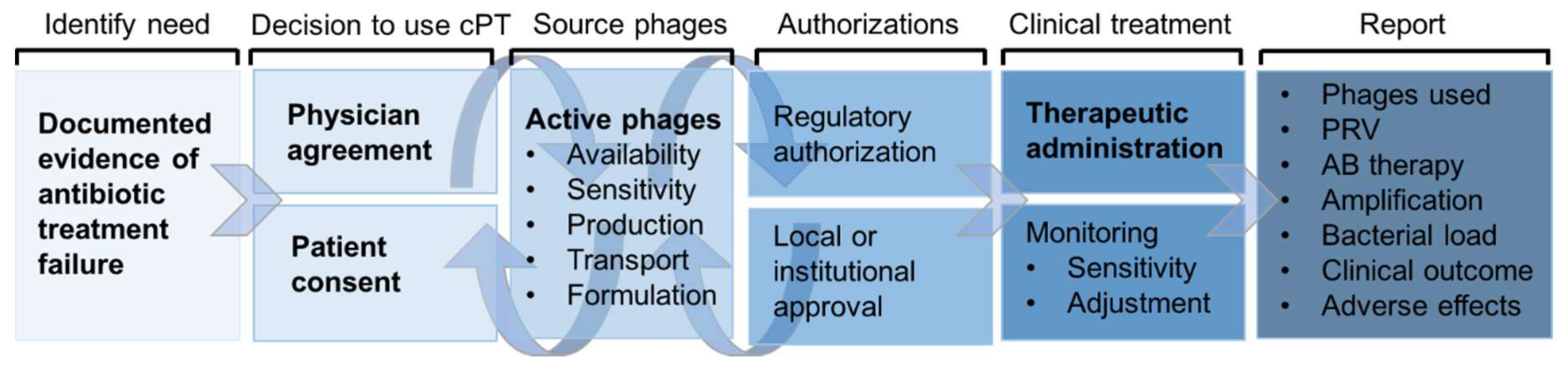
2. Compassionate Use
3. Compassionate Phage Therapy (cPT)
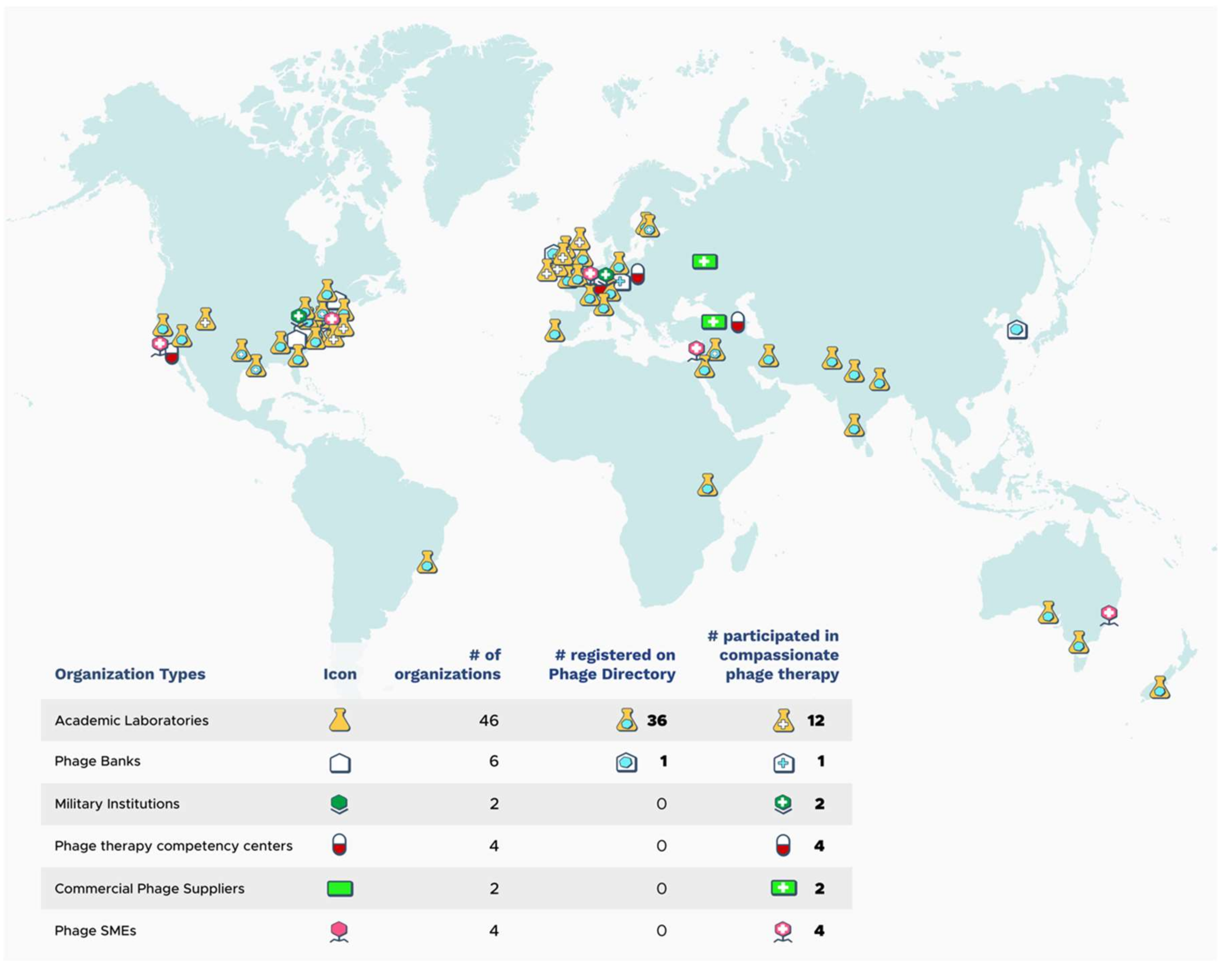
4. Sources and Availability of Phages for cPT
4.1. Environmentally-Sourced Phages
4.2. Academically-Sourced Phages
4.3. Phage Products in Clinical Development
4.4. Eastern European Phage Products
4.5. Crowd-Sourcing Phages
4.6. Logistical Constraints
5. Beyond Availability
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- D’Herelle, F. Bacteriophage as a Treatment in Acute Medical and Surgical Infections. Bull. N. Y. Acad. Med. 1931, 7, 329–348. [Google Scholar] [PubMed]
- Summers, W.C. Cholera and plague in India: The bacteriophage inquiry of 1927–1936. J. Hist. Med. Allied Sci. 1993, 48, 275–301. [Google Scholar] [CrossRef] [PubMed]
- D’Hérelle, F. Sur un microbe invisible antagoniste des bacilles dysentériques. Acad. Sci. Paris 1917, 165, 373–375. [Google Scholar]
- Bruynoghe, R.; Maisin, J. Essais de thérapeutique au moyen du bactériophage. CR Soc. Biol. 1922, 85, 1120–1121. [Google Scholar]
- Krueger, A.P.; Scribner, E.J. The Bacteriophage: Its Nature and Therapeutic Use. JAMA 1941, 116, 2269–2277. [Google Scholar] [CrossRef]
- Eaton, M.D.; Bayne-Jones, S. Bacteriophage Therapy: Review of the Principles and Results of the use of Bacteriophages in the Treatment of Infections. JAMA 1934, 103, 1769–1776. [Google Scholar] [CrossRef]
- Parfitt, T. Georgia: An unlikely stronghold for bacteriophage therapy. Lancet 2005, 365, 2166–2167. [Google Scholar] [CrossRef]
- Dublanchet, A. Autobiographie de Félix d’Hérelle. Les pérégrinations d’un bactériologiste; Pech, K., Ed.; Lavoisier: Paris, France, 2017; p. 347. [Google Scholar]
- Kutateladze, M. Experience of the Eliava Institute in bacteriophage therapy. Virol. Sin. 2015, 30, 80–81. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.M.; Koskella, B.; Lin, H.C. Phage therapy: An alternative to antibiotics in the age of multi-drug resistance. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 162–173. [Google Scholar] [CrossRef] [PubMed]
- McCallin, S.; Brüssow, H. Phage therapy: An alternative or adjunct to antibiotics? Emerg. Top. Life Sci. 2017. [Google Scholar] [CrossRef]
- Nobrega, F.L.; Costa, A.R.; Kluskens, L.D.; Azeredo, J. Revisiting phage therapy: new applications for old resources. Trends Microbiol. 2015, 23, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Reardon, S. Phage therapy gets revitalized. Nature 2014, 510, 15–16. [Google Scholar] [CrossRef]
- Kortright, K.E.; Chan, B.K.; Koff, J.L.; Turner, P.E. Phage Therapy: A Renewed Approach to Combat Antibiotic-Resistant Bacteria. Cell Host Microbe. 2019, 25, 219–232. [Google Scholar] [CrossRef]
- Jault, P.; Leclerc, T.; Jennes, S.; Pirnay, J.P.; Que, Y.A.; Resch, G.; Rousseau, A.F.; Ravat, F.; Carsin, H.; Le Floch, R.; et al. Efficacy and tolerability of a cocktail of bacteriophages to treat burn wounds infected by Pseudomonas aeruginosa (PhagoBurn): A randomised, controlled, double-blind phase 1/2 trial. Lancet Infect. Dis. 2019, 19, 35–45. [Google Scholar] [CrossRef]
- Sarker, S.A.; Sultana, S.; Reuteler, G.; Moine, D.; Descombes, P.; Charton, F.; Bourdin, G.; McCallin, S.; Ngom-Bru, C.; Neville, T.; et al. Oral Phage Therapy of Acute Bacterial Diarrhea With Two Coliphage Preparations: A Randomized Trial in Children From Bangladesh. EBioMedicine 2016, 4, 124–137. [Google Scholar] [CrossRef]
- Wright, A.; Hawkins, C.H.; Anggard, E.E.; Harper, D.R. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clin. Otolaryngol. 2009, 34, 349–357. [Google Scholar] [CrossRef]
- World Medical, A. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Carlson, R.V.; Boyd, K.M.; Webb, D.J. The revision of the Declaration of Helsinki: past, present and future. Br. J. Clin. Pharmacol. 2004, 57, 695–713. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, G.; Morampudi, S.; Chhabra, P.; Gowda, A.; Zomorodi, B. An overview of Compassionate Use Programs in the European Union member states. Intractable Rare Dis. Res. 2016, 5, 244–254. [Google Scholar] [CrossRef]
- Jarow, J.P.; Lurie, P.; Ikenberry, S.C.; Lemery, S. Overview of FDA’s Expanded Access Program for Investigational Drugs. Ther. Innov. Regul. Sci. 2017, 51, 177–179. [Google Scholar] [CrossRef]
- Donovan, P. Access to unregistered drugs in Australia. Aust. Prescr. 2017, 40, 194–196. [Google Scholar] [CrossRef]
- Borysowski, J.; Ehni, H.J.; Gorski, A. Ethics review in compassionate use. BMC Med. 2017, 15, 136. [Google Scholar] [CrossRef]
- Bunnik, E.M.; Aarts, N.; van de Vathorst, S. The changing landscape of expanded access to investigational drugs for patients with unmet medical needs: Ethical implications. J. Pharm. Policy Pract. 2017, 10, 10. [Google Scholar] [CrossRef]
- Holbein, M.E.; Berglund, J.P.; Weatherwax, K.; Gerber, D.E.; Adamo, J.E. Access to Investigational Drugs: FDA Expanded Access Programs or “Right-to-Try’’ Legislation? Clin. Transl. Sci. 2015, 8, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Gaffney, A. FDA Sees 92% Increase in Requests for Compassionate Access to Medicines in 2014. Regulatory Focus 2015. Available online: http://www.raps.org/Regulatory-Focus/News/2015/01/23/21151/Compassionate-Use-Requests-Increase-92-Percent/## (accessed on 5 January 2019).
- Mackey, T.K.; Schoenfeld, V.J. Going “social” to access experimental and potentially life-saving treatment: an assessment of the policy and online patient advocacy environment for expanded access. BMC Med. 2016, 14, 17. [Google Scholar] [CrossRef] [PubMed]
- Joffe, S.; Lynch, H.F. Federal Right-to-Try Legislation—Threatening the FDA’s Public Health Mission. N. Engl. J. Med. 2018, 378, 695–697. [Google Scholar] [CrossRef]
- Corporation, A.B. AmpliPhi to Collaborate with Western Sydney Local Health District and Westmead Institute for Medical Research on Expanded Access for Investigational Bacteriophage Therapeutics AB-SA01 and AB-PA01. San Diego, CA, 2018. Available online: https://investor.ampliphibio.com/press-release/featured/ampliphi-collaborate-western-sydney-local-health-district-and-westmead (accessed on 5 January 2019).
- Corporation, A.B. AmpliPhi Biosciences Announces Presentation of Positive Clinical Data from Its Expanded Access Program for Serious Saureus Infections at IDWeek 2018 Conference. San Diego, CA, 2018. Available online: https://investor.ampliphibio.com/press-release/featured/ampliphi-biosciences-announces-presentation-positive-clinical-data-its (accessed on 5 January 2019).
- Sacher, J.; Zheng, J.; McCallin, S. Sourcing phages for compassionate use. Microbiol. Aust. 2019. [Google Scholar] [CrossRef]
- Fadlallah, A.; Chelala, E.; Legeais, J.-M.M. Corneal Infection Therapy with Topical Bacteriophage Administration. Open Ophthalmol. J. 2015, 9, 167–168. [Google Scholar] [CrossRef]
- Patey, O.; McCallin, S.; Mazure, H.; Liddle, M.; Smithyman, A.; Dublanchet, A. Clinical Indications and Compassionate Use of Phage Therapy: Personal Experience and Literature Review with a Focus on Osteoarticular Infections. Viruses 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Zhvania, P.; Hoyle, N.S.; Nadareishvili, L.; Nizharadze, D.; Kutateladze, M. Phage Therapy in a 16-Year-Old Boy with Netherton Syndrome. Front. Med. (Lausanne) 2017, 4, 94. [Google Scholar] [CrossRef] [PubMed]
- Leszczyński, P.; Weber-Dabrowska, B.; Kohutnicka, K.M.; Łuzcak, M.; Górecki, A.; Górski, A. Successful eradication of methicillin-resistant Staphylococcus aureus (MRSA) intestinal carrier status in a healthcare worker—Case report. Folia. Microbiol. 2006, 51, 336–338. [Google Scholar] [CrossRef]
- Letkiewicz, S.; Międzybrodzki, R.; Fortuna, W.; Weber-Dąbrowska, B.; Górski, A. Eradication of Enterococcus faecalis by phage therapy in chronic bacterial prostatitis—case report. FEMS Immunol. Med. Microbiol. 2010, 54, 457–461. [Google Scholar] [CrossRef]
- Weber-Dabrowska, B.; Mulczyk, M.; Gorski, A. Bacteriophage therapy of bacterial infections: an update of our institute’s experience. Arch. Immunol. Ther. Exp. (Warsz) 2000, 48, 547–551. [Google Scholar] [PubMed]
- Weber-Dabrowska, B.; Mulczyk, M.; Gorski, A. Bacteriophage therapy for infections in cancer patients. Clin. Appl. Immunol. Rev. 2001, 1, 4. [Google Scholar] [CrossRef]
- Weber-Dabrowska, B.; Mulczyk, M.; Gorski, A. Bacteriophages as an efficient therapy for antibiotic-resistant septicemia in man. Transplant. Proc. 2003, 35, 1385–1386. [Google Scholar] [CrossRef]
- Miedzybrodzki, R.; Borysowski, J.; Weber-Dabrowska, B.; Fortuna, W.; Letkiewicz, S.; Szufnarowski, K.; Pawelczyk, Z.; Rogoz, P.; Klak, M.; Wojtasik, E.; et al. Clinical aspects of phage therapy. Adv. Virus Res. 2012, 83, 73–121. [Google Scholar] [CrossRef]
- Center for Innovative Phage Applications and Therapeutics. Available online: http://ipath.ucsd.edu/ (accessed on 5 January 2019).
- Pirnay, J.P.; Verbeken, G.; Ceyssens, P.J.; Huys, I.; De Vos, D.; Ameloot, C.; Fauconnier, A. The Magistral Phage. Viruses 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Chan, B.K.; Turner, P.E.; Kim, S.; Mojibian, H.R.; Elefteriades, J.A.; Narayan, D. Phage treatment of an aortic graft infected with Pseudomonas aeruginosa. Evol. Med. Public Health 2018, 2018, 60–66. [Google Scholar] [CrossRef]
- Duplessis, C.; Biswas, B.; Hanisch, B.; Perkins, M.; Henry, M.; Quinones, J.; Wolfe, D.; Estrella, L.; Hamilton, T. Refractory Pseudomonas Bacteremia in a 2-Year-Old Sterilized by Bacteriophage Therapy. J. Pediatric. Infect. Dis. Soc. 2017, 7, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Fish, R.; Kutter, E.; Bryan, D.; Wheat, G.; Kuhl, S. Resolving Digital Staphylococcal Osteomyelitis Using Bacteriophage—A Case Report. Antibiotics (Basel) 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Fish, R.; Kutter, E.; Wheat, G.; Blasdel, B.; Kutateladze, M.; Kuhl, S. Compassionate Use of Bacteriophage Therapy for Foot Ulcer Treatment as an Effective Step for Moving Toward Clinical Trials. Methods Mol. Biol. 2018, 1693, 159–170. [Google Scholar] [CrossRef] [PubMed]
- Khawaldeh, A.; Morales, S.; Dillon, B.; Alavidze, Z.; Ginn, A.N.; Thomas, L.; Chapman, S.J.; Dublanchet, A.; Smithyman, A.; Iredell, J.R. Bacteriophage therapy for refractory Pseudomonas aeruginosa urinary tract infection. J. Med. Microbiol. 2011, 60, 1697–1700. [Google Scholar] [CrossRef]
- Corporation, A.B. AmpliPhi Biosciences Announces First Intravenous Treatment of a Patient with AB-SA01 Targeting Staphylococcus aureus. San Diego, USA. 2017. Available online: https://www.ampliphibio.com/ampliphi-biosciences-announces-first-intravenous-treatment-of-a-patient-with-ab-sa01-targeting-staphylococcus-aureus/ (accessed on 5 January 2019).
- Nir-Paz, R.; Gelman, D.; Khouri, A.; Sisson, B.M.; Fackler, J.; Alkalay-Oren, S.; Khalifa, L.; Rimon, A.; Yerushalmy, O.; Bader, R.; et al. Successful treatment of antibiotic resistant poly-microbial bone infection with bacteriophages and antibiotics combination. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Ujmajuridze, A.; Chanishvili, N.; Goderdzishvili, M.; Leitner, L.; Mehnert, U.; Chkhotua, A.; Kessler, T.M.; Sybesma, W. Adapted Bacteriophages for Treating Urinary Tract Infections. Front. Microbiol. 2018, 9, 1832. [Google Scholar] [CrossRef] [PubMed]
- Hoyle, N.; Zhvaniya, P.; Balarjishvili, N.; Bolkvadze, D.; Nadareishvili, L.; Nizharadze, D.; Wittmann, J.; Rohde, C.; Kutateladze, M. Phage therapy against Achromobacter xylosoxidans lung infection in a patient with cystic fibrosis: a case report. Res. Microbiol. 2018, 169, 540–542. [Google Scholar] [CrossRef] [PubMed]
- Aslam, S.; Yung, J.; Dan, S.; Reed, S.; LeFebvre, M.; Logan, C.; Taplitz, R.; Law, N.; Golts, E.; Afshar, S.; et al. Bacteriophage Treatment in a Lung Transplant Recipient. J. Heart Lung Transplant. 2018, 37, S155–S156. [Google Scholar] [CrossRef]
- Ferry, T.; Boucher, F.; Fevre, C.; Perpoint, T.; Chateau, J.; Petitjean, C.; Josse, J.; Chidiac, C.; L’Hostis, G.; Leboucher, G.; et al. Innovations for the treatment of a complex bone and joint infection due to XDR Pseudomonas aeruginosa including local application of a selected cocktail of bacteriophages. J. Antimicrob. Chemother. 2018, 73, 2901–2903. [Google Scholar] [CrossRef] [PubMed]
- Ferry, T.; Leboucher, G.; Fevre, C.; Herry, Y.; Conrad, A.; Josse, J.; Batailler, C.; Chidiac, C.; Medina, M.; Lustig, S.; et al. Salvage Debridement, Antibiotics and Implant Retention (“DAIR”) With Local Injection of a Selected Cocktail of Bacteriophages: Is It an Option for an Elderly Patient With Relapsing Staphylococcus aureus Prosthetic-Joint Infection? Open Forum. Infect. Dis. 2018, 5, ofy269. [Google Scholar] [CrossRef]
- Morozova, V.V.; Vlassov, V.V.; Tikunova, N.V. Applications of Bacteriophages in the Treatment of Localized Infections in Humans. Front. Microbiol. 2018, 9, 1696. [Google Scholar] [CrossRef]
- LaVergne, S.; Hamilton, T.; Biswas, B.; Kumaraswamy, M.; Schooley, R.T.; Wooten, D. Phage Therapy for a Multidrug-Resistant Acinetobacter baumannii Craniectomy Site Infection. Open Forum. Infect. Dis. 2018, 5, ofy064. [Google Scholar] [CrossRef]
- Schooley, R.T.; Biswas, B.; Gill, J.J.; Hernandez-Morales, A.; Lancaster, J.; Lessor, L.; Barr, J.J.; Reed, S.L.; Rohwer, F.; Benler, S.; et al. Development and Use of Personalized Bacteriophage-Based Therapeutic Cocktails To Treat a Patient with a Disseminated Resistant Acinetobacter baumannii Infection. Antimicrob. Agents Chemother. 2017, 61, 17. [Google Scholar] [CrossRef] [PubMed]
- Jennes, S.; Merabishvili, M.; Soentjens, P.; Pang, K.; Rose, T.; Keersebilck, E.; Soete, O.; François, P.-M.; Teodorescu, S.; Verween, G.; et al. Use of bacteriophages in the treatment of colistin-only-sensitive Pseudomonas aeruginosa septicaemia in a patient with acute kidney injury—a case report. Critical. Care 2017, 21, 129. [Google Scholar] [CrossRef]
- Fish, R.; Kutter, E.; Wheat, G.; Blasdel, B.; Kutateladze, M.; Kuhl, S. Bacteriophage treatment of intransigent diabetic toe ulcers: A case series. J. Wound Care 2016, 25 (Suppl. 7), S27–33. [Google Scholar] [CrossRef]
- Rose, T.; Verbeken, G.; Vos, D.D.; Merabishvili, M.; Vaneechoutte, M.; Lavigne, R.; Jennes, S.; Zizi, M.; Pirnay, J.P. Experimental phage therapy of burn wound infection: difficult first steps. Int. J. Burns Trauma 2014, 4, 66–73. [Google Scholar] [PubMed]
- Marza, J.A.; Soothill, J.S.; Boydell, P.; Collyns, T.A. Multiplication of therapeutically administered bacteriophages in Pseudomonas aeruginosa infected patients. Burns 2006, 32, 644–646. [Google Scholar] [CrossRef]
- Jikia, D.; Chkhaidze, N.; Imedashvili, E.; Mgaloblishvili, I.; Tsitlanadze, G.; Katsarava, R.; Morris, G.J.; Sulakvelidze, A. The use of a novel biodegradable preparation capable of the sustained release of bacteriophages and ciprofloxacin, in the complex treatment of multidrug-resistant Staphylococcus aureus-infected local radiation injuries caused by exposure to Sr90. Clin. Exp. Dermatol. 2005, 30, 23–26. [Google Scholar] [CrossRef] [PubMed]
- Markoishvili, K.; Tsitlanadze, G.; Katsarava, R.; Morris, J.G., Jr.; Sulakvelidze, A. A novel sustained-release matrix based on biodegradable poly(ester amide)s and impregnated with bacteriophages and an antibiotic shows promise in management of infected venous stasis ulcers and other poorly healing wounds. Int. J. Dermatol. 2002, 41, 453–458. [Google Scholar] [CrossRef]
- Weber-Dąbrowska, B.; Jończyk-Matysiak, E.; Żaczek, M.; Łobocka, M.; Łusiak-Szelachowska, M.; Górski, A. Bacteriophage Procurement for Therapeutic Purposes. Front. Microbiol. 2016, 7, 1177. [Google Scholar] [CrossRef]
- Félix d’Hérelle Reference Center for Bacterial Viruses. Available online: https://www.phage.ulaval.ca/en/home/ (accessed on 5 January 2019).
- Phages. Available online: https://www.dsmz.de/catalogues/catalogue-microorganisms/groups-of-organisms-and-their-applications/phages.html (accessed on 5 January 2019).
- The Bacteriophage Bank of Korea. Available online: http://www.phagebank.or.kr/intro/eng_intro.jsp (accessed on 5 January 2019).
- Phage Directory. Available online: https://phage.directory/ (accessed on 5 January 2019).
- Fruciano, D.E.; Bourne, S. Phage as an antimicrobial agent: D’Herelle’s heretical theories and their role in the decline of phage prophylaxis in the West. Can. J. Infect. Dis. Med. Microbiol. 2007, 18, 19–26. [Google Scholar] [CrossRef]
- Sulakvelidze, A.; Alavidze, Z.; Morris, J.G., Jr. Bacteriophage therapy. Antimicrob. Agents Chemother. 2001, 45, 649–659. [Google Scholar] [CrossRef]
- Summers, W.C. The strange history of phage therapy. Bacteriophage 2012, 2, 130–133. [Google Scholar] [CrossRef] [PubMed]
- Calandra, G.B.; Garelik, J.P.; Kohler, P.T.; Brown, K.R. Problems and benefits of an antibiotic compassionate therapy program. Rev. Infect. Dis. 1987, 9, 1095–1101. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abedon, S.T. Information Phage Therapy Research Should Report. Pharmaceuticals (Basel) 2017, 10. [Google Scholar] [CrossRef]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; Review on Antimicrobial Resistance: London, UK, 2016. [Google Scholar]
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect Dis. 19, 56–66. [CrossRef]
| Pathogen | Infection | Admin Route | N* | Clinical Outcome | AB (N*) | Failure /PRV+ | Phage Source | Ref. |
|---|---|---|---|---|---|---|---|---|
| A. baumannii, K. pneumoniae | Bone | iv | 1 | Success | Yes | na/no | Military | [49] |
| S. aureus; P. aeruginosa; E. coli; ProteusPM | Bone; GI; ENT; urogenital | Local; oral; rectal; joint injection | 15 | High success rate (12/15); all cases improved | Yes | 2° pathogen for 1 patient; unclear results for 2 patients | Mostly commercial | [33] |
| S. aureus | Bone | Soft-tissue injection | 1 | Success | Int. | na/nr | Commercial (Eliava) | [45] |
| S. aureus; E. coli; Proteus; Streptococcus; P. aeruginosa | UTI | Local via catheter | 9 | Bacterial load decrease in 67% (6/9); pathogen clearance for 3 patients | Yes (1) | No decrease for 1 patient; 2° infection for 1 patient/ nr | Commercial (Eliava); adapted to strains | [50] |
| Achromabacter xylosoxidans | Cystic Fibrosis infection | Inhaled; oral | 1 | Improved lung function and general condition | Yes, post | na/nr | Environ. | [51] |
| P. aeruginosa | Recurrent pneumonia | Inhaled; iv | 1 | Success | Yes | na/ Yes (PS) | Environ., biotech; military | [52] |
| S. aureus | Bone | Local | 1 | Success | Yes | na/nr | Biotech | |
| S. aureus, P. aeruginosaPM | Bone | Local | 3 | nr | nr | nr/nr | Biotech | [53] |
| P. aeruginosa | Bone | Local | 1 | Success for bacterial clearance† | Yes | na/nr | Biotech | [54] |
| E. coli; Proteus; S. aureus; P. aeruginosa; Streptococcus; Enterococcus | Burns, ulcers, wounds | Topical; sc | 234: (27; 90; 94; 23) | Overall high success rate; varied by study | Varied with study | Varied with study/nr | Commercial; unspecified | Review of 4 cases in Russian [55] |
| P. aeruginosa | Aortic valve graft | Direct via fistula | 1 | Success | Yes | na/nr | Academic | [43] |
| A. baumanii | Post-operative cranial infection | iv | 1 | Infection site cleared; blood cultures negative† | No | Treatment discontinued/nr | Military | [56] |
| S. aureus | Chronic skin infection | Topical; oral | 1 | Decreased bacterial load; improved clinical condition | No | Prolonged treatment/Yes (PS) | Commercial (Eliava) | [34] |
| A. baumanii | Necrotizing pancreatitis | iv; local | 1 | Success | Yes | na/Yes (PS) | Environ., military; biotech; phage bank | [57] |
| P. aeruginosa | Infected wound/ septicemia | iv; local | 1 | Wounds remained colonized, blood cultures were negative† | Int | Bacteremia resolved, but local infection persisted/nr | Military | [58] |
| P. aeruginosa | Bacteremia | iv | 1 | Bacteremia eradicated twice; subsequent regrowth † | Yes | Slow bacterial regrowth/PRV likely | Military | [44] |
| S. aureus | Diabetic toe ulcer infection | Topical | 6 | Success; avoided amputation | nr | na/nr | Commercial (Eliava) | [59] |
| S. aureus | Corneal abscess | Topical, nasal, iv | 1 | Success | nr | na/nr | Commercial (Eliava) | [32] |
| P. aeruginosa; S. aureusPM | Burn wound infections | Topical | 9 | Modest reduction in bacterial load for 8 patients | Just prior | nr/nr | Military | [60] |
| Staphylococcus; Enterococcus; Pseudomonas; E. coli; Proteus; Enterococcus; etcPM | UTI; urogenital; soft tissue; skin; orthopedic; respiratory; bacteremia; etc. | Topical, oral, rectal, vaginal, inhaled | 157 | Good clinical outcomes for 44% of patients (success for 18%) | Yes (29%) | Inadequate response for 60% of patients/Yes | In-house | [40] |
| P. aeruginosa | UTI | Local in bladder | 1 | Success | Yes | na/No | Commercial (Eliava) | [47] |
| Enterococcus faecalis | Prostatitis | Rectal | 3 | Success | No | na/nr | In-house | [36] |
| S. aureus | GI Carrier status | Oral | 1 | Success | No | na/nr | In-house | [35] |
| P. aeruginosa | Burn wound | Topical | 1 | Successful grafting | Yes | na/nr | Academic | [61] |
| S. aureus | Wounds | Topical | 2 | Success | Yes | na/nr | Commercial | [62] |
| S. aureus; E. coli; P. aeruginosa; Klebsiella; etcPM | Septicemia | Oral | 94 | 85% success rate | Yes (n = 71) | Phage ineffective for 15% of patients/nr | In-house | [39] |
| Staphylococcus; E. coli; Proteus; Streptococcus; P. aeruginosa PM | Venous ulcers and wounds | Topical | 96 | 70% healing | Yes | No clinical improvement for 5 patients | Commercial | [63] |
| S. aureus; E. coli; P. aeruginosa; Klebsiella PM | Various infections in cancer patients | Oral, local | 20 | Healing in all patients | nr | na/nr | In-house | [38] |
| S. aureus; E. coli; Proteus; P. aeruginosa; Klebsiella; EnterobacterPM | Septicemia; ENT; UTI; meningitis; respiratory; wounds; bone; etc. | Oral; topical; local | 1307 | Full recovery 86%; 11% transient improvement | nr | No effect in 3.8% of study population (n = 50) | In-house | [37] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCallin, S.; Sacher, J.C.; Zheng, J.; Chan, B.K. Current State of Compassionate Phage Therapy. Viruses 2019, 11, 343. https://doi.org/10.3390/v11040343
McCallin S, Sacher JC, Zheng J, Chan BK. Current State of Compassionate Phage Therapy. Viruses. 2019; 11(4):343. https://doi.org/10.3390/v11040343
Chicago/Turabian StyleMcCallin, Shawna, Jessica C. Sacher, Jan Zheng, and Benjamin K. Chan. 2019. "Current State of Compassionate Phage Therapy" Viruses 11, no. 4: 343. https://doi.org/10.3390/v11040343
APA StyleMcCallin, S., Sacher, J. C., Zheng, J., & Chan, B. K. (2019). Current State of Compassionate Phage Therapy. Viruses, 11(4), 343. https://doi.org/10.3390/v11040343






